Abstract
Background
Cord Blood (CB) has been considered a promising source of natural killer (NK) cells for cellular immunotherapy. However, it is difficult to expand the large numbers of highly pure NK cells from CB without cell sorting and feeder cells/multiple cytokines. In this study, we try to develop a simple, safe and economical method for ex vivo expansion and purification of NK cells from CB without cell sorting and feeder cells/multiple cytokines.
Results
The large numbers (mean: 1.59 × 1010) of highly pure (≥90%) NK cells from CB could be obtained through interleukin-2, group A streptococcus and zoledronate stimulation of mononuclear cells using the 21-day culture approach. When compared to resting NK cells, expanded NK cells were a higher expression of activating receptors CD16, NKG2D, NKp30, NKp44, NKp46 and activating markers CD62L and CD69, while the inhibitory receptors, CD158a and CD158b remained largely unchanged. In addition, these cells showed a higher concentration of IFN-γ, TNF-α and GM-CSF secretion and cytotoxicity to K562 cells and acute myeloid leukemia targets than resting NK cells.
Conclusion
We develop a simple, safe and economical method to obtain high yield, purity, and functionality NK cells from CB without cell sorting and feeder cells/multiple cytokines.
Keywords: Cord blood, Natural killer cells, Expansion, Cytotoxicity, Immunotherapy
Background
Allogeneic natural killer (NK) cell infusion is promising for cancer immunotherapy because of the “missing self” hypothesis [1]. Cord blood (CB), serves as an immediate “off-the-shelf” source of NK cells, has been considered an attractive source of allogeneic NK cells for therapeutic infusion [2, 3]. However, a major challenge of cell therapy with NK cells is to attain sufficient amount of highly pure cells (> 70% pure, > 1 × 109) because of the low frequency and number (<20% pure, <1 × 108) of NK cells in the CB [3, 4]. To provide allogeneic NK cells with high yield, purity and functionality, some methods have been developed to purify and expand NK cells from CB ex vivo [5–10].
To date, most methods for in vitro preparation of NK cells from CB require to selecte NK cells with immune-selection techniques because of low frequency [11]. In order to avoid the limitations in low number and immature state of NK cells in CB, ex vivo expansion and activation is necessary [12]. NK cells are generally isolated from CB through immunomagnetic beads selection protocols to enrich CD56-positive cells and/or deplete CD3-positive cells, and then cultured for functional expansion and activation using feeder cells, such as Epstein-Barr virus-transformed lymphoblastoid cell lines, mesenchymal stromal cells, gene-modified K562 cells expressing 4-1BB ligand and IL-15, and other irradiated tumor cell lines [5, 13]. In addition, NK cells are originally generated from CD34+ hematopoietic stem cells (HSCs), some studies have described an alternative method to generate NK cells with high yield, purity and functionality from CB-derived CD34+ HSCs under feeder cells-based conditions [10, 14–16]. Recently, a feeder cells-free method has been successfully performed for the generation of NK cells from CB-derived CD34+ HSCs [7, 17]. However, it needs delicate culture regimens and multiple cytokine cocktails, which may lead to high cost-effectiveness. Generally, these methods require a complicated technology of cell sorting in an initial step, and it may increase the risk of cell trauma and contamination. Furthermore, the use of feeder cells or multiple cytokines during longer-term cultures would lead to NK cell apoptosis in vivo when optimum culturing conditions are eliminated after adoptive transfer [18]. In addition, these methods are also more costly because of complex operations and supplements.
Although several methods have been proposed to generate clinically relevant NK cell products (mean: 2 × 109 cells) with high purity (> 90%) from CB [13, 19], it is still difficult to obtain the sufficient numbers of highly pure NK cells from CB without cell sorting and feeder cells/multiple cytokines [13]. Previously, we had found that zoledronate could increase enrichment, expansion and activation of NK cells from CB-derived mononuclear cells (MNCs) [20]. Some studies have reported that interleukin (IL)-2 expansion could recruit and activate key regulators involved in lytic immunological synapse formation of CB-derived NK cells, enabling effective cytotoxicity against killing of acute myeloid leukemia (AML) cells in vitro and in vivo [21, 22]. Group A streptococcus preparation, which is widely used as an immunopotentiator with considerable success in patients with malignant diseases, strongly augmented human NK cell activity in vivo as well as in vitro [23]. Therefore, we try to use develop a simple method with the capability of generating NK cells with high yield, purity and functionality from CB through using zoledronate, group A streptococcus and IL-2 stimulation of MNCs without cell sorting and feeder cells/multiple cytokines.
Results
Preparation of NK cells from CB
After the isolation process by Ficoll, an average of 5.02% CD56+CD3− NK cells (range, 1.92 to 9.66%) was obtained in MNCs, whereas CD56−CD3+ T cells constituted 84.53% (range, 72.98 to 96.34%). Expansion of CD56+CD3− NK cells was much higher compared with other types of cells, so NK cells dominated at the end of the culture, reaching on average 92.37% of the total cell populations (range: 88.91 to 96.37%; Additional file 1) by day 21. The frequency of CD56+CD3+ NKT cells remained largely unchanged before and after culture (day 0: 1.05%, day 21: 1.35%), whereas the frequency of CD56−CD3+ T cells declined, decreasing to an average of 4.56% (range: 1.30 to 9.63%; Fig. 1a and b). By day 21, the total cell count had expanded on average 101-fold (range: 65–137-fold) and, among these, CD56+CD3− NK cell population had expanded on average 1561-fold (range: 695–2387-fold), reaching on average 1.59 × 1010 (range: 0.84–2.23 × 109; Fig. 1c). At this time point the expansion potential reached a plateau, thus the cells reached a quiescence phase when measured later on day 28. These data demonstrate that NK cells with high yield and purity could be expanded efficiently from CB-derived MNCs ex vivo using the method described here. .
Fig. 1.
Characterization of expanded NK cells from CB. Cell proportion (CD56+CD3− NK cell, CD56−CD3+ T cell and CD56+CD3+ NKT cell) was analyzed by flow cytometric analyses, representative FACS dot plots are presented (a). The kinetics of (b) cell proportion and (c) cell population (total cell and CD56+CD3− NK cell) during culture. ***P < 0.001, indicates statistical significance increase in NK cell proportion and total and NK cell population compared to previous time points of assessment; #P > 0.05, indicates no statistical significance between day 28 and 21. Data are shown as mean ± standard deviation, n = 5
Surface expression of NK receptors and markers
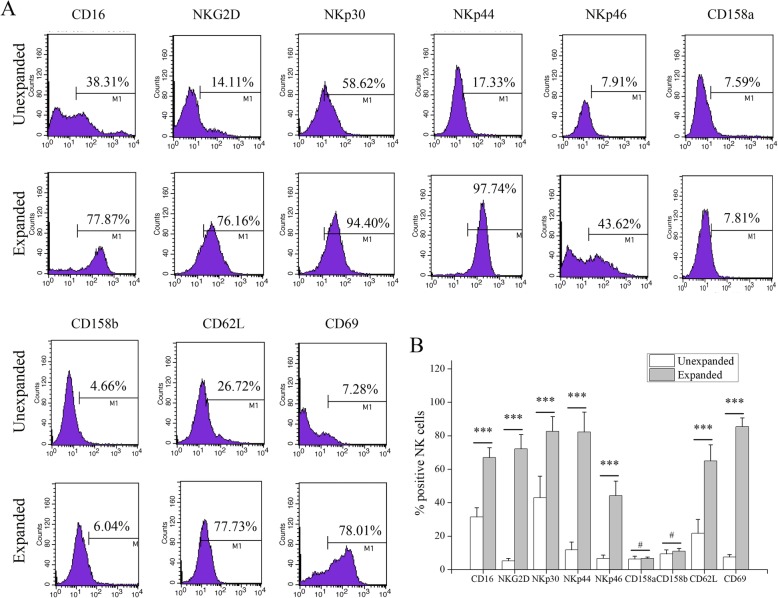
The expression of receptors and markers in expanded NK cells was assessed and compared to unexpanded NK cells. Among activating receptors, CD16 (day 0: 31.40%, day 21: 66.91%; P < 0.001), NKp30 (day 0: 42.99%, day 21: 82.61%; P < 0.001), NKp44 (day 0: 11.72%, day 21: 82.19%; P < 0.001), NKp46 (day 0: 6.68%, day 21: 44.20%; P < 0.001) and NKG2D (day 0: 5.14%, day 21: 72.20%; P < 0.001) significantly increased during the expansion while expression of the inhibitory receptors, CD158a (day 0: 6.33%, day 21: 6.68%; P > 0.05) and CD158b (day 0: 9.40%, day 21: 10.91%; P > 0.05) remained largely unchanged during the culture period. Also, activation markers of the NK cells including CD62L (day 0: 21.66%, day 21: 64.91%; P < 0.001) and CD69 (day 0: 7.47%, day 21: 85.43%; P < 0.001) were increased on the surface of expanded NK cells (Fig. 2). These data demonstrate that NK cells are markedly activated after expansion.
Fig. 2.
Receptors and markers of unexpanded and expanded NK cells from CB. (a) Representative histograms and (b) percentages of CD16, NKG2D, NKp30, NKp44, NKp46, CD158a, CD158b, CD62L, and CD69 expression on CD56+CD3− NK cells of the unexpanded and expanded NK cells from CB. ***P < 0.001, indicates statistical significance increase in receptors and markers compared to unexpanded NK cells; #P > 0.05, indicates no statistical significance. Data are shown as mean ± standard deviation, n = 5
Cytokines production of expanded NK cells
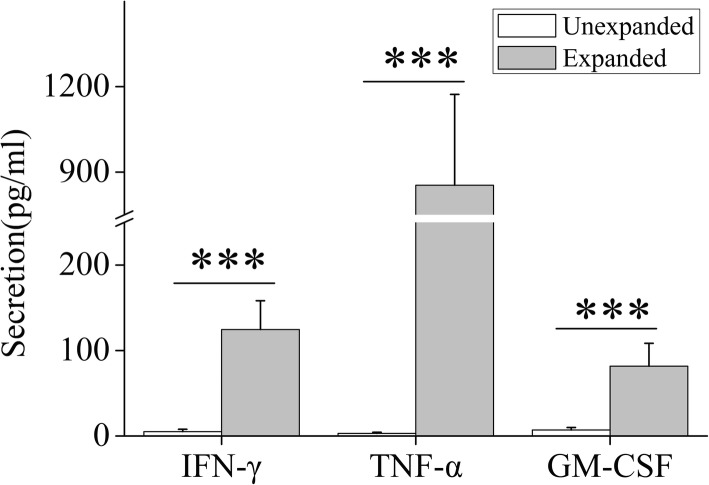
The secretion cytokines including granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon-gamma (IFN-γ), and tumor necrosis factor-alpha (TNF-α) were also assessed before and after culture. The very low level of IFN-γ (mean: 5.21 pg/mL, range: 1.37–8.45 pg/mL), TNF-α (mean: 2.99 pg/mL, range: 1.16–5.04 pg/mL) and GM-CSF (mean: 6.95 pg/mL, range: 4.38–9.81 pg/mL) secretion was observed in unexpanded NK cells. Expanded NK cells exhibited significantly increased IFN-γ (mean: 124.67 pg/mL, range: 57.95–183.12 pg/mL), TNF-α (mean: 853.94 pg/mL, range: 479.67–1201.26 pg/mL) and GM-CSF (mean: 81.67 pg/mL, range: 55.43–108.42 pg/mL) secretion compared to unexpanded NK cells (all P < 0.001, Fig. 3). These results demonstrate that the patterns of cytokine secretion are markedly increased after culture.
Fig. 3.
Cytokine production of unexpanded and expanded NK cells from CB. ***P < 0.001, indicates statistical significance increase in cytokine production compared to unexpanded NK cells. Data are shown as mean ± standard deviation, n = 5
Cytotoxicity of expanded NK cells
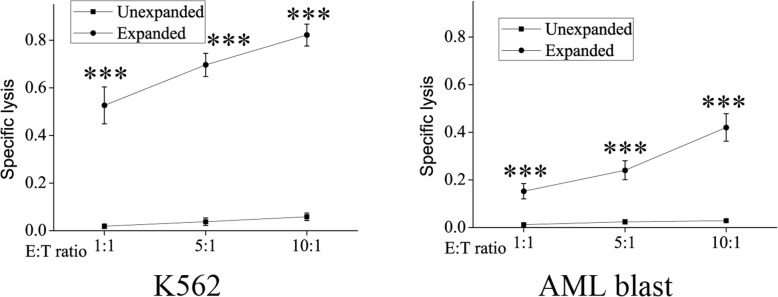
The cytotoxicity levels of expanded vs. unexpanded NK cells against leukemia cell line (K562) and primary patient AML blasts were evaluated. The unexpanded NK cells were poor cytotoxicity against K562 cells (range: 1.91–5.86%) and primary patient AML blasts (range: 1.24–2.89%) at a variety of E:T ratios, but the expanded NK cells showed high levels of cytotoxicity against K562 (range: 52.63–82.18%) and AML targets (range: 15.28–24.07%). NK cells expanded in culture consistently exhibited a high level of cytotoxicity against K562 and AML targets compared to unexpanded NK cells at a wide range of E:T ratios (all P < 0.001, Fig. 4). These results show that cytotoxicity of NK cells are markedly elevated after expansion.
Fig. 4.
Cytolytic activities of unexpanded and expanded NK cells from CB. ***P < 0.001, indicates statistical significance increase in cytolytic activities compared to unexpanded NK cells. Data are shown as mean ± standard deviation, n = 5
Discussion
In this study, we develop a simple and economical method for in vitro preparation of NK cells through IL-2, group A streptococcus and zoledronate stimulation of CB-derived MNCs. it is a practically advantageous method that does not need cell sorting and feeder cells/multiple cytokines, which could lead to reduction in cost. Furthermore, IL-2, streptococcus A group and zoledronate have also been approved for human use, which could increase the safety. In addition, this simple method is more easily standardized and exportability. To our knowledge, this is the first report of a simple method for in vitro preparation of NK cells from CB without cell sorting and feeder cells/multiple cytokines.
Using this method, a mean number of 1.59 × 1010 NK cells with an average purity of 92.37% are generated after 21 days of culture. Previously, we found that zoledronate had a favorable effect on expansion of NK cells from CB-derived MNCs [20]. Furthermore, the addition of group A streptococcus induced a higher increase in the expansion fold (1561 vs. 1286) of NK cells, showing that both have the synergism. A study has reported a multiple cytokines-based delicate culture method for up to 6 weeks to generate therapeutic NK cell products (mean: 2 × 109 cells) with high purity (> 90%) from CB-derived CD34+ HSCs [9]. Compared with this method, our method yields much higher number (8-times) of NK cells with less time. We hypothesize that this is related to very low proportion of CD34+ HSCs in CB and cumbersome procedures including HSCs expansion and NK cell differentiation and expansion. Other group has reported that a feeder cell-based NK cultivation method is established with the capability of generating a clinically relevant dose (mean: 1.2 × 109 cells) with a purity of > 80% of CB-derived NK cells [6]. Compared with this method, our method also yields much higher number (15-times) of NK cells with the same time. A possible explanation might be that accessory cells such as monocytes in CB-derived MNCs can support NK cell expansion [24]. Therefore, A large amount of NK cells generated in vitro by our method can be, allowed for multiple infusions of NK cells.
Purity, as one of crucial release criteria for NK cell-based therapies, has not yet been standardized, but > 90% CD56+CD3− for allogenic NK cells have been suggested in recently published study [9, 25]. Previously, we found that zoledronate had a favorable effect on enrichment of NK cells from CB-derived MNCs [20]. Furthermore, the addition of group A streptococcus induced a greater increase in the frequency (92.37% vs. 80.46%) of NK cells, showing that both have the synergism. The purity of NK cells produced by our method is comparable to cytokine-based culture method (> 90%) [9], but higher compared to feeder cell-based method (> 80%) [6].
Another crucial obstacle is the immaturity state of NK cells in CB [3]. It has been known that NK cell cytotoxicity is regulated by the complex balance between activating and inhibitory receptors. NK cells expended by our method exhibit high expression level of activating receptors CD16, NKG2D, NKp30, NKp44 and NKp46, but unchanged expression level of inhibitory receptors CD158a and CD158b compared with unexpended NK cells. This leads to high-level expression of activating markers CD62L and CD69, showing that NK cells expanded in this study have activated a cellular mechanism controlling effector function. A recent study also reported no changes in expression of CD158a and CD158b by CB-derived NK cells after incubation with IL-2, IL-12 or IL-18 [26]. This confirms that killer cell Ig-like receptor acquisition does not require group A streptococcus and zoledronate activation.
Previous studies have shown that IL-2-activated NK cells from CB exhibited significantly increased frequency of IFN-γ and TNF-α secretion compared with unexpended NK cells [21, 26]. In previous study, we showed that zoledronate increased IFN-γ, TNF-α and GM-CSF secretion by IL-2-activated NK cells from CB [20]. Early study showed that group A streptococcus stimulated human lymphocytes to produce IFN (both a- and γ-types) and IL-2 and that both factors were primarily responsible for the NK augmentation by group A streptococcus [23]. It has been previously shown that cytokines including IFN-γ, TNF-α and GM-CSF play key roles in NK cell functions [27]. To evaluate the impact of IL-2, zoledronate and group A streptococcus on the secretion of cytokines by CB NK cells, cytokines secretion was measured in the supernatants of unexpended and expanded NK cell cultures. The result shows that NK cells expanded by our method can produce robust inflammatory cytokine IFN-γ, TNF-α and GM-CSF. We further investigate that NK cells expanded by our method can induce lytic function against K562 and primary AML targets compared with unexpended NK cells, showing that ex vivo generated NK cells from CB could contribute greatly to the elimination of tumor during adoptive NK cell immunotherapy. These results are comparable to data from zoledronate only, showing no synergy between them. This might be related to MNCs rather than purified NK cells. However, in vivo functional studies are needed to explore in the next study.
Conclusion
We have developed a simple, safe and economical method to obtain high yield, purity, and functionality NK cells from CB through IL-2, streptococcus A group and zoledronate stimulation of MNCs without cell sorting and feeder cells/ multiple cytokines. Adoptive transfer of NK cells generated from CB by this method may therefore provide a promising new paradigm for the treatment of patients with AML and other hematological malignancies. This simple method satisfies the standards for in vitro preparation of NK cells as a potential therapeutic product, is more accessible for clinical practice, and may hold valuable potential for adoptive cellular immunotherapy for hematological malignancies like AML.
Methods
NK cells preparation
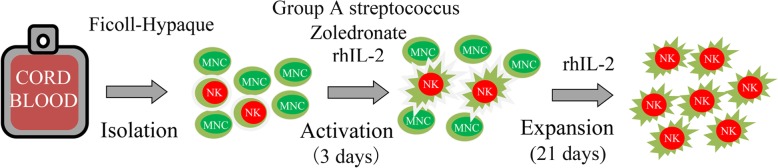
All frozen CB samples were drawn from the Shandong Cord Blood Bank. MNCs were isolated from CB using Ficoll-Hypaque density gradient centrifugation. For activation, MNCs were cultured for 3 days in T175 flasks at 2 × 106 cells/mL in AIM-V serum free media (Life Technologies) supplemented with 2000 IU IL-2/mL (Four Rings Biopharma, China), 0.01KE/mL group A streptococcus (Lu Ya Pharma, China) and 5 μM zoledronate (Novartis Pharma). For expansion, the fresh medium containing 2000 IU IL-2/mL was added every 2 to 3 days for 21 days. An outline of the preparation protocol is summarized in Fig. 5.
Fig. 5.
Schematic diagram of the preparation protocol for NK cells from CB
Expansion kinetics assessment
Total cell counts were determined by trypan blue staining on days 0, 7, 14, 21 and 28. Absolute cell numbers were calculated by multiplying the total cell count by the percentage of each cell type determined by flow cytometry. Cell expansion efficiency was expressed as “fold expansion” which was determined by dividing the number of total cells or absolute NK cells on days 7, 14, 21 and 28 by the number on day 0.
Flow Cytometr
The cells were labeled with fluorochrome-conjugated mouse mAbs against human cluster of differentiation CD3, CD56, CD16, CD158a, CD158b and NKG2D (BD Biosciences); NKp30, NKp44 and NKp46 (Beckman Coulter); CD69, CD62L (Miltenyi Biotec). Isotype-matched antibodies were used as controls. Flow cytometry analysis was performed using a MACSQuant Analyzer (Miltenyi Biotec). Data were analyzed with MACSQuantify Software.
Cytokines release assay
The cell culture supernatants were collected, the level of cytokines including IFN-γ, TNF-α and GM-CSF in supernatants was analyzed using enzyme-linked immunosorbent assay kits (eBioscience, Inc) according to the manufacturer’s instructions.
Cytotoxicity assay
The cytotoxicity of NK cells was analyzed using the standard 4-h 51Cr release assay. The NK-sensitive leukemia cell line (K562) and primary AML blasts (target) were labeled with 51Cr sodium chromate and incubated with NK cells (effector) at different target-to-effector (E:T) ratios (1:1, 5:1, and 10:1) in U-bottomed 96-well plates. 51Cr-release was determined in the supernatant after co-culture for 4 h. The maximum release was measured by treating target cells with 2% Triton X-100 and spontaneous release was determined with medium alone. The specific lysis was calculated according to the formula: (experimental release - spontaneous release) / (maximum release - spontaneous release).
Statistical analysis
Data are presented as the mean ± standard deviation. Statistical difference was determined using a student’s t test when comparing two groups, or a one-way ANOVA analysis when more than two groups (Graphpad Prism 5.0). P < 0.05 was considered significant.
Supplementary information
Additional file 1: Figure S1. FACS analysis gating strategy for NK cells expanded on day 21.
Acknowledgements
We acknowledged the kind support from Department of Technology, Beijing.
JingMeng Stem Cell Technology. Co. Ltd., Beijing, China.
Abbreviations
- AML
Acute myeloid leukemia
- CB
Cord blood
- E:T
Effector-to-target
- GM-CSF
Granulocyte-macrophage colony-stimulating factor
- HSCs
Hematopoietic stem cells
- IFN-γ
Interferon-gamma
- IL
Interleukin
- MNCs
Mononuclear cells
- NK
Natural killer
- TNF-α
Tumor necrosis factor-alpha
Authors’ contributions
All authors participated in the design, interpretation of the results, and review of the manuscript; YXM and XYW were involved in the experimentation, and wrote the manuscript; YXZ, HJB and BYL were involved in the analysis of the data. HJ, XLQ and LYB generated the figures and contributed to editing the manuscript, the study was conceived by YHW and ZGL who also edited the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by National Natural Science Foundation of China (No. 81860157) and Natural Science Foundation of Inner Mongolia (No. 2017MS0314).
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
All frozen CB samples were obtained with prior consent and ethical committee approval from the Shandong Cord Blood Bank. The study had full ethical approval from the institutional ethics committee of Beijing Friendship Hospital.
Consent for publication
Not applicable.
Competing interests
A patent application for the composition for expanding NK cells and the use of them has been filed with Xiao Yun Wu as a first inventor. Other authors declare that they have no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yong Xu Mu and Yu Xia Zhao contributed equally to this work.
Contributor Information
Zhi Jie Ma, Email: 13811647091@163.com.
Xiao Yun Wu, Email: stemcells@foxmail.com.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12896-019-0564-0.
References
- 1.Lim O, Jung MY, Hwang YK, Shin EC. Present and future of allogeneic natural killer cell therapy. Front Immunol. 2015;6:286. doi: 10.3389/fimmu.2015.00286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shaim H, Yvon E. Cord blood: a promising source of allogeneic natural killer cells for immunotherapy. Cytotherapy. 2015;17(1):1–2. doi: 10.1016/j.jcyt.2014.12.001. [DOI] [PubMed] [Google Scholar]
- 3.Mehta RS, Shpall EJ, Rezvani K. Cord blood as a source of natural killer cells. Front Med. 2015;2:93. doi: 10.3389/fmed.2015.00089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Klingemann H. Challenges of cancer therapy with natural killer cells. Cytotherapy. 2015;17(3):245–249. doi: 10.1016/j.jcyt.2014.09.007. [DOI] [PubMed] [Google Scholar]
- 5.Hosseini E, Ghasemzadeh M, Kamalizad M, Schwarer AP. Ex vivo expansion of CD3depleted cord blood-MNCs in the presence of bone marrow stromal cells; an appropriate strategy to provide functional NK cells applicable for cellular therapy. Stem Cell Res. 2017;19:148–155. doi: 10.1016/j.scr.2017.01.010. [DOI] [PubMed] [Google Scholar]
- 6.Kang L, Voskinarian-Berse V, Law E, Reddin T, Bhatia M, Hariri A, Ning Y, Dong D, Maguire T, Yarmush M, et al. Characterization and ex vivo expansion of human placenta-derived natural killer cells for Cancer immunotherapy. Front Immunol. 2013;4:101. doi: 10.3389/fimmu.2013.00101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cany J, van der Waart AB, Tordoir M, Franssen GM, Hangalapura BN, de Vries J, Boerman O, Schaap N, van der Voort R, Spanholtz J, et al. Natural killer cells generated from cord blood hematopoietic progenitor cells efficiently target bone marrow-residing human leukemia cells in NOD/SCID/IL2Rg(null) mice. PLoS One. 2013;8(6):e64384. doi: 10.1371/journal.pone.0064384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dezell SA, Ahn YO, Spanholtz J, Wang H, Weeres M, Jackson S, Cooley S, Dolstra H, Miller JS, Verneris MR. Natural killer cell differentiation from hematopoietic stem cells: a comparative analysis of heparin- and stromal cell-supported methods. Biol Blood Marrow Transplant. 2012;18(4):536–545. doi: 10.1016/j.bbmt.2011.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spanholtz J, Preijers F, Tordoir M, Trilsbeek C, Paardekooper J, de Witte T, Schaap N, Dolstra H. Clinical-grade generation of active NK cells from cord blood hematopoietic progenitor cells for immunotherapy using a closed-system culture process. PLoS One. 2011;6(6):e20740. doi: 10.1371/journal.pone.0020740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Frias AM, Porada CD, Crapnell KB, Cabral JM, Zanjani ED, Almeida-Porada G. Generation of functional natural killer and dendritic cells in a human stromal-based serum-free culture system designed for cord blood expansion. Exp Hematol. 2008;36(1):61–68. doi: 10.1016/j.exphem.2007.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Becker PS, Suck G, Nowakowska P, Ullrich E, Seifried E, Bader P, Tonn T, Seidl C. Selection and expansion of natural killer cells for NK cell-based immunotherapy. Cancer Immunol Immunother. 2016;65(4):477–484. doi: 10.1007/s00262-016-1792-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lapteva N, Szmania SM, van Rhee F, Rooney CM. Clinical grade purification and expansion of natural killer cells. Crit Rev Oncog. 2014;19(1–2):121–132. doi: 10.1615/CritRevOncog.2014010931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chabannon C, Mfarrej B, Guia S, Ugolini S, Devillier R, Blaise D, Vivier E, Calmels B. Manufacturing natural killer cells as medicinal products. Front Immunol. 2016;7:504. doi: 10.3389/fimmu.2016.00504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pinho MJ, Punzel M, Sousa M, Barros A. Ex vivo differentiation of natural killer cells from human umbilical cord blood CD34+ progenitor cells. Cell Commun Adhes. 2011;18(3):45–55. doi: 10.3109/15419061.2011.610911. [DOI] [PubMed] [Google Scholar]
- 15.Boissel L, Tuncer HH, Betancur M, Wolfberg A, Klingemann H. Umbilical cord mesenchymal stem cells increase expansion of cord blood natural killer cells. Biol Blood Marrow Transplant. 2008;14(9):1031–1038. doi: 10.1016/j.bbmt.2008.06.016. [DOI] [PubMed] [Google Scholar]
- 16.Kao IT, Yao CL, Kong ZL, Wu ML, Chuang TL, Hwang SM. Generation of natural killer cells from serum-free, expanded human umbilical cord blood CD34+ cells. Stem Cells Dev. 2007;16(6):1043–1051. doi: 10.1089/scd.2007.0033. [DOI] [PubMed] [Google Scholar]
- 17.Domogala A, Blundell M, Thrasher A, Lowdell MW, Madrigal JA, Saudemont A. Natural killer cells differentiated in vitro from cord blood CD34+ cells are more advantageous for use as an immunotherapy than peripheral blood and cord blood natural killer cells. Cytotherapy. 2017;19(6):710–720. doi: 10.1016/j.jcyt.2017.03.068. [DOI] [PubMed] [Google Scholar]
- 18.Childs RW, Berg M. Bringing natural killer cells to the clinic: ex vivo manipulation. Hematology Am Soc Hematol Educ Program. 2013;2013:234–246. doi: 10.1182/asheducation-2013.1.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Koehl U, Kalberer C, Spanholtz J, Lee DA, Miller JS, Cooley S, Lowdell M, Uharek L, Klingemann H, Curti A, et al. Advances in clinical NK cell studies: donor selection, manufacturing and quality control. Oncoimmunology. 2016;5(4):e1115178. doi: 10.1080/2162402X.2015.1115178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ma Zhijie, Wang Yunhong, Kang Huiyan, Wu Xiaoyun. Zoledronate increases enrichment, activation and expansion of natural killer cells from umbilical cord blood. Human Cell. 2018;31(4):310–312. doi: 10.1007/s13577-018-0213-2. [DOI] [PubMed] [Google Scholar]
- 21.Xing D, Ramsay AG, Gribben JG, Decker WK, Burks JK, Munsell M, Li S, Robinson SN, Yang H, Steiner D, et al. Cord blood natural killer cells exhibit impaired lytic immunological synapse formation that is reversed with IL-2 exvivo expansion. J Immunother. 2010;33(7):684–696. doi: 10.1097/CJI.0b013e3181e475e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Khaziri N, Mohammadi M, Aliyari Z, Soleimani Rad J, Tayefi Nasrabadi H, Nozad Charoudeh H. Cord blood mononuclear cells have a potential to produce NK cells using IL2Rg cytokines. Adv Pharm Bull. 2016;6(1):5–8. doi: 10.15171/apb.2016.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wakasugi H, Kasahara T, Minato N, Hamuro J, Miyata M, Morioka Y. In vitro potentiation of human natural killer cell activity by a streptococcal preparation, OK-432: interferon and interleukin-2 participation in the stimulation with OK-432. J Natl Cancer Inst. 1982;69(4):807–812. [PubMed] [Google Scholar]
- 24.Koehl U, Brehm C, Huenecke S, Zimmermann SY, Kloess S, Bremm M, Ullrich E, Soerensen J, Quaiser A, Erben S, et al. Clinical grade purification and expansion of NK cell products for an optimized manufacturing protocol. Front Oncol. 2013;3:118. doi: 10.3389/fonc.2013.00118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Berg M, Childs R. Ex-vivo expansion of NK cells: what is the priority--high yield or high purity? Cytotherapy. 2010;12(8):969–970. doi: 10.3109/14653249.2010.536216. [DOI] [PubMed] [Google Scholar]
- 26.Alnabhan R, Madrigal A, Saudemont A. Differential activation of cord blood and peripheral blood natural killer cells by cytokines. Cytotherapy. 2015;17(1):73–85. doi: 10.1016/j.jcyt.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 27.Yoon SR, Kim TD, Choi I. Understanding of molecular mechanisms in natural killer cell therapy. Exp Mol Med. 2015;47:e141. doi: 10.1038/emm.2014.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. FACS analysis gating strategy for NK cells expanded on day 21.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.