Abstract
In a retrospective analysis of 38,504 lymphoma diagnoses in Georgia between 2001 and 2015, we found that, compared with whites, blacks with diffuse large B-cell lymphoma and follicular lymphoma were more likely to present at a younger age and experience B symptoms. Future research utilizing population-based data to address disparities in presentation and outcomes within a catchment area is needed.
Background:
Racial disparities in non-Hodgkin lymphoma (NHL) are not well-elucidated for specific catchment areas, which can influence outcomes. Leveraging regional data from a population-based cancer registry may provide unique opportunities to quantify NHL disparities.
Materials and Methods:
Using Surveillance, Epidemiology, and End Results (SEER) data for NHL cases diagnosed in Georgia from 2001 to 2015, we examined NHL incidence rates by lymphoma subtype and racial differences in baseline characteristics and outcomes for diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL). Cox regression models identified predictors of overall survival (OS).
Results:
SEER documented 38,504 NHL cases in Georgia from 2001 to 2015. The age-adjusted incidence rate for NHL in Georgia increased 1.03% per year, and the annual percentage change was 1.72 in blacks compared with 0.84 in whites. Compared with whites, blacks with DLBCL and FL were more likely to be diagnosed at a younger age (DLBCL, 54.1 vs. 65.5 years; P < .0001; FL, 58.4 vs. 64.0 years; P < .0001) and with B symptoms (DLBCL, 44.4% vs. 33.4%; P < .0001; FL, 28.5% vs. 21.4%; P = .004). Across racial categories, age at diagnosis > 60 years, advanced stage, and B symptoms predicted worse OS in DLBCL and FL. Blacks with DLBCL more commonly were diagnosed with stage III/IV disease (55.5% vs. 48.1%; P < .0001) and had worse 5-year relative survival (58.8% vs. 62.3%; P = .01).
Conclusions:
Regional cancer registry data can be used to define incidence patterns and disparities in outcomes across NHL subtypes to help define key targets for interventions in a catchment area.
Keywords: Diffuse large B-cell lymphoma, Follicular lymphoma, Non-Hodgkin lymphoma, Racial differences, SEER
Introduction
Non-Hodgkin lymphoma (NHL) is the seventh most common cancer, and the ninth leading cause of cancer death for both men and women in the United States (US).1 In 2019, 74,200 new cases of NHL are expected, representing 4.2% of all new cancer cases in the US.1 Although the incidence of NHL in the United States has decreased and survival has improved in recent years,2 disparities on the basis of race, gender, age, insurance status, demographic location, socioeconomic status (SES), and other sociodemographic characteristics remain.2–12 Racial disparities have been shown to be a critical issue in cancer care, and arise across the cancer control continuum for many malignancies.2,3,6,9,13 Thus, such disparities have become increasingly recognized as critically important targets for improvement in cancer care across populations. However, few analyses have examined cancer subtype incidence and population-level outcomes for catchment areas where interventions can be specifically designed to address disparities.
There is emerging evidence that blacks, particularly black males, are more likely to have poorer survival than whites for certain NHL subtypes.7,9,10,12,14 Shenoy and colleagues examined the differences in incidence, patterns of presentation, and survival for diffuse large B-cell lymphoma (DLBCL) using Surveillance, Epidemiology, and End Results (SEER) data from 1981 to 2010.9 Their study suggested that, although the incidence of DLBCL was lower in blacks, black patients with DLBCL were more likely to be diagnosed at a younger age, at a more advanced stage, and have inferior survival. Notable racial differences in survival have also been described in other subtypes, including mycosis fungoides, peripheral T-cell lymphoma, Burkitt lymphoma, and chronic lymphocytic leukemia/ small lymphocytic lymphoma.2 Various factors may account for racial disparities in NHL. However, studies that identify population-based racial disparities in NHL and elucidate the mechanisms underlying them are needed to develop strategies aimed at closing these gaps.
The state of Georgia provides an ideal opportunity to study racial disparities in NHL owing to its unique demographic makeup, the presence of a population-based statewide cancer registry, and a National Cancer Institute (NCI)-designated Comprehensive Cancer Center with a catchment area that matches the registry. According to the 2018 US Census Bureau population estimates, the US has a demographic composition that is 76.6% white alone, 13.4% black alone, and 10.0% other.15 In comparison, Georgia has a demographic composition that is 60.8% white alone, 32.2% black alone, and 7.0% individuals who identified as other racial group, thus providing a population-based setting enriched for examining racial disparities in cancer incidence and outcomes. In addition, the Georgia registry comprise the NCI-SEER program. SEER registries are designed to collect high-quality cancer surveillance data with longitudinal survival outcomes and are specifically equipped to support research activities involving the populations covered by each registry.16 These characteristics make Georgia’s population of unique importance in broadening our understanding of racial disparities in NHL. To systematically describe racial disparities for NHL in Georgia, we performed a retrospective analysis of Georgia NHL cases registered from 2001 to 2015.
Materials and Methods
Study Population
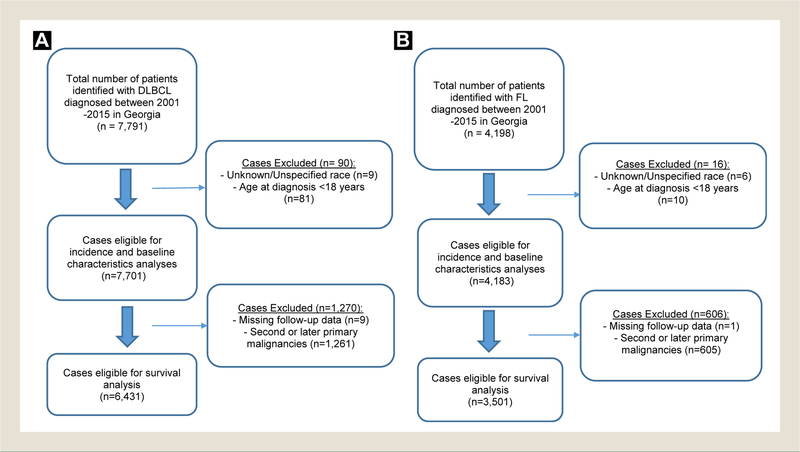
NHL cases diagnosed between 2001 and 2015 in Georgia were identified and categorized using International Classification of Diseases for Oncology (ICD-O-3) codes updated based on the World Health Organization (WHO) Classification of Tumours of Haematopoietic and Lymphoid Tissues.17 These data were accessed using SEER*Stat18 from the November 2017 submission.17 Cases with unknown or unspecified race and cases with unknown age or younger than the age of 18 were excluded. Cases were classified into 3 groups: white, black, and other (eg,19 American Indian/Alaska Native, Asian, or Pacific Islander). DLBCL diagnoses included the following groups according to ICD-O-3: DLBCL, not otherwise specified (NOS) (9680 except site C49.9); intravascular large B-cell lymphoma (9680 of site C49.9 and 9712); primary effusion lymphoma (PEL) (9678); mediastinal large B-cell lymphoma (9679); immunoblastic B-cell lymphoma (9684 B); T-cell/histiocyte rich large B-cell lymphoma (9688); ALK-positive large B-cell lymphoma (9737); large B-cell (plasmablastic) lymphoma arising from HHV-8 associated multicentric Castleman disease (9738). Follicular lymphoma (FL) diagnoses included the following groups according to ICD-O-3: FL, NOS (9690 except site C44.0–44.9); FL, grade I to II (9691, 9695); FL, grade III (9698). For the survival analysis of DLBCL and FL, we excluded patients with second primaries and patients with patients diagnosed based on death certificate or autopsy alone and patients who were alive with missing survival time information. Figure 1 shows the selection of patients with DLBCL and FL for inclusion in this study. Inclusion criteria were consistent among all subtypes.
Figure 1. Selection of DLBCL and FL Study Cohorts. A, Selection of DLBCL Cohort. B, Selection of FL Cohort.
Abbreviations: DLBCL = diffuse large B-cell lymphoma; FL = follicular lymphoma.
Statistical Analysis
Age-adjusted incidence rates were expressed as new cases per 100,000 person-years. Comparison of age-adjusted incidence rates and age-specific incidence rates by race and gender was expressed using incidence rate ratios (IRRs) with 95% confidence intervals (CIs), with whites as the reference group. Temporal trends in the age-adjusted incidence rate by race, gender, and subtype were described as the annual percent change (APC) in the rate, calculated using weighted least squares log-linear regression. Differences in baseline characteristics at diagnosis across racial groups were analyzed (ie, age, gender, stage, site of disease, and presence of B symptoms). χ2 tests and the 2-sided t test were used for categorical and continuous variables, respectively. Two-year and 5-year relative survival (RS) were calculated by actuarial methods. Differences in 2-year and 5-year RS by race, gender, and subtype were analyzed using a z-test to evaluate the equivalence of RS. Multivariable Cox proportional hazards models were fitted to evaluate the covariates as predictors of overall survival (OS). Analyses of incidence, temporal trends, and RS were performed using the NCI’s SEER*Stat software. Analyses for differences in baseline characteristics at diagnosis and the multivariable Cox proportional hazards models were performed using SAS (version 9.4).
Results
Age-adjusted Incidence Rate and Temporal Trends for NHL
After excluding cases, as specified above, there were 38,504 cases of lymphoid cancers reported in Georgia between 2001 and 2015. The most common subtype of lymphoid cancer was plasma cell neoplasm, followed by DLBCL, chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), and FL (see Supplemental Table 1A [in the online version] and Table 1). Although blacks comprise 32.2% of the population in Georgia in 2018, black patients represented 25.6% of all lymphoid cancer diagnoses in Georgia between 2001 and 2015 (see Supplemental Tables 1B and 1C in the online version). During the period from 2001 to 2015, the age-adjusted incidence rate of lymphoid malignancies in Georgia increased 1.03% per year (95% CI, 0.50%−1.56%). The APC in incidence rate was more pronounced in blacks (1.72; 95% CI, 0.95–2.50) compared with whites (0.84; 95% CI, 0.24–1.45). Among all racial groups, the largest increase in B-cell NHL was seen in the B-cell NHL, NOS group (APC, 4.75; 95% CI, 2.93–6.60), whereas the largest increase in T-cell NHL was seen in adult T-cell lymphoma/leukemia (APC, 8.24; 95% CI, 3.63–13.05). Rates also increased for plasma cell neoplasms, mantle cell lymphoma, marginal zone lymphoma, mycosis fungoides/Sezary syndrome, and NK/T-cell lymphoma (see Supplemental Table 2 in the online version).
Table 1.
Distribution of and Racial Difference in Age at Diagnosis in Lymphoid Neoplasms by Subtype in Georgia, 2001 to 2015
| Lymphoid Neoplasm Subtypes | N | % | Mean Age at Diagnosis (95% CI) |
P Value |
|||
|---|---|---|---|---|---|---|---|
| White | Black | Other | W vs. B | W vs. O | |||
| Mature B-cell and T-/NK-cell neoplasms | 38,504 | 100.0 | |||||
| Mature B-cell lymphoid neoplasms | |||||||
| Chronic lymphocytic leukemia/small lymphocytic lymphoma | 6351 | 16.5 | 69.6 (69.2–69.9) | 66.9 (66.2–67.6) | 64.9 (60.7–69.1) | <.0001 | .0171 |
| Mantle cell lymphoma | 860 | 2.2 | 67.0 (66.2–67.8) | 61.0 (57.9–64.1) | 63.1 (55.8–70.4) | <.0001 | .2851 |
| Lymphoplasmacytic lymphoma/Waldenstrom | 501 | 1.3 | 69.6 (68.4–70.8) | 62.5 (59.8–65.2) | 64.5 (52.4–76.6) | <.0001 | .0002 |
| Diffuse large B-cell lymphoma | 7701 | 20.0 | 65.5 (65.1–65.9) | 54.1 (53.4–54.9) | 60.5 (58.1–62.9) | <.0001 | .8343 |
| Burkitt lymphoma/leukemia | 425 | 1.1 | 51.5 (49.6–53.5) | 43.5 (40.3–46.7) | 56.0 (40.8–71.2) | <.0001 | .4550 |
| Marginal zone lymphoma | 1734 | 4.5 | 66.5 (65.8–67.2) | 59.1 (57.7–64.5) | 62.0 (54.7–69.3) | <.0001 | .0027 |
| Follicular lymphoma | 4183 | 10.9 | 64.0 (63.5–64.4) | 58.4 (57.2–59.6) | 57.6 (53.8–61.4) | <.0001 | .9847 |
| Hairy-cell leukemia | 321 | 0.8 | 61.8 (60.0–63.5) | 60.9 (56.3–65.8) | 60.0 (39.1–80.9) | .9386 | .0007 |
| Mature T-/NK-cell lymphoid neoplasms | |||||||
| Mycosis fungoides/Sezary syndrome | 522 | 1.4 | 60.6 (58.8–62.4) | 51.9 (49.8–53.9) | 54.1 (42.4–65.9) | <.0001 | .5411 |
| Peripheral T-cell lymphoma | 1550 | 4.0 | 60.4 (59.4–61.4) | 54.5 (53.1–55.9) | 57.2 (50.8–63.6) | <.0001 | .6020 |
| Other mature B-cell and T-/NK-cell neoplasms | 3922 | 10.2 | |||||
| Adult T-cell lymphoma/leukemia | 46.9 (42.8–51.0) | 47.5 (43.6–51.4) | 44.8 (27.0–62.6) | .9787 | .9723 | ||
| NK/T-cell lymphoma, nasal-type/aggressive NK leukemia | 53.8 (48.9–58.8) | 45.3 (36.3–54.2) | 64.0 (44.6–83.4) | .2235 | .5693 | ||
| T-cell large granular lymphocytic leukemia | 70.1 (60.9–79.3) | N/A | N/A | N/A | N/A | ||
| Prolymphocytic T-cell leukemia | 70.7 (63.9–77.4) | 66.5 (57.9–75.1) | N/A | .4474 | N/A | ||
Abbreviations: B = Black; CI = confidence interval; O = other; W = white.
Baseline Characteristics at Diagnosis for DLBCL and FL
Patients classified in the race groups black and other were diagnosed at a younger age compared with whites for both DLBCL and FL. The mean age at diagnosis for DLBCL and FL in blacks was significantly younger than for whites (DLBCL, 54.1 vs. 65.5 years, respectively; FL, 58.4 vs. 64.0, respectively; all P < .001). The mean age at diagnosis for DLBCL and FL in the other race category was significantly younger than whites (DLBCL, 60.5 vs. 65.5 years, respectively and FL, 57.6 vs. 64.0, respectively; all P < .001). Differences between whites and blacks in age at diagnosis were observed for most B-cell NHLs, mycosis fungoides/Sezary syndrome, and peripheral T-cell lymphoma (Table 1). The mean age at diagnosis for DLBCL and FL in blacks was significantly younger than for whites (DLBCL, 54.1 vs. 65.5 years, respectively; FL, 58.4 vs. 64.0, respectively; all P < .001). Patients in the other racial category were also significantly younger than whites at diagnosis (DLBCL, 60.5 years; FL, 57.6 years; all P < .001 when compared with whites).
Black patients were more likely to be diagnosed before the age of 60 years for both DLBCL and FL (all P < .05) (Table 2). Black patients with DLBCL were more likely than whites to present with stage III/IV disease (55.5% vs. 48.1%; P < .001) and B symptoms (44.4% vs. 33.4%; P < .001), and less likely to present with an extranodal primary site (35.6% vs. 39.9%; P = .0020). Blacks with FL were more likely to present with B symptoms at diagnosis when compared with whites (28.5% vs. 21.4%; P = .0035). There was no significant difference in stage at diagnosis and extranodal primary site of FL across all racial groups.
Table 2.
Racial Difference in Baseline Characteristics at Diagnosis of DLBCL and FL in Georgia, 2001 to 2015
| DLBCL (n =7701) |
FL (n = 4183) |
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Whitea (n = 5951) |
Black (n = 1592) |
Otherb (n = 158) |
P Value |
Whitea (n = 3656) |
Black (n = 479) |
Otherb (n = 48) |
P Value |
|||||||||
| N | % | N | % | N | % | N | % | N | % | N | % | N | % | B vs. W | O vs. W | |
| Age, y | ||||||||||||||||
| 18–59 | 1867 | 24.24 | 1004 | 63.07 | 66 | 41.77 | <.0001 | .0055 | 1323 | 36.19 | 263 | 54.91 | 26 | 54.17 | <.0001 | .0101 |
| ≤60 | 4084 | 68.63 | 588 | 36.93 | 92 | 58.23 | 2333 | 63.81 | 216 | 45.09 | 22 | 45.83 | ||||
| Gender | ||||||||||||||||
| Female | 2697 | 45.32 | 699 | 43.91 | 77 | 48.73 | .3141 | .3949 | 1877 | 51.34 | 227 | 47.39 | 17 | 35.42 | .1040 | .0283 |
| Male | 3254 | 54.68 | 893 | 56.09 | 81 | 51.27 | 1779 | 48.66 | 252 | 52.61 | 31 | 64.58 | ||||
| Stage | ||||||||||||||||
| I/II | 2945 | 51.86 | 685 | 44.48 | 86 | 54.78 | <.0001 | .4702 | 1692 | 49.02 | 214 | 46.93 | 21 | 44.65 | .4024 | .6504 |
| III/IV | 2734 | 48.14 | 855 | 55.52 | 71 | 45.22 | 1760 | 50.98 | 242 | 53.07 | 25 | 54.35 | ||||
| Missing | 272 | 52 | 1 | 204 | 23 | 2 | ||||||||||
| Lymph site | ||||||||||||||||
| Extranodal | 2365 | 39.87 | 566 | 35.62 | 72 | 45.86 | .0020 | .1304 | 650 | 17.82 | 93 | 19.46 | 4 | 8.33 | .3808 | .0871 |
| Nodal | 3567 | 60.13 | 1023 | 64.38 | 85 | 54.14 | 2998 | 82.18 | 385 | 80.54 | 44 | 91.67 | ||||
| Missing | 19 | 3 | 1 | 8 | 1 | 0 | ||||||||||
| B symptoms | ||||||||||||||||
| Yes | 1350 | 33.38 | 531 | 44.40 | 28 | 25.69 | <.0001 | .0923 | 516 | 21.43 | 95 | 28.53 | 5 | 14.71 | .0035 | .3420 |
| No | 2694 | 66.62 | 665 | 55.60 | 81 | 74.31 | 1892 | 78.57 | 238 | 71.47 | 29 | 85.29 | ||||
| Missing | 1907 | 396 | 49 | 1248 | 146 | 14 | ||||||||||
Information on stage, extranodal disease, and B symptoms was missing in 4.2%, 0.3%, and 30.5% of cases with DLBCL, and was missing in 5.5%, 0.2%, and 33.7% of cases with FL. Abbreviations: B = black; DLBCL = diffuse large B-cell lymphoma; FL = follicular lymphoma; O = other; W = white.
Reference group.
Other includes American Indian/Alaska Native and Asian or Pacific Islander.
Age-specific Incidence Rate for DLBCL and FL
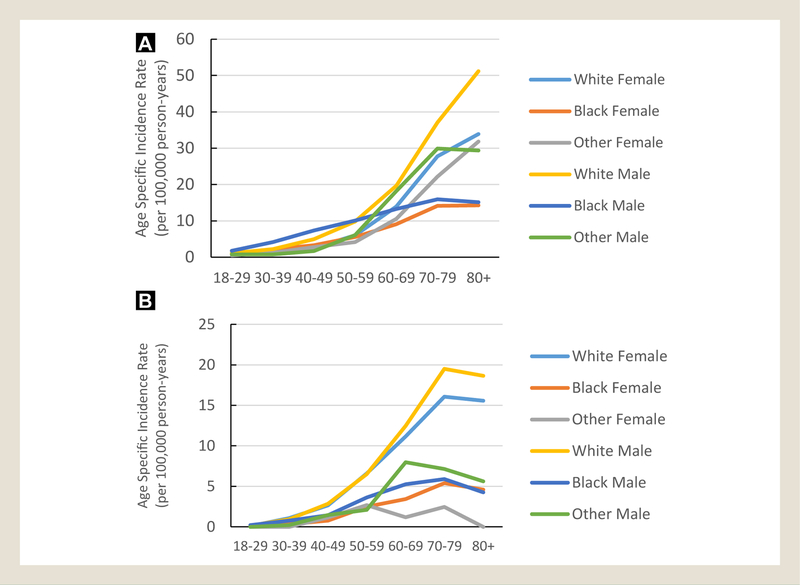
The incidence rate of DLBCL increased with age for all race and gender subgroups, except for the slight drop in the incidence among black males after the age of 80 years (Figure 2A). The incidence rate of FL increased with age for whites and blacks, except for the slight drop in the incidence rate after the age of 80 years (Figure 2B).
Figure 2.
Age-specific Incidence Rate of Diffuse Large B-cell Lymphoma (A) and Follicular Lymphoma (B) by Gender and Race in Georgia (2001–2015)
Survival Analysis
The 2- and 5-year RS for all patients with DLBCL were 67.4% and 61.6%, respectively. Black patients with DLBCL had a significantly worse 2- and 5-year RS compared with white patients (2-year RS, 64.1% vs. 68.2%; P = .0083; 5-year RS, 58.8% vs. 62.3%; P = .012) (see Supplemental Table 3 in the online version). There was no significant difference in RS for DLBCL between whites and individuals in the other race group. Patients with mediastinal large B-cell lymphoma had higher 2- and 5-year RS compared with those in DLBCL, NOS group (2-year RS, 88.6% vs. 67.0%; P = .0022; 5-year RS, 88.6% vs. 61.2%; P = .0006); whereas patients with intravascular large B-cell lymphoma had a lower 2-year RS (33.4% vs. 67.0%; P = .0397). There was no significant difference in gender in the 2- and 5-year RS for patients with DLBCL. The 2- and 5-year RS for patients with FL were 92.6% and 87.8%, respectively. Female patients with FL had significantly higher 5-year RS compared with male FL patients (89.4% vs. 86.2%; P = .047). Among the FL subtypes, FL grade I to II had the highest 2- and 5-year RS (94.0% and 90.0, respectively), whereas FL grade III had the lowest 2-year RS (90.0%) and FL, NOS had the lowest 5-year RS (85.3%). There was no significant racial difference in RS for FL. Multivariable Cox regression demonstrated that patients > 60 years, with advanced stage disease and presenting with B symptoms had worse survival for both DLBCL and FL. Additionally, black race, male gender, and presence of an extranodal primary site were associated with worse OS in DLBCL (Table 3).
Table 3.
Multivariable Cox Regression Models of Predictors of Mortality in Patients With DLBCL and FL
| Factors | DLBCL (n = 7701; 20.0%) |
FL (n = 4183; 10.9%) |
||
|---|---|---|---|---|
| HR | 95% CI | HR | 95% CI | |
| Age, y | ||||
| 18–60 | 1.00 | 1.00 | ||
| >60 | 2.09 | 1.89–2.31 | 3.24 | 2.64–3.97 |
| Gender | ||||
| Female | 1.00 | 1.00 | ||
| Male | 1.13 | 1.03–1.24 | 1.15 | 0.97,1.36 |
| Race | ||||
| White | 1.00 | 1.00 | ||
| Black | 1.20 | 1.07–1.34 | 1.13 | 0.87–1.46 |
| Other | 0.79 | 0.54–1.14 | 0.17 | 0.02–1.21 |
| Disease stage | ||||
| I/II | 1.00 | 1.00 | ||
| III/IV | 1.61 | 1.45–1.78 | 1.22 | 1.02–1.45 |
| Extranodal primary site | ||||
| No | 1.00 | 1.00 | ||
| Yes | 1.17 | 1.06–1.28 | 1.08 | 0.86–1.38 |
| B symptoms | ||||
| No | 1.00 | 1.00 | ||
| Yes | 1.17 | 1.06–1.28 | 1.50 | 1.24–1.81 |
The multivariable model included each of the variables in the table: age, gender, race, disease stage, extranodal site of disease, and B-symptoms.
Abbreviations: CI = confidence interval; DLBCL = diffuse large B-cell lymphoma; FL = follicular lymphoma; HR = hazard ratio.
Discussion
Studies investigating lymphoma incidence patterns in the US commonly focus on analyzing nationwide data to explore trends over time,2,20 but similar studies at the state and regional levels are lacking. Studies at the state and regional level are particularly important for population health surveillance and designing targeted interventions capable of addressing regional disparities. Additionally, the drivers of disparities in incidence and outcomes may vary regionally, and granular data could help to uncover the etiology of geographic differences. We discovered only one study examining time trends of NHL incidence at the state-level in Pennsylvania. This study found that increases in NHL incidence in Pennsylvania between 1985 and 2004 were similar to the national trends.21 However, analyzing state-level data in Georgia also is important owing to the unique characteristics of its population. With a larger percentage of minorities compared with the US population, an NCI-designated Comprehensive Cancer Center with a catchment area that matches the population of the state, and a SEER cancer registry, Georgia has a unique opportunity to match population-based registry data to a cancer center catchment area, which can allow for the design of local interventions to address the state findings.
Our study is, to our knowledge, the first to analyze the temporal trends in lymphoid malignancies at the state level in Georgia. Our research indicated that there was a significant increase in NHL incidence in Georgia from 2001 to 2015, which was different from the overall declining time trends in the recent 10 years based on the national SEER data.1 Although the overall incidence of NHL in the US has declined in the past decade, regional and racial disparities still exist and need to be better understood so that targeted interventions may be developed. In our study, even though overall NHL incidence from 2001 to 2015 was higher in whites than in blacks, further analyses should be performed to assess whether racial disparities exist in the rate of increased incidence over time. Although these analysis pinpoint key targets to be addressed in the catchment area for addressing important disparities in incidence and outcome for the region, further work is needed to assess possible factors underlying this disparity, including behaviors and exposures to environmental risk factors, as well as socioeconomic determinants. We have begun to examine some of these factors in Georgia including using geospatial epidemiology to identify exposures such as benzene that are associated with clusters of NHL, DLBCL, and other lymphomas.14,18,19 Racial and socioeconomic difference in residential and occupational environment may contribute to geospatial differences in exposures for these and other factors that can lead to disparities in the incidence trends for lymphoid malignancies. In addition, a multi-level model and approach4 can be applied to identify and delineate, specific disparities where intervention can address the root causes and improve outcomes. Possible interventions for potential etiologies of disparities include: social support, housing, and financial support to reduce patient-level barriers in access to care; connections to patient advocacy groups and internet-based patient education to overcome inequities in disease-specific information; and molecularly targeted therapies and clinical trials to address disparities owing to biological differences in molecular subtypes or immunologic responses to treatment.
Previous studies have reported that males had a higher incidence of NHL than females.2,20,21 Our study found that males had a significant excess risk in both DLBCL and FL. Possible reasons for male predominance in NHL incidence may be that occupational exposures related to lymphomagenesis usually exist in male-driven industries,2,22,23 whereas gravidity provides a protective role against NHL occurrence in females.24,25 Identifying racial and gender differences in incidence for a specific catchment area like Georgia can delineate areas for focused epidemiologic investigation and eventually promote risk reduction strategies.
Our research also indicates that the overall incidence rates for both DLBCL and FL in Georgia were significantly lower in black individuals compared with whites. This echoes our previous work2,9,26 describing different racial patterns for age-specific incidence rates for DLBCL and FL. This finding warrants further consideration owing to the multifactorial etiologies of both DLBCL and FL identified in international studies.27,28 The InterLymph group performed the most comprehensive evaluation of lymphoma epidemiology ever, involving pooled individual-level data from 17,471 NHL cases and 23,096 controls from 20 case-control studies to identify common and heterogeneous sets of risk factors for DLBCL and FL, which included family history of NHL, occupational exposure, active autoimmune disease, smoking, HCV infection, blood transfusion, SES, high body mass index as a young adult, genetic factors, and others.23,27,28 However, very few African American patients were represented in these studies, thus prohibiting discussion of risk factors for NHL in this population. The distribution of specific risk factors23,29,30 could differ by race and subtype, which may partially explain the observed differences in the incidence patterns across subtypes and races. Alternatively, African Americans (and other racial groups) may have race/ethnic-specific risk factors that have not been previously uncovered because these groups are traditionally under-represented in epidemiologic studies. Our current work with the Lymphoma Epidemiology of Outcomes (LEO) study, aims to accurately represent race and ethnicity distributions across NHLs, and leverage population-based cancer registries to assist in identifying and improving lymphoma outcomes at a state and national level.31
In addition, several recent studies have demonstrated racial disparities in the presentation and outcomes of NHL subtypes.7,9,10,13,32,33 In this study, we demonstrated the existence of racial disparities in clinical presentation and outcomes for both DLBCL and FL in Georgia. Based on Georgia NHL data in SEER, black individuals were more likely to be diagnosed at a younger age across most B-cell NHLs, mycosis fungoides/Sezary syndrome and peripheral T-cell lymphoma. Blacks with DLBCL presented nearly 11 years younger than whites with DLBCL, which was the largest difference in age at diagnosis for all NHL subtypes across race. Nearly two-thirds of blacks versus one-fourth of whites were diagnosed with DLBCL at age < 60 years, whereas nearly one-half of black patients versus one-third of white patients were diagnosed with FL at age < 60 years. Future analyses should examine whether these racial differences in the age of onset across lymphoma subtypes persist or change over time.
We also found that blacks with DLBCL were more likely to be diagnosed at an advanced stage, present with B symptoms, and have inferior survival compared with whites, which was in accordance with previous research using national SEER data and the National Cancer DataBase.3,9 However, we found that blacks were less likely to present with an extranodal primary site in Georgia, which conflicted with results of previous studies based on nationwide data. Several factors may impact the outcomes of patients with DLBCL and contribute to racial disparities in DLBCL survival. At present, the reasons for these racial differences in disease status at presentation remain unknown. Multi-level models are needed to structure suspected relationships between factors and lymphoma outcomes and guide future research.4,32,33 We suggest that leveraging data from population-based resources like cancer registries and linking patient-level data to epidemiologic and biological studies may provide opportunities to explain these differences.
Disparities in treatment, SES, insurance status, and genetic variations may be additional factors that impact survival in different racial groups. We proposed a multi-level model to examine and disentangle the interactions between these factors on lymphoma patient outcomes.4 Our previous study demonstrated that blacks with DLBCL were less likely to receive chemotherapy or chemo-immunotherapy compared with whites.3 Tao and colleagues reported that living in lower SES neighborhoods was associated with a worse survival after DLBCL, and this disparity was greater in patients < 65 years.34 As racial inequality in SES continue to exist, we suspect that racial differences in SES may play an increasing role in racial disparities in DLBCL outcomes. However, research on the effect of changes in population demographics, insurance status, and SES over time on cancer outcomes will require building on longitudinal surveillance datasets, such as the LEO cohort, with enhancements to address changes in the availability of novel therapies.
In addition, racial differences in genetic variants may contribute to racial disparities in lymphoma presentation and survival. One study reported a lower frequency of the t(14;18) translocation in DLBCL in Chinese patients than in Western patients.35 Other studies involving patients from China, Turkey, Malaysia, Japan, and the US have suggested differences in the frequency of the favorable biological subtype, germinal-center like DLBCL and the unfavorable subtype, activated B-cell like DLBCL across racial/ethnic groups.31,35–40 Recent studies provided detailed characterization of DLBCL genomics,41–45 but it remains unknown how these findings apply across racial and ethnic groups. In our study, significant racial differences in clinical manifestations of FL were only observed in age at diagnosis and the presentation of B symptoms. Although blacks had the lowest 2-year and 5-year RS among the 3 racial groups in FL, the OS for all racial groups was > 85% at 5 years. These results were in accordance with our previous report from the National LymphoCare Study, indicating that, although racial differences in the treatment patterns for FL existed, there was no significant difference in FL OS across racial groups.7 An additional study examined the impact of race/ethnicity, age, gender, and SES on FL outcomes based on national SEER data (1992–2009).46 Inferior overall survival for Hispanic patients was observed when compared with whites during the period 2001 to 2009, and patients with lower SES had an inferior survival.
Examining outcomes across cancer subtypes, with the help of local cancer registries, may help in prioritizing catchment area research questions based on their expected impact on population outcomes. The collaboration between local cancer registries and their cancer centers could help to address and reduce regional disparities by uncovering regional-specific outcomes through cancer registry data, and designing and implementing interventions through the cancer centers to address these disparities. Studies of race and ethnicity have become increasingly complex in the modern era, challenging our efforts to distinguish biological, socioeconomic, and other determinants of cancer outcomes. Future studies investigating NHL disparities across racial and ethnicity categories should include biological measures of admixture and ancestry as well as robust capture of data on SES, health literacy, social support, and other known social determinants of health.
Limitations
Owing to the complexity and heterogeneity of lymphoid malignancies, as well as the deepening understanding of lymphoma, there have been many changes in the coding schema of lymphoma subtypes over time. New subtypes have been added as scientific knowledge of the disease expands, and some subtypes are re-categorized as entity definitions are modified. Such changes might infiuence the classification of NHL across the study period and impact the results of our analyses. The lack of surveillance data on recurrence in the SEER registries is also a limitation of this analysis. As more therapies are developed for second relapses and beyond, the relationships between recurrence and mortality has diminished, especially in FL. For more aggressive diseases like DLBCL, the capture of recurrence is critical because access to potentially curative therapies such as stem cell transplantation and chimeric antigen receptor T-cell therapy may vary by SES and insurance status. Another limitation of this analysis was the amount of missing data on stage (5%−13%) and B symptoms (> 30%). Future research should strive to enhance population-based datasets to corroborate these results and augment registry data for research question important to a catchment area. In addition, some NHL and DLBCL subtypes are particularly rare or have an extremely low incidence in certain racial groups, resulting in a lack of sample size to provide robust results. Future research should strive to enhance population-based datasets to corroborate these results and augment registry data for research questions important to a catchment area. Additional biological characterization of patient samples using modern genomics based on recently published approaches41–44 are needed to understand the relationships between biological factors and other determinants of lymphoma outcomes.
Conclusion
Our analyses indicate that racial disparities exist in NHL incidence rates, disease presentation in DLBCL and FL, and survival outcomes for patients with DLBCL in Georgia. However, the factors that account for racial disparities remain unclear, and further research is essential for clarifying possible mechanisms. Although our study harbors some limitations, our findings provide a better understanding of regional NHL racial disparities. Ongoing research in necessary to facilitate the development of targeted interventions and strategies to overcome the existing NHL disparities in a catchment region.
Supplementary Material
Clinical Practice Points.
Most current research has explored racial disparities in lymphoma at a national level. This study sought to identify and describe racial disparities in NHL in a specific catchment area where its findings could be used to implement new clinical practices aimed at addressing these disparities.
Black patients in Georgia with DLBCL were more likely than whites to present with stage III/IV disease and B symptoms and had significantly worse 2-year and 5-year RS.
Regional information on racial differences in lymphoma incidence and presentation can help to inform future clinical practices directed at improving outcomes in poor-risk populations.
Acknowledgments
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award numbers K24CA208132 and U01CA195568, and by a Diversity Supplement to U01CA195568. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Disclosure
Dr Flowers has served as a consultant for: Abbvie, Astra Zeneca, Bayer, BeiGene, Celgene, Denovo Biopharma, Genentech/Roche (unpaid), Gilead, OptumRx, Karyopharm, MEI Pharmaceuticals, Pharmacyclics/Janssen, and Spectrum. Related to Dr Flowers’ research, Emory University has received research funding from: Abbvie, Acerta, BeiGene, Celgene, Gilead, Genentech/Roche, Janssen Pharmaceutical, Millennium/Takeda, M2Gen, Pharmacyclics, TG Therapeutics, Burroughs Wellcome Fund, Eastern Cooperative Oncology Group, National Cancer Institute, and the V Foundation. The remaining authors have stated that they have no conflicts of interest.
Supplemental Data
Supplemental tables accompanying this article can be found in the online version at https://doi.org/10.1016/j.clml.2019.06.009.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin 2019; 69: 7–34. [DOI] [PubMed] [Google Scholar]
- 2.Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin 2016; 66:443–59. [DOI] [PubMed] [Google Scholar]
- 3.Flowers CR, Fedewa SA, Chen AY, et al. Disparities in the early adoption of chemoimmunotherapy for diffuse large B-cell lymphoma in the United States. Cancer Epidemiol Biomarkers Prev 2012; 21:1520–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Flowers CR, Nastoupil LJ. Socioeconomic disparities in lymphoma. Blood 2014; 123:3530–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goldstein JS, Nastoupil LJ, Han X, Jemal A, Ward E, Flowers CR. Disparities in survival by insurance status in follicular lymphoma. Blood 2018; 132:1159–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Keegan THM, Li Q, Steele A, et al. Sociodemographic disparities in the occurrence of medical conditions among adolescent and young adult Hodgkin lymphoma survivors. Cancer Causes Control 2018; 29:551–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nabhan C, Byrtek M, Taylor MD, et al. Racial differences in presentation and management of follicular non-Hodgkin lymphoma in the United States: report from the National LymphoCare Study. Cancer 2012; 118:4842–50. [DOI] [PubMed] [Google Scholar]
- 8.Ritter AJ, Goldstein JS, Ayers AA, Flowers CR. Rural and urban patients with diffuse large B-cell and follicular lymphoma experience reduced overall survival: a National Cancer DataBase study. Leuk Lymphoma 2019; 60:1656–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shenoy PJ, Malik N, Nooka A, et al. Racial differences in the presentation and outcomes of diffuse large B-cell lymphoma in the United States. Cancer 2011; 117: 2530–40. [DOI] [PubMed] [Google Scholar]
- 10.Shenoy PJ, Malik N, Sinha R, et al. Racial differences in the presentation and outcomes of chronic lymphocytic leukemia and variants in the United States. Clin Lymphoma Myeloma Leuk 2011; 11:498–506. [DOI] [PubMed] [Google Scholar]
- 11.Caglayan C, Terawaki H, Ayer T, et al. Assessing the effectiveness of treatment sequences for older patients with high-risk follicular lymphoma with a multistate model. Clin Lymphoma Myeloma Leuk 2019; 19:300–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen Q, Ayer T, Nastoupil LJ, et al. Population-specific prognostic models are needed to stratify outcomes for African-Americans with diffuse large B-cell lymphoma. Leuk Lymphoma 2016; 57:842–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Flowers CR, Glover R, Lonial S, Brawley OW. Racial differences in the incidence and outcomes for patients with hematological malignancies. Curr Probl Cancer 2007; 31:182–201. [DOI] [PubMed] [Google Scholar]
- 14.Bulka C, Nastoupil LJ, McClellan W, et al. Residence proximity to benzene release sites is associated with increased incidence of non-Hodgkin lymphoma. Cancer 2013; 119:3309–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.United States Census Bureau. United States Census Bureau: QuickFacts 2019; Vol. 2019 Available at: https://www.census.gov/quickfacts/fact/table/US/PST045218. Accessed: March 19, 2019. [Google Scholar]
- 16.Howlader N, Noone A, Krapcho M, et al. SEER Cancer Statistics Review, 1975–2012 Bethesda, MD: National Cancer Institute; 2015. [Google Scholar]
- 17.Surveillance E, and End Results (SEER) Program. SEER*Stat Database: Incidence - SEER 18 Regs Research Data Hurricane Katrina Impacted Louisiana Cases, Nov 2017 Sub (1973–2015 varying) - Linked To County Attributes - Total U.S., 1969–2016 Counties. In: National Cancer Institute, Surveillance Research Program ed Vol. released April 2018, based on the November 2017 submission 2018. [Google Scholar]
- 18.Switchenko JM, Bulka C, Ward K, et al. Resolving uncertainty in the spatial relationships between passive benzene exposure and risk of non-Hodgkin lymphoma. Cancer Epidemiol 2016; 41:139–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bulka C, Nastoupil LJ, Koff JL, et al. Relations between residential proximity to EPA-designated toxic release sites and diffuse large B-cell lymphoma incidence. South Med J 2016; 109:606–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee JS, Bracci PM, Holly EA. Non-Hodgkin lymphoma in women: reproductive factors and exogenous hormone use. Am J Epidemiol 2008; 168:278–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Morton LM, Wang SS, Devesa SS, Hartge P, Weisenburger DD, Linet MS. Lymphoma incidence patterns by WHO subtype in the United States, 1992–2001. Blood 2006; 107:265–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Monnereau A, Slager SL, Hughes AM, et al. Medical history, lifestyle, and occupational risk factors for hairy cell leukemia: the InterLymph Non-Hodgkin Lymphoma Subtypes Project. J Natl Cancer Inst Monogr 2014; 2014:115–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morton LM, Slager SL, Cerhan JR, et al. Etiologic heterogeneity among non-Hodgkin lymphoma subtypes: the InterLymph Non-Hodgkin Lymphoma Subtypes Project. J Natl Cancer Inst Monogr 2014; 2014:130–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Horesh N, Horowitz NA. Does gender matter in non-hodgkin lymphoma? Differences in epidemiology, clinical behavior, and therapy. Rambam Maimonides Med J 2014; 5:e0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Prescott J, Lu Y, Chang ET, et al. Reproductive factors and non-Hodgkin lymphoma risk in the California Teachers Study. PLoS One 2009; 4:e8135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li Y, Wang Y, Wang Z, Yi D, Ma S. Racial differences in three major NHL subtypes: descriptive epidemiology. Cancer Epidemiol 2015; 39:8–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cerhan JR, Kricker A, Paltiel O, et al. Medical history, lifestyle, family history, and occupational risk factors for diffuse large B-cell lymphoma: the InterLymph Non-Hodgkin Lymphoma Subtypes Project. J Natl Cancer Inst Monogr 2014; 2014:15–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Linet MS, Vajdic CM, Morton LM, et al. Medical history, lifestyle, family history, and occupational risk factors for follicular lymphoma: the InterLymph Non-Hodgkin Lymphoma Subtypes Project. J Natl Cancer Inst Monogr 2014; 2014:26–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chihara D, Nastoupil LJ, Williams JN, Lee P, Koff JL, Flowers CR. New insights into the epidemiology of non-Hodgkin lymphoma and implications for therapy. Expert Rev Anticancer Ther 2015; 15:531–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Koff JL, Chihara D, Phan A, Nastoupil LJ, Williams JN, Flowers CR. To each its own: linking the biology and epidemiology of NHL subtypes. Curr Hematol Malig Rep 2015; 10:244–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Flowers CR, Nastoupil L, Borate U, et al. Racial disparities in cell of origin among DLBCL patients. Blood 2012; 120. [Google Scholar]
- 32.Flowers CR, Nastoupil LJ. Identifying racial differences in nodular lymphocyte-predominant Hodgkin lymphoma. Cancer 2015; 121:3387–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Flowers CR, Pro B. Racial differences in chronic lymphocytic leukemia. Digging deeper. Cancer 2013; 119:3593–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tao L, Foran JM, Clarke CA, Gomez SL, Keegan TH. Socioeconomic disparities in mortality after diffuse large B-cell lymphoma in the modern treatment era. Blood 2014; 123:3553–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yamauchi A, Fujita S, Ikeda J, et al. Diffuse large B-cell lymphoma in the young in Japan: a study by the Osaka Lymphoma Study Group. Am J Hematol 2007; 82: 893–7. [DOI] [PubMed] [Google Scholar]
- 36.Chen Y, Han T, Iqbal J, et al. Diffuse large B-cell lymphoma in Chinese patients: immunophenotypic and cytogenetic analyses of 124 cases. Am J Clin Pathol 2010; 133:305–13. [DOI] [PubMed] [Google Scholar]
- 37.Flowers CR, Nastoupil LJ, Bernal-Mizrachi L, Rose AC, Sinha R. New challenges in the management of diffuse large B-cell lymphoma. Treat Strateg Hematol 2012; 2:68–73. [PMC free article] [PubMed] [Google Scholar]
- 38.Peh SC, Gan GG, Lee LK, Eow GI. Clinical relevance of CD10, BCL-6 and multiple myeloma-1 expression in diffuse large B-cell lymphomas in Malaysia. Pathol Int 2008; 58:572–9. [DOI] [PubMed] [Google Scholar]
- 39.Shia AK, Gan GG, Jairaman S, Peh SC. High frequency of germinal centre derivation in diffuse large B cell lymphoma from Asian patients. J Clin Pathol 2005; 58:962–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Alacacioglu I, Ozcan MA, Ozkal S, et al. Prognostic significance of immunohisto-chemical classification of diffuse large B-cell lymphoma. Hematology 2009; 14:84–9. [DOI] [PubMed] [Google Scholar]
- 41.Schmitz R, Wright GW, Huang DW, et al. Genetics and pathogenesis of diffuse large B-cell lymphoma. N Engl J Med 2018; 378:1396–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arthur SE, Jiang A, Grande BM, et al. Genome-wide discovery of somatic regulatory variants in diffuse large B-cell lymphoma. Nat Commun 2018; 9: 4001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chapuy B, Stewart C, Dunford AJ, et al. Molecular subtypes of diffuse large B cell lymphoma are associated with distinct pathogenic mechanisms and outcomes. Nat Med 2018; 24:679–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Reddy A, Zhang J, Davis NS, et al. Genetic and functional drivers of diffuse large B cell lymphoma. Cell 2017; 171:481–94.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhang J, Grubor V, Love CL, et al. Genetic heterogeneity of diffuse large B-cell lymphoma. Proc Natl Acad Sci U S A 2013; 110:1398–403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nabhan C, Aschebrook-Kilfoy B, Chiu BC, Kruczek K, Smith SM, Evens AM. The impact of race, age, and sex in follicular lymphoma: a comprehensive SEER analysis across consecutive treatment eras. Am J Hematol 2014; 89:633–8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.