Abstract
Previous neuroimaging studies have suggested similar neural activations for word reading in native and second languages. However, such similarities were qualitatively determined (i.e., overlapping activation based on traditional univariate activation analysis). In this study, using representational similarity analysis and an artificial language training paradigm, we quantitatively computed cross‐language neural pattern similarity to examine the modulatory effect of proficiency in the new language. Twenty‐four native Chinese speakers were trained to learn 30 words in a logographic artificial language for 12 days and scanned while performing a semantic decision task after 4‐day training and after 12‐day training. Results showed that higher proficiency in the new language was associated with higher cross‐language pattern similarity in select regions of the reading network.
Keywords: fMRI, language learning, native language, pattern similarity, second language
1. INTRODUCTION
Learning to read one or more foreign languages has become increasingly important in this era of globalization. One of the fundamental questions about bilingualism is how words in the native and second languages are represented in the brain. Most neuroimaging studies have revealed similar activations in the prefrontal cortex, temporoparietal cortex, and occipitotemporal regions when reading in native and second languages (Buchweitz, Shinkareva, Mason, Mitchell, & Just, 2012; Liu, Hu, Guo, & Peng, 2010; Nakada, Fujii, & Kwee, 2001; Wartenburger et al., 2003), especially at the word level (Briellmann et al., 2004; Cao, Tao, Liu, Perfetti, & Booth, 2013; Kim et al., 2016; Mei et al., 2015b; Nelson, Liu, Fiez, & Perfetti, 2009; Van de Putte, De Baene, Brass, & Duyck, 2017; Videsott et al., 2010; Xue, Dong, Jin, Zhang, & Wang, 2004). Other studies, however, have reported differences in certain regions (Jamal, Piche, Napoliello, Perfetti, & Eden, 2012; H. Liu & Cao, 2016; Y. Liu, Dunlap, Fiez, & Perfetti, 2007; Sun et al., 2015; Tham et al., 2005). For instance, Tham et al. (2005) found that the neural activations for Chinese and English words differed in a number of brain regions when Chinese–English bilinguals performed a homophone matching task. Specifically, distinct activations for Chinese were found in the bilateral frontal lobe, right occipital lobe, and left temporal lobe, while distinct activations for English were found in the bilateral frontal and parietal lobes.
Further studies have suggested that proficiency in second language plays an important role in determining the similarities and differences in neural activations between native and second languages (Bowden, Steinhauer, Sanz, & Ullman, 2013; Cao et al., 2013; Gao et al., 2017; Ojima, Nakamura, Matsuba‐Kurita, Hoshino, & Hagiwara, 2011; Ojima, Nakata, & Kakigi, 2005; Rossi, Gugler, Friederici, & Hahne, 2006; Stein et al., 2009; Wartenburger et al., 2003). For example, a number of studies have found that bilinguals with higher second‐language proficiency showed more native‐like brain processing mechanisms when reading words in second language, compared to second‐language learners with lower proficiency (Cao et al., 2013; Gao et al., 2017; Stein et al., 2009).
Although much is known about how words in native and second languages are represented in the brain, previous studies mainly used traditional univariate activation analysis, which determines the degree of similarity in neural activation qualitatively based on the co‐activation or activation differences between native and second languages. Such an approach has missed rich representational space information (Kriegeskorte & Kievit, 2013). For example, a recent study found that word reading in native and second languages showed activations in common regions, but different activation patterns in the same regions when multi‐voxel pattern analysis was conducted (Xu, Baldauf, Chang, Desimone, & Tan, 2017). Therefore, multivariate approaches (e.g., representational similarity analysis, RSA) (Kriegeskorte, Mur, & Bandettini, 2008) are needed to quantify neural pattern similarity between native and nonnative languages (e.g., second and third languages). Moreover, it is unclear how non‐native‐language experience modulates neural pattern similarity between native and nonnative languages.
The modulatory effect of language experience has typically been studied by comparing two groups of bilinguals with different levels of proficiency in second language (Cao et al., 2013; Gao et al., 2017; S. Y. Kim, Liu, & Cao, 2017; Wartenburger et al., 2003). Such comparisons are likely to be confounded by factors such as learning methods (different methods might have been used to attain different levels of proficiency) and individual differences (different levels of proficiency might have resulted from cognitive differences). To control for those confounding factors, this study used a well‐designed artificial language paradigm, in which all participants were instructed to learn words in a new language using the same procedure for the same amount of time. This paradigm allows us to examine the changes in cross‐language pattern similarity as a result of learning the new language.
Specifically, we trained 24 Chinese college students to learn an artificial language, which was created by borrowing visual forms and phonologies of 30 Korean Hangul characters. These artificial language words were then assigned arbitrary meanings using 30 pictures of objects that belong to two semantic categories (i.e., man‐made and natural objects). The same two semantic categories were used for Chinese and English words in this study. The training lasted for 12 days. After 4‐ and 12‐day training, participants were scanned while performing a semantic decision task, which has been widely used in reading research (Daselaar et al., 2002; Guo & Burgund, 2010; Hulten, Vihla, Laine, & Salmelin, 2009; Whatmough, Verret, Fung, & Chertkow, 2004; Wu et al., 2009), especially when studying neural representations of native and second languages in bilinguals (Ding et al., 2003; Illes et al., 1999; Xue et al., 2004).
The modulatory effect of proficiency in the new language was examined by comparing pattern similarity between Chinese and artificial language across the two learning sessions [i.e., after 4‐day training (lower proficiency) and after 12‐day training (higher proficiency)]. In this analysis, pattern similarity between Chinese and English words was included to control potential time‐related variances across the two learning sessions. In addition, we also examined the modulatory effect of language proficiency by comparing pattern similarity between Chinese and English (higher proficiency) with that between Chinese and artificial language (lower proficiency). Both whole‐brain RSA and ROI‐based RSA were used. Whole‐brain RSA was used to detect all potential brain regions showing the modulatory effect of language experience on cross‐language pattern similarity, while ROI‐based RSA was used to further confirm whether the effect of language experience occurred in brain regions critically involved in word reading, including the bilateral inferior frontal gyrus (i.e., pars opercularis and pars triangularis), inferior temporal gyrus, fusiform gyrus, and angular gyrus (Binder, Desai, Graves, & Conant, 2009; Borghesani et al., 2016; Dehaene & Cohen, 2011; Fischer‐Baum, Bruggemann, Gallego, Li, & Tamez, 2017; Liuzzi et al., 2017; Taylor, Rastle, & Davis, 2013). We expected that higher proficiency in the new language (as well as in English as second language) was associated with higher cross‐language pattern similarity within brain regions for word reading.
2. MATERIALS AND METHODS
2.1. Participants
Twenty‐four college students (11 males, mean age = 19.46, SD = 0.93, range = 18–22) participated in the study. All participants were native Chinese speakers and had learned English as second language for at least 10 years. Their proficiency in the two languages was self‐evaluated on a 7‐point scale (1 = “quite poor,” 7 = “highly proficient”). The average ratings were 5.5 (SD = 1.06) for Chinese and 3.47 (SD = 0.69) for English. Thus, participants were unbalanced Chinese–English bilinguals with intermediate proficiency in English.
None of participants had prior experience of Korean language. All participants were strongly right‐handed, as assessed by Snyder and Harris's handedness inventory (Snyder & Harris, 1993). They had normal or corrected‐to‐normal vision. None of the participants had a history of head trauma and neurological or psychiatric illness. Each participant gave a written informed consent before the experiment. The study was approved by the IRB of School of Psychology at South China Normal University.
2.2. Materials
Thirty Chinese words, 30 English words, and 30 artificial language words were used in this study. For each type of words, half of the stimuli represented man‐made objects, and the other half represented natural objects. The Chinese words were selected from a Chinese database (Cai & Brysbaert, 2010). They were medium‐ to high‐frequency (mean = 56.83 per million, SD = 49.29), and consisted of 2–3 units (mean = 3, SD = 0.69) and 6–9 strokes (mean = 7, SD = 1.23).
The English materials were selected from the MRC Psycholinguistic Database (http://websites.psychology.uwa.edu.au/school/MRCDatabase/uwa_mrc.htm). These words were also medium‐ to high‐frequency words (mean = 54.63 per million, SD = 33.53), and contained 3–6 letters (mean = 4.5, SD = 1.11). Before the main experiment, 10 Chinese college students were instructed to evaluate the familiarity of all English words on a 5‐point scale. The scores were higher than 4 (4.6 ± 0.24), suggesting Chinese participants were familiar with those English words.
The artificial language was created by adopting the visual forms and sounds of 30 Korean Hangul characters. They were assigned arbitrary meanings through pictures of 30 different objects (half are natural objects and the other half are man‐made objects) (Figure 1a). These words were composed of 2–3 units and 5–9 strokes to match with Chinese words in visual complexity. The sounds of the artificial language words were in the same length (600 ms) and loudness.
Figure 1.
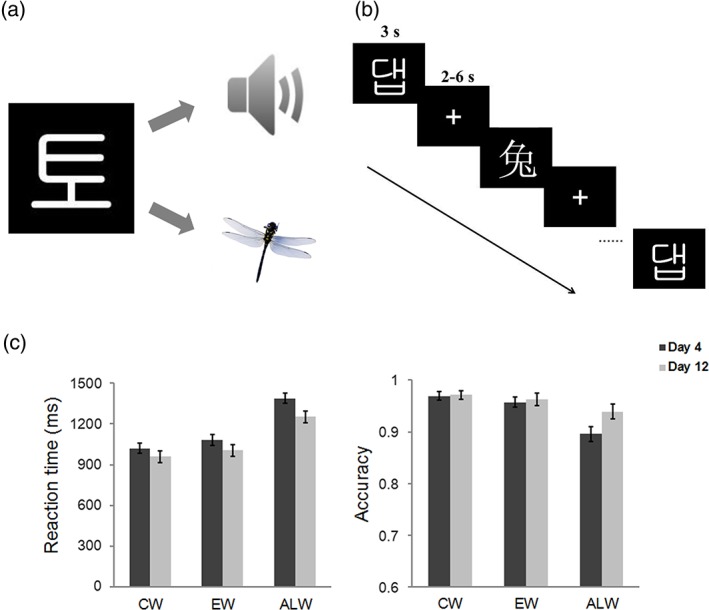
Experimental design and behavioral performance. Participants received artificial language training (a) for 12 days (1 hr per day). (b) A semantic decision task was administered after 4‐day training and after 12‐day training. (c) Reaction time and accuracy of artificial language words increased with training. Error bars represent the standard error of the mean [Color figure can be viewed at http://wileyonlinelibrary.com]
2.3. Training procedure
In this study, we trained participants to learn the visual forms, sounds, and semantics of the artificial language for 12 days, about 1 hr per day. Several learning tasks were used to facilitate the acquisition of the artificial language by using a computerized program. The tasks included character learning, phonological choice, semantic choice, free learning, naming with feedback, fast matching, and fast naming.
The first five tasks were used over the 12‐day training. Specifically, participants had to learn and associate each artificial language visual word with its sound and meaning in the character learning task, and were asked to choose the correct sound/meaning out of four to match the target word in the phonological/semantic choice task. The free learning task was designed to relearn any words that participants had difficulties in the phonological/semantic choice tasks. During naming with feedback task, participants were asked to read a word aloud followed by its correct pronunciation. After Day 4, two speed tasks were additionally designed to facilitate the efficient access to the phonologies and semantics of artificial languages words. In the fast matching task, participants were asked to match 10 visual words with 10 pictures as fast and accurately as possible. The fast naming task required participants to read 10 words or name 10 pictures as fast and accurately as possible. It should be noted that all training tasks were designed to facilitate the acquisition of visual forms, phonologies, semantics, and their associations. Although the meanings of artificial language words consisted of two categories of objects (i.e., man‐made and natural objects), participants were not explicitly instructed to categorize the words based on their meanings during training. In other words, artificial language training in this study focused on lexical learning, but not category learning.
2.4. fMRI task
Participants were scanned twice, one after the 4‐day training and another after the 12‐day training. Day 4 was chosen for the first fMRI scan based on the behavioral performance of another small sample (3 Chinese college students) in a preliminary experiment (Supporting Information Figure S1). Specifically, on Day 4, the learning curves became smooth and participants mastered most of the artificial language words (the mean accuracy was 84.4% for the word naming task). The relatively high behavioral performance during the first scan would minimize the possibility that the differences in pattern similarity across the two learning sessions reflected the differences between known and unknown words, rather than the effect of language proficiency.
During scanning, participants performed a semantic decision task. They were instructed to judge whether the meaning of the presented word was a man‐made object or a natural object (Figure 1b). In the semantic decision task, three types of stimuli (i.e., Chinese, English, and artificial language words) were included. Each type of stimuli consisted of 30 items and each item was presented for three times.
A rapid event‐related design was used for the fMRI scan. The stimuli were pseudorandomly presented, and the sequences of the trials were optimized with OPTSEQ2 (http://surfer.nmr.mgh.harvard.edu/optseq/). There were two functional runs in each scan. Each run began with a 6 s blank screen. Each word was presented for 3 s, followed by a fixation varying randomly from 2 to 6 s (mean = 3 s) to improve design efficiency (Dale, 1999). In total, each run contained 135 trials and lasted for 816 s.
2.5. MRI data acquisition
All MRI images were collected using a 3.0 T Siemens MRI scanner in the MRI Center at South China Normal University. Functional images were acquired with a single‐shot T2*‐weighted gradient‐echo EPI sequence. The specific scanning parameters were: TR = 2,000 ms, TE = 25 ms, flip angle = 90°, FOV = 192 × 192 mm, matrix size = 64 × 64, and slice thickness = 3.5 mm. Thirty‐five contiguous axial slices parallel to the anterior commissure‐posterior commissure plane were obtained to cover the whole brain. A T1‐weighted, three‐dimensional, gradient‐echo pulse‐sequence was used for anatomical MRI with the following parameters: TR = 2,300 ms, TE = 3.24 ms, flip angle = 9°, FOV = 256 × 256 mm, matrix size = 256 × 256, and slice thickness = 1 mm. One hundred and seventy‐six sagittal slices were acquired to provide a high‐resolution structural image of the whole brain.
2.6. Image preprocessing and statistical analysis
Image processing was carried out using the FEAT (FMRI Expert Analysis Tool) Version 6.00, part of FSL (FMRIB's Software Library, http://www.fmrib.ox.ac.uk/fsl). The first three volumes in each time series were discarded to remove nonequilibrium effects of T1. The remaining images were then realigned, and no translational movement parameters exceeded 1 voxel in any direction for any participant or run. All imaging data were spatially smoothed using a Gaussian kernel of 5 mm full‐width‐half‐maximum (FWHM) and then temporally filtered by using a nonlinear high‐pass filter with a 60 s cutoff. The functional images were registered to standard Montreal Neurological Institute (MNI) space using a two‐step registration from functional to the MPRAGE structural image to MNI‐template (Jenkinson & Smith, 2001). Registration from MPRAGE structural image to the standard MNI space was further refined using FNIRT nonlinear registration (Andersson, Jenkinson, & Smith, 2007a, 2007b).
At the first level, the data were modeled using the general linear models for each participant, for each learning session, and for each run. The onsets and durations (3 s) of the events were convolved with the double‐gamma hemodynamic response function to generate the regressors used in the general linear model. To improve statistical sensitivity, the six motion parameters and temporal derivatives were included as covariates of no interest. Fixations served as the baseline condition. The contrast images for the three conditions (i.e., Chinese words, English words, and artificial language words) were computed separately for each run, learning session, and participant.
A second‐level analysis was performed on the four runs in the two scans to get the averaged activation in each scan by using a fixed‐effects model. These data were then entered into the third‐level analyses. In these analyses, we firstly computed the group activation for Chinese, English, and artificial language words after the 12 days training, and performed a conjunction analysis to reveal common activations across the three types of words, using the procedure suggested by Nichols, Brett, Andersson, Wager, and Poline (2005). Specifically, group activation maps for the three types of words were thresholded individually at p < .001, and then binarized and multiplied to reveal brain regions that were significantly activated for all three conditions. Then, we performed a two‐way (language: Chinese, English, and artificial language; learning session: Day 4 and Day 12) analysis of variance (ANOVA) to examine the training effect. Follow‐up simple‐effect analysis was used to compute the differences across the three languages, and differences between Day 4 and Day 12 for each language. A random‐effects model with FLAME stage 1 only was used in the third‐level analysis (Beckmann, Jenkinson, & Smith, 2003; M. Woolrich, 2008; Woolrich, Behrens, Beckmann, Jenkinson, & Smith, 2004). Unless otherwise stated, all reported group images were thresholded with a voxel‐level threshold of p < .001 and a cluster probability of p < .05, corrected for whole‐brain multiple comparisons using the Gaussian random field theory (Worsley, 2001).
2.7. Representational similarity analysis
To examine the effect of language experience on the cross‐language pattern similarity, we further computed neural pattern similarity using both whole‐brain representation similarity analysis (RSA) and ROI‐based RSA. In both analyses, we first re‐estimated the above mentioned first‐level models with unsmoothed data.
In whole‐brain RSA, a searchlight method was used to locate brain regions (Kriegeskorte et al., 2008; Xue et al., 2013). At each voxel, activation patterns were extracted from the spherical ROI with a radius of 4 mm centered on that voxel (Kriegeskorte, Goebel, & Bandettini, 2006), separately for Chinese, English, and artificial language words. Pearson correlation analysis was used to compute cross‐language pattern similarity (i.e., pattern similarity between Chinese and English words, and that between Chinese and artificial language words). These correlation coefficients were then transformed into Fisher's z‐scores. The searchlight method was conducted at participants' native space, separately for each run. The results were then transformed into standard space and concatenated across runs. The brain maps of individual participants were spatially smoothed at 5 mm FWHM and were then subjected to random‐effect group analysis. To examine the validity of representational similarity analysis, we also computed within‐language and between‐language pattern similarity by correlating activation patterns across the two runs. Within‐language pattern similarity was calculated by averaging pattern similarity of all within‐language pairs and between‐language pattern similarity was calculated by averaging pattern similarity of all between‐language pairs.
To confirm that the modulatory effect of the proficiency of nonnative languages occurred in brain regions critically involved in word reading, we further performed ROI‐based RSA. In this analysis, 10 brain regions that have been repeatedly reported to be involved in word reading were defined as regions of interest (ROIs). They included four regions in the inferior frontal gyrus [the bilateral pars opercularis (PO) and bilateral pars triangularis (PT)], four regions in the occipitotemporal areas [the bilateral fusiform gyrus (FG) and inferior temporal gyrus (ITG)], and two regions in the temporoparietal cortex [bilateral angular gyrus (AG)] (Binder et al., 2009; Bolger, Perfetti, & Schneider, 2005; Dehaene & Cohen, 2011; Mei et al., 2010; Price, 2012; Taylor et al., 2013). Anatomical ROIs were defined based on Harvard‐Oxford probabilistic atlas (Maximal Probability Threshold: 25%) within FSL. Within each ROI, we extracted averaged voxel‐wise activation pattern across all stimuli in each condition and in each run for each participant. Then, we used Pearson correlation analysis to compute cross‐language pattern similarity across the three types of words on the averaged activation patterns across stimuli (Qu et al., 2017; Xue et al., 2010). All correlation coefficients were transformed into Fisher's z‐scores and averaged across the two runs in each scan.
3. RESULTS
3.1. Training improved behavioral performance on artificial language words
Behavioral results showed that participants correctly responded to 89.6% and 93.9% artificial language words after 4‐day and 12‐day training, respectively, suggesting our training was effective. We further conducted two‐way repeated‐measure ANOVAs to examine the behavioral differences across the three types of materials and across the two learning sessions. For reaction time, the main effect of material was significant [F(2, 46) = 364.79, p < .001]. Post hoc comparisons revealed that the reaction time for Chinese words was shorter than that for English words, which was in turn shorter than the reaction time for artificial language words (Figure 1c). The main effect of learning session was also significant [F(1, 23) = 31.18, p < .001]. Specifically, the reaction time significantly decreased from Day 4 to Day 12. Furthermore, the reaction time for artificial language words decreased more than that for Chinese words and English words [material‐by‐learning‐session interaction: F(2, 46) = 7.44, p = .002], because of extensive training on artificial language words.
Similar results were found for accuracy. The main effect of material was significant [F(2, 46) = 21.67, p < .001]. The accuracy of artificial language words was lower than that of Chinese words and English words (ps < .05), but there was no difference between Chinese words and English words (Figure 1c). In addition, the material‐by‐learning‐session interaction was significant [F(2, 46) = 7.62, p = .001]. Simple effect analysis revealed that the accuracy of artificial language words was lower on Day 4 than Day 12 [F(1, 23) = 11.14, p = .003], but there were no differences across the two learning sessions for Chinese and English words [Chinese words: F(1, 23) = 0.03, p = .864; English words: F(1, 23) = 0.25, p = .622]. These results suggest that training improved proficiency in artificial language word reading, and that the level of proficiency was lower in artificial language relative to native language (i.e., Chinese) and second language (i.e., English).
3.2. Neural activations for Chinese, English, and artificial language words across the two learning sessions
We first conducted the whole‐brain analysis to explore neural activations of Chinese, English, and artificial language words after the 12‐day training. Results showed that the three types of words elicited similar activation patterns in brain regions for general cognitive control (i.e., anterior cingulate cortex and bilateral middle frontal gyrus) (Abutalebi & Green, 2007; Seo, Stocco, & Prat, 2018; Sierpowska et al., 2018) and for word reading (i.e., the bilateral inferior frontal gyrus, occipitoparietal cortex, and occipitotemporal cortex) (Bolger et al., 2005; Cattinelli, Borghese, Gallucci, & Paulesu, 2013; Price, 2012) (Figure 2a–c). The common activations in those regions were confirmed by a conjunction analysis (Figure 2d).
Figure 2.
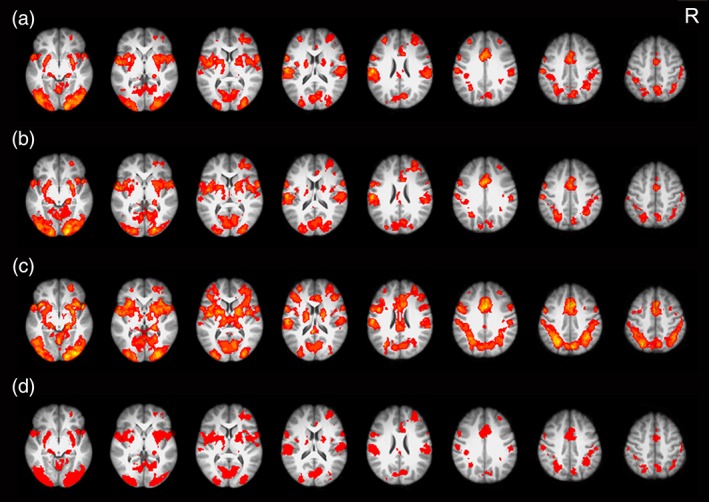
Brain activations for (a) Chinese words, (b) English words, and (c) artificial language words after 12‐day training. (d) Conjunction analysis showed that the three types of words elicited similar activation in the typical reading network. All activations were thresholded at p < .001 (whole‐brain corrected) and overlaid onto the group‐averaged anatomical map. R = right [Color figure can be viewed at http://wileyonlinelibrary.com]
We further conducted a language‐by‐learning‐session ANOVA on the imaging data to examine the training effect and the language effect (Supporting Information Figure S2). Results showed that the main effect of language was significant in the bilateral cingulate gyrus, prefrontal cortex, occipitoparietal cortex, occipitotemporal cortex, and angular gyrus. Post hoc comparisons revealed that artificial language words elicited greater activation in the bilateral cingulate gyrus, inferior frontal gyrus, middle frontal gyrus, superior parietal lobule, inferior temporal gyrus than did Chinese words and English words, whereas Chinese and English words elicited more activation in the bilateral medial frontal cortex and angular gyrus. English words elicited greater activation in bilateral occipital cortex than did Chinese words, whereas Chinese words elicited greater activation in bilateral fusiform gyrus than did English words (Supporting Information Figure S3). These results suggest that more cognitive resources were required to process the newly acquired artificial language words relative to words in familiar languages. Neither the main effect of learning session nor the language‐by‐learning‐session interaction was significant in any regions.
To detect the subtle effects of training, we directly compared the neural activations after 4‐day training with those after 12‐day training separately for the three types of words. Results showed that compared with Day 4, artificial language words elicited less activations in the bilateral occipitotemporal cortex and occipitoparietal cortex in Day 12. No regions showed significant changes in activation for Chinese words and English words (Supporting Information Figure S4).
3.3. Artificial language training enhances pattern similarity between native language and artificial language
Before examining the effect of language training on pattern similarity, we first needed to establish validity of RSA in our study by comparing within‐language pattern similarity with between‐language pattern similarity across the two runs. For both Days 4 and 12, whole‐brain RSA showed that within‐language pattern similarity was higher than between‐language pattern similarity in a wide neural network, including the anterior cingulate cortex, bilateral prefrontal gyrus, occipitotemporal areas, and occipitoparietal cortex (Supporting Information Figure S5). No regions showed higher between‐language pattern similarity than within‐language pattern similarity. These results suggest that the representational similarity analysis used in this study had good validity.
To examine the effect of the proficiency in nonnative languages on cross‐language neural pattern similarity, we performed both whole‐brain and ROI‐based representational similarity analysis (RSA). We first compared pattern similarity between Chinese and artificial language words (C–A) on Day 4 with that on Day 12. Results showed that artificial language training enhanced C–A pattern similarity in several regions, including the bilateral orbital frontal cortex, superior occipital gyrus, right middle frontal gyrus, and superior parietal lobule (Figure 3a). After subtracting the pattern similarity between Chinese and English words (C–E), the bilateral superior occipital gyrus remained significant. The bilateral orbital frontal cortex was additionally activated when a liberal threshold was used (p < .01, whole‐brain corrected). In addition, we compared neural pattern similarity of C‐E (higher proficiency) with that of C–A (lower proficiency) after 12‐day training. Results showed that pattern similarity of C–E was higher than that of C–A in the anterior cingulate cortex, bilateral inferior frontal gyrus (extending to precentral gyrus), left superior parietal lobule, and right superior occipital gyrus (Figure 3b).
Figure 3.
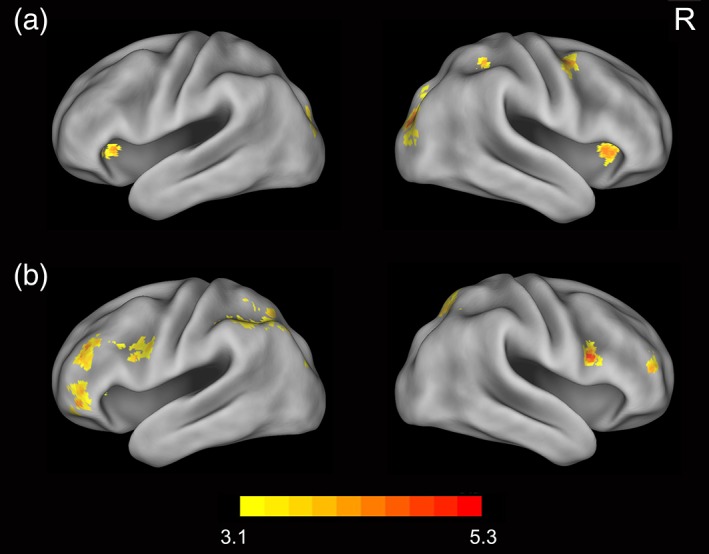
The effect of language experience on cross‐language pattern similarity. (a) Results showed that artificial language training enhanced pattern similarity between Chinese words and artificial language words in several regions, including the bilateral orbital frontal cortex, superior occipital gyrus, right middle frontal gyrus, and superior parietal lobule. (b) Pattern similarity between Chinese and English words was higher than that between Chinese and artificial language words in the anterior cingulate cortex, bilateral inferior frontal gyrus (extending to precentral gyrus), left superior parietal lobule, and right superior occipital gyrus. R = right [Color figure can be viewed at http://wileyonlinelibrary.com]
To ensure that language experience specifically enhanced cross‐language pattern similarity within brain regions for word reading, we further conducted ROI‐based RSA. As described in Methods, 10 brain regions for word reading were defined as ROIs. In this analysis, we performed two‐way (comparison: C–E and C–A; learning session: Day 4 and Day 12) repeated‐measures ANOVA to directly compare pattern similarity of C–E with that of C–A, and compare pattern similarity of C–A across the two learning sessions. As shown in Figure 4 and Table 1, the main effect of comparison was significant in eight ROIs, including the left pars opercularis [F(1, 23) = 31.21, p < .001], right pars opercularis [F(1, 23) = 42.23, p < .001], left pars triangularis [F(1, 23) = 23.51, p < .001], right pars triangularis [F(1, 23) = 18.89, p < .001], left inferior temporal gyrus [F(1, 23) = 37.50, p < .001], right inferior temporal gyrus [F(1, 23) = 14.64, p = .001], and left angular gyrus [F(1, 23) = 36.71, p < .001], right angular gyrus [F(1, 23) = 13.92, p = .001]. Specifically, pattern similarity of C–E was higher than that of C–A in those regions for word reading. A significant main effect of learning session was found only in the inferior temporal gyrus [F(1, 23) = 6.33, p = .019].
Figure 4.
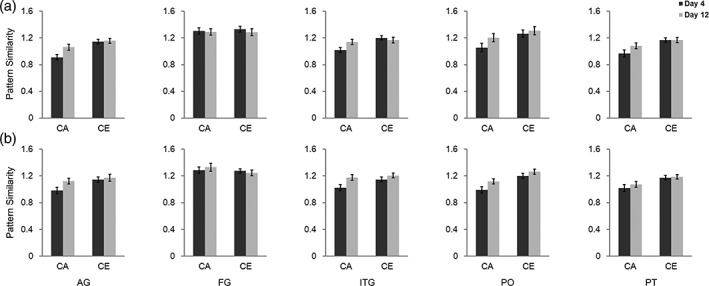
Cross‐language pattern similarity in the 10 predefined ROIs. (a) The upper panels show results of 5 ROIs in the left hemisphere, (b) while the lower panels show results in the right hemisphere. AG = angular gyrus; FG = fusiform gyrus; ITG = inferior temporal gyrus; PO = pars opercularis; and PT = pars triangularis. CA = pattern similarity between Chinese and artificial language words; CE = pattern similarity between Chinese and English words
Table 1.
Summary statistics of the comparison‐by‐learning‐session ANOVA
| Interaction | Main effect of comparison | Main effect of learning session | ||||
|---|---|---|---|---|---|---|
| ROIs | F | P | F | P | F | P |
| Left | ||||||
| Angular gyrus | 9.01 | .006** | 36.71 | <.001*** | 3.72 | .066 |
| Fusiform gyrus | 0.94 | .342 | 0.26 | .616 | 0.48 | .493 |
| Inferior temporal gyrus | 15.51 | .001** | 37.50 | <.001*** | 1.19 | .287 |
| Pars opercularis | 4.95 | .036* | 31.21 | <.001*** | 2.89 | .103 |
| Pars triangularis | 9.29 | .006** | 23.51 | <.001*** | 1.24 | .276 |
| Right | ||||||
| Angular gyrus | 6.37 | .019* | 13.92 | .001** | 2.18 | .153 |
| Fusiform gyrus | 5.10 | .034* | 3.16 | .089 | 0.03 | .860 |
| Inferior temporal gyrus | 4.69 | .041* | 14.64 | .001** | 6.33 | .019* |
| Pars opercularis | 2.32 | .141 | 42.23 | <.001*** | 3.12 | .091 |
| Pars triangularis | 0.74 | .398 | 18.89 | <.001*** | 0.70 | .410 |
p < .05;
p < .01;
p < .001.
More importantly, seven ROIs showed significant comparison‐by‐learning‐session interaction [left pars opercularis: F(1, 23) = 4.95, p = .036; left pars triangularis: F(1, 23) = 9.29, p = .006; right fusiform gyrus: F(1, 23) = 5.1, p = .034; left inferior temporal gyrus: F(1, 23) = 15.51, p = .001; right inferior temporal gyrus: F(1, 23) = 4.69, p = .041; and left angular gyrus: F(1, 23) = 9.01, p = .006; right angular gyrus: F(1, 23) = 6.37, p = .019]. Simple‐effect analysis revealed that pattern similarity of C–A increased accompanying training in the left pars opercularis [F(1, 23) = 4.94, p = .036], left inferior temporal gyrus [F(1, 23) = 6.48, p = .018], right inferior temporal gyrus [F(1, 23) = 8.55, p = .008], left angular gyrus [F(1, 23) = 9.26, p = .006], and right angular gyrus [F(1, 23) = 6.13, p = .021], whereas that of C–E did not change between the two training sessions (Supporting Information Table S1). These results provide direct evidence for the modulatory effect of nonnative‐language experience on pattern similarity between native and nonnative languages in brain regions for word reading.
In the above analysis, neural responses across the three presentations for each word were averaged to get higher signal‐to‐noise ratio of BOLD signals. To examine whether the improvement of cross‐language pattern similarity with training was consistent across the three presentations, we further computed pattern similarity of C–A in Day 4 and in Day 12, separately for the three presentations. Repetition‐by‐learning‐session ANOVA showed that the main effect of learning session (i.e., the enhancement of pattern similarity) was less robust, but was still evident in the left pars opercularis, angular gyrus, and bilateral inferior temporal gyrus (Supporting Information Table S2 and Figure S6). Although pattern similarity of C–A decreased within increasing repetition in 6 of 10 ROIs, none of ROIs showed significant repetition‐by‐learning‐session interaction (all ps > .1). These results suggest that the enhancement of cross‐language pattern similarity with training was consistent across the three presentations.
3.4. Changes in pattern similarity during training were associated with improvement in behavioral performance
Finally, we conducted correlational analyses to examine the associations between neural pattern similarity and behavioral performance. Results showed that changes in pattern similarity from Day 4 to Day 12 were associated with improvement in behavioral performance. Specifically, more changes of pattern similarity between Chinese words and artificial language words in the bilateral pars triangularis, and right pars opercularis were associated with greater improvement (accuracy) for artificial language words in the semantic decision task (Figure 5). It is worth noting that there were two participants showing slight decreases in accuracy from Day 4 to Day 12 (0.08 and 0.07), perhaps because of forgetting or interference or instability in measurement. To exclude the possibility that the brain–behavior relationship was driven by those two participants, we performed additional correlational analyses by excluding data of those two participants. The correlation coefficients remained significant in all three regions (left pars triangularis: r = .65, p = .001; right pars triangularis: r = .55, p < .01; right pars opercularis: r = .56, p < .01).
Figure 5.
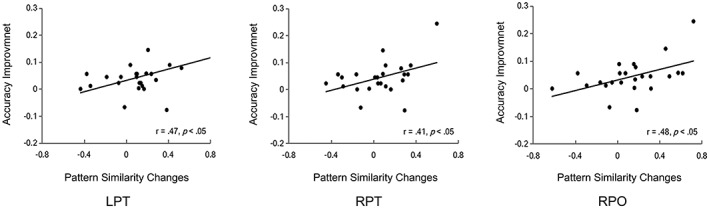
The correlations between neural pattern similarity and behavioral performance on the semantic decision task. Changes in pattern similarity between Chinese words and artificial language words in the bilateral pars triangularis, and right pars opercularis were positively associated with the improvement in accuracy of artificial language words in the semantic decision task. LPT = left pars triangularis; RPT = right pars triangularis; RPO = right pars opercularis
4. DISCUSSION
Using an artificial language training paradigm and representational similarity analysis, this study aimed to investigate whether lexical learning in a language led to neural pattern similarity with word reading in native language. Consistent with previous studies (S. Y. Kim et al., 2017; Mei et al., 2015b; Sun et al., 2015; Xue et al., 2004), this study found that different languages were generally processed in common regions, such as the anterior cingulate cortex, bilateral prefrontal cortex, occipitoparietal cortex, and occipitotemporal cortex. Furthermore, training reduced neural activations in bilateral occipitotemporal cortex for artificial language words, but not for words in native (i.e., Chinese) and second languages (i.e., English). More importantly, we found that higher proficiency in the nonnative language (i.e., second and third languages) was associated with higher pattern similarity between native and nonnative languages. These results suggest that pattern similarity between words in native and nonnative languages reflect proficiency in the latter.
Previous neuroimaging studies on bilingualism have generally revealed that common regions are activated during word reading in native and second languages (Berken et al., 2015; Chee, Tan, & Thiel, 1999; S. Y. Kim et al., 2017; Kovelman, Baker, & Petitto, 2008; Marian et al., 2007; Wong, Yin, & O'Brien, 2016; Xue et al., 2004). Further studies have observed that learners with higher second‐language proficiency showed more native‐like brain processing mechanisms (Cao, 2015; Cao et al., 2013; S. Y. Kim et al., 2016; Ojima et al., 2005; Rossi et al., 2006; Stein et al., 2009). Nevertheless, these studies mainly used the traditional univariate activation analysis to compute neural activity at the level of individual voxels, and consequently missed the information of multi‐voxel neural pattern. Here, by adopting representational similarity analysis that computes multi‐voxel pattern similarity (Kriegeskorte et al., 2008), our study quantitatively examined neural pattern similarity between native and nonnative languages. Specifically, we first conducted whole‐brain RSA to examine the modulatory effect of proficiency in nonnative languages on the cross‐language pattern similarity. Results showed that pattern similarity of C–A increased with training in the bilateral prefrontal cortex and occipitoparietal cortex. In addition, pattern similarity of C–E (higher proficiency) was higher than that of C–A (lower proficiency) in the anterior cingulate cortex, bilateral inferior frontal gyrus, left superior parietal lobule, and right superior occipital gyrus. Next, the enhancement of cross‐language pattern similarity was specifically confirmed in regions for word reading by using ROI‐based RSA. Specifically, pattern similarity of C–E was higher than that of C–A in the bilateral inferior frontal gyrus, inferior temporal gyrus, and angular gyrus. Furthermore, by comparing pattern similarity between the two learning sessions for C–A and C–E separately, we found that artificial language training enhanced pattern similarity of C–A in bilateral inferior temporal gyrus, angular gyrus, and left pars opercularis, whereas that of C–E did not change. Finally, the modulatory effect of non‐native‐language proficiency was further confirmed by the results of brain–behavior correlational analysis. We found that the changes in C–A pattern similarity in the bilateral inferior frontal gyrus were associated with improvement in behavioral performance. These results converge to suggest that proficiency in nonnative languages enhances neural pattern similarity between native and nonnative languages.
It is worth noting that although the increased cross‐language pattern similarity was consistently observed in both whole‐brain and ROI‐based RSA, we did not find increased activations induced by training in any brain regions. On the contrary, univariate activation analysis showed that training reduced activations for artificial language words in the bilateral occipitotemporal and occipitoparietal cortex, probably because of increased familiarity with artificial language words (Ischebeck et al., 2004; Kronbichler et al., 2007; Twomey et al., 2013). Those inconsistent results between univariate and multivariate analyses are likely a reflection of the advantages of RSA over univariate activation analysis. Specifically, the univariate activation analysis typically uses spatially smoothed images and treats each voxel independently, and consequently loses fine‐grained pattern information (Haxby, 2012; Haynes, 2015), while multivariate methods (e.g., RSA) compute patterns of neural activity across multiple voxels, which are thought to reflect neuronal population codes (Mur, Bandettini, & Kriegeskorte, 2009). The multivariate methods are sensitive to voxel‐level variance within participants (Davis et al., 2014), and are able to detect fine‐grained pattern differences even if there are no regional‐average differences (Mur et al., 2009). Consistent with this view, previous studies have revealed that multi‐voxel patterns differ across conditions without differences in mean activation in several cognitive domains such as cognitive control (Esterman, Chiu, Tamber‐Rosenau, & Yantis, 2009; Tamber‐Rosenau, Esterman, Chiu, & Yantis, 2011) and language (Xu et al., 2017).
Our results of increased pattern similarity accompanying the lexical learning in a new language are consistent with previous findings of cross‐language interactions during word reading in bilinguals. Previous studies have revealed that native language experience can shape the neural mechanisms underlying second‐language learning and processing (Cao et al., 2013; S. Y. Kim et al., 2017; Mei et al., 2015b; Nakada et al., 2001; Nelson et al., 2009; Tan et al., 2003). Specifically, it has been revealed that Chinese speakers recruit the left middle frontal gyrus (Y. Liu et al., 2007; Tan et al., 2003) and bilateral occipitotemporal regions (Nelson et al., 2009) to process English, which is different from English speakers (Bolger et al., 2005; Price, 2012; Tan, Laird, Li, & Fox, 2005). Similarly, one recent study has found that Chinese speakers and English speakers differ in the involvement of addressed and assembled phonologies in learning a new language (Mei et al., 2015b). Those findings suggest that the human brain applies the neural network of native language to learn and process words in second language (Perfetti et al., 2007; Perfetti & Liu, 2005). On the other hand, learning to read a second language also affects the neural mechanisms of native language (Mei et al., 2014; Mei et al., 2015a; Parker Jones et al., 2012; Timmer, Ganushchak, Ceusters, & Schiller, 2014; Zou et al., 2012). For example, it has been reported that, compared to English speakers without experience on Chinese, English speakers with Chinese experience recruit bilateral posterior fusiform gyrus to process English words, which is similar to the way Chinese characters are processed by Chinese speakers (Mei et al., 2015a). These two lines of research suggest that native language interacts with second language during the acquisition of second language words, which leads to common neural mechanisms of words in native and nonnative languages. From this perspective, our findings of increased cross‐language pattern similarity might have resulted from the cross‐language interactions during the learning of a new language. Consistent with this view, there is evidence that proficiency in second language modulates the involvement of the native‐language neural network in the processing of second language (Cao et al., 2013).
Our results of increased pattern similarity seem to contradict with the findings of one recent study (Xu et al., 2017). Xu et al. investigated how different languages were represented in the brain of Chinese‐English bilinguals using multivariate pattern analyses (MVPA). They found that native and second languages were processed in common regions but in distinguishable neural patterns of activity. Two differences between Xu et al.’s and our study might account for the inconsistent results. First, the two studies adopted different tasks. Xu et al. used an implicit reading task (i.e., same‐different judgment task) which emphasizes perceptual processing, while our study adopted an explicit reading task (i.e., semantic decision task) which requires heavy engagement of semantic processing. One prevalent model of bilingual representation (i.e., the Revised Hierarchical Model) has proposed that the native and second languages share a common conceptual system (i.e., semantics), but their lexical forms (i.e., orthography and phonology) are represented separately (J. F. Kroll & Stewart, 1994; J. F. Kroll, van Hell, Tokowicz, & Green, 2010). Consistent with this model, neuroimaging studies have consistently observed common activations for the two languages when semantic tasks are used (Ding et al., 2003; Illes et al., 1999; Xue et al., 2004), even for low‐proficiency second‐language learners (Xue et al., 2004), but less consistent results have been reported when tasks focusing on other aspects (e.g., phonology, grammar) are used (Feng, Chen, Zhu, He, & Wang, 2015; H. Liu et al., 2010; Saur et al., 2009; Tham et al., 2005). Therefore, inconsistent results might be accounted by the task differences. Future studies should directly examine the task effect on cross‐language neural pattern similarity. Second, the two studies differed in the methods in data analysis. Xu et al.'s study used MVPA to discriminate the two languages, whereas our study used representational similarity analysis to explore the neural pattern similarity between the two languages and the modulatory effect of proficiency. In other words, Xu et al. focused on the differences between native and second languages, whereas our study focused on their similarities. Therefore, inconsistent results might be accounted by the differences in our foci and associated data analytical strategies.
Given that the fMRI task (i.e., semantic decision task) required participants to categorize the artificial language words based on their meanings, it is possible that the enhancement of cross‐language pattern similarity reflects the effect of category learning instead of language proficiency. Previous studies have revealed that category learning shapes the pattern similarity across stimuli. Specifically, category learning leads to more similar responses to stimuli from the same category and more dissimilar responses to stimuli from different categories (De Baene, Ons, Wagemans, & Vogels, 2008; Sigala & Logothetis, 2002). Furthermore, there is evidence that patterns of representational similarity become increasingly similar across different categorization tasks (i.e., rule‐based and information‐integration categorization tasks) after extensive training (Soto, Waldschmidt, Helie, & Ashby, 2013). Our results of the increased cross‐language pattern similarity were not likely to reflect the category‐learning effect for two reasons. First, all learning tasks used in this study focused on lexical learning without any explicit requirement of categorization. The semantic decision task requiring categorization was only performed twice for the two fMRI scans separated by 8 days. Second, both pattern similarity of C–A and that of C–E would increase if limited practice on categorization did affect the cross‐language pattern similarity. Contrary to this expectation, we only observed the enhancement of pattern similarity for C–A, but not for C–E.
There are two limitations that should be addressed in the future. First, unlike natural language, the artificial language used in this study was limited in its vocabulary size and morphology, and it lacked syntax. Such differences might limit the generalization of our findings to natural languages to some extent, although researchers have found high positive correlations between natural language learning and artificial language learning (Ettlinger, Morgan‐Short, Faretta‐Stutenberg, & Wong, 2016). Therefore, future research should test the modulatory effect of second‐language proficiency on the cross‐language pattern similarity in the course of natural language acquisition. Second, in addition to proficiency level, age of acquisition (AOA) may also affect the cross‐language pattern similarity. For example, using univariate activation analyses, previous neuroimaging studies have revealed that, compared with late bilinguals, early bilinguals showed more similar activation patterns for the two languages (Berken et al., 2015; K. H. Kim, Relkin, Lee, & Hirsch, 1997; Wartenburger et al., 2003). Participants in this study were all college students with small variations in age, and consequently precluded us from examining the AOA effect. Future studies should investigate the AOA effect by comparing the cross‐language pattern similarity in early bilinguals with that in late bilinguals.
5. CONCLUSION
In conclusion, using an artificial language training paradigm and representational similarity analysis, this study revealed that higher proficiency in nonnative languages was associated with higher pattern similarity between native and nonnative languages.
Supporting information
Figure S1. The reaction times and accuracies of 3 Chinese college students in word naming task during training in a preliminary experiment. D = Day.
Figure S2. Brain maps from language‐by‐learning‐session ANOVA. Results showed significant language main effects (a) in the bilateral cingulate gyrus, prefrontal cortex, occipitoparietal cortex, occipitotemporal cortex, and angular gyrus. Neither the main effect of learning sessions (b) nor the language‐by‐learning‐session interaction (c) was significant in any regions. R = right. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S3. Neural activation differences across the three types of words. CW = Chinese words; EW = English words; ALW = artificial language words. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S4. Neural activation differences between Day 4 and Day 12 for (a) Chinese, (b) English, and (c) artificial language words. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S5. The differences between within‐language and between‐language pattern similarity in the two scans. Within‐language pattern similarity was higher than between‐language pattern similarity in a wide neural network both (a) after 4‐day training and (b) after 12‐day training. No regions were found in the reverse contrast. R = right.
Figure S6. Pattern similarity between Chinese and artificial language words shown separately for the three presentations in the 10 predefined ROIs. The upper and lower panels show results (a) in the left hemisphere and (b) those in the right hemisphere, respectively. AG = angular gyrus; FG = fusiform gyrus; ITG = inferior temporal gyrus; PO = pars opercularis; and PT = pars triangularis. R1 = first presentation; R2 = second presentation; R3 = third presentation.
Table S1. Summary statistics of the comparison‐by‐learning‐session ANOVA.
Table S2. Summary statistics of the repetition‐by‐learning‐session ANOVA for C‐A.
ACKNOWLEDGMENT
This work was supported by the National Natural Science Foundation of China (numbers 31771199, 31400867), the Foundation for innovation team in Guangdong Higher Education (2017WCXTD002), and the Humanities and Social Science Research Base Project from the Ministry of Education of China (16JJD88025).
Li H, Qu J, Chen C, et al. Lexical learning in a new language leads to neural pattern similarity with word reading in native language. Hum Brain Mapp. 2019;40:98–109. 10.1002/hbm.24357
Funding information National Natural Science Foundation of China, Grant/Award Numbers: 31771199, 31400867; Foundation for innovation team in Guangdong Higher Education, Grant/Award Number: 2017WCXTD002; Humanities and Social Science Research Base Project from the Ministry of Education of China, Grant/Award Number: 16JJD88025
REFERENCES
- Abutalebi, J. , & Green, D. (2007). Bilingual language production: The neurocognition of language representation and control. Journal of Neurolinguistics, 20(3), 242–275. [Google Scholar]
- Andersson, J. L. R. , Jenkinson, M. , & Smith, S. (2007a). Non‐linear optimisation. FMRIB Technial Report TR07JA1. Oxford(UK): FMRIB Centre. [Google Scholar]
- Andersson, J. L. R. , Jenkinson, M. , & Smith, S. (2007b). Non‐linear registration. aka Spatial normalisation FMRIB Technial Report TR07JA2. FMRIB Analysis Group of the University of Oxford.
- Beckmann, C. F. , Jenkinson, M. , & Smith, S. M. (2003). General multilevel linear modeling for group analysis in FMRI. NeuroImage, 20(2), 1052–1063. [DOI] [PubMed] [Google Scholar]
- Berken, J. A. , Gracco, V. L. , Chen, J. K. , Soles, J. , Watkins, K. E. , Baum, S. , … Klein, D. (2015). Neural activation in speech production and reading aloud in native and non‐native languages. NeuroImage, 112, 208–217. [DOI] [PubMed] [Google Scholar]
- Binder, J. R. , Desai, R. H. , Graves, W. W. , & Conant, L. L. (2009). Where is the semantic system? A critical review and meta‐analysis of 120 functional neuroimaging studies. Cerebral Cortex, 19(12), 2767–2796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolger, D. J. , Perfetti, C. A. , & Schneider, W. (2005). Cross‐cultural effect on the brain revisited: Universal structures plus writing system variation. Human Brain Mapping, 25(1), 92–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghesani, V. , Pedregosa, F. , Buiatti, M. , Amadon, A. , Eger, E. , & Piazza, M. (2016). Word meaning in the ventral visual path: A perceptual to conceptual gradient of semantic coding. NeuroImage, 143, 128–140. [DOI] [PubMed] [Google Scholar]
- Bowden, H. W. , Steinhauer, K. , Sanz, C. , & Ullman, M. T. (2013). Native‐like brain processing of syntax can be attained by university foreign language learners. Neuropsychologia, 51(13), 2492–2511. [DOI] [PubMed] [Google Scholar]
- Briellmann, R. S. , Saling, M. M. , Connell, A. B. , Waites, A. B. , Abbott, D. F. , & Jackson, G. D. (2004). A high‐field functional MRI study of quadri‐lingual subjects. Brain and Language, 89(3), 531–542. [DOI] [PubMed] [Google Scholar]
- Buchweitz, A. , Shinkareva, S. V. , Mason, R. A. , Mitchell, T. M. , & Just, M. A. (2012). Identifying bilingual semantic neural representations across languages. Brain and Language, 120(3), 282–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai, Q. , & Brysbaert, M. (2010). SUBTLEX‐CH: Chinese word and character frequencies based on film subtitles. PLoS One, 5(6), e10729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao, F. (2015). Neuroimaging studies of reading in bilinguals. Bilingualism: Language and Cognition, 19(04), 683–688. [Google Scholar]
- Cao, F. , Tao, R. , Liu, L. , Perfetti, C. A. , & Booth, J. R. (2013). High proficiency in a second language is characterized by greater involvement of the first language network: Evidence from Chinese learners of English. Journal of Cognitive Neuroscience, 25(10), 1649–1663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattinelli, I. , Borghese, N. A. , Gallucci, M. , & Paulesu, E. (2013). Reading the reading brain: A new meta‐analysis of functional imaging data on reading. Journal of Neurolinguistics, 26(1), 214–238. [Google Scholar]
- Chee, M. W. , Tan, E. W. , & Thiel, T. (1999). Mandarin and English single word processing studied with functional magnetic resonance imaging. The Journal of Neuroscience, 19(8), 3050–3056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dale, A. M. (1999). Optimal experimental design for event‐related fMRI. Human Brain Mapping, 8(2–3), 109–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daselaar, S. M. , Veltman, D. J. , Rombouts, S. A. , Raaijmakers, J. G. , Lazeron, R. H. , & Jonker, C. (2002). Medial temporal lobe activity during semantic classification using a flexible fMRI design. Behavioural Brain Research, 136(2), 399–404. [DOI] [PubMed] [Google Scholar]
- Davis, T. , LaRocque, K. F. , Mumford, J. A. , Norman, K. A. , Wagner, A. D. , & Poldrack, R. A. (2014). What do differences between multi‐voxel and univariate analysis mean? How subject‐, voxel‐, and trial‐level variance impact fMRI analysis. NeuroImage, 97, 271–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Baene, W. , Ons, B. , Wagemans, J. , & Vogels, R. (2008). Effects of category learning on the stimulus selectivity of macaque inferior temporal neurons. Learning & Memory, 15(9), 717–727. [DOI] [PubMed] [Google Scholar]
- Dehaene, S. , & Cohen, L. (2011). The unique role of the visual word form area in reading. Trends in Cognitive Sciences, 15(6), 254–262. [DOI] [PubMed] [Google Scholar]
- Ding, G. , Perry, C. , Peng, D. , Ma, L. , Li, D. , Xu, S. , … Yang, J. (2003). Neural mechanisms underlying semantic and orthographic processing in Chinese‐English bilinguals. NeuroReport, 14(12), 1557–1562. [DOI] [PubMed] [Google Scholar]
- Esterman, M. , Chiu, Y. C. , Tamber‐Rosenau, B. J. , & Yantis, S. (2009). Decoding cognitive control in human parietal cortex. Proceedings of the National Academy of Sciences of the United States of America, 106(42), 17974–17979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ettlinger, M. , Morgan‐Short, K. , Faretta‐Stutenberg, M. , & Wong, P. C. (2016). The relationship between artificial and second language learning. Cognitive Science, 40(4), 822–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng, G. , Chen, H. C. , Zhu, Z. , He, Y. , & Wang, S. (2015). Dynamic brain architectures in local brain activity and functional network efficiency associate with efficient reading in bilinguals. NeuroImage, 119, 103–118. [DOI] [PubMed] [Google Scholar]
- Fischer‐Baum, S. , Bruggemann, D. , Gallego, I. F. , Li, D. S. P. , & Tamez, E. R. (2017). Decoding levels of representation in reading: A representational similarity approach. Cortex, 90, 88–102. [DOI] [PubMed] [Google Scholar]
- Gao, Y. , Sun, Y. , Lu, C. , Ding, G. , Guo, T. , Malins, J. G. , … Liu, L. (2017). Dynamic spatial organization of the occipito‐temporal word form area for second language processing. Neuropsychologia, 103, 20–28. [DOI] [PubMed] [Google Scholar]
- Guo, Y. , & Burgund, E. D. (2010). Task effects in the mid‐fusiform gyrus: A comparison of orthographic, phonological, and semantic processing of Chinese characters. Brain and Language, 115(2), 113–120. [DOI] [PubMed] [Google Scholar]
- Haxby, J. V. (2012). Multivariate pattern analysis of fMRI: The early beginnings. NeuroImage, 62(2), 852–855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haynes, J. D. (2015). A primer on pattern‐based approaches to fMRI: Principles, pitfalls, and perspectives. Neuron, 87(2), 257–270. [DOI] [PubMed] [Google Scholar]
- Hulten, A. , Vihla, M. , Laine, M. , & Salmelin, R. (2009). Accessing newly learned names and meanings in the native language. Human Brain Mapping, 30(3), 976–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Illes, J. , Francis, W. S. , Desmond, J. E. , Gabrieli, J. D. , Glover, G. H. , Poldrack, R. , … Wagner, A. D. (1999). Convergent cortical representation of semantic processing in bilinguals. Brain and Language, 70(3), 347–363. [DOI] [PubMed] [Google Scholar]
- Ischebeck, A. , Indefrey, P. , Usui, N. , Hellwig, F. , Hellwig, F. , & Taira, M. (2004). Reading in a regular orthography: An fMRI study investigating the role of visual familiarity. Journal of Cognitive Neuroscience, 16(5), 727–741. [DOI] [PubMed] [Google Scholar]
- Jamal, N. I. , Piche, A. W. , Napoliello, E. M. , Perfetti, C. A. , & Eden, G. F. (2012). Neural basis of single‐word reading in Spanish‐English bilinguals. Human Brain Mapping, 33(1), 235–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson, M. , & Smith, S. (2001). A global optimisation method for robust affine registration of brain images. Medical Image Analysis, 5(2), 143–156. [DOI] [PubMed] [Google Scholar]
- Kim, K. H. , Relkin, N. R. , Lee, K. M. , & Hirsch, J. (1997). Distinct cortical areas associated with native and second languages. Nature, 388(6638), 171–174. [DOI] [PubMed] [Google Scholar]
- Kim, S. Y. , Liu, L. , & Cao, F. (2017). How does first language (L1) influence second language (L2) reading in the brain? Evidence from Korean‐English and Chinese‐English bilinguals. Brain and Language, 171, 1–13. [DOI] [PubMed] [Google Scholar]
- Kim, S. Y. , Qi, T. , Feng, X. , Ding, G. , Liu, L. , & Cao, F. (2016). How does language distance between L1 and L2 affect the L2 brain network? An fMRI study of Korean‐Chinese‐English trilinguals. NeuroImage, 129, 25–39. [DOI] [PubMed] [Google Scholar]
- Kovelman, I. , Baker, S. A. , & Petitto, L. A. (2008). Bilingual and monolingual brains compared: A functional magnetic resonance imaging investigation of syntactic processing and a possible "neural signature" of bilingualism. Journal of Cognitive Neuroscience, 20(1), 153–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegeskorte, N. , Goebel, R. , & Bandettini, P. (2006). Information‐based functional brain mapping. Proceedings of the National Academy of Sciences of the United States of America, 103(10), 3863–3868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegeskorte, N. , & Kievit, R. A. (2013). Representational geometry: Integrating cognition, computation, and the brain. Trends in Cognitive Sciences, 17(8), 401–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegeskorte, N. , Mur, M. , & Bandettini, P. (2008). Representational similarity analysis: Connecting the branches of systems neuroscience. Frontiers in Systems Neuroscience, 2, 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroll, J. F. , & Stewart, E. (1994). Category interference in translation and picture naming: Evidence for asymmetric connections between bilingual memory representations. Journal of Memory and Language, 33(2), 149–174. [Google Scholar]
- Kroll, J. F. , van Hell, J. G. , Tokowicz, N. , & Green, D. W. (2010). The revised hierarchical model: A critical review and assessment. Bilingualism (Cambridge, England), 13(3), 373–381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kronbichler, M. , Bergmann, J. , Hutzler, F. , Staffen, W. , Mair, A. , Ladurner, G. , & Wimmer, H. (2007). Taxi vs. taksi: On orthographic word recognition in the left ventral occipitotemporal cortex. Journal of Cognitive Neuroscience, 19(10), 1584–1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, H. , & Cao, F. (2016). L1 and L2 processing in the bilingual brain: A meta‐analysis of neuroimaging studies. Brain and Language, 159, 60–73. [DOI] [PubMed] [Google Scholar]
- Liu, H. , Hu, Z. , Guo, T. , & Peng, D. (2010). Speaking words in two languages with one brain: Neural overlap and dissociation. Brain Research, 1316, 75–82. [DOI] [PubMed] [Google Scholar]
- Liu, Y. , Dunlap, S. , Fiez, J. , & Perfetti, C. (2007). Evidence for neural accommodation to a writing system following learning. Human Brain Mapping, 28(11), 1223–1234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liuzzi, A. G. , Bruffaerts, R. , Peeters, R. , Adamczuk, K. , Keuleers, E. , De Deyne, S. , … Vandenberghe, R. (2017). Cross‐modal representation of spoken and written word meaning in left pars triangularis. NeuroImage, 150, 292–307. [DOI] [PubMed] [Google Scholar]
- Marian, V. , Shildkrot, Y. , Blumenfeld, H. K. , Kaushanskaya, M. , Faroqi‐Shah, Y. , & Hirsch, J. (2007). Cortical activation during word processing in late bilinguals: Similarities and differences as revealed by functional magnetic resonance imaging. Journal of Clinical and Experimental Neuropsychology, 29(3), 247–265. [DOI] [PubMed] [Google Scholar]
- Mei, L. , Xue, G. , Chen, C. , Xue, F. , Zhang, M. , & Dong, Q. (2010). The "visual word form area" is involved in successful memory encoding of both words and faces. NeuroImage, 52(1), 371–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mei, L. , Xue, G. , Lu, Z. L. , Chen, C. , Wei, M. , He, Q. , & Dong, Q. (2015a). Long‐term experience with Chinese language shapes the fusiform asymmetry of English reading. NeuroImage, 110, 3–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mei, L. , Xue, G. , Lu, Z. L. , Chen, C. , Zhang, M. , He, Q. , … Dong, Q. (2014). Learning to read words in a new language shapes the neural organization of the prior languages. Neuropsychologia, 65, 156–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mei, L. , Xue, G. , Lu, Z. L. , He, Q. , Wei, M. , Zhang, M. , … Chen, C. (2015b). Native language experience shapes neural basis of addressed and assembled phonologies. NeuroImage, 114, 38–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mur, M. , Bandettini, P. A. , & Kriegeskorte, N. (2009). Revealing representational content with pattern‐information fMRI‐‐an introductory guide. Social Cognitive and Affective Neuroscience, 4(1), 101–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakada, T. , Fujii, Y. , & Kwee, I. L. (2001). Brain strategies for reading in the second language are determined by the first language. Neuroscience Research, 40(4), 351–358. [DOI] [PubMed] [Google Scholar]
- Nelson, J. R. , Liu, Y. , Fiez, J. , & Perfetti, C. A. (2009). Assimilation and accommodation patterns in ventral occipitotemporal cortex in learning a second writing system. Human Brain Mapping, 30(3), 810–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols, T. , Brett, M. , Andersson, J. , Wager, T. , & Poline, J. B. (2005). Valid conjunction inference with the minimum statistic. NeuroImage, 25(3), 653–660. [DOI] [PubMed] [Google Scholar]
- Ojima, S. , Nakamura, N. , Matsuba‐Kurita, H. , Hoshino, T. , & Hagiwara, H. (2011). Neural correlates of foreign‐language learning in childhood: A 3‐year longitudinal ERP study. Journal of Cognitive Neuroscience, 23(1), 183–199. [DOI] [PubMed] [Google Scholar]
- Ojima, S. , Nakata, H. , & Kakigi, R. (2005). An ERP study of second language learning after childhood: Effects of proficiency. Journal of Cognitive Neuroscience, 17(8), 1212–1228. [DOI] [PubMed] [Google Scholar]
- Parker Jones, O. , Green, D. W. , Grogan, A. , Pliatsikas, C. , Filippopolitis, K. , Ali, N. , … Price, C. J. (2012). Where, when and why brain activation differs for bilinguals and monolinguals during picture naming and reading aloud. Cerebral Cortex, 22(4), 892–902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perfetti, C. A. , & Liu, Y. (2005). Orthography to phonology and meaning: Comparisons across and within writing systems. Reading and Writing, 18(3), 193–210. [Google Scholar]
- Perfetti, C. A. , Liu, Y. , Fiez, J. , Nelson, J. , Bolger, D. J. , & Tan, L.‐H. (2007). Reading in two writing systems: Accommodation and assimilation of the brain's reading network. Bilingualism: Language and Cognition, 10(02), 131. [Google Scholar]
- Price, C. J. (2012). A review and synthesis of the first 20 years of PET and fMRI studies of heard speech, spoken language and reading. NeuroImage, 62(2), 816–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu, J. , Qian, L. , Chen, C. , Xue, G. , Li, H. , Xie, P. , & Mei, L. (2017). Neural pattern similarity in the left IFG and fusiform is associated with novel word learning. Frontiers in Human Neuroscience, 11, 424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi, S. , Gugler, M. F. , Friederici, A. D. , & Hahne, A. (2006). The impact of proficiency on syntactic second‐language processing of German and Italian: Evidence from event‐related potentials. Journal of Cognitive Neuroscience, 18(12), 2030–2048. [DOI] [PubMed] [Google Scholar]
- Saur, D. , Baumgaertner, A. , Moehring, A. , Buchel, C. , Bonnesen, M. , Rose, M. , … Meisel, J. M. (2009). Word order processing in the bilingual brain. Neuropsychologia, 47(1), 158–168. [DOI] [PubMed] [Google Scholar]
- Seo, R. , Stocco, A. , & Prat, C. S. (2018). The bilingual language network: Differential involvement of anterior cingulate, basal ganglia and prefrontal cortex in preparation, monitoring, and execution. NeuroImage, 174, 44–56. [DOI] [PubMed] [Google Scholar]
- Sierpowska, J. , Fernandez‐Coello, A. , Gomez‐Andres, A. , Camins, A. , Castaner, S. , Juncadella, M. , … Rodriguez‐Fornells, A. (2018). Involvement of the middle frontal gyrus in language switching as revealed by electrical stimulation mapping and functional magnetic resonance imaging in bilingual brain tumor patients. Cortex, 99, 78–92. [DOI] [PubMed] [Google Scholar]
- Sigala, N. , & Logothetis, N. K. (2002). Visual categorization shapes feature selectivity in the primate temporal cortex. Nature, 415(6869), 318–320. [DOI] [PubMed] [Google Scholar]
- Snyder, P. J. , & Harris, L. J. (1993). Handedness, sex, and familial sinistrality effects on spatial tasks. Cortex, 29, 115–134. [DOI] [PubMed] [Google Scholar]
- Soto, F. A. , Waldschmidt, J. G. , Helie, S. , & Ashby, F. G. (2013). Brain activity across the development of automatic categorization: A comparison of categorization tasks using multi‐voxel pattern analysis. NeuroImage, 71, 284–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein, M. , Federspiel, A. , Koenig, T. , Wirth, M. , Lehmann, C. , Wiest, R. , … Dierks, T. (2009). Reduced frontal activation with increasing 2nd language proficiency. Neuropsychologia, 47(13), 2712–2720. [DOI] [PubMed] [Google Scholar]
- Sun, Y. , Peng, D. , Ding, G. , Qi, T. , Desroches, A. S. , & Liu, L. (2015). The dynamic nature of assimilation and accommodation procedures in the brains of Chinese‐English and English‐Chinese bilinguals. Human Brain Mapping, 36(10), 4144–4157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamber‐Rosenau, B. J. , Esterman, M. , Chiu, Y. C. , & Yantis, S. (2011). Cortical mechanisms of cognitive control for shifting attention in vision and working memory. Journal of Cognitive Neuroscience, 23(10), 2905–2919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan, L. H. , Laird, A. R. , Li, K. , & Fox, P. T. (2005). Neuroanatomical correlates of phonological processing of Chinese characters and alphabetic words: A meta‐analysis. Human Brain Mapping, 25(1), 83–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan, L. H. , Spinks, J. A. , Feng, C. M. , Siok, W. T. , Perfetti, C. A. , Xiong, J. , … Gao, J. H. (2003). Neural systems of second language reading are shaped by native language. Human Brain Mapping, 18(3), 158–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor, J. S. , Rastle, K. , & Davis, M. H. (2013). Can cognitive models explain brain activation during word and pseudoword reading? A meta‐analysis of 36 neuroimaging studies. Psychological Bulletin, 139(4), 766–791. [DOI] [PubMed] [Google Scholar]
- Tham, W. W. , Rickard Liow, S. J. , Rajapakse, J. C. , Choong Leong, T. , Ng, S. E. , Lim, W. E. , & Ho, L. G. (2005). Phonological processing in Chinese‐English bilingual biscriptals: An fMRI study. NeuroImage, 28(3), 579–587. [DOI] [PubMed] [Google Scholar]
- Timmer, K. , Ganushchak, L. Y. , Ceusters, I. , & Schiller, N. O. (2014). Second language phonology influences first language word naming. Brain and Language, 133, 14–25. [DOI] [PubMed] [Google Scholar]
- Twomey, T. , Kawabata Duncan, K. J. , Hogan, J. S. , Morita, K. , Umeda, K. , Sakai, K. , & Devlin, J. T. (2013). Dissociating visual form from lexical frequency using Japanese. Brain and Language, 125(2), 184–193. [DOI] [PubMed] [Google Scholar]
- Van de Putte, E. , De Baene, W. , Brass, M. , & Duyck, W. (2017). Neural overlap of L1 and L2 semantic representations in speech: A decoding approach. NeuroImage, 162, 106–116. [DOI] [PubMed] [Google Scholar]
- Videsott, G. , Herrnberger, B. , Hoenig, K. , Schilly, E. , Grothe, J. , Wiater, W. , … Kiefer, M. (2010). Speaking in multiple languages: Neural correlates of language proficiency in multilingual word production. Brain and Language, 113(3), 103–112. [DOI] [PubMed] [Google Scholar]
- Wartenburger, I. , Heekeren, H. R. , Abutalebi, J. , Cappa, S. F. , Villringer, A. , & Perani, D. (2003). Early setting of grammatical processing in the bilingual brain. Neuron, 37(1), 159–170. [DOI] [PubMed] [Google Scholar]
- Whatmough, C. , Verret, L. , Fung, D. , & Chertkow, H. (2004). Common and contrasting areas of activation for abstract and concrete concepts: An H 2 15 O PET study. Journal of Cognitive Neuroscience, 16(7), 1211–1226. [DOI] [PubMed] [Google Scholar]
- Wong, B. , Yin, B. , & O'Brien, B. (2016). Neurolinguistics: Structure, function, and connectivity in the bilingual brain. BioMed Research International, 2016, 7069274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolrich, M. (2008). Robust group analysis using outlier inference. NeuroImage, 41(2), 286–301. [DOI] [PubMed] [Google Scholar]
- Woolrich, M. W. , Behrens, T. E. , Beckmann, C. F. , Jenkinson, M. , & Smith, S. M. (2004). Multilevel linear modelling for FMRI group analysis using Bayesian inference. NeuroImage, 21(4), 1732–1747. [DOI] [PubMed] [Google Scholar]
- Worsley, K. J. (2001). Stastistical analysis of activation images. Oxford: Oxford University Press. [Google Scholar]
- Wu, X. , Lu, J. , Chen, K. , Long, Z. , Wang, X. , Shu, H. , … Yao, L. (2009). Multiple neural networks supporting a semantic task: An fMRI study using independent component analysis. NeuroImage, 45(4), 1347–1358. [DOI] [PubMed] [Google Scholar]
- Xu, M. , Baldauf, D. , Chang, C. Q. , Desimone, R. , & Tan, L. H. (2017). Distinct distributed patterns of neural activity are associated with two languages in the bilingual brain. Science Advances, 3(7), e1603309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue, G. , Dong, Q. , Chen, C. , Lu, Z. , Mumford, J. A. , & Poldrack, R. A. (2010). Greater neural pattern similarity across repetitions is associated with better memory. Science, 330(6000), 97–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue, G. , Dong, Q. , Chen, C. , Lu, Z. L. , Mumford, J. A. , & Poldrack, R. A. (2013). Complementary role of frontoparietal activity and cortical pattern similarity in successful episodic memory encoding. Cerebral Cortex, 23(7), 1562–1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue, G. , Dong, Q. , Jin, Z. , Zhang, L. , & Wang, Y. (2004). An fMRI study with semantic access in low proficiency second language learners. NeuroReport, 15(5), 791–796. [DOI] [PubMed] [Google Scholar]
- Zou, L. , Abutalebi, J. , Zinszer, B. , Yan, X. , Shu, H. , Peng, D. , & Ding, G. (2012). Second language experience modulates functional brain network for the native language production in bimodal bilinguals. NeuroImage, 62(3), 1367–1375. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. The reaction times and accuracies of 3 Chinese college students in word naming task during training in a preliminary experiment. D = Day.
Figure S2. Brain maps from language‐by‐learning‐session ANOVA. Results showed significant language main effects (a) in the bilateral cingulate gyrus, prefrontal cortex, occipitoparietal cortex, occipitotemporal cortex, and angular gyrus. Neither the main effect of learning sessions (b) nor the language‐by‐learning‐session interaction (c) was significant in any regions. R = right. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S3. Neural activation differences across the three types of words. CW = Chinese words; EW = English words; ALW = artificial language words. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S4. Neural activation differences between Day 4 and Day 12 for (a) Chinese, (b) English, and (c) artificial language words. All activations were thresholded at p < .001 (whole‐brain corrected). R = right.
Figure S5. The differences between within‐language and between‐language pattern similarity in the two scans. Within‐language pattern similarity was higher than between‐language pattern similarity in a wide neural network both (a) after 4‐day training and (b) after 12‐day training. No regions were found in the reverse contrast. R = right.
Figure S6. Pattern similarity between Chinese and artificial language words shown separately for the three presentations in the 10 predefined ROIs. The upper and lower panels show results (a) in the left hemisphere and (b) those in the right hemisphere, respectively. AG = angular gyrus; FG = fusiform gyrus; ITG = inferior temporal gyrus; PO = pars opercularis; and PT = pars triangularis. R1 = first presentation; R2 = second presentation; R3 = third presentation.
Table S1. Summary statistics of the comparison‐by‐learning‐session ANOVA.
Table S2. Summary statistics of the repetition‐by‐learning‐session ANOVA for C‐A.


