Abstract
The limbic system is a network of interconnected brain regions regulating emotion, memory, and behavior. Pathology of the limbic system can manifest as psychiatric disease, including obsessive-compulsive disorder and major depressive disorder. For patients with these disorders who have not responded to standard pharmacological and cognitive behavioral therapy, ablative surgery is a neurosurgical treatment option. The major ablative limbic system procedures currently used are anterior capsulotomy, dorsal anterior cingulotomy, subcaudate tractotomy, and limbic leucotomy. In this review, we include a brief history of ablative limbic system surgery leading up to its current form. Mechanistic justification for these procedures is considered in a discussion of the pathophysiology of psychiatric disease. We then discuss therapeutic efficacy as demonstrated by recent trials. Finally, we consider future directions, including the search for predictors of treatment response, the development of more precise targeting methods, and the use of advances in neuroimaging to track treatment response.
Keywords: limbic system, neuroimaging, psychosurgery, obsessive-compulsive disorder, major depressive disorder, mental disorders, psychiatry, neurosurgery, stereotaxis
Introduction
The limbic system plays a major role in the regulation of emotion, memory, and behavior [1]. Originally described by Papez in 1937 [2] and further characterized by Maclean in the early 1950s [3] , the limbic system is currently understood to include subcortical structures (such as the hippocampus, amygdala, and ventral striatum) and cortical structures (including orbitofrontal cortex, cingulate cortex, and insula) [1]. The past decades have brought a refinement in functional understanding of limbic circuits. The current model of the limbic system includes multiple subcircuits, including hippocampal-diencephalic and parahippocampal-retrosplenial networks mediating memory and spatial orientation, and a temporo-amygdala-orbitofrontal-cingulate network broadly involved in emotion and its link to behavior [1]. The complex neuroanatomy of the limbic system parallels its requirement to support these diverse functions.
Because of its complex structure and function, the limbic system is uniquely vulnerable to dysfunction, which can manifest behaviorally as neurological or psychiatric diseases. At their conceptual core, these disorders are characterized by a variety of emotional, attentional, and behavioral perturbations, functions that are governed by the limbic system. Obsessive-compulsive disorder (OCD), major depressive disorder (MDD), and bipolar disorder, among many others, are all psychiatric disorders with increasingly well-understood mechanistic underpinnings in limbic system dysfunction.
The majority of patients with these disorders improve with traditional pharmacological and cognitive-behavioral therapies. However, in both OCD and MDD, about 10-20% of patients do not respond adequately to these conventional approaches [4–7]. For these refractory patients, neuromodulatory surgical procedures, including stereotactic ablative lesions and deep brain stimulation, can be effective treatment options. The goal of this review is to discuss this first category of procedures, ablative lesions, which are believed to disrupt dysfunctional networks implicated in disorders of the limbic system. Here we will briefly discuss the history of ablative limbic system surgery through its initial implementations, then focus on the current utilizations of and trends in cingulotomy, capsulotomy, subcaudate tractotomy, and limbic leucotomy. In doing so, we will discuss the pathophysiological circuits in disorders of the limbic system that motivate the use of these procedures, the studies defining that pathophysiology, and future directions for limbic system surgery.
History
In 1891, Burckhardt published the results of a series of six resections of left frontotemporal cortex in patients with various psychiatric disorders [8]. Primate studies implicating the frontal lobe in behavioral pathology by John Fulton inspired Egas Moniz's development of the prefrontal leucotomy in the 1930s, in which the frontal white matter was surgically interrupted [9]. In this era before antipsychotic drugs, patients with severe psychiatric disease such as schizophrenia had few options outside of overcrowded asylums. For some of these patients, Moniz's procedure provided symptomatic relief and the possibility of moving on from lifelong psychiatric institutionalization. It was seen as a remarkable accomplishment for a challenging situation and, in 1949, Moniz was awarded the Nobel Prize in Physiology or Medicine [10]. Moniz's accomplishment was particularly noteworthy because it underscored the emerging understanding that psychiatric diseases have an anatomical and functional basis in the brain.
However, the innovation of the prefrontal leucotomy was controversial at the time of its development, and went through a period of widespread adoption, followed by disfavor. In the 1940s, Freeman and Watts adapted Moniz's procedure to a technically simpler procedure that could be performed through burr holes, terming it the “lobotomy.” Freeman eventually further simplified the technique to a transorbital procedure that could be performed at the bedside, totaling approximately 20,000 procedures by 1951, without neurosurgical support or backup [11]. Again, therapeutic efficacy was seen for some patients, but its use became increasingly indiscriminate, and many patients were neurologically devastated. The advent of antipsychotics in the early 1950s led to a marked decrease in the use of the lobotomy.
Why did these procedures provide benefit for some patients? Broadly speaking, the interruption of pathological frontolimbic circuits is likely what mediated symptomatic improvement. Analysis of historical lesions, limited as it is without contemporary imaging, has identified damage to many of the same mediobasal frontal structures and pathways included in modern lesions [12,13]. The procedures in the 1940s and 50s were performed without the benefit of stereotaxy or radiography and were therefore heterogeneous, imprecise, and difficult to study, and often associated with major neurological morbidity [14]. However, it was noted that those procedures that featured smaller lesions without major cortical destruction could be effective in the treatment of psychiatric disease while sparing neurological function [15]. At around the same time, the advent of stereotactic techniques made it possible to safely and reproducibly create lesions, with minimal anatomical disruption [15].
Stereotaxis: The Advent of Target Definition
In the early 1900s, Horsley and Clarke pioneered the development of stereotactic procedures, in which a rigid head-mounted frame is adjusted in the Cartesian x, y, and z planes to target a specific location in the brain [16]. In 1947, Spiegel and Wycis were the first to apply the “Horsley apparatus” to humans when they performed a stereotactic medial thalamotomy [17], which was intended as a treatment for a patient with intractable “emotional reactivity.” Today's stereotaxy has improved over the decades since Spiegel and Wycis’ plaster-and-metal frame, providing increasingly precise and targeted means of creating reproducible lesions in a minimally invasive manner.
Stereotaxy is one of numerous technical advances since the 1940s that have made psychiatric neurosurgery safer and more effective. Advancement in the understanding of the pathophysiology of psychiatric disease has also been a major development. Thus, before discussing the major ablative neurosurgical procedures for limbic system pathology, we provide an update on the circuits involved in psychiatric disease. While these targets were identified and refined by trial-and-error, we advance the rationale for currently utilized targets based on the current understanding of the pathophysiology of refractory OCD and depression. Initially, we focus on the role of cortico-striato-thalamo-cortical (CSTC) loops and how, when dysfunctional, their interruption may be therapeutic.
Brain Circuits Targeted by Ablative Limbic Surgery: The CSTC Loops
CSTC circuits are classically described as “loops” that bind together regions of cortex, basal ganglia, and thalamus subserving related functions, such as movement, memory, or behavioral regulation [18]. As described by Alexander, Delong, and Strick, there are a number of circuits projecting from cortex to striatum to thalamus back to cortex, although their exact number, as well as the functional rigidity of their boundaries, is under debate [19]. One circuit, involving the dorsal anterior cingulate cortex, orbitofrontal cortex, ventral striatum, mediodorsal thalamus, and subcortical structures such as hippocampus and amygdala, integrates the limbic system, basal ganglia, and frontal circuits in a feedback-dependent manner to regulate behavior, specifically its affective and cognitive components [20–22] (Figure 1A). Dysfunction within the limbic loop may therefore result in aberrant affective and cognitive processing of stimuli (the nature of that aberrance dependent on the exact disorder), resulting in abnormal behavior. The limbic loop of the CSTC circuits is therefore implicated in a number of the psychiatric disorders that limbic system modulation attempts to relieve [21,23,24].
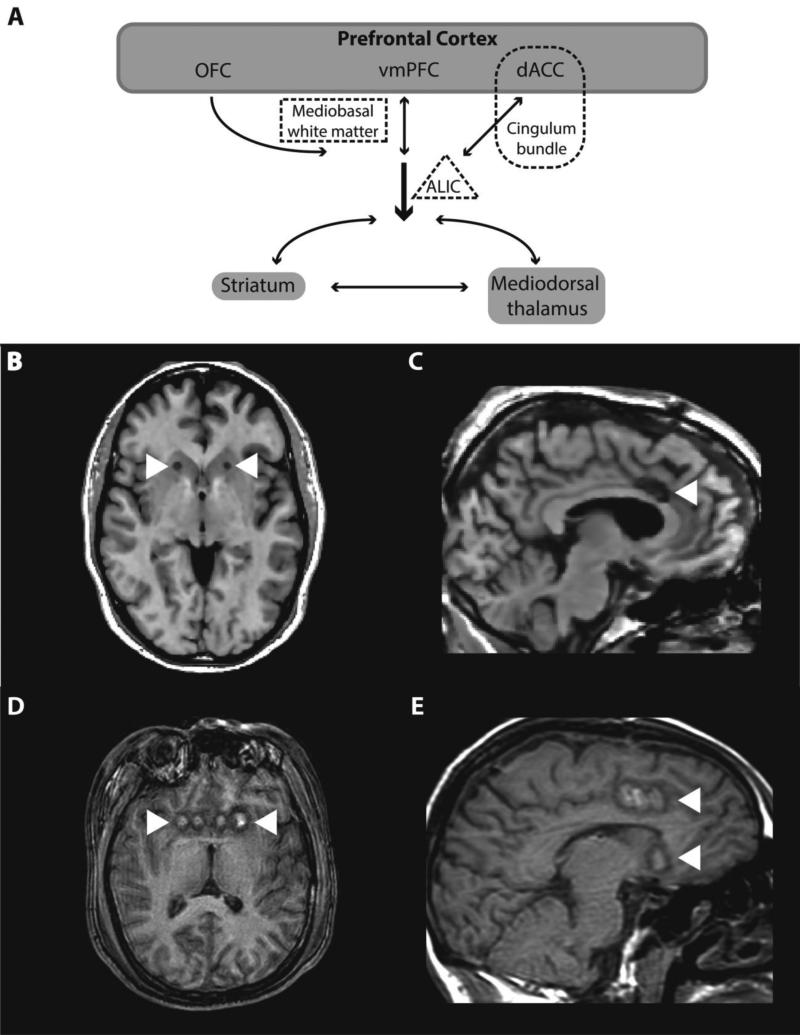
Fig. 1.
Schematic of the limbic loop of the cortico-striato-thalamo-cortical (CSTC) circuits with representative T1-weighted MR images of the major ablative limbic system surgeries. (A) Like other CSTC circuits, the limbic CSTC loop consists of input from cortex to striatum, from striatum to thalamus, then from thalamus back to cortex. There is also involvement of subcortical limbic structures such as the amygdala and hippocampal formation (not shown). The prefrontal cortex components of the limbic CSTC loop are orbitofrontal cortex (OFC), ventromedial prefrontal cortex (vmPFC), and dorsal anterior cingulate cortex (dACC). These cortical structures communicate reciprocally and also send projections through the anterior limb of the internal capsule (ALIC) to the mediodorsal thalamus and striatum. Three major lesion locations for ablative limbic system surgery are shown in dashed outlines. Respective MR images are shown with white arrowheads indicating lesion location. Anterior capsulotomy (B, MRI at 6 months post-op) is performed by lesion of the ventral aspect of the anterior limb of the internal capsule (dashed triangle). Dorsal anterior cingulotomy (C, MRI at 12 months post-op) is performed by lesion of the dACC and its corresponding white matter tract, the cingulum bundle (dashed round rectangle). Subcaudate tractotomy (D, MRI immediately post-op) is performed by lesion of the mediobasal white matter (dashed rectangle). Limbic leucotomy (E, MRI immediately postop) is a combination of dorsal anterior cingulotomy and subcaudate tractotomy
Indeed, appreciation of the CSTC framework has informed the study of the anatomic and functional underpinnings of many psychiatric disorders. Functional and metabolic neuroimaging of OCD, for example, has identified dysfunction in a number of regions characterized as part of CSTC loops. Hyperactivity of orbitofrontal cortex (OFC) [19], dorsal anterior cingulate cortex (dACC) [25], caudate [26], and thalamus has been described [27], with demonstrable decreases in metabolism on PET after treatment [28]. Symptom provocation neuroimaging studies of OCD patients have demonstrated increased OFC and ACC activity with trigger stimuli for OCD behavior [29,30]. Finally, hyperconnectivity within CSTC circuit regions has been seen in OFC, dACC, thalamus, and striatum [31], although pathological hypoconnectivity has also been reported [19]. The concept underlying ablative surgery for OCD is thus based on this pathological hyperactivity and dysfunctional connectivity within the limbic CSTC loop.
Other psychiatric disorders for which ablative limbic system surgery has been utilized also exhibit limbic CSTC-related dysfunction. Affective disorders, such as major depressive disorder (MDD) and bipolar disorder, have been explained in the context of dorsal, ventral, and modulatory components consisting of integrated rather than segregated CSTC circuits [15,32]. According to one such account, the ventral component controls affect by encoding emotional salience through the subgenual cingulate cortex, OFC, and insula, with reciprocal connections to anterior and mediodorsal thalamic nuclei through the ventral striatum. This component is typically found to be hyperactive in functional neuroimaging studies of affective disorders [33]. The dorsal component, on other hand, has been shown to mediate cognitive and motor aspects of behavior, and consists of the dACC, dlPFC, and premotor cortices, with connections to the anterior and mediodorsal thalamic nuclei through the dorsal striatum. This component has demonstrated hypoactivity in neuroimaging studies of MDD, which may partly explain the affective blunting observed in some forms of the disorder [15]. Finally, the modulatory component, comprised of pregenual ACC, amygdala, and the hypothalamic-pituitary axis, appears to modulate dorsal and ventral components. Though the exact method of framing CSTC circuit involvement may be different, the existence of pathological activity within associated brain regions is evident in psychiatric disease.
We now consider each of the major ablative limbic system surgeries in turn, with considerations of surgical technique, applications, and further pathophysiologic discussion.
Anterior Capsulotomy
Following Spiegel and Wycis’ use of stereotaxy in humans, Lars Leksell and Jean Talairach developed the anterior capsulotomy in 1949 [34]. This procedure targets the ventral aspect of the anterior limb of the internal capsule (ALIC) bilaterally (Figure 1B). Disruption of this white matter structure is thought to have provided at least some of the therapeutic relief of early lobotomies [15]. Since then, studies of this ventral ALIC target have demonstrated orbitofrontal cortex (OFC) and subgenual cingulate projections traveling to medial thalamic nuclei [15].
Capsulotomy is currently performed with either radiofrequency thermoablation or stereotactic radiosurgery. The latter, often performed using the Gamma Knife device (“Gamma capsulotomy”), is growing in popularity recently due to its less invasive nature [35] and amenability to randomized, blinded trials [36]. Capsulotomy has been studied in OCD, in which symptom improvement for OCD is typically measured by decreases in scores on the Yale-Brown Obsessive Compulsive Disorder Scale (Y-BOCS) [37]. Using this scale, a “full” response to treatment is typically defined as a > 35% decrease in score from baseline. “Partial” response is typically defined as a decrease of 25-35% from baseline Y-BOCS score. A recent retrospective analysis of 19 patients treated over a decade with thermoablation found that 36.8% were full responders and 10.5% were partial responders [38]. Other reports of response rate range from 48-80% [39–42] (Table 1). Thermoablative capsulotomy has also been used for intractable MDD and mood disorders. A prospective study of 20 MDD patients undergoing capsulotomy over a period of 7 years demonstrated marked improvement in 55% of patients [43]. Adverse effects in all studies of thermoablative capsulotomy can be categorized as short-term vs. long-term, and minor vs. serious. Serious adverse events, which are becoming increasingly less common, include hemiplegia and deficits in executive function and cognition [38]. The large majority of reported adverse events tend to be minor and short-term, including asymptomatic intracranial hemorrhage, abulia, and weight gain [40,43].
| Procedure | Study | Sample Size | Disorder Treated | Mean time of LFU* | Mean % change† | Adverse events• |
|---|---|---|---|---|---|---|
| Anterior capsulotomy (thermoablative) | Zhan et al, 2014 | 53 | OCD | 12 | −73.7 |
S: Memory impairment (13), UI (11) L: Personality change (3), hypersexuality (3) |
| D'Astous, 2013 | 19 | OCD | 84 | −32 |
S: Frontal syndrome (5), hemorrhage (3) L: Hemiplegia (1), cognitive deficits (1) |
|
| Hurwitz, 2012‡ | 8 | MDD | 24 | −57.4 |
S: Confusion (2), akinetic mutism (1) L: Executive dysfunction (1), fatigue (1) |
|
| Christmas, 2011^ | 20 | MDD | 84 | −50.7 |
S: Confusion (5), HA (3), UI (3) L: Amotivation (4), weight gain (3) |
|
| Liu, 2008 | 35 | OCD | 36 | −79.2 |
S: UI (3), intracerebral hematoma (1) L: Apathy, abulia (2) |
|
| Rück, 2008 | 25 | OCD | 33.5 | −52.5 |
S: Weight gain (mean 6 kg/1 yr) L: UI (2), apathy (1), disinhibition (1) |
|
| Oliver, 2003 | 15 | OCD | 24 | −39 |
S: Hallucinations (1), seizure (1) L: Behavioral disorder (1) |
|
| Gamma capsulotomy | Lopes, 2014 | 16 | OCD | 12 | −51.9 |
S: Weight gain (4), HA (4), manic episodes (2) L: Brain cyst (1), drug abuse (1) |
| Sheehan, 2013 | 5 | OCD | 22 | −50 | None reported | |
| Kondziolka, 2011 | 3 | OCD | 42 | −54 | None reported | |
| Lopes, 2009 | 5 | OCD | 48 | −36 |
S: HA, nausea L: Weight gain |
|
| Dorsal anterior cingulotomy | Sheth, 2013§ | 64 | OCD | 63.8 | −28 |
S: Memory deficit (5), abulia (4), UR (2) L: Suicide (2), infection (1) |
| Shields, 2008‡ | 33 | MDD | 30 | −20.8 | S: UI (4), memory deficit (1), infection (1) | |
| Jung, 2006 | 16 | OCD | 24 | −48 | None reported | |
| Kim, 2003 | 14 | OCD | 12 | −36 | S: Insomnia (4), weight change (4), HA (3) | |
| Limbic leucotomy | Cho, 2008‡ | 16 | Bipolar | 84 | −40.6 | S: Wound infection (1), hallucinations (1), extrapyramidal symptoms (1) |
| Subcaudate tractotomy | Bourne, 2013¶ | 11 | OCD | 50 | −42 | S: Abulia (3), memory deficit (1) |
KEY
LFU = last follow up, months.
% change refers to % change on a given behavioral scale between preoperative administration and LFU administration. For all OCD studies, Yale-Brown Obsessive Compulsive Scale (Y-BOCS) was used. See notation for other studies. Conventionally, 35% change is regarded as a full treatment response.
Abbreviations: S = short term, L = long term, UI = urinary incontinence, UR = urinary retention, HA = headache.
Includes patients from Baer et al, 1995 and Doughery et al, 2002.
Used Beck Depression Inventory as behavioral scale.
Used HRSD-17 as behavioral scale.
Subcaudate tractotomy was performed as a follow up for cingulotomy non-responders, effectively producing a limbic leucotomy. Given the relative paucity of studies of rigidly tested patients who have undergone subcaudate tractotomy, we present these results for completeness.
Gamma capsulotomy was first used in 1976 [44] and has seen increasing use over the last several years. The first blinded, sham-controlled, randomized controlled trial of Gamma capsulotomy for refractory OCD was recently published [36]. As in thermoablative capsulotomy, the ventral aspect of the ALIC was targeted. Of 8 patients undergoing the procedure, 3 responded within 12 months and 2 more responded within 54 months (response defined by >35% improvement of Y-BOCS score). None of the sham patients responded. Four patients within the sham group elected to undergo Gamma capsulotomy in the open label follow-up period after removal of blinding, two of whom responded. A recent retrospective study assessed Gamma capsulotomy for refractory OCD in 5 patients. Four of these patients demonstrated “marked clinical improvement” at a median of 24 months follow up. Another study of 3 patients found that all responded to GK capsulotomy [45]. Adverse events are similar when comparing the two capsulotomy techniques. Although the Gamma capsulotomy procedure itself is relatively noninvasive, the impact of ionizing radiation must be considered. The high doses of radiation required for these procedures can lead to the development of cystic changes in the brain [36], as well as a slightly increased risk of secondary malignancies [46]. Cognitive events were also seen, including a manic episode in patients with previous hypomania, the development of drug abuse in one patient, and insomnia [36].
Neuroimaging findings in patients who have undergone capsulotomy have confirmed and extended theories about hyperactivity in cortical and subcortical areas in psychiatric disease, providing valuable information about pathophysiological circuits. A recent PET study of 13 patients who had undergone capsulotomy for OCD confirmed metabolic changes in previously described areas, including subgenual cingulate cortex, caudate, and dACC [47,48]. These structures all have corresponding fibers that travel through the ALIC, providing further support for a ventral ALIC target. Structural studies have noted postoperative changes in brain volume, including decreases in ALIC, ventral striatum, thalamus, and occasionally hippocampus [49]. Another study noted novel post-operative white matter changes at 2 months after capsulotomy in 5 patients, which included increases in T1 signal potentially signifying axonal change [50]. Longer-term studies are needed to better characterize the relationship between structural changes and clinical response. These findings will inform design of future interventions for refractory psychiatric disease.
Summary and Future Directions
Anterior capsulotomy has demonstrated efficacy for refractory OCD and mood disorders. Techniques for Gamma capsulotomy, which offers the benefit of noninvasiveness, continue to be refined. Moreover, as it is the only ablative surgical modality for psychiatric disease for which a blinded trial has been performed, Gamma capsulotomy has the strongest evidence supporting it. Recent trends in Gamma capsulotomy include a reduction in radiation dose from 180 Gy to 140-160 Gy and a reduction in the number of isocenters used [51]. Older studies typically delivered 3 bilateral doses, whereas a recent study used just one bilateral dose [51]. If similar therapeutic benefit can be achieved with less radiation, adverse events will decline as well. Moreover, further investigation is needed to determine the typical time course of response to Gamma capsulotomy. In particular, time to symptomatic relief varies between individuals and studies, but typically tends to follow on the order of months to over a year. In one study, two patients who did not initially respond were considered responders at two-year follow-up [36]. Moreover, the durability of the response to capsulotomy has not been clearly defined in the literature. Long-term follow-up studies will be needed to address these questions. Transcranial focused ultrasound (tFUS) has recently been advanced as a promising new technique for creating intracranial lesions [52,53]. It has been used for OCD [54], but similar long-term studies are needed to better understand its efficacy and long-term potential.
Dorsal Anterior Cingulotomy
Cingulotomy was first proposed in the 1950s by Fulton and subsequently refined and popularized by Ballantine [9,55]. In dorsal anterior cingulotomy, the anterior cingulate cortex and the cingulum bundle are ablated with stereotactic guidance (Figure 1C). The ACC is a region within the medial prefrontal cortex consisting of the anterior aspect of the cingulate cortex as it wraps around the rostral corpus callosum [56]. It is subdivided into rostral and dorsal ACC (rACC and dACC) regions. In broad terms, these regions appear to have emotional and cognitive functions, respectively. ACC is known to have extensive connectivity with dorsolateral prefrontal, orbitofrontal, primary premotor, and insular cortices, along with thalamic nuclei, especially the mediodorsal thalamus. This complex connectivity enables the ACC to play a role in cognitive control [57,58], pain [59,60], and emotional processing [61]. Though it was commonly thought that the rostral and dorsal anatomic subdivisions represented distinct functional units, recent analysis instead suggests functional integration across these regions [62].
This combination of cognitive and emotional processing makes the ACC a theoretically ideal focus of therapeutic intervention for psychiatric disorders in which these processes are impaired. Several disorders have been treated with cingulotomy, including treatment-refractory OCD, MDD, and chronic neuropathic pain [9,15]. Cingulotomy lesions are typically made via radiofrequency thermocoagulation under stereotactic guidance. Though it is the most commonly used psychiatric neurosurgical procedure in the United States and Canada [15], it is still not widely performed. However, a relatively concise literature suggests it is safe and effective in the treatment of OCD and depression.
Studies of the effectiveness of cingulotomy are relatively small but roughly uniform in results. A recent retrospective study with a cohort of 64 patients who had undergone cingulotomy for refractory OCD showed a full response rate of 47% and partial response rate of 22% after mean follow-up of over 5 years [63]. Other studies of cingulotomy for OCD have described response rates of 48% after 2 years [64], 43% after 1 year [65], and 5 of 7 (71%) patients after 1 year [66] (Table 1). Mood disorders show a similar response rate, with a recent study of anterior cingulotomy for intractable MDD in 33 patients showing 41% improvement and 33% partial improvement after 30 months [14].
Adverse events can again be categorized as short-term vs. long-term and minor vs. serious. Recent studies have demonstrated some short-term adverse events, but far fewer long-term adverse events. Adverse events in both categories are typically minor. One study reported the development of long-term post-operative epilepsy that required medication control [67]. Reported short-term adverse events include both urinary retention [63] and urinary incontinence [68,69]. Short-term cognitive changes can also occur, including blunted affect, abulia, and post-operative amnesia; these usually resolve within days [56]. Some impairment in performance on cognitive interference tasks, such as the Stroop test and Multi-Source Interference Task (MSIT) [70], have been reported in the months following cingulotomy, although these tend to resolve fairly rapidly as well [56,71].
Similar to anterior capsulotomy, studies suggest that it takes time for the efficacy of cingulotomy to develop. In most studies, it takes from months to over a year for therapeutic response. In one study, for example, nearly half of patients who eventually exhibited response to cingulotomy did not show appreciable symptomatic improvement for multiple follow up visits [63]. Mechanistically, this delay suggests potential long-term plasticity measures required for symptomatic improvement. Response appears to be durable, as the same study demonstrated long-term maintenance of response status [63].
Summary and Future Directions
Dorsal anterior cingulotomy is thus an efficacious procedure for the treatment of refractory psychiatric disease. However, further work remains to be done in regards to defining optimal targets. dACC function continues to be investigated, and the current emerging picture of this region's functionality is an overlap in the processing of cognitive control, negative affect, and pain [62]. Indeed, the dACC of different patients with psychiatric disease may be differentially affected, leading to heterogeneous manifestations of disease and similarly heterogeneous optimal therapeutic targets. Through advances in neuroimaging combined with empirically observed symptomatic changes, further narrowing of lesion extent may be possible on an individualized basis. Moreover, despite evidence for the clinical benefit of cingulotomy, a number of questions remain about patient selection, predictors of response, and mechanism of action. Potential anatomic predictors of response were recently described, including gray matter partial volume in the dACC, as well as asymmetry of CBTC connectivity [72]. These types of neuroimaging studies will also be helpful in clarifying the mechanism of action of these procedures, which may involve changes in CSTC circuits that develop over time.
Subcaudate Tractotomy
The subcaudate tractotomy (SCT), first reported in 1960 by Knight [69], is a procedure with similar principles and indications as capsulotomy and anterior cingulotomy. Lesions are made in the frontal mediobasal white matter, which is located ventral to the head of the caudate nucleus and contains corticothalamic fibers [15,69] (Figure 1D). Although the target is precisely defined in the modern era using stereotaxy, retrospective data on SCT lesions has suggested that there may be substantial patient-to-patient variation in anatomy in this brain region. A recent analysis of SCT procedures showed variable involvement of the uncinate fasciculus, medial orbitofrontal cortex, and nucleus accumbens [73].
SCT as a standalone procedure is not commonly used in the United States, and is only slightly more popular in England [69]. In the past, it has been used for both mood and anxiety disorders, including MDD and OCD. A study of 23 patients with affective disorders demonstrated symptomatic improvement but significant cognitive changes on postoperative neuropsychological testing [74].
Despite its limited clinical use, SCT has had scientific value in its contribution to anatomic understanding of relevant pathways. Recent tractographic analyses of OFC-related white matter tracts in non-human primates [75] and healthy humans [76] have shed light on the anatomic organization of OFC subdivisions projecting to the thalamus and brainstem. Building on these characterizations, a similar study of OCD patients who had undergone SCT recapitulated these findings and found that the best clinical outcomes may be associated with a target in the ventral aspect of medial OFC white matter tracts [12]. This study proves, in principle, the ability of current modalities to localize an optimal target along a white matter tract for therapeutic relief, and can in theory be applied to more psychiatric disorders.
Limbic Leucotomy
Performed since the 1970s with demonstrated efficacy for mood disorders and OCD [77,78], the limbic leucotomy is a combination of anterior cingulotomy and subcaudate tractotomy. Depending on the clinical context, the limbic leucotomy can be performed as one procedure, in which frontothalamic white matter tracts are lesioned in the basal medial frontal lobes, along with a lesion of the dACC as described above for anterior cingulotomy [69] (Figure 1E). Alternatively, it can be performed by following up an anterior cingulotomy with SCT. One group performing the latter demonstrated a 73% rate of symptomatic improvement in patients with OCD and intractable MDD who did not initially respond to anterior cingulotomy [79]. Another group released a 7-year prospective study of patients who received limbic leucotomy in one procedure for intractable bipolar disorder, and found a significant and stable decrease in depressive, but not manic symptoms [78].
Side effects of limbic leucotomy appear to be short-term and spontaneously resolving, and include transient hallucinations, amnesia and mania. Notably, however, abulia appears to develop at a greater rate following limbic leucotomy than cingulotomy [63], but is still typically self-limited. Long-term adverse events include extrapyramidal symptoms treated with medication [78].
Future Directions
Over its 60-year history, ablative limbic system surgery appears to be an efficacious intervention for refractory psychiatric disorders. However, a number of clinical and scientific questions have not been answered. Three major questions, in particular, deserve mention:
-
1)
Do neuroimaging features or other biomarkers exist that predict clinical response?
-
2)
What is the mechanism of efficacy of ablative limbic system surgery?
-
3)
Can this information be harnessed to individualize procedures?
Prospective databases of patients undergoing psychiatric neurosurgery, including standardized clinical and neurocognitive evaluations, neuroimaging protocols, and follow-up data are needed to address these questions. Promising routes of exploring each question already exist however.
The ability to predict response to an invasive intervention, especially a permanent one such as a brain lesion, would be extremely useful. Progress towards this goal continues to be made. One potential modality for predicting response is preoperative F-18-fluorodeoxyglucose-positron emission tomography (FDG-PET). FDG-PET has been utilized to demonstrate loci of hypermetabolism that may correlate to treatment response in patients undergoing cingulotomy for OCD [80] and MDD [81]. Patient anatomy itself may also provide indices of prediction of response to cingulotomy. In a retrospective assessment of structural imaging of responders and non-responders to cingulotomy, dACC grey matter volume, as well as laterality differences in connectivity of key CSTC nodes, predicted clinical response [72]. Taken together, these results indicate the potential for using structural and metabolic brain imaging as a means of predicting response to neurosurgical intervention for refractory psychiatric disease.
Neuroimaging approaches can further provide information about how the therapeutic response develops. Therapeutic efficacy in ablative limbic system surgery typically occurs over many months [36,63]. Thus an important question to address is whether anatomical and functional plasticity changes can be observed longitudinally with modalities likes structural MRI, PET, DTI, and resting state fMRI. Plasticity within CSTC circuits may mediate specific symptomatic changes based on lesion location. Tracking changes in neuroimaging would thus allow for better definition of how lesion location affects clinical symptomatology. Moreover, multiple lesion procedures seem to provide benefit in some patients. For example, in some patients who were not responsive to cingulotomy, completion of limbic leucotomy provided therapeutic benefit (12,63,78). What differences in symptomatology and plasticity are at play in these patients compared to those who respond to an alternative procedure? Further information about response patterns could presumably lead to optimal targets as defined by the specific clinical manifestations of the patient (i.e. patients with certain symptoms respond better to certain procedures).
Indeed, a major goal of psychiatric neurosurgery is targeting on an individualized basis. The diverse symptomatology of psychiatric disorders may reflect differential involvement of CSTC circuits. As a result, a lesion in a specific location may be greatly beneficial for one patient, but marginally so in someone else. Thus, better understanding of dysfunctional circuits is needed in conjunction with how they correspond to symptomatology. In doing so, more specific targets may theoretically be designed that vary based on individual symptom constellations. Accomplishing such individualization of treatment will require parallel progress in defining pathological circuits and improving efficacy of procedures. The involvement of multi-disciplinary teams, with psychiatrists, neurosurgeons, neuropsychologists, neurologists, and cognitive scientists, is therefore essential. Rationally designed targets defined with this level of detail will ideally reduce adverse events as well as provide better outcomes for patients.
Conclusion
In the decades since the initial forays into psychiatric neurosurgery, much progress has been made in understanding the physiology and pathophysiology of the limbic system. Importantly, the limbic subset of the CSTC network has been elucidated in progressively greater detail. Dysfunction in this circuit serves as a mechanistic common link between psychiatric disorders such as obsessive-compulsive disorder, major depressive disorder, and many others. Using progressively more refined neuroimaging tools, structural and functional correlates of CSTC network pathology have been elaborated in order to more precisely define targets. Current ablative limbic system procedures, including dorsal anterior cingulotomy, anterior capsulotomy, subcaudate tractotomy, and limbic leucotomy, thus serve as efficacious therapeutic interventions for those who have exhausted all conventional options. Moreover, the neuroimaging and behavioral data that arise from these procedures are a means of probing pathological circuits to better define disease pathways. With better capability to predict response, better methods of tracking response, and individualized patient targeting, efficacy and safety will improve as distinct patient symptomatology is targeted.
Footnotes
Compliance with Ethics Guidelines
Conflict of Interest
Saurabh Sinha, Garrett Banks, Robert McGovern, Charles Mikell and Sameer Sheth have no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by the author.
References
Recent papers of interest have been marked with an asterisk.
- 1.Catani M, Dell'acqua F, Thiebaut de Schotten M. A revised limbic system model for memory, emotion and behaviour. Neurosci Biobehav Rev. 2013;37(8):1724–37. doi: 10.1016/j.neubiorev.2013.07.001. [DOI] [PubMed] [Google Scholar]
- 2.Papez JW. A proposed mechanism of emotion. 1937. J Neuropsychiatry Clin Neurosci. 1995;7(1):103–12. doi: 10.1176/jnp.7.1.103. [DOI] [PubMed] [Google Scholar]
- 3.Maclean PD. Some psychiatric implications of physiological studies on frontotemporal portion of limbic system (visceral brain). Electroencephalogr Clin Neurophysiol. 1952 Nov;4(4):407–18. doi: 10.1016/0013-4694(52)90073-4. [DOI] [PubMed] [Google Scholar]
- 4.Saxena S, Rauch SL. Functional neuroimaging and the neuroanatomy of obsessive-compulsive disorder. Psychiatr Clin North Am. 2000 Sep;23(3):563–86. doi: 10.1016/s0193-953x(05)70181-7. [DOI] [PubMed] [Google Scholar]
- 5.Sackeim HA. The definition and meaning of treatment-resistant depression. J Clin Psychiatry. 2001;62(Suppl 16):10–7. [PubMed] [Google Scholar]
- 6.Pallanti S, Quercioli L. Treatment-refractory obsessive-compulsive disorder: methodological issues, operational definitions and therapeutic lines. Prog Neuropsychopharmacol Biol Psychiatry. 2006 May;30(3):400–12. doi: 10.1016/j.pnpbp.2005.11.028. [DOI] [PubMed] [Google Scholar]
- 7.Berlim MT, Turecki G. Definition, assessment, and staging of treatment-resistant refractory major depression: a review of current concepts and methods. Can J Psychiatry Rev Can Psychiatr. 2007 Jan;52(1):46–54. doi: 10.1177/070674370705200108. [DOI] [PubMed] [Google Scholar]
- 8.Luigjes J, de Kwaasteniet BP, de Koning PP, Oudijn MS, van den Munckhof P, Schuurman PR, et al. Surgery for psychiatric disorders. World Neurosurg. 2013 Oct;80(3- 4):S31.e17–28. doi: 10.1016/j.wneu.2012.03.009. [DOI] [PubMed] [Google Scholar]
- 9.Mashour GA, Walker EE, Martuza RL. Psychosurgery: past, present, and future. Brain Res Brain Res Rev. 2005;48(3):409–19. doi: 10.1016/j.brainresrev.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 10.Raju TN. The Nobel chronicles. 1949: Walter Rudolf Hess (1881-1973); and Antônio Egas Moniz (1874-1955). Lancet. 1999 Apr 10;353(9160):1281. doi: 10.1016/s0140-6736(05)75208-9. [DOI] [PubMed] [Google Scholar]
- 11.El-Hai J. The Lobotomist: A Maverick Medical Genius and His Tragic Quest to Rid the World of Mental Illness. Wiley; Hoboken, N.J.: 2007. p. 368. [Google Scholar]
- 12.Yang JC, Ginat DT, Dougherty DD, Makris N, Eskandar EN. Lesion analysis for cingulotomy and limbic leucotomy: comparison and correlation with clinical outcomes. J Neurosurg. 2014;120(1):152–63. doi: 10.3171/2013.9.JNS13839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schoene-Bake J-C, Parpaley Y, Weber B, Panksepp J, Hurwitz TA, Coenen VA. Tractographic analysis of historical lesion surgery for depression. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol. 2010 Dec;35(13):2553–63. doi: 10.1038/npp.2010.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shields DC, Asaad W, Eskandar EN, Jain FA, Cosgrove GR, Flaherty AW, et al. Prospective assessment of stereotactic ablative surgery for intractable major depression. Biol Psychiatry. 2008;64(6):449–54. doi: 10.1016/j.biopsych.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 15*.Lapidus KAB, Kopell BH, Ben-Haim S, Rezai AR, Goodman WK. History of psychosurgery: a psychiatrist’s perspective. World Neurosurg. 2013;80(3-4):S27, e1–16. doi: 10.1016/j.wneu.2013.02.053. [This is an accessible review of psychiatric neurosurgery from the perspective of psychiatry that discusses its history with an overview of each procedure.] [DOI] [PubMed] [Google Scholar]
- 16.Pereira EAC, Green AL, Nandi D, Aziz TZ. Stereotactic neurosurgery in the United Kingdom: the hundred years from Horsley to Hariz. Neurosurgery. 2008 Sep;63(3):594–606. doi: 10.1227/01.NEU.0000316854.29571.40. discussion 606–7. [DOI] [PubMed] [Google Scholar]
- 17.Spiegel EA, Wycis HT, Marks M, Lee AJ. Stereotaxic Apparatus for Operations on the Human Brain. Science. 1947;106(2754):349–50. doi: 10.1126/science.106.2754.349. [DOI] [PubMed] [Google Scholar]
- 18.Alexander GE, Crutcher MD. Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci. 1990 Jul;13(7):266–71. doi: 10.1016/0166-2236(90)90107-l. [DOI] [PubMed] [Google Scholar]
- 19.Posner J, Marsh R, Maia TV, Peterson BS, Gruber A, Simpson HB. Reduced Functional Connectivity Within the Limbic Cortico-Striato-Thalamo-Cortical Loop in Unmedicated Adults With Obsessive-Compulsive Disorder. Hum Brain Mapp. 2014 Jun;35(6):2852–60. doi: 10.1002/hbm.22371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci. 1986;9:357–81. doi: 10.1146/annurev.ne.09.030186.002041. [DOI] [PubMed] [Google Scholar]
- 21.Menzies L, Chamberlain SR, Laird AR, Thelen SM, Sahakian BJ, Bullmore ET. Integrating evidence from neuroimaging and neuropsychological studies of obsessive- compulsive disorder: the orbitofronto-striatal model revisited. Neurosci Biobehav Rev. 2008;32(3):525–49. doi: 10.1016/j.neubiorev.2007.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Göttlich M, Krämer UM, Kordon A, Hohagen F, Zurowski B. Decreased limbic and increased fronto-parietal connectivity in unmedicated patients with obsessive- compulsive disorder. Hum Brain Mapp. 2014;35(11):5617–32. doi: 10.1002/hbm.22574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Marchand WR. Cortico-basal ganglia circuitry: a review of key research and implications for functional connectivity studies of mood and anxiety disorders. Brain Struct Funct. 2010 Aug;215(2):73–96. doi: 10.1007/s00429-010-0280-y. [DOI] [PubMed] [Google Scholar]
- 24.Furman DJ, Hamilton JP, Gotlib IH. Frontostriatal functional connectivity in major depressive disorder. [2014 Nov 22];Biol Mood Anxiety Disord [Internet] 2011 Dec 8; doi: 10.1186/2045-5380-1-11. 1. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3384258/ [DOI] [PMC free article] [PubMed]
- 25.Swedo SE, Schapiro MB, Grady CL, Cheslow DL, Leonard HL, Kumar A, et al. Cerebral glucose metabolism in childhood-onset obsessive-compulsive disorder. Arch Gen Psychiatry. 1989 Jun;46(6):518–23. doi: 10.1001/archpsyc.1989.01810060038007. [DOI] [PubMed] [Google Scholar]
- 26.Saxena S, Brody AL, Ho ML, Zohrabi N, Maidment KM, Baxter LR. Differential brain metabolic predictors of response to paroxetine in obsessive-compulsive disorder versus major depression. Am J Psychiatry. 2003 Mar;160(3):522–32. doi: 10.1176/appi.ajp.160.3.522. [DOI] [PubMed] [Google Scholar]
- 27.Nakao T, Okada K, Kanba S. Neurobiological model of obsessive-compulsive disorder: evidence from recent neuropsychological and neuroimaging findings. Psychiatry Clin Neurosci. 2014;68(8):587–605. doi: 10.1111/pcn.12195. [DOI] [PubMed] [Google Scholar]
- 28.Baxter LR, Phelps ME, Mazziotta JC, Guze BH, Schwartz JM, Selin CE. Local cerebral glucose metabolic rates in obsessive-compulsive disorder. A comparison with rates in unipolar depression and in normal controls. Arch Gen Psychiatry. 1987 Mar;44(3):211–8. doi: 10.1001/archpsyc.1987.01800150017003. [DOI] [PubMed] [Google Scholar]
- 29.Breiter HC, Rauch SL, Kwong KK, et al. FUnctional magnetic resonance imaging of symptom provocation in obsessive-compulsive disorder. Arch Gen Psychiatry. 1996 Jul 1;53(7):595–606. doi: 10.1001/archpsyc.1996.01830070041008. [DOI] [PubMed] [Google Scholar]
- 30.Adler CM, McDonough-Ryan P, Sax KW, Holland SK, Arndt S, Strakowski SM. fMRI of neuronal activation with symptom provocation in unmedicated patients with obsessive compulsive disorder. J Psychiatr Res. 2000 Oct;34(4-5):317–24. doi: 10.1016/s0022-3956(00)00022-4. [DOI] [PubMed] [Google Scholar]
- 31.Hou J-M, Zhao M, Zhang W, Song L-H, Wu W-J, Wang J, et al. Resting-state functional connectivity abnormalities in patients with obsessive-compulsive disorder and their healthy first-degree relatives. J Psychiatry Neurosci JPN. 2014;39(5):304–11. doi: 10.1503/jpn.130220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Milad MR, Rauch SL. Obsessive-compulsive disorder: beyond segregated cortico striatal pathways. Trends Cogn Sci. 2012 Jan;16(1):43–51. doi: 10.1016/j.tics.2011.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kopell BH, Greenberg B, Rezai AR. Deep brain stimulation for psychiatric disorders. J Clin Neurophysiol Off Publ Am Electroencephalogr Soc. 2004 Feb;21(1):51–67. doi: 10.1097/00004691-200401000-00007. [DOI] [PubMed] [Google Scholar]
- 34.Leksell L, Backlund EO. [Radiosurgical capsulotomy--a closed surgical method for psychiatric surgery]. Läkartidningen. 1978 Feb 15;75(7):546–7. [PubMed] [Google Scholar]
- 35.Lévêque M, Carron R, Régis J. Radiosurgery for the treatment of psychiatric disorders: a review. World Neurosurg. 2013;80(3-4):S32, e1–9. doi: 10.1016/j.wneu.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 36*.Lopes AC, Greenberg BD, Canteras MM, Batistuzzo MC, Hoexter MQ, Gentil AF, et al. Gamma ventral capsulotomy for obsessive-compulsive disorder: a randomized clinical trial. JAMA Psychiatry. 2014 Sep;71(9):1066–76. doi: 10.1001/jamapsychiatry.2014.1193. [This is the first randomized controlled trial of an ablative limbic system surgery (Gamma capsulotomy), and features thorough explanations of patient selection post-operative clinical course.] [DOI] [PubMed] [Google Scholar]
- 37.Goodman WK, Price LH, Rasmussen SA, Mazure C, Fleischmann RL, Hill CL, et al. The Yale-Brown Obsessive Compulsive Scale. I. Development, use, and reliability. Arch Gen Psychiatry. 1989 Nov;46(11):1006–11. doi: 10.1001/archpsyc.1989.01810110048007. [DOI] [PubMed] [Google Scholar]
- 38.D'Astous M, Cottin S, Roy M, Picard C, Cantin L. Bilateral stereotactic anterior capsulotomy for obsessive-compulsive disorder: long-term follow-up. J Neurol Neurosurg Psychiatry. 2013;84(11):1208–13. doi: 10.1136/jnnp-2012-303826. [DOI] [PubMed] [Google Scholar]
- 39.Oliver B, Gascón J, Aparicio A, Ayats E, Rodriguez R, Maestro De León JL, et al. Bilateral anterior capsulotomy for refractory obsessive-compulsive disorders. Stereotact Funct Neurosurg. 2003;81(1-4):90–5. doi: 10.1159/000075110. [DOI] [PubMed] [Google Scholar]
- 40.Rück C, Andréewitch S, Flyckt K, Edman G, Nyman H, Meyerson BA, et al. Capsulotomy for refractory anxiety disorders: long-term follow-up of 26 patients. Am J Psychiatry. 2003 Mar;160(3):513–21. doi: 10.1176/appi.ajp.160.3.513. [DOI] [PubMed] [Google Scholar]
- 41.Liu K, Zhang H, Liu C, Guan Y, Lang L, Cheng Y, et al. Stereotactic treatment of refractory obsessive compulsive disorder by bilateral capsulotomy with 3 years follow-up. J Clin Neurosci Off J Neurosurg Soc Australas. 2008 Jun;15(6):622–9. doi: 10.1016/j.jocn.2007.07.086. [DOI] [PubMed] [Google Scholar]
- 42.Zhan S, Liu W, Li D, Pan S, Pan Y, Li Y, et al. Long-term follow-up of bilateral anterior capsulotomy in patients with refractory obsessive-compulsive disorder. Clin Neurol Neurosurg. 2014 Apr;119:91–5. doi: 10.1016/j.clineuro.2014.01.009. [DOI] [PubMed] [Google Scholar]
- 43.Christmas D, Eljamel MS, Butler S, Hazari H, MacVicar R, Steele JD, et al. Long term outcome of thermal anterior capsulotomy for chronic, treatment refractory depression. J Neurol Neurosurg Psychiatry. 2011;82(6):594–600. doi: 10.1136/jnnp.2010.217901. [DOI] [PubMed] [Google Scholar]
- 44.Mindus null, Jenike MA. Neurosurgical treatment of malignant obsessive compulsive disorder. Psychiatr Clin North Am. 1992 Dec;15(4):921–38. [PubMed] [Google Scholar]
- 45.Kondziolka D, Flickinger JC, Hudak R. Results following gamma knife radiosurgical anterior capsulotomies for obsessive compulsive disorder. Neurosurgery. 2011;68(1):28–32. doi: 10.1227/NEU.0b013e3181fc5c8b. –discussion 23–3. [DOI] [PubMed] [Google Scholar]
- 46.Loeffler JS, Niemierko A, Chapman PH. Second tumors after radiosurgery: tip of the iceberg or a bump in the road? Neurosurgery. 2003 Jun;52(6):1436–40. doi: 10.1227/01.neu.0000064809.59806.e8. discussion 1440–2. [DOI] [PubMed] [Google Scholar]
- 47.Suetens K, Nuttin B, Gabriëls L, Van Laere K. Differences in Metabolic Network Modulation Between Capsulotomy and Deep-Brain Stimulation for Refractory Obsessive-Compulsive Disorder. J Nucl Med Off Publ Soc Nucl Med. 2014;55(6):951–9. doi: 10.2967/jnumed.113.126409. [DOI] [PubMed] [Google Scholar]
- 48.Zuo C, Ma Y, Sun B, Peng S, Zhang H, Eidelberg D, et al. Metabolic imaging of bilateral anterior capsulotomy in refractory obsessive compulsive disorder: an FDG PET study. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab. 2013;33(6):880–7. doi: 10.1038/jcbfm.2013.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Taren JA, Curtis GC, Gebarski SS. Late local and remote structural changes after capsulotomy for obsessive compulsive disorder. Stereotact Funct Neurosurg. 1994;63(1-4):1–6. [PubMed] [Google Scholar]
- 50.Hurwitz TA, Mandat T, Forster B, Honey C. Tract identification by novel MRI signal changes following stereotactic anterior capsulotomy. Stereotact Funct Neurosurg. 2006;84(5-6):228–35. doi: 10.1159/000096496. [DOI] [PubMed] [Google Scholar]
- 51.Sheehan JP, Patterson G, Schlesinger D, Xu Z. γ knife surgery anterior capsulotomy for severe and refractory obsessive-compulsive disorder. J Neurosurg. 2013;119(5):1112–8. doi: 10.3171/2013.5.JNS13201. [DOI] [PubMed] [Google Scholar]
- 52.Martin E, Jeanmonod D, Morel A, Zadicario E, Werner B. High-intensity focused ultrasound for noninvasive functional neurosurgery. Ann Neurol. 2009 Dec;66(6):858–61. doi: 10.1002/ana.21801. [DOI] [PubMed] [Google Scholar]
- 53.Jeanmonod D, Werner B, Morel A, Michels L, Zadicario E, Schiff G, et al. Transcranial magnetic resonance imaging-guided focused ultrasound: noninvasive central lateral thalamotomy for chronic neuropathic pain. Neurosurg Focus. 2012;32(1):E1. doi: 10.3171/2011.10.FOCUS11248. [DOI] [PubMed] [Google Scholar]
- 54.Jung HH, Chang WS, Rachmilevitch I, Tlusty T, Zadicario E, Chang JW. Different magnetic resonance imaging patterns after transcranial magnetic resonance-guided focused ultrasound of the ventral intermediate nucleus of the thalamus and anterior limb of the internal capsule in patients with essential tremor or obsessive-compulsive disorder. J Neurosurg. 2014:1–7. doi: 10.3171/2014.8.JNS132603. [DOI] [PubMed] [Google Scholar]
- 55.Ballantine HT, Bouckoms AJ, Thomas EK, Giriunas IE. Treatment of psychiatric illness by stereotactic cingulotomy. Biol Psychiatry. 1987;22(7):807–19. doi: 10.1016/0006-3223(87)90080-1. [DOI] [PubMed] [Google Scholar]
- 56.Gasquoine PG. Localization of function in anterior cingulate cortex: from psychosurgery to functional neuroimaging. Neurosci Biobehav Rev. 2013;37(3):340–8. doi: 10.1016/j.neubiorev.2013.01.002. [DOI] [PubMed] [Google Scholar]
- 57.Sheth SA, Mian MK, Patel SR, Asaad WF, Williams ZM, Dougherty DD, et al. Human dorsal anterior cingulate cortex neurons mediate ongoing behavioural adaptation. Nature. 2012;488(7410):218–21. doi: 10.1038/nature11239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shenhav A, Botvinick MM, Cohen JD. The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron. 2013 Jul 24;79(2):217–40. doi: 10.1016/j.neuron.2013.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rainville P, Duncan GH, Price DD, Carrier B, Bushnell MC. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science. 1997 Aug 15;277(5328):968–71. doi: 10.1126/science.277.5328.968. [DOI] [PubMed] [Google Scholar]
- 60.Emmert K, Breimhorst M, Bauermann T, Birklein F, Van De Ville D, Haller S. Comparison of anterior cingulate vs. insular cortex as targets for real-time fMRI regulation during pain stimulation. Front Behav Neurosci. 2014;8:350. doi: 10.3389/fnbeh.2014.00350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Morita T, Tanabe HC, Sasaki AT, Shimada K, Kakigi R, Sadato N. The anterior insular and anterior cingulate cortices in emotional processing for self-face recognition. Soc Cogn Affect Neurosci. 2014 May;9(5):570–9. doi: 10.1093/scan/nst011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Shackman AJ, Salomons TV, Slagter HA, Fox AS, Winter JJ, Davidson RJ. The integration of negative affect, pain and cognitive control in the cingulate cortex. Nat Rev Neurosci. 2011 Mar;12(3):154–67. doi: 10.1038/nrn2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63*.Sheth SA, Neal J, Tangherlini F, Mian MK, Gentil A, Cosgrove GR, et al. Limbic system surgery for treatment-refractory obsessive-compulsive disorder: a prospective long-term follow-up of 64 patients. J Neurosurg. 2013 Mar;118(3):491–7. doi: 10.3171/2012.11.JNS12389. [This prospective study features the largest sample size of patients treated with dorsal anterior cingulotomy. It follows up and adds patients to previous similar studies, demonstrating the long-term therapeutic course of limbic system surgeries.] [DOI] [PubMed] [Google Scholar]
- 64.Jung HH, Kim C-H, Chang JH, Park YG, Chung SS, Chang JW. Bilateral anterior cingulotomy for refractory obsessive-compulsive disorder: Long-term follow-up results. Stereotact Funct Neurosurg. 2006;84(4):184–9. doi: 10.1159/000095031. [DOI] [PubMed] [Google Scholar]
- 65.Kim C-H, Chang JW, Koo M-S, Kim JW, Suh HS, Park IH, et al. Anterior cingulotomy for refractory obsessive-compulsive disorder. Acta Psychiatr Scand. 2003 Apr;107(4):283–90. doi: 10.1034/j.1600-0447.2003.00087.x. [DOI] [PubMed] [Google Scholar]
- 66.Zhang QJ, Wang WH, Wei XP. Long-term efficacy of stereotactic bilateral anterior cingulotomy and bilateral anterior capsulotomy as a treatment for refractory obsessive-compulsive disorder. Stereotact Funct Neurosurg. 2013;91(4):258–61. doi: 10.1159/000348275. [DOI] [PubMed] [Google Scholar]
- 67.Dougherty DD, Baer L, Cosgrove GR, Cassem EH, Price BH, Nierenberg AA, et al. Prospective long-term follow-up of 44 patients who received cingulotomy for treatment-refractory obsessive-compulsive disorder. Am J Psychiatry. 2002 Feb;159(2):269–75. doi: 10.1176/appi.ajp.159.2.269. [DOI] [PubMed] [Google Scholar]
- 68.Baer L, Rauch SL, Ballantine HT, Martuza R, Cosgrove R, Cassem E, et al. Cingulotomy for intractable obsessive-compulsive disorder. Prospective long-term follow-up of 18 patients. Arch Gen Psychiatry. 1995 May;52(5):384–92. doi: 10.1001/archpsyc.1995.03950170058008. [DOI] [PubMed] [Google Scholar]
- 69.Mashour GA, Walker EE, Martuza RL. Psychosurgery: past, present, and future. Brain Res Brain Res Rev. 2005;48(3):409–19. doi: 10.1016/j.brainresrev.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 70.Bush G, Shin LM, Holmes J, Rosen BR, Vogt BA. The Multi-Source Interference Task: validation study with fMRI in individual subjects. Mol Psychiatry. 2003 Jan;8(1):60–70. doi: 10.1038/sj.mp.4001217. [DOI] [PubMed] [Google Scholar]
- 71.Janer KW, Pardo JV. Deficits in selective attention following bilateral anterior cingulotomy. J Cogn Neurosci. 1991;3(3):231–41. doi: 10.1162/jocn.1991.3.3.231. [DOI] [PubMed] [Google Scholar]
- 72.Banks GP, Mikell CB, Youngerman BE, Henriques B, Kelly KM, Chan AK, et al. Neuroanatomical Characteristics Associated With Response to Dorsal Anterior Cingulotomy for Obsessive-Compulsive Disorder. JAMA Psychiatry. 2014 Dec 23; doi: 10.1001/jamapsychiatry.2014.2216. [DOI] [PubMed] [Google Scholar]
- 73.Yang JC, Papadimitriou G, Eckbo R, Yeterian EH, Liang L, Dougherty DD, et al. Multi- tensor investigation of orbitofrontal cortex tracts affected in subcaudate tractotomy. Brain Imaging Behav [Internet] 2014 doi: 10.1007/s11682-014-9314-z. Available from: http://link.springer.com/10.1007/s11682-014-9314-z. [DOI] [PMC free article] [PubMed]
- 74.Kartsounis LD, Poynton A, Bridges PK, Bartlett JR. Neuropsychological correlates of stereotactic subcaudate tractotomy. A prospective study. Brain J Neurol. 1991 Dec;114(Pt 6):2657–73. doi: 10.1093/brain/114.6.2657. [DOI] [PubMed] [Google Scholar]
- 75.Lehman JF, Greenberg BD, McIntyre CC, Rasmussen SA, Haber SN. Rules ventral prefrontal cortical axons use to reach their targets: implications for diffusion tensor imaging tractography and deep brain stimulation for psychiatric illness. J Neurosci Off J Soc Neurosci. 2011 Jul 13;31(28):10392–402. doi: 10.1523/JNEUROSCI.0595-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Jbabdi S, Lehman JF, Haber SN, Behrens TE. Human and monkey ventral prefrontal fibers use the same organizational principles to reach their targets: tracing versus tractography. J Neurosci Off J Soc Neurosci. 2013 Feb 13;33(7):3190–201. doi: 10.1523/JNEUROSCI.2457-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kelly D, Richardson A, Mitchell-Heggs N, Greenup J, Chen C, Hafner RJ. Stereotactic limbic leucotomy: a preliminary report on forty patients. Br J Psychiatry J Ment Sci. 1973;123(573):141–8. doi: 10.1192/bjp.123.2.141. [DOI] [PubMed] [Google Scholar]
- 78.Cho D-Y, Lee W-Y, Chen C-C. Limbic leukotomy for intractable major affective disorders: a 7-year follow-up study using nine comprehensive psychiatric test evaluations. J Clin Neurosci Off J Neurosurg Soc Australas. 2008 Feb;15(2):138–42. doi: 10.1016/j.jocn.2006.10.017. [DOI] [PubMed] [Google Scholar]
- 79.Bourne SK, Sheth SA, Neal J, Strong C, Mian MK, Cosgrove GR, et al. Beneficial effect of subsequent lesion procedures after nonresponse to initial cingulotomy for severe, treatment-refractory obsessive-compulsive disorder. Neurosurgery. 2013;72(2):196–202. doi: 10.1227/NEU.0b013e31827b9c7c. –discussion 202. [DOI] [PubMed] [Google Scholar]
- 80.Rauch SL, Dougherty DD, Cosgrove GR, Cassem EH, Alpert NM, Price BH, et al. Cerebral metabolic correlates as potential predictors of response to anterior cingulotomy for obsessive compulsive disorder. Biol Psychiatry. 2001;50(9):659–67. doi: 10.1016/s0006-3223(01)01188-x. [DOI] [PubMed] [Google Scholar]
- 81.Dougherty DD, Weiss AP, Cosgrove GR, Alpert NM, Cassem EH, Nierenberg AA, et al. Cerebral metabolic correlates as potential predictors of response to anterior cingulotomy for treatment of major depression. J Neurosurg. 2003 Dec;99(6):1010–7. doi: 10.3171/jns.2003.99.6.1010. [DOI] [PubMed] [Google Scholar]