Unstructured abstract
Endothelial to mesenchymal transition (EndMT) is a process whereby an endothelial cell undergoes a series of molecular events that lead to a change in phenotype toward a mesenchymal cell (e.g. myofibroblast, smooth muscle cell). EndMT plays a fundamental role during development, and mounting evidence indicates that EndMT is involved in adult cardiovascular diseases (CVDs), including atherosclerosis, pulmonary hypertension, valvular disease and fibroelastosis. Therefore, the targeting of EndMT may hold therapeutic promise for treating CVD. However, the field faces a number of challenges, including the lack of a precise functional and molecular definition, a lack of understanding of the causative pathological role of EndMT in CVDs (versus being a ‘bystander-phenomenon’), and a lack of robust human data corroborating the extent and causality of EndMT in adult CVDs. Here, we review this emerging but exciting field, and propose a framework for its systematic advancement at the molecular and translational levels.
Keywords: Endothelial to mesenchymal transition, EndMT, cardiovascular
Condensed abstract
Endothelial to mesenchymal transition (EndMT) is a process whereby an endothelial cell undergoes a series of molecular changes that lead to a mesenchymal-like cellular phenotype (e.g. myofibroblast). EndMT plays a fundamental role during development, and mounting evidence indicates that EndMT is involved in adult cardiovascular diseases, raising the possibility that targeting of EndMT holds therapeutic promise. However, this field faces several challenges, including the lack of a precise definition, a lack of understanding of its disease-causal role, and a lack of robust human data. Here, we review this exciting scientific field, and propose a framework for its advancement.
The endothelium is arguably one of thelargest organ systems, and data continue to emerge regarding its heterogeneity and the many complex functions that it performs. Importantly, substantial evidence has implicated ‘endothelial dysfunction’ as contributing to a range of cardiovascular diseases (CVDs). However, the broader programs whereby ‘endothelial dysfunction’ leads to CVD pathogenesis have been challenging to define. Here, we review a rapidly expanding body of literature implicating endothelial to mesenchymal transition (EndMT) as a common and potentially disease-causal biologic program in CVD, highlighting the gaps in knowledge and therapeutic opportunities (Central Illustration).
Central Illustration. EndMT in CVD: Key Mechanisms and Clinical Translation Opportunities.
Summary of major concepts elucidated in this article. Adapted from (144) with permission.
To place EndMT in context, it is important to first consider epithelial to mesenchymal transition (EMT). Our understanding of EMT has its origins in seminal studies of embryonic development from the 1920s and the work of Johannes Holtfreter (1). However, it was not until the 1960’s that chick embryo studies conducted by Elizabeth Hay led to the understanding that epithelial cells can undergo a ‘transformation’ and give rise to embryonic mesoderm (2). It was later appreciated that EMT is reversible (mesenchymal to epithelial transition [MET]), and gradually the term ‘transition’ has replaced ‘transformation.’ Given these initial studies, it is not surprising that a great deal is known about the indispensable roles of EMT/MET during embryonic development (which we previously reviewed from a cardiovascular perspective (3)). However, an equally impressive body of research also attests to the importance of EMT/MET during adult life. While many examples exist, such as the role played by EMT in organ fibrosis (4), perhaps the most relevant from a translational perspective is the role of EMT in cancer (5). While EMT is implicated in multiple aspects of cancer, and in particular epithelial tumor metastasis (5), it is notable that multiple targeted therapies aiming to inhibit EMT in cancer are already undergoing clinical evaluation (5). Furthermore, the inhibition of EMT is a partial effect of several FDA-approved chemotherapeutic agents that are already in use (6).
While a vast amount has been learned about EMT/MET, our knowledge of EndMT is far more rudimentary. However, the endothelium is a specialized sub-type of epithelium and therefore, as highlighted throughout this review, it has been possible to extend some of the prior knowledge regarding EMT to EndMT.
A functional and molecular definition of EndMT
Conceptually, EndMT involves a transition from an endothelial to a mesenchymal-like cellular state. However, at a molecular level, there are no agreed upon criteria for defining EndMT. This is rapidly becoming a hindrance, as there is no standardization and often little cross-comparability among data from different model systems and laboratories. Moreover, with respect to both development and CVD, the field must take account of endothelial cellular origins and their significant heterogeneity when considering formal EndMT definitions. Here we review the current methods and systems used to study and define EndMT.
In vitro EndMT models
EndMT is readily studied using in vitro cell culture systems. Typically, primary endothelial cells (ECs) or EC lines are induced to undergo EndMT by chemical or physical stimuli, with the most widely used being the application of transforming growth factor-beta (TGF-β) for 5–8 days. Again, while a lack of standardization is problematic, an increasing tendency has been to use TGF-β with an additional stimulus, such as interleukin (IL)-1β (7) or hydrogen peroxide (H2O2) (8). These in vitro models have the advantage of providing a convenient and controllable environment to test novel factors and study molecular aspects of EndMT. They also provide a supply of cells that have undergone EndMT, which can be studied in downstream molecular and functional assays. However, a major limitation is that cell culture conditions (e.g. media, supplements) impact the extent and phenotype of EndMT.
In vivo EndMT models
At present, 3 principal methods are used for studying EndMT in vivo. The simplest is to perform immunostaining for endothelial and mesenchymal proteins, which allows co-localization of these markers on individual cells that is suggestive of ‘transitioning’ cells undergoing EndMT. However, this approach cannot identify cells that have substantially reduced or lost EC marker expression, and it is also dependent on the specificity and sensitivity of the antibodies used for immunostaining. Furthermore, under light microscopy, the superimposition of an EC and mesenchymal cell can be erroneously interpreted as a single cell undergoing EndMT.
While generally only applicable to mouse models, endothelial-specific Cre-lox lineage tracking systems are a more rigorous approach for studying EndMT in vivo (9). Such mice are able to activate Cre-recombinase, which can be placed under the control of an endothelial-specific gene (e.g. VE-Cadherin). Cre activation is used to trigger defined genetic events, like the expression of a fluorescent marker protein that can be used to track ECs. With careful selection, Cre-lox systems can achieve permanent fluorescent marking of ECs, such that they continue to exhibit the fluorescent signal even if they undergo EndMT and suppress endothelial gene/protein expression. Alternatively, EC-specific Cre mouse strains can be crossed to “floxed” strains, where Cre activation leads to the deletion of a gene of interest. EC-specific Cre-lox gene deletion strategies can be used to selectively delete genes of interest that regulate EndMT, and thus, the effects of these genes and EndMT on differing biologic processes can be determined (10,11).
As a further method for studying EndMT in vivo (and also in vitro), high-throughput RNA-sequencing, of bulk or single cell preparations, is a powerful tool for studying the cellular transcriptome, whereby endothelial and mesenchymal gene expression patterns can be profiled to define the extent of EndMT. For example, bulk RNA deep sequencing of purified murine cells showed that following myocardial infarction or tissue hypoxia, ECs undergo clonal expansion and express mesenchymal genes such as SM22α in vivo (12). In addition, due to its potential to resolve EC signatures while concurrently showing mesenchymal gene upregulation at the single cell level, it is anticipated that single cell RNA-sequencing will be another useful tool for studying EndMT in vivo in human samples. Furthermore, RNA-sequencing holds promise for providing insights on EC plasticity, which is the ability of an EC to switch its identity, including to additional phenotypes other than mesenchymal cells and also, having changed identity, to revert back to an EC state (see review (13)).
Cellular and molecular analysis of EndMT
A diverse selection of readouts has been used to demonstrate EndMT, but obligatory characteristics are either: i) reduced expression of endothelial genes/proteins; ii) increased expression of mesenchymal genes/proteins, or iii) ideally, both of these. Typically, most investigators present 2–3 each of endothelial and mesenchymal genes/proteins. Common examples include: Endothelial; CD31, VE-Cadherin, endothelial nitric oxide synthase (NOS3); Mesenchymal; α-smooth muscle actin (α-SMA), calponin, SM22α, versican. However, there is no agreement on which genes/proteins should be studied, or how many, and the level of change required. Additional features that are sometimes also studied include increased expression of EndMT-associated transcription factors such as TWIST, SMAD3, ZEB2, SNAI1 and SNAI2.
Looking ahead, we propose that future studies should seek to provide more comprehensive transcriptomic and proteomic profiles of any proposed EndMT phenomenon. Furthermore, in any individual cell or cell population undergoing EndMT, gradations of EndMT exist (i.e. partial versus more complete EndMT, reversible, transient etc.), and there may be relative differences in the extent of endothelial gene/protein downregulation versus mesenchymal upregulation (12). This heightens the importance of a complete portrayal of EndMT using high-throughput techniques, whereby the balance of endothelial gene/protein downregulation versus mesenchymal upregulation is fully appreciated.
Functional and phenotypic cell changes during EndMT are also fundamental to this process and, thereby, to a definition of EndMT. Accordingly, studies of EndMT are increasingly demonstrating relevant changes in phenotypic traits (Table 1). However, yet again, no standardization exists. Indeed, reaching a consensus on these functional cellular aspects may be challenging, because certain EndMT-related phenotypic features may be important in specific contexts, but irrelevant or even opposing in others. For example, reduced tubule formation (i.e. angiogenesis) has been associated with EndMT (14,15), but as an apparent paradox, at least partial EndMT is necessary for angiogenesis (16). Highlighting this paradox at a molecular level, the transcription factor SNAI2 is expressed in angiogenic ECs and mediates angiogenesis (16), but SNAI2 is also a key mediator of EndMT (11). A full reconciliation of these functional aspects will likely remain challenging until more is understood about EndMT as a whole.
Table 1.
Functional studies to support an altered cellular phenotype with EndMT.
| Assay | Cell characteristic | References |
|---|---|---|
| Reduction of endothelial characteristics | ||
| EC tubule formation in culture | Cellular ability to form tubules in culture – a defining EC characteristic | (14,15) |
| Thrombin generation | Cellular ability to inhibit thrombin formation | (14,15) |
| Lectin binding | Lectin binding is a defining EC characteristic | (15) |
| LDL-uptake | Ability of cells to uptake low density lipoprotein (LDL) cholesterol – a defining characteristic of ECs | (15) |
| Enhancement of mesenchymal characteristics | ||
| Invasion | Ability to invade through matrix and other substrates | (8) |
| Migration | Ability of cells to migrate across a transwell and/or though micropores | (8,14) |
| Contraction | Enhanced cell contractility with mesenchymal phenotype | (14,15,60) |
| Collagen production | Enhanced collagen production with mesenchymal phenotype | (108) |
EndMT in cardiac development
Heart progenitor cells arise within the embryo from newly formed mesoderm that originates from the primitive streak. After heart tube formation, the endocardium and endothelium of the great vessels are created by vasculogenesis (17–19); whereby vessels form de novo from endothelial progenitors. The endocardium likely has heterogeneous origins, arising from endocardial-myocardial heart field progenitors (17,18,20–22), and also cells that migrate in from the yolk sac mesoderm (an extra-embryonic tissue) (19). Some endocardial cells express markers of hemogenic endothelium, perhaps reflecting their origins from yolk sac hemangioblasts (a common progenitor of blood and vessels) (19) and the activation of the hematopoietic program within embryonic endothelium (23).
As the heart develops, the endocardium retains remarkable cellular plasticity. For example, the endocardium associated with the forming ventricles undergoes a process that resembles angiogenic sprouting in developing vascular beds (24), leading to the formation of endocardial domes which, together with myocardium and extracellular matrix (ECM), define the morphological units of trabeculation. The endothelium of the coronary arteries and veins is also formed by sprouting, in this case from the sinus venosus endocardium into the myocardium (25). This process leads to formation of an endothelial plexus within the sub-epicardial ECM, which then extends deeper into the myocardial walls (26,27). Developmental patterning of the coronary vessels may then be supplemented by adaptive angiogenesis initiated by sprouting of endocardium from the inter-trabecular crypts into the myocardial wall, driven by hypoxia (27–30). Trabecular endocardium also contributes to the coronary arterial tree postnatally, as the outer “compact” myocardial layer undergoes expansion (29). Blood islands form on the ventral surface of the heart through a budding process and these contain an endothelium that is also derived from the endocardium (27). Blood island endothelium expresses hemogenic markers (31), suggesting that the blood cells found within these islands are derived from the endothelium (and hence endocardium). Endocardial lineage plasticity is further highlighted by its ability to trans-differentiate into adipocytes and mural cells in distinct settings (32,33).
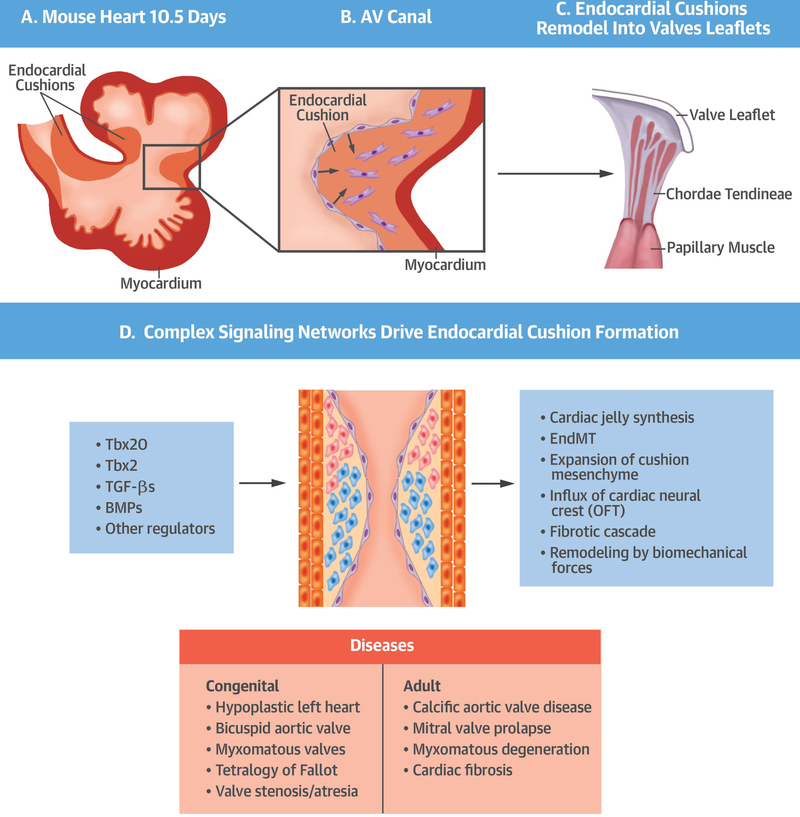
The touchstone of EndMT occurs during the formation of the endocardial cushions, which are the precursor structures for the cardiac valves (Figure 1) (34,35). The endocardial cushions first appear as prominent swellings of ECM (called cardiac jelly) located between endocardium and myocardium in the valve-forming regions of the atrioventricular (AV) canal and outflow tract. Cushions are induced locally by TGF-β signaling from adjacent myocardium, which increases the synthesis of cardiac jelly ECM and pathways that induce EndMT (34,36). At around embryonic day 8.5–9.0 in the mouse, following heart looping, a subset of endocardial cells lining the cushions undergo EndMT and migrate into the cushion ECM (34). Genetic lineage tracking shows that the majority of mesenchymal cells infiltrating the cushions are derived from endocardium (37), although the lateral (parietal) AV valve leaflets are composed of epicardium-derived cells (38). Cushion infiltration is mediated by metalloproteinases and ECM receptor signaling, and is accompanied by both new synthesis and degradation of ECM. During further development, cellularized cushions are remodeled into valve leaflets with stratified mesenchymal and ECM layers (34), a process that relies on genetic and hemodynamic cues (39). Cushion mesenchyme also contributes to the structural fibrotic tissue that knits together the inter-atrial and inter-ventricular septal structures with the valvular complexes (36). The outflow tract cushions are also infiltrated by migratory cranial neural crest cells, which contribute to outflow tract septation forming the aortic and pulmonary trunks (34,35).
Figure 1. EndMT during cardiac development.
(A) Schematic cross-section of the developing mouse heart at ~10.5 days showing major chambers, forming septa, and the outflow tract (OFT). (B) Schematic enlargement of one side of the AV canal (boxed region in A). During EndMT, ECs of the AV canal and OFT become mesenchymal and occupy the prominent ECM swellings separating myocardium and endocardium (endocardial cushions) in those regions. (C) Cellularized endocardial cushions are later remodeled into stratified valve leaflets. Here, a mitral valve leaflet is shown stratified into atrialis [A], spongiosa [S] and fibrosa [F] layers, and tethered to the ventricle by papillary muscles and chordae tendineae. (D) Complex signaling networks drive endocardial cushion formation, EndMT, expansion of cushion mesenchyme and cardiac remodeling, which are further modulated by biomechanical forces associated with heart contraction and blood flow. Key molecular factors (see text and Figure 2) and relevant human congenital and adult valvular diseases are indicated.
The involvement of EndMT in formation of the AV-septal complex, and its critical role in valvulogenesis, septation, alignment of the cardiac chambers and vessels, and hemodynamics, creates a vulnerability that underlies both congenital heart disease and adult valvular disease. At the severe end of the spectrum, aberrant cushion development may lead to complete AV canal defect, which is typically fatal. More subtle forms of endocardial and cushion mal-development may contribute to a variety of congenital heart diseases. For example, in the rare but serious hypoplastic left heart, aortic and/or mitral valve stenosis may be a contributing factor. Likewise, pulmonary valve stenosis is a part of the tetralogy of Fallot (Figure 1). We focus specifically on EndMT in valvular disease later in this article.
Signaling pathways and mechanisms controlling EndMT
EndMT signaling during development
A large number of signaling pathways govern EndMT during cardiac development (see (34–36,40)). Briefly, signaling through bone morphogenetic proteins (BMPs) and TGF-β ligands and receptors, that is modulated by the Hippo pathway (41), leads to endocardial expression of Snai1, Snai2 and Twist, which encode archetypal transcription factors regulating EndMT (36,40). The NOTCH pathway is also essential for EndMT, although it is not required for the initial formation of ECM swellings (42). NOTCH ligands Delta-like 4 and NOTCH receptors (NOTCH1–4) are expressed on AV canal and outflow tract endocardial cells before and throughout EndMT. When membrane-bound NOTCH1 receptor is engaged by ligand, sequential protease cleavages release the NOTCH1 intracellular domain (N1ICD) which migrates to the nucleus and acts as a transcription co-regulator, activating and repressing genes that define cell identity. NOTCH1 signaling through an alternate ligand Jagged 1, expressed from myocardium, restrains BMP-mediated EndMT, highlighting the presence of negative feedback mechanisms (43). NOTCH1 intracellular domain binds directly to and positively regulates Snai1 and Snai2, and the expressed Snai1 and Snai2 proteins repress Ve-Cadherin transcription to allow EndMT. Signaling pathways involving WNT/β-catenin, VEGFA/VEGFR and neuregulin 1/ERBB2/ERBB3, as well as transcription factors NFATC1, GATA4 and SOX9, and ECM proteins hyaluronan and versican, are also involved in EndMT and subsequent valve maturation (34–36,40). As a result of these signaling pathways, endocardial-derived cells within the cushions undergo EndMT and adopt a fibroblastic fate. Like fibroblasts in other connective tissues, valvular fibroblasts undergo a maturational process akin to bone, cartilage and tendon formation, and the transcription factor SOX9, which is induced by BMPs, acts as a central regulator of ECM gene expression networks (44).
TGF-β and the TGF-β superfamily
The TGF-β superfamily is an extensive signaling network that is considered a master regulator of EndMT and which comprises TGF-β isoforms 1–4, BMPs, activins and related proteins (Figure 2). Among these, while TGF-β3 and TGF-β4 are less studied, both TGF-β1 (11,45) and TGF-β2 (8) promote EndMT. TGF-β and other ligands from the superfamily signal via TGF-β receptor complexes (46). These receptor complexes combine and are comprised of two type I and two type II receptor components (four components in total), which include activin receptor-like kinases (ALKs) and BMP receptor components. Also among these are TGF-β receptor 1 and 2 (TGF-βR1 and TGF-βR2), with TGF-βR2 being a type II receptor component. Type I receptor components are comprised of the ALK family, which include TGF-βR1 (also known as ALK5). In the complex, type II receptors phosphorylate and active type I components, which then propagate the signal intracellularly. There are seven type I and five type II receptor complexes in humans, however, the binding possibilities are restricted in ECs, where TGF-β binding to TGF-βR2 can activate either of two type I receptors; ALK1 which is largely restricted to ECs, or the broadly expressed ALK5 (47,48). Accessory TGF-β receptors may also become involved, like endoglin or betaglycan, which modulate signaling through type I and II receptors.
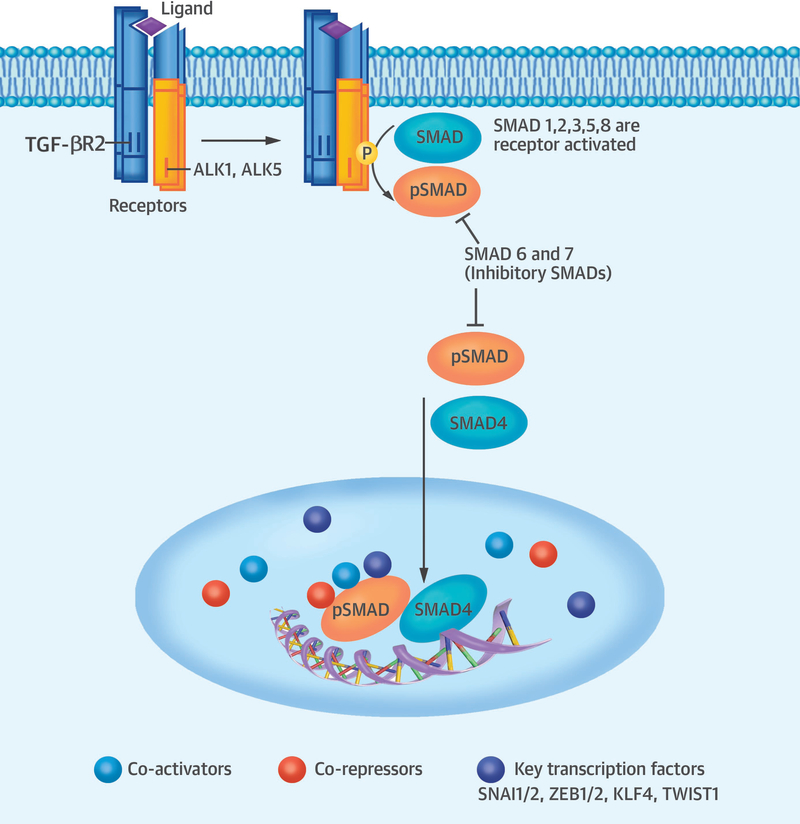
Figure 2. TGF-β signaling and EndMT.
In ECs, TGF-β classically signals via TGF-βR2 (a type II receptor component) and AKL1 or ALK5 (type I receptor complexes). Receptor complexes combine on the cell surface and are comprised of two type I and two type II components. TGF-βR2 phosphorylates (activates) type I components, which then propagate the signal intracellularly via activation (phosphorylation; pSMAD) of SMAD 1,2,3,5 or 8. Activated SMAD proteins form complexes that include the common mediator SMAD4, and which may be inhibited by SMAD6 or SMAD7. SMAD complexes shuttle to the nucleus, where they interact with co-activators, co-repressors and additional transcription factors, the latter including key EndMT gatekeepers SNAI1/2, ZEB1/2, KLF4, TCF3 and TWIST. These interactions culminate in chromatin rearrangements and transcription factor binding to endothelial, mesenchymal and other gene promoter regions that ultimately bring about EndMT.
Upon type I receptor activation, TGF-β family members regulate gene expression via SMAD transcription factor activation (i.e. via phosphorylation) (46,49). Activated SMAD proteins form complexes and shuttle to the nucleus, where they interact with additional transcription factors that include key regulators of EndMT: SNAI1, SNAI2, ZEB1, ZEB2, KLF4, TCF3 and TWIST. These interactions culminate in chromatin rearrangements and transcription factor binding to endothelial, mesenchymal and other relevant gene promoter regions which induce EndMT (46,49) (Figure 2).
There are multiple checkpoints in this system including the ligand BMP7 which inhibits EndMT (45), and SMAD7, which exerts an inhibitory effect at the transcriptional level (7). In addition, although TGF-βs signal mainly via the SMADs (termed “canonical TGF-β signaling”), they may also activate other complimentary pathways (“non-canonical TGF-β signaling”).
The TGF-β signaling system also acts as a final common mechanism for other pathways. Important factors that intersect with TGF-β signaling include mitogen-activated protein kinases (MAPKs), the phosphoinositide 3-kinase (PI3K) pathway, inhibitory micro RNAs (miRNAs) such as the miR-200 family, and others. Therefore, as well as canonical and non-canonical TGF-β signaling, the TGF-β signaling system serves to integrate these other pathways and to fine-tune the ultimate regulatory changes governing EndMT (7).
Metabolic regulation of EndMT
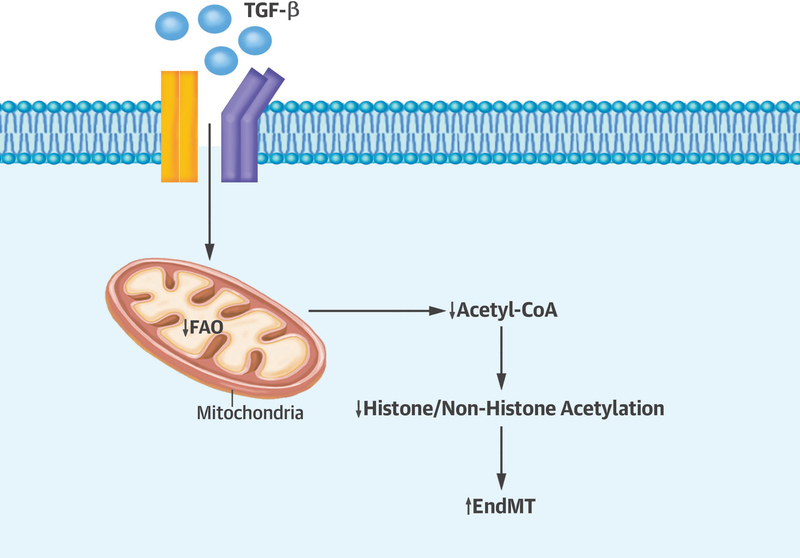
There is a growing appreciation that cellular fate is mechanistically associated with intracellular metabolism. However, the mechanisms linking these processes are imprecisely understood. As a new development, a recent study suggests that EndMT may have metabolic underpinnings (7). Using TGF-β1 to induce EndMT in vitro, it was shown that TGF-β1 triggered a reduction in mitochondrial-dependent fatty acid oxidation (FAO) (7). In other cell types and paradigms, TGF-β signaling had been shown to modulate glucose metabolism (50), lipid metabolism (51) and mitochondrial function (52). For the case of ECs, the TGF-β-stimulated inhibition of FAO resulted in a decline in acetyl-CoA (7). Indeed, this fall in acetyl-CoA was an important metabolic stimulus for EndMT, as other genetic or pharmacological strategies that reduced cytosolic acetyl-CoA levels could recapitulate the effects of TGF-β signaling (7) (Figure 3). Notably, although FAO inhibition would be expected to primarily alter mitochondrial acetyl-CoA levels, the authors found that it was the cytoplasmic pool of acetyl-CoA that was modulating cellular fate. These pools are not in equilibrium and there is growing evidence that acetyl-CoA modulates its effects under strict spatiotemporal control (53). These observations likely have in vivo relevance since it was further shown that genetic disruption of endothelial FAO augmented the contribution of EndMT to mitral valve development in a mouse model (7), suggesting that targeting of endothelial metabolism might be a therapeutic strategy to modulate EndMT in other pathological settings.
Figure 3. Fatty acid oxidation regulates EndMT.
Key elements of the role of FAO in regulating EndMT.
Finally, there is an additional potential link between metabolism and EndMT. There is increasing evidence for a role of EndMT in fibrotic disease, including the fibrosis associated with chronic kidney disease (54). In that sense, other studies have suggested that fibrosis in chronic kidney disease is somehow mediated by a fall in FAO (55). It is tempting to speculate that the mechanistic link between a fall in FAO and the increase in fibrosis is somehow related to an altered threshold for EndMT, or through the related process of EMT.
Non-coding RNAs in EndMT
Non-protein coding RNAs (ncRNAs) play a major role in cell fate decisions and recent advances have also underlined their critical role in regulating EndMT. NcRNAs include micro RNAs (miRNAs), long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs), which together could influence the entire EndMT regulatory program.
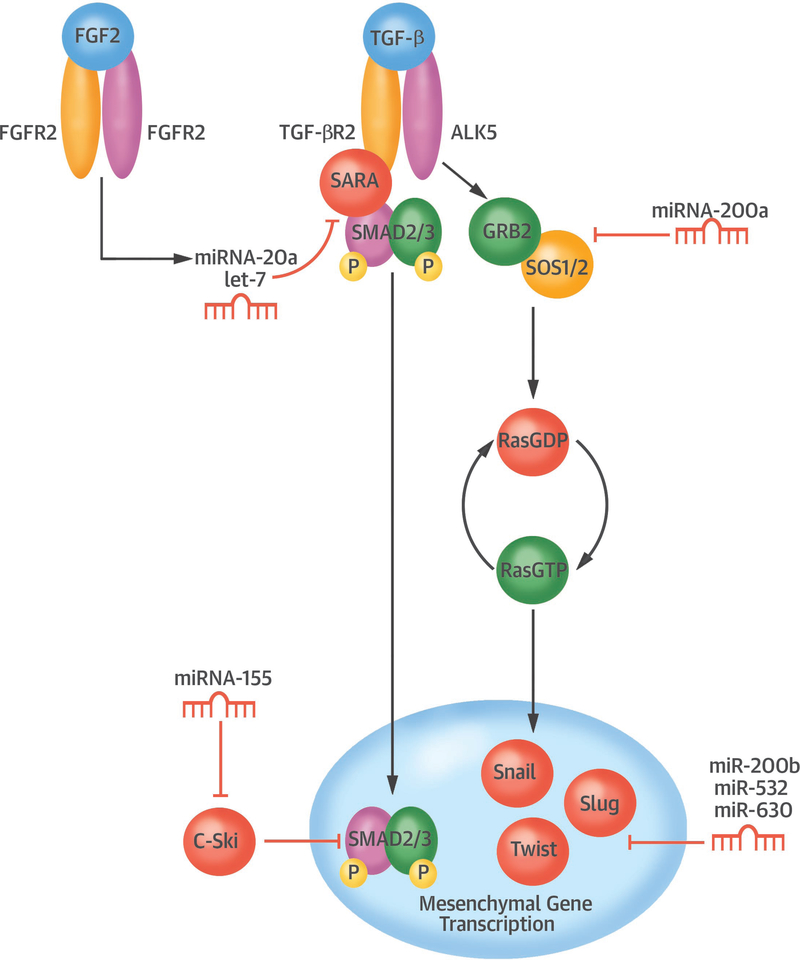
MiRNAs are small, non-coding RNAs that inhibit the expression of their gene targets, predominantly by inducing messenger RNA (mRNA) degradation or inhibiting mRNA translation. In the context of EndMT, TGF-β induces a distinct shift in EC miRNA expression (56), suggesting their importance in the overarching regulation of EndMT. Notably, several miRNAs have been identified that antagonize the EndMT transcriptional program, which are transcriptionally suppressed by TGF-β signaling (57–62). For example, fibroblast growth factor (FGF)2, an antagonist of TGF-β signaling in ECs (63), induces the expression of miR-20a which then silences TGF-βR1 and TGF-βR2 expression, effectively blunting canonical TGF-β signaling (60). Chen et al. also showed that miRNA let-7 negatively regulates TGF-βR1 expression (59). Similarly, miR-200a can reduce the expression of growth factor receptor-bound 2 (GRB2), a mediator of non-canonical TGF-β signaling (62). GRB2 plays a vital role in the development of cardiac fibrosis (64), a condition wherein EndMT may be present (45), and the ectopic expression of miR-200a in ECs treated with TGF-β blunted the EndMT response (62). Downstream of TGF-β receptors, miRNAs also affect the expression of SNAI1 (e.g. miR-200b and miR-532 (57,58)), and SNAI2 (e.g. miR-630 (61)).
As well as miRNAs that directly suppress EndMT, TGF-β induces the expression of miRNAs that affect endothelial gene expression or that suppress inhibitors of mesenchymal gene transcription (65–67). In ECs, mesenchymal gene transcription is kept inactive by transcriptional repressors, including the SKI proto-oncogene (c-Ski) and the ternary complex factor ELK1. C-SKI represses TGF-β signaling by stabilization of inactive SMAD complexes on SMAD-binding elements (68), which is inhibited by miR-155 upon TGF-β signaling (67). Similarly, ELK1 is repressed by miR-27b upon TGF-β signaling (66). ELK1 competes with the mesenchymal transcription factor MRTF in binding to serum response factor, thereby acting as a myogenic repressor (69). The loss of ELK1 from ECs leads to increased MRTF activity (70) and mesenchymal gene transcription (71). Besides miRNAs that affect mesenchymal gene expression, TGF-β also increases the expression of miRNAs that suppress endothelial protein expression. Sustained AKT activation facilitates EndMT (72) and culminates in elevated expression of matrix metalloproteinases (73,74) that can degrade VE-Cadherin (75). PTEN is an endogenous inhibitor of AKT activation (76) and a target of miR-21 (65), suggesting that miR-21 inhibition can inhibit EndMT. Similarly, the systemic delivery of miR-21 antagonists reduced the number of cells undergoing EndMT in the cardiac microvasculature, and altered cardiac fibrosis in mice (65). Notably, the regulation of EndMT by miRNAs is not limited to these examples (see also Figure 4), and the list of miRNAs implicated in EndMT appears certain to expand.
Figure 4. The role of non-coding RNAs in regulating EndMT.
Key elements of what is known about how EndMT may be controlled by non-coding RNAs. Abbreviations not previously defined: FGFR, FGF receptor; SARA, SMAD anchor for receptor activation; SOS, Son of sevenless homolog.
LncRNAs are a vast additional class of ncRNA that regulate gene transcription by a variety of mechanisms. Recently, GATA6-AS, a long non-coding antisense transcript of GATA6, was shown to facilitate EndMT by interacting with the histone deaminase LOXL2 (Lysyl oxidase homolog 2) to regulate endothelial gene expression via chromatin remodeling (77). Moreover, the lncRNA MALAT1 was shown to suppress the function of miR-145, which culminated in increased expression of TGF-βR2 and SMAD3, facilitating EndMT (78). However, little is currently known about how lncRNAs are regulated and functionally relevant in EndMT; an area that is important to pursue with the improving knowledge of lncRNA biology. Notably, lncRNAs are generally poorly conserved across species; adding difficulty to proving in vivo evidence of their function. This may be particularly relevant when considering translational animal studies targeting lncRNAs as a route to human therapeutics.
CircRNAs are a poorly understood subset of lncRNA that are characterized by their covalently closed loop structures (79), with current research suggesting a possible regulatory role for circRNAs in EMT (80). If a regulatory role for circRNAs in EndMT is also demonstrated, this will assuredly be a rich area for further basic research.
Epigenetic control of EndMT
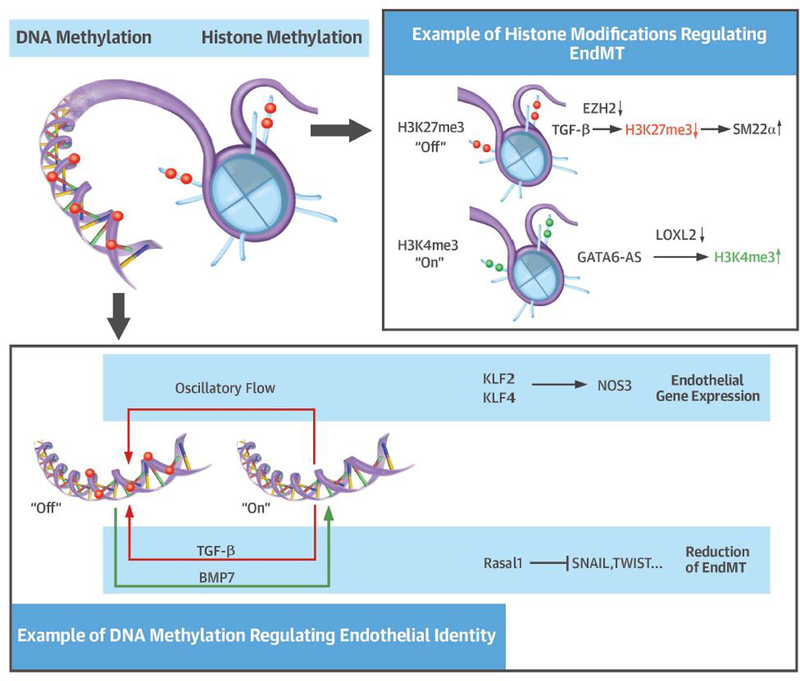
‘Epigenetic’ refers to heritable control of gene expression that does not involve changes to the underlying DNA sequence. Epigenetic control can occur at the level of DNA, where DNA methylation induced by DNA methyltransferases (Dnmts) results in silencing of gene expression, a process which can be reversed by DNA demethylases (e.g. TETs). In addition, various histone modifications, including acetylation and methylation, control accessibility of transcription factors to target gene promoter regions. Whereas multiple studies have elucidated the epigenetic control of EMT, little is known regarding the epigenetic control of EndMT (Figure 5).
Figure 5. Epigenetic mechanisms and control of EndMT.
Key elements of what is known of how epigenetic changes modulate EndMT.
In EMT, expression of the SNAI1/2 family, TWIST and ZEB1/2 is controlled by DNA methylation as well as histone acetylation and methylation (81). Also, the effects of transcription factors on their target genes (e.g. E-Cadherin) are regulated by corepressors, including the histone deacetylases (HDACs), histone methyltransferase G9a or SUV39H1, and Dnmts (81). In ECs, epigenetic mechanisms at the level of DNA methylation or histone modifications play a crucial role in the expression of EC-specific genes and up-stream regulators. For example, DNA methylation represses the flow-induced transcription factors of the Krüppel-like family Klf2 and Klf4 (82–84), which are important for maintaining endothelial function and are involved in EndMT (13). Likewise, the promoter of Nos3 is repressed in non-ECs by DNA methylation and is controlled by histone acetylation and methylation (Figure 5) (85,86). These mechanisms regulate endothelial-specific gene expression in response to differing stimuli, however, whether EndMT is associated with complete, direct and long-lasting silencing of endothelial genes via epigenetic mechanisms is unclear.
DNA methylation patterns are modulated under conditions of EndMT and can indirectly interfere with EndMT signaling. Altered DNA methylation in response to oscillatory flow was reported in aortic intima-media tissues from patients with aortic valve disease (87). This study showed that methylation patterns are distinct in dilated versus non-dilated ascending aortas, and specifically that non-dilated aortas from patients with bicuspid aortic valve disease show a methylation signature associated with cell transformation and differentiation. Conversely, the flow response in ascending aortas from patients with bicuspid aortic valves involved hypomethylation and increased expression of Wnt/β-catenin genes, while an angiogenic profile was observed in the aortas of patients with tricuspid aortic valves (87). Whether these changes in DNA methylation are solely due to ECs and how they causally contribute to aneurysm formation in patients with bicuspid aortic valves will be important to understand. Additional insights regarding the epigenetic control mechanisms of EndMT were gained in animal models of cardiac fibrosis. Here, TGF-β1 induced DNA methylation of the promoter of the Ras inhibitor RASAL1, thereby increasing the expression of SNAI1, SNAI2 and TWIST and promoting EndMT in vitro and in vivo. Interestingly, BMP7 reversed the TGF-β1-induced RASAL1 promoter methylation and subsequent silencing of gene expression via induction of the DNA demethylase TET3 (88). At the level of histones, enhancer of zeste homolog-2 (EZH2), a methyltransferase of the polycomb complex, was shown to regulate SM22α expression (89). TGF-β2 reduced EZH2 levels in ECs, leading to a decrease in silencing H3K27me3 marks at the SM22α promoter (89). Furthermore, the HDAC3 isoform HD3α was shown to induce EndMT (90). However this effect was likely not caused by epigenetic control mechanisms, but was mediated via HD3α interactions with Akt and regulation of TGF-β2 (90). Finally, as mentioned earlier, lncRNAs may control EndMT by interfering with histone modifications, where GATA6-AS was shown to regulate EndMT and modulate H3K4m3-dependent gene expression by binding to LOXL2 (77).
Other factors and pathways influencing EndMT
It is notable that TGF-β signaling only partly induces EndMT (8,11,45), suggesting that additional mechanisms are also involved. Although several stimuli, including glucose (91), endothelin-1 (92), angiotensin II (93) and advanced glycation end-products (94) induce EndMT by converging with TGF-β signaling, alternate pathways of EndMT induction also exist. Among these, Jagged/NOTCH signaling can directly induce the expression of SNAI2, TWIST and the mesenchymal transcription factor RUNX3 (95–97). In addition, Wnt/β-Catenin signaling drives EndMT via increased SNAI2 expression (98). Interestingly, Wnt/β-Catenin-induced EndMT via SNAI2 induction does not change SNAI1 transcripts (99), indicating that not all transcription factors are required for EndMT induction.
Oxidative stress is another factor that promotes EndMT. Specifically, hydrogen peroxide (H2O2), a classic inducer of oxidative stress, promotes EndMT (8). Furthermore, the effect of H2O2 is additive to TGF-β (8), and the inhibition of reactive oxygen species can decrease oxidative stress-induced EndMT in vitro (100). Consistent with this, EndMT is also promoted by the inhibition of nitric oxide synthase, which reduces the bioavailability of nitric oxide and enhances oxidative stress (101). The importance of oxidative stress in EndMT is being further explored, with recent studies suggesting that oxidative stress may promote EndMT in the setting of atherosclerosis and renal fibrosis (102,103).
As mentioned, endogenous inhibitors of EndMT also exist, although their mechanisms of action are incompletely understood. FGF signaling in ECs abrogates TGF-β signaling by suppressing the transcriptional activity of SMAD2 (104) and the induction of miRNAs that silence TGF-β receptor expression (59,60). BMP7 can antagonize TGF-β signaling by induction of ID proteins (105), which are dominant negative helix–loop–helix proteins that lack a DNA-binding domain. ID proteins can heterodimerize with SMAD2 and SMAD3, resulting in the formation of inactive transcription factor complexes (106). Of note, ID protein expression is reduced during EndMT (15) and the restoration of ID protein expression can inhibit EMT in certain tumors (107). Although these data suggest a role for ID proteins in EndMT, this is yet to be confirmed. Undoubtedly, many additional pathways controlling EndMT remain to be disclosed.
Adult cardiovascular diseases and pathologic processes where EndMT has been implicated, and clinical translation opportunities
Atherosclerosis and plaque erosion
The accumulation of mesenchymal cells, including myofibroblasts, smooth muscle cells and osteoblasts, is central to plaque formation and atherosclerosis. Mesenchymal cells play key roles in this disease including proinflammatory molecule secretion; matrix, collagen and metalloproteinase production; plaque calcification; and fibrous cap formation. As early evidence suggesting EndMT is involved in atherosclerosis, co-staining of human atherosclerotic plaques and porcine vessels for endothelial and mesenchymal markers identified co-positive cells in the intima and within neointimal tissues (108). In addition, while uniform laminar shear stress was found to inhibit EndMT, ECs exposed to disturbed flow (as is typical in atherosclerosis-prone regions) underwent EndMT and showed atherogenic differentiation. Gain- and loss-of-function studies established a role for ERK5 signaling in the inhibition of EndMT with uniform laminar shear stress (108). Supporting these findings, Mahmoud et al. showed that low, oscillatory shear stress promotes EndMT, while high shear stress is protective (109). In this case, low-shear related EndMT was under the control of SNAI1 (109) and TWIST1 (110), while co-staining was again suggestive of EndMT in human atherosclerotic plaques (109). Importantly, the link between EndMT and disturbed flow indirectly suggests that EndMT may be causal for atherosclerosis. As a sidebar, but also indirectly suggesting that EndMT may be causal for atherosclerosis, it was recently shown that athero-protective high-density lipoproteins inhibit EndMT (111).
Two studies have used Cre-lox mouse models to study EndMT in atherosclerosis, with both showing that EndMT plays an important role. Of these, Evrard et al. (8) showed that the predominant EndMT-derived cell population in atherosclerosis is fibroblast-like cells, with a lesser contribution to smooth muscle-like cells (Figure 6). Overall, EndMT-derived cells comprised almost half of the fibroblast population in advanced atherosclerotic lesions. In addition, they showed that EndMT is associated with increased plaque vulnerability. On the other hand, Chen et al. (10) studied a potential link between disrupted FGF signaling, EndMT and atherosclerosis. In addition to lineage tracking, they also created atherosclerotic mice with endothelial-specific deletion of FGF receptor substrate 2α (Frs2α). These knockout mice exhibited extensive EndMT and developed atherosclerosis earlier than control mice, eventually demonstrating an 84% increase in total plaque burden. As a whole their study suggested a link between loss of protective endothelial FGF signaling, development of EndMT, and progression of atherosclerosis.
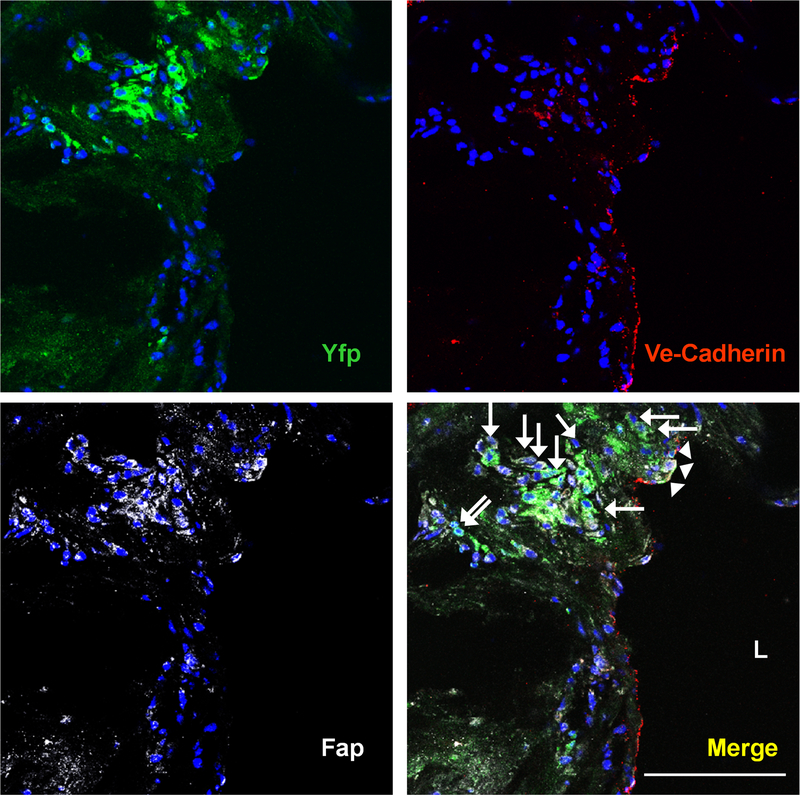
Figure 6. EndMT in atherosclerosis and plaque erosion.
Confocal microscopy of thoracic aortic sections from tamoxifen-induced end.SclCreERT;R26RstopYfp;ApoE−/− mice after Western diet feeding. In this model of advanced atherosclerosis, ECs are permanently marked using a Cre-lox system such that ECs, and all EC-derived cells, permanently express yellow fluorescence protein (Yfp). Staining for Ve-Cadherin is in red, with staining for fibroblast activation protein (Fap), a fibroblast marker, in white. DAPI nuclear staining is in blue. Yfp+Fap+Ve-Cadherin+ cells (arrowheads) represent endothelial-derived cells expressing endothelial and fibroblast proteins. Yfp+Fap+Ve-Cadherin- cells (arrows) represent endothelial-derived cells that express Fap, but which have lost Ve-Cadherin expression. L=lumen; scale bars, 100 μm. Reproduced from (8) with permission.
It is provocative and exciting that these studies, involving mice, large animals and humans, have consistently shown that EndMT is prominent in atherosclerosis. An important next step will be to define the exact functional role of EndMT in the development and progression of atherosclerotic disease (versus being an epi- or bystander-phenomenon). Furthermore, we believe that another important step is to investigate the role of EndMT in ‘plaque erosion.’ In brief, plaque erosion may lead to arterial thrombosis and accounts for ~30% of acute coronary events (112). Mechanistically, plaque erosion occurs without fibrous cap disruption, where blood comes into contact with an intimal surface lacking ECs. Supporting the hypothesis that EndMT is involved, plaque erosion is more common in arterial bifurcations and areas of disturbed blood flow (112). We speculate that if a significant proportion of ECs undergo EndMT, this may lead to a disrupted endothelial layer that culminates in plaque erosion. Subjectively, images obtained during lineage tracking of EndMT in atherosclerosis (8), give the impression that the loss of ECs over the surface of plaques is related to their migration into the plaque’s inner aspects (Figure 6).
Valvular disease
While EndMT is critical to valve development, low levels of EndMT likely persist in postnatal and adult cardiac valves. As gauged by CD31/α-SMA co-expression, ~10% of ECs in human fetal valves undergo EndMT, decreasing to ~1% in human adult valves (113). Importantly, this raises the hypothesis that the adult valvular endothelium contains a subset of cells that can undergo EndMT to replenish the turnover of valvular interstitial cells (114), thus maintaining valve tissue homeostasis.
Recent evidence implicates EndMT in valvulopathies (115), and many transcriptional regulatory mechanisms of heart valve development actively respond to valve injury, stress, and disease (Figure 7). Whereas interstitial valvular cells are quiescent fibroblasts in healthy adult valves, during disease progression they transform into activated myofibroblast-like cells that express α-SMA (116), and subsequently differentiate into osteoblast- and chondrocyte-like cells characteristic of calcific aortic valve disease (117). Since many abovementioned fundamental pathways involved in valvulogenesis (e.g., NOTCH, Wnt, BMP, TGF-β) also participate directly in valvular calcification, the question arises whether EndMT can generate osteogenic cells. The discovery that cadherin-11, which is important for cushion formation, is re-expressed in the endothelium and osteoblast-like interstitial cells in adult human aortic valves may support this notion (118). Moreover, in vivo and in vitro studies have demonstrated the osteogenic potential of a subpopulation of mitral valve ECs (119). Furthermore, Hjortneas et al. (120) showed that EndMT precedes osteogenesis and that valvular interstitial cells suppress calcification of valvular ECs undergoing EndMT. Recent studies also implicated inflammation and mechanical stress in potentiating valvular EndMT (121–123). To recapitulate the microenvironment of mechanical strain, 2D microcontact printing was used to mimic regions of healthy and diseased leaflets, and to measure EndMT in sheep valve ECs responding to low (10%, healthy) and high (20%, disease) strain. The results suggest that dual strain-dependent pathways regulate EndMT: increased TGF-β yields low-strain EndMT and increased Wnt/β-catenin signaling yields high-strain EndMT. Furthermore, a surgical model of ischemic mitral regurgitation in adult sheep revealed elevated levels of α-SMA within the endothelium and interstitium, indicative of EndMT (122). After myocardial infarction, higher levels of collagen-producing α-SMA-positive cells in malfunctioning mitral valve subendothelium indicated a dramatically exaggerated EndMT process (123), which could be modulated by losartan without reducing adaptive growth (124).
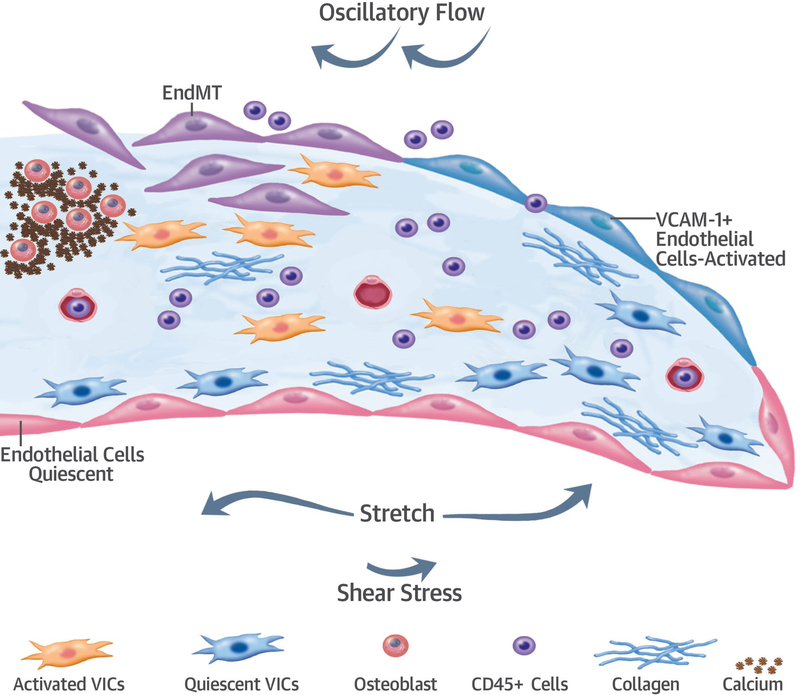
Figure 7. EndMT in heart valve disease.
In normal valves, valvular interstitial cells (VICs) possess a quiescent phenotype. VICs become activated in disease. EndMT in heart valves could be initiated by mechanical stress and inflammation (CD45-positive cells). Activated VCAM1-positive endothelium undergoes EndMT, which generates more interstitial cells. Some of these cells may undergo osteogenic transformation and activation resulting in various diseases.
Using clinically-relevant large animal models, collectively these reports suggest that: (i) EndMT participates in the initial adaptive response to an altered environment and may result in pathological processes such as fibrosis, leading to suboptimal valve function; and (ii) pro-inflammatory conditions and mechanical stress/strain might regulate EndMT in adult valves. Furthermore, these studies demonstrate that EndMT plays an important role in maintaining the phenotype of valvular cells in adults, and that certain environmental conditions may predispose valvular endothelium to enhanced EndMT.
Fibroelastosis
Endocardial fibroelastosis (EFE) is a rare disorder characterized by a unique fibrosis involving the ventricular endocardium which restricts ventricular growth in infants and children. EFE is typically associated with prenatal cardiac abnormalities, most notably in lesions with left heart obstruction including Barth and hypoplastic left heart syndrome (125). Often, the only therapeutic option is surgical univentricular palliation, which is associated with high mortality rates (126). Hence, EFE is of major clinical importance, yet the mechanisms underlying this disease are poorly understood.
Novel mouse models that mimic human EFE now permit studies of the origin of EFE tissues and their mechanisms of formation (127,128). As discussed, during development the endocardium undergoes EndMT to form the cardiac valves and septa (Figure 1). This indirectly suggests that, if aberrantly activated, the endocardium might also form the fibroelastic tissue found in EFE. Supporting this hypothesis, endothelial lineage tracking studies in EFE mice have shown that a proportion of EFE cells are derived via EndMT. Moreover, using immunofluorescence staining for endothelial and mesenchymal markers, EndMT was identified in human EFE tissues (129).
Interestingly, hypermethylation of BMP7 (an endogenous EndMT inhibitor) was found in human EFE tissues, and exogenous recombinant BMP7 was able to inhibit EndMT and EFE development in the mouse model (129), suggesting that drugs targeting epigenetic mechanisms (DNA methyltransferase inhibitors or DNA demethylase activators; see Epigenetics section) might by efficacious for preventing EFE.
Vein graft remodeling
Veins are commonly used conduits in arterial bypass graft surgery, however, 20–30% of vein grafts may fail within 12–18 months (130). Vein graft failure is largely due to adverse vascular remodeling, and the modulation of “early” activators of this process could be targeted to block the entire downstream complications that lead to graft failure (130). Cooley et al. (11) have shown that EndMT is important in vein graft remodeling and neointimal formation, which is the maladaptive smooth muscle cell hyperplasia that arises after a vein is exposed to arterial pressure. Specifically, with the adaptation to arterial pressure, they observed that ~50% of neointimal cells were EndMT-derived (11). EndMT-derived cells were found to be typical synthetic SMCs, expressing α-SMA and SM22α. EndMT in this setting was dependent on TGF-β signaling, with early activation of Smad2/3-Snai2. Correspondingly, antagonism of TGF-β signaling resulted in decreased EndMT and less neointimal formation. Cooley et al. (11) further identified that both Smad2 and Smad3 regulate Snai2, with Smad3 shown to directly bind the Snai2 promoter. Histological examination of postmortem human vein grafts corroborated these findings, suggesting that EndMT is operative during human vein graft remodeling (11).
Cardiac fibrosis
If there is a controversial aspect of EndMT, it is its contribution to cardiac fibrosis. In 2007 the first major publication emerged about EndMT in adult animals, suggesting that cardiac fibrosis was associated with EndMT (45). Employing a Tie1 Cre-lox endothelial lineage tracking system in a model of cardiac overload and fibrosis, cells that once expressed Tie1 (an endothelial marker) contributed to 27–33% of cardiac fibroblasts. The use of Smad3 deficient mice or administration of BMP7 inhibited EndMT and cardiac fibrosis in vivo (45). This study catalyzed significant interest in the field and subsequently, using cellular co-staining (65,88,92,131) and Cre-lox systems (132), others investigators recapitulated the finding that EndMT contributes to cardiac fibrosis. For example, Murdoch et al. used co-staining and changes in protein expression to conclude that EndMT is involved in cardiac fibrosis and diastolic dysfunction, which was mediated by endothelial nicotinamide adenine dinucleotide phosphate oxidase-2 (NOX2) activation (131). However, other studies have refuted these claims, suggesting that de novo EndMT plays little role in cardiac fibrosis in the adult (133,134). Adding complexity, a challenge faced by these studies is the need to distinguish between cardiac fibroblasts that are developmentally derived via EndMT from the endocardial cushions (for which there is consensus agreement (133,134)), versus de novo cardiac EndMT from adult ECs (where the controversy resides).
A potential explanation may lie in the fact that during EndMT in the adult, cells with a fully mature mesenchymal phenotype may be rarely achieved (8). In other words, as already mentioned, EndMT in the adult is likely associated with a partial transition to a mesenchymal-like phenotype, but not fully mature mesenchymal cells. This is consistent with recent studies in the kidney, where de novo EMT gave rise to partially transitioned fibroblast-like cells (4). Nevertheless, the fact the EndMT in the adult may be an incomplete process likely cannot account for all of the discrepancies in these studies, and further research is required to fully define the contribution (or not) of de novo EndMT to cardiac fibrosis in the adult.
Pulmonary hypertension
Primary pulmonary arterial hypertension (PAH) is a rare condition mediated by distal pulmonary vasculature vasoconstriction, aberrant vascular remodeling, vascular occlusions, and the formation of characteristic plexiform lesions (Figure 8) (135). In addition, endothelial dysfunction is a hallmark of PAH (135). Many cases of PAH are caused by BMP type II receptor gene (BMPR2) mutations, resulting in increased TGF-β signaling, including both non-canonical and canonical Smad-mediated signaling (135), although other rare variants are also implicated (136).
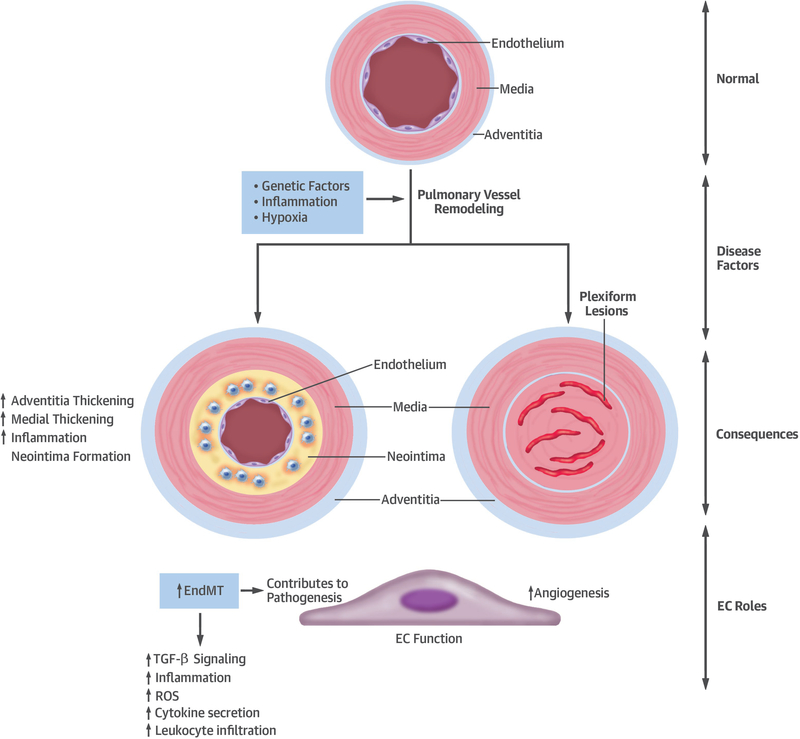
Figure 8. EndMT in PAH.
Key features of PAH and the role of EndMT.
EndMT was first identified in PAH based on in situ analyses of endothelial and mesenchymal markers, as well as an intervention study using rapamycin that reversed protein markers that are characteristic of EndMT (137). Further evidence demonstrated both the presence of EndMT in PAH and also the induction of EndMT by IL-1β, TGF-β and TNFα. Notably, EndMT-derived cells secreted high levels of cytokines and supported a greater extent of immune cell transmigration (138). An association of pathological mechanisms came with the finding that EndMT in PAH was related to high motility group AT-hook 1 (HMGA1), demonstrated through association of HMGA1 protein expression with cells undergoing EndMT. Interestingly and as a link to EndMT, HMGA1 expression was associated with reduced BMPR2 levels (139). Further mechanistic understanding has come through an association of EndMT in PAH with Twist expression and function (140). Moreover, a study of HIF-2α in human and experimental models has demonstrated a link with hypoxia, and mechanistically through induction of Snai1/2. Notably, endothelial loss of the prolyl hydroxylase domain protein 2 gene (this protein promotes HIF-2α degradation) led to severe PAH even in normoxia conditions (141). A separate study showed that loss of HIF-1α inhibited EndMT induction and normalized endothelial CD31 levels (142). Finally, a detailed characterization of pulmonary ECs undergoing EndMT has highlighted the contribution of the cells themselves, and also the paracrine signaling that such cells induce in the lung vasculature (143).
A growing list of other CVDs are also associated with EndMT. While in some cases the evidence is perhaps not as robust as the studies and diseases mentioned above, these are summarized in Table 2.
Table 2.
Additional disease states where EndMT has been implicated
| Disease | Potential role of EndMT | Reference |
|---|---|---|
| Fibrodysplasia ossificans progressiva | Murine lineage tracking and human cell characterization experiments showed an endothelial origin of osteoblasts and chonrdocytes via EndMT | (145) |
| Kidney fibrosis and kidney transplant failure | EndMT may participate in renal fibrosis | (146) |
| Cardiac transplant vasculopathy | Somewhat similar to atherosclerosis, EndMT may participate in cardiac transplant vasculopathy | (63) |
Concluding remarks and next steps
EndMT is involved in numerous CVDs, which collectively are a major cause of global morbidity and mortality. Hence, the manipulation of EndMT for therapeutic gain is a tantalizing prospect. Nevertheless, a number of obstacles remain to be overcome before the full therapeutic potential of manipulating EndMT can be realized, as described in Table 3. Undoubtedly, beyond these issues there are still further unknown challenges to be met and unforeseen obstacles to be resolved. However, with a collaborative and focused effort, we believe that over the next decade enormous advances can be made with respect to our understanding, and future manipulation of EndMT as a potential clinical therapy.
Table 3.
Major near-term obstacles and challenges that remain to be overcome in the investigation and clinical translation of EndMT.
| Challenge / Obstacle |
Comments | Solution |
|---|---|---|
| Lack of a robust functional and molecular definition of EndMT | The lack of a functional and molecular definition of EndMT is fostering scientific confusion, hampering research and interpretation, limiting comparability of data and facilitating the publication of studies with sub-optimal endpoints. | Research teams should work together to achieve a functional and molecular definition in the near term. This will require sharing of data, pooling and combined analyses of high-throughput datasets (i.e. RNA-sequencing, proteomics), and consensus agreement on definitions. This will be an ongoing process that will need refinement as further data and knowledge emerge. Embedded within this task is the understanding of additional molecular issues such as the reversibility of EndMT, or whether it is a clonal phenomenon. |
| Lack of understanding of the contribution of EndMT to disease causation (versus being a disease association or epi-phenomenon) | This problem is challenging to address in the human context, but genetic mouse models are well suited to this task. However, these studies require significant resources and meticulous scientific approaches that must be relevant to EndMT in human pathology. | We propose extensive, well designed and meticulously conducted genetic mouse studies, with validation and reproducibility achieved among collaborating laboratories. |
| Lack of robust human data on EndMT | While mouse studies are the most effective model system for rigorous proof of concept and for demonstrating causality, findings must be validated (ideally) in larger animals and (absolutely) in humans. Furthermore, some critical questions that drive our interest in EndMT, such as the role of EndMT in plaque erosion, can only be tackled in humans as there are no true animal models. | We propose detailed human studies using explanted and surplus surgical tissues from relevant disease states, with the application of cutting-edge techniques such as single cell RNA-sequencing to explore the contribution and extent of EndMT. Cross-validation among collaborating laboratories of key findings will be essential. |
| Lack of translational proof of concept | Few studies have attempted to manipulate EndMT in larger animals (122–124). While large animal translational proof-of-concept studies are clearly necessary, they require extensive resources and clear scientific objectives. A critical consideration is the existence of a suitable model, and whether regulatory authorities would require large animal studies before clinical trials could commence. Moreover, this may be disease- and context-specific, because EndMT appears relevant across a range of CVDs. | Consortia should be formed with a view to prioritizing EndMT targets in disease states amenable to large animal models, with a view to systematically studying the utility of manipulating EndMT for therapeutic gain. It will be essential to engage with regulatory authorities to assess need and appropriate nature of such models in the functional disease context. |
Acknowledgements:
Jason Kovacic acknowledges research support from the National Institutes of Health (R01HL130423), the American Heart Association (14SFRN20490315; 14SFRN20840000) and The Leducq Foundation (Transatlantic Network of Excellence Award). Stefanie Dimmeler is supported by the German Research Foundation (SFB834, Project B5), the LOEWE Center for Cell- and Gene Therapy (State of Hesse) and the ERC Advanced Grant Angiolnc. Richard Harvey acknowledges research support from the National Health and Medical Research Council of Australia (NHMRC; APP1118576, 1074386), the Australian Research Counsel (ARC; DP160104858, SR110001002), Foundation Leducq Transatlantic Networks of Excellence in Cardiovascular Research (15 CVD 03, 13 CVD 01) and the New South Wales Government Department of Health. Elena Aikawa is supported by National Institutes of Health grants R01HL114805, R01HL141917 and R01HL136431. Guido Krenning acknowledges research support from the Netherlands Organization for Health Research and Development (917.16.446) and the Dutch Kidney Foundation (15OP13). Toren Finkel is supported by the Leducq Foundation (Transatlantic Network of Excellence Award) and the Progeria Research Foundation. Andrew Baker is supported by the BHF Chair of Translational Cardiovascular Sciences and grants RG/14/3/30706 and ERC Advanced Grant VASCMIR.
Abbreviations
- α-SMA
α-smooth muscle actin
- AV
atrioventricular
- BMP
bone morphogenetic protein
- BMPRII
bone morphogenetic protein type II receptor
- circRNA
circular RNA
- CVD
cardiovascular disease
- Dnmt
DNA methyltransferase
- EC
endothelial cell
- ECM
extracellular matrix
- EFE
endocardial fibroelastosis
- EMT
epithelial to mesenchymal transition
- EndMT
endothelial to mesenchymal transition
- FAO
fatty acid oxidation
- FGF
fibroblast growth factor
- GRB2
growth factor receptor-bound 2
- IL
interleukin
- lncRNA
long non-coding RNA
- LOXL2
Lysyl oxidase homolog 2
- mRNA
messenger RNA
- miRNA
micro RNA
- ncRNA
non-protein coding RNAs
- NOS3
nitric oxide synthase 3 – also termed endothelial nitric oxide synthase
- PAH
pulmonary arterial hypertension
- TGF-β(R)
transforming growth factor-β (receptor)
Footnotes
Disclosures:
The authors declare no conflicts of interest.
References
- 1.Hamburger V Introduction: Johannes Holtfreter, pioneer in experimental embryology. Dev Dyn 1996;205:214–6. [DOI] [PubMed] [Google Scholar]
- 2.Trelstad RL, Hay ED, Revel JD. Cell contact during early morphogenesis in the chick embryo. Dev Biol 1967;16:78–106. [DOI] [PubMed] [Google Scholar]
- 3.Kovacic JC, Mercader N, Torres M, Boehm M, Fuster V. Epithelial-to-mesenchymal and endothelial-to-mesenchymal transition: from cardiovascular development to disease. Circulation 2012;125:1795–808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grande MT, Sanchez-Laorden B, Lopez-Blau C et al. Snail1-induced partial epithelial-to-mesenchymal transition drives renal fibrosis in mice and can be targeted to reverse established disease. Nat Med 2015;21:989–97. [DOI] [PubMed] [Google Scholar]
- 5.Santamaria PG, Moreno-Bueno G, Portillo F, Cano A. EMT: Present and future in clinical oncology. Molecular oncology 2017;11:718–738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fan LC, Teng HW, Shiau CW et al. Regorafenib (Stivarga) pharmacologically targets epithelial-mesenchymal transition in colorectal cancer. Oncotarget 2016;7:64136–64147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xiong J, Kawagishi H, Yan Y et al. A Metabolic Basis for Endothelial-to-Mesenchymal Transition. Mol Cell 2018;69:689–698 e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Evrard SM, Lecce L, Michelis KC et al. Endothelial to mesenchymal transition is common in atherosclerotic lesions and is associated with plaque instability. Nat Commun 2016;7:11853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Souilhol C, Harmsen MC, Evans PC, Krenning G. Endothelial-mesenchymal transition in atherosclerosis. Cardiovasc Res 2018;114:565–577. [DOI] [PubMed] [Google Scholar]
- 10.Chen PY, Qin L, Baeyens N et al. Endothelial-to-mesenchymal transition drives atherosclerosis progression. J Clin Invest 2015;125:4514–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cooley BC, Nevado J, Mellad J et al. TGF-beta signaling mediates endothelial-to-mesenchymal transition (EndMT) during vein graft remodeling. Sci Transl Med 2014;6:227ra34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Manavski Y, Lucas T, Glaser SF et al. Clonal Expansion of Endothelial Cells Contributes to Ischemia-Induced Neovascularization. Circ Res 2018;122:670–677. [DOI] [PubMed] [Google Scholar]
- 13.Dejana E, Hirschi KK, Simons M. The molecular basis of endothelial cell plasticity. Nat Commun 2017;8:14361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Krenning G, Moonen JR, van Luyn MJ, Harmsen MC. Vascular smooth muscle cells for use in vascular tissue engineering obtained by endothelial-to-mesenchymal transdifferentiation (EnMT) on collagen matrices. Biomaterials 2008;29:3703–11. [DOI] [PubMed] [Google Scholar]
- 15.Moonen JR, Krenning G, Brinker MG, Koerts JA, van Luyn MJ, Harmsen MC. Endothelial progenitor cells give rise to pro-angiogenic smooth muscle-like progeny. Cardiovasc Res 2010;86:506–15. [DOI] [PubMed] [Google Scholar]
- 16.Welch-Reardon KM, Ehsan SM, Wang K et al. Angiogenic sprouting is regulated by endothelial cell expression of Slug. J Cell Sci 2014;127:2017–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Milgrom-Hoffman M, Harrelson Z, Ferrara N, Zelzer E, Evans SM, Tzahor E. The heart endocardium is derived from vascular endothelial progenitors. Development 2011;138:4777–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paffett-Lugassy N, Singh R, Nevis KR et al. Heart field origin of great vessel precursors relies on nkx2.5-mediated vasculogenesis. Nat Cell Biol 2013;15:1362–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zamir L, Singh R, Nathan E et al. Nkx2.5 marks angioblasts that contribute to hemogenic endothelium of the endocardium and dorsal aorta. Elife 2017;6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hutson MR, Zeng XL, Kim AJ, Antoon E, Harward S, Kirby ML. Arterial pole progenitors interpret opposing FGF/BMP signals to proliferate or differentiate. Development 2010;137:3001–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kattman SJ, Huber TL, Keller GM. Multipotent flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev Cell 2006;11:723–32. [DOI] [PubMed] [Google Scholar]
- 22.Moretti A, Caron L, Nakano A et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 2006;127:1151–65. [DOI] [PubMed] [Google Scholar]
- 23.Gordon-Keylock S, Sobiesiak M, Rybtsov S, Moore K, Medvinsky A. Mouse extraembryonic arterial vessels harbor precursors capable of maturing into definitive HSCs. Blood 2013;122:2338–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.del Monteo-Nieto G, Ramialison M, Adam AAS et al. Control of cardiac jelly dynamics by NOTCH1 and NRG1 defines the building plan for trabeculation. Nature 2018;557:439–45. [DOI] [PubMed] [Google Scholar]
- 25.Red-Horse K, Ueno H, Weissman IL, Krasnow MA. Coronary arteries form by developmental reprogramming of venous cells. Nature 2010;464:549–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen HI, Sharma B, Akerberg BN et al. The sinus venosus contributes to coronary vasculature through VEGFC-stimulated angiogenesis. Development 2014;141:4500–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sharma B, Ho L, Ford GH et al. Alternative Progenitor Cells Compensate to Rebuild the Coronary Vasculature in Elabela- and Apj-Deficient Hearts. Dev Cell 2017;42:655–666 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Harrison MR, Bussmann J, Huang Y et al. Chemokine-guided angiogenesis directs coronary vasculature formation in zebrafish. Dev Cell 2015;33:442–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tian X, Hu T, Zhang H et al. Vessel formation. De novo formation of a distinct coronary vascular population in neonatal heart. Science 2014;345:90–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Y, Wu B, Lu P et al. Uncontrolled angiogenic precursor expansion causes coronary artery anomalies in mice lacking Pofut1. Nat Commun 2017;8:578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yzaguirre AD, Padmanabhan A, de Groh ED et al. Loss of neurofibromin Ras-GAP activity enhances the formation of cardiac blood islands in murine embryos. Elife 2015;4:e07780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen Q, Zhang H, Liu Y et al. Endothelial cells are progenitors of cardiac pericytes and vascular smooth muscle cells. Nat Commun 2016;7:12422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang H, Pu W, Liu Q et al. Endocardium Contributes to Cardiac Fat. Circ Res 2016;118:254–65. [DOI] [PubMed] [Google Scholar]
- 34.Camenisch TD, Runyan RB, Markwald RR. Molecular Regulation of Cushion Morphogenesis In: Rosenthal N, Harvey RP, editors. Heart Development and Regeneration. London: Academic Press, 2010:363–387. [Google Scholar]
- 35.de Vlaming A, Sauls K, Hajdu Z et al. Atrioventricular valve development: new perspectives on an old theme. Differentiation 2012;84:103–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang H, Lui KO, Zhou B. Endocardial Cell Plasticity in Cardiac Development, Diseases and Regeneration. Circ Res 2018;122:774–789. [DOI] [PubMed] [Google Scholar]
- 37.Snarr BS, Kern CB, Wessels A. Origin and fate of cardiac mesenchyme. Dev Dyn 2008;237:2804–19. [DOI] [PubMed] [Google Scholar]
- 38.Wessels A, van den Hoff MJ, Adamo RF et al. Epicardially derived fibroblasts preferentially contribute to the parietal leaflets of the atrioventricular valves in the murine heart. Dev Biol 2012;366:111–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Goddard LM, Duchemin AL, Ramalingan H et al. Hemodynamic Forces Sculpt Developing Heart Valves through a KLF2-WNT9B Paracrine Signaling Axis. Dev Cell 2017;43:274–289 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.von Gise A, Pu WT. Endocardial and epicardial epithelial to mesenchymal transitions in heart development and disease. Circ Res 2012;110:1628–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang H, von Gise A, Liu Q et al. Yap1 is required for endothelial to mesenchymal transition of the atrioventricular cushion. J Biol Chem 2014;289:18681–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Luxan G, D’Amato G, MacGrogan D, de la Pompa JL. Endocardial Notch Signaling in Cardiac Development and Disease. Circ Res 2016;118:e1–e18. [DOI] [PubMed] [Google Scholar]
- 43.MacGrogan D, D’Amato G, Travisano S et al. Sequential Ligand-Dependent Notch Signaling Activation Regulates Valve Primordium Formation and Morphogenesis. Circ Res 2016;118:1480–97. [DOI] [PubMed] [Google Scholar]
- 44.Lincoln J, Lange AW, Yutzey KE. Hearts and bones: shared regulatory mechanisms in heart valve, cartilage, tendon, and bone development. Dev Biol 2006;294:292–302. [DOI] [PubMed] [Google Scholar]
- 45.Zeisberg EM, Tarnavski O, Zeisberg M et al. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med 2007;13:952–61. [DOI] [PubMed] [Google Scholar]
- 46.Hata A, Chen YG. TGF-beta Signaling from Receptors to Smads. Cold Spring Harb Perspect Biol 2016;8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lebrin F, Deckers M, Bertolino P, Ten Dijke P. TGF-beta receptor function in the endothelium. Cardiovasc Res 2005;65:599–608. [DOI] [PubMed] [Google Scholar]
- 48.Pardali E, Sanchez-Duffhues G, Gomez-Puerto MC, Ten Dijke P. TGF-beta-Induced Endothelial-Mesenchymal Transition in Fibrotic Diseases. Int J Mol Sci 2017;18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Derynck R, Zhang YE. Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 2003;425:577–84. [DOI] [PubMed] [Google Scholar]
- 50.Rodriguez-Garcia A, Samso P, Fontova P et al. TGF-beta1 targets Smad, p38 MAPK, and PI3K/Akt signaling pathways to induce PFKFB3 gene expression and glycolysis in glioblastoma cells. FEBS J 2017;284:3437–3454. [DOI] [PubMed] [Google Scholar]
- 51.Jung MY, Kang JH, Hernandez DM et al. Fatty acid synthase is required for profibrotic TGF-beta signaling. FASEB J 2018:fj201701187R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Guo Q Changes in mitochondrial function during EMT induced by TGFbeta-1 in pancreatic cancer. Oncol Lett 2017;13:1575–1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sivanand S, Viney I, Wellen KE. Spatiotemporal Control of Acetyl-CoA Metabolism in Chromatin Regulation. Trends Biochem Sci 2018;43:61–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Piera-Velazquez S, Mendoza FA, Jimenez SA. Endothelial to Mesenchymal Transition (EndoMT) in the Pathogenesis of Human Fibrotic Diseases. J Clin Med 2016;5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kang HM, Ahn SH, Choi P et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat Med 2015;21:37–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ghosh AK, Nagpal V, Covington JW, Michaels MA, Vaughan DE. Molecular basis of cardiac endothelial-to-mesenchymal transition (EndMT): differential expression of microRNAs during EndMT. Cell Signal 2012;24:1031–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bayoumi AS, Teoh JP, Aonuma T et al. MicroRNA-532 protects the heart in acute myocardial infarction, and represses prss23, a positive regulator of endothelial-to-mesenchymal transition. Cardiovasc Res 2017;113:1603–1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cao Y, Feng B, Chen S, Chu Y, Chakrabarti S. Mechanisms of endothelial to mesenchymal transition in the retina in diabetes. Invest Ophthalmol Vis Sci 2014;55:7321–31. [DOI] [PubMed] [Google Scholar]
- 59.Chen PY, Qin L, Barnes C et al. FGF regulates TGF-beta signaling and endothelial-to-mesenchymal transition via control of let-7 miRNA expression. Cell Rep 2012;2:1684–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Correia AC, Moonen JR, Brinker MG, Krenning G. FGF2 inhibits endothelial-mesenchymal transition through microRNA-20a-mediated repression of canonical TGF-beta signaling. J Cell Sci 2016;129:569–79. [DOI] [PubMed] [Google Scholar]
- 61.Sun Y, Cai J, Yu S, Chen S, Li F, Fan C. MiR-630 Inhibits Endothelial-Mesenchymal Transition by Targeting Slug in Traumatic Heterotopic Ossification. Sci Rep 2016;6:22729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang H, Hu J, Liu L. MiR-200a modulates TGF-beta1-induced endothelial-to-mesenchymal shift via suppression of GRB2 in HAECs. Biomed Pharmacother 2017;95:215–222. [DOI] [PubMed] [Google Scholar]
- 63.Chen PY, Qin L, Tellides G, Simons M. Fibroblast growth factor receptor 1 is a key inhibitor of TGFbeta signaling in the endothelium. Sci Signal 2014;7:ra90. [DOI] [PubMed] [Google Scholar]
- 64.Zhang S, Weinheimer C, Courtois M et al. The role of the Grb2-p38 MAPK signaling pathway in cardiac hypertrophy and fibrosis. J Clin Invest 2003;111:833–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kumarswamy R, Volkmann I, Jazbutyte V, Dangwal S, Park DH, Thum T. Transforming growth factor-beta-induced endothelial-to-mesenchymal transition is partly mediated by microRNA-21. Arterioscler Thromb Vasc Biol 2012;32:361–9. [DOI] [PubMed] [Google Scholar]
- 66.Suzuki HI, Katsura A, Mihira H, Horie M, Saito A, Miyazono K. Regulation of TGF-beta-mediated endothelial-mesenchymal transition by microRNA-27. J Biochem 2017;161:417–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wang J, He W, Xu X et al. The mechanism of TGF-beta/miR-155/c-Ski regulates endothelial-mesenchymal transition in human coronary artery endothelial cells. Biosci Rep 2017;37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Suzuki H, Yagi K, Kondo M, Kato M, Miyazono K, Miyazawa K. c-Ski inhibits the TGF-beta signaling pathway through stabilization of inactive Smad complexes on Smad-binding elements. Oncogene 2004;23:5068–76. [DOI] [PubMed] [Google Scholar]
- 69.Wang Z, Wang DZ, Hockemeyer D, McAnally J, Nordheim A, Olson EN. Myocardin and ternary complex factors compete for SRF to control smooth muscle gene expression. Nature 2004;428:185–9. [DOI] [PubMed] [Google Scholar]
- 70.Sharma V, Dogra N, Saikia UN, Khullar M. Transcriptional regulation of endothelial-to-mesenchymal transition in cardiac fibrosis: role of myocardin-related transcription factor A and activating transcription factor 3. Can J Physiol Pharmacol 2017;95:1263–1270. [DOI] [PubMed] [Google Scholar]
- 71.Mihira H, Suzuki HI, Akatsu Y et al. TGF-beta-induced mesenchymal transition of MS-1 endothelial cells requires Smad-dependent cooperative activation of Rho signals and MRTF-A. J Biochem 2012;151:145–56. [DOI] [PubMed] [Google Scholar]
- 72.Meadows KN, Iyer S, Stevens MV et al. Akt promotes endocardial-mesenchyme transition. J Angiogenes Res 2009;1:2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kim D, Kim S, Koh H et al. Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J 2001;15:1953–62. [DOI] [PubMed] [Google Scholar]
- 74.Park BK, Zeng X, Glazer RI. Akt1 induces extracellular matrix invasion and matrix metalloproteinase-2 activity in mouse mammary epithelial cells. Cancer Res 2001;61:7647–53. [PubMed] [Google Scholar]
- 75.Navaratna D, McGuire PG, Menicucci G, Das A. Proteolytic degradation of VE-cadherin alters the blood-retinal barrier in diabetes. Diabetes 2007;56:2380–7. [DOI] [PubMed] [Google Scholar]
- 76.Hamada K, Sasaki T, Koni PA et al. The PTEN/PI3K pathway governs normal vascular development and tumor angiogenesis. Genes Dev 2005;19:2054–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Neumann P, Jae N, Knau A et al. The lncRNA GATA6-AS epigenetically regulates endothelial gene expression via interaction with LOXL2. Nat Commun 2018;9:237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Xiang Y, Zhang Y, Tang Y, Li Q. MALAT1 Modulates TGF-beta1-Induced Endothelial-to-Mesenchymal Transition through Downregulation of miR-145. Cell Physiol Biochem 2017;42:357–372. [DOI] [PubMed] [Google Scholar]
- 79.Meng S, Zhou H, Feng Z et al. CircRNA: functions and properties of a novel potential biomarker for cancer. Mol Cancer 2017;16:94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Conn SJ, Pillman KA, Toubia J et al. The RNA binding protein quaking regulates formation of circRNAs. Cell 2015;160:1125–34. [DOI] [PubMed] [Google Scholar]
- 81.Lee JY, Kong G. Roles and epigenetic regulation of epithelial-mesenchymal transition and its transcription factors in cancer initiation and progression. Cell Mol Life Sci 2016;73:4643–4660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kumar A, Kumar S, Vikram A et al. Histone and DNA methylation-mediated epigenetic downregulation of endothelial Kruppel-like factor 2 by low-density lipoprotein cholesterol. Arterioscler Thromb Vasc Biol 2013;33:1936–42. [DOI] [PubMed] [Google Scholar]
- 83.Dunn J, Thabet S, Jo H. Flow-Dependent Epigenetic DNA Methylation in Endothelial Gene Expression and Atherosclerosis. Arterioscler Thromb Vasc Biol 2015;35:1562–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jiang YZ, Jimenez JM, Ou K, McCormick ME, Zhang LD, Davies PF. Hemodynamic disturbed flow induces differential DNA methylation of endothelial Kruppel-Like Factor 4 promoter in vitro and in vivo. Circ Res 2014;115:32–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Man HS, Yan MS, Lee JJ, Marsden PA. Epigenetic determinants of cardiovascular gene expression: vascular endothelium. Epigenomics 2016;8:959–79. [DOI] [PubMed] [Google Scholar]
- 86.Ohtani K, Vlachojannis GJ, Koyanagi M et al. Epigenetic regulation of endothelial lineage committed genes in pro-angiogenic hematopoietic and endothelial progenitor cells. Circ Res 2011;109:1219–29. [DOI] [PubMed] [Google Scholar]
- 87.Bjorck HM, Du L, Pulignani S et al. Altered DNA methylation indicates an oscillatory flow mediated epithelial-to-mesenchymal transition signature in ascending aorta of patients with bicuspid aortic valve. Sci Rep 2018;8:2777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Xu X, Tan X, Tampe B et al. Epigenetic balance of aberrant Rasal1 promoter methylation and hydroxymethylation regulates cardiac fibrosis. Cardiovasc Res 2015;105:279–91. [DOI] [PubMed] [Google Scholar]
- 89.Maleszewska M, Gjaltema RA, Krenning G, Harmsen MC. Enhancer of zeste homolog-2 (EZH2) methyltransferase regulates transgelin/smooth muscle-22alpha expression in endothelial cells in response to interleukin-1beta and transforming growth factor-beta2. Cell Signal 2015;27:1589–96. [DOI] [PubMed] [Google Scholar]
- 90.Zeng L, Wang G, Ummarino D et al. Histone deacetylase 3 unconventional splicing mediates endothelial-to-mesenchymal transition through transforming growth factor beta2. J Biol Chem 2013;288:31853–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Yu CH, Suriguga Gong M et al. High glucose induced endothelial to mesenchymal transition in human umbilical vein endothelial cell. Exp Mol Pathol 2017;102:377–383. [DOI] [PubMed] [Google Scholar]
- 92.Widyantoro B, Emoto N, Nakayama K et al. Endothelial cell-derived endothelin-1 promotes cardiac fibrosis in diabetic hearts through stimulation of endothelial-to-mesenchymal transition. Circulation 2010;121:2407–18. [DOI] [PubMed] [Google Scholar]
- 93.Tang RN, Lv LL, Zhang JD et al. Effects of angiotensin II receptor blocker on myocardial endothelial-to-mesenchymal transition in diabetic rats. Int J Cardiol 2013;162:92–9. [DOI] [PubMed] [Google Scholar]
- 94.Ma J, Liu T, Dong X. Advanced glycation end products of bovine serum albumin-induced endothelial-to-mesenchymal transition in cultured human and monkey endothelial cells via protein kinase B signaling cascades. Mol Vis 2010;16:2669–79. [PMC free article] [PubMed] [Google Scholar]
- 95.Niessen K, Fu Y, Chang L, Hoodless PA, McFadden D, Karsan A. Slug is a direct Notch target required for initiation of cardiac cushion cellularization. J Cell Biol 2008;182:315–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Reichman D, Man L, Park L et al. Notch hyper-activation drives trans-differentiation of hESC-derived endothelium. Stem Cell Res 2016;17:391–400. [DOI] [PubMed] [Google Scholar]
- 97.Tian Y, Xu Y, Fu Q et al. Notch inhibits chondrogenic differentiation of mesenchymal progenitor cells by targeting Twist1. Mol Cell Endocrinol 2015;403:30–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Conacci-Sorrell M, Simcha I, Ben-Yedidia T, Blechman J, Savagner P, Ben-Ze’ev A. Autoregulation of E-cadherin expression by cadherin-cadherin interactions: the roles of beta-catenin signaling, Slug, and MAPK. J Cell Biol 2003;163:847–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Liebner S, Cattelino A, Gallini R et al. Beta-catenin is required for endothelial-mesenchymal transformation during heart cushion development in the mouse. J Cell Biol 2004;166:359–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Li J, Zhang Q, Ren C et al. Low-Intensity Pulsed Ultrasound Prevents the Oxidative Stress Induced Endothelial-Mesenchymal Transition in Human Aortic Endothelial Cells. Cell Physiol Biochem 2018;45:1350–1365. [DOI] [PubMed] [Google Scholar]
- 101.O’Riordan E, Mendelev N, Patschan S et al. Chronic NOS inhibition actuates endothelial-mesenchymal transformation. American journal of physiology Heart and circulatory physiology 2007;292:H285–94. [DOI] [PubMed] [Google Scholar]
- 102.Liang X, Duan N, Wang Y et al. Advanced oxidation protein products induce endothelial-to-mesenchymal transition in human renal glomerular endothelial cells through induction of endoplasmic reticulum stress. J Diabetes Complications 2016;30:573–9. [DOI] [PubMed] [Google Scholar]
- 103.Ma Z, Zhu L, Liu Y et al. Lovastatin Alleviates Endothelial-to-Mesenchymal Transition in Glomeruli via Suppression of Oxidative Stress and TGF-beta1 Signaling. Front Pharmacol 2017;8:473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ichise T, Yoshida N, Ichise H. FGF2-induced Ras-MAPK signalling maintains lymphatic endothelial cell identity by upregulating endothelial-cell-specific gene expression and suppressing TGFbeta signalling through Smad2. J Cell Sci 2014;127:845–57. [DOI] [PubMed] [Google Scholar]
- 105.Goumans MJ, Valdimarsdottir G, Itoh S, Rosendahl A, Sideras P, ten Dijke P. Balancing the activation state of the endothelium via two distinct TGF-beta type I receptors. EMBO J 2002;21:1743–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ling F, Kang B, Sun XH. Id proteins: small molecules, mighty regulators. Curr Top Dev Biol 2014;110:189–216. [DOI] [PubMed] [Google Scholar]
- 107.Stankic M, Pavlovic S, Chin Y et al. TGF-beta-Id1 signaling opposes Twist1 and promotes metastatic colonization via a mesenchymal-to-epithelial transition. Cell Rep 2013;5:1228–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Moonen JR, Lee ES, Schmidt M et al. Endothelial-to-mesenchymal transition contributes to fibro-proliferative vascular disease and is modulated by fluid shear stress. Cardiovasc Res 2015;108:377–86. [DOI] [PubMed] [Google Scholar]
- 109.Mahmoud MM, Serbanovic-Canic J, Feng S et al. Shear stress induces endothelial-to-mesenchymal transition via the transcription factor Snail. Sci Rep 2017;7:3375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mahmoud MM, Kim HR, Xing R et al. TWIST1 Integrates Endothelial Responses to Flow in Vascular Dysfunction and Atherosclerosis. Circ Res 2016;119:450–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Spillmann F, Miteva K, Pieske B, Tschope C, Van Linthout S. High-density lipoproteins reduce endothelial-to-mesenchymal transition. Arterioscler Thromb Vasc Biol 2015;35:1774–7. [DOI] [PubMed] [Google Scholar]
- 112.Dai J, Xing L, Jia H et al. In vivo predictors of plaque erosion in patients with ST-segment elevation myocardial infarction: a clinical, angiographical, and intravascular optical coherence tomography study. Eur Heart J 2018. [DOI] [PubMed] [Google Scholar]
- 113.Paruchuri S, Yang JH, Aikawa E et al. Human pulmonary valve progenitor cells exhibit endothelial/mesenchymal plasticity in response to vascular endothelial growth factor-A and transforming growth factor-beta2. Circ Res 2006;99:861–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Aikawa E, Whittaker P, Farber M et al. Human semilunar cardiac valve remodeling by activated cells from fetus to adult: implications for postnatal adaptation, pathology, and tissue engineering. Circulation 2006;113:1344–52. [DOI] [PubMed] [Google Scholar]
- 115.Wirrig EE, Yutzey KE. Conserved transcriptional regulatory mechanisms in aortic valve development and disease. Arterioscler Thromb Vasc Biol 2014;34:737–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Rabkin E, Aikawa M, Stone JR, Fukumoto Y, Libby P, Schoen FJ. Activated interstitial myofibroblasts express catabolic enzymes and mediate matrix remodeling in myxomatous heart valves. Circulation 2001;104:2525–32. [DOI] [PubMed] [Google Scholar]
- 117.Yutzey KE, Demer LL, Body SC et al. Calcific aortic valve disease: a consensus summary from the Alliance of Investigators on Calcific Aortic Valve Disease. Arterioscler Thromb Vasc Biol 2014;34:2387–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Zhou J, Bowen C, Lu G et al. Cadherin-11 expression patterns in heart valves associate with key functions during embryonic cushion formation, valve maturation and calcification. Cells Tissues Organs 2013;198:300–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wylie-Sears J, Aikawa E, Levine RA, Yang JH, Bischoff J. Mitral valve endothelial cells with osteogenic differentiation potential. Arterioscler Thromb Vasc Biol 2011;31:598–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Hjortnaes J, Shapero K, Goettsch C et al. Valvular interstitial cells suppress calcification of valvular endothelial cells. Atherosclerosis 2015;242:251–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Balachandran K, Alford PW, Wylie-Sears J et al. Cyclic strain induces dual-mode endothelial-mesenchymal transformation of the cardiac valve. Proc Natl Acad Sci U S A 2011;108:19943–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Dal-Bianco JP, Aikawa E, Bischoff J et al. Active adaptation of the tethered mitral valve: insights into a compensatory mechanism for functional mitral regurgitation. Circulation 2009;120:334–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Dal-Bianco JP, Aikawa E, Bischoff J et al. Myocardial Infarction Alters Adaptation of the Tethered Mitral Valve. J Am Coll Cardiol 2016;67:275–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Bartko PE, Dal-Bianco JP, Guerrero JL et al. Effect of Losartan on Mitral Valve Changes After Myocardial Infarction. J Am Coll Cardiol 2017;70:1232–1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Seki A, Patel S, Ashraf S, Perens G, Fishbein MC. Primary endocardial fibroelastosis: an underappreciated cause of cardiomyopathy in children. Cardiovasc Pathol 2013;22:345–50. [DOI] [PubMed] [Google Scholar]
- 126.d’Udekem Y, Xu MY, Galati JC et al. Predictors of survival after single-ventricle palliation: the impact of right ventricular dominance. J Am Coll Cardiol 2012;59:1178–85. [DOI] [PubMed] [Google Scholar]
- 127.Friehs I, Illigens B, Melnychenko I, Zhong-Hu T, Zeisberg E, Del Nido PJ. An animal model of endocardial fibroelastosis. J Surg Res 2013;182:94–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Shimada S, Robles C, Illigens BM, Casar Berazaluce AM, del Nido PJ, Friehs I. Distention of the Immature Left Ventricle Triggers Development of Endocardial Fibroelastosis: An Animal Model of Endocardial Fibroelastosis Introducing Morphopathological Features of Evolving Fetal Hypoplastic Left Heart Syndrome. Biomed Res Int 2015;2015:462469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Xu X, Friehs I, Zhong Hu T et al. Endocardial fibroelastosis is caused by aberrant endothelial to mesenchymal transition. Circ Res 2015;116:857–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wan S, George SJ, Berry C, Baker AH. Vein graft failure: current clinical practice and potential for gene therapeutics. Gene Ther 2012;19:630–6. [DOI] [PubMed] [Google Scholar]
- 131.Murdoch CE, Chaubey S, Zeng L et al. Endothelial NADPH oxidase-2 promotes interstitial cardiac fibrosis and diastolic dysfunction through proinflammatory effects and endothelial-mesenchymal transition. J Am Coll Cardiol 2014;63:2734–41. [DOI] [PubMed] [Google Scholar]
- 132.Jeong D, Lee MA, Li Y et al. Matricellular Protein CCN5 Reverses Established Cardiac Fibrosis. J Am Coll Cardiol 2016;67:1556–1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ali SR, Ranjbarvaziri S, Talkhabi M et al. Developmental heterogeneity of cardiac fibroblasts does not predict pathological proliferation and activation. Circ Res 2014;115:625–35. [DOI] [PubMed] [Google Scholar]
- 134.Moore-Morris T, Guimaraes-Camboa N, Banerjee I et al. Resident fibroblast lineages mediate pressure overload-induced cardiac fibrosis. J Clin Invest 2014;124:2921–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Humbert M, Morrell NW, Archer SL et al. Cellular and molecular pathobiology of pulmonary arterial hypertension. J Am Coll Cardiol 2004;43:13S–24S. [DOI] [PubMed] [Google Scholar]
- 136.Graf S, Haimel M, Bleda M et al. Identification of rare sequence variation underlying heritable pulmonary arterial hypertension. Nat Commun 2018;9:1416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ranchoux B, Antigny F, Rucker-Martin C et al. Endothelial-to-mesenchymal transition in pulmonary hypertension. Circulation 2015;131:1006–18. [DOI] [PubMed] [Google Scholar]
- 138.Good RB, Gilbane AJ, Trinder SL et al. Endothelial to Mesenchymal Transition Contributes to Endothelial Dysfunction in Pulmonary Arterial Hypertension. Am J Pathol 2015;185:1850–8. [DOI] [PubMed] [Google Scholar]
- 139.Hopper RK, Moonen JR, Diebold I et al. In Pulmonary Arterial Hypertension, Reduced BMPR2 Promotes Endothelial-to-Mesenchymal Transition via HMGA1 and Its Target Slug. Circulation 2016;133:1783–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mammoto T, Muyleart M, Konduri GG, Mammoto A. Twist1 in Hypoxia-induced Pulmonary Hypertension through Transforming Growth Factor-beta-Smad Signaling. Am J Respir Cell Mol Biol 2018;58:194–207. [DOI] [PubMed] [Google Scholar]
- 141.Tang H, Babicheva A, McDermott KM et al. Endothelial HIF-2alpha contributes to severe pulmonary hypertension due to endothelial-to-mesenchymal transition. Am J Physiol Lung Cell Mol Physiol 2018;314:L256–L275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zhang B, Niu W, Dong HY, Liu ML, Luo Y, Li ZC. Hypoxia induces endothelialmesenchymal transition in pulmonary vascular remodeling. Int J Mol Med 2018;42:270–278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Suzuki T, Carrier EJ, Talati MH et al. Isolation and characterization of endothelial-to-mesenchymal transition cells in pulmonary arterial hypertension. Am J Physiol Lung Cell Mol Physiol 2018;314:L118–L126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kovacic JC. The Endothelial-Metabolic Axis: A Novel Cardiometabolic Disease Target. Trends Endocrinol Metab 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Medici D, Shore EM, Lounev VY, Kaplan FS, Kalluri R, Olsen BR. Conversion of vascular endothelial cells into multipotent stem-like cells. Nat Med 2010;16:1400–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Wang Z, Han Z, Tao J et al. Role of endothelial-to-mesenchymal transition induced by TGF-beta1 in transplant kidney interstitial fibrosis. J Cell Mol Med 2017;21:2359–2369. [DOI] [PMC free article] [PubMed] [Google Scholar]