Abstract
Solena amplexicaulis (Lam.) Gandhi (Family Cucurbitaceae) is one of the important plant species used by the Irula tribes of Walayar valley of southern Western Ghats, India for the management of diabetes. To confirm the antidiabetic property of S. amplexicaulis, the present study was addressed using crude methanolic leaf extract of S. amplexicaulis (MeOHSa) and its isolated compound, Forskolin against streptozotocin (STZ) induced diabetic rats. The oral glucose tolerance test (OGTT), blood glucose, lipid profile, serum liver markers, antioxidants, hemoglobin and glycogen were evaluated using standard procedure. The oral administration of Forskolin and MeOHSa (600 mg/kg b.w.) for 30 days resulted in significant restoration of all these parameters supported by histopathological observations too. The results clearly suggest that the Forskolin (diterpene) possess potent antidiabetic and antihyperlipidemic activities, which may be considered as a lead molecule for therapeutic purposes, and the source of Forskolin i.e. S. amplexicaulis can be further exploited for pharmaceutical industries.
Keywords: Solena amplexicaulis, Cucurbitaceae, Forskolin, Antidiabetic, Antihyperlipidemic, Antioxidant
1. Introduction
Diabetes mellitus (-a group of the metabolic disorders), is characterized by hyperglycemia, and is resulting from the defects in the secretion and action of the insulin [-a protein (hormone)] or both produced by the pancreas (Maritim et al., 2003). The global burden of the disease diabetes in the year 2017 only was approximately 425 million, which have been estimated to increase up to 629 million by 2045 (Ogurtsova et al., 2017). There have been several insulin as well as oral hypoglycemic agents which are under trade for the management of diabetes (Newman and Cragg, 2016); however, its use lead to various side effects (Kameswararao et al., 2003). Therefore, identification of natural products having antidiabetic activity with least side effects become immense importance nowadays (Newman and Cragg, 2016).
Traditional knowledge of ethnic communities is the most reliable source to know the healing properties of plant species for various ailments (Venkatachalapathi et al., 2015, Venkatachalapathi et al., 2018). Recently bioinformatics tools like ‘Prediction of Activity Spectra for Substances’ (PASS) are much helpful to find the possible medicinal uses of certain bioactive compounds of ethnobotanical significance (Filimonov et al., 2014). The information collected from the ethnic communities and the tools of bioinformatics are used to identify and select plants of medicinal importance for further scientific validation. The ‘Irula’ (-one of the oldest tribal communities of India, migrated from Africa) belongs to the Negrito race, has wide knowledge in using plants for their health care (Von, 1982, Venkatachalapathi et al., 2015, Venkatachalapathi et al., 2018). Solena amplexicaulis (Lam.) Gandhi (family Cucurbitaceae) is the rare sighted climber, inhabiting in the dry deciduous forests of southern India, is being used by Irula tribes of Walayar valley of the Western Ghats of Tamil Nadu, India for the management of diabetes since long back (Paulsamy and Karthika, 2014).
Diabetes is affiliated with the increased ROS (free radicals) formation of tilting the balance of oxidant/antioxidant defense system of the human body (Nazirogilu and Butterworth, 2005); as a result, there is alteration in enzymatic systems, impaired Glutathione metabolism, lipid peroxidation and lower level of Vitamin C. In the diabetes, catalane, lipids, proteins, Glutathione, DNA damage, and superoxide dismutase are various biomarkers of oxidative stress (Asmat et al., 2016). We have previously been reported that S. amplexicaulis is rich in polyphenols with strong antioxidant activity (Karthika et al., 2012), and the bioinformatics software ‘PASS’ also predicted that the Forskolin (-an isolated compound from S. amplexicaulis) have antidiabetic property.; this has prompted us for the in vivo evaluation of antidiabetic and antihyperlipidemic properties of methanolic extract of S. amplexicaulis and Forskolin (-the major components of methanolic extract of S. amplexicaulis) using streptozotocin (STZ) induced diabetic rats.
2. Materials and methods
2.1. Preparation of test samples
The leaves of S. amplexicaulis were collected from Madukkarai (Coimbatore, India), and the taxonomic authentication was confirmed by comparing the reference specimen (Vide No: CPS 313) housed at Botanical Survey of India, Coimbatore. The powdered leaves (1000 g) were exhaustively extracted with methanol after de-waxing with petroleum ether using soxhlet apparatus, concentrated up to dryness using rotary evaporator, and then stored at −20 °C temperature until used in the experiments. The methanolic crude extract (MeOHSa) yield was 18.6% w/w (dry weight basis). The spectral data and also our report published earlier confirmed that the chemical structure of the isolated compound as Forskolin [((3R,4aR,5S,6S,6aS,10S,10aR,10bS)-6,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-3-vinyldodecahydro-1H-benzo[f]chromen-5-yl acetate)] (Karthika et al., 2016). The isolation of Forskolin was made using wet packing method of silica gel (60–120 mesh) using column chromatography (Reid and Sarker, 2012).
2.2. Experimental animals
The animal studies conducted on male wistar albino rats (150–250 g) were approved (No: 659/02/a/CPCSEA) by Institutional Animal Ethical Committee (IAEC). In order to evaluate the behavioral and toxicological effects, the acute toxicity was evaluated following Organization for Economic Cooperation and Development (OECD) guideline 423 (2001). The rats were divided into eleven groups (n = 6) each (the first group served as control, the groups 2–8 were orally treated with MeOHSa at the doses of 50, 150, 300, 500, 1000, 2000 and 3000 mg/kg body weight (b.w.) respectively, and the groups 9–11 received oral doses of 10, 50 and 100 mg/kg b.w. of Forskolin respectively). The observations were made for mortality and clinical signs up to 14 days.
2.3. Oral glucose tolerance test (OGTT)
A total number of 24 rats (assigned randomly into four equal groups) were fasted for 16 h, and then were orally treated [Group 1: 1 ml of distilled water (control), Group 2: 600 mg/kg b.w. of MeOHSa, Group 3: 10 mg/kg b.w. Forskolin, Group 4: 600 μg/kg b.w. of standard Glibenclamide (Sigma- Aldrich Chemicals, India)] followed by 5 ml/kg of 50% (w/v) glucose solution after 1 h, and then the blood glucose levels were estimated (using Accu-chek, Roche Diagnostics, USA) after 60, 120 and 240 min.
2.4. Antidiabetic effect of MeOHSa and Forskolin in type 1 diabetes induced rats
2.4.1. Induction of IDDM
The insulin dependent diabetes mellitus was induced in overnight fasted wistar albino rats by the single dose of intraperitoneal injection of Streptozotocin (Sigma- Aldrich Chemicals, India) (50 mg/kg b.w.) (0.01 M, pH 4.5). After Streptozotocin administration, in order to stave off the hypoglycaemic shock, the rats were given 5% (w/v) glucose orally. The massive glycosuria and hyperglycemia were noted in the rats within two days after administration of Streptozotocin. The rats with blood glucose levels above 250 mg/dl were considered diabetic, and were selected for the further study.
2.4.2. Experimental regime
The diabetic rats were divided into five groups (n = 6), and were fed orally by gastric intubation once daily for 30 days in the following manner: Group 1 (Normoglycemic, control group), Group 2 (Streptozotocin-induced diabetic rats), Group 3 (MeOHSa, 600 mg/kg b.w.), Group 4 (Forskolin, 10 mg/kg b.w.), Group 5 (Glibenclamide, 600 μg/kg b.w.). The third day of induction was designated as day 1 for extract administration in diabetic rats. The fasting blood glucose levels were monitored at the interval of 10 days of administration using glucometer elite (glucose oxidase method). All the animals were sacrificed at 30th days, the blood samples were collected. The fresh serum was collected, and stored at −20 °C until further analysis of blood. The pancreas, liver and kidney were carefully excised, and fixed in 10% Formolsaline. The body weight of animals was recorded.
2.4.3. Biochemical analysis
The Serum Glutamate Oxaloacetate Transaminase (SGOT), Serum Glutamate Pyruvate Transaminase (SGPT) and the levels of High and Low Density Lipoproteins (HDL and LDL), Total Cholesterol (TC) and Triglycerides (TG) in serum were assessed in an autoanalyzer (MISPA EXCEL, India). The fresh blood samples were drawn in heparinized tube, and their total haemoglobin content was estimated (Rastogi, 2005). The hepatic glycogen level estimated following method of Sadasivam and Manickam (1996). The activities of Lipid Peroxidation (Niehius and Samuelsson, 1968), Catalase (CAT) (Sinha, 1972), Superoxide Dismutase (SOD) (Marklund and Marklund, 1974), Glutathione-S-Transferase (GST) (Habiget al., 1974), Glutathione Peroxidase (GPx) (Rotruck et al., 1973), Reduced Glutathione (Moron et al., 1979) and Vitamin C (Omayeet al., 1979) were determined in hepatic and renal tissues.
2.4.4. Histopathological studies
The whole pancreas from each animal were fixed in 10% Formolsaline, and were cut into ultra-thin sections, stained and mounted, and the histological architecture at 10× and 40× magnification in Axiostar plus microscope (Carl Zeiss, Germany).
2.5. Statistical analysis
The results were presented as mean ± S.E.M (n = 6), one-way analysis of variance (ANOVA) were performed followed by Tukey’s multiple comparison test, with the help of Graph Pad Prism version 4.0 (Graph Pad software, San Diego, California, USA). P < 0.001 was considered as significant.
3. Results and discussion
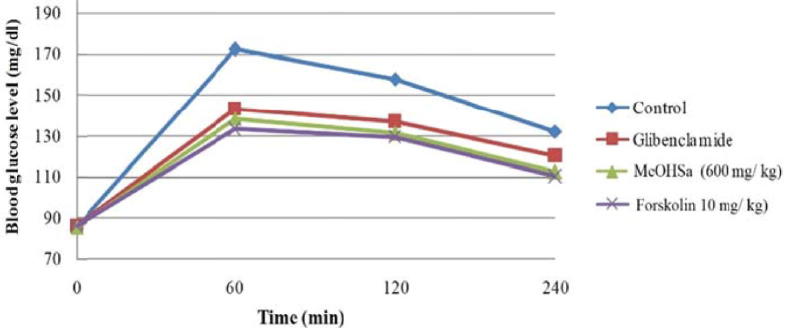
An oral load of 2.5 gm glucose resulted in nearly two fold raise of blood glucose level in 60 min which progressively decreased towards normalcy over the period of time; however, pretreatment with MeOHSa) and Forskolin or Glibenclamide significantly lead to the blood glucose levels towards normalcy. Interestingly, the efficacy of Forskolin was lower as compared to Glibenclamide (Fig. 1). The hypoglycemic effect of Forskolin might be due to the enhanced glucose-mediated stimulus to release insulin; this effect produced by the elevation of CAMP which activates main signaling pathways in beta cells viz., Protein Kinase (PKA) pathway and guanine nucleotide pathway regulated by CAMP (Rios-Silva et al., 2014). Further, the administration of the MeOHSa and Forskolin significantly increases the body weight (Table 1) and decreases the blood glucose levels (Table 2). The significant climb in the level of the total haemoglobin content, the reversal of the transaminase enzyme (SGOT and SGPT) activities towards their respective normal levels, the significant restoration of the lipid profile to the normalcy, increase in the glycogen content of the liver after the administration of Forskolin (Table 3) further strengthen its antidiabetogenic effect which might be due to ability of antidiabetic compound to reduce hyperglycemia (Shirwaikar et al., 2005) through insulin release stimulatory effect (Vijayakumar et al., 2006) and the sensitization of insulin receptors and reactivation of glycogen synthase system involved in the glycogen synthesis (Huang et al., 2000, Rotimi et al., 2014).
Fig. 1.
Effect of MeOHSa and Forskolin on OGT. Values are mean ± SEM (n = 6). aP < 0.001, significantly different from the respective control group.
Table 1.
Effect of MeOHSa and Forskolin on body weight in diabetic rats.
| Treatment group | Body weight (g) |
|
|---|---|---|
| Initial (*Day 1) | Final (Day 30) | |
| Control | 233.3 ± 10.5 | 271.7 ± 11.8 |
| Diabetic control | 183.3 ± 9.8 | 115.0 ± 11.2a (−37.3%) |
| Diabetic + MeOHSa (600 mg/kg b.w.) | 176.7 ± 12.0 | 195.0 ± 7.2a,b (+10.4%) |
| Diabetic + Forskolin (10 mg/kg b.w.) | 173.3 ± 13.3 | 220.0 ± 9.1b (+26.9%) |
| Diabetic + Glibenclamide (600 µg/kg b.w.) | 176.7 ± 10.5 | 228.3 ± 12.1b (+29.2%) |
Values are mean ± SEM (n = 6). Percent increase or decrease in body weights are given in the parenthesis.
Day 3 of induction of diabetes.
P < 0.001, significantly different from the control group of day 30.
P < 0.001, significantly different from the diabetic control group of day 30.
Table 2.
Effect of MeOHSa and Forskolin on blood glucose level in STZ induced diabetic rats.
| Treatment group | Blood glucose level (mg/dL) |
||||
|---|---|---|---|---|---|
| 0 day | 1st day* | 10th day | 20th day | 30th day | |
| Control | 83.3 ± 1.4 | 82.0 ± 2.1 | 85.0 ± 2.1 | 86.5 ± 1.5 | 86.3 ± 2.3 |
| Diabetic control | 78.5 ± 4.2 | 420.5 ± 1.8 | 432.8 ± 3.0a (+2.9%) | 450.0 ± 5.3a (+7.0%) | 467.0 ± 1.0a (+11.1%) |
| Diabetic + MeOHSa (600 mg/kg b.w.) | 86.0 ± 3.0 | 415.0 ± 0.7 | 356.0 ± 2.4a (−14.2%) | 320.0 ± 1.5a,e (−23.0%) | 235.3 ± 1.4b,d (−43.3%) |
| Diabetic + Forskolin (10 mg/kg b.w.) | 83.0 ± 2.0 | 412.8 ± 2.1 | 334.8 ± 1.8a,f (−18.9%) | 262.0 ± 1.5b,d (−36.5%) | 156.5 ± 1.7d (−62.1%) |
| Diabetic + Glibenclamide (600 µg/kg) | 79.0 ± 2.4 | 418.5 ± 0.6 | 307.3 ± 3.8a,e (−26.6%) | 219.8 ± 1.0b,d (−47.5%) | 118.3 ± 2.9d (−71.7%) |
Values are mean ± SEM (n = 6). The values given in the parenthesis indicates percentage of increase or decrease in glucose concentration over 1st day.
cP < 0.05, significantly different from the control group of the respective day.
Day 3 of induction of diabetes.
P < 0.001, significantly different from the control group of the respective day.
P < 0.01, significantly different from the control group of the respective day.
P < 0.001, significantly different from the diabetic control group of the respective day.
P < 0.01, significantly different from the diabetic control group of the respective day.
P < 0.05, significantly different from the diabetic control group of the respective day.
Table 3.
Effect of MeOHSa and Forskolin on various biochemical markers in STZ induced diabetic rats.
| Parameter* | Treatment group |
||||
|---|---|---|---|---|---|
| Control | Diabetic control | Diabetic + MeOHSa (600 mg/kg b.w.) | Diabetic + Forskolin (10 mg/kg b.w.) | Diabetic + Glibenclamide (600 µg/kg b.w.) | |
| Haemoglobin (%) | 14.5 ± 0.4 | 6.9 ± 0.2a | 12.5 ± 0.5b,d | 13.3 ± 0.4d | 13.9 ± 0.3d |
| SGOT (IU/L) | 41.6 ± 2.8 | 112.3 ± 3.6a | 49.4 ± 1.5d | 44.2 ± 1.7d | 42.6 ± 4.7d |
| SGPT (IU/L) | 28.2 ± 6.4 | 102.6 ± 1.4a | 57.1 ± 5.2bd | 30.6 ± 2.5d | 32.9 ± 7.5d |
| HDL (mg/dL) | 52.8 ± 8.7 | 11.4 ± 2.0a | 43.5 ± 7.6e | 51.8 ± 5.0d | 49.1 ± 0.1d |
| LDL (mg/dL) | 82.8 ± 5.6 | 231.9 ± 2.5a | 95.2 ± 4.3d | 84.5 ± 1.3d | 88.2 ± 8.7d |
| TC (mg/dL) | 156.3 ± 4.6 | 280.4 ± 1.4a | 161.6 ± 5.2d | 157.8 ± 2.7d | 159.3 ± 2.5d |
| TG (mg/dL) | 103.3 ± 1.1 | 185.7 ± 1.6a | 114.7 ± 8.1d | 107.4 ± 8.1d | 109.8 ± 5.3d |
| Glycogen (mg/g liver tissue) | 19.0 ± 1.9 | 5.2 ± 1.4a | 14.4 ± 0.5f | 18.8 ± 3.7d | 18.6 ± 0.7d |
Values are mean ± SEM (n = 6).
cP < 0.05, significantly different from the control group.
SGOT - serum glutamate oxaloacetate transaminase; SGPT - serum glutamate pyruvate transaminase; HDL - high-density lipoproteins; LDL - low density lipoproteins; TC - total cholesterol; TG - triglycerides.
P < 0.001, significantly different from the control group.
P < 0.01, significantly different from the control group.
P < 0.001, significantly different from the diabetic control group.
P < 0.01, significantly different from the diabetic control group.
P < 0.05, significantly different from the diabetic control group.
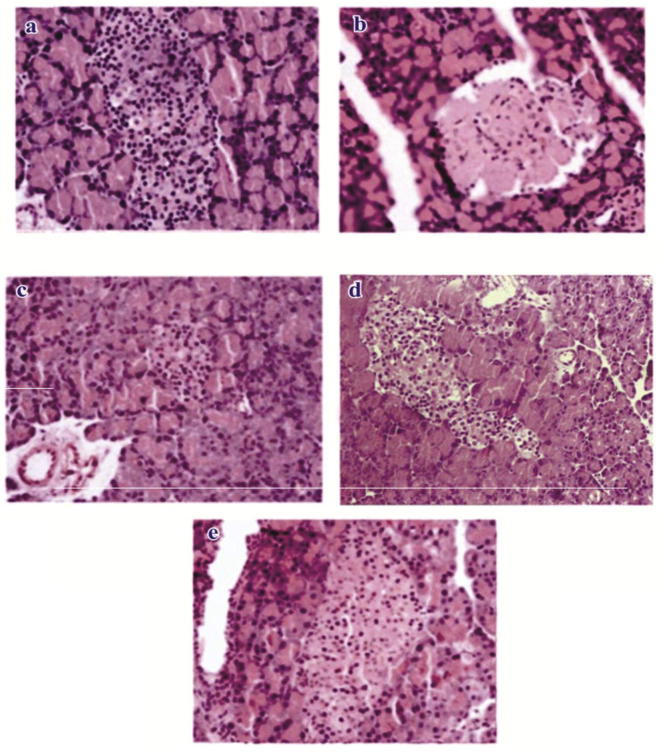
Diabetes is associated with the increased in the formation of free radicals, tilting the balance of oxidant/antioxidant defense system (Nazirogilu and Butterworth, 2005). The administration of the MeOHSa and Forskolin significantly (P < 0.05) improved the activities of enzymic and non-enzymic antioxidants in the diabetic rats (Table 4). In addition, the administration of Forskolin markedly ameliorated islet damage comparable with Glibenclamide (Fig. 2).
Table 4.
Effect of MeOHSa and forskolin on antioxidant markers in STZ induced diabetic rats.
| Parameter* | Control | Diabetic control | Diabetic + MeOHSa(600 mg/kg b.w.) | Diabetic + forskolin (10 mg/kg b.w.) | Diabetic + glibenclamide (600 µg/kg b.w.) |
|---|---|---|---|---|---|
| LPO (µ moles of malondialdehyde (MDA) formed/mg protein) | |||||
| Liver | 8.4 ± 0.9 | 17.8 ± 0.7a | 10.3 ± 0.3d | 9.0 ± 0.2d | 8.9 ± 0.8d |
| Kidney | 7.1 ± 0.4 | 16.2 ± 1.4a | 8.4 ± 1.2e | 7.9 ± 0.5d | 7.9 ± 2.1d |
| SOD (units/mg protein) | |||||
| Liver | 38.4 ± 0.9 | 17.6 ± 1.3a | 28.5 ± 3.2bd | 37.7 ± 0.7d | 35.8 ± 0.6d |
| Kidney | 38.8 ± 1.9 | 15.3 ± 3.0a | 29.1 ± 1.9 cd | 35.5 ± 2.0d | 32.5 ± 1.1d |
| CAT (µ moles of hydrogen peroxide consumed/min/mg protein) | |||||
| Liver | 62.8 ± 4.1 | 21.2 ± 0.1a | 51.2 ± 0.2b,d | 59.5 ± 0.3d | 60.4 ± 0.4d |
| Kidney | 53.0 ± 3.6 | 18.5 ± 0.1a | 45.0 ± 0.2c,d | 52.0 ± 2.2d | 51.4 ± 0.4d |
| GPx (µ moles of reduced glutathione utilized/min/mg protein) | |||||
| Liver | 3.1 ± 0.2 | 1.1 ± 0.1a | 1.9 ± 0.1b,f | 3.0 ± 0.3d | 2.8 ± 0.2d |
| Kidney | 2.2 ± 0.1 | 0.9 ± 0.1a | 1.7 ± 0.1c,d | 2.1 ± 0.1d | 2.0 ± 0.1d |
| GST (units/mg protein) | |||||
| Liver | 6.3 ± 0.2 | 2.1 ± 0.2a | 5.8 ± 0.1c,d | 6.2 ± 0.1d | 6.0 ± 0.2d |
| Kidney | 5.2 ± 0.2 | 1.8 ± 0.1a | 3.9 ± 0.1b,d | 4.9 ± 0.1d | 4.8 ± 0.4d |
| Vitamin C (µ moles/mg protein) | |||||
| Liver | 13.4 ± 0.6 | 3.9 ± 0.6a | 9.4 ± 0.3c,d | 10.3 ± 0.3d | 11.5 ± 1.5d |
| Kidney | 7.3 ± 0.6 | 4.5 ± 0.2a | 6.5 ± 0.2e | 7.0 ± 0.1d | 6.9 ± 0.2d |
| GSH (µ moles/mg protein) | |||||
| Liver | 40.8 ± 1.0 | 20.6 ± 2.5a | 34.7 ± 1.0d | 38.7 ± 0.5d | 36.8 ± 1.6d |
| Kidney | 33.7 ± 3.7 | 12.9 ± 2.7a | 24.0 ± 1.0c,e | 31.9 ± 0.8d | 28.5 ± 0.7d |
Values are mean ± SEM (n = 6).
LPO - lipid peroxides; SOD - superoxide dismutase; CAT - catalase; GPx - glutathione peroxidase; GST - glutathione-s-transferase; GSH - reduced glutathione.
P < 0.001, significantly different from the control group.
P < 0.01, significantly different from the control group.
P < 0.05, significantly different from the control group.
P < 0.001, significantly different from the diabetic control group.
P < 0.01, significantly different from the diabetic control group.
P < 0.05, significantly different from the diabetic control group.
Fig. 2.
Photomicrograph showing (a) pancreatic islet of normal untreated control group, (b) STZ-induced diabetic group, (c) diabetic + MeOHSa (600 mg/kg b.w.) treated group, (d) diabetic + Forskolin (10 mg/kg b.w.) treated group and (e) diabetic + Glibenclamide (600 µg/kg b.w.) treated group.
There are several cucurbits are well known for their antidiabetic properties viz. Bryonia alba L. (Karagenzyan et al., 1998), Citrullus colocynthis Schrad. (Nmila et al., 2000, Abdel-Hassan et al., 2000), Coccina indica Wight & Am. (Hossain et al., 1992, Kumar et al., 1993), Cucurbita ficifolia Bouché (Roman-Ramos et al., 1992a, Roman-Ramos et al., 1992b), Momordica charantia L. (Day et al., 1990, Higashino et al., 1992, Cakici et al., 1994, Tennekoom et al., 1994, Sarkar et al., 1996, Raza et al., 1996, Sitasawad et al., 2000, Jayasooriya et al., 2000), Momordica cymbalaria (Rao et al., 1999). S. amplexicaulis is known to be rich in polyphenols with strong antioxidant activity (Karthika et al., 2012). Moreover, Forskolin isolated from Coleus forskohlii (family Lamiaceae) have previously been reported as the active antidiabetic agent (Rios-Silva et al., 2014). As such, it is assumed that apart from its strong antioxidant potential, Forskolin might stimulate the insulin secretion from β-cells by the mechanism similar to oral hypoglycemic drugs.
Acknowledgments
Acknowledgements
The authors gratefully acknowledge the financial support from University Grants Commission, Government of India, New Delhi (Grant No. F. 41-415/2012(SR). The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding the work through the research group project (RG-1439-84).
Conflicts of interest
We declare that we have no conflicts of interest.
Footnotes
Peer review under responsibility of King Saud University.
Contributor Information
Arjunan Venkatachalapathi, Email: avenkatachalapathi161@gmail.com.
Mohammad Ajmal Ali, Email: ajmalpdrc@gmail.com.
References
- Abdel-Hassan I.A., Abdel-Barry J.A., Tariq M.S. The hypoglycaemic and antihyperglycaemic effect of Citrullus colocynthis fruit aqueous extract in normal and alloxan diabetic rabbits. J. Ethnopharmacol. 2000;71:325–330. doi: 10.1016/s0378-8741(99)00215-9. [DOI] [PubMed] [Google Scholar]
- Asmat U., Abad K., Ismail K. Diabetes mellitus and oxidative stress-a concise review. Saudi Pharm. J. 2016;24(5):547–553. doi: 10.1016/j.jsps.2015.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cakici I., Hurmoğlu C., Tunçtan B., Abacioğlu N., Kanzik I., Sener B. Hypoglycaemic effect of Momordica charantia extracts in normoglycaemic or cyproheptadine-induced hyperglycaemic mice. J. Ethnopharmacol. 1994;44:117–121. doi: 10.1016/0378-8741(94)90077-9. [DOI] [PubMed] [Google Scholar]
- Day C., Cartwright T., Provost J., Bailey C.J. Hypoglycaemic effect of Momordica charantia extracts. Planta Med. 1990;56:426–429. doi: 10.1055/s-2006-961003. [DOI] [PubMed] [Google Scholar]
- Filimonov D.A., Lagunin A.A., Gloriozova T.A., Rudik A.V., Druzhilovskii D.S., Pogodin P.V., Poroikov V.V. Prediction of the biological activity spectra of organic compounds using the PASS online web resource. Chem. Heterocycl. Compd. 2014;50:444–457. [Google Scholar]
- Habig W.H., Pabst M.J., Jakpoby W.B. Glutathione transferase: a first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974;249:7130–7139. [PubMed] [Google Scholar]
- Higashino H., Suzuki A., Tanaka P.K. Hypoglycaemic effects of Siamese Momordica Charantia and phyllantus urinaria extracts in streptozotocin-induced diabetic rats. Nippon Yakurigaku Zasshi. 1992;100:515–521. doi: 10.1254/fpj.100.415. [DOI] [PubMed] [Google Scholar]
- Hossain M.Z., Shibib B.A., Rahman R. Hypoglycaemic effects of Coccina indica: inhibition of key gluconeogenic enzyme, glucose-6-phosphatase. Indian J. Exp. Biol. 1992;30:418–420. [PubMed] [Google Scholar]
- Huang X., Vaag A., Hanson M., Weng J., Goop L. Impaired insulin stimulated expression of the glycogen synthase gene in skeletal muscle of type 2 diabetic patients is acquired rather than inherited. J. Clin. Endocrinol. Metab. 2000;85:1584–1590. doi: 10.1210/jcem.85.4.6535. [DOI] [PubMed] [Google Scholar]
- Jayasooriya A.P., Sakono M., Yukizaki C., Kawano M., Yamamoto K., Fukuda N. Effects of Momordica charantia powder on serum glucose levels and various lipid parameters in rats fed with cholesterol-free and cholesterol-enriched diets. J. Ethnopharmacol. 2000;72:331–336. doi: 10.1016/s0378-8741(00)00259-2. [DOI] [PubMed] [Google Scholar]
- Karagenzyan K.G., Vartanyan G.S., Agadjanov M.I., Panossian A.G., Hoult J.R. Restoration of the disordered glucose fatty acid cycle in alloxan-diabetic rats by trihydroxyoctadecadienoic acids from Bryonia alba, a native Armenian medicinal plant. Planta Med. 1998;64:417–422. doi: 10.1055/s-2006-957472. [DOI] [PubMed] [Google Scholar]
- Karthika K., Jamuna S., Abinaya G., Venkatachalapathi A., Thenmozhi K., Paulsamy S. Evaluation of antiinflammatory and antioxidant properties of crude extract and Forskolin from Solena amplexicaulis Leaf. Indian J. Pharm. Sci. 2016;78:377–387. [Google Scholar]
- Karthika K., Paulsamy S., Jamuna S. Evaluation of in vitro antioxidant potential of methanolic leaf and stem extracts of Solena amplexicaulis (Lam.) Gandhi. J. Chem. Pharm. Res. 2012;4:3254–3258. [Google Scholar]
- Kameswararao B., Kesavulu M.M., Apparao C. Evaluation of antidiabetic effect of Momordica cymbalaria fruit in alloxan-induced diabetic rats. Fitoterapia. 2003;74:7–13. doi: 10.1016/s0367-326x(02)00297-6. [DOI] [PubMed] [Google Scholar]
- Kumar G.P., Sudheesh S., Vijayalakshmi N.R. Hypoglycaemic effect of Coccina indica: mechanism of action. Planta Med. 1993;59:330–332. doi: 10.1055/s-2006-959693. [DOI] [PubMed] [Google Scholar]
- Maritim A.C., Sanders R.A., Watkins J.B. Diabetes, oxidative stress, and antioxidants: a review. J. Biochem. Mol. Toxicol. 2003;17(1):24–38. doi: 10.1002/jbt.10058. [DOI] [PubMed] [Google Scholar]
- Marklund S., Marklund G. Involvement of the superoxide anion radical in the auto oxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974;47:469–474. doi: 10.1111/j.1432-1033.1974.tb03714.x. [DOI] [PubMed] [Google Scholar]
- Moron M.S., Depierre J.W., Mannervik B. Levels of glutathione, glutathione reductase and glutathione-S-transferase activities in rat lung and liver. Biochim. Biophysica. Acta. 1979;582:67–78. doi: 10.1016/0304-4165(79)90289-7. [DOI] [PubMed] [Google Scholar]
- Nazirogilu M., Butterworth P. Protective effects of moderate exercise with dietary vitamin C and E on blood antioxidative defense mechanism in rats with streptozotocin-induced diabetes. Can. J. Appl. Physiol. 2005;30:172–185. doi: 10.1139/h05-113. [DOI] [PubMed] [Google Scholar]
- Newman D.J., Cragg G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016;79(3):629–661. doi: 10.1021/acs.jnatprod.5b01055. [DOI] [PubMed] [Google Scholar]
- Niehius W.G., Samuelsson D. Formation of malondialdehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur. J. Biochem. 1968;6:126–130. doi: 10.1111/j.1432-1033.1968.tb00428.x. [DOI] [PubMed] [Google Scholar]
- Nmila R., Gross R., Rchid H., Roye M., Manteghetti M., Petit P., Tijane M., Ribes G., Sauvaire Y. Insulinotropic effect of Citrullus colocynthis fruit extracts. Planta Med. 2000;66:418–423. doi: 10.1055/s-2000-8586. [DOI] [PubMed] [Google Scholar]
- Ogurtsova K., da Rocha Fernandes J.D., Huang Y., Linnenkamp U., Guariguata L., Cho N.H., Cavan D., Shaw J.E., Makaroff L.E. IDF diabetes atlas: global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017:40–50. doi: 10.1016/j.diabres.2017.03.024. [DOI] [PubMed] [Google Scholar]
- Omaye S.T., Turbull J.D., Sauberlich H.E. Selected methods for the determination of ascorbic acid in animal cells, tissues and fluids. In: Mc Cormick D.B., Wright L.D., editors. Methods in Enzymology. Academic Press; New York: 1979. [DOI] [PubMed] [Google Scholar]
- Paulsamy S., Karthika K. Distribution and density of the climber, Solena amplexicaulis (Lam.) Gandhi in certain dry deciduous forests of southern Western Ghats. India. Int. J. Ecol. Dev. 2014;28:63–78. [Google Scholar]
- Rao B.K., Kesavulu M.M., Giri R., Appa Rao C. Antidiabetic and hypolipidemic effects of Momordica cymbalaria Hook. Fruit powder in alloxan-diabetic rats. J. Ethnopharmacol. 1999;67:103–119. doi: 10.1016/s0378-8741(99)00004-5. [DOI] [PubMed] [Google Scholar]
- Rastogi S.C. 2nd ed. New Age International Pvt. Ltd; India: 2005. Experimental Physiology. [Google Scholar]
- Raza H., Ahmed I., Lakhani M.S., Sharma A.K., Pallot D., Montague W. Effect of bitter melon (Momordica charantia) fruit juice on the hepatic cytochrome P450-dependent monooxygenase and glutathione S-transferase in streptozotocin-induced diabetic rats. Biochem. Pharmacol. 1996;52:1639–1642. doi: 10.1016/s0006-2952(96)00526-6. [DOI] [PubMed] [Google Scholar]
- Reid R.G., Sarker S.D. Isolation of natural products by low-pressure column chromatography. Methods. Mol. Biol. 2012;864:155–187. doi: 10.1007/978-1-61779-624-1_7. [DOI] [PubMed] [Google Scholar]
- Rios-Silva M., Trujillo X., Trujillo-Hernandez B., Sanchez-Pastor E., Urzua Z., Mancilla E., Huerta M. Effect of chronic administration of forskolin on glycemia and oxidative stress in rats with and without experimental diabetes. Int. J. Med. Sci. 2014;11:448–452. doi: 10.7150/ijms.8034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman-Ramos R., Alarcon-Aguilar F., Lara-Lemus A., Flores-Saenz J.L. Hypoglycaemic effect of plants used in Mexico as antidiabetics. Arch. Med. Res. 1992;23:59–64. [PubMed] [Google Scholar]
- Roman-Ramos R., Lara-Lemus A., Alarcon-Aguilar F. Hypoglycaemic activity of some antidiabetic plants. Arch. Med. Res. 1992;23:105–109. [PubMed] [Google Scholar]
- Rotimi O.A., Aderounmu I.G., Oluwafemi O.O. Lipid profile, antidiabetic and antioxidant activity of Acacia ataxacantha bark extract in Streptozotocin-induced diabetic rats. In: Oguntibeju Oluwafemi., editor. Antioxidant-Antidiabetic Agents and Human Health. InTech Publisher; Croatia: 2014. [Google Scholar]
- Rotruck J.T., Pope A.L., Ganther H.E., Swanson A.B., Hafeman D.G., Hoekstra W.G. Selenium: biochemical roles as a component of glutathione peroxidase. Science. 1973;179:588–590. doi: 10.1126/science.179.4073.588. [DOI] [PubMed] [Google Scholar]
- Sadasivam S., Manickam A. Carbohydrates. In: Sadasivam S., Manickam A., editors. Methods in Biochemistry. New Age International Pvt. Ltd.; New Delhi: 1996. [Google Scholar]
- Sarkar S., Pranava M., Marita R. Demonstration of the hypoglycaemic action of Momordica Charantia in a validated model of diabetes. Pharmacol. Res. 1996;33:1–4. doi: 10.1006/phrs.1996.0001. [DOI] [PubMed] [Google Scholar]
- Shirwaikar A., Rajendran K., Punitha I.S.R. Antidiabetic activity of alcoholic stem extract of Coscinium fenestratum in streptozotocin-nicotinamide induced type 2 diabetic rats. J. Ethnopharmacol. 2005;97:369–374. doi: 10.1016/j.jep.2004.11.034. [DOI] [PubMed] [Google Scholar]
- Sinha A.K. Colorimetric assay of catalase. Anal. Biochem. 1972;47:389–394. doi: 10.1016/0003-2697(72)90132-7. [DOI] [PubMed] [Google Scholar]
- Sitasawad S.L., Shewade Y., Bhonde R. Role of bittergourd fruit juice in STZ-induced diabetic state in vivo and in vitro. J. Ethnopharmacol. 2000;73:71–79. doi: 10.1016/s0378-8741(00)00282-8. [DOI] [PubMed] [Google Scholar]
- Tennekoom K.H., Jeevathayaparan S., Angunawala P., Karunanayake E.H., Jayasinghe K.S. Effect of Momordica charantia on key hepatic enzymes. J. Ethnopharmacol. 1994;44:93–97. doi: 10.1016/0378-8741(94)90074-4. [DOI] [PubMed] [Google Scholar]
- Venkatachalapathi A., Sangeeth T., Paulsamy S. Ethnobotanical informations on the species of selected areas in Nilgiri Biosphere Reserve, the Western Ghats, India. J. Res. Biol. 2015;5:43–57. [Google Scholar]
- Venkatachalapathi A., Sangeeth T., Ali M.A., Tamilselvi S.S., Paulsamy S. Ethnomedicinal assessment of Irula tribes of Walayar valley of Southern Western Ghats, India. Saudi J. Biol. Sci. 2018;25:760–775. doi: 10.1016/j.sjbs.2016.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vijayakumar M., Govindarajan R., Rao G.M.M., Rao C.H.V., Shirwaikar A., Mehrotra S., Pushpangadan P. Action of Hygrophila auriculata against streptozotocin-induced oxidative stress. J. Ethnopharmacol. 2006;104:356–361. doi: 10.1016/j.jep.2005.09.030. [DOI] [PubMed] [Google Scholar]
- Von F.H.C. University of California Press; Berkeley: 1982. Tribes of India: The Struggle for Survival. [Google Scholar]