Abstract
Background:
The VITamin D and OmegA-3 TriaL (VITAL) is a completed randomized, placebo-controlled trial of vitamin D3 (2000 IU/day) and marine omega-3 (1 g/day) supplements in the primary prevention of cancer and cardiovascular disease. Here we examine baseline and change in 25-hydroxyvitamin D (25(OH)D) and related biomarkers with randomized treatment and by clinical factors.
Methods:
Baseline 25(OH)D was measured in 15,804 participants (mean age 68 yrs; 50.8% women; 15.7% African Americans) and in 1,660 1-year follow-up samples using liquid chromatography-tandem mass spectrometry and chemiluminescence. Calcium and parathyroid hormone (iPTH) were measured by chemiluminescence and spectrophotometry respectively.
Results:
Mean baseline total 25(OH)D (ng/mL±SD) was 30.8±10.0 ng/mL, and correlated inversely with iPTH (r=−0.28), p<0.001. After adjusting for clinical factors, 25(OH)D (ng/mL±SE) was lower in men vs women (29.7±0.30 vs 31.4±0.30, p<0.0001) and in African Americans vs whites (27.9±0.29 vs 32.5±0.22, p<0.0001). It was also lower with increasing BMI, smoking, and latitude, and varied by season. Mean 1-year 25(OH)D increased by 11.9 ng/mL in the active group and decreased by 0.7 ng/mL in placebo. The largest increases were noted among individuals with low baseline and African Americans. Results were similar for chemiluminescent immunoassay. Mean calcium was unchanged, and iPTH decreased with treatment.
Conclusion:
In VITAL, baseline 25(OH)D varied by clinical subgroups, was lower in men and African Americans. Concentrations increased with vitamin D supplementation, with the greatest increases in those with lower baseline 25(OH)D. The seasonal trends in 25(OH)D, iPTH, and calcium may be relevant when interpreting 25(OH)D levels for clinical treatment decisions.
Keywords: 25-hydroxyvitamin D, Vitamin D, parathyroid hormone, VITAL trial, seasonal effects
Introduction
Vitamin D plays an important role in regulating calcium and phosphate metabolism for maintenance of metabolic functions and for skeletal health. Observational studies have reported associations between low levels of serum 25-hydroxyvitamin D (25(OH)D) and increased risk of cardiovascular disease (CVD), cancer, and other disorders. (1–4) There is controversy, however, on whether optimizing vitamin D levels can improve these non-skeletal clinical outcomes. (5) After absorption through the intestines or synthesis in the skin, vitamin D is metabolized in the liver to 25(OH)D and subsequently metabolized in the kidneys to the biologically active metabolite 1,25(OH)2 D which has a short half-life. Although other biomarkers on the vitamin D pathway (e.g. vitamin D binding protein, free and bioavailable 25(OH)D) might also be clinically important, clinical guidelines on vitamin D adequacy, insufficiency, and deficiency are based primarily on the total 25(OH)D. (1)
Serum 25(OH)D concentrations are influenced by numerous factors including season, latitude, skin pigmentation, and genetics. Other clinical risk factors include age, nutritional status and intake, body-mass index (BMI), race/ethnicity, and sex. (6,7) Similar to other steroid hormones, serum 25(OH)D is mostly bound to proteins, predominantly to vitamin D binding protein and to a lesser extent to albumin. (8) Assays differ in methods and reagents used to extract and measure vitamin D. (9) These technical challenges and biological variation in vitamin D levels may have broad public health implications, as the variability in vitamin D measurement can lead to differences in the measured 25(OH)D concentrations, which in turn could mean the difference between a clinical diagnosis of insufficiency, sufficiency, or even toxicity.
We recently reported the principal results of the VITamin D and OmegA-3 TriaL (VITAL), an NIH-funded, randomized, double-blind, placebo-controlled trial that tested high-dose (2000 IU/day) vitamin D among 25,871 U.S. men aged ≥50 and women aged ≥55, including 5,106 African Americans (10). Over a 5.3 year intervention period, vitamin D supplementation did not significantly reduce the primary endpoints of CVD or cancer, and no excess risks of hypercalcemia or other adverse events were identified.
The purpose of the current analysis is to examine the randomized treatment effects of daily vitamin D 2000 IU vs placebo on total serum 25(OH)D, as well as its components (25(OH)D3 and 25(OH)D2), and other vitamin D-related biomarkers including intact parathyroid hormone (iPTH), and calcium levels among 15,804 VITAL participants at baseline and on a subset of 1,660 participants at 1-year of follow-up. This was evaluated using two different vitamin D assays (liquid chromatography-tandem mass spectrometry (LC-MS/MS ) and an immunochemiluminescent assay), which were both calibrated using the vitamin D standardization program of the Centers for Disease Control and Prevention (CDC). Furthermore, we examined relationships of demographic and relevant clinical factors including age, sex, race/ethnicity, body mass index (BMI), latitude/geographic location, and season on baseline and change in serum 25(OH)D concentrations.
Materials and Methods
Study Population
VITAL is a nationwide, randomized, double-blind, placebo-controlled, 2×2 factorial trial of vitamin D (in the form of vitamin D3 (cholecalciferol), 2000 IU/day) and marine omega-3 fatty acid (Omacor® fish oil, eicosapentaenoic acid (EPA)+docosahexaenoic acid (DHA), 1 g/day) supplements in the primary prevention of cancer and CVD among 25,871 men and women, aged ≥50 and ≥55, respectively, with an oversampling of African Americans. (11,12) Participants were permitted up to 800 IU/d of additional vitamin D supplementation (non-randomized) and up to 1200 mg/d of elemental calcium from all sources. At the time of enrollment participants provided written informed consent, completed baseline questionnaires and were asked to provide a blood sample. Sex, age, race/ethnicity, use of non-randomized supplements or medications, smoking, alcohol consumption, history of hypertension, and other relevant health history were collected from baseline questionnaires. Self-reported weight and height were recorded, and body-mass index (BMI) was calculated. The study protocol was approved by the Partners Institutional Review Board, Boston. The first authors had full access to the data and take full responsibility for its integrity. All authors have read and agree to the manuscript as written.
Blood collection and Laboratory Analysis
Prior to randomization, participants were invited to provide a voluntary blood sample. A total of 16,903 (65.3 % of randomized) participants provided a (pre-randomization) blood sample at baseline between October 2010 and March 2014. As part of an ancillary biomarker study, a subset of participants who returned baseline blood during the winter or early spring of 2011-12 and 2012-13 were invited to additionally provide a follow-up blood sample one year later. Baseline collection in the follow-up cohort took place from November to April, and 1-year collection mainly from January to May. A total of 1,686 participants provided both baseline and 1-year follow-up samples.
A blood collection kit, including a freezer pack and an informed consent form was mailed to each willing participant. Samples were shipped overnight within 24 hours of venipuncture to the laboratory in Boston where they were centrifuged to separate plasma, serum, red blood cells, and buffy coat and stored in nitrogen freezers (−170°C) within 36 hours of venipuncture. To minimize potential batch effects, all baseline and 1-year sample pairs were shipped and measured at the same time in tandem, in a manner blinded to the analyzing labs.
Total 25(OH)D was measured in 15,804 baseline and 1,660 1-year samples using LC-MS/MS (Thermo Fisher, Quest Diagnostics Nichols Institute, Valencia, CA) with both 25(OH)D2 and D3 quantitated, and also summed as total serum 25(OH)D. Most of the baseline samples (n=13,149, 83%), including those from the 1-year cohort, plus all the 1-year follow-up samples, were also analyzed with a second assay in a separate laboratory (Atherotech Diagnostics, Inc., Birmingham, AL) using the Abbott ARCHITECT 25(OH)D chemiluminescent microparticle immunoassay (CMIA), referred to as Chemiflex. In addition, we measured intact PTH (iPTH) using a chemiluminescent-based assay (Beckman Coulter, Fullerton, CA) and calcium by spectrophotometry (Quest Diagnostics Nichols Institute, San Juan Capistrano, CA).
Quality Control and Calibration of Assays
In a pilot analysis on blinded duplicate assessments of 20 VITAL participant samples divided into two aliquots, the resulting coefficients of variations (CVs) were 4.1% for total 25(OH)D and 4.9% for 25(OH)D3 measured by LC-MS/MS, 3.6% for total 25(OH)D measured by CMIA, 7.6% for iPTH, and 2.0% for calcium.
We also participated in the vitamin D standardization program of the Centers for Disease Control and Prevention (CDC) in order to calibrate 25(OH)D results to the CDC reference method. The CDC vitamin D standardization program aims to standardize vitamin D measurements such that results are comparable over time and across various laboratories. The CDC created aliquots of authentic human serum material at specified low (19.7 ng/mL) and high (40.7 ng/mL) 25(OH)D concentrations (measured by the CDC gold standard) to support VITAL’s vitamin D assay measurements. We included 2.5% CDC standards, in pairs of low/high, blinded for the analyzing laboratories. CDC performance criteria, derived from biological variability, to evaluate laboratory performance imprecision and bias of measurements, recommended a bias of less than ±5.0% and a CV <10% for total 25(OH)D.(13) We derived batch specific calibration equations based on the measured 25(OH)D of the CDC QC samples and their “true” concentration as measured by the CDC reference method.(14) After calibration of CDC samples in batches with initial CVs outside their required ranges, we achieved CVs of 8.9% / 6.5 % (low/high) for the LC-MS/MS assay, and 3.3% / 2.7 % (low/high) for the Chemiflex assay. We applied the same calibration equation to all the study samples
Season Adjustment
Because blood 25(OH)D concentrations vary with sun exposure, we adjusted our analyses for season of blood draw. We estimated the seasonal influence based on pre-randomization levels of 7,502 VITAL participants who reported no (non-randomized) vitamin D supplement use at baseline. We fitted a linear model with one-year periodic sine and cosine functions of the date of blood draw as follows: (sin((2π÷365) × calendar day) and cos((2π÷365) × calendar day)) to the 25(OH)D levels. (15) Additionally we included age, sex, race/ethnicity, BMI, smoking status, and latitude of the participants’ residential zip code in the model. The model estimates resulted in a seasonal maximum on Aug 10, a seasonal minimum on February 10 and a peak-to-peak amplitude of 5.3 ng/mL (Figure 1). The individual measurements were adjusted by subtracting the estimated season effects (sine and cosine terms) from the measured 25(OH)D.
Figure 1:
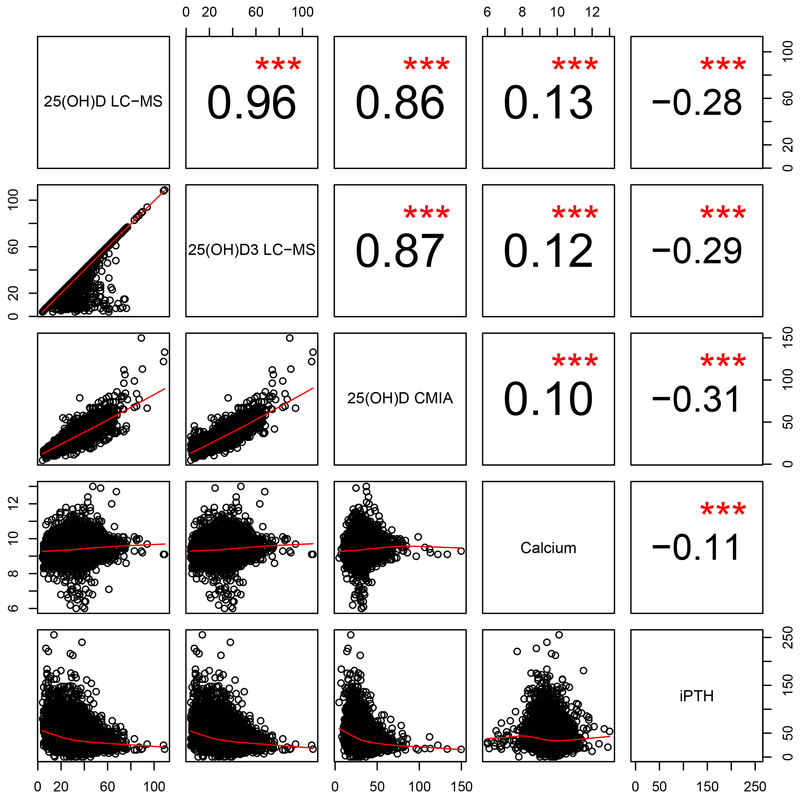
Spearman correlation coefficients between 25(OH)D total and 25(OH)D3 measured by LC-MS/MS (ng/mL), 25(OH)D total measured by chemiluminescent microparticle immunoassay (CMIA) (ng/mL), Calcium (mg/dL) and iPTH (pg/ml). Based on 15804 measurements at baseline.
Statistical Methods
Quantitative characteristics of the study population are shown as means and standard deviations (SD), and categorical variables are shown as numbers or percentages. For biomarker concentrations, medians and interquartile ranges (IQR) are also presented. Visual inspection of the untransformed and log-transformed distributions of 25(OH)D, calcium, and iPTH showed normal distributions for 25(OH)D, calcium and a slightly right-skewed distribution for iPTH, but we decided to keep iPTH untransformed for consistency. We assessed the correlation between biomarkers with Spearman correlation coefficients.
We evaluated the effects of randomized treatment on the biomarker absolute and percent changes from baseline to 1-year, using the natural logs of the biomarkers to calculate the geometric means of the percent changes over time. We used paired t-tests and Wilcoxon signed-rank tests to evaluate the change between baseline and 1-year concentrations within treatment groups. We used two-sample t-tests to compare changes in mean concentrations from baseline to 1-year between the randomized treatment groups. Based on clinical cut-points, 25(OH)D concentrations (ng/mL) were additionally categorized as <20, 20 to 29.9, and 30 or greater, and iPTH into two categories (≤65, >65 pg/mL); and we compared the change in frequency distribution of these clinical categories from baseline to 1-year between the treatment arms.
We used linear regression to evaluate associations of baseline 25(OH)D with a priori specified clinical characteristics (age, sex, race/ethnicity, BMI), and selected covariates including lifestyle (alcohol consumption, smoking, non-randomized vitamin D supplement use), health history (diabetes, hypertension), and medication use (aspirin, statin and non-statin lipid lowering medications). Using interaction terms, we examined patient characteristics as potential modifiers of the association between randomized vitamin D treatment effect and 25(OH)D change. Regression results are reported as beta coefficients, standard errors (SE), and 95% confidence intervals (CIs). For the interaction models, the beta coefficients represent the estimated differences in 25(OH)D change and the 95% CIs associated with the active vs. placebo vitamin D treatment arm. In a sensitivity analysis, the longitudinal data were restricted to participants who were compliant (taking at least two-thirds of their pills at 1-year, 97%) with similar results. All analyses were performed with SAS software version 9.4 (SAS Institute, Inc; Cary, NC) and all reported p-values were 2-tailed.
Results
Study Population Characteristics
Baseline 25(OH)D was measured for 15,804 participants of the 25,871 VITAL participants. Characteristics of the total randomized cohort, the participants with baseline 25(OH)D, and the cohort with 1-year follow-up measurements are reported in Table 1. The cohort with baseline blood was slightly older, and more likely to use nonrandomized vitamin D supplements. African Americans were less likely to provide a baseline blood sample than other racial/ethnic groups. The 1-year cohort included a higher percentage of African Americans and Hispanics than the overall cohort with baseline blood but was similar to the overall trial population in racial/ethnic distribution. This is also reflected in the geographic distribution with higher numbers of 1-year participants from Southern states. Non-randomized vitamin D supplement use (up to the recommended dietary allowance for older adults of 800 IU/day) was present among approximately 43% of the study population. Pill compliance, defined as taking at least two-thirds of their pills at 1-year, was 91.5% among all participants, 93.2 % among participants with baseline blood, and 97.2% among participants with 1-year blood. Median seasonal difference between baseline and 1-year was +26 days.
Table 1:
Baseline characteristics of the VITAL cohort, the cohort with baseline blood and the 1-year follow-up cohort, by randomized treatmenta,b
| All Participants (n=25871) | Cohort with baseline blood (n=15804) | Cohort with year-1 blood (n=1660) | ||||||
|---|---|---|---|---|---|---|---|---|
| VitaminD (n=12927) | Placebo (n=12944) | VitaminD (n=7905) | Placebo (n=7899) | VitaminD (n=844) | Placebo (n=816) | |||
| Age at randomization in years, [mean (SD)] | 67.1 (7.0) | 67.1 (7.1) | 67.9 (7.0) | 68.0 (7.0) | 65.5 (7.4) | 65.8 (7.2) | ||
| Age categories, [%] | ||||||||
| < 65 years | 38.0 | 38.0 | 32.2 | 32.5 | 48.1 | 47.7 | ||
| 65 - <75 | 49.2 | 49.1 | 53.2 | 52.8 | 41.6 | 41.4 | ||
| 75+ | 12.8 | 12.8 | 14.6 | 14.7 | 10.3 | 10.9 | ||
| Female, [%] | 50.6 | 50.5 | 51.0 | 50.7 | 52.5 | 50.0 | ||
| Race/Ethnicity, [%] | ||||||||
| Non-Hispanic white | 71.3 | 71.4 | 76.5 | 76.1 | 70.0 | 72.4 | ||
| African American | 20.2 | 20.2 | 15.7 | 15.7 | 18.6 | 18.3 | ||
| Hispanic | 4.1 | 3.9 | 3.6 | 3.7 | 6.5 | 5.4 | ||
| Asian/Pacific Islander | 1.5 | 1.6 | 1.4 | 1.5 | 1.6 | 1.8 | ||
| Native American | 0.9 | 0.9 | 0.8 | 0.8 | 1.1 | 0.4 | ||
| Other/unknown | 2.0 | 2.1 | 2.0 | 2.2 | 2.3 | 1.6 | ||
| Body Mass Index in kg/m2, [mean (SD)] | 28.1 (5.7) | 28.1 (5.8) | 27.9 (5.6) | 27.8 (5.6) | 28.6 (6.0) | 28.2 (5.6) | ||
| Body Mass Index in kg/m2, [%] | ||||||||
| <25 | 30.8 | 31.4 | 31.6 | 33.0 | 27.8 | 31.4 | ||
| 25 - <30 | 40.1 | 40.1 | 40.7 | 40.3 | 41.1 | 37.3 | ||
| 30+ | 29.1 | 28.6 | 27.8 | 26.6 | 31.1 | 31.3 | ||
| Alcohol consumption, [%] | ||||||||
| Never | 31.3 | 31.5 | 30.2 | 30.1 | 35.3 | 32.9 | ||
| Rarely | 7.4 | 7.6 | 7.2 | 7.4 | 5.9 | 7.0 | ||
| 1-6 /week | 35.5 | 34.4 | 35.4 | 35.0 | 36.1 | 35.0 | ||
| Daily | 25.8 | 26.4 | 27.3 | 27.5 | 22.7 | 25.1 | ||
| Smoking status, [%] | ||||||||
| never | 51.6 | 51.9 | 52.1 | 51.6 | 55.9 | 51.2 | ||
| past | 41.2 | 40.9 | 42.0 | 42.5 | 38.2 | 42.8 | ||
| Current | 7.2 | 7.2 | 5.9 | 5.9 | 5.9 | 5.9 | ||
| Non-randomized Supplement use, [%] | ||||||||
| Vitamin D (≤800 IU/day) | 42.5 | 42.7 | 46.2 | 46.2 | 43.6 | 46.3 | ||
| Calcium | 19.4 | 18.8 | 21.5 | 21.0 | 23.5 | 19.3 | ||
| Health History / Medications, [%] | ||||||||
| Diabetes | 13.5 | 13.0 | 13.8 | 12.5 | 15.5 | 13.7 | ||
| Hypertension | 53.3 | 54.0 | 53.2 | 53.0 | 54.8 | 52.4 | ||
| Aspirin | 45.2 | 45.6 | 46.4 | 46.9 | 46.5 | 43.2 | ||
| Statins | 35.5 | 34.4 | 37.6 | 36.0 | 39.5 | 36.6 | ||
| Non-statin lipid lowering | 4.8 | 4.5 | 5.2 | 4.8 | 5.3 | 5.6 | ||
| Geographic Region, [%] | ||||||||
| West | 16.9 | 16.9 | 18.7 | 18.2 | 14.2 | 15.0 | ||
| South | 38.9 | 39.2 | 38.7 | 39.5 | 50.6 | 49.6 | ||
| Midwest | 21.5 | 21.4 | 23.1 | 22.8 | 16.8 | 18.0 | ||
| Northwest | 22.8 | 22.6 | 19.5 | 19.5 | 18.4 | 17.4 | ||
| Latitude of participants’ residence, zip code centroid in degrees, [mean (SD), min-max] | 38.0 (5.1) 18.2 – 71.3 |
37.0 (5.1) 18.1 - 61.7 |
38.0 (5.1) 18.3-71.3 |
37.9 (5.1) 18.3-61.7 |
36.9 (4.9) 21.3-48.6 |
37.2 (5.0) 19.9-61.2 |
||
| Pill Compliance at 1-year (≥ 2/3 of study pills), [%] | 91.9 | 91.2 | 93.4 | 93.1 | 96.9 | 97.5 | ||
| Study Compliance at 1-year (≥ 2/3 of study pills & (≤800 IU/day non-randomized Vitamin D)[%] | 88.6 | 86.6 | 90.1 | 88.3 | 93.3 | 92.1 | ||
| Season of blood draw, [%] | BL | BL | BL | 1YR | BL | 1YR | ||
| Winter (Dec-Feb) | 19.3 | 19.5 | 61.0 | 32.0 | 62.5 | 34.4 | ||
| Spring (March-May) | 17.4 | 16.6 | 38.6 | 63.7 | 37.0 | 61.3 | ||
| Summer (June-Aug) | 26.1 | 26.5 | 0 | 3.9 | 0.1 | 3.9 | ||
| Fall (Sept-Nov) | 37.2 | 37.4 | 0.4 | 0.4 | 0.4 | 0.4 | ||
Abbreviations: SD=standard deviation, BL=baseline, 1YR = 1-year
Time-variant characteristics are shown at baseline, unless noted otherwise
Effect of Randomized Vitamin D Supplementation
Mean and median biomarker concentrations at baseline and at 1-year follow-up are shown in Table 2. Baseline mean (SD) total 25(OH)D was 30.8 (10.0) ng/mL, and correlated positively with 25(OH)D3 (r=0.96) and inversely with iPTH (r=−0.28), p<0.001 (Figure 2). The inverse correlation between 25(OH)D and iPTH levels was consistent among whites and African Americans (results not shown). Total 25(OH)D measured by CMIA was highly correlated with the LC-MS measure (r=0.86, Figure 2). Total 25(OH)D, measured by LC-MS/MS, increased by a mean (SD) of 11.9 (9.0) ng/mL (45%) in the vitamin D treatment group and decreased slightly over time in the placebo group (p across treatment groups<0.001). As only 4.6% of the samples had detectable 25(OH)D2 concentrations (lower limit of detection = 4 ng/mL) by LC-MS/MS and 25(OH)D2 contributed 2% to the total 25(OH) concentration, the results are not shown.
Table 2:
Baseline and 1-year follow-up lab measurements, among placebo- and vitamin D-treated participants a
| Cohort with baseline and 1-year measurements | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline in blood cohort (n=15805) | Baseline in 1YR cohort (n=1660) | 1-year (n=1660) | Change from BL to 1-year (n=1660) | Percent Change b (n=1660) | ||||||||
| Treatment | med(IQR) | mean (SD) | med(IQR) | mean (SD) | med(IQR) | mean (SD) | med(IQR)1 | mean (CI) 2 | p-value3 | mean (CI)2 | p-value3 | |
| 25(OH)D total, LC-MS/MS, | Placebo | 31.0 (13.0) | 30.8 (10.0) | 29.2 (12.0) | 29.7 (9.8) | 29.2 (12.0) | 29.0 (9.6) | −0.9 (6.8)*** | −0.7** (−1.2, −0.2) | <.0001 | −2.6** (−4.3, −0.8) | <.0001 |
| [ng/mL] | Vitamin D | 31.0 (12.9) | 30.9 (10.0) | 30.0 (14.5) | 29.8 (10.4) | 40.3 (12.0) | 41.7 (10.0) | 11.1 (11.0)*** | 11.9*** (11.3, 12.5) | 45.2*** (42.1, 48.4) | ||
| 25(OH)D3, LC-MS/MS | Placebo | 30.0 (12.0) | 30.2 (10.2) | 29.2 (12.8) | 29.0 (9.9) | 28.3 (12.0) | 28.2 (9.7) | −0.9 (6.8)*** | −0.9*** (−1.3, −0.4) | <.0001 | −3.6*** (−5.3, −1.9) | <.0001 |
| [ng/mL] | Vitamin D | 30.0 (13.0) | 30.4 (10.2) | 29.2 (13.7) | 29.3 (10.6) | 40.3 (11.9) | 41.4 (10.1) | 11.1 (10.3)*** | 12.1*** (11.5, 12.7) | 47.4*** (44.0, 50.9) | ||
| 25(OH)D total, CMIA | Placebo | 31.1 (10.3) | 31.8 (8.5) | 30.5 (11.4) | 31.4 (8.3) | 30.2 (10.1) | 30.7 (8.0) | −0.8 (5.1)*** | −0.7*** (−1.0, −0.3) | <.0001 | −2.0** (−3.1, −0.8) | <.0001 |
| [ng/mL] | Vitamin D | 31.1 (10.2) | 31.9 (8.7) | 30.8 (10.8) | 31.6 (9.9) | 41.4 (12.3) | 43.1 (10.6) | 10.8 (9.7)*** | 11.6 *** (11.0, 12.1) | 38.5 *** (36.4, 40.7) | ||
| Calcium [mg/dL] | Placebo | 9.4 (0.5) | 9.4 (0.4) | 9.5 (0.7) | 9.6 (0.6) | 9.5 (0.6) | 9.5 (0.6) | 0 (0.6) | 0 (−0.05,0.03) | 0.790 | −0.1 (−0.5, 0.3) | 0.836 |
| Vitamin D | 9.4 (0.5) | 9.4 (0.4) | 9.5 (0.7) | 9.6 (0.5) | 9.5 (0.7) | 9.6 (0.5) | 0 (0.6) | 0 (−0.04,0.04) | 0 (−0.4, 0.4) | |||
| iPTH [pg/ml] | Placebo | 35.0 (21.0) | 39.3 (18.6) | 40.7 (27.8) | 44.7 (23.6) | 40.7 (25.1) | 44.9 (24.5) | 1.0 (21.3) | 0.3 (−1.2, 1.7) | <.0001 | 0.1 (−4.5, 4.9) | 0.004 |
| Vitamin D | 35.0 (20.0) | 39.2 (19.2) | 38.6 (25.7) | 43.8 (25.6) | 34.5 (21.6) | 39.1 (22.6) | −3.1 (17.5)*** | −4.8*** (−6.1, −3.4) | −8.6*** (−12.5, −4.6) | |||
Abbreviations: SD=standard deviation: IQR=interquartile range; CI=95% confidence interval; 25(OH)D=25-hydroxivitamin D; LC-MS/MS= liquid chromatography-tandem mass spectrometry; CMIA=chemiluminescent microparticle immunoassay.
Percent change reported as geometric mean, calculated based on log-transformed data
Nonparametric Wilcoxon signed-rank test for comparison between baseline and 1-year median
Paired t-test for comparison between baseline and 1-year mean
Two sample t-test for comparison of mean change between treatment groups
p<0.05,
p<0.01,
p<0.001
Figure 2:
Seasonal trend of weekly baseline (pre-randomization) total 25(OH)D (LC-MS/MS), Calcium and iPTH averages over the course of the study. Fitted curve was modeled as a linear function of sine and cosine terms with period of one year, adjusted for age, gender, race, BMI, smoking status, and latitude.
Based on a total of 7502 (25(OH)D), 7447 (Ca), and 7474 (iPTH) baseline measurements from subjects without non-randomized vitamin D or calcium supplements; red dots: weekly means based on 3 or less measurements.
Concomitantly, iPTH levels decreased by 4.7 (19.4) pg/mL with vitamin D active treatment and did not change with placebo (Table 2). Calcium levels did not change over time in either treatment group.
Seasonal trends were observed in 25(OH)D as well as in the corresponding iPTH and calcium (Figure 1), with 25(OH)D peaks noted during summer and troughs noted in winter. The season-adjusted mean change in 25(OH)D from baseline to 1-year with randomized vitamin treatment was +11.4 ng/mL or 39.8% (data not shown), slightly lower than the unadjusted treatment effect (+11.9, or 45.2%, Table 2), as part of the increase was related to the seasonally later 1-year collection dates.
The percentage of subjects with vitamin D <20 ng/mL among the cohort with both baseline and 1-year blood samples decreased from 17.8% at baseline to 0.6% in the active treatment arm, and increased from 15.2 to 16.9% in the placebo arm (Table 3). A total of 91.9% of the participants with active treatment achieved blood levels of 25(OH)D at 1-year of at least 30 ng/mL total 25(OH)D.
Table 3:
Categorized baseline and 1-year follow-up serum 25(OH)D total (LC-MS/MS) and iPTH among Placebo- and Vitamin D-treated Participants
| Baseline in blood cohort | Baseline in 1-year cohort | 1-year | ||||
|---|---|---|---|---|---|---|
| VitaminD (n=7905) | Placebo (n=7899) | VitaminD (n=844) | Placebo (n=816) | VitaminD (n=844) | Placebo (n=816) | |
| 25(OH)D total, LC-MS/MS, [%] | ||||||
| <20 ng/mL | 12.3 | 13.0 | 17.8 | 15.2 | 0.6 | 16.0 |
| 20-29.9 ng/mL | 32.1 | 32.3 | 31.5 | 36.4 | 7.5 | 36.9 |
| 30+ ng/mL | 55.6 | 54.6 | 50.7 | 48.4 | 91.9 | 46.2 |
| iPTH, [%] | ||||||
| ≤65 pg/mL | 92.2 | 92.1 | 84.6 | 85.8 | 90.2 | 83.7 |
| > 65 pg/mL | 7.8 | 8.0 | 15.4 | 14.2 | 9.8 | 16.3 |
Assessment of Clinical Characteristics with Baseline and Temporal Changes in 25(OH)D
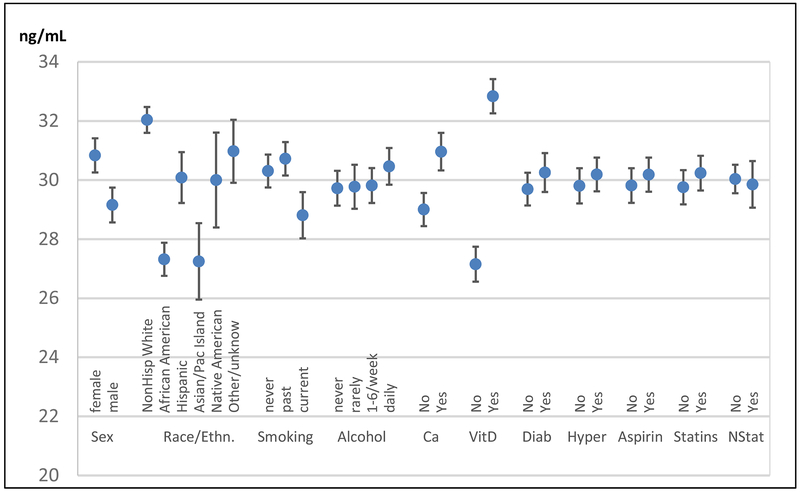
Baseline 25(OH)D levels were significantly associated with sex, race, BMI, smoking, non-randomized vitamin D intake, health history, latitude, and season (Table 4, Figure 3). In multivariable regression models that included all the clinical factors listed in Table 4 and further adjusted for season, women had 1.7 (SE: 0.16) ng/mL higher mean levels than men. Compared to whites, the 25(OH)D levels in African Americans, Hispanics, and Asians were significantly lower by 4.6 (0.23) ng/mL, 1.5 (0.40) ng/mL, and 3.9 (0.63) ng/mL, respectively (all p≤0.0001). Daily alcohol consumption was associated with slightly higher (0.8 (0.21) ng/mL) levels compared with never users, and current smoking with 1.3 (0.32) ng/mL lower 25(OH)D levels. Not surprisingly, vitamin D supplement use at a dose lower than 800 IU/day (otherwise excluded from study) was associated with 5.7 (0.17) ng/mL higher 25(OH)D levels and calcium supplement use (any intake) was associated with a 2.0 (0.21) ng/mL higher 25(OH)D levels. Every degree increase in latitude was associated with a slightly lower 25(OH)D level (−0.15 (0.01)); for example, the latitude difference between Miami, FL, and Boston, MA corresponded to a 2.5 ng/mL lower 25(OH)D.
Table 4:
Association of total serum 25(OH)D (LC-MS/MS), measured at baseline, with clinical characteristics, demographics, and medical historya.
| N | Beta (SE) | 95% CI | p-value | |
|---|---|---|---|---|
| Age in years | −0.006 (0.012) | (−0.03, 0.02) | 0.629 | |
| Female (reference: male) | 6996 | 1.667 (0.164) | (1.35, 1.99) | <.0001 |
| Race/Ethnicity (reference: non-Hispanic white) | 10800 | |||
| African American | 2044 | −4.595 (0.229) | (−5.04, −4.15) | <.0001 |
| Hispanic | 490 | −1.528 (0.404) | (−2.32, −0.74) | 0.0001 |
| Asian/Pacific Islander | 188 | −3.931 (0.634) | (−5.17, −2.69) | <.0001 |
| Native American | 117 | −0.864 (0.798) | (−2.43, 0.70) | 0.279 |
| Other/unknown | 290 | −0.432 (0.510) | (−1.43, 0.57) | 0.398 |
| BMI in kg/m2 [per 1 unit higher] | −0.314 (0.015) | (−0.34, −0.29) | <.0001 | |
| Alcohol consumption [reference=never] | 4128 | |||
| Rarely | 1005 | 0.207 (0.302) | (−0.38, 0.80) | 0.492 |
| 1-6/week | 4927 | 0.094 (0.184) | (−0.27, 0.46) | 0.612 |
| Daily | 3869 | 0.773 (0.205) | (0.37, 1.17) | 0.0002 |
| Smoking status [reference: never] | 7210 | |||
| Past | 5894 | 0.424 (0.153) | (0.12, 0.73) | 0.0057 |
| Current | 825 | −1.273 (0.319) | (−1.90, −0.65 ) | 0.0001 |
| Non-randomized Supplement use of | ||||
| Calcium | 2978 | 2.035 (0.212) | (1.62, 2.45) | <.0001 |
| Vitamin D (<800 IU/day) | 6467 | 5.679 (0.166) | (5.35, 6.00) | <.0001 |
| Health History / Medications | ||||
| Diabetes | 1777 | 0.663 (0.236) | (0.20, 1.12) | 0.0049 |
| Hypertension | 7286 | 0.357 (0.159) | (0.05, 0.67) | 0.0246 |
| Aspirin | 6459 | 0.360 (0.153) | (0.06, 0.66) | 0.0187 |
| Statins | 5103 | 0.485 (0.161) | (0.17, 0.80) | 0.0026 |
| Non-statin lipid lowering | 680 | 0.122 (0.339) | (−0.54, 0.79) | 0.7188 |
| Latitude [increase of 1 degree] b | −0.152 (0.014) | (−0.18, −0.12) | <.0001 |
multivariable linear regression model of total 25(OH)D levels and all covariates listed in this table, additionally adjusted for season (sine and cosine terms), based on 13929 observations complete in all covariates
one degree latitude corresponds to approximately 111 kilometers (69 miles)
Figure 3:
Mean baseline 25(O)D (+95% confidence intervals) associated with subject characteristic.
Multivariable linear regression model of total 25(OH)D levels and all listed covariates, additionally adjusted for age, BMI, latitude, and season.
Ca= non-randomized calcium supplements, VitD=non-randomized VitD supplement, Diab=Diabetes, Hyper=Hypertension, NStat=non-statin lipid-lowering
Adjusted for baseline level and non-randomized supplement intake at 1-year, the change from baseline to 1-year 25(OH)D with randomized vitamin treatment was +12.7 (0.35) ng/mL (Table 5), equivalent to the difference between the mean unadjusted increases (−0.7 for placebo and +11.9 for treatment group, Table 2). There was significant effect modification by race, compliance, non-randomized vitamin D supplementation, and baseline 25(OH)D concentration on the changes in serum levels with treatment (Table 6). The increases in 25(OH)D levels with treatment were greater in African Americans (+15.7 (0.84) ng/mL), Hispanics (+12. (1.47) ng/ml), and other race/ethnic groups (+13.9 (1.72) ng/ml) compared to whites (+12.0 (0.42) ng/mL). Compliant participants (taking at least two-thirds of the study pills) showed a 13.1 (0.37) ng/mL increase compared to 7.7 (1.34) ng/mL in non-compliant participants. No or low use of non-randomized vitamin D supplementation led to greater increases in serum 25(OH)D (+14.6 (0.47) ng/mL). Similarly, lower baseline concentrations of 25(OH)D were associated with greater treatment-related increases in levels, which ranged from a 17.3 (0.89) ng/mL mean increase for participants with baseline levels <20 ng/mL to an 11.4 (0.51) ng/mL mean increase for those with baseline levels ≥30 ng/mL. There was no significant treatment and serum level interaction by BMI categories (p for interaction 0.102). In contrast to the effect modification of race on the 25(OH)D change with vitamin D supplement use (higher increases in African Americans than in whites), we observed greater reductions in iPTH among African Americans (−10.5 (−14.8, −6.2) pg/ml) than among whites −4.6(−6.7, −2.4) (data not shown), consistent with the inverse correlation between the two biomarkers.
Table 5:
Effects of randomized treatment and non-randomized supplement use on season-adjusted change in serum 25(OH)D concentrations from baseline to 1-year a
| Beta (SE) | 95% CI | p-value | |
|---|---|---|---|
| Randomized Vitamin D Treatment | 12.74 (0.35) | (12.04, 13.43) | <.0001 |
| Randomized Omega-3 Treatment | −0.56 (0.35) | (−1.25, 0.13) | 0.115 |
| Non-randomized Supplement use at 1-year | |||
| Calcium | −0.10 (0.51) | (−1.09, 0.90) | 0.850 |
| Vitamin D (reference = none) | |||
| ≤400 IU/day | 0.61 (0.51) | (−0.38, 1.60) | 0.230 |
| 401-800 IU/day | 2.48 (0.51) | (1.48, 3.48) | <.0001 |
| >800 IU/day | 6.18 (0.87) | (4.48, 7.88) | <.0001 |
| Baseline 25(OH)D | −0.37 (0.02) | (−0.41, −0.33) | <.0001 |
multivariable linear regression model of change in total 25(OH)D levels from baseline to 1-year, including Vitamin D treatment, omega-3 treatment and non-randomized Supplement use at 1-year. Based on 1623 observations complete in all covariates.
Table 6:
Effect modification by clinical characteristics, demographics, and medical history on season-adjusted change in serum 25(OH)D concentrations from baseline to 1-year a,b
| Characteristics included as effect modifiers: | N | Beta (SE) | 95% CI | p-value | p-value interaction |
|---|---|---|---|---|---|
| Age categories | 0.1014 | ||||
| < 65 years | 782 | 13.44 (0.51) | (12.4, 14.4) | <.0001 | |
| 65 - <75 | 667 | 11.85 (0.55) | (10.8, 12.9) | <.0001 | |
| 75+ | 174 | 12.92 (1.08) | (10.8, 15.0) | <.0001 | |
| Gender | 0.007 | ||||
| Male | 791 | 11.75 (0.51) | (10.8, 12.7) | <.0001 | |
| Female | 832 | 13.67 (0.49) | (12.7, 14.6) | <.0001 | |
| Race/Ethnicity | 0.001 | ||||
| Non-Hispanic white | 1133 | 11.99 (0.42) | (11.2, 12.8) | <.0001 | |
| African American | 287 | 15.66 (0.84) | (14.0, 17.3) | <.0001 | |
| Hispanic | 94 | 12.12 (1.47) | (9.2, 15.0) | <.0001 | |
| Other | 70 | 13.87 (1.72) | (10.5, 17.2) | <.0001 | |
| BMI categories at 1-year | 0.102 | ||||
| <25 kg/m2 | 460 | 13.98 (0.66) | (12.7, 15.3) | <.0001 | |
| 25 - <30 kg/m2 | 614 | 12.50 (0.57) | (11.4, 13.6) | <.0001 | |
| 30+ kg/m2 | 490 | 12.14 (0.64) | (10.9, 13.4) | <.0001 | |
| Non-randomized vitamin D supplement use at 1-year | <.0001 | ||||
| None | 878 | 14.64 (0.47) | (13.7, 15.6) | <.0001 | |
| ≤400 IU/day | 308 | 10.71 (0.75) | (9.2, 12.2) | <.0001 | |
| 401-800 IU/day | 357 | 11.00 (0.80) | (9.4, 12.6) | <.0001 | |
| >800 IU/day | 80 | 7.55 (1.58) | (4.5, 10.7) | < 0.001 | |
| Compliance at 1-year | 0.0001 | ||||
| <2/3 of study pills or non-rand Vitamin D>800 IU/day | 113 | 7.69 (1.34) | (5.10, 10.3) | <.0001 | |
| ≥2/3 of study pills & non-rand Vitamin D≤800 IU/day | 1510 | 13.11 (0.37) | (12.4, 13.8) | <.0001 | |
| 25(OH)D level at baseline | <.0001 | ||||
| <20 ng/mL | 268 | 17.31 (0.89) | (15.6, 19.0) | <.0001 | |
| 20 – 29.9 ng/mL | 549 | 12.19 (0.62) | (11.0 . 13.4 ) | <.0001 | |
| ≥30 ng/mL | 806 | 11.38 (0.51) | (10.4, 12.4) | <.0001 | |
Main multivariable linear regression model contains all covariates listed in table 4. One variable of interest (e.g. gender, race/ethnicity, etc.) and an interaction term with vitamin D randomized treatment were (one at a time) additionally included in the model.
No significant interaction for alcohol consumption, smoking, diabetes, hypertension, or medication use.
Discussion
In the VITAL randomized trial conducted in a contemporary US population, baseline 25(OH)D levels varied by clinical subgroups, were lower in men and racial/ethnic minorities, and also varied by BMI, latitude, and season. After one year of randomized supplementation with vitamin D 2000 IU/day versus placebo, 25(OH)D levels (total and D3) increased appropriately, with >90% of participants achieving 25(OH)D levels of 30ng/mL or higher and 53% achieving levels that were 40 ng/mL or higher (results not shown). The greatest increases with treatment were noted among individuals with baseline levels <20 ng/mL and in African Americans. Results for 25(OH)D measured using a chemiluminescent immunoassay (CMIA) were similar to the LC-MS/MS assay after both assays were calibrated with the CDC vitamin D standardization program. We also noted seasonal trends for iPTH and calcium in addition to 25(OH)D, which may be relevant for clinicians when interpreting 25(OH)D levels for clinical treatment decisions.
VITAL is the largest randomized trial of moderate- or high-dose vitamin D in U.S. adults (2,16), and is the only large trial worldwide (one of two trials with 10,000 or more participants) with an appreciable number of African American participants, who are at increased risk for vitamin D insufficiency. Levels of total 25(OH)D are the most commonly measured clinical tests for assessment of adequacy of vitamin D levels and the basis for clinical recommendations, although controversy exists regarding optimal levels for both skeletal and extra-skeletal clinical outcomes.(1) In the VITAL study population, baseline 25(OH)D <20 and 20 to <30 ng/mL were present in 12.7% and 32.2%, respectively, while <1% of the 1-year 25(OH)D concentrations were <20 ng/mL after randomized allocation to 2000 IU of vitamin D3 (cholecalciferol). The levels of 25(OH)D (both total and D3) increased appropriately and consistently with the dose of vitamin D administered, and the participant compliance with randomized treatment was high (>97% at 1-year in the blood follow-up cohort, compared to 91% in the overall trial). Prior studies have found a curvilinear dose response increase in 25(OH)D with vitamin D supplementation, with an average increase in levels by 0.7 to 1.0 ng/mL per 100 unit higher vitamin D3 administered dose. (17) Furthermore, it is controversial whether there is a safe upper bound level of 25(OH)D. In VITAL, while the vast majority of participants had levels of 30 ng/mL or greater at 1-year, there was no evidence of toxicity or adverse effects of vitamin D supplementation. (10).
Important considerations when measuring 25(OH)D levels are the season and latitude, as these have been noted to affect 25(OH)D levels, with a peak in 25(OH)D level noted in the summer and a trough in the winter based on variations in ultraviolet light exposure, and lower levels in the north vs south. (18,19) We adjusted our results (particularly relevant for interpreting the temporal changes in levels) for season (using a cosine/sine approach) as well as latitude, among other factors, which increased the rigor of the current results. We also noted seasonal trends for the corresponding iPTH and calcium levels in addition to 25(OH)D, which may be relevant for clinicians when interpreting 25(OH)D levels for clinical treatment decisions. Furthermore, the relative secondary hyperparathyroidemia was decreased after 1-year of randomized vitamin D supplementation versus placebo, consistent with prior studies. (17)
We also noted ethnic and racial differences, with minorities – in particular African Americans - having lower levels compared with whites, although fewer African Americans provided blood samples compared with whites. Prior studies that have also noted African Americans have lower 25(OH)D than whites, but paradoxically they have also been noted to have higher bone mineral density.(20) However, a recent small study found that free 25(OH)D levels were not influenced by race/ethnicity (21) and another recent study found that vitamin D binding protein was comparable between African-ancestry subjects and whites when measured by assays using polyclonal antibodies. (22) We encourage future studies to clarify the effects of vitamin D supplementation, not only on 25(OH)D, but also on emerging biomarkers of vitamin D activity, and to assess their association with cancer and CVD outcomes and their potential modifying effect on the vitamin D intervention. We plan to perform such analyses in VITAL.
Strengths of the current study include the large sample size of 15,804 participants, with a large proportion of African Americans, the availability of both baseline and follow-up bloods on a subset of participants (n=1,660), and the availability of two different laboratory assays for 25(OH)D and related biomarkers available at the same time points in the trial. We also participated in the CDC vitamin D standardization program that improved laboratory performance parameters such as precision and reduced bias of measurements and gives us the flexibility in future studies to calibrate results with respect to the CDC reference method.(23) Other strengths include that blood samples could be used to confirm intervention compliance, monitor changes in background intake in the study population over time (placebo group), and determine whether treatment effects are modified by baseline and post-randomization blood levels (10). Potential limitations should also be acknowledged. While we measured total 25(OH)D (with both 25(OH)D2 and D3 quantitated), as well as iPTH and calcium, we did not measure other emerging biomarkers of vitamin D activity such as free or bioavailable vitamin D at this time. Not all participants provided blood samples and African Americans had lower rates of blood collection than non-Hispanic whites. Finally, VITAL study participants, as study volunteers, were healthier on average, than the general population, (12) which may result in higher 25(OH)D levels and a potentially smaller increase in 25(OH)D with 2000 IU of vitamin D3 supplementation than in clinical practice general populations.
In conclusion, in the VITAL randomized trial, serum 25(OH)D concentrations varied by clinical subgroups, were lower in men and racial/ethnic minorities, and varied by BMI, season, and latitude. Levels increased appropriately with vitamin D supplementation, with the greatest increases in those with lower baseline levels, including African Americans. Future studies should evaluate the role of other emerging biomarkers of vitamin D activity and their relevance for modifying the effects of vitamin D supplementation and predicting both skeletal and non-skeletal clinical outcomes.
Acknowledgements
We thank the VITAL participants and staff for their dedicated and conscientious collaboration. Pharmavite LLC of Northridge, California (vitamin D) and Pronova BioPharma of Norway and BASF (Omacor fish oil) donated the study agents, matching placebos, and packaging in the form of calendar packs. The collaborating laboratories (Quest Diagnostics and Atherotech Diagnostics) performed the laboratory measurements at no additional costs to the VITAL study. We thank Dr. Michael Caulfield and Jennifer Boring from Quest Diagnostics for their contributions. We also thank the Center for Disease Control and Prevention (Dr. Hubert Vesper and Dr. Julianne Cook Botelho) for their collaboration on the standardization and calibration of the 25(OH)D measurements throughout the study.
Funding Sources
VITAL was supported by grants U01 CA138962 and R01 CA138962, which included support from the National Cancer Institute, National Heart, Lung and Blood Institute, Office of Dietary Supplements, National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. The ancillary studies are supported by grants from multiple Institutes, including the National Heart, Lung and Blood Institute; the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institute on Aging; the National Institute of Arthritis and Musculoskeletal and Skin Diseases; the National Institute of Mental Health; and others. Dr. Mora was supported by the research grants from the National Institute of Diabetes and Digestive and Kidney Diseases (DK112940), National Heart, Lung, and Blood Institute (R01HL134811 and K24 HL136852), and the Molino Family Trust. Dr. Demler was supported by a research grant from the National Heart, Lung, and Blood Institute (5K01HL135342). The funding sources had no role in the design and conduct of this study or the interpretation of the data. The opinions expressed in the manuscript are those of the study authors.
Disclosures: HG, CC, NC, OD, VB, CL, GF, IL, and JB have nothing to declare. SM received research grant support from Atherotech Diagnostics for the measurement of vitamin D in the VITAL trial, with additional support from the Molino Family Trust, and the NIH, and has served as consultant (modest) to Quest Diagnostics. JL and JP are employed by Quest Diagnostics. AG and JW are employees and stockholders of Quest. KK and MC were previously employed by Atherotech Diagnostics. JM received research grant support from the National Institutes of Health. Pharmavite LLC of Northridge, California donated the (vitamin D) study pills for VITAL and Quest Diagnostics and Atherotech Diagnostics conducted relevant assays free of charge.
Footnotes
Clinical Trial Registration: VITAL ClinicalTrials.gov number: .
References
- 1.Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK, Durazo-Arvizu RA, Gallagher JC, Gallo RL, Jones G, Kovacs CS, Mayne ST, Rosen CJ, Shapses SA. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96(1):53–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Manson JE, Bassuk SS, Lee IM, Cook NR, Albert MA, Gordon D, Zaharris E, Macfadyen JG, Danielson E, Lin J, Zhang SM, Buring JE. The VITamin D and OmegA-3 TriaL (VITAL): rationale and design of a large randomized controlled trial of vitamin D and marine omega-3 fatty acid supplements for the primary prevention of cancer and cardiovascular disease. Contemp Clin Trials. 2012;33(1):159–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yin L, Ordonez-Mena JM, Chen T, Schottker B, Arndt V, Brenner H. Circulating 25-hydroxyvitamin D serum concentration and total cancer incidence and mortality: a systematic review and meta-analysis. Prev Med. 2013;57(6):753–764. [DOI] [PubMed] [Google Scholar]
- 4.Zhang R, Li B, Gao X, Tian R, Pan Y, Jiang Y, Gu H, Wang Y, Wang Y, Liu G. Serum 25-hydroxyvitamin D and the risk of cardiovascular disease: dose-response meta-analysis of prospective studies. Am J Clin Nutr. 2017;105(4):810–819. [DOI] [PubMed] [Google Scholar]
- 5.Giustina A, Adler RA, Binkley N, Bouillon R, Ebeling PR, Lazaretti-Castro M, Marcocci C, Rizzoli R, Sempos CT, Bilezikian JP. Controversies in Vitamin D: Summary Statement From an International Conference. J Clin Endocrinol Metab. 2019;104(2):234–240. [DOI] [PubMed] [Google Scholar]
- 6.LeBoff MS, Yue AY, Copeland T, Cook NR, Buring JE, Manson JE. VITAL-Bone Health: rationale and design of two ancillary studies evaluating the effects of vitamin D and/or omega-3 fatty acid supplements on incident fractures and bone health outcomes in the VITamin D and OmegA-3 TriaL (VITAL). Contemp Clin Trials. 2015;41:259–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wacker M, Holick MF. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol. 2013;5(1):51–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Atef SH. Vitamin D assays in clinical laboratory: Past, present and future challenges. J Steroid Biochem Mol Biol. 2018;175:136–137. [DOI] [PubMed] [Google Scholar]
- 9.Farrell CJ, Martin S, McWhinney B, Straub I, Williams P, Herrmann M. State-of-the-art vitamin D assays: a comparison of automated immunoassays with liquid chromatography-tandem mass spectrometry methods. Clin Chem. 2012;58(3):531–542. [DOI] [PubMed] [Google Scholar]
- 10.Manson JE, Cook NR, Lee IM, Christen W, Bassuk SS, Mora S, Gibson H, Gordon D, Copeland T, D’Agostino D, Friedenberg G, Ridge C, Bubes V, Giovannucci EL, Willett WC, Buring JE, Group VR. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N Engl J Med. 2018. [DOI] [PubMed] [Google Scholar]
- 11.Manson JE, Bassuk SS. Determining the Value of Vitamin D Supplementation--Reply. JAMA. 2015;314(6):627–628. [DOI] [PubMed] [Google Scholar]
- 12.Bassuk SS, Manson JE, Lee IM, Cook NR, Christen WG, Bubes VY, Gordon DS, Copeland T, Friedenberg G, D’Agostino DM, Ridge CY, MacFadyen JG, Kalan K, Buring JE. Baseline characteristics of participants in the VITamin D and OmegA-3 TriaL (VITAL). Contemp Clin Trials. 2016;47:235–243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stockl D, Sluss PM, Thienpont LM. Specifications for trueness and precision of a reference measurement system for serum/plasma 25-hydroxyvitamin D analysis. Clin Chim Acta. 2009;408(1–2):8–13. [DOI] [PubMed] [Google Scholar]
- 14.Binkley N, Dawson-Hughes B, Durazo-Arvizu R, Thamm M, Tian L, Merkel JM, Jones JC, Carter GD, Sempos CT. Vitamin D measurement standardization: The way out of the chaos. J Steroid Biochem Mol Biol. 2017;173:117–121. [DOI] [PubMed] [Google Scholar]
- 15.Sachs MC, Shoben A, Levin GP, Robinson-Cohen C, Hoofnagle AN, Swords-Jenny N, Ix JH, Budoff M, Lutsey PL, Siscovick DS, Kestenbaum B, de Boer IH. Estimating mean annual 25-hydroxyvitamin D concentrations from single measurements: the Multi-Ethnic Study of Atherosclerosis. Am J Clin Nutr. 2013;97(6):1243–1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pradhan AD, Manson JE. Update on the Vitamin D and OmegA-3 trial (VITAL). J Steroid Biochem Mol Biol. 2016;155(Pt B):252–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gallagher JC, Sai A, Templin T 2nd, Smith L. Dose response to vitamin D supplementation in postmenopausal women: a randomized trial. Ann Intern Med. 2012;156(6):425–437. [DOI] [PubMed] [Google Scholar]
- 18.Jukic AMZ, Hoofnagle AN, Lutsey PL. Measurement of Vitamin D for Epidemiologic and Clinical Research: Shining Light on a Complex Decision. Am J Epidemiol. 2018;187(4):879–890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kroll MH, Bi C, Garber CC, Kaufman HW, Liu D, Caston-Balderrama A, Zhang K, Clarke N, Xie M, Reitz RE, Suffin SC, Holick MF. Temporal relationship between vitamin D status and parathyroid hormone in the United States. PLoS One. 2015;10(3):e0118108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Powe CE, Karumanchi SA, Thadhani R. Vitamin D-binding protein and vitamin D in blacks and whites. The New England journal of medicine. 2014;370(9):880–881. [DOI] [PubMed] [Google Scholar]
- 21.Schwartz JB, Lai J, Lizaola B, Kane L, Markova S, Weyland P, Terrault NA, Stotland N, Bikle D. A comparison of measured and calculated free 25(OH) vitamin D levels in clinical populations. The Journal of clinical endocrinology and metabolism. 2014;99(5):1631–1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nielson CM, Jones KS, Chun RF, Jacobs JM, Wang Y, Hewison M, Adams JS, Swanson CM, Lee CG, Vanderschueren D, Pauwels S, Prentice A, Smith RD, Shi T, Gao Y, Schepmoes AA, Zmuda JM, Lapidus J, Cauley JA, Bouillon R, Schoenmakers I, Orwoll ES, Osteoporotic Fractures in Men Research G. Free 25-Hydroxyvitamin D: Impact of Vitamin D Binding Protein Assays on Racial-Genotypic Associations. J Clin Endocrinol Metab. 2016;101(5):2226–2234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Binkley N, Carter GD. Toward Clarity in Clinical Vitamin D Status Assessment: 25(OH)D Assay Standardization. Endocrinol Metab Clin North Am. 2017;46(4):885–899. [DOI] [PubMed] [Google Scholar]