Abstract
MicroRNAs (miRNAs) have emerged as important regulators of bone development and regeneration. The aim of the present study was to determine whether miR-203a-3p.1 is involved in osteogenic differentiation of multiple myeloma (MM)-mesenchymal stem cells (MSCs) and the potential underlying mechanism. MSCs were isolated from patients with MM and normal subjects and confirmed by flow cytometry using specific surface markers. The osteogenic differentiation capacity of MM-MSCs was identified by Alizarin Red S calcium deposition staining and reverse transcription-quantitative PCR (RT-qPCR) of typical osteoblast differentiation markers. The role of miR-203a-3p.1 in the osteoblast differentiation of MM-MSCs was determined by gain or loss of function experiments. The target of miR-203a-3p.1 was identified using bioinformatics (including the miRNA target prediction database TargetScan, miRDB, DIANA TOOLS and venny 2.1.0), luciferase reporter assay, RT-qPCR and western blotting. The expression levels of proteins involved in the Wnt3a/β-catenin signaling pathway were detected by western blot analysis. The results revealed that the osteogenic differentiation capacity of MM-MSCs was reduced when compared with normal (N)-MSCs, as demonstrated by a decrease in calcium deposition and mRNA expression of typical osteoblast differentiation markers, including ALP, OPN and OC. In addition, miR-203a-3p.1 was downregulated in N-MSCs following osteoblast induction, whereas no changes were observed in MM-MSCs. The downregulation of miR-203a-3p.1 resulted in increased osteogenic potential, as indicated by the increase in the mRNA expression levels of the typical osteoblast differentiation markers, including alkaline phosphatase (ALP), osteopontin (OPN) and osteocalcin (OC). Bioinformatics and luciferase reporter assay analysis indicated that mothers against decapentaplegic homolog 9 (Smad9) may be a direct target of miR-203a-3p.1 in N-MSCs. The RT-qPCR and western blot assays revealed that overexpression of smad9 significantly enhanced the effect of miR-203a-3p.1 inhibitors on osteoblast markers, which indicated that miR-203a-3p.1 inhibitors may regulate the osteogenic differentiation of MM-MSCs by upregulating Smad9. In addition, the Wnt3a/β-catenin signaling pathway was activated following miR-203a-3p.1 inhibition. These results suggest that miR-203a-3p.1 may serve an important role in the osteogenic differentiation of MM-MSCs by regulating Smad9 expression.
Keywords: multiple myeloma, mesenchymal stem cells, microRNA-203a-3p.1, mothers against decapentaplegic homolog 9, Wnt3a/β-catenin
Introduction
Multiple myeloma (MM) is a clonal plasma cell malignancy that strongly depends on interactions with its microenvironment (1). A major complication of MM is the development of osteolytic lesions, which are caused by an imbalance between osteoclastic bone resorption and impaired osteoblastic bone formation and lead to severe bone pain, fractures, osteoporosis and hypercalcemia (2). Mesenchymal stem cells (MSCs) are involved in bone repair and regeneration as they can differentiate into osteoblasts and osteocytes (3). A previous study has indicated that the osteogenic differentiation of MSCs obtained from patients with MM (MM-MSCs) is impaired (4). Thus, an efficient strategy that induces osteogenic differentiation of MM-MSCs is required to improve the wellbeing of patients with MM.
MicroRNAs (miRNAs/miRs) are a large class of small, noncoding RNA molecules of 17–25 nucleotides that are involved in gene regulation at the post-transcriptional level by binding to the 3′-untranslated regions (UTRs) of target mRNAs and have been demonstrated to serve critical roles in a number of biological processes (5). Previous studies have suggested that miRNAs contribute to bone development (6–8). Several miRNAs have been identified as regulators of osteogenesis; for instance, miR-133a-5p has been demonstrated to target runt-related transcription factor 2 (RUNX2) and inhibit the expression of osteoblast differentiation-associated markers (9). In addition, miR-20a promotes osteoblast differentiation and bone formation of human MSCs by co-regulating bone morphogenetic protein signaling (10). miR-203a is expressed in keratinocytes and affects their growth, differentiation and function (11). Additionally, miR-203a has been demonstrated to increase tumor growth in a number of types of cancer (12,13). A recent study reported that miR-203-3p inhibited osteogenesis in the jaws of diabetic rats and in rat bone marrow mesenchymal stem cells cultured in high-glucose medium by directly targeting mothers against decapentaplegic (Smad) homolog 1 (Smad1) (14). However, to the best of our knowledge, the role of miR-203a-3p.1 in the osteogenic differentiation of MM-MSCs has not been identified. Thus, the aim of the present study was to characterize the expression of miR-203a-3p.1 in MM-MSCs and to investigate its effects on osteoblast differentiation, as well as the potential molecular mechanisms.
Materials and methods
Patients and subjects
A total of five patients with newly diagnosed stage IIIA-IIIB of MM (age range, 40–63 years; 2 males, 3 females) and 5 normal healthy subjects (age range, 32–48 years; 3 males, 2 females) were recruited in the present study. The bone marrow samples were obtained at The General Hospital of Western Theater Command (Chengdu, China) from April 2017 to April 2018 according to the institutional guidelines. The present study was approved by the General Hospital of Western Theater Command (Chengdu, China). All volunteers provided written informed consent.
Bone marrow (BM)-MSC isolation and propagation
MSCs were isolated from BM samples. Briefly, the BM fluid was mixed with an equal volume of Ficoll (Tianjin Haoyang Biological Products Technology Co., Ltd.), and mononuclear cells were obtained following centrifugation at 450 × g for 20 min at room temperature. The cells were seeded in a T25 cell culture bottle at 5,000 cells/cm2 with Dulbecco's modified Eagle's medium (DMEM; HyClone; GE Healthcare Life Sciences) supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.). The medium was replaced twice weekly until the cultures attained 80% confluence.
MSC identification by flow cytometry
In the primary cells, CD44-PE, CD90-FITC and CD105-PC5.5 were used to isolate MSCs. Briefly, MSCs were resuspended in 4 ml PBS following digestion and centrifugation. Subsequently, MSCs were incubated with 40 µl mouse anti-human CD90-FITC (1:100), CD44-PE (1:100) and CD105-PerCP-Cy™5.5 (1:100) for 30 min at 4°C. MSCs were isolated using a FACScalibur flow cytometer (BD Biosciences). Following 4 culture passages, the MSCs with very low fluorescence value (Blank) were sorted, and single staining was used to identify MSCs. Following washing, trypsin digestion and centrifugation, the sorted MSCs were resuspended in 100 µl PBS (1×106 cells) and stained with 5 µl mouse anti-human CD90-FITC (1:20), CD44-PE (1:20), CD34-FITC (1:20), CD105-PerCP-Cy™5.5 (1:20) and CD45-PE antibodies (1:20) for 30 min at 4°C. After rinsing twice with PBS and resuspending in 500 µl of PBS, the cells were analyzed using a FACScalibur flow cytometer (BD Biosciences) and Cell Quest 3.3 software (BD Biosciences), based on their characteristic immunophenotype of CD44+, CD90+, CD105+, CD34− and CD45−. The antibodies against CD45-PE (cat. no. 304008) and CD34-FITC (cat. no. 343503) were purchased from BioLegend, Inc. The BD Stemflow™ Human MSC Analysis kit (cat. no. 562245; BD Biosciences) included CD90-FITC, CD105- PerCP-Cy™5.5 and CD44-PE antibodies.
MSC differentiation
Following 3 passages, MSCs were seeded in 6-well plates at 1×105 cells/well with DMEM supplemented with 10% FBS and cultured until cell confluence reached 60–80%. Fresh DMEM containing 10% FBS, 1×10−8 mol/l dexamethasone, 10 mmol/l sodium β-glycerophosphate and 50 µg/ml vitamin C was used to incubate the MSCs for 2 weeks (15) and replaced twice a week to induce MSC differentiation into osteoblasts (16).
Alizarin Red S staining
Following osteogenesis differentiation, MSCs were washed with PBS and fixed in 4% paraformaldehyde for 20 min at room temperature. MSCs were stained with Alizarin Red S (1%, pH 4.2; Novon Scientific; Beijing Xinhua Luyuan Technology Co., Ltd.) for 5 min at room temperature. Images of the cells were captured using a light microscope.
Cell transfection
MSCs were seeded into a 6-well plate at a density of 1×105 cells/well the day prior to transfection. Transfection was performed using Lipofectamine® 2000 (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol with 50 pmol/ml miR-203a-3p inhibitor or mimic (Guangzhou RiboBio Co., Ltd.) at 37°C. Fresh medium was added at 6 h. Cells were harvested for analysis following transfection for 24 h.
For the transfection of Smad9, lentiviruses overexpressing Smad9 (lv-Smad9; Smad9 mRNA sequence, NM_001127217; vector name, GV492) and the corresponding control lentiviruses (lv-green fluorescent protein) were purchased from Shanghai GeneChem Co., Ltd. MSCs were seeded into a 6-well plate at a density of 1×105 cells/well. Lentivirus (1×108 TU/ml) infection was performed when the cells reached 20–40% confluence. The lentiviruses were transfected into MSCs with a multiplicity of infection of 30. After transfection for 8–12 h, fresh medium was added for further incubation. Cells were collected for RT-qPCR or western blotting analysis 72 h after transfection.
Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was isolated from the cells (1×105 cells/well) using TRIzol® reagent (Takara Biotechnology, Co., Ltd.). Following isolation, 4 µg RNA was reverse-transcribed into cDNA using PrimeScript™ RT reagent kit (Takara Biotechnology, Co., Ltd.) according to the manufacturer's protocol at 37°C for 15 min and 85°C for 5 sec. qPCR was performed on an Applied Biosystems 7500 Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.) using SYBR Premix Ex Taq II (Takara Biotechnology, Co., Ltd.) in a 25 µl mixture containing 2 µl cDNA templates, 12.5 µl 2X SYBR Premix Ex Taq II, 1 µl each primer and 8.5 µl DNase/RNase-free H2O. The thermocycling conditions were as follows: 3 min at 95°C; 40 cycles of 95°C for 5 sec and 60°C for 30 sec; followed by 72°C for 30 sec. For the quantification of miRNA expression, RT was performed at 42°C for 60 min and 70°C for 10 min using Bulge-Loop™ miRNA RT-qPCR Primer and Bulge-Loop™ miRNA RT-qPCR Starter kit (cat. no. C10211-1; Guangzhou RiboBio Co., Ltd.). Gene expression levels were quantified at 95°C for 10 min, followed by 40 cycles of 95°C for 2 sec, 60°C for 20 sec and 70°C for 10 sec. The 2−∆∆Cq value was used for comparative quantitation (17). β-actin and U6 small nuclear RNA genes were used as endogenous normalization controls. The primer sequences (Sangon Biotech Co., Ltd.) are listed in Table I. The catalogue number of miR-203a-3p primer was miRA0000264-1-200, and the catalogue number of U6 was miRAN0002-1-200.
Table I.
Primer sequences for reverse transcription-quantitative PCR.
| Gene | Primer sequence (5′→3′) |
|---|---|
| ALP | F: GACCTCCTCGGAAGACACTCTG |
| R: CGCCTGGTAGTTGTTGTGAGC | |
| OPN | F: GCCGACCAAGGAAAACTCACT |
| R: GGCACAGGTGATGCCTAGGA | |
| OC | F: CCAGGCGCTACCTGTATCAATG |
| R: ATGTGGTCAGCCAACTCGTCA | |
| Smad9 | F: GCAGCCTCAAGGTCTTCAACAAC |
| R: CATGAAGATGAATCTCAATCCAGGA | |
| β-actin | F: TGGCACCCAGCACAATGAA |
| R: CTAAGTCATAGTCCGCCTAGAAGCA |
ALP, alkaline phosphatase; OPN, osteopontin; OC, osteocalcin; Smad9, mothers against decapentaplegic homolog 9.
Western blot analysis
The MSCs from three normal healthy subjects were randomly selected and seeded into 6-well plates at 1×105 cells/well after four culture passages. Total protein was extracted using radioimmunoprecipitation assay lysis buffer (Wuhan Boster Biological Technology, Ltd.). Protein concentration was quantified using a BCA Protein Assay kit (Wuhan Boster Biological Technology, Ltd.). The protein samples (20 µg) were separated by 10% SDS-PAGE and transferred onto a polyvinylidene difluoride membrane (EMD Millipore). Following blocking with 5% skimmed milk powder at 37°C for 1 h, the membranes were incubated with primary antibodies against Smad9 (cat. no. ab115900; 1:500; Abcam), Wnt3a (cat. no. ab28472; 1:500; Abcam), β-catenin (cat. no. 8480; 1:1,000; Cell Signaling Technology, Inc.), glycogen synthase kinase (GSK)-3β (cat. no. 12456; 1:1,000; Cell Signaling Technology, Inc.) and β-actin (cat. no. 4970; 1:1,000; Cell Signaling Technology, Inc.) at 4°C overnight, followed by a HRP-conjugated goat anti-rabbit immunoglobulin G secondary antibody (cat. no. BA1054; 1:5,000; Wuhan Boster Biological Technology, Ltd.) at room temperature for 1 h. The protein bands were visualized using a ChemiDoc™ MP imaging system (Bio-Rad Laboratories, Inc.). Protein levels were calculated relative to β-actin.
Luciferase assay
miR-203a-3p.1 targets were predicted using bioinformatics software, including TargetScan (http://www.targetscan.org/), miRDB (http://mirdb.org), DIANA TOOLS (http://diana.imis.athena-innovation.gr) and venny 2.1.0 (http://bioinfogp.cnb.csic.es/tools/venny/index.html). MSCs were plated in 24-well plates (4×104 cells/well). When the cultures attained 50% confluence, cells were co-transfected with the Renilla luciferase pRL-TK plasmid (100 ng/ml; Shanghai GenePharma Co., Ltd.) plus the recombinant Firefly luciferase pGL3 reporters containing the 3′-untranslated region (3′-UTR) of human Smad9 (2 µg/ml; Shanghai GenePharma Co., Ltd.) in combination with miR-203a-3p.1 mimic and miR-203a-3p.1 inhibitor using Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.). Luciferase activity was detected at 24 h using a Dual-Luciferase Reporter Assay kit (cat. no. E1910; Promega Corporation). Firefly luciferase activity was normalized to Renilla luciferase activity for each tested well.
Statistical analysis
Statistical analysis was performed using SPSS 20.0 (IBM Corp.). The data are expressed as the mean ± standard deviation. Comparisons between two groups were analyzed by unpaired Student's t-test (for parametric data) or Mann-Whitney U test (for non-parametric data). Differences among multiple groups were compared by one-way analysis of variance (ANOVA) with Dunnett's post hoc test or two-way ANOVA with Bonferroni's post hoc test. P<0.05 was considered to indicate a statistically significant difference, and P<0.01 was considered to indicate a highly significant difference.
Results
MSCs from patients with MM exhibit decreased osteogenic differentiation
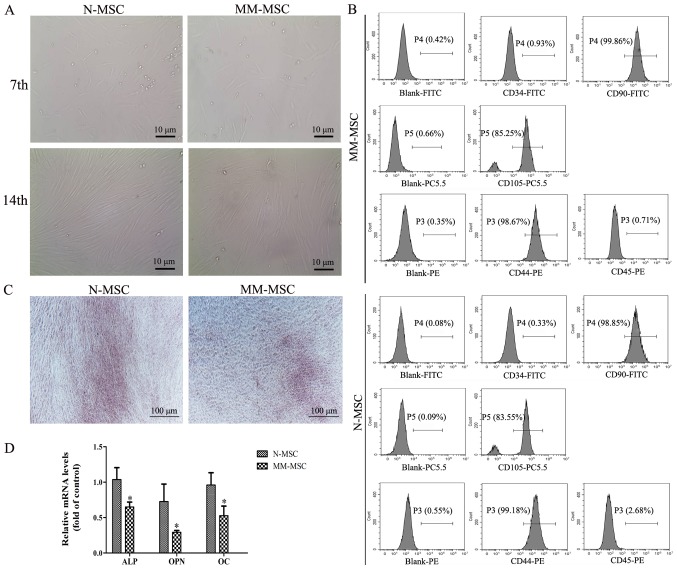
Following 7 days of primary culture, the adherent cells exhibited colony growth and reached >40% confluence. The cells were fusiform and pleomorphic. Following 14 days of primary culture, the cells attained 60–70% confluence and had regular morphology and a long spindle shape (Fig. 1A). After 4 culture passages, the cell surface markers were detected by flow cytometry and the results revealed that MSCs were negative for CD34 and CD45, but positive for the CD44, CD90 and CD105 markers (Fig. 1B). These results suggested that the cultured cells were MSCs.
Figure 1.
MM-MSCs exhibit a reduced osteogenic differentiation capacity. (A) Images of MSCs in primary culture on the 7th and 14th day captured using an inverted microscope (magnification, ×400). (B) The surface markers of the third generation MM-MSCs and N-MSCs were identified by flow cytometry Blank group, isolated MSCs from the primary cells at the fourth generation with no fluorescence detected. (C) Following 21 days of osteogenic induction, calcium deposition was evaluated using Alizarin Red S staining (magnification, ×40). (D) Reverse transcription-quantitative PCR was performed to detect the mRNA levels of ALP, OPN and OC following osteogenic induction in N-MSCs and MM-MSCs. *P<0.05 vs. N-MSC. MM, multiple myeloma; N, normal; MSCs, mesenchymal stem cells; ALP, alkaline phosphatase; OPN, osteopontin; OC, osteocalcin; CD, cluster of differentiation.
The osteogenic differentiation capacity of MSCs from patients with MM and normal subjects was investigated using Alizarin Red S staining, which revealed that the calcium deposition of MM-MSCs was lower compared with MSCs derived from normal healthy subjects (N-MSCs) (Fig. 1C). RT-qPCR results demonstrated an decrease in mRNA expression levels of typical osteoblast differentiation markers in MM-MSCs compared with N-MSCs, including alkaline phosphatase (ALP), osteopontin (OPN) and osteocalcin (OC) (Fig. 1D). These results indicated that the osteogenic differentiation capacity of MM-MSCs may be reduced.
Effects of miR-203a-3p.1 on osteogenic differentiation in MM-MSCs
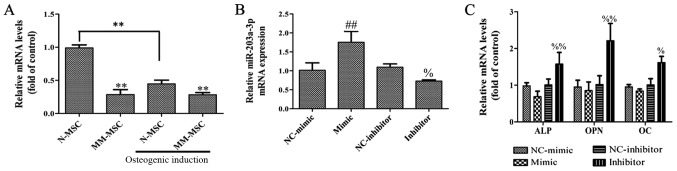
To determine whether miR-203a-3p.1 is associated with osteogenesis, the expression levels of miR-203a-3p.1 in MSCs from patients with MM and normal subjects were analyzed by RT-qPCR. The results revealed that the expression of miR-203a-3p.1 in MM-MSCs was significantly lower compared with that in N-MSCs. In addition, the expression of miR-203a-3p.1 in N-MSCs was significantly decreased following osteoblast induction, whereas no evident change was observed in MM-MSCs; therefore, it was hypothesized that the decrease in the expression of miR-203a-3p.1 contributes to osteogenic differentiation in N-MSCs, whereas in MM-MSCs, the decreased expression of miR-203a-3p.1 was inhibited, which resulted in the reduced osteogenic differentiation capacity of MM-MSCs. To study the role of miR-203a-3p.1 on osteogenic differentiation, MM-MSCs cells were transfected with an miR-203a-3p.1 mimic and inhibitor. RT-qPCR results revealed that the miR-203a-3p.1 mRNA expression levels were significantly increased in the mimic group and decreased in the inhibitor group compared with the corresponding negative control groups (Fig. 2B). In N-MSCs transfected with the miR-203a-3p.1 inhibitor, mRNA expression levels of ALP, OPN and OC increased; however, the overexpression of miR-203a-3p.1 had no significant effects on osteogenic differentiation marker expression (Fig. 2C). These results indicate that miR-203a-3p.1 inhibition may increase osteogenic differentiation.
Figure 2.
Effects of miR-203a-3p.1 inhibition on the osteogenic differentiation of N-MSCs. (A) The miR-203a-3p.1 mRNA expression levels in N-MSCs and MM-MSCs were detected by RT-qPCR prior to and following osteoblast induction. (B) The miR-203a-3p.1 mRNA expression levels in N-MSCs were detected by RT-qPCR following transfection with miR-203a-3p.1 mimic or inhibitor. (C) The mRNA expression levels of ALP, OPN and OC in N-MSCs were detected by RT-qPCR following transfection with miR-203a-3p.1 mimic or inhibitor. **P<0.01 vs. N-MSC; ##P<0.01 vs. NC-mimic; %P<0.05 and %%P<0.01 vs. NC-inhibitor. miR, microRNA; MM, multiple myeloma; N, normal; MSCs, mesenchymal stem cells; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; ALP, alkaline phosphatase; OPN, osteopontin; OC, osteocalcin; NC, negative control; mimic, miR-203a-3p.1-mimic; inhibitor, miR-203a-3p.1-inhibitor.
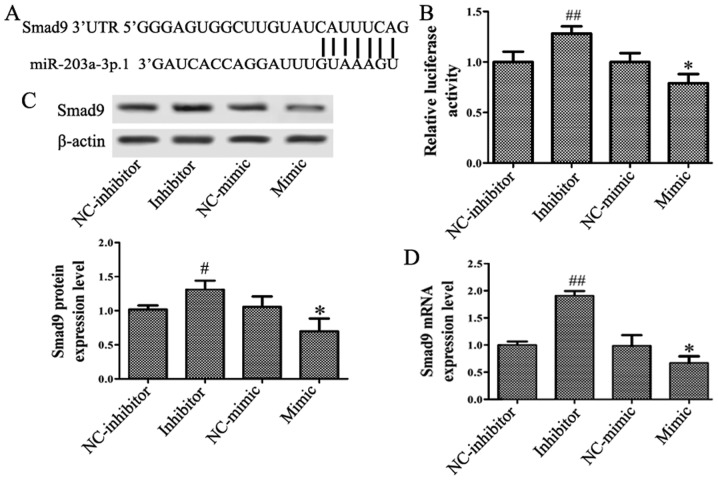
Smad9 is a target of miR-203a-3p.1
The miRNA target prediction databases TargetScan, miRDB, DIANA TOOLS and venny 2.1.0 were used to identify the target genes of miR-203a-3p.1 in osteogenesis. A conserved putative target site for miR-203a-3p.1 was identified in the 3′-UTR of the Smad9 gene (Fig. 3A). To assess whether Smad9 may be regulated by miR-203a-3p.1, the N-MSCs were transfected with miR-203a-3p.1 mimic and inhibitor, respectively. The luciferase assay revealed that the miR-203a-3p.1 mimic significantly repressed the luciferase activity and the miR-203a-3p.1 inhibitor increased luciferase activity (Fig. 3B). RT-qPCR and western blotting results demonstrated that the mRNA and protein levels of Smad9 were reduced when miR-203a-3p.1 was overexpressed; by contrast, the expression of Smad9 increased following treatment with miR-203a-3p.1 inhibitor (Fig. 3C and D). These results indicated that miR-203a-3p.1 may target Smad9 and negatively regulate Smad9 expression.
Figure 3.
miR-203a-3p.1 targets Smad9 in N-MSCs. (A) Putative miR-203a-3p.1-binding sequence of Smad9. (B) N-MSCs were co-transfected with miR-203a-3p.1 mimic or inhibitor and the reporter plasmid containing the 3′-UTR of Smad9. miR-203a-3p.1 inhibitor enhanced luciferase activity, while the miR-203a-3p.1 mimic reduced luciferase activity. (C and D) Smad9 protein and mRNA expression levels were detected by western blot and RT-qPCR following transfection with miR-203a-3p.1 inhibitor or mimic, respectively. miR-203a-3p.1 inhibitor increased the expression levels of Smad9 protein and mRNA, while miR-203a-3p.1 mimic decreased the expression levels of Smad9 protein and mRNA. *P<0.05 vs. NC-mimic; #P<0.05 and ##P<0.01 vs. NC-inhibitor. miR, microRNA; MM, multiple myeloma; MSCs, mesenchymal stem cells; UTR, untranslated region; Smad9, mothers against decapentaplegic homolog 9; NC, negative control; mimic, miR-203a-3p.1-mimic; inhibitor, miR-203a-3p.1-inhibitor.
Inhibition of miR-203a-3p.1 mediates osteogenic differentiation through Smad9
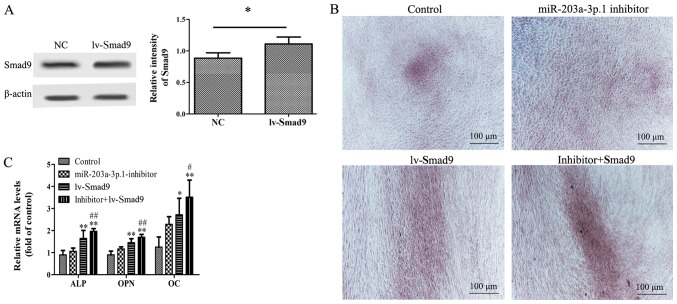
To investigate the association between miR-203a-3p.1 and Smad9 during osteogenic differentiation, a lentiviral vector overexpressing Smad9 was used. Western blotting results demonstrated that Smad9 protein expression levels were significantly increased in N-MSCs following lentiviral vector transfection (Fig. 4A). Furthermore, co-transfection of miR-203a-3p.1 inhibitor with lv-Smad9 increased calcium deposition and mRNA expression levels of ALP, OPN and OC in MM-MSCs (Fig. 4B and C).
Figure 4.
Effects of Smad9 overexpression on miR-203a-3p.1 inhibitor-mediated osteogenic differentiation in N-MSCs. (A) Smad9 protein expression levels were detected by western blot analysis following transfection with lentivirus. (B) N-MSCs transfected with miR-203a-3p.1 inhibitors were infected with lv-Smad9. Images of Alizarin Red S staining were captured using an inverted microscope (magnification, ×40). Co-transfection of miR-203a-3p.1 inhibitor with lv-Smad9 increased the calcium deposition. (C) The ALP, OPN and OC mRNA expression levels were detected by reverse transcription-quantitative polymerase chain reaction following transfection with miR-203a-3p.1 inhibitor or lentiviral vector. *P<0.05 and **P<0.01 vs. control group; #P<0.05 and ##P<0.01 vs. miR-203a-3p.1 inhibitor group. miR, microRNA; MM, multiple myeloma; MSCs, mesenchymal stem cells; ALP, alkaline phosphatase; OPN, osteopontin; OC, osteocalcin; Smad9, mothers against decapentaplegic homolog 9; lv-, lentivirus; inhibitor: miR-203a-3p.1-inhibitor.
Thus, overexpression of Smad9 appeared to enhance the miR-203a-3p.1-knockdown-mediated promotion of osteogenic differentiation. These results indicated that the inhibition of miR-203a-3p.1 increased osteogenic differentiation, in part via the upregulation of Smad9 expression.
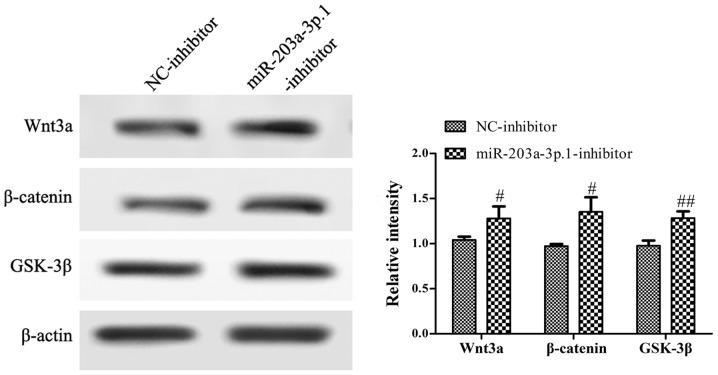
miR-203a-3p.1 may inhibit the osteogenic differentiation of N-MSCs by inhibiting the Wnt3a/β-catenin signaling pathway
To assess whether the impairment of osteogenic differentiation in N-MSCs was due to abnormalities in the Wnt3a/β-catenin signaling pathway, the protein expression levels of genes involved in the Wnt3a/β-catenin signaling pathway were analyzed by western blotting. The results revealed that the miR-203a-3p.1 inhibitor significantly increased the expression of Wnt3a, β-catenin and GSK-3β (Fig. 5).
Figure 5.
Inhibition of miR-203a-3p.1 activates the Wnt3a/β-catenin signaling pathway. The key proteins of the Wnt3a/β-catenin signaling pathway were detected using western blotting following N-MSC transfection with miR-203a-3p.1-inhibitor. #P<0.05 and ##P<0.01. miR, microRNA; MM, multiple myeloma; MSC, mesenchymal stem cells; NC, negative control; inhibitor, miR-203a-3p.1-inhibitor; GSK-3β, glycogen synthase kinase 3β.
Discussion
MM is a common malignancy characterized by the abnormal proliferation of clonal plasma cells in BM (18). One of the characteristics of MM is bone lesions due to a severe imbalance in bone remodeling (19). MSCs serve as a basic cellular unit of embryonic bone formation, which has a key role in fracture repair and bone regeneration (20). However, the osteogenic differentiation of MSCs obtained from patients with MM is impaired, which leads to decreased osteogenesis, increased adipogenesis and osteonecrosis (4,21,22). Maintaining a balance between the levels of osteoclasts and osteoblasts regulates bone homeostasis (23). Osteoblasts promote bone formation by secreting ALP and the bone matrix protein that induces bone matrix mineralization (24). In the present study, the osteogenic differentiation capacity of MM-MSCs was significantly lower compared with that of N-MSCs, as indicated by the decrease in calcium deposition and the mRNA expression levels of the typical osteoblast differentiation markers, including ALP, OPN and OC. These results demonstrated that the osteogenic differentiation of MSCs was inhibited in patients with MM. Thus, understanding the mechanisms leading to the decreased osteogenic differentiation capability of MM-MSCs may explain the osteogenesis defects in patients with MM. In further studies, more bone marrow samples will be used to verify this result and further study the mechanism underlying decreased osteogenic differentiation of MSCs in patients with MM.
miRNAs have emerged as essential regulatory molecules of gene expression that participate in the regulation of bone homeostasis through transcriptional inhibition or mRNA cleavage (25). The majority of miRNAs have been described as signaling network nodes that serve a vital role in osteoblastic differentiation processes; for instance, miR-99a serves as a novel regulator of lysine demethylase 6B to regulate the osteogenic differentiation of BM stromal cells (26). In addition, miR-590-5p promotes osteoblast differentiation by indirectly protecting and stabilizing the Runx2 protein by targeting Smad7 gene expression in MSCs (27). Tang et al (14) demonstrated that miR-203-3p participates in the suppression of diabetes-associated osteogenesis in the jaw bone through targeting smad1. The results of the present study revealed decreased expression levels of miR-203a-3p.1 in MM-MSCs. Following osteoblast induction, the miR-203a-3p.1 mRNA expression level in N-MSCs was significantly decreased, whereas no change was observed in MM-MSCs. These results indicated that the downregulation of miR-203a-3p.1 may contribute to the osteogenic differentiation of normal MSCs. In agreement with these results, the typical osteoblast differentiation markers ALP, OPN and OC were upregulated in MM-MSCs following treatment with the miR-203a-3p.1 inhibitor. No changes were observed in ALP, OPN and OC mRNA levels in MM-MSCs overexpressing miR-203a-3p.1. These results suggested that the inhibition of miR-203a-3p.1 may increase osteoblast differentiation of MM-MSCs.
Transforming growth factor (TGF)-β signaling is an important pathway in osteoblastic differentiation. Through putative target prediction, the present study identified that miR-203a-3p.1 may target Smad9, which is an important component of the TGF-β signaling pathway. Previous studies have reported that several SMADs, including Smad3, Smad7 and Smad5, are involved in bone formation, remodeling and maintenance (27–29). Smad9 is upregulated during chondrocyte differentiation (30), whereas its expression and role in osteoclast differentiation has not been studied. In the present study, a conserved putative target site for miR-203a-3p.1 was identified in the 3′-UTR of Smad9. Furthermore, the RT-qPCR and western blotting assays revealed a negative association between Smad9 and miR-203a-3p.1. Luciferase reporter analysis indicated that Smad9 may be a direct target of miR-203a-3p.1 in MM-MSCs. Rescue experiments demonstrated that overexpression of Smad9 significantly enhanced the effect of the miR-203a-3p.1 inhibitor on osteoblast marker expression, which indicated that inhibition of miR-203a-3p.1 mediated promotion of osteogenic differentiation partially by upregulating Smad9. In further studies, Smad9 gene silencing will be used to validate the effects of Smad9 overexpression on osteoblast marker expression.
Wnt proteins are a large family of highly conserved secreted signaling molecules that mediate essential biological processes such as embryogenesis, organogenesis and tumorigenesis (31). In addition, Wnt/β-catenin is an important signaling pathway that regulates osteoblast differentiation and bone formation. Chen et al (32) reported that the knockdown of Sirtuin-7 increased the osteogenic differentiation of human BM-MSCs by activating the Wnt/β-catenin signaling pathway. In addition, baicalein promoted the osteogenic differentiation of human periodontal ligament cells by activating the Wnt/β-catenin signaling pathway (33). In the present study, the partial inhibition of miR-203a-3p.1 increased the protein expression levels of Wnt3a, β-catenin and GSK-3β in N-MSCs. These results indicated that the miR-203a-3p.1 inhibitor enhanced the osteoblast differentiation of MM-MSCs potentially by activating the Wnt/β-catenin signaling pathway.
In conclusion, the present study demonstrated that miR-203a-3p.1 was downregulated in MM-MSCs and may participate in osteogenic differentiation. Furthermore, the inhibition of miR-203a-3p.1 increased osteoblast differentiation by directly targeting Smad9. The potential mechanism may be associated with the activation of the Wnt3a/β-catenin signaling pathway. The present study revealed a potential function of miR-203a-3p.1 in the osteogenic differentiation of MM-MSCs, which may be targeted to develop a promising therapeutic against myeloma bone disease in the future.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Key Research and Development Program of China (grant no. 2017YFA0105502), the Surface Project of National Natural Science Foundation of China (grant no. 81570097), the Basic and Frontier Research Project of Chongqing (grant no. cstc2015jcyjBX0077) and the Science and Technology Innovation Special Project of Social Undertakings and People's Livelihood Security of Chongqing (grant no. cstc2016shms-ztzx10003).
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
FYF, RD, YS and XZ conceived and designed the experiments. FYF, RD, LQ, QW, YZ and LG performed the experiments. CZ, PK, JZ, NZ and ZL analyzed the data and assisted with the experiments. FYF, RD, YS and XZ wrote the manuscript. YS and XZ revised the manuscript and supervised the study. All authors have read and approved the final version of this manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of General Hospital of Western Theater Command (Chengdu, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.López-Corral L, Gutiérrez NC, Vidriales MB, Mateos MV, Rasillo A, García-Sanz R, Paiva B, San Miguel JF. The progression from MGUS to smoldering myeloma and eventually to multiple myeloma involves a clonal expansion of genetically abnormal plasma cells. Clin Cancer Res. 2011;17:1692–1700. doi: 10.1158/1078-0432.CCR-10-1066. [DOI] [PubMed] [Google Scholar]
- 2.Johnson DC, Weinhold N, Mitchell J, Chen B, Stephens OW, Försti A, Nickel J, Kaiser M, Gregory WA, Cairns D, et al. Genetic factors influencing the risk of multiple myeloma bone disease. Leukemia. 2016;30:883–888. doi: 10.1038/leu.2015.342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Robey PG, Kuznetsov SA, Ren J, Klein HG, Sabatino M, Stroncek DF. Generation of clinical grade human bone marrow stromal cells for use in bone regeneration. Bone. 2015;70:87–92. doi: 10.1016/j.bone.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Raje N, Roodman GD. Advances in the biology and treatment of bone disease in multiple myeloma. Clin Cancer Res. 2011;17:1278–1286. doi: 10.1158/1078-0432.CCR-10-1804. [DOI] [PubMed] [Google Scholar]
- 5.Gregory RI, Shiekhattar R. MicroRNA biogenesis and cancer. Cancer Res. 2005;65:3509–3512. doi: 10.1158/0008-5472.CAN-05-0298. [DOI] [PubMed] [Google Scholar]
- 6.Chen J, Qiu M, Dou C, Cao Z, Dong S. MicroRNAs in bone balance and osteoporosis. Drug Dev Res. 2015;76:235–245. doi: 10.1002/ddr.21260. [DOI] [PubMed] [Google Scholar]
- 7.Fang S, Deng Y, Gu P, Fan X. MicroRNAs regulate bone development and regeneration. Int J Mol Sci. 2015;16:8227–8253. doi: 10.3390/ijms16048227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lian JB, Stein GS, van Wijnen AJ, Stein JL, Hassan MQ, Gaur T, Zhang Y. MicroRNA control of bone formation and homeostasis. Nat Rev Endocrinol. 2012;8:212–227. doi: 10.1038/nrendo.2011.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang W, Wu Y, Shiozaki Y, Sugimoto Y, Takigawa T, Tanaka M, Matsukawa A, Ozaki T. miRNA-133a-5p inhibits the expression of osteoblast differentiation-associated markers by targeting the 3′ UTR of RUNX2. DNA Cell Biol. 2018;37:199–209. doi: 10.1089/dna.2017.3936. [DOI] [PubMed] [Google Scholar]
- 10.Zhang JF, Fu WM, He ML, Xie WD, Lv Q, Wan G, Li G, Wang H, Lu G, Hu X, et al. MiRNA-20a promotes osteogenic differentiation of human mesenchymal stem cells by co-regulating BMP signaling. RNA Biol. 2011;8:829–838. doi: 10.4161/rna.8.5.16043. [DOI] [PubMed] [Google Scholar]
- 11.Ma X, Li L, Jia T, Chen M, Liu G, Li C, Li N, Yang D. miR-203a controls keratinocyte proliferation and differentiation via targeting the stemness-associated factor ΔNp63 and establishing a regulatory circuit with SNAI2. Biochem Biophys Res Commun. 2017;491:241–249. doi: 10.1016/j.bbrc.2017.07.131. [DOI] [PubMed] [Google Scholar]
- 12.Hu G, Lai P, Liu M, Xu L, Guo Z, Liu H, Li W, Wang G, Yao X, Zheng J, Xu Y. miR-203a regulates proliferation, migration, and apoptosis by targeting glycogen synthase kinase-3β in human renal cell carcinoma. Tumor Biol. 2014;35:11443–11453. doi: 10.1007/s13277-014-2476-x. [DOI] [PubMed] [Google Scholar]
- 13.Huo W, Du M, Pan X, Zhu X, Gao Y, Li Z. miR-203a-3p.1 targets IL-24 to modulate hepatocellular carcinoma cell growth and metastasis. FEBS Open Bio. 2017;7:1085–1091. doi: 10.1002/2211-5463.12248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tang Y, Zheng L, Zhou J, Chen Y, Yang L, Deng F, Hu Y. miR-203-3p participates in the suppression of diabetes-associated osteogenesis in the jaw bone through targeting Smad1. Int J Mol Med. 2018;41:1595–1607. doi: 10.3892/ijmm.2018.3373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shi YY, Wang GL, Yang HL, Lu SZ, Zhang Y, Cai X. Repairing rabbit femur bone defects by porous silk fibroin/hydroxyapatite combined with adipose-derived stromal cells. J Clin Rehabil Tissue Eng Res. 2010;14:1341–1344. [Google Scholar]
- 16.Langenbach F, Handschel J. Effects of dexamethasone, ascorbic acid and β-glycerophosphate on the osteogenic differentiation of stem cells in vitro. Stem Cell Res Ther. 2013;4:117. doi: 10.1186/scrt328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 18.Guo J, Fei C, Zhao Y, Zhao S, Zheng Q, Su J, Wu D, Li X, Chang C. Lenalidomide restores the osteogenic differentiation of bone marrow mesenchymal stem cells from multiple myeloma patients via deactivating Notch signaling pathway. Oncotarget. 2017;121:55405–55421. doi: 10.18632/oncotarget.19265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yaccoby S. Advances in the understanding of myeloma bone disease and tumour growth. Br J Haematol. 2010;149:311–321. doi: 10.1111/j.1365-2141.2010.08141.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.He X, Wang H, Jin T, Xu Y, Mei L, Yang J. TLR4 activation promotes bone marrow MSC proliferation and osteogenic differentiation via Wnt3a and Wnt5a signaling. PLoS One. 2016;11:e0149876. doi: 10.1371/journal.pone.0149876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Giuliani N, Rizzoli V, Roodman GD. Multiple myeloma bone disease: Pathophysiology of osteoblast inhibition. Blood. 2006;108:3992–3996. doi: 10.1182/blood-2006-05-026112. [DOI] [PubMed] [Google Scholar]
- 22.Zhuang W, Ge X, Yang S, Huang M, Zhuang W, Chen P, Zhang X, Fu J, Qu J, Li B. Upregulation of lncRNA MEG3 promotes osteogenic differentiation of mesenchymal stem cells from multiple myeloma patients by targeting BMP4 transcription. Stem Cells. 2015;33:1985–1997. doi: 10.1002/stem.1989. [DOI] [PubMed] [Google Scholar]
- 23.Tanaka Y, Nakayamada S, Okada Y. Osteoblasts and osteoclasts in bone remodeling and inflammation. Curr Drug Targets Inflamm Allergy. 2005;4:325–328. doi: 10.2174/1568010054022015. [DOI] [PubMed] [Google Scholar]
- 24.Rodan GA, Martin TJ. Therapeutic approaches to bone diseases. Science. 2000;289:1508–1514. doi: 10.1126/science.289.5484.1508. [DOI] [PubMed] [Google Scholar]
- 25.Pi C, Li YP, Zhou X, Gao B. The expression and function of microRNAs in bone homeostasis. Front Biosci (Landmark Ed) 2015;20:119–138. doi: 10.2741/4301. [DOI] [PubMed] [Google Scholar]
- 26.Tang Y, Zhang L, Tu T, Li Y, Murray D, Tu Q, Chen JJ. MicroRNA-99a is a novel regulator of KDM6B-mediated osteogenic differentiation of BMSCs. J Cell Mol Med. 2018;22:2162–2176. doi: 10.1111/jcmm.13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vishal M, Vimalraj S, Ajeetha R, Gokulnath M, Keerthana R, He Z, Partridge NC, Selvamurugan N. MicroRNA-590-5p stabilizes Runx2 by targeting Smad7 during osteoblast differentiation. J Cell Physiol. 2016;232:371–380. doi: 10.1002/jcp.25434. [DOI] [PubMed] [Google Scholar]
- 28.Hao C, Yang S, Xu W, Shen JK, Ye S, Liu X, Dong Z, Xiao B, Feng Y. MiR-708 promotes steroid-induced osteonecrosis of femoral head, suppresses osteogenic differentiation by targeting SMAD3. Sci Rep. 2016;6:22599. doi: 10.1038/srep22599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ishibashi O, Ikegame M, Takizawa F, Yoshizawa T, Moksed MA, Iizawa F, Mera H, Matsuda A, Kawashima H. Endoglin is involved in BMP-2-induced osteogenic differentiation of periodontal ligament cells through a pathway independent of Smad-1/5/8 phosphorylation. J Cell Physiol. 2010;222:465–473. doi: 10.1002/jcp.21968. [DOI] [PubMed] [Google Scholar]
- 30.Dexheimer V, Gabler J, Bomans K, Sims T, Omlor G, Richter W. Differential expression of TGF-β superfamily members and role of Smad1/5/9-signalling in chondral versus endochondral chondrocyte differentiation. Sci Rep. 2016;6:36655. doi: 10.1038/srep36655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang H, Sun W, Ma J, Pan Y, Wang L, Zhang WB. Biglycan mediates suture expansion osteogenesis via potentiation of Wnt/β-catenin signaling. J Biomech. 2015;48:432–440. doi: 10.1016/j.jbiomech.2014.12.032. [DOI] [PubMed] [Google Scholar]
- 32.Chen EEM, Zhang W, Ye CCY, Gao X, Jiang LLJ, Zhao TTF, Pan ZZJ, Xue DDT. Knockdown of SIRT7 enhances the osteogenic differentiation of human bone marrow mesenchymal stem cells partly via activation of the Wnt/b-catenin signaling pathway. Cell Death Dis. 2017;8:e3042. doi: 10.1038/cddis.2017.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen LJ, Hu BB, Shi XL, Ren MM, Yu WB, Cen SD, Hu RD, Deng H. Baicalein enhances the osteogenic differentiation of human periodontal ligament cells by activating the Wnt/β-catenin signaling pathway. Arch Oral Biol. 2017;78:100–108. doi: 10.1016/j.archoralbio.2017.01.019. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.