Abstract
Increasing serostatus awareness is a key HIV prevention strategy. Despite expanded testing efforts, some men who have sex with men (MSM) remain unaware of their HIV status. This study explored demographic characteristics, sexual identity, sexual role, and behavioral factors associated with unknown HIV status among MSM in the United States. Data from 9,170 MSM in the 2014 American Men’s Internet Survey were analyzed using logistic regression to identify correlates of unknown HIV status. Young age, race, low education, rural residence, and lack of recent healthcare visits were significantly associated with unknown HIV status. In addition, nondisclosure of one’s sexual orientation (OR=3.70, 95% CI=2.99–4.59) and a self-identified sexual role as “bottom” (OR=1.45, 95% CI=1.24–1.70) were predictors of unknown HIV status. Post-hoc analysis showed HIV-negative MSM not tested in the last year had fewer self-reported risks behaviors than recent testers, suggesting that repeat testing among MSM may be aligned with individual risk.
Keywords: HIV testing, unknown HIV status, men who have sex with men (MSM), internet survey
INTRODUCTION
Despite ongoing prevention efforts, the HIV epidemic continues to be a major public health concern in the United States (US) with approximately 40,000 new HIV infections occurring each year.(1) It is estimated that one-third of new cases is attributed to transmission involving HIV-positive individuals who are unaware of their infection.(2) Consequently, increasing serostatus awareness is a key step in preventing new HIV infections. When HIV-positive individuals become aware of their infection, not only can they take steps to improve their own health by seeking treatment, but they may also practice measures to reduce the risk of transmitting the infection to others. Several studies have shown that newly diagnosed HIV-positive individuals make behavioral changes such as reducing the number of sex partners, decreasing the frequency of condomless anal intercourse, practicing harm reduction strategies such as serosorting or seropositioning, and/or abstaining from sex during the period following diagnosis.(3–9) These behaviors may be long-term and sustained for several years following diagnosis.(3, 5) In addition, as HIV-infected individuals begin antiretroviral therapy, their viral loads decrease, often dramatically, and the risk of infection to others also declines.(10–14) Thus, promoting HIV serostatus awareness remains an important public health objective.
As part of a comprehensive plan to prevent new HIV infections, the Office of National AIDS Policy has identified increasing the percentage of individuals living with HIV who know their status as a key priority in the National HIV/AIDS Strategy. The goal is to increase serostatus awareness among HIV-infected persons to 90% by the year 2020.(15) Although great strides have been made toward this goal, recent estimates suggest that 12.8% of HIV-infected individuals in the US are still unaware of their HIV status, and this proportion is slightly higher (14.8%) among infected men who have sex with men (MSM).(16) As we continue working to improve serostatus awareness, it becomes increasingly critical to focus efforts on HIV testing, as this is the first step in the HIV care continuum. Increasing HIV testing will raise serostatus awareness, and in turn lower the proportion of HIV-infected individuals who remain unaware of their infection. Therefore, reaching populations who are still not being tested and identifying barriers to HIV testing are vital to both HIV prevention and treatment.
In the US, male-to-male sexual contact accounts for the highest number of new HIV infections. It is estimated that in 2015, 82.4% of all new infections among males were attributed to male-to-male sexual contact.(1) As a result, MSM are often the focus of targeted testing initiatives. The current HIV testing recommendations for MSM include testing for all men aged 13–64 at least once in their lifetime, plus repeated testing for high-risk groups such as injection drug users, sex partners of HIV-infected individuals, individuals who exchange sex for money or drugs, or those with multiple sex partners. Yet even among this at-risk and highly targeted population, 8.6 – 35.6% of MSM report never being tested for HIV.(17–21)
In order to increase testing among MSM with unknown HIV status, it is imperative to define the characteristics of this group. Studying MSM who have never been tested for HIV is of particular interest so that interventions may be properly developed and targeted to address testing barriers specific to these individuals. Research in this area is mounting but is often limited to studies that have small sample sizes, include non-MSM or only specific subgroups of MSM, were conducted outside the US, or examine only demographic risk factors associated with HIV testing behaviors.(17, 19, 22, 23) The current study aims to explore a broader set of correlates for unknown HIV status including sexual identity, sexual role, various behavioral risk factors, and perceived peer HIV testing behavior by analyzing data collected from a large sample of MSM participating in the 2014 American Men’s Internet Survey (AMIS). Results from this study will help to better characterize the population of MSM with unknown HIV status and inform strategies to increase HIV testing in this group.
METHODS
The study population was MSM living in the US. Data used for this analysis were collected from the 2014 American Men’s Internet Survey (AMIS), an on-line survey assessing the behaviors of men who have sex with men (MSM) in the US. Full details of survey administration and key indicators have been described elsewhere.(24) Briefly, MSM were recruited to participate in AMIS from a variety of social networking websites between November 2014 and April 2015. Eligible participants were 15 years of age or older at the time of survey administration, identify as male, reside in the US or a dependent area, and reported at least one lifetime sex act (oral or anal) with a male partner. The survey collected information about demographics, sexual behaviors, substance use behaviors, HIV status, HIV testing behaviors, history of sexually transmitted infections (STIs), and awareness of HIV prevention services and educational campaigns. Some participants were also randomized to complete a series of questions about HIV pre-exposure prophylaxis (PrEP) and post-exposure prophylaxis (PEP).
For this analysis, the outcome of interest was unknown HIV status, which was assessed through two survey items—self-reported history of ever being tested for HIV and self-reported results of one’s most recent HIV test. Unknown HIV status was defined as an individual who a) had never been tested for HIV, b) was tested for HIV but never received the results, or c) was tested for HIV but had test results that were unknown or indeterminate. Inclusion of these last two groups is valuable as it helps to describe a broader spectrum of MSM with unknown HIV status. Men with unknown HIV status were compared to men with known HIV-negative status.
Data for several predictor variables were analyzed. Demographic factors included age in years (continuous), race (White, Black, Hispanic/Latino, American Indian/Native Alaskan, Asian/Hawaiian/Pacific Islander, or other/multiple), education level (less than high school graduate, high school graduate or equivalent, some college/technical school, or beyond college graduate), and zip code of residence. Zip code information was subsequently used to determine the participant’s geographic region of residence in accordance with the HIV surveillance categories used by the Centers for Disease Control and Prevention and whether it was considered a rural or urban area. Two healthcare indicators were included—health insurance status (private, public, other, or none) and visit with a healthcare provider in the past year (yes/no). Sexual risk predictors included sexual identity (heterosexual, homosexual, or bisexual), disclosure status or “outness” (defined as disclosing to anyone an attraction to or history of sex with another man), sexual position (self-identifying as a “top,” “bottom,” or “versatile”), type of sex acts experienced in the past 12 months (oral, anal, both, or none), number of sex partners in the past year (continuous), history of sex with a female in the past year, history of condomless sex in the past year, serosorting (defined as only having sex with HIV-negative men), and diagnosis with an STI in the past year. Two substance use risk factors were also included—history of ever injecting drugs, and non-injection drug use in the past 12 months. Finally, perception of peer HIV testing behavior was assessed through a five-item Likert-type response to the statement, “Most gay men I know get tested for HIV at least every 3–6 months” (1-strongly agree to 5-strongly disagree).
Descriptive statistics were calculated for each variable using frequencies for categorical predictors and means with standard deviations (SDs) for continuous predictors. Next, bivariate logistic regression models were run to determine associations between unknown HIV status and each predictor. Then, a multivariate logistic regression model was built using a backward selection approach to reduce model complexity. All predictors with a bivariate association of p-value < .20 were included in the initial model. Variables were removed one at a time until only predictors with a multivariate association of p-value < .05 remained. Odds ratios (OR) and 95% confidence intervals (CIs) were calculated. Fit of the final model was assessed using the Hosmer-Lemeshow goodness of fit test. Descriptive analyses and backward selection logistic regression were carried out using SAS University Edition version 9.2.(25)
In order to account for missing data across predictors, the final logistic regression model resulting from the backward selection procedure was used for Full Information Maximum Likelihood (FIML) estimation in Mplus version 7.4.(26) This allowed for retention of all participants in the dataset and utilized the full information available to estimate the population parameters most likely to result in the observed data. The estimates from the FIML model did not differ significantly from the backward elimination model using the limited dataset, and so the FIML results are reported here.
A post-hoc analysis was also conducted to compare HIV-negative MSM who tested for HIV more than 12 months ago to HIV-negative MSM who tested within the last 12 months (recent testers). This additional analysis provided a more comprehensive description of the different risk profiles associated with unknown HIV status resulting from testing behaviors. Model selection was the same as described above with the following exceptions. Men living in US territories were excluded from the analysis of region of residence due to low cell counts. Additionally, men who reported having only anal sex in the last 12 months were combined with men who reported both oral and anal sex due to insufficient sample sizes in the anal sex only category. Similarly, response options for the peer HIV testing behavior item were combined (strongly agree with agree and strongly disagree with disagree) due to low cell counts for the extreme response categories. All other variables examined in the initial analysis were included. Finally, a descriptive analysis was conducted to characterize the HIV testing behaviors of high-risk MSM in the study sample. Participants were classified as high-risk according to risk factors identified by the CDC in the 2006 HIV screening recommendations for adults and adolescents in health-care settings. This included any of the following self-reported factors in the last 12 months: injection drug use, exchanging sex for money or drugs, condomless anal sex with an HIV-positive individual, and having multiple sex partners (> 1 partner) but no HIV test in the last 12 months. Additionally, participants who reported having an STI in the last 12 months were considered high-risk. Sex-partners of injection drug users were not included as a high-risk group for this analysis, as this risk factor was not assessed in the AMIS questionnaire.
RESULTS
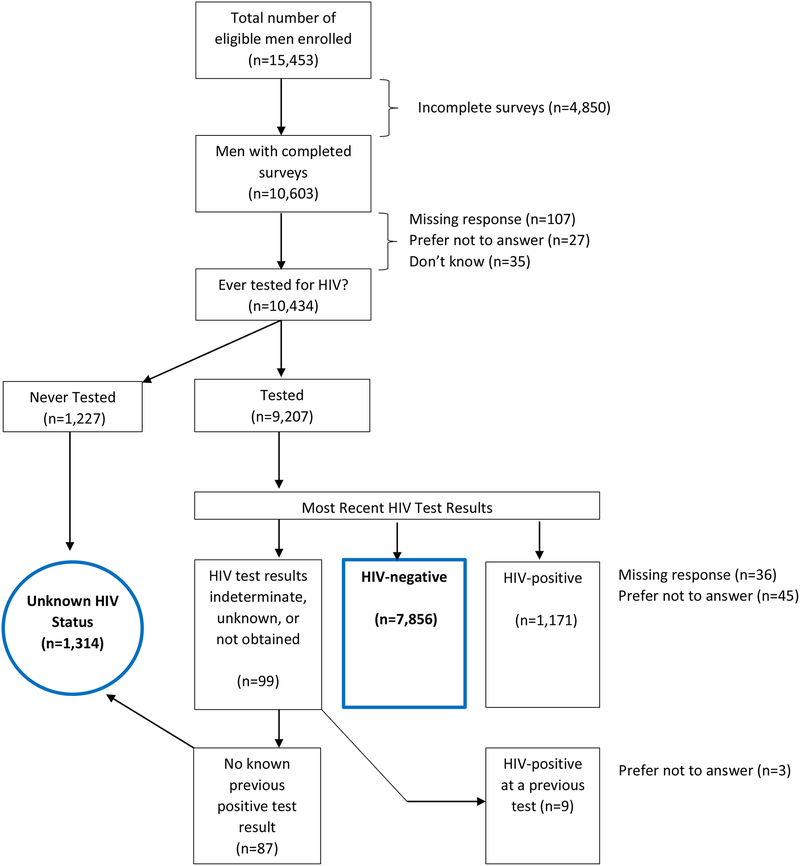
The selection of participants for this analysis is shown in Figure 1. A total of 15,453 eligible men participated in the 2014 AMIS survey. Of these, 10,603 (68.6%) records were considered to be mostly complete, which included records that did not have missing data for the first item of two consecutive sections. Participants who did not provide a response (n=107), or reported “I don’t know” (n=35) or “I prefer not to answer” (n=27) to the item regarding history of ever testing for HIV were excluded from the analysis. Additionally, men who were tested for HIV but whose most recent test result was missing (n=36) or reported as “I prefer not to answer” (n=45) were also excluded. Finally, all HIV-positive men (n=1,180) and men selecting “I prefer not to answer” in response to ever testing positive (n=3) were excluded, leaving a final sample of 9,170 records for analysis.
Figure 1.
Flowchart outlining AMIS participants selected for analysis. Blue outline indicates groups included in analysis
Characteristics of the study sample are shown in Table I. Participants ranged in age from 15–101 years (mean=40.7 years, SD=14.3 years). Respondents were mostly White (75.9%) and highly educated, with most participants (58.3%) being college graduates. Geographically, 38.6% of respondents resided in the Southern US, followed by the West (22.4%), Midwest (21.5%), Northwest (17.4%), and US dependent areas (0.1%). The majority of respondents (67.7%) lived in urban areas. Most respondents reported having private health insurance and visiting a healthcare provider within the past 12 months (73.4% and 87.3% respectively). Those with unknown HIV status accounted for 14.3% of the sample. Compared to participants with known HIV-negative status, this group was slightly younger and had lower education. MSM with unknown HIV status also reported fewer sexual partners, and included a lower proportion of men who identify as homosexual.
Table I.
Characteristics of 2014 American Men’s Internet Survey (AMIS) participants with Unknown and Known-Negative HIV status (n=9,170)
| Participant Characteristic | Total (n=9,170) | Unknown HIV status (n=1,314) | Known-Negative HIV status (n=7,856) |
|---|---|---|---|
| Age (years) | |||
| Range | 15 – 101 | 15 – 101 | 15 – 86 |
| Mean (standard deviation) | 40.7 (14.3) | 35.1 (16.4) | 41.6 (13.6) |
| N (%) | N (%)* | N (%)* | |
| Race | |||
| White | 6,879 (75.9) | 995 (75.7) | 5,884 (74.9) |
| American Indian/Alaska Native | 72 (0.8) | 9 (0.7) | 63 (0.8) |
| Asian/Hawaiian/Pacific Islander | 221 (2.4) | 41 (3.1) | 180 (2.3) |
| Black | 355 (3.9) | 46 (3.5) | 309 (3.9) |
| Hispanic/Latino | 1,214 (13.4) | 178 (13.6) | 1,036 (13.2) |
| Other/multiple | 323 (3.6) | 35 (2.7) | 288 (3.7) |
| Region | |||
| Northwest | 1,597 (17.4) | 219 (16.7) | 1,378 (17.5) |
| Midwest | 1,967 (21.5) | 342 (26.0) | 1,625 (20.7) |
| South | 3,540 (38.6) | 478 (36.4) | 3,062 (39.0) |
| West | 2,055 (22.4) | 274 (20.9) | 1,781 (22.7) |
| US Territory | 11 (0.1) | 1 (0.1) | 10 (0.1) |
| Rural/Urban Residence | |||
| Urban | 6,098 (67.7) | 718 (54.6) | 5,371 (68.4) |
| Rural | 2,909 (32.3) | 566 (43.1) | 2,343 (29.8) |
| Education | |||
| Less than high school graduate | 174 (1.9) | 87 (6.6) | 87 (1.1) |
| High school graduate/GED | 758 (8.3) | 224 (17.1) | 534 (6.8) |
| Some college/Technical degree | 2,861 (31.5) | 485 (36.9) | 2,376 (30.2) |
| College graduate and beyond | 5,291 (58.3) | 492 (37.4) | 4,799 (61.1) |
| Health Insurance | |||
| No insurance | 806 (9.3) | 106 (8.1) | 700 (8.9) |
| Private insurance | 6,364 (73.4) | 809 (61.6) | 5,555 (70.7) |
| Public insurance | 773 (8.9) | 138 (10.5) | 635 (8.1) |
| Other/Multiple insurance | 731 (8.4) | 127 (9.7) | 604 (7.7) |
| Visited healthcare provider in last 12 months | |||
| Yes | 7,353 (87.3) | 945 (71.9) | 6,408 (81.6) |
| No | 1052 (12.5) | 265 (20.2) | 787 (10.0) |
| Sexual Identity | |||
| Heterosexual | 159 (1.4) | 67 (5.1) | 92 (1.2) |
| Homosexual | 6,837 (81.2) | 806 (61.3) | 6,031 (76.8) |
| Bisexual | 1342 (15.9) | 308 (23.4) | 1034 (13.2) |
| Sexually active in last 12 months | |||
| Yes | 8,191 (89.3) | 1,045 (11.4) | 7,146 (77.9) |
| No | 911 (10.0) | 255 (19.4) | 656 (8.4) |
| Number of sex partners in last 12 months (among sexually active respondents) | |||
| Range | 1 – 1,334 | 1 – 480 | 1 – 1,334 |
| Mean (standard deviation) | 9.3 (27.2) | 6.5 (23.3) | 9.68 (27.7) |
| Sex with a female in last 12 months | |||
| Yes | 1,070 (11.7) | 229 (17.4) | 841 (10.7) |
| No | 8,045 (88.3) | 1,074 (81.7) | 6,971 (88.7) |
| Sexually transmitted infection in last 12 months | |||
| Yes | 624 (6.8) | 36 (2.7) | 588 (7.5) |
| No | 8,546 (93.2) | 1,278 (97.3) | 7,268 (92.5) |
| Serosorting - only sex with HIV-negative men | |||
| Yes | 4,368 (47.6) | 549 (41.8) | 3,819 (48.6) |
| No | 4,802 (52.4) | 765 (58.2) | 4,037 (51.4) |
| Ever injected drugs | |||
| Yes | 296 (3.2) | 20 (1.5) | 276 (3.5) |
| No | 8,837 (96.7) | 1,286 (97.9) | 7,551 (96.1) |
| Non-injection drug use in last 12 months | |||
| Yes | 2,488 (27.3) | 299 (22.8) | 2,189 (27.9) |
| No | 6,601 (72.4) | 1,005 (76.5) | 5,596 (71.2) |
column percentages do not sum to 100% due to missing data
The results of the logistic regression analyses comparing MSM with unknown HIV status to HIV-negative MSM are shown in Table II. All bivariate associations between unknown HIV status and the predictor variables were significant at a p-value <.20 and were entered into the initial model for backward elimination. Of the demographic variables analyzed, region of residence dropped out of the model. Of the two healthcare indicators, visiting a healthcare provider in the past 12 months remained significant, but health insurance status dropped out of the adjusted model. Several of the sexual identity and sexual risk behavior predictors that had significant bivariate associations with unknown HIV status were dropped after controlling for the other covariates in the adjusted model, leaving only disclosure status, sexual position, and type of sexual activity as significant. Perceived peer HIV testing behavior was not significantly associated with unknown HIV status in the adjusted model.
Table II.
Bivariate and multivariate logistic regression models predicting Unknown HIV Status among men who have sex with men (MSM)
| Bivariate models | Final adjusted model* | |||||
|---|---|---|---|---|---|---|
| Participant Characteristic | OR | 95% CI | p-value | OR | 95% CI | p-value |
| Age (years) | 0.97 | 0.96–0.97 | <.001 | 0.96 | 0.96–0.97 | <.001 |
| Race | ||||||
| White | Ref. | -- | -- | Ref. | -- | -- |
| American Indian/Native Alaskan | 0.85 | 0.42–1.70 | .638 | 0.66 | 0.31–1.39 | .273 |
| Asian/Hawaiian/Pacific Islander | 1.35 | 0.95–1.90 | .091 | 1.21 | 0.83–1.77 | .332 |
| Black | 0.88 | 0.64–1.21 | .431 | 0.56 | 0.39–0.79 | .001 |
| Hispanic/Latino | 1.02 | 0.86–1.21 | .857 | 0.72 | 0.59–0.87 | .001 |
| Other/multiple | 0.72 | 0.50–1.03 | .070 | 0.48 | 0.32–0.71 | <.001 |
| Region | ||||||
| Northwest | 1.03 | 0.85–1.25 | .739 | |||
| Midwest | 1.37 | 1.12–1.63 | <.001 | |||
| South | 1.02 | 0.87–1.19 | .858 | |||
| West | Ref. | -- | --- | |||
| US Territory | 0.65 | 0.08–5.10 | .684 | |||
| Education | ||||||
| Less than high school graduate | 9.75 | 7.15–13.32 | <.001 | 5.45 | 3.86–7.70 | <.001 |
| High school graduate/GED | 4.09 | 3.41–4.91 | <.001 | 2.58 | 2.11–3.15 | <.001 |
| Some college/Technical degree | 1.99 | 1.74–2.28 | <.001 | 1.64 | 1.42–1.89 | <.001 |
| College grad graduate and beyond | Ref. | -- | -- | Ref. | -- | -- |
| Rural Residence | ||||||
| Yes | 1.81 | 1.60–2.04 | <.001 | 1.54 | 1.35–1.75 | <.001 |
| No | Ref. | -- | -- | Ref. | -- | -- |
| Health Insurance | ||||||
| Private insurance | Ref. | -- | -- | |||
| Public insurance | 1.49 | 1.22–1.82 | <.001 | |||
| Other/Multiple insurance | 1.44 | 1.18–1.77 | <.001 | |||
| No insurance | 1.04 | 0.84–1.29 | .725 | |||
| Visited healthcare provider in last 12 months | ||||||
| Yes | 0.56 | 0.47–0.65 | <.001 | 0.57 | 0.48–0.67 | <.001 |
| No | Ref. | -- | -- | Ref. | -- | -- |
| Sexual Identity | ||||||
| Heterosexual | 2.45 | 1.74–3.43 | <.001 | |||
| Homosexual | Ref. | -- | -- | |||
| Bisexual | 0.45 | 0.39–0.52 | <.001 | |||
| Disclosure status (“outness”) | ||||||
| Disclosed | Ref. | -- | -- | Ref. | -- | -- |
| Not disclosed | 4.79 | 3.96–5.79 | <.001 | 3.70 | 2.99–4.59 | <.001 |
| Ever had sex with a female | ||||||
| Yes | 1.77 | 1.51–2.07 | <.001 | |||
| No | Ref. | -- | -- | |||
| Sexual position | ||||||
| Identifies as top | 0.90 | 0.76–1.06 | .194 | 1.01 | 0.84–1.20 | .957 |
| Identifies as bottom | 1.41 | 1.22–1.64 | <.001 | 1.45 | 1.24–1.70 | <.001 |
| Identifies as versatile | Ref. | -- | -- | Ref. | -- | -- |
| Number of sex partners last 12 months | 0.97 | 0.97–0.98 | <.001 | |||
| Type of sex last 12 months | ||||||
| Oral only | 0.77 | 0.63–0.94 | .011 | 0.83 | 0.66–1.04 | .107 |
| Anal only | 0.74 | 0.33–1.63 | .450 | 0.43 | 0.17–1.12 | .085 |
| Both oral and anal | 0.32 | 0.27–0.38 | <.001 | 0.28 | 0.23–0.34 | <.001 |
| None | Ref. | -- | -- | Ref. | -- | -- |
| Condomless anal intercourse (CAI) in last 12 months (n=8,191) | ||||||
| CAI with partner of unknown HIV status | 1.28 | 1.02–1.61 | .034 | |||
| No CAI with partner of unknown status | Ref. | -- | -- | |||
| CAI with HIV-positive partner | 0.34 | 0.20–0.55 | <.001 | |||
| No CAI with HIV-positive partner | Ref. | -- | -- | |||
| CAI with HIV-negative partner | 0.68 | 0.42–1.10 | .115 | |||
| No CAI with HIV-negative partner | Ref. | -- | -- | |||
| Sexually transmitted infection in last 12 months | ||||||
| Yes | 0.35 | 0.25–0.49 | <.001 | |||
| No | Ref. | -- | -- | |||
| Serosorting - only sex with HIV-negative men | ||||||
| Yes | 0.76 | 0.67–0.85 | <.001 | |||
| No | Ref. | -- | -- | |||
| Ever injected drugs | ||||||
| Yes | 0.43 | 0.27–0.67 | <.001 | |||
| No | Ref. | -- | -- | |||
| Non-injection drug use in last 12 months | ||||||
| Yes | 0.76 | 0.66–0.87 | <.001 | |||
| No | Ref. | -- | -- | |||
| Peers get HIV tested every 3–6 months | ||||||
| Neither Agree/Disagree | Ref. | -- | -- | |||
| Strongly Agree | 0.46 | 0.32–0.67 | <.001 | |||
| Agree | 0.70 | 0.57–0.87 | <.001 | |||
| Disagree | 0.75 | 0.62–0.90 | .003 | |||
| Strongly Disagree | 1.05 | 0.81–1.37 | .712 | |||
logistic regression model adjusting for all other covariates using Full Information Maximum Likelihood estimation
In the final adjusted model using FIML estimation, younger age (OR=0.96, 95% CI=0.96–0.97) was associated with unknown HIV status. Translating this to a more interpretable scale, the regression coefficient was converted to a 10-year age scale resulting in an OR=0.71 for each 10-year increase in age. Regarding race, participants reporting Black (OR=0.56, 95% CI=0.39–0.79), Hispanic/Latino (OR=0.72, 95% CI=0.59–0.87), or other/multiple race (OR=0.48, 95% CI=0.32–0.71) were less likely to have unknown HIV status compared to White participants. MSM living in rural areas were more likely to have unknown HIV status compared to those living in urban areas (OR=1.54, 95% CI=1.35–1.75). Odds of unknown HIV status also increased as education level decreased, with men receiving less than a high school education being over 5 times more likely to have unknown status than men with a college degree (OR=5.45, 95% CI=3.86–7.70). Of the healthcare-related factors assessed, seeing a healthcare provider in the last 12 months was associated with a decreased odds of unknown HIV status (OR=0.57, 95% CI=0.48–0.67). The two sexual identity factors that retained significance in the final model were disclosure status and sexual position. Men who had not disclosed their sexual orientation and men who reported their sexual role as “bottom” were more likely to have unknown HIV status compared to the reference group (OR=3.70, 95% CI=2.99–4.59 and OR=1.45, 95% CI=1.24–1.70 respectively). Finally, men reporting both oral and anal sex in the last 12 months were significantly less likely to have unknown HIV status than men reporting no sexual activity (OR=0.28, 95% CI=0.23–0.34).
Results from the post-hoc comparison of HIV-negative MSM who tested more than a year ago versus those who tested within the last year are shown in Table III. Men who did not test within the past year were more likely to be older in age (OR=1.02, 95% CI=1.01–1.02) and less likely to be Asian/Hawaiian/Pacific Islander than White (OR=0.63, 95% CI=0.42–0.95). They were also less likely to have seen a healthcare provider in the last 12 months (OR=0.34, 95% CI=0.29–0.41), were less likely to report oral or anal sex in the past year (OR=0.23, 95% CI=0.19–0.29), had fewer sex partners (OR=0.98, 95% CI=0.98–0.99), were less likely to report sex with an HIV-positive partner (OR=0.59, 95% CI=0.43–0.82), were less likely to have had an STI in the last year (OR=0.34, 95% CI=0.22–0.42), and were less likely to agree that their MSM peers test regularly for HIV (OR=0.45, 95% CI=0.38–0.53).
Table III.
Bivariate and multivariate logistic regression models comparing HIV-negative men who have sex with men (MSM) who tested for HIV > 1 year ago (n=2,400) vs ≤ 1 year ago (n=4,566)
| Bivariate models | Final adjusted model* | |||||
|---|---|---|---|---|---|---|
| Participant Characteristic | OR | 95% CI | p-value | OR | 95% CI | p-value |
| Age (years) | 1.03 | 1.02–1.03 | <.001 | 1.02 | 1.01–1.02 | <.001 |
| Race | ||||||
| White | Ref. | -- | -- | Ref. | -- | -- |
| American Indian/Native Alaskan | 0.06 | 0.28–0.91 | .020 | 0.65 | 0.32–1.35 | .249 |
| Asian/Hawaiian/Pacific Islander | 0.52 | 0.37–0.73 | <.001 | 0.63 | 0.42–0.95 | .026 |
| Black | 0.83 | 0.66–1.03 | .086 | 0.78 | 0.57–1.07 | .119 |
| Hispanic/Latino | 0.66 | 0.58–0.76 | <.001 | 0.91 | 0.76–1.08 | .283 |
| Other/multiple | 0.80 | 0.63–1.02 | .072 | 0.82 | 0.61–1.10 | .184 |
| Region | ||||||
| Northwest | 1.12 | 0.97–0.29 | .128 | |||
| Midwest | 1.28 | 1.12–1.47 | <.001 | |||
| South | 1.03 | 0.92–1.16 | .593 | |||
| West | Ref. | -- | -- | |||
| US Territory | 0.41 | 0.09 −1.89 | .252 | |||
| Education | ||||||
| Less than high school graduate | 1.07 | 0.97–1.18 | .198 | |||
| High school graduate/GED | 1.01 | 0.85 −1.21 | .899 | |||
| Some college/Technical degree | 0.72 | 0.46 −1.14 | .162 | |||
| College graduate and beyond | Ref. | -- | -- | |||
| Rural Residence | ||||||
| Yes | 1.23 | 1.11–1.35 | <.001 | |||
| No | Ref. | -- | -- | |||
| Health Insurance | ||||||
| Private insurance | Ref. | -- | -- | Ref. | -- | -- |
| Public insurance | 1.19 | 1.03–0.39 | .023 | 0.83 | 0.67–1.02 | .073 |
| Other/Multiple insurance | 1.22 | 1.04–1.43 | .017 | 0.87 | 0.71–1.08 | .209 |
| No insurance | 0.96 | 0.81–1.15 | .666 | 0.64 | 0.50–0.82 | <.001 |
| Visited healthcare provider in last 12 months | ||||||
| Yes | 0.43 | 0.37–0.50 | <.001 | 0.34 | 0.29–0.41 | <.001 |
| No | Ref. | -- | -- | Ref. | -- | -- |
| Sexual Identity | ||||||
| Heterosexual | 3.44 | 2.13–5.57 | <.001 | |||
| Homosexual | Ref. | -- | -- | |||
| Bisexual | 0.97 | 0.84–1.11 | .640 | |||
| Disclosure status (“outness”) | ||||||
| Disclosed | Ref. | -- | -- | |||
| Not disclosed | 1.34 | 1.04–1.72 | .022 | |||
| Ever had sex with a female | ||||||
| Yes | 0.90 | 0.77–1.05 | .178 | |||
| No | Ref. | -- | -- | |||
| Sexual position | ||||||
| Identifies as top | 0.99 | 0.88–1.12 | .868 | |||
| Identifies as bottom | 1.10 | 0.98–1.20 | .112 | |||
| Identifies as versatile | Ref. | -- | -- | |||
| Number of sex partners last 12 months | 0.98 | 0.98 −0.99 | <.001 | 0.98 | 0.98–0.99 | <.001 |
| Type of sex last 12 months | ||||||
| Oral only | 0.42 | 0.33–0.52 | <.001 | 0.39 | 0.31–0.51 | <.001 |
| Anal only | 0.38 | 0.18–0.78 | .009 | |||
| Both oral and anal | 0.19 | 0.16–0.23 | <.001 | 0.23 | 0.19–0.29 | <.001 |
| None | Ref. | -- | -- | Ref. | -- | -- |
| Condomless anal intercourse (CAI) in last 12 months (n=8,191) | ||||||
| CAI with partner of unknown HIV status | 0.59 | 0.52–0.68 | <.001 | |||
| No CAI with partner of unknown status | Ref. | -- | -- | |||
| CAI with HIV-positive partner | 0.88 | 0.74–1.03 | .108 | 0.59 | 0.43–0.82 | .002 |
| No CAI with HIV-positive partner | Ref. | -- | -- | Ref. | -- | -- |
| CAI with HIV-negative partner | 0.22 | 0.19–0.25 | <.001 | |||
| No CAI with HIV-negative partner | Ref. | -- | -- | |||
| Sexually transmitted infection in last 12 months | ||||||
| Yes | 0.40 | 0.34–0.48 | <.001 | 0.34 | 0.22–0.42 | <.001 |
| No | Ref. | -- | -- | Ref. | -- | -- |
| Serosorting - only sex with HIV-negative men | ||||||
| Yes | 0.90 | 0.82–1.00 | .042 | |||
| No | Ref. | -- | -- | |||
| Ever injected drugs | ||||||
| Yes | 1.52 | 1.23–1.86 | <.001 | |||
| No | Ref. | -- | -- | |||
| Non-injection drug use in last 12 months | ||||||
| Yes | 0.78 | 0.71–0.87 | <.001 | |||
| No | Ref. | -- | -- | |||
| Peers get HIV tested every 3–6 months | ||||||
| Neither Agree/Disagree | Ref. | -- | -- | Ref. | -- | -- |
| Agree | 0.45 | 0.39–0.51 | <.001 | 0.45 | 0.38–0.53 | <.001 |
| Disagree | 0.97 | 0.85–1.11 | .671 | 1.06 | 0.91–1.22 | .464 |
logistic regression model adjusting for all other covariates using Full Information Maximum Likelihood estimation
Finally, descriptive analysis of high-risk behaviors showed that 2,087 (22.8%) men in the study sample reported behaviors in the last 12 months that placed them at increased risk for HIV infection. The most common high-risk behavior was having multiple sex partners but no HIV test in the last 12 months (39.2%), followed by history of STI (29.9%), and exchanging sex for money or drugs (25.5%) (see Table IV). Men engaging in high-risk behaviors were generally more likely to be HIV tested in the last 12 months than not, but some subgroups of high-risk MSM had substantial proportions of men not being tested. About half of high-risk MSM living in rural areas were not HIV tested in the last 12 months. Similarly, 47.1% of high-risk MSM living in the Midwest and 45.4% of those living in the South were not tested in the last 12 months. The majority of high-risk MSM visited a healthcare provider in the last 12 months (82.3%), and 53.9% of these men were offered an HIV test by their provider. Among high-risk MSM offered HIV testing by a provider, 77.7% reported being tested in the last 12 months.
Table IV.
HIV testing history among high-risk HIV-negative men who have sex with men (MSM), (n=2,087)
| Total* n=2,087 | Tested in the last 12 months^ n=1,016 (48.7%)† | Not tested in the last 12 months^ n=876 (42.0 %)† | |
|---|---|---|---|
| N (%) | N (%) | N (%) | |
| Risk reported in the last 12 months | |||
| Injection Drug Use | 75 (3.6) | 58 (77.3) | 12 (16.0) |
| Condomless anal sex with HIV-positive partner | 438 (21.0) | 367 (83.8) | 42 (9.6) |
| Exchange sex for money/drugs | 532 (25.5) | 312 (58.7) | 117 (22.0) |
| Multiple (> 1) sex partners AND no HIV test in last 12 months | 819 (39.2) | -- | 819 (100.0) |
| History of sexually transmitted infection | 624 (29.9) | 499 (80.0) | 47 (7.5) |
| Age – mean (standard deviation) | 39.59 (13.2) | 38.86 (12.6) | 40.91 (13.2) |
| Race | |||
| White | 1,505 (72.1) | 706 (46.9) | 661 (43.9) |
| American Indian/Native Alaskan | 18 (0.9) | 13 (72.2) | 5 (27.8) |
| Asian/Hawaiian/Pacific Islander | 52 (2.5) | 35 (67.3) | 12 (23.1) |
| Black | 85 (4.1) | 41 (48.2) | 33 (38.8) |
| Hispanic/Latino | 338 (16.2) | 179 (53.0) | 127 (37.6) |
| Other/multiple | 65 (3.1) | 34 (52.3) | 24 (36.9) |
| Region of residence | |||
| Northwest | 335 (16.1) | 170 (50.8) | 130 (38.8) |
| Midwest | 440 (21.1) | 189 (43.0) | 207 (47.1) |
| South | 778 (37.3) | 355 (43.1) | 353 (45.4) |
| West | 533 (25.5) | 301 (56.5) | 186 (34.9) |
| US Territory | 1 (0.1) | 1 (100.0) | 0 (0.0) |
| Education | |||
| Less than high school graduate | 33 (1.6) | 13 (39.4) | 13 (39.4) |
| High school graduate/GED | 167 (8.0) | 72 (43.1) | 66 (39.4) |
| Some college/Technical degree | 706 (33.8) | 320 (45.3) | 319 (45.2) |
| College graduate and beyond | 1,169 (55.9) | 606 (51.8) | 475 (40.6) |
| Rural residence | 598 (28.7) | 225 (37.6) | 302 (50.5) |
| Urban residence | 1,446 (69.3) | 770 (53.3) | 555 (38.4) |
| Visited healthcare provider | 1,717 (82.3) | 913 (53.2) | 660 (38.4) |
| Offered HIV test by healthcare provider | 926 (53.9) | 719 (77.7) | 149 (16.1) |
percentages reflect proportion of all high-risk participants
percentages reflect proportion among participants with given risk factor
percentages do not sum to 100% due to missing data
DISCUSSION
The results of this analysis show several consistencies with previous studies examining unknown HIV status. First, the association between younger age and unknown HIV status has been well-documented and is likely related to increased cumulative opportunities for HIV testing as age increases.(19, 20, 23, 27, 28) This finding, however, is concerning because young MSM have a disproportionately high incidence of HIV infection in the US. In 2015, 13–24 year olds accounted for 22% of all new HIV infections. Of these new cases, 81% were gay or bisexual males.(1) Increasing serostatus awareness in this group should be a key priority for HIV testing programs and prevention services. Second, the increased odds of unknown HIV status among residents of rural areas has also been previously reported.(19, 28) This suggests a lack of access to testing services outside large, urban centers or lower perceived risk of HIV in rural areas. Third, lower education level has been known to predict unknown HIV status and underscores the importance of HIV education and awareness components of prevention programs. Fourth, history of a recent visit to a healthcare provider was associated with lower odds of unknown HIV status, which has been shown in both MSM and non-MSM populations in the US.(19, 23) This indicates that engagement with the healthcare system provides greater opportunities for HIV testing and supports the rationale behind routine HIV screening in healthcare settings as a strategy to increase serostatus awareness.
This analysis also identified new factors associated with unknown HIV status among MSM. This is the first study to find that that men who identify their sexual role as “bottom,” typically indicating their sexual position as the receptive partner during anal intercourse, are more likely to have unknown HIV status compared to men who identify as “top” or “versatile.” This is troubling because the risk of acquiring HIV through unprotected anal intercourse is higher for the receptive anal sex partner.(29, 30) Thus, men who self-identify their sexual role as “bottom” might be expected to test more frequently for HIV because their sexual position carries greater risk, but this was not the case. Further research into sexual positioning and the meaning of self-reported labels such as “top” and “bottom” is necessary to fully understand the implications of this association. The finding that MSM who have not disclosed their sexual orientation are more likely to have unknown HIV status, a finding that has also been identified in some European studies, (22, 27) highlights the importance of social support and acceptance in the MSM community. Men who are more comfortable disclosing their sexual preferences toward other men may be more likely to self-identify as members of the MSM community, better identify their risk behaviors, and recognize the importance of HIV testing. They may also receive social support from other MSM who reinforce the importance of HIV testing. This could help to explain this study’s finding that MSM were more likely to have tested recently if they perceived that their peers test regularly for HIV. Conversely, MSM who have not disclosed their sexual orientation may feel stigmatized and therefore be less likely to access services targeted toward MSM or disclose their sexual risks to healthcare providers.(31)
This study also detected an association between race and unknown HIV status. MSM who reported being of Black, Hispanic/Latino, or other/multiple race were less likely to have unknown status compared to White MSM. This is likely due to the result of increased HIV testing among minority groups, who are less likely to report never testing for HIV.(32) It should be noted, however, that lower odds of unknown HIV status does not suggest minority MSM are less likely have an undiagnosed HIV infection. It is more likely a reflection of testing services often targeted toward minority populations, and perhaps it is the proportion of those populations who are not being tested that remain unaware of their infection.
Interestingly, aside from differences in unknown HIV status between men engaging in both oral and anal sex versus men who were not sexually active in the past 12 months, none of the other sexual risk factors retained significance in the adjusted model. This suggests that risky sexual behaviors such as condomless anal intercourse and multiple sex partners do not necessarily predict HIV testing history among MSM, and perhaps risk level is not the primary factor in the decision to seek out an initial HIV test. Rather, psychosocial factors related to sexual preference disclosure and sexual positioning may be more important predictors.(33, 34)
With regards to MSM who do have a history of HIV testing, however, the post-hoc analysis indicates that there are distinct differences in risk behaviors between men who tested more than 12 months ago and those who tested within the past 12 months. Men who tested over a year ago had fewer sex partners, were less likely to be sexually active within the past year, were less likely to have a recent STI, and were less likely to report sex with an HIV-positive partner. These results are consistent with other reports suggesting that HIV testing frequency among men who have a history of HIV testing is dictated by level of sexual risk.(35) MSM may not undergo annual testing without experiencing a risk event. MSM testing more than a year ago were also older in age, and were less likely to have seen a healthcare provider compared to recent testers. It is possible that men did not test because they did not have a recent visit with a healthcare provider who may have recommended HIV testing as part of routine care. Thus, the decision to undergo an initial HIV test may have more to do with psychosocial factors, whereas the decision to undergo repeat testing is based more on an individual’s assessment of their perceived risk. However, even though MSM engaging in risky behaviors may be more likely to seek frequent HIV testing, the analysis of high-risk groups shows that some subgroups are still not testing despite their risks. Among MSM who reported exchanging sex for money or drugs in the past year, 22.0% did not have an HIV test in the last 12 months. Among injection drug users in this sample, 16.0% were not tested in the last 12 months. This demonstrates the need for continued targeted testing efforts in order to reach risk groups who may be missed through traditional testing mechanisms and would stand to benefit from expanded testing programs.
The major strength of this study is the large sample size associated with the AMIS survey which provides robust evidence for the statistical associations identified in this analysis. This study is also the first to explore such a broad range of correlates for unknown HIV status among MSM in the US. The inclusion of sexual identity, sexual risk behaviors, and perceived peer testing behaviors provides new insight into the complexity of factors that predict HIV testing behaviors among MSM. Another strength of this analysis is the use of FIML to address missing data within the sample. Nonresponse is a chief limitation in survey research, but FIML provides a robust statistical method of estimation without dropping observations such as in listwise deletion. While missing data may have affected the model selection procedure, all available data were incorporated into the final model estimates presented.
A primary limitation of this study is that the sample is comprised of a predominantly White, highly-educated group of MSM engaged in social networking via the internet. Thus, the results of this analysis may not be generalizable to other MSM populations. Additionally, data in the AMIS study are obtained by self-report and may subject to certain biases. For example, participants may underreport certain risk behaviors that are viewed as unfavorable resulting in social desirability bias. Accuracy of responses may also be affected by recall bias, although this is unlikely to be a significant factor since most risk behaviors assessed in this analysis were measured over the past 12 months. Another limitation of this study is the cross-sectional design, precluding the identification of causal relationships between variables. For example, it cannot be determined if certain health-seeking or sexual risk behaviors are influenced by HIV status awareness, or if serostatus awareness leads to changes in such behaviors. Finally, the use of stepwise model selection has certain disadvantages including possible spurious associations resulting from multiple hypothesis testing, the risk of overfitting the data, and the potential for biased parameter estimates. Although this method is reasonable given the exploratory nature of the research question, interpretation of model estimates should be made with caution. Despite these limitations, the results of this study provide support for further research, specifically on the association of sexual identity and sexual role on HIV testing behaviors.
CONCLUSIONS
This study provides supporting evidence that young age, race, low education, rural residence, and absence of recent healthcare visits are significant factors associated with unknown HIV status among MSM. Additionally, it identifies the influence of sexual orientation disclosure and sexual positioning on the decision to test for HIV. Men who have not disclosed their sexual orientation and men who identify their sexual role as “bottom” are less likely to be tested for HIV than other MSM, yet they may be at higher risk for HIV due to sexual risk behaviors and stigmatizing social factors. This points to the need for additional research on the psychosocial factors associated with sexual identity, sexual risk, and entry into the continuum of HIV prevention services. Among MSM who have a history of HIV testing, recent HIV testing is more closely related to risk behavior, suggesting that repeat testing behaviors among MSM are consistent with HIV testing recommendations.
Acknowledgements:
The data used in this study were derived from the American Men’s Internet Survey conducted by PRISM Health at Emory University.
Funding: The data for this analysis were collected through funding received from the National Institutes of Health (Emory Center for AIDS Research, P30AI050409) and the MAC AIDS Fund.
Footnotes
Conflict of Interest: The authors declare that they have no conflict of interest.
Ethical Approval: All procedures performed as part of the American Men’s Internet Survey study involving human participants were conducted in accordance with the ethical standards of the Emory University Institutional Review Board (IRB) and in compliance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this secondary analysis of de-identified data, formal consent was not required as determined by the University of Miami IRB.
References
- 1.Centers for Disease Control and Prevention. HIV Surveillance Report, 2015; vol. 27 http://www.cdc.gov/hiv/library/reports/hiv-surveillance.html. Published November 2016. Accessed June 15, 2017. [Google Scholar]
- 2.Skarbinski J, Rosenberg E, Paz-Bailey G, Hall HI, Rose CE, Viall AH, et al. Human immunodeficiency virus transmission at each step of the care continuum in the United States. JAMA Intern Med. 2015;175(4):588–96. [DOI] [PubMed] [Google Scholar]
- 3.Khosropour CM, Dombrowski JC, Kerani RP, Katz DA, Barbee LA, Golden MR. Changes in condomless sex and serosorting among men who have sex with men after HIV diagnosis. J Acquir Immune Defic Syndr. 2016;73(4):475–81. [DOI] [PubMed] [Google Scholar]
- 4.Rodger AJ, Lampe FC, Grulich AE, Fisher M, Friedland G, Phanuphak N, et al. Transmission risk behaviour at enrolment in participants in the INSIGHT Strategic Timing of AntiRetroviral Treatment (START) trial. HIV Med. 2015;16 Suppl 1:64–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vallabhaneni S, McConnell JJ, Loeb L, Hartogensis W, Hecht FM, Grant RM, et al. Changes in seroadaptive practices from before to after diagnosis of recent HIV infection among men who have sex with men. PLoS ONE. 2013;8(2):e55397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gorbach PM, Drumright LN, Daar ES, Little SJ. Transmission behaviors of recently HIV-infected men who have sex with men. J Acquir Immune Defic Syndr. 2006;42(1):80–5. [DOI] [PubMed] [Google Scholar]
- 7.Steward WT, Remien RH, Higgins JA, Dubrow R, Pinkerton SD, Sikkema KJ, et al. Behavior change following diagnosis with acute/early HIV infection-a move to serosorting with other HIV-infected individuals. The NIMH Multisite Acute HIV Infection Study: III. AIDS Behav. 2009;13(6):1054–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eaton LA, Kalichman SC. Changes in transmission risk behaviors across stages of HIV disease among people living with HIV. J Assoc Nurses AIDS Care. 2009;20(1):39–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wolitski RJ, MacGowan RJ, Higgins DL, Jorgensen CM. The effects of HIV counseling and testing on risk-related practices and help-seeking behavior. AIDS Educ Prev. 1997;9(3 Suppl):52–67. [PubMed] [Google Scholar]
- 10.Attia S, Egger M, Muller M, Zwahlen M, Low N. Sexual transmission of HIV according to viral load and antiretroviral therapy: systematic review and meta-analysis. AIDS. 2009;23(11):1397–404. [DOI] [PubMed] [Google Scholar]
- 11.Rodger AJ, Cambiano V, Bruun T, Vernazza P, Collins S, van Lunzen J, et al. Sexual activity without condoms and risk of HIV transmission in serodifferent couples when the HIV-positive partner is using suppressive antiretroviral therapy. JAMA. 2016;316(2):171–81. [DOI] [PubMed] [Google Scholar]
- 12.Cohen MS, Chen YQ, McCauley M, Gamble T, Hosseinipour MC, Kumarasamy N, et al. Antiretroviral therapy for the prevention of HIV-1 transmission. N Engl J Med. 2016;365(6):493–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Donnell D, Baeten JM, Kiarie J, Thomas KK, Stevens W, Cohen CR, et al. Heterosexual HIV-1 transmission after initiation of antiretroviral therapy: a prospective cohort analysis. Lancet. 2010;375(9731):2092–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cohen MS, Chen YQ, McCauley M, Gamble T, Hosseinipour MC, Kumarasamy N, et al. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med. 2011;365(6):493–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.White House Office of National AIDS Policy. National HIV/AIDS Strategy for the United States: Updated to 2020. Published July 2015.
- 16.Hall HI, An Q, Tang T, Song R, Chen M, Green T, et al. Prevalence of diagnosed and undiagnosed HIV infection--United States, 2008–2012. MMWR. 2015;64(24):657–62. [PMC free article] [PubMed] [Google Scholar]
- 17.Mackellar DA, Hou SI, Whalen CC, Samuelsen K, Sanchez T, Smith A, et al. Reasons for not HIV testing, testing intentions, and potential use of an over-the-counter rapid HIV test in an internet sample of men who have sex with men who have never tested for HIV. Sex Transm Dis. 2011;38(5):419–28. [DOI] [PubMed] [Google Scholar]
- 18.Finlayson TJ, Le B, Smith A, Bowles K, Cribbin M, Miles I, et al. HIV risk, prevention, and testing behaviors among men who have sex with men--National HIV Behavioral Surveillance System, 21 U.S. cities, United States, 2008. MMWR Surveill Summ. 2011;60(14):1–34. [PubMed] [Google Scholar]
- 19.Margolis AD, Joseph H, Belcher L, Hirshfield S, Chiasson MA. ‘Never testing for HIV’ among men who have sex with men recruited from a sexual networking website, United States. AIDS Behav. 2012;16(1):23–9. [DOI] [PubMed] [Google Scholar]
- 20.Veinot TC, Caldwell E, Loveluck J, Arnold MP, Bauermeister J. HIV testing behavior and social network characteristics and functions among young men who have sex with men (YMSM) in Metropolitan Detroit. AIDS Behav. 2016;20(11):2739–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.White D, Stephenson R. Correlates of perceived HIV prevalence and associations with HIV testing behavior among men who have sex with men in the United States. Am J Mens Health. 2016;10(2):90–9. [DOI] [PubMed] [Google Scholar]
- 22.den Daas C, Doppen M, Schmidt AJ, Op de Coul E. Determinants of never having tested for HIV among MSM in the Netherlands. BMJ Open. 2016;6(1):e009480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Conserve DF, Oraka E, Abara WE, Wafula E, Turo A. Correlates of never testing for HIV among Non-Hispanic Black men in the United States: National Survey of Family Growth, 2011–2013. AIDS Behav. 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sanchez T, Zlotorzynska M, Sineath C, Kahle E, Sullivan P. The annual American Men’s Internet Survey of behaviors of men who have sex with men in the United States: 2014 Key Indicators Report. JMIR Public Health Surveill. 2016;2(1):e23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.SAS Institute, Inc., SAS 9.3.1 Help and Documentation, Cary, NC: 2002–2004. [Google Scholar]
- 26.Muthén LK, & Muthén BO Mplus User’s Guide, 7th ed Los Angeles, CA: 1998–2015. [Google Scholar]
- 27.Carvalho C, Fuertes R, Lucas R, Martins A, Campos MJ, Mendao L, et al. HIV testing among Portuguese men who have sex with men--results from the European MSM Internet Survey (EMIS). HIV Med. 2013;14 Suppl 3:15–8. [DOI] [PubMed] [Google Scholar]
- 28.Zablotska I, Holt M, de Wit J, McKechnie M, Mao L, Prestage G. Gay men who are not getting tested for HIV. AIDS Behav. 2012;16(7):1887–94. [DOI] [PubMed] [Google Scholar]
- 29.Jin F, Crawford J, Prestage GP, Zablotska I, Imrie J, Kippax SC, et al. HIV risk reduction behaviours in gay men: Unprotected anal intercourse, risk reduction behaviours, and subsequent HIV infection in a cohort of homosexual men. AIDS. 2009;23(2):243–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Beyrer C, Baral SD, van Griensven F, Goodreau SM, Chariyalertsak S, Wirtz AL, et al. Global epidemiology of HIV infection in men who have sex with men. Lancet. 2012;380(9839):367–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pachankis JE, Hatzenbuehler ML, Hickson F, Weatherburn P, Berg RC, Marcus U, et al. Hidden from health: structural stigma, sexual orientation concealment, and HIV across 38 countries in the European MSM Internet Survey. AIDS. 2015;29(10):1239–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Johnson BA, McKenney J, Ricca AV, Rosenberg ES, Liu C, Sharma A, et al. Risk factors associated with repeated HIV testing among internet-using men who have sex with men. AIDS Educ Prev. 2016;28(6):511–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dangerfield DT 2nd, Smith LR, Williams J, Unger J, Bluthenthal R. Sexual positioning among men who have sex with men: a narrative review. Arch of Sex Behav. 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.White D, Stephenson R. Identity formation, outness, and sexual risk among gay and bisexual men. Am J Mens Health. 2014;8(2):98–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Witzel TC, Melendez-Torres GJ, Hickson F, Weatherburn P. HIV testing history and preferences for future tests among gay men, bisexual men and other MSM in England: results from a cross-sectional study. BMJ Open. 2016;6(9):e011372. [DOI] [PMC free article] [PubMed] [Google Scholar]