Abstract
Background
The importance of functional outcome (FO) in the treatment of patients with extremity soft tissue sarcoma (STS) has been increasingly recognized in the last three decades. This systematic review aimed to investigate how FO is measured in surgically treated lower-extremity STS patients.
Methods
A systematic search of PubMed, Web of Science, and Scopus was performed based on the PRISMA guidelines. The methodologic quality of the publications was measured using the MINORS tool. The results from the included studies examining measurement types, measures, and time of FO measurement were compiled. The FO pooled mean and standard deviation were calculated as a weighted average for the groups. The validity of the applied measures is reported.
Results
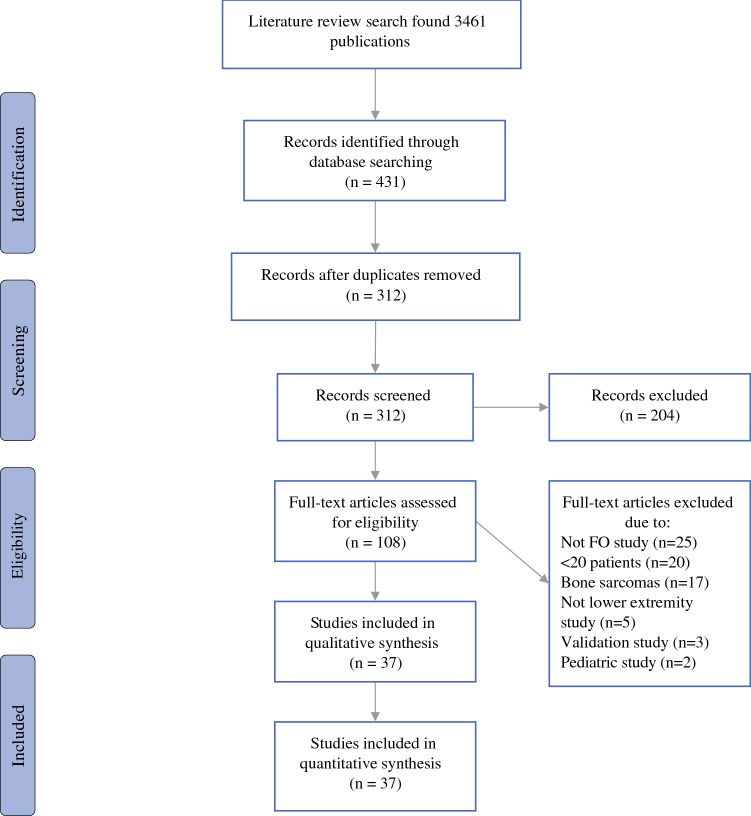
The literature search found 3461 publications, 37 of which met the inclusion criteria. The measurement types used were clinician-reported outcomes (n = 27), patient-reported outcomes (n = 20), and observer-reported outcomes (n = 2). The most frequently used measures were the Toronto Extremity Salvage Score (TESS) (n = 16) and the Musculoskeletal Tumor Society (MSTS) score 1993 (n = 12). The postoperative FO was relatively good. The pooled mean TESS and MSTS 1993 scores were respectively 83.3 and 86.2 (out of 100). Of the 10 previously reported measures, 3 provide validated FO scores. The methodologic quality of publications was generally low.
Conclusions
Based on this systematic review, several different methods exist for assessing FO in patients with lower-extremity sarcoma. The most frequently used measure is a validated TESS. The postoperative FO of patients with lower-extremity STS seems to increase to the preoperative baseline level during long-term follow-up evaluation.
In the last 30 years, limb salvage has become the standard of care in the treatment of extremity sarcoma, and amputations are rare. This has been achieved by improved diagnostics, pre- and postoperative radiotherapy, and more refined reconstructive surgical methods. Reports on functional outcome (FO) have been increasing.1–3
Several methods for assessing FO have been described, including subjective and objective measures.2–4 Functional outcome measures should be valid, reliable, accurate, and clinically meaningful for the population in which the measurement is made.5,6 Consistent usage of the same measures allows benchmarking and comparison of study results over time and between research centers.
A few previous review studies have considered the topic from different perspectives. The review by Davis4 focused on FO of all extremity tissue sarcoma patients, including upper extremities and bone sarcomas. Tang et al.3 investigated quality-of-life studies in adult extremity sarcomas (also including upper extremities and bone sarcomas) and all quality-of-life studies. Furtado et al.2 reviewed physical functioning after treatment for lower- and upper-extremity sarcoma patients, including both bone and soft tissue sarcoma (STS). Only objective measures investigating postural balance, gait, and physical activity were included. All patient-reported outcome (PRO) measurement studies were excluded. Groundland et al.7 investigated pediatric patients. Wilson et al.8 studied pelvic sarcoma patients, and Winnette et al.9 investigated all patients with STS, including abdominal sarcomas. Thus, no systematic literature review has previously focused specifically on measurement of FO after surgical treatment of adult lower-extremity STS patients.
This study aimed to identify how FO has been measured in patients with surgically treated lower-extremity STS. More specifically, we sought to determine the type of methods and measures used to measure FO, whether the measures used had been tested for validity, FO for lower-extremity STS patients, and quality of the publications that report FO.
Materials and Methods
Overview and Eligibility Criteria for Review
A systematic literature review was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.10 A review protocol was created by the authors and is available on request.
The study included all publications concerning patients with surgically treated lower-extremity STS whose FO was measured. The exclusion criteria ruled out duplication, studies that included fewer than 20 lower-extremity STS patients (considered pilot studies),11 non-adult study populations, and publications in languages other than English.
In the current review, the measures for assessing FO were classified as “previously developed and reported measures” or “new measures developed by the authors.” The “previously developed and reported measures” were defined as measures developed to assess FO and published previously. Measures, scales, or questionnaires developed by the authors themselves with the purpose to assess FO only in the reviewed article were considered “new measures developed by the authors.”
To report the type of measurement applied for assessment of FO, we used the terms “patient-reported outcome,” “clinician-reported outcome,” “observer-reported outcome,” and “performance outcome” measures.12 Patient-reported outcome (PRO) measures are based on a patient’s subjective assessment. Clinician-reported outcome measures are based on evaluation by a trained health care professional. Observer-reported outcome measures are based on observation by a person other than the patient or a health professional. Performance outcome is based on measurement of performance in specific tasks and are considered objective measures.
Search Methods
PubMed, Scopus, and Web of Science search engines were used for the search. All published articles were retrieved without a search time constraint on 5 May 2018. The keywords combined with Medical Subject Headings (MeSH) terms were “lower AND (limb OR limbs OR leg OR legs OR extremity OR extremities OR foot) AND sarcoma AND (functional OR functionality OR function OR outcome).”
Two authors (G.K. and M.K.) independently reviewed all titles and appropriate abstracts. All unsuitable articles were excluded by the previously mentioned exclusion criteria. A manual search was performed for all references of suitable studies by review of titles and appropriate abstracts. The included studies were reviewed and added to the final list by the set inclusion criteria. Disagreements in data extraction were resolved by discussion and consensus of the authors (G.K., M.K., K.K., J.R., and I.B.R.).
Study Data
Two authors (G.K. and M.K.) independently collected the following information from the included publications: study period, origin of the study, article type, anatomic location of the tumor, number of patients, age, diagnosis, measures and measurement types used for assessing FO, results of FO, and follow-up time. In case of missing data, an e-mail requesting additional information was sent to the corresponding authors.
Validity Assessment
In this review, a validated FO measure is defined as a measure that has been scientifically validated to assess FO in extremity tumor patients. A literature search was performed to detect suitable literature concerning the validity of the FO measures.
Functional Outcome
In this review, FO is reported from publications that used validated FO measures, and FO results are presented as means ± standard deviations (SDs). The pooled mean and SD were calculated as a weighted average of SDs for the groups. Publications reporting FO for bone sarcoma or upper-extremity STS patients in addition to lower-extremity STS patients were included in the FO report.
Quality of Publications Assessment
Because the quality of a publication can be measured according to many different criteria, and because the validity of these criteria have not been determined, quality was not used to exclude studies in this review.13 The Methodological Index for Nonrandomized Studies (MINORS) quality assessment tool was used to assess the quality of publications.14 The MINORS tool is a valid instrument designed to assess the methodologic quality of nonrandomized surgical studies, either comparative or noncomparative.14 We used the MINORS tool to assess the quality of randomized controlled trials (RCTs), as has been done in previous literature.15
The MINORS tool consists of the following 12 methodologic items for studies: a clearly stated aim, inclusion of consecutive patients, prospective collection of data, end points appropriate for the aim of the study, unbiased assessment of the study end point, follow-up period appropriate for the aim of the study, less than a 5% loss to follow-up evaluation, and prospective calculation of the study size. Additional criteria for comparative studies include adequate statistical analyses, an adequate control group, contemporary groups, and baseline equivalence of groups. The items are scored as follows; 0 (not reported), 1 (reported but inadequate), or 2 (reported and adequate). The maximum score is 16 for non-comparative studies and 24 for comparative studies.14 Results are reported as percentages from 0 to 100.
Results
Study Selection
Details of the literature search are presented in Fig. 1. For the final review, 37 publications were selected.16–52
Fig. 1.
Flow diagram showing flow of studies retrieved for systematic review of functional outcome measurement in patients with lower-extremity soft tissue sarcoma
Study Characteristics
The included studies were published between 1984 and 2018. Of the 31 retrospective and 6 prospective studies, 3 were RCTs. Of the 37 studies, 25 were cross-sectional, 7 were cohort, and 2 were case–control studies. The sample sizes of the studies ranged from 25 to 728 patients. The characteristics of the articles and the FO measurements are presented in Table 1.
Table 1.
Characteristic of the articles and functional outcome measurements
| Author | Year | Country | Study type | Patients | Tumor location | Number of patients | Age (years)a | Measure | Measurement type | Measurement time points | MINORS score |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Slump et al.18 | 2018 | NET/CAN | Re, C | STS, free or pedicled flap reconstruction | LE; UE | 266. LE 181 | 59.2 ± 18.6 | TESS, MSTS 1987, 1993 | PRO, ClinRO | Pre- and postoperatively at 9–12 months | 75 |
| Podleska et al.16 | 2017 | GER | Re, CS | STS, previously nonresectable sarcoma, chemotherapy | LE | 27 | 52.7 (12–73) | SMFA-D | PRO | Minimum 5 months after surgery | 69 |
| Stoeckle et al.17 | 2017 | F | Re, CS | STS, primary operated non-metastatic STS | LE | 728, LE 480 | Median 59 (16–91) | Own | ClinRO | N/P | 44 |
| Saebye et al.19 | 2017 | DEN | Re, CS | STS, treated with first-time limb-sparing surgery without bone resection | LE; UE | 128, LE 90 | Median 61 (IQR 47–70) | TESS, MSTS 1993 | PRO, ClinRO | Minimum 1 year after surgery | 75 |
| Harati20 | 2016 | GER | Re; CS | STS, extending distally to the level of the knee joints | LE | 120 | 57.4 (17–89) | TESS, FF I, SF–36 | PRO | Minimum 1.3 years after surgery | 50 |
| Heaver et al.21 | 2016 | UK | Re, CS | BTS/STS/other tumors, patients underwent limb-salvage surgery | LE; UE | 198, LE 152 | LE: 52.5 (14–86) | TESS | PRO | Minimum 3 months after surgery | 38 |
| Chang et al.22 | 2016 | USA | Re, CS | All tumor types, 77 muscle only flaps, 67 MC flaps and 76 FC flaps | LE;UE | 220. LE 156 | LE: 49.5 ± 18.1 | MSTS 1993, Karnof-sky score | ClinRO | N/P | 71 |
| Furtado et al.23 | 2015 | UK | Re, CS | BTS/STS, primary tumor, management by primary or secondary amputation | LE | 100, LE STS 37 | 53.6 (19–91) | TESS | PRO | Minimum 1 year after surgery | 56 |
| Rivard et al.24 | 2015 | CAN | Pr, C | STS, primary or recurrent sarcoma | LE;UE; T | 52, LE 35 | 57 (24–83) | TESS, MSTS 1993, SF–36 | PRO, ClinRO | Preoperatively and at 6 and 12 months postoperatively | 92 |
| Kang et al.25 | 2014 | S-K | Re, CC | STS, flap reconstruction case-matched with primary wound-closure group | LE; UE | 148, LE 104 | 48.1 ± 19.3 | MSTS 1993 | ClinRO | 12–15 Months after surgery | 71 |
| Townley et al.26 | 2013 | CAN | Re, C | BTS/STS, preoperatively irradiated compared with non-irradiated patients | 46, LE 24 | 57 ± 18 | TESS, MSTS 1987 | PRO, ClinRO | Minimum 1 year after surgery | 63 | |
| Kozawa & Nishida27 | 2012 | JAP | Re, CS | BTS/STS, foot sarcoma. Limb salvage surgery or amputation | LE | 31, 24 STS | 43 (11–76) | ISOLS | ClinRO | Minimum 2 years after surgery | 56 |
| Friedmann et al.28 | 2011 | CAN | Re, C | STS, limb salvage surgery with or without adjuvant external beam radiotherapy | LE; UE | 288. LE 207 | 53 (16–88) | MSTS 1993, TESS | PRO, ClinRO | Minimum 1 year after surgery | 63 |
| Thacker et al.29 | 2008 | USA | Re, CS | STS, foot- and ankle-treated sarcoma patients | LE | 52 | 38 (11–96) | MSTS 1993 | ClinRO | Minimum 2 years after surgery | 63 |
| Tsukushi et al.30 | 2008 | JAP | Re, CS | STS, investigation of results of arterial vs arteriovenous reconstruction | LE | 25 | 46 (18–77) | MSTS 1993 | ClinRO | Minimum 8 months after surgery | 71 |
| Hoy et al.31 | 2006 | USA | Re, CS | BTS/STS/other, sarcoma resection, limb salvage and MFR | 102 MFR, STS 36 | 39 (4–90) | MSTS 1993 | ClinRO | Minimum 4 months after surgery | 50 | |
| Pardasaney et al.32 | 2006 | USA | Re, CS | BTS/STS, amputations and limb salvage | LE | 408 | 49 (11–96) | Own | PRO, ObsRO | Minimum 2 years after surgery | 71 |
| Pradhan et al.33 | 2006 | USA, CAN, UK | Re, CS | STS, treatment of the adductor compartment tumors, 3-center comparative study | LE | 184 | 56 (13–88) | TESS | PRO | N/P | 50 |
| Nelson et al.34 | 2006 | USA | Re, CS | STS, wide resection, functional restoration surgery or soft-tissue coverage with flaps | LE; UE | 67, 48 LE | 52 (13–84) | TESS | PRO | N/P | 54 |
| Thijssens et al.35 | 2006 | NET | Re, CS | STS, limb salvage vs amputation | LE;UE | O 39, LE 33 | Median 59 (15–78) | SF-36b | PRO | Minimum 1 year after surgery | 75 |
| Davis et al.36 | 2005 | CAN | RCT | STS, surgery and pre- or postoperative radiotherapy. Function and radiation morbidity | LE; UE | 129, LE 100 | N/P | MSTS 1987, TESS | PRO, ClinRO | 2 Years | 88 |
| Gerrand et al.37 | 2004 | CAN | Re, C | STS, limb-sparing surgery | LE | 207 | 54 (15–89) | MSTS 1993, TESS | PRO, ClinRO | Pre- and postoperatively within 1–2 years | 71 |
| Rachbauer et al.38 | 2003 | AUS | Pr, CS | STS, marginal surgical resection, combined with both IOHDR and EBRT | LE; UP; T; R; HN | 39, LE 29 | Median 58 (17–87) | MSTS 1993 | ClinRO | Minimum 3 months after surgery | 69 |
| O’Sullivan et al.52 | 2002 | CAN | RCT | STS, randomized by tumor size dichotomized at 10 cm. Pre- and postoperative radiotherapy groups. Wound healing | LE; UE | 182, LE 145 | 54.7 | MSTS 1987, TESS, SF-36 | PRO, ClinRO | Preoperatively and at 6 weeks and at 3, 6, 12 and 24 months postoperatively | 96 |
| Davis et al.39 | 2002 | CAN | RCT | STS, randomized by tumor size dichotomized at 10 cm. Pre- and postoperative radiotherapy groups. Functional outcome | LE; UE | 185, LE 147 | 54.7 | MSTS 1987, TESS, SF-36 | PRO, ClinRO | Preoperatively and at 6 weeks and 3, 6, 12 and 24 months postoperatively | 96 |
| Refaat et al.40 | 2002 | USA | Re, CS | BTS/STS, limb salvage or amputation | LE | 408 | 55 | Own | PRO, ObsRO | N/P | 67 |
| Davis et al.41 | 2000 | CAN | Re, CS | STS, limb-preservation surgery | 172 | 51 | MSTS 1987, 1993, TESS, SF-36 | PRO, ClinRO | Minimum 1 year after surgery | 56 | |
| Davis et al.42 | 1999 | CAN | Re, CC | STS/BTS, amputation matched with limb-sparing surgery | LE | 36 | 32 | TESS, SF-36, RNL | PRO | Minimum 1 year after surgery | 75 |
| Colterjohn et al.43 | 1997 | CAN | Re, C | STS/fibromatosis, limb-salvage surgery for foot and ankle | LE | 30 | 51 | MSTS 1987 | ClinRO | See comment | 56 |
| Pitcher & Thomas44 | 1994 | UK | Re, CS | STS, “functional compartmental resection” | LE | 24 | Median 47 (12–79) | Own | ClinRO | Minimum 1 month after surgery | 50 |
| Keus et al.45 | 1994 | NET | Re, CS | STS, local treatment with curative intent and no known distant metastases | LE; UE | 156, LE 115 | Median 46 (4–89) | Own | ClinRO | N/P | 38 |
| Moseley46 | 1992 | USA | Pr, C | STS, limb salvage using neoadjuvant chemotherapy, 2 groups | LE; UE | 38, LE 27 | Median 46/54c | Own | ClinRO | N/P | 79 |
| Karasek et al.47 | 1992 | USA | Re, CS | STS/fibromatosis | LE; UE; T | 41, LE 25 | 50 (13–85) | Own | ClinRO | Minimum 7 months after surgery | 63 |
| Robinson et al.48 | 1991 | UK | Re, CS | STS, treated with combination of surgery and radiotherapy; surgery alone; irradiation and intra-arterial doxorubicin | LE; UE; P | 54, LE 46 | 52 (23–82) | Own | ClinRO | Minimum 24 months after surgery | 31 |
| Stinson et al.49 | 1991 | USA | Re, CS | STS, limb-sparing surgery, radiation therapy with or without adjuvant chemotherapy | LE; UE | 145 | N/P | Own | ClinRO | Minimum 1 year after surgery | 38 |
| Talbert et al.50 | 1990 | USA | Re, CS | STS/unspecified, non-metastatic patients | LE; UE | 78, LE 39 | Age zones presented | Own | ClinRO | Minimum 28 months after surgery | 44 |
| Lampert et al.51 | 1984 | USA | Re, CS | STS, wide local resection and radiation therapy | LE; UP; T; HN | 40, 20 LE STS | 37 (15–67) | Own, Convery scale | ClinRO | Minimum 7 months after surgery | 56 |
NET The Netherlands, CAN Canada, GER Germany, F France, DEN Denmark, UK United Kingdom, USA United States of America, S-K South Korea, JAP Japan, AUS Australia, Re retrospective study, C cohort study, CS cross-sectional study, Pr prospective study, CC case–control study, RCT randomized control trial, STS soft tissue sarcoma, BTS bone tissue sarcoma, IOHDR intraoperative brachytherapy, EBRT external beam irradiation, LE lower-extremity, UE upper extremity, T trunk, R retroperitoneum, HN head and neck, P pelvic, MFR muscle flap reconstructions, TESS Toronto extremity salvage score, MSTS Musculoskeletal Tumor Society, SMFA-D the German Short Musculoskeletal Function Assessment questionnaire, FFI Foot Function Index, ISOLS International Society Of Limb Salvage, RNL Reintegration to Normal Living, PRO patient-reported outcome, ClinRO clinician-reported outcome, ObsRO observer-reported outcome
aPresented as mean age, unless otherwise stated
bMeasure used was RAND-36
cGroup A median, 46 years; Group B median, 54 years
Type of Measurement to Assess FO
A single PRO measurement was used in 8 of the 37 publications, and a clinician-reported outcome measurement was used in 17 of the 37 publications. The PRO and clinician-reported outcome measurement types were used together in 10 studies. The PRO and observer-reported outcome measures were used in two studies. No performance outcome measurement was used. All six prospective studies used clinician-reported outcome measures, and four of these also used a PRO measure (Table 2).
Table 2.
Previously developed and reported patient-reported outcome (PRO) and clinician-reported outcome (ClinRO) tools
| Measure | Measurement type | n | Studies | Valid | |||||
|---|---|---|---|---|---|---|---|---|---|
| R (n = 31) | P (n = 6) | CS (n = 25) | C (n = 7) | CC (n = 2) | RCT (n = 3) | ||||
| TESS | PRO | 16 | 12 | 4 | 7 | 5 | 1 | 3 | + |
| MSTS 1993 | ClinRO | 12 | 10 | 2 | 7 | 4 | 1 | 0 | + |
| MSTS 1987 | ClinRO | 7 | 4 | 3 | 1 | 3 | 0 | 3 | + |
| SF-36 | PRO | 7 | 5 | 2 | 3 | 1 | 1 | 2 | |
| SMFA | PRO | 1 | 1 | 0 | 1 | 0 | 0 | 0 | |
| FFI | PRO | 1 | 1 | 0 | 1 | 0 | 0 | 0 | |
| Karnofsky score | ClinRO | 1 | 1 | 0 | 1 | 0 | 0 | 0 | |
| Modified MSTS 1993 (ISOLS) | ClinRO | 1 | 1 | 0 | 1 | 0 | 0 | 0 | |
| RNL | PRO | 1 | 1 | 0 | 0 | 0 | 1 | 0 | |
| Convery scale | ClinRO | 1 | 1 | 0 | 1 | 0 | 0 | 0 | |
R retrospective study, P prospective study, CS cross-sectional study, C Cohort study, CC Case–control study, RCT randomized clinical trial, TESS Toronto extremity salvage score, MSTS Musculoskeletal Tumor Society, SMFA Short Musculoskeletal Function Assessment, FFI Foot Function Index, ISOLS International Society Of Limb Salvage, RNL Reintegration to Normal Living, PRO patient-reported outcome, ClinRO clinician-reported outcome
FO Measures
The majority of the publications (27/37) used previously reported measures. The most common measures were the Toronto Extremity Salvage Score (TESS) (43%, n = 16) and the Musculoskeletal Tumor Society (MSTS) score 1993 (32%, n = 12). A total of 10 different previously reported measurement tools were used (Table 2). New measures developed by the authors were used in 10 publications. Of the 27 publications, one previously reported measure alone was used in 14 publications, two and three measures in 6 publications each, and four measures in 1 publication.
The RAND-36 is multidimensional PRO questionnaire identical to the Short Form 36 (SF-36) but uses a different scoring method.35 In this review, RAND-36 and SF-36 are reported together.
Timing of FO Measurement
In 4 (11%) of the 37 studies, FO was measured both pre- and postoperatively.24,37,39,52 More than one postoperative time point of measurement was used in 3 (8%) of the 37 studies.24,39,52 The time of FO measurement varied in the 31 retrospective studies. The inclusion criteria required a minimum follow-up period of 1 year in most of the studies. In 18 (58%) of the 31 retrospective studies and 12 (50%) of the 24 retrospective cross-sectional studies, the FO was measured at least 1 year after the surgery. The time point of measurement was not reported in seven studies, and additional information was not available. Additional details are presented in Table 1.
Validity of the Measures
Validated FO measures were used in 23 (62%) of the 37 publications. the validated measures were the TESS1,53 and MSTS 19854,71 and 19931,55 (Table 2). The SF-3656 validation also has been studied, but its validity as a extremity FO measure in this population is questionable.1 In addition, the validity of the Short Musculoskeletal Function Assessment (SMFA) questionnaire,57,58 the Foot Function Index (FFI),59,60 the Karnofsky score,61 and the Reintegration to Normal Living Index (RNL)62,63 measures also has been studied. However, these measures were studies in a different population of patients or as a general FO measure, or both. No studies on the validity or reliability of the modified International Society Of Limb Salvage (ISOLS)/MSTS 199327 scoring system or the modified Convery scale64 were found.
Functional Outcome
The TESS, MSTS 1993, or MSTS 1987 results were presented in 23 of the 37 studies. In some studies, not all the required results (mean or SD of FO results, number of lower- vs upper-extremity patients, number of STS vs other tumors) were presented. Additional information was requested from 15 corresponding authors. Five replied, with two supplying the requested additional information.
Of the 23 studies, 3 were excluded from our FO report. The reasons were as follows: only the difference in means was reported;18 several TESS scores from the same patients were included (in lower-extremity patients, a mean of 2.6 TESS results for one patient was included);21 and two studies used the same data set (a study by O’Sullivan was excluded).39,52 Means and SDs for a study by Saebye et al.19 were calculated using median and IQR results.65,66 The SDs for studies by Friedmann et al.28 and Pradhan et al.33 were approximated based on range using the range rule calculation formula (SD = [max − min]/4). The pre- and postoperative function results are presented in Table 3.
Table 3.
Pre- and postoperative function scores. Results of the TESS, MSTS 1993 and MSTS 1987 measures
| Author | Year | LE patients (n) | Mean TESS (SD)a | Mean MSTS 1993 (SD)a | Mean MSTS 1987 (SD)b | Comments |
|---|---|---|---|---|---|---|
| Preoperative | ||||||
| Rivard | 2015 | 35 | 79.8 (20.6) | 78.8 (20.7) | NA | FO results including UE STS patients (n = 15). Results available for MSTS from 45 and for TESS from 48 patients |
| Gerrand | 2004 | 207 | 83.1 (20.1) | 87.9 (19.6) | NA | Results available for MSTS from 203 and for TESS from 172 patients |
| Davis | 2002 | 147 | 84.4 (19.0) | NA | 26.8 (4.4) | FO results including UE STS patients (n = 38) |
| Postoperative | ||||||
| Saebye | 2017 | 90 | 94 (11.3) | 92.8 (13.6) | NA | FO results including UE STS patients (n = 38) |
| Harati | 2016 | 120 | 63.8 (17.0) | NA | NA | FO results available for 30 patients |
| Chang | 2016 | 129 | NA | 80.2 (NP) | NA | FO results including UE STS and other tumors. Excluded from the pooled mean and SD analysis due to missing SD data |
| Furtado | 2015 | 37 | 56.4 (23.3) | NA | NA | TESS results including bone sarcoma patients (n = 63) |
| Rivard | 2015 | 35 | 87.1 (16.6) | 88.8 (11.9) | NA | FO results including UE STS patients (n = 15). Results available for MSTS from 37 and for TESS from 41 patients |
| Kang | 2014 | 104 | NA | 85.4 (13.9) | NA | FO result including UE STS patients (n = 44) |
| Townley | 2013 | 21 | 84.76 (NP) | NA | 29.73 (NP) | FO results including bone sarcoma patients. Excluded from the pooled mean and SD analysis due to missing SD data |
| Friedmann | 2011 | 204 | 89.4 (32.4–100)c | NA | 32 (11–35)c | FO results including UE STS patients (n = 59) |
| Thacker | 2008 | 52 | NA | 83.3 (11.5) | NA | FO results available for 30 sarcoma patients |
| Tsukushi | 2008 | 25 | NA | 70 (NP) | NA | |
| Hoy | 2006 | 70 | NA | 90.3 (NP) | NA | FO results including UE STS (n = 6) and bone sarcoma (n = 39) patients |
| Pradhan | 2006 | 184 | 77 (23–100)c | NA | NA | FO results available for 70 patients |
| Nelson | 2006 | 48 | 85.1 (19.3) | NA | NA | FO results available for 34 patients |
| Davis | 2005 | 100 | 83.2 (21.8) | NA | 28.9 (9.2) | FO results including UE STS patients (n = 29) |
| Gerrand | 2004 | 207 | 82.7 (17.7) | 85.8 (19.0) | NA | Results available for MSTS from 189 and for TESS from 155 patients |
| Rachbauer | 2003 | 29 | NA | 88.5 (NA) | NA | FO results including UE STS (n = 6) and other anatomic location sarcomas (n = 4) |
| Davis | 2002 | 147 | 80.3 (21.1) | NA | 28.6 (7.8) | FO results including UE STS patients (n = 38). Results available for MSTS from 163 and for TESS from 156 patients |
| Davis | 2000 | 172 | 82.7 (18.7) | 84.8 (20.4) | 30.0 (6.2) | |
| Davis | 1999 | 29 | 81.6 (17.8) | NA | NA | FO results including bone sarcoma patients (n = 7) |
| Colterjohn | 1997 | 29 | NA | NA | 31.4 (4.6) | FO results available for 26 patients |
TESS Toronto extremity salvage score, MSTS Musculoskeletal Tumor Society, LE lower-extremity, SD standard deviation, NA not available, NP not presented, UE upper extremity, STS soft tissue sarcoma, FO functional outcome
aScores are presented as range of 0 (minimum) to 100 (maximum)
bScores are presented as range of 0 (minimum) to 35 (maximum)
cRange (min–max)
After study selection and additional gathering of information, some studies still included upper-extremity or bone sarcoma patients.
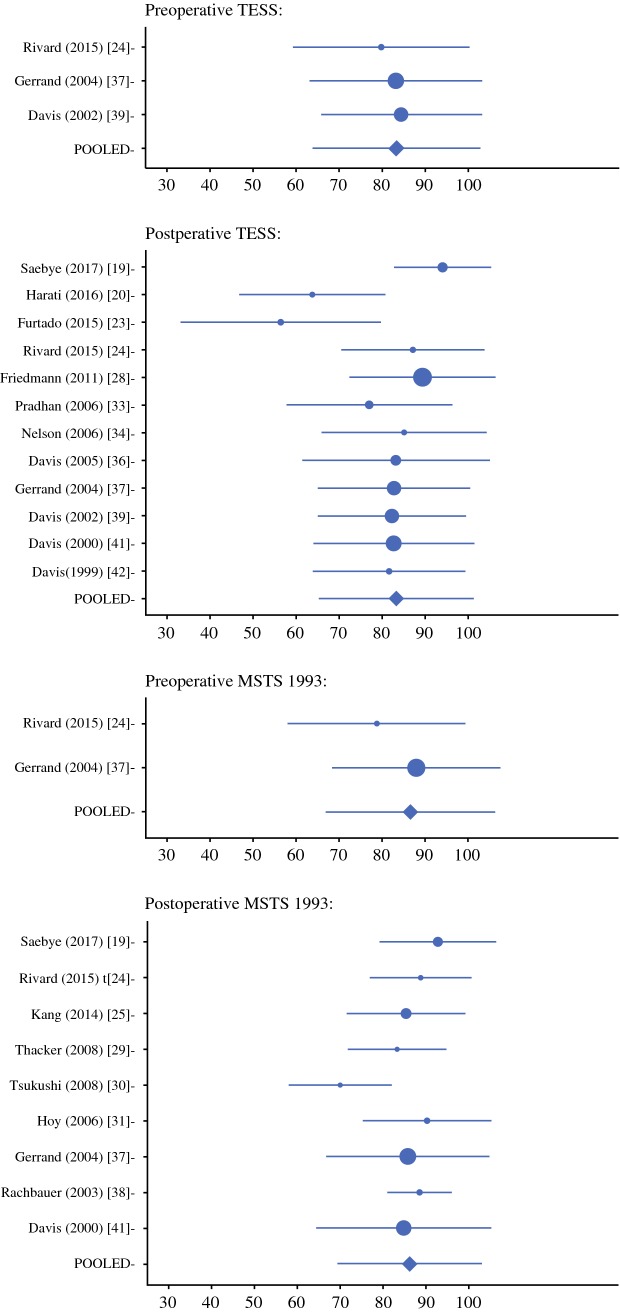
The pooled mean and SD were calculated for TESS and MSTS 1993 results. Of 19 studies, 2 were excluded due to missing SD data. The pre- and postoperative TESS and MSTS 1993 pooled mean and SD results are presented in Fig. 2. Because not all the publications included only lower-extremity patients, TESS results for both lower- and upper-extremity STS patients were included in 2 of 3 preoperative and 5 of 12 postoperative studies. Similarly, MSTS 1993 results included both lower- and upper-extremity STS patients in 1 of 2 preoperative and 5 of 9 postoperative studies. In 3 of 17 studies, the TESS (n = 2) and MSTS 1993 (n = 1) results included other extremity tumor patients in addition to STS patients. The number of included patients is described in Table 3.
Fig. 2.
Pre- and postoperative TESS and MSTS 1993 pooled mean and SD results
The preoperative pooled mean TESS and MSTS 1993 scores were respectively 83.3 and 86.6, and the postoperative scores were respectively 83.3 and 86.2 (out of 100). In the pooled mean and SD analysis, the proportion of lower-extremity and STS patients were respectively 88% and 94%.
As a sensitivity analysis, the mean overall postoperative FO in publications including also upper-extremity or bone sarcoma patients was investigated. In publications including upper-extremity patients (7 of 17 publications), the mean overall TESS score was 86.7 (5 publications), and the MSTS score was 89.0 (5 publications). In publications that included also bone sarcomas (3 of 17 publications), the mean overall TESS score was 63.1 (2 publications), and the MSTS score was 90.3 (1 publication).
Quality of Publications
The mean MINORS score was 62.2 (median, 62.4; range, 31–92). For the three RCTs, the respective scores were 96,39 96,52 and 88.36 For the observational studies, the mean MINOR score was 60.2 (median, 62.5; range, 31–92). The MINORS scores for each study are presented in Table 1.
Discussion
Based on the current systematic literature review, the most frequently used measurement types used to measure FO for surgically treated adult lower-extremity STS patients are clinician-reported outcome and PRO measurements. The most frequently used measures are the TESS and the MSTS 1993 questionnaires. Of the 10 previously reported measures, 3 were proven to provide valid scores for lower-extremity sarcoma patients. Most of the studies on lower-extremity STS FO have poor methodologic quality.
Methods and Measures of FO
The majority of the included studies used clinician-reported outcome measurement. In the last 10 years, the use of PRO measurements has increased. Furtado et al.2 considered performance outcome an important component of FO in sarcoma patients. This clearly is a minority position in their study because no performance outcome measurement type was found. Performance outcome measurement type has been used more frequently in publications with smaller study samples.2 This may be due to the greater time requirements and equipment and personnel costs required for performance outcome measurements.2
In the literature review of Furtado et al.2 that investigated performance outcome measurement techniques, the study size ranged from 4 to 82 patients. In the current review, one of the inclusion criteria was a minimum of 20 lower-extremity STS patients. In our study, the sample size ranged from 25 to 728 patients.
When FO is assessed in a study, it is important to avoid loss to follow-up evaluation. Loss to follow-up evaluation less than 5% is indicated in the MINORS publications’ quality measurement tool as an indicator of a good-quality study.14 More important than the measurement type is that the outcome measure provide valid and reliable scores in measuring FO. However, the type of measurement should be chosen based on the objectives of the assessment, the available resources, and the aspects of FO that are of interest.
According to the current review, the TESS and MSTS 1993 questionnaires have been used most frequently. Tang et al.3 observed that the most frequently used outcome measures were the TESS, the MSTS 1987, and the SF-36. However, they observed that the TESS was used only four times, the MSTS 1987 three times, and the MSTS 1993 two times. They also found that when the MSTS was used, the 1987 version was preferred. Likewise, Groundland et al.7 found that the MSTS is the most widely used FO measure for pediatric patients after limb-preservation surgery.
Winnette et al.9 investigated patient experience with STS in all anatomic locations, including abdominal sarcomas. They found that in extremity patients, the RNL and the TESS were used three times each. The MSTS questionnaire was not presented in their review because it examined only PRO measures.
Wilson et al.8 found that the MSTS was the most frequently used measure and that the TESS was presented only once for pelvic sarcoma patients. Some studies have used both the older and newer versions of the MSTS questionnaires in the same study.18,41 Other measures such as the SF-36, the FFI, the Karnofsky score, the RNL, the SMFA, the modified MSTS 93/ISOLS, and the modified Convery scale64 have been used but much less frequently.
The MSTS 198754 is a clinician-reported outcome assessment that evaluates seven parameters of FO (mobility, pain, stability, deformity, strength, functional, and emotional acceptance). The MSTS 1993,55 a revised version of the MSTS 1987, also is assessed by physicians. However, the MSTS 1993 is more limb-specific than the older version and includes six parameters. Pain, function, and emotional acceptance are measured for both extremities. For the lower-extremity, use of walking aids, gait, and walking are evaluated. Hand positioning, dexterity, and lifting ability are evaluated for the upper extremity.
The TESS was developed for limb sarcoma patients. As a PRO questionnaire, it measures physical disability and performance in activities of daily living.53 Janssen et al.67 found that the TESS has adequate coverage and is more reliable than the MSTS questionnaire. In addition, they found that the MSTS score is the least reliable, as indicated by a high standard error of measurement for the complete range of ability scores. Although both versions of MSTS are the most frequently used tool for assessing FO, the TESS tool is more frequently used than the two MSTS versions separately. The TESS also was developed later than the MSTS measures.53–55
The study by Tang et al.3 included only articles published in last 10 years. The TESS was used in four publications, the MSTS 87 in three publications, and the MSTS 93 in two publications. Because of the relatively wide use of MSTS scores, the MSTS questionnaires permit comparison of results more easily and widely than in other studies. On the other hand, the MSTS questionnaire must be completed by a clinician, thus limiting its use in studies with larger samples.
The TESS questionnaire was designed to be completed by patients and can therefore be administered by mail or electronically. This is important particularly in long-term follow-up studies. Using the TESS may avoid the need for a physician consultation, thus saving resources. Using PRO measures may make it easier for patients to participate in the study. Also, minimal clinically important differences (MCIDs) are calculated for the TESS.68 Because the MSTS measure was developed by orthopedics for surgically treated bone and soft tissue musculoskeletal tumors, it may not capture the effects of radiotherapy, chemotherapy, and other factors that also affect FO.16,36,48,49,69 Although all FO measurements have limitations, the use of standardized instruments is important. Using the TESS and MSTS measures allows for benchmarking and comparison of results with other studies.
According to the results of our literature review, most studies used only one FO measure. The use of more than one FO measure provides more precise information on FO. For example, the TESS measures activity limitations, whereas the MSTS measures impairment in extremity sarcoma patients.53,55 On the other hand, using too many time-consuming questionnaires could lead to decreased participation and loss to follow-up evaluation. In addition, using more than one FO measuring tool might not be clinically relevant.70 Study participants should be informed about the patient burden, including how many items must be completed, how long it takes to complete the questionnaires for participation in the assessment, and how many assessments are made. Questionnaires implemented in a clinical study should be carefully chosen to ensure that they are fit for the purpose.71–74
Most retrospective studies required a minimum follow-up period of 1 year after surgery. In addition, FO showed a decrease up to 6 months after the surgery. Beyond 1 year, FO plateaued before the scores returned to approximate pre-treatment levels 1 year after surgery.39,43,75
Rivard et al.24 did not observe significant changes in the TESS and MSTS scores from the preoperative period to 6 months after surgery. By 12 months, the scores showed significant improvement. This is an important factor when prospective data measurement in retrospective samples is planned. In cross-sectional studies and other prospective studies, it is important to consider measuring FO at least 1 year after the surgery, in addition to other measurement time points.
Validity of Measures
The validity of a measurement tool is a multi-dimensional term. The most important measurement property is content validity. The measure should be relevant, comprehensive, and comprehensible with respect to the construct of interest and the study population.76 Structural validity is the degree to which the scores adequately reflect the dimensionality of the construct to be measured.39,77 This review considered the measure to be validated when it was tested in lower-extremity patients and reported to be a valid measurement tool.
Half of all the studies reviewed (23/37 studies) used validated tools. In 1999, Davis4 found that studies often did not use standardized, validated measures. Several measurement tools currently in routine use had been available for only a few years in 1999. Based on our review and on the previous literature, it seems that although the psychometric properties of several PRO and clinician-reported outcome measures have been extensively studied in the last decade, performance outcome measures lack quality in this field.2
In a systematic review of objective measurement methods, Furtado et al.2 found that only a few studies investigated aspects of validity of outcome measures. For example, they found that only 1 in 18 studies investigated reliability. They concluded that this raises questions about the accuracy of the objective (in our terminology, performance outcome) measures and veracity of the results.2
FO of Lower-Extremity STS Patients
The data sample in the current review included a heterogeneous group of lower-extremity STS patients. According to the current review and analysis, the postoperative FO for patients is relatively good. The mean postoperative FO measured by MSTS for patients with extremity osteosarcoma is reported to range between 40% and 76.6%.78 In pediatric bone sarcoma patients, the reported postoperative MSTS mean scores ranged from 76% to 82.5%.7 In a review of pelvic sarcoma patients, the mean MSTS score was 65%.8
Amputation seems to decrease FO.23,78 Our FO data included relatively few articles on amputation. The MSTS 1993 FO analysis did not include any articles and the TESS analysis included only two articles on amputations.23,42 In the work of Davis et al.42 the mean TESS score for the patients with amputations was 74.5 versus 85.1 for the patients with limb-sparing procedures. In the study by Furtado et al.23 the mean TESS score was only 56.4. Han et al.78 performed a meta-analysis of osteosarcoma patients and observed that the number of amputation patients was relatively higher. The mean MSTS for the amputation patients ranged from 41.1% to 71% versus 70% to 76.6% for the patients with limb-sparing procedures, which is lower than the result of the current review. As expected, some studies show that amputation decreases FO,23,78 whereas others have failed to show a significant difference compared with limb-sparing treatment.79–81 Based on this review, lower-extremity STS patients seem to achieve preoperative function levels postoperatively during long-term follow-up evaluation (> 1 year).
The Quality of the Publications Reporting FO
This review included different types of studies examining varied methodologic quality. The use of the MINORS tool showed that most studies investigating lower-extremity STS FO are lacking in methodologic quality (median MINORS score, 62.4%; range, 31–92).
This is the first systematic review of FO measurement to focus on lower-extremity STS patients. The strengths of this review were the use of PRISMA guidelines and the methodologic quality assessment for this topic. The main weakness of the current review was the nonstructural search strategy for validity of FO measurement tools used. In addition, one exclusion criterion was to have “fewer than 20 lower-extremity STS patients in the study (considered a pilot study).” This might have excluded some relevant studies with small samples. Because the authors attempted to overview the existing literature on FO measurement of lower-extremity STS patients as thoroughly as possible, they included some studies containing small amounts of upper-extremity and bone sarcomas. Because the number of non–lower extremity STS patients in the reviewed studies was small (88% lower-extremity and 94% STS patients), and because the sensitivity analysis presented similar FO results for publications reporting on upper-extremity or bone sarcoma patients, the effect on FO results was small.
Conclusion
The most frequently used FO measurements for surgically treated adult lower-extremity STS patients are clinician-reported outcome and PRO measurements. The most widely used measure is the patient-reported TESS instrument, which has been shown to produce reliable and valid scores in assessing FO for lower-extremity sarcoma patients. Using the TESS and MSTS measures allows for benchmarking and comparison of results with other studies. Functional outcome scores seem to return to pretreatment levels 1 year after surgery. Thus, measurement of FO also should be performed at least once 1 year after surgery or later in addition to other time points. This review indicates that quality is lacking in FO studies examining surgical treatment of lower-extremity STS.
Acknowledgments
Open access funding provided by University of Helsinki including Helsinki University Central Hospital.
Disclosure
There are no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Davis AM, Bell RS, Badley EM, et al. Evaluating functional outcome in patients with lower extremity sarcoma. Clin Orthop Relat Res. 1999;90–100. [PubMed]
- 2.Furtado S, Errington L, Godfrey A, et al. Objective clinical measurement of physical functioning after treatment for lower extremity sarcoma: a systematic review. Eur J Surg Oncol. 2017;43:968–993. doi: 10.1016/j.ejso.2016.10.002. [DOI] [PubMed] [Google Scholar]
- 3.Tang MH, Pan DJ, Castle DJ, Choong PF. A systematic review of the recent quality-of-life studies in adult extremity sarcoma survivors. Sarcoma. 2012;2012:171342. doi: 10.1155/2012/171342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Davis AM. Functional outcome in extremity soft tissue sarcoma. Semin Radiat Oncol. 1999;9:360–368. doi: 10.1016/s1053-4296(99)80030-7. [DOI] [PubMed] [Google Scholar]
- 5.MacDermid JC, Grewal R, MacIntyre NJ. Using an evidence-based approach to measure outcomes in clinical practice. Hand Clin. 2009;25(97–111):vii. doi: 10.1016/j.hcl.2008.11.001. [DOI] [PubMed] [Google Scholar]
- 6.van Dam MS, Kok GJ, Munneke M, et al. Measuring physical activity in patients after surgery for a malignant tumour in the leg: the reliability and validity of a continuous ambulatory activity monitor. J Bone Joint Surg Br. 2001;83:1015–1019. doi: 10.1302/0301-620x.83b7.12195. [DOI] [PubMed] [Google Scholar]
- 7.Groundland JS, Ambler SB, Houskamp LD, et al. Surgical and functional outcomes after limb-preservation surgery for tumor in pediatric patients: a systematic review. JBJS Rev. 2016 doi: 10.2106/JBJS.RVW.O.00013. [DOI] [PubMed] [Google Scholar]
- 8.Wilson RJ, Freeman TH, Jr, Halpern JL, et al. Surgical outcomes after limb-sparing resection and reconstruction for pelvic sarcoma: a systematic review. JBJS Rev. 2018;6:e10. doi: 10.2106/JBJS.RVW.17.00072. [DOI] [PubMed] [Google Scholar]
- 9.Winnette R, Hess LM, Nicol SJ, et al. The patient experience with soft tissue sarcoma: a systematic review of the literature. Patient. 2017;10:153–162. doi: 10.1007/s40271-016-0200-1. [DOI] [PubMed] [Google Scholar]
- 10.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62:1006–1012. doi: 10.1016/j.jclinepi.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 11.Stephen Isaac WBM. Handbook in Research and Evaluation: A Collection of Principles, Methods, and Strategies Useful in the Planning, Design, and Evaluation of Studies in Education and the Behavioral Sciences. San Diego, CA: Educational and Industrial Testing Services; 1995. [Google Scholar]
- 12.Center for Drug Evaluation and Research (CDER), Food and Drug Administration. Qualification Process for Drug Development Tools. Guidance document, 2014.
- 13.Elwood M. Critical Appraisal of Epidemiological Studies and Clinical Trial. Oxford: Oxford University Press; 2007. [Google Scholar]
- 14.Slim K, Nini E, Forestier D, et al. Methodological index for nonrandomized studies (MINORS): development and validation of a new instrument. ANZ J Surg. 2003;73:712–716. doi: 10.1046/j.1445-2197.2003.02748.x. [DOI] [PubMed] [Google Scholar]
- 15.Smeeing DPJ, van der Ven DJC, Hietbrink F, et al. Surgical versus nonsurgical treatment for midshaft clavicle fractures in patients aged 16 years and older: a systematic review, meta-analysis, and comparison of randomized controlled trials and observational studies. Am J Sports Med. 2017;45:1937–1945. doi: 10.1177/0363546516673615. [DOI] [PubMed] [Google Scholar]
- 16.Podleska LE, Kaya N, Farzaliyev F, et al. Lower limb function and quality of life after ILP for soft-tissue sarcoma. World J Surg Oncol. 2017;15:84. doi: 10.1186/s12957-017-1150-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stoeckle E, Michot A, Rigal L, et al. The risk of postoperative complications and functional impairment after multimodality treatment for limb and trunk wall soft tissue sarcoma: long-term results from a monocentric series. Eur J Surg Oncol. 2017;43:1117–1125. doi: 10.1016/j.ejso.2017.01.018. [DOI] [PubMed] [Google Scholar]
- 18.Slump J, Hofer SOP, Ferguson PC, et al. Flap choice does not affect complication rates or functional outcomes following extremity soft tissue sarcoma reconstruction. J Plast Reconstr Aesthet Surg. 2018;71:989–996. doi: 10.1016/j.bjps.2018.04.002. [DOI] [PubMed] [Google Scholar]
- 19.Saebye C, Fugloe HM, Nymark T, et al. Factors associated with reduced functional outcome and quality of life in patients having limb-sparing surgery for soft tissue sarcomas: a national multicenter study of 128 patients. Acta Oncol. 2017;56:239–244. doi: 10.1080/0284186X.2016.1268267. [DOI] [PubMed] [Google Scholar]
- 20.Harati K, Kirchhoff P, Behr B, et al. Soft tissue sarcomas of the distal lower extremities: a single-institutional analysis of the prognostic significance of surgical margins in 120 patients. Oncol Rep. 2016;36:863–870. doi: 10.3892/or.2016.4862. [DOI] [PubMed] [Google Scholar]
- 21.Heaver C, Isaacson A, Gregory JJ, et al. Patient factors affecting the Toronto extremity salvage score following limb salvage surgery for bone and soft tissue tumors. J Surg Oncol. 2016;113:804–810. doi: 10.1002/jso.24247. [DOI] [PubMed] [Google Scholar]
- 22.Chang EI, Nguyen AT, Hughes JK, et al. Optimization of free-flap limb salvage and maximizing function and quality of life following oncologic resection: 12-year experience. Ann Surg Oncol. 2016;23:1036–1043. doi: 10.1245/s10434-015-4905-5. [DOI] [PubMed] [Google Scholar]
- 23.Furtado S, Grimer RJ, Cool P, et al. Physical functioning, pain, and quality of life after amputation for musculoskeletal tumours: a national survey. Bone Joint J. 2015;97-B:1284–90. [DOI] [PubMed]
- 24.Rivard JD, Puloski SS, Temple WJ, et al. Quality of life, functional outcomes, and wound complications in patients with soft tissue sarcomas treated with preoperative chemoradiation: a prospective study. Ann Surg Oncol. 2015;22:2869–2875. doi: 10.1245/s10434-015-4490-7. [DOI] [PubMed] [Google Scholar]
- 25.Kang S, Han I, Kim S, et al. Outcomes after flap reconstruction for extremity soft tissue sarcoma: a case-control study using propensity score analysis. Eur J Surg Oncol. 2014;40:1101–1108. doi: 10.1016/j.ejso.2014.05.004. [DOI] [PubMed] [Google Scholar]
- 26.Townley WA, Mah E, O’Neill AC, et al. Reconstruction of sarcoma defects following pre-operative radiation: free tissue transfer is safe and reliable. J Plast Reconstr Aesthet Surg. 2013;66:1575–1579. doi: 10.1016/j.bjps.2013.06.029. [DOI] [PubMed] [Google Scholar]
- 27.Kozawa E, Nishida Y, Nakashima H, et al. Foot sarcomas: factors affecting oncological and functional outcomes. Oncol Lett. 2012;3:82–88. doi: 10.3892/ol.2011.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Friedmann D, Wunder JS, Ferguson P, et al. Incidence and severity of lymphoedema following limb salvage of extremity soft tissue sarcoma. Sarcoma. 2011;2011:289673. doi: 10.1155/2011/289673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Thacker MM, Potter BK, Pitcher JD, Temple HT. Soft tissue sarcomas of the foot and ankle: impact of unplanned excision, limb salvage, and multimodality therapy. Foot Ankle Int. 2008;29:690–698. doi: 10.3113/FAI.2008.0690. [DOI] [PubMed] [Google Scholar]
- 30.Tsukushi S, Nishida Y, Sugiura H, et al. Results of limb-salvage surgery with vascular reconstruction for soft tissue sarcoma in the lower extremity: comparison between only arterial and arterovenous reconstruction. J Surg Oncol. 2008;97:216–220. doi: 10.1002/jso.20945. [DOI] [PubMed] [Google Scholar]
- 31.Hoy E, Granick M, Benevenia J, et al. Reconstruction of musculoskeletal defects following oncologic resection in 76 patients. Ann Plast Surg. 2006;57:190–194. doi: 10.1097/01.sap.0000216255.18106.e1. [DOI] [PubMed] [Google Scholar]
- 32.Pardasaney PK, Sullivan PE, Portney LG, Mankin HJ. Advantage of limb salvage over amputation for proximal lower-extremity tumors. Clin Orthop Relat Res. 2006;444:201–208. doi: 10.1097/01.blo.0000195413.16150.bc. [DOI] [PubMed] [Google Scholar]
- 33.Pradhan A, Cheung YC, Grimer RJ, et al. Does the method of treatment affect the outcome in soft-tissue sarcomas of the adductor compartment? J Bone Joint Surg Br. 2006;88:1480–1486. doi: 10.1302/0301-620X.88B11.17424. [DOI] [PubMed] [Google Scholar]
- 34.Nelson AA, Frassica FJ, Gordon TA, Deune EG. Cost analysis of functional restoration surgery for extremity soft-tissue sarcoma. Plast Reconstr Surg. 2006;117:277–283. doi: 10.1097/01.prs.0000187140.83705.cf. [DOI] [PubMed] [Google Scholar]
- 35.Thijssens KM, Hoekstra-Weebers JE, van Ginkel RJ, Hoekstra HJ. Quality of life after hyperthermic isolated limb perfusion for locally advanced extremity soft tissue sarcoma. Ann Surg Oncol. 2006;13:864–871. doi: 10.1245/ASO.2006.05.023. [DOI] [PubMed] [Google Scholar]
- 36.Davis AM, O’Sullivan B, Turcotte R, et al. Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol. 2005;75:48–53. doi: 10.1016/j.radonc.2004.12.020. [DOI] [PubMed] [Google Scholar]
- 37.Gerrand CH, Wunder JS, Kandel RA, et al. The influence of anatomic location on functional outcome in lower-extremity soft tissue sarcoma. Ann Surg Oncol. 2004;11:476–482. doi: 10.1245/ASO.2004.07.016. [DOI] [PubMed] [Google Scholar]
- 38.Rachbauer F, Sztankay A, Kreczy A, et al. High-dose-rate intraoperative brachytherapy (IOHDR) using flab technique in the treatment of soft tissue sarcomas. Strahlenther Onkol. 2003;179:480–485. doi: 10.1007/s00066-003-1063-7. [DOI] [PubMed] [Google Scholar]
- 39.Davis AM, O’Sullivan B, Bell RS, et al. Function and health status outcomes in a randomized trial comparing preoperative and postoperative radiotherapy in extremity soft tissue sarcoma. J Clin Oncol. 2002;20:4472–4477. doi: 10.1200/JCO.2002.03.084. [DOI] [PubMed] [Google Scholar]
- 40.Refaat Y, Gunnoe J, Hornicek FJ, Mankin HJ. Comparison of quality of life after amputation or limb salvage. Clin Orthop Relat Res. 2002;397:298–305. doi: 10.1097/00003086-200204000-00034. [DOI] [PubMed] [Google Scholar]
- 41.Davis AM, Sennik S, Griffin AM, et al. Predictors of functional outcomes following limb salvage surgery for lower-extremity soft tissue sarcoma. J Surg Oncol. 2000;73:206–211. doi: 10.1002/(sici)1096-9098(200004)73:4<206::aid-jso4>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 42.Davis AM, Devlin M, Griffin AM, et al. Functional outcome in amputation versus limb sparing of patients with lower-extremity sarcoma: a matched case-control study. Arch Phys Med Rehabil. 1999;80:615–618. doi: 10.1016/s0003-9993(99)90161-2. [DOI] [PubMed] [Google Scholar]
- 43.Colterjohn NR, Davis AM, O’Sullivan B, et al. Functional outcome in limb-salvage surgery for soft tissue tumours of the foot and ankle. Sarcoma. 1997;1:67–74. doi: 10.1080/13577149778326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pitcher ME, Thomas JM. Functional compartmental resection for soft tissue sarcomas. Eur J Surg Oncol. 1994;20:441–445. [PubMed] [Google Scholar]
- 45.Keus RB, Rutgers EJ, Ho GH, et al. Limb-sparing therapy of extremity soft tissue sarcomas: treatment outcome and long-term functional results. Eur J Cancer. 1994;30A:1459–1463. doi: 10.1016/0959-8049(94)00302-l. [DOI] [PubMed] [Google Scholar]
- 46.Moseley HS. An evaluation of two methods of limb salvage in extremity soft tissue sarcomas. Arch Surg. 1992;127:1169–73; discussion 1173–1164. [DOI] [PubMed]
- 47.Karasek K, Constine LS, Rosier R. Sarcoma therapy: functional outcome and relationship to treatment parameters. Int J Radiat Oncol Biol Phys. 1992;24:651–656. doi: 10.1016/0360-3016(92)90710-y. [DOI] [PubMed] [Google Scholar]
- 48.Robinson MH, Spruce L, Eeles R, et al. Limb function following conservation treatment of adult soft tissue sarcoma. Eur J Cancer. 1991;27:1567–1574. doi: 10.1016/0277-5379(91)90417-c. [DOI] [PubMed] [Google Scholar]
- 49.Stinson SF, DeLaney TF, Greenberg J, et al. Acute and long-term effects on limb function of combined modality limb-sparing therapy for extremity soft tissue sarcoma. Int J Radiat Oncol Biol Phys. 1991;21:1493–1499. doi: 10.1016/0360-3016(91)90324-w. [DOI] [PubMed] [Google Scholar]
- 50.Talbert ML, Zagars GK, Sherman NE, Romsdahl MM. Conservative surgery and radiation therapy for soft tissue sarcoma of the wrist, hand, ankle, and foot. Cancer. 1990;66:2482–2491. doi: 10.1002/1097-0142(19901215)66:12<2482::aid-cncr2820661207>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 51.Lampert MH, Gerber LH, Glatstein E, et al. Soft tissue sarcoma: functional outcome after wide local excision and radiation therapy. Arch Phys Med Rehabil. 1984;65:477–480. [PubMed] [Google Scholar]
- 52.O’Sullivan B, Davis AM, Turcotte R, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359:2235–2241. doi: 10.1016/S0140-6736(02)09292-9. [DOI] [PubMed] [Google Scholar]
- 53.Davis AM, Wright JG, Williams JI, et al. Development of a measure of physical function for patients with bone and soft tissue sarcoma. Qual Life Res. 1996;5:508–516. doi: 10.1007/BF00540024. [DOI] [PubMed] [Google Scholar]
- 54.Enneking W, ed. Modification of the system for functional evaluation in the surgical management of musculoskeletal tumours. In: Limb Salvage in Musculoskeletal Oncology. Churchill Livingstone, New York, NY, 1987.
- 55.Davis WF, Dunham W, Gebhardt MC, et al. A system for the functional evaluation of reconstructive procedures after surgical treatment of tumors of the musculoskeletal system. Clin Orthop Relat Res. 1993;286:241–246. [PubMed] [Google Scholar]
- 56.Ware JE. SF-36 Health Survey: Manual and Interpretation Guide. Boston: Health Institute, New England Medical Center; 1993. [Google Scholar]
- 57.Wollmerstedt N, Kirschner S, Faller H, Konig A. Reliability, validity, and responsiveness of the German Short Musculoskeletal Function Assessment Questionnaire in patients undergoing surgical or conservative inpatient treatment. Qual Life Res. 2006;15:1233–1241. doi: 10.1007/s11136-006-0066-0. [DOI] [PubMed] [Google Scholar]
- 58.Swiontkowski MF, Engelberg R, Martin DP, Agel J. Short musculoskeletal function assessment questionnaire: validity, reliability, and responsiveness. J Bone Joint Surg Am. 1999;81:1245–1260. doi: 10.2106/00004623-199909000-00006. [DOI] [PubMed] [Google Scholar]
- 59.Bennett PJ, Patterson C, Wearing S, Baglioni T. Development and validation of a questionnaire designed to measure foot-health status. J Am Podiatr Med Assoc. 1998;88:419–428. doi: 10.7547/87507315-88-9-419. [DOI] [PubMed] [Google Scholar]
- 60.Landorf KB, Keenan AM. An evaluation of two foot-specific, health-related quality-of-life measuring instruments. Foot Ankle Int. 2002;23:538–546. doi: 10.1177/107110070202300611. [DOI] [PubMed] [Google Scholar]
- 61.Schag CC, Heinrich RL, Ganz PA. Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol. 1984;2:187–193. doi: 10.1200/JCO.1984.2.3.187. [DOI] [PubMed] [Google Scholar]
- 62.Wood-Dauphinee SL, Opzoomer MA, Williams JI, et al. Assessment of global function: the Reintegration to Normal Living Index. Arch Phys Med Rehabil. 1988;69:583–590. [PubMed] [Google Scholar]
- 63.Wood-Dauphinee S, Williams JI. Reintegration to Normal Living as a proxy to quality of life. J Chronic Dis. 1987;40:491–502. doi: 10.1016/0021-9681(87)90005-1. [DOI] [PubMed] [Google Scholar]
- 64.Convery FR, Minteer MA, Amiel D, Connett KL. Polyarticular disability: a functional assessment. Arch Phys Med Rehabil. 1977;58:494–499. [PubMed] [Google Scholar]
- 65.Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range, and/or interquartile range. BMC Med Res Methodol. 2014;14:135. doi: 10.1186/1471-2288-14-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Luo D, Wan X, Liu J, Tong T. Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res. 2018;27:1785–1805. doi: 10.1177/0962280216669183. [DOI] [PubMed] [Google Scholar]
- 67.Janssen SJ, Paulino Pereira NR, Raskin KA, et al. A comparison of questionnaires for assessing physical function in patients with lower extremity bone metastases. J Surg Oncol. 2016;114:691–696. doi: 10.1002/jso.24400. [DOI] [PubMed] [Google Scholar]
- 68.Ogura K, Uehara K, Akiyama T, et al. Minimal clinically important differences in Toronto Extremity Salvage Score for patients with lower-extremity sarcoma. J Orthop Sci. 2019 doi: 10.1016/j.jos.2019.03.022. [DOI] [PubMed] [Google Scholar]
- 69.Chang AE, Steinberg SM, Culnane M, et al. Functional and psychosocial effects of multimodality limb-sparing therapy in patients with soft tissue sarcomas. J Clin Oncol. 1989;7:1217–1228. doi: 10.1200/JCO.1989.7.9.1217. [DOI] [PubMed] [Google Scholar]
- 70.Mercieca-Bebber R, Palmer MJ, Brundage M, et al. Design, implementation, and reporting strategies to reduce the instance and impact of missing patient-reported outcome (PRO) data: a systematic review. BMJ Open. 2016;6:e010938. doi: 10.1136/bmjopen-2015-010938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Health USDo, Human Services FDACfDE, Research, et al. Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes. 2006;4:79. [DOI] [PMC free article] [PubMed]
- 72.Frost MH, Reeve BB, Liepa AM, et al. What is sufficient evidence for the reliability and validity of patient-reported outcome measures? Value Health. 2007;10(Suppl 2):S94–S105. doi: 10.1111/j.1524-4733.2007.00272.x. [DOI] [PubMed] [Google Scholar]
- 73.Patrick DL, Burke LB, Powers JH, et al. Patient-reported outcomes to support medical product labeling claims: FDA perspective. Value Health. 2007;10(Suppl 2):S125–S137. doi: 10.1111/j.1524-4733.2007.00275.x. [DOI] [PubMed] [Google Scholar]
- 74.Calvert M, Kyte D, Mercieca-Bebber R, et al. Guidelines for inclusion of patient-reported outcomes in clinical trial protocols: the SPIRIT-PRO extension. JAMA. 2018;319:483–494. doi: 10.1001/jama.2017.21903. [DOI] [PubMed] [Google Scholar]
- 75.Bell RS, O’Sullivan B, Davis A, et al. Functional outcome in patients treated with surgery and irradiation for soft tissue tumours. J Surg Oncol. 1991;48:224–231. doi: 10.1002/jso.2930480404. [DOI] [PubMed] [Google Scholar]
- 76.Prinsen CAC, Mokkink LB, Bouter LM, et al. COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res. 2018;27:1147–1157. doi: 10.1007/s11136-018-1798-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J Clin Epidemiol. 2010;63:737–745. doi: 10.1016/j.jclinepi.2010.02.006. [DOI] [PubMed] [Google Scholar]
- 78.Han G, Bi WZ, Xu M, et al. Amputation versus limb-salvage surgery in patients with osteosarcoma: a meta-analysis. World J Surg. 2016;40:2016–2027. doi: 10.1007/s00268-016-3500-7. [DOI] [PubMed] [Google Scholar]
- 79.Mei J, Zhu XZ, Wang ZY, Cai XS. Functional outcomes and quality of life in patients with osteosarcoma treated with amputation versus limb-salvage surgery: a systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1507–1516. doi: 10.1007/s00402-014-2086-5. [DOI] [PubMed] [Google Scholar]
- 80.Malek F, Somerson JS, Mitchel S, Williams RP. Does limb-salvage surgery offer patients better quality of life and functional capacity than amputation? Clin Orthop Relat Res. 2012;470:2000–2006. doi: 10.1007/s11999-012-2271-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ladlow P, Phillip R, Coppack R, et al. Influence of immediate and delayed lower-limb amputation compared with lower-limb salvage on functional and mental health outcomes post-rehabilitation in the UK military. J Bone Joint Surg Am. 2016;98:1996–2005. doi: 10.2106/JBJS.15.01210. [DOI] [PubMed] [Google Scholar]