Abstract
IMPORTANCE
Metabolomics is the broad and parallel study of metabolites within an organism and provides a contemporaneous snapshot of physiologic state. Use of metabolomics in the clinical setting may help achieve precision medicine for those who have experienced trauma, where diagnosis and treatment are tailored to the individual patient.
OBJECTIVE
To examine whether metabolomics can (1) distinguish healthy volunteers from trauma patients and (2) quantify changes in catabolic metabolites over time after injury.
DESIGN, SETTING, AND PARTICIPANTS
Prospective cohort study with enrollment from September 2014 to May 2015 at an urban, level 1 trauma center. Included in the study were 10 patients with severe blunt trauma admitted within 12 hours of injury with systolic blood pressure less than 90 mm Hgor base deficit greater than 6 mEq/Land 5 healthy volunteers. Plasma samples (n = 35) were obtained on days 1, 3, and 7, and they were analyzed using mass spectrometry.
MAIN OUTCOMES AND MEASURES
Principal component analyses, multiple linear regression, and paired t tests were used to select biomarkers of interest. A broad-based metabolite profile comparison between trauma patients and healthy volunteers was performed. Specific biomarkers of interest were oxidative catabolites.
RESULTS
Trauma patients had a median age of 45 years and a median injury severity score of 43 (interquartile range, 34–50). Healthy fasting volunteers had a median age of 33 years. Compared with healthy volunteers, trauma patients showed oxidative stress on day 1: niacinamide concentrations were a mean (interquartile range) of 0.95 (0.30–1.45) relative units for trauma patients vs 1.06 (0.96–1.09) relative units for healthy volunteers (P = .02), biotin concentrations, 0.43 (0.27–0.58) relative units for trauma patients vs 1.21 (0.93–1.56) relative units for healthy volunteers (P = .049); and choline concentrations, 0.17 (0.09–0.22) relative units for trauma patients vs 0.21 (0.18–0.22) relative units for healthy volunteers (P = .004). Trauma patients showed lower nucleotide synthesis on day 1: adenylosuccinate concentrations were 0.08 (0.04–0.12) relative units for trauma patients vs 0.15 (0.14–0.17) relative units for healthy volunteers (P = .02) and cytidine concentrations were 1.44 (0.95–1.73) relative units for trauma patients vs 1.74(1.62–1.98) relative units for healthy volunteers (P = .05). From trauma day 1 to day 7, trauma patients showed increasing muscle catabolism: serine levels increased from 42.03 (31.20–54.95) μM to 79.37 (50.29–106.37) μM (P = .002), leucine levels increased from 69.21 (48.36–99.89) μM to 114.16 (92.89–143.52) μM (P = .004), isoleucine levels increased from 20.43 (10.92–27.41) μM to 48.72 (36.28–64.84) μM (P < .001), and valine levels increased from 122.56 (95.63–140.61) μM to 190.52 (136.68–226.07) μM (P = .004). There was an incomplete reversal of oxidative stress.
CONCLUSIONS AND RELEVANCE
Metabolomics can function as a serial, comprehensive, and potentially personalized tool to characterize metabolism after injury. A targeted metabolomics approach was associated with ongoing oxidative stress, impaired nucleotide synthesis, and initial suppression of protein metabolism followed by increased nitrogen turnover. This technique may provide new therapeutic and nutrition targets in critically injured patients.
Precision medicine is the most recent evolution in health care,1,2 where diagnosis and treatment are specifically tailored to an individual patient rather than to a broader category of disease. The fields of genomics, proteomics, and now metabolomics provide the tools for systems biology investigations, detailed biological insights, and ultimately personalized medicine. Metabolomics is the high-throughput study of small molecules such as amino acids, nitrates, sugars, and lipids. Within an organism, these metabolites provide an instantaneous view of the physiologic state and represent the ultimate downstream products of the genome and its interaction with the environment.3 Traditional laboratory medicine has relied on individual biomarkers, but metabolomics is a systemwide approach, involving simultaneous assessment of the substrate/product balance in multiple metabolic pathways.4 Mass spectrometry (MS) or nuclear magnetic resonance metabolomics analyses of biofluids have the potential to quantify metabolic states in individuals to better characterize disease and treatment response. Prior studies have identified metabolic markers in biofluids, which have helped to provide earlier diagnoses and improved treatment monitoring for various cancers and common critical illnesses.4–12 These studies have shown that hundreds of metabolites can be quantified per biologic sample, but data interpretation is difficult in the context of complex biologic pathway interactions.4 More research is needed to explore the reliability, validity, and interpretability of these methods.
Although trauma-related morbidity and mortality have steadily decreased over the past decade,13 many trauma patients still succumb to the ultimate consequences of overactive immune responses, catabolism, oxidation, and multiple-organ failure.14–16 Metabolomics may be the ideal tool to rapidly characterize, anticipate, and ameliorate these acute changes in the trauma patient’s metabolism and energy needs. However, to our knowledge, there are few studies that have applied metabolomics to critically injured patients.17–19 In this study, our goal was to assess the ability of broad-based, targeted metabolomics to characterize the metabolic response to injury. We hypothesized that metabolomics would reliably distinguish healthy volunteers from trauma patients, and that it would be able to quantify changes in catabolic and oxidative metabolites over time after injury.
Methods
Study Design, Participants, and Setting
Blunt trauma patients admitted to Harborview Medical Center in Seattle, Washington, from September 2014 to May 2015 were recruited, provided written informed consent, and were enrolled in this prospective cohort study. Inclusion criteria were (1) age between 18 and 60 years, (2) enrollment within 12 hours of injury, and (3) circulatory shock, defined as hypotension (systolic blood pressure <90 mm Hg) or base deficit of 6 mEq/L or greater. Five healthy volunteers were also recruited from among the surgery staff, provided written informed consent, and enrolled as a comparison group. Individuals were excluded from either group if they were pregnant, obese (body mass index [BMI, calculated as weight in kilograms divided by height in meters squared] ≥35), or had a history of cancer, prior organ dysfunction, or endocrine disorders. Trauma patients were excluded if they required an exploratory laparotomy, if they had a prolonged nothing-by-mouth period, or if they required parenteral nutrition. These exclusion criteria were designed to minimize confounding variables, consistent with the precedents established in other metabolomics studies.4,19 All study procedures were approved by the University of Washington institutional review board.
Sample Collection and Processing
Venous blood samples were obtained in 5-mL K2 EDTA Vacutainer tubes (BD) within 24 hours of injury and on hospital days 3 and 7. Healthy volunteers donated samples the morning after 12 hours or more of fasting. Samples were immediately placed on ice and centrifuged at 2500g for 10 minutes within 1 hour of collection. The plasma supernatant was then removed, divided into 0.5-mL aliquots, and stored at −80°C. In addition, medical records for trauma patients were reviewed to obtain hospital-based plasma glucose levels acquired within 4 hours of venous blood sampling for this study.
Metabolite Measurements
Plasma samples were thawed for MS-based analysis in 2 processing batches of15 and 20 samples. Detailed protocols for metabolomics analyses are reported in the eAppendix in the Supplement. Briefly, samples were analyzed using a targeted liquid chromatography and quadrupole MS (Sciex 5500 Qtrap) approach, and metabolites were identified by comparison with known standards to obtain relative concentrations. All processing batches included 3 quality-control (QC) samples derived from the same pooled serum sample. All metabolite signals were normalized to these standardized QCs using the mean QC value in each batch, consistent with established techniques.20 A total of 210 metabolites were assayed (eTable 1 in the Supplement).
Exposures and Outcomes
Our primary exposures were participant type (trauma patient or volunteer) and time since injury. The main outcome measures were the plasma metabolite concentrations. Specific biomarkers of interest were oxidation products and protein catabolites. For most metabolites, quantified results were recorded as relative concentrations. For some metabolites, such as amino acids and some organic acids, absolute quantification (micromolar) was possible because isotope-labeled internal standards for these compounds have been developed.21,22
Statistical Analysis and Data Presentation
Continuous data are presented as medians with interquartile ranges (IQRs). Categorical data are presented as counts and percentages.
Initial broad metabolic comparisons between day-1 trauma samples and healthy volunteer samples and among trauma samples over time were made using principal component analysis (PCA),4 with oblique rotation to allow for sample clustering, as appropriate. To partially assess the validity of metabolomic techniques in a trauma population, Pearson correlation was used to assess concordance between hospital-based plasma measures and metabolome-based plasma measures for glucose.
In addition, a supervised subset of 5 day-1 trauma samples was selected in an attempt to match these samples to volunteer samples on the basis of age, sex, and BMI. Statistical inference for this comparison was based on postestimation Wald tests from linear regression with robust SEs for each metabolite; the model included exposure to trauma as the independent variable, metabolite concentration as the outcome variable, and categorical variables for both BMI and processing batch as covariates. A comparison between trauma samples from day 1 and day 7 was also performed; a paired t test for each metabolite was used for statistical inference in this comparison. The Benjamini-Hochberg false discovery rate correction23 was applied to all P values to account for multiple testing, and statistical significance was set at α < .05. All statistical analyses were performed using Stata version 12.1 (StataCorp) and the R software environment version 3.2.3 (R Foundation for Statistical Computing).24 Metabolite expression heat maps were generated using GENE-E software 3.0.2 (Broad Institute of MIT and Harvard University),25 with scaling of relative concentrations by row. Metaboanalyst 3.0 software (Metabolomics Innovation Center)26 incorporates both enrichment analysis and topology analysis and was used for all pathway-level analyses.
Results
In total, 30 plasma samples from 10 trauma patients and 5 plasma samples from 5 healthy fasting volunteers were obtained. In these samples, 102 metabolites were reliably identified and quantified out of a possible 210 measured. The trauma cohort was severely injured, as evidenced by relatively high injury severity scores (median, 43; IQR, 34–50) and long length of hospital stays (median, 31 days; IQR, 15–50) (Table). All patients required mechanical ventilation on admission, and all were treated with enteral nutrition in the first week. The mean coefficient of variance for relative concentrations of QC samples was 4.4% and for isotope-labeled internal standards, the coefficient of variance was 2.8%. Hospital-based measures of plasma glucose and MS-based measures of glucose demonstrated good correlation (r = 0.84; eFigure in the Supplement).
Table.
Demographic and Clinical Characteristics for the Cohort of Blunt Trauma Patients and Healthy Volunteers Who Underwent Plasma Sampling for Metabolomic Analysis
| Characteristic | Trauma Patients (n = 10) |
Healthy Volunteers (n = 5) |
|---|---|---|
| Plasma samples, No. | 30 | 5 |
| Age, median (IQR), y | 45 (30–55) | 33 (29–36) |
| Female, No. (%) | 4 (40) | 3 (60) |
| White, No. (%) | 10 (100) | 5 (100) |
| BMI, median (IQR) | 26.5 (22.1–27.5) | 23.7 (23.2–24.1) |
| Injury mechanism, No. (%) | ||
| MVC occupant | 5 (50) | NA |
| Pedestrian struck | 3 (30) | NA |
| Fall | 1 (10) | NA |
| Motorcycle crash | 1 (10) | NA |
| ISS, median (IQR) | 43 (34–50) | NA |
| Severe injury (AIS >4), No. (%) | ||
| Head | 3 (30) | NA |
| Chest | 4 (40) | NA |
| Abdomen | 3 (30) | NA |
| Extremity | 8 (80) | NA |
| Admission, median (IQR) | ||
| BD, mEq/L | 6.8 (3.1–12.3) | NA |
| SBP, mmHg | 75 (66–81) | NA |
| Blood products, median (IQR)a | 9.5 (8–13) | NA |
| ICU LOS, median (IQR) | 14 (9–18) | NA |
| Duration ventilation, median (IQR) | 8.5 (4–15) | NA |
| Hospital LOS, median (IQR) | 31 (15–50) | NA |
Abbreviations: AIS, Abbreviated Injury Score; BD, base deficit; BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); ICU, intensive care unit; IQR, interquartile range; ISS, Injury Severity Score;
LOS, length of stay; MVC, motorvehicle crash; NA, not applicable; SBP, systolic blood pressure.
Units of total blood products.
Plasma Metabolomics in Trauma Patients and Healthy Volunteers at Early and Later Points
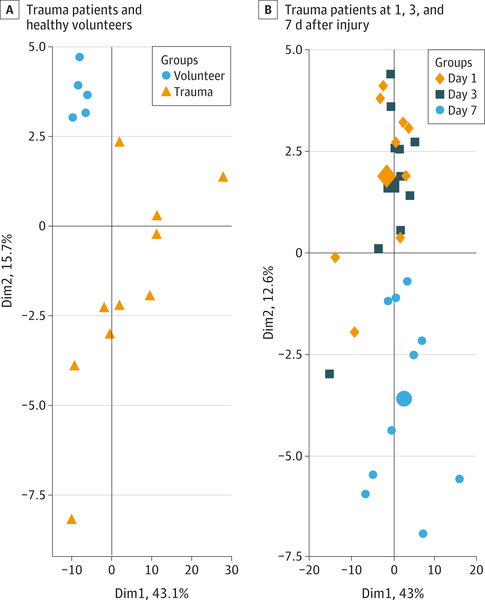
Principal component analysis is a pattern recognition method that selects groups of metabolites that best explain the variance in the data.4 Principal component analysis of plasma metabolic profiles for day-1 trauma patients and healthy volunteers demonstrated consistent differences between groups (Figure 1A). The first principle component accounted for 43% of the total variance among samples from trauma patients and healthy volunteers. Changes in methionine, citrulline, pipecolate, threonine, and isoleucine concentrations contributed to the separation between groups. The second principle component accounted for an additional 16% of the total variance among samples from trauma patients and healthy volunteers. Glucoronate, N-acetylneuraminate, aconitate, hydroxygluta-rate, and acetylcarnitine contributed to the separation. The metabolites from these 2 components are involved in either amino acid metabolism, sugar metabolism, or fatty acid oxidation.
Figure 1. Principle Component Analysis of Plasma Samples.
Each data point represents an individual plasma sample. Points that are close together represent samples with similar metabolic phenotypes; points that are far away have dissimilar metabolic phenotypes. B, The large symbols represent the mean values of the sample scores for a given group. Dim indicates dimension.
Principal component analysis of trauma patient samples on days 1 (referent), 3, and 7 also demonstrated differences in metabolic profiles over-time (Figure 1B). The first principle component showed a good separation for the day-7 patients, with cysteine, hippuric acid, maleic acid, arginine, and urate contributing to the distinction. All of these metabolites are involved in glucose metabolism, amino acid metabolism, or oxidative stress.
Metabolites that did not vary significantly among sub-groups included those involved in bile acid degradation, gut microflora metabolism, and the pentose phosphate pathway.
Oxidative Stress, Nucleotide Synthesis, and Protein and Fatty Acid Turnover in Trauma Patients
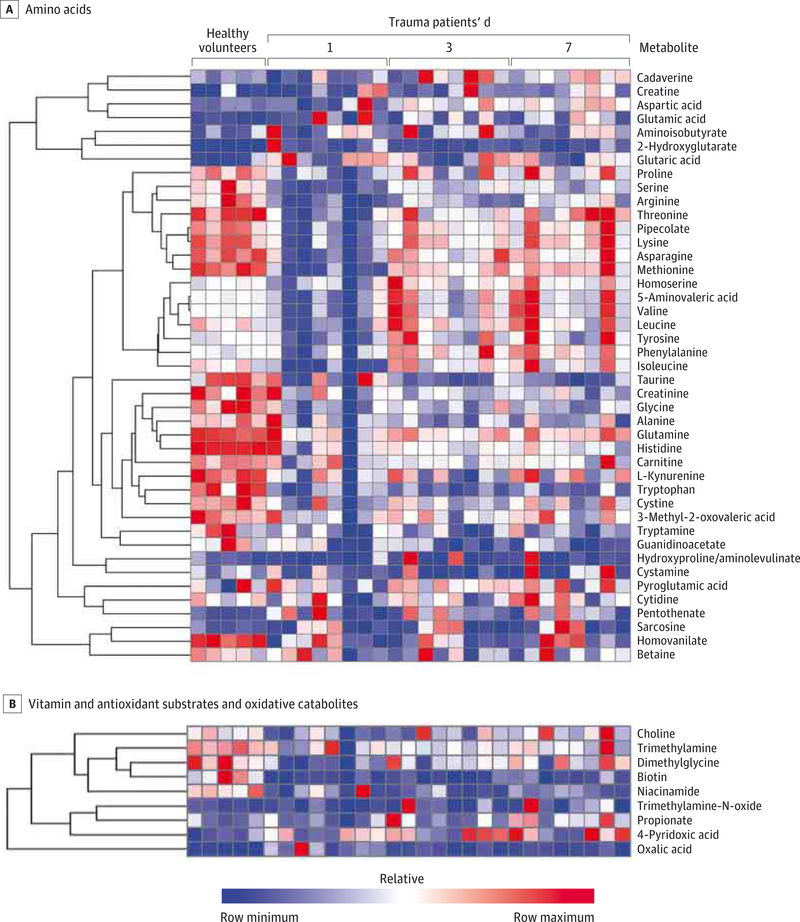
We next determined which patterns of metabolite levels differed among trauma patients and healthy volunteers and among trauma patients over time after injury. First, relative to healthy volunteers, trauma patients initially showed lower levels of circulating amino acids (Figure 2A) and higher concentrations of oxidative stress metabolites and vitamin catabolites (Figure 2B).
Figure 2. Plasma Metabolome Heat Maps for Trauma Patients and Healthy Volunteers.
The heat maps demonstrate (A) relatively decreased amino acids, with a rise over time, and (B) reduced vitamin and antioxidant substrates (niacinamide and biotin) and increased oxidative catabolites (propionate and 4-pyridoxic acid). The heat maps are generated using mass spectrometry-based metabolomics. Two trauma patients are omitted owing to high concentrations, which obscure all other relative concentrations.
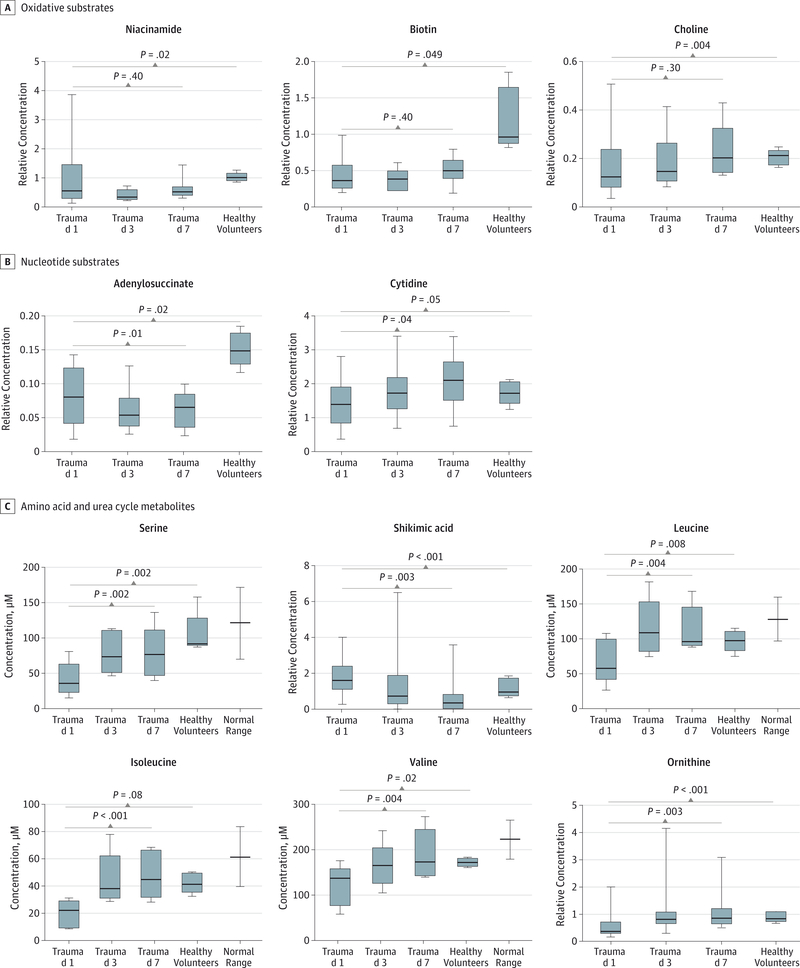
A supervised subset of trauma patients was exactly matched to volunteers on the basis of sex (60% female) and race (100% white), and they were well-matched on the basis of nutritional status (all fasting about 12 hours), BMI (median, 26.3 vs 23.7), and age (median, 33 years vs 31 years). A comparison of individual quantified metabolites demonstrated that trauma patients had lower levels of oxidative substrates relative to healthy volunteers (mean [IQR], niacinamide concentrations: 0.95 [0.30–1.45] relative units for trauma patients vs 1.06 [0.96–1.09] relative units for healthy volunteers, P = .02; biotin concentrations: 0.43 [0.27–0.58] relative units for trauma patients vs 1.21 [0.93–1.56] relative units for healthy volunteers, P = .049; and choline concentrations: 0.17 [0.09–0.22] relative units for trauma patients vs 0.21 [0.18–0.22] relative units for healthy volunteers, P = .004; Figure 3A). Trauma patients showed relatively lower substrates for ribonucleic acid and DNA synthesis (mean [IQR], adenylosuccinate concentrations: 0.08 [0.04–0.12] relative units fortrauma patients vs 0.15 [0.14–0.17] relative units for healthy volunteers, P = .02; and cytidine concentrations: 1.44 [0.95–1.73] relative units for trauma patients vs 1.74 [1.62–1.98] relative units for healthy volunteers, P = .05; Figure 3B). Finally, as time from injury increased, there was a progressive elevation in urea cycle products and circulating amino acids, particularly in branched-chain amino acids (BCAAs) (mean [IQR], ornithine levels increased from 0.59 [0.30–0.65] relative units to 1.10 [0.67–1.22] relative units, P = .003; serine levels increased from 42.03 [31.20–54.95] μM to 79.37 [50.29–106.37] μM, P = .002; shkimic acid levels decreased from 1.90 [1.29–2.07] relative units to 0.74 [0.12–0.83] relative units, P = .003; leucine levels increased from 69.21 [48.36–99.89] μM to 114.16 [92.89–143.52] μM, P = .004; isoleucine levels increased from 20.43 [10.92–27.41] μM to 48.72 [36.28–64.84] μM, P < .001; andvaline levels increased from 122.56 [95.63–140.61] μM to 190.52 [136.68–226.07] μM, P = .004; Figure 3C).
Figure 3. Plasma Concentrations in Blunt Trauma Patients and Healthy Volunteers.
Generated using mass spectrometry-based metabolomics. Box and whisker plots display median, interquartile range, maximum, and minimumvalues. Comparisons between trauma patients and healthy volunteers are based on multiple regression with adjustments for body mass indexand processing batch. Comparisons between day-1 and day-7 trauma patients are based on paired t tests. Multiple testing is accounted for by a Benjamini-Hochberg false discovery rate method.23
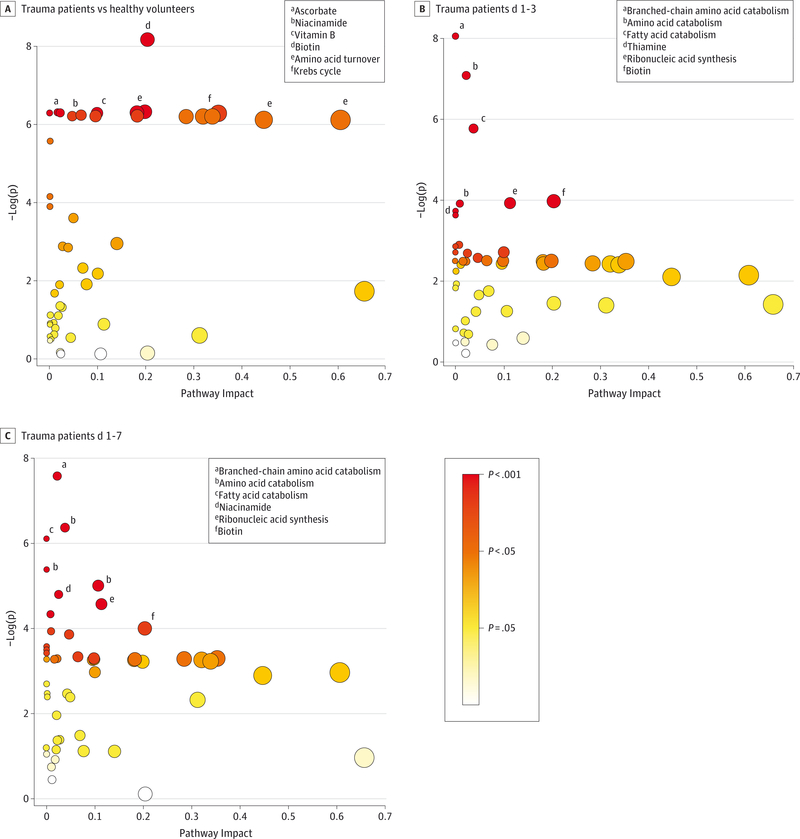
Pathway analysis results were consistent with the afore-mentioned individual metabolite results. Relative to volunteers, trauma patients showed alterations in pathways associated with vitamins and antioxidants, ribonucleic acid/DNA synthesis, and amino acids (Figure 4A). Over time, trauma patients demonstrated alterations in pathways associated with BCAAs, oxidative products, and ribonucleic acid synthesis (Figure 4B and C).
Figure 4. Pathway Analyses of the Plasma Metabolome.
Generated using mass spectrometry-based metabolomics. Each data point represents a biologic pathway with quantified plasma metabolites. Based on the available metabolite’s centrality and importance to the pathway of interest, a level of pathway impact is assigned. Larger pathway impact is communicated by moving rightward on the x-axis and by increasing diameter of circles. Based on paired differences in metabolite concentration between comparison groups, a P value is assigned to the pathway. A more significant result is communicated by moving upward on they-axis and by a color scale, which goes from white (low significance) to yellow to orange to red (high significance).
In total, after false discovery rate correction, 36 of 102 identified metabolites showed statistically significant variation between day-1 trauma patients and healthy volunteers (eTable 2 in the Supplement), and 15 showed statistically significant variation between day-1 and day-7 trauma patients (eTable 3 in the Supplement).
Discussion
Patient families and caregivers alike are often caught off-guard when an expected recovery after traumatic injury is displaced by a turn toward multiple-organ failure, prolonged critical illness, and even death. Despite similar injury patterns and evidence-based treatments, individual patients may follow entirely different clinical paths. A more comprehensive understanding of the individual patient’s metabolic response to trauma may help us understand this perplexing variation in outcomes. Metabolomics, the study of the small molecules involved in metabolism, may provide an improved tool for directing more real-time, personalized, and comprehensive treatment of an individual trauma patient with rapidly changing physiology. Calorimetry and nitrogen-balance studies help characterize metabolism in critically ill patients, but these methods are relatively resource-and time-intensive. Using MS-based metabolomics, data acquisition for a targeted metabolite panel can now be achieved in less than 3 hours, making bedside application a real possibility.27 Metabolomics could help move beyond algorithm-based care and help better understand which critically injured patients actually require additional enteral calories, antioxidant supplementation, fluid boluses, or transfusions. Indeed, metabolomics may help realize the goals of precision medicine1,2 in the trauma setting, whereby health is improved by matching treatment to the needs of the individual.
We demonstrated that MS-based metabolomics can identify, quantify, and measure trends of metabolites for key biologic pathways following traumatic injury. There are no gold-standard measurements to help validate plasma metabolomics in this setting, but for at least 1 marker (glucose), a relatively good association was shown between hospital-based methods and MS-based methods. The 2 glucose measurements were taken within 4 hours of each other in all cases; if all samples were obtained at exactly the same time, the agreement would likely have been greater.
Overall, plasma metabolomics demonstrated that amino acid turnover and nucleotide metabolism are suppressed in the initial phase of injury, followed by a gradual increase to normal and even catabolic levels. This is consistent with the original ebb-flow hypothesis of the metabolic response to injury proposed by Cuthbertson28 in 1932. Under this construct, initial metabolism is globally suppressed following traumatic injury and then progresses to a hyperactive, catabolic state within 24 hours. Using MS-based methods in the current study, we have found evidence that not only supports the ebb-flow hypothesis, but also identifies specific metabolites and metabolic pathways that may be integral to this phenomenon.
Specifically, as time from injury increased, the concentration of most circulating amino acids and urea cycle products progressively rose, suggesting a transition in metabolism from initial suppression to muscle catabolism. This finding is consistent with prior literature that linked postinjury cortisol and epinephrine levels to hyperaminoacidemia29 and perturbations in BCAA metabolism.19 In our study cohort, concentrations of BCAAs (valine, leucine, and isoleucine) were particularly elevated, and these amino acids are considered specific to proteolysis.30–32 Over the first week following injury, a severely injured individual’s energy requirement is nearly double the baseline energy requirement in an uninjured individual.16 Following injury, muscle catabolism yields BCAAs to serve as carbon chains for unmet energy needs; valine is converted to carbohydrates, leucine to fats, and isoleucine to fats and carbohydrates.30,31,33 In addition, for all but 1 case, trauma patients were treated with enteral nutrition by day 3, and some of the observed changes in amino acids (apart from BCAAs) may be associated with the start of nutrition.
We also observed persistent upregulation of oxidative pathways and ongoing depletion of antioxidant stores in trauma patients. Circulating biotin, also known as vitamin B7, was low in the trauma patients’ plasma. Biotin is a cofactor in carboxylase-based reactions for proteolysis and is used in BCAA catabolism.33,34 Although biotin is a known cofactor in a myriad of other biologic pathways,34 interpretation of this marker in the context of other changes in the trauma metabolome supports the notion that biotin is depleted after injury due, in part, to increased demand for BCAA catabolism. Concentrations of niacinamide, another oxidation substrate and vitamin derivative, were lower in trauma patients. This metabolite is involved in nicotinamide adenine dinucleotide redox reactions, which are essential to the tricarboxylic acid cycle and oxidative phosphorylation.33 In addition, niacinamide is a known free-radical scavenger and antioxidant.35,36 Finally, choline was relatively depleted in trauma patient plasma. This is a product of oxidation reactions and a substrate for detoxification reactions using methyl donors.33 Interpreted as a whole, these data suggest that trauma patients have ongoing oxidative stress and high demand for antioxidants, and these derangements persist throughout the first week following injury.
Metabolomics represents an advance over other systemwide approaches, yet several limitations are relevant to the interpretation of our study and are related to the current capacities of MS and pathway analysis. First, individual metabolites may be involved in several pathways, and changes should be interpreted with caution. For a given metabolite, elevation in its concentration could represent increased production, decreased use, or both. The complexity and large volume of data produced in metabolomics may increase the chance for false-positives, which can be compounded by unmeasured confounding variables. For this reason, interpretation of individual metabolites must occur in concert with other biomarkers in the pathway of interest. This process is expected to improve because the field of metabolomics-based pathway analysis continues to evolve at a rapid pace.37 Second, close examination of the data from this study will reveal that not all patients in the cohort follow the exact pathway trends that we emphasized. Individual variation is the rule rather than the exception, and interpretation of this variation is limited by the small sample size. This study is an important first step toward identifying changes in metabolic pathways over-time after trauma. Larger independent studies are needed to confirm our results and characterize the typical trends and the important deviations. Third, it may be suspected that elevation in the plasma amino acid concentrations for this trauma cohort was simply due to initiation of enteral nutrition. However, this is unlikely to be the primary driver because gut absorption of amino acids is tightly regulated; most enteral amino acids are metabolized via a first pass in the liver and are thereby prevented from entering systemic circulation.38 Moreover, concentrations of BCAAs became elevated over time; BCAAs are relatively more specific to muscle catabolism30–32 and are not supplemented in our enteral formulas.
Conclusions
Metabolomics may provide a path for achieving more comprehensive and personalized trauma care. This technology can function as a rapid, serial, and precise tool to characterize metabolism in trauma patients as they recover from injury. Preliminary data from this study suggest that metabolism after injury is characterized by protein catabolism, persistent oxidative stress, and increases in nucleotide turnover. These insights may provide new therapeutic and nutrition targets in this population. Future studies of interventions in critically injured populations should be paired with plasma metabolome analyses to fully explore the potential of this novel technology.
Supplementary Material
Key Points.
Question
Can metabolomics help achieve precision medicine for critically ill trauma patients?
Findings
In this cohort study, 35 plasma samples from 10 patients with severe trauma and 5 healthy volunteers were analyzed. Mass spectrometry-based metabolomics was able to quantify and trend significantly elevated plasma markers for oxidative stress, muscle catabolism, and impaired nucleotide synthesis in trauma patients over time.
Meaning
Metabolomics is a rapid and comprehensive tool that can characterize metabolism after injury and may provide new personalized therapeutic targets in critically ill trauma patients.
Acknowledgments
Funding/Support: The University of Washington and Harborview Medical Center Department of Surgery provided funding for the data analysis.
Role of the Funder/Sponsor: The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Previous Presentation: This study was presented at the 87th Annuam Meeting of the Pacific Coast Surgical Association; February 14,2016; Kohala Coast, Hawaii.
Footnotes
Conflict of Interest Disclosures: Drs O’Keefe, Parent, and Raftery are University of Washington CoMotion patent holders for the use of metabolomics in trending recovery after trauma. No other disclosures were reported.
Contributor Information
Brodie A. Parent, Harborview Department of Surgery, University of Washington Medical Center, Seattle.
Max Seaton, Harborview Department of Surgery, University of Washington Medical Center, Seattle.
Ravi F. Sood, Harborview Department of Surgery, University of Washington Medical Center, Seattle.
Haiwei Gu, Mitochondria and Metabolism Center, Department of Anesthesiology and Pain Medicine, University of Washington, Seattle..
Danijel Djukovic, Mitochondria and Metabolism Center, Department of Anesthesiology and Pain Medicine, University of Washington, Seattle..
Daniel Raftery, Mitochondria and Metabolism Center, Department of Anesthesiology and Pain Medicine, University of Washington, Seattle..
Grant E. O’Keefe, Harborview Department of Surgery, University of Washington Medical Center, Seattle.
REFERENCES
- 1.Tanaka H Omics-based medicine and systems pathology: a new perspective for personalized and predictive medicine. Methods Inf Med 2010;49(2): 173–185. [DOI] [PubMed] [Google Scholar]
- 2.Hawgood S, Hook-Barnard IG, O’Brien TC, Yamamoto KR. Precision medicine: beyond the inflection point. Sci Transi Med 2015;7(300): 300ps17. [DOI] [PubMed] [Google Scholar]
- 3.Llorach R, Garcia-Aloy M, Tulipani S, Vazquez-Fresno R, Andres-Lacueva C. Nutrimetabolomic strategies to develop new biomarkers of intake and health effects. J Agric FoodChem 2012;60(36):8797–8808. [DOI] [PubMed] [Google Scholar]
- 4.Gowda GA, Zhang S, Gu H, Asiago V, Shanaiah N, Raftery D. Metabolomics-based methods for early disease diagnostics. Expert Rev Moi Diagn 2008;8 (5):617–633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Koeth RA, Wang Z, Levison BS, et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 2013; 19(5):576–585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhu J, Djukovic D, Deng L, et al. Colorectal cancer detection using targeted serum metabolic profiling. J Proteome Res 2014;13(9):4120–4130. [DOI] [PubMed] [Google Scholar]
- 7.Bos LD, Weda H, Wang Y, et al. Exhaled breath metabolomics as a noninvasive diagnostictool for acute respiratory distress syndrome. Eur Respir J 2014;44(1):188–19. [DOI] [PubMed] [Google Scholar]
- 8.Wang Z, Levison BS, Hazen JE, Donahue L, Li XM, Hazen SL. Measurement of trimethylamine-N-oxide by stable isotope dilution liquid chromatography tandem mass spectrometry. Anal Biochem 2014;455:35–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin ZY, Xu PB, Yan SK, et al. A metabonomic approach to early prognostic evaluation of experimental sepsis by (1)H NMR and pattern recognition. NMR Biomed 2009;22(6):601–608. [DOI] [PubMed] [Google Scholar]
- 10.Dai DF, Karunadharma PP, Chiao YA, et al. Altered proteome turnover and remodeling by short-term caloric restriction or rapamycin rejuvenate the aging heart. Aging Cell 2014;13(3): 529–539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sood RF, Gu H, Djukovic D, et al. Targeted metabolic profiling of wounds in diabetic and nondiabetic mice. Wound Repair Regen 2015;23(3): 423–434. [DOI] [PubMed] [Google Scholar]
- 12.Nicholson JK, Holmes E, Kinross JM, Darzi AW, Takats Z, Lindon JC. Metabolic phenotyping in clinical and surgical environments. Nature 2012; 491(7424):384–392. [DOI] [PubMed] [Google Scholar]
- 13.Cuschieri J, Johnson JL, Sperry J, et al. ; Inflammation and Host Response to Injury, Large Scale Collaborative Research Program. Benchmarking outcomes in the critically injured trauma patient and the effect of implementing standard operating procedures. Ann Surg 2012;255 (5):993–999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Keel M, Trentz O. Pathophysiology of polytrauma. Injury 2005;36(6):691–709. [DOI] [PubMed] [Google Scholar]
- 15.Hirsiger S, Simmen HP, Werner CM, Wanner GA, Rittirsch D. Danger signals activating the immune response after trauma. Mediators Inflamm 2012; 2012:315941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Plank LD, Hill GL. Sequential metabolic changes following induction of systemic inflammatory response in patients with severe sepsis or major blunt trauma. World J Surg 2000;24(6):630–638. [DOI] [PubMed] [Google Scholar]
- 17.Peltz ED, D’Alessandro A, Moore EE, et al. Pathologic metabolism: an exploratory study of the plasma metabolome of critical injury. J Trauma Acute Care Surg 2015;78(4):742–751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cohen MJ, Serkova NJ, Wiener-Kronish J, Pittet JF, Niemann CU. 1H-NMR-based metabolic signatures ofclinical outcomes in trauma patients: beyond lactate and base deficit. J Trauma 2010;69 (1):31–40. [DOI] [PubMed] [Google Scholar]
- 19.Mao H, Wang H, Wang B, et al. Systemic metabolic changes of traumatic critically ill patients revealed by an NMR-based metabonomic approach. J Proteome Res 2009;8(12):5423–5430. [DOI] [PubMed] [Google Scholar]
- 20.Kuligowski J, Sanchez-Illana A, Sanjuan-Herraez D, Vento M, Quintas G. Intra-batch effect correction in liquid chromatography-mass spectrometry using quality control samples and supportvectorregression(QC-SVRC). Analyst 2015;140(22):7810–781. [DOI] [PubMed] [Google Scholar]
- 21.Bueschl C, Krska R, Kluger B, Schuhmacher R. Isotopic labeling-assisted metabolomics using LC-MS. Anal Bioanal Chem 2013;405(1):27–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gu H, Du J, Carnevale Neto F, et al. Metabolomics method to comprehensively analyze amino acids in different domains. Analyst 2015;140 (8):2726–2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 1995;57(1):289–300. [Google Scholar]
- 24.R Development Core Team. R: A language and environment for statistical computing Vienna, Austria: R Foundation for Statistical Computing; http://www.R-project.org/ Published 2014. Accessed February 4, 2016. [Google Scholar]
- 25.Broad Institute of MIT; Harvard University. GENE-E. Version 3.0.204 http://www.broadinstitute.org/cancer/software/GENE-E/. Published 2015. Accessed January 17,2016.
- 26.Wishart D, Xia J. MetaboAnalyst 3.0: a comprehensive tool suite for metabolomic data analysis Alberta, Canada: The Metabolomics Innovation Center; http://www.metaboanalyst.ca/. Published 2015. Accessed November 14,2015. [Google Scholar]
- 27.Rinehart D, Johnson CH, Nguyen T, et al. Metabolomic data streaming for biology-dependent data acquisition. Nat Biotechnol 2014; 32(6):524–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cuthbertson D Observations on the disturbance of metabolism produced by injury to the limbs. Q J Med 1932;1:233–246. [Google Scholar]
- 29.Gelfand RA, Matthews DE, Bier DM, Sherwin RS. Role of counterregulatory hormones in the catabolic response to stress. J Clin Invest 1984;74 (6):2238–2248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cynober LA. Plasma amino acid levels with a note on membrane transport: characteristics, regulation, and metabolic significance. Nutrition 2002;18(9):761–766. [DOI] [PubMed] [Google Scholar]
- 31.Cals MJ, Bories PN, Devanlay M, et al. ; Research Group on Aging. Extensive laboratory assessment of nutritional status in fit, health-conscious, elderly people living in the Paris area. J Am Coll Nutr 1994;13(6):646–65. [DOI] [PubMed] [Google Scholar]
- 32.Le Boucher J, Cynober L. Protein metabolism and therapy in burn injury. Ann NutrMetab 1997;41 (2):69–82. [DOI] [PubMed] [Google Scholar]
- 33.Wishart DS, Jewison T, Guo AC, et al. HMDB 3.0: The Human Metabolome Database in 2013. Nucleic Acids Res 2013;41(database issue):D801–D80 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gravel RA, Narang MA. Molecular genetics of biotin metabolism: old vitamin, new science. J Nutr Biochem 2005;16(7):428–431. [DOI] [PubMed] [Google Scholar]
- 35.Sonee M, Martens JR, Mukherjee SK. Nicotinamide protects HCN2 cells from the free radical generating toxin, tertiary butylhydroperoxide (t-BuOOH). Neurotox Res 2002;4(7–8):595–599. [DOI] [PubMed] [Google Scholar]
- 36.Ruggieri S, Orsomando G, Sorci L, Raffaelli N. Regulation of NAD biosynthetic enzymes modulates NAD-sensing processes to shape mammalian cell physiology under varying biological cues. Biochim Biophys Acta 2015;1854(9):1138–1149. [DOI] [PubMed] [Google Scholar]
- 37.Alonso A, Marsal S, Julia A. Analytical methods in untargeted metabolomics: state of the art in 2015. Front Bioeng Biotechnol 2015;3:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Abumrad NN, Miller B. The physiologic and nutritional significance of plasma-free amino acid levels. JPEN J Parenter Enteral Nutr 1983;7(2):163–170. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.