Abstract
The enantioselective dichlorination of alkenes is a continuing challenge in organic synthesis owing to the limitations of selective and independent antarafacial delivery of both electrophilic chlorenium and nucleophilic chloride to an olefin. Development of a general method for the enantioselective dichlorination of isolated alkenes would allow access to a wide variety of polyhalogenated natural products. Accordingly, the enantioselective suprafacial dichlorination of alkenes catalyzed by electrophilic organoselenium reagents has been developed to address these limitations. The evaluation of twenty-three diselenides as precatalysts for enantioselective dichlorination is described, with a maximum e.r. of 76:24 Additionally, mechanistic studies suggest an unexpected Dynamic Kinetic Asymmetric Transformation (DyKAT) process may be operative.
Keywords: Enantioselective dichlorination, Electrophilic selenium, Redox catalysis
1. Introduction
The vicinal dihalogenation of alkenes is familiar to every organic chemist and is taught in every introductory organic chemistry course. This special stature is a consequence of its generality for a wide variety of alkenes and the predictable diastereoselectivity associated with it. The relative configuration of the dihalide products and the mechanistic explanation for the diastereoselectivity observed were a matter of great debate in the early 20th century [1,2]. The intermediacy of haliranium ions and the stereochemical requirement of invertive opening eventually gained acceptance, and much later the isolation and characterization of stable bromiranium ions established their existence with certainty [3,4].
Despite this foundational mechanistic understanding and the high diastereoselectivity obtained with many electrophilic halogen sources, methods available for catalytic, enantioselective vicinal halogenation of alkenes are distinctly lacking when compared to the myriad other vicinal, difunctionalization reactions of alkenes such as epoxidation [5–7], dihydroxylation [8–10], hydrogenation [11–13], and intramolecular halofunctionalization [14–16].
This deficiency is not for a lack of necessity; several classes of vicinally polyhalogenated (usually marine) natural products could be efficiently prepared were a selective alkene dihalogenation available (Fig. 1) [17]. Owing in part to this limited synthetic accessibility, much less is known about the biological activities of these natural products compared to terrestrial bacterial, plant, and fungi isolates.
Fig. 1.
Representative chlorosulfolipids, a subclass of halogenated natural products.
Synthetic approaches to these natural products involve diastereo- and enantioselective generation of oxygen-bearing stereocenters through historically well-developed asymmetric oxidations and subsequent stereospecific replacement of oxygen with chlorine or alcohol-directed diastereoselective dichlorination [18–22]. Although several of these natural products have been obtained in what are undoubtedly impressive feats of total synthesis, the multiple steps required to enantioselectively transform an alkene into a vicinal dichloride highlight a major shortcoming in the synthetic organic chemist’s toolkit.
Although several attempts have been made to address this challenge in the last decade (vide infra), all are reliant on halenium ion (or equivalent) delivery to an alkene, resulting in a haliranium ion, which is subsequently opened by a halide anion to afford the vicinal dihalide in an overall antarafacial fashion. Numerous hurdles must be overcome to design an alkene dihalogenation by this approach (Scheme 1) [23,24]. In a simplified system, chiral catalyst (Cat*), coordinated by either hydrogen bonding, Lewis base association, or ion pairing to the electrophilic halogen source (X+), delivers the halenium ion to the olefin with enantiofacial selectivity. The possibility for enantiofacial selectivity in this transfer may be attenuated by the stereoelectronic requirement of interaction between the alkene π-orbital and the Cat*–X σ* orbital, placing the most significant steric effects of the chiral catalyst far from the alkene, axially opposite the halenium ion (Scheme 1, right). The haliranium ion thus formed is opened by halide (X−) to afford the dihalide product. To achieve high enantioselectivity (assuming high enantiofacial selectivity), first, halenium ion transfer to the alkene should be irreversible [25]. Second, the haliranium ion thus formed should be configurationally stable. Finally, nucleophilic trapping of the haliranium ion must be biased toward one carbon terminus of the ion, as the two possible ring-opened products are enantiomeric. Consequently, both haliranium ion formation and capture must be highly selective to afford highly enantioenriched vicinal dihalide product.
In spite of the hurdles to the development of an enantioselective anti-dihalogenation [23], four methods for catalytic, enantioselective dichlorination following this process have been developed (Fig. 2) [26–30].
Fig. 2.
State of the art for catalytic enantioselective dichlorination.
Nicolaou and coworkers first achieved moderate to good enantioselectivities for the dichlorination of cinnamyl alcohols 1 [26]. This method employs Lewis base (ligand-accelerated) catalysis to activate an iodosobenzene dichloride derivative as the chlorenium ion and chloride source. Both the alcohol and aromatic ring on the substrate alkene are necessary to achieve good selectivity, most likely due to the requirement for hydrogen-bonded ligand association to the substrate and a strong electronic bias for selective chloriranium ion opening.
Adaptation of these conditions by Borhan and coworkers to the dichlorination of allylic amides 3 afforded a wide variety of E and Z-disubstituted alkenes with excellent yields and selectivities [27]. In contrast to the method of Nicolaou and coworkers, Cinnamyl and 3,3-dialkyl (trisubstituted) alkene substrates afforded dichloride products with lower enantiomeric ratios, suggesting that the electronic bias of chloriranium opening generally favors the carbon proximal to nitrogen, and stabilization of partial positive charge at the distal carbon reduces that selectivity.
Most recently, Hennecke and coworkers have further adapted this method, through modification of cinchona alkaloid-derived ligand, to afford dichloride products in modest to excellent selectivities from alkenes 5 not bearing allylic hydrogen-bonding functional groups [28]. The method does nonetheless still rely on styrenyl substrates to bias chloriranium opening toward the carbon proximal to the benzene ring. Furthermore, the substrate scope was limited to Z-olefins and only a select few electron-poor dihydronaphthalenes gave dichloride products with e.r. greater than 90:10.
Perhaps the most general method, developed by Burns and coworkers, requires allylic alcohol substrates 7 but has a much broader scope. Here, a halotitanium triisopropoxide complex and a halenium ion source (t-BuOCl or NBS) with catalytic amounts of a Salen-type ligand provide vicinal anti-dihalide products. Exquisite selectivity is obtained in bromochlorination, dibromination, and dichlorination across a wide variety of 1,1-di-, 1,2-E- and Z-di-, and trisubstituted olefins. The scope and selectivity of the method rivals the Sharpless asymmetric epoxidation (Scheme 2) [29,30].
Similar to the method of Nicolaou et al., this transformation is suggested to operate within a similar manifold of Lewis base, or ligand accelerated, catalysis. The halenium ion source is only reactive enough to oxidize the alkene once it has been coordinated to a ligated titanium center (coordination to chlorotriisopropoxytitanium is insufficient as little background reaction is observed in the absence of ligand). A structural limitation to the scope of this antarafacial dihalogenation is the requirement for an allylic alcohol to direct halenium ion and halide addition to the double bond. Although this feature can be leveraged to their advantage as evidenced by the presence of simple alkenes left untouched in a variety of products, the limitation does emphasize the need to overcome unselective haliranium ion opening by tethering the halide source, tightly bound to titanium, to the alcohol.
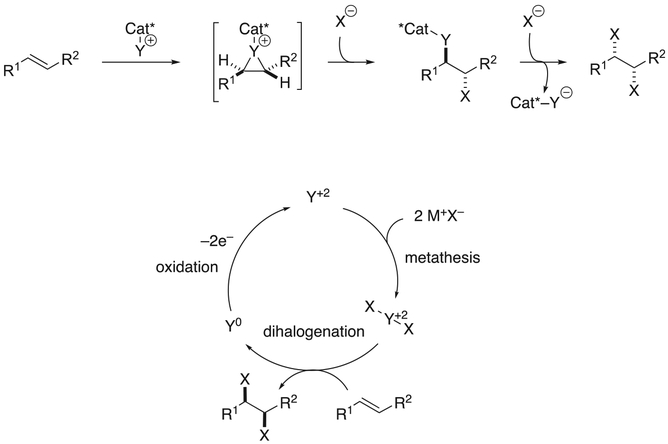
In contrast to these antarafacial vicinal dihalogenations, suprafacial dihalogenation (pictured below as proceeding through a different -iranium ion intermediate but true for all suprafacial dihalogenations) [31–34] converges to a single enantiomer, or does not proceed through an intermediate that could diverge (Scheme 3). Here, an electrophilic complex Cat*–Y reacts with the alkene to form a non-halo -iranium ion. This intermediate is then opened at either constitutionally heterotopic carbon to afford a mixture of isomers. This isomeric mixture converges to a single dihalide product by displacement of Cat*–Y (now in reduced form) from the alkane backbone of the product. Thus, the only requirements for a selective process are now irreversible -iranium ion formation and configurational stability of that intermediate.
Fig. 3.
General structure for a chiral, enantioenriched organoselenium reagent. Y = O, N, S; R1 = R2 = alkyl, aryl; X = Cl, Br, OTf, HSO4, PF6, etc.
This alternative approach is distinguished by the presence of the chiral, enantioenriched catalyst backbone Cat* in the -iranium ion and ring-opened intermediates, as Y is, unlike X, an atom with valence greater than 1. This higher valence also allows for greater variety in the transition state geometry of -iranium ion formation (Scheme 3, right) and the portion of the catalyst bearing features that convey stereochemical information can be brought in closer proximity to the olefin than in the necessarily linear Cat*–X–alkene complex (Scheme 1, right).
The mode by which two halogen atoms are incorporated into the product molecule is also unique compared to the antarafacial dihalogenations, as both equivalents of X are introduced as halide anions. The second halide equivalent displaces Cat*–Y from the product and results in the formation of anionic Cat*–Y− (Scheme 4). This catalyst must be reoxidized for the cycle to turn over, standing in contrast to the requirement of separate halide and halenium ion sources in conjunction for antarafacial vicinal dihalogenations. This type of transformation has been previously categorized as a member of the larger group of “group-transfer catalysis,” [35] but could be more precisely referred to as “redox catalysis.”
Fig. 4.
Scrambling of arylselenium(IV) chloride adduct with external olefin.
The structural limitations to the scope of antarafacial enantioselective dichlorination reactions could be overcome by employing a suprafacial dichlorination wherein the alkene need not be electronically (or otherwise) biased. In the case of a catalytic, enantioselective syn-dichlorination proceeding through an alternative -iranium ion, and provided that -iranium ion formation is irreversible, then formation of that ion alone would be stereo-determining. The ring-opening of this cationic intermediate is inconsequential to the final enantiomeric ratio, simplifying the challenge in simultaneous installation of adjacent halogenated stereocenters.
2. Background
As a prelude to the development of an enantioselective vicinal dihalogenation, these laboratories recently disclosed the stereospecific syn-dichlorination of alkenes using redox catalysis (Scheme 5) [24]. The reaction employs diphenyl diselenide (PhSeSePh 13) as the precatalyst, with benzyltriethylammonium chloride ([BnNEt3]+[Cl]− 14) as the chloride source and N-fluoropyridinium tetrafluoroborate ([PyF]+[BF4]− 15) as the oxidant. Trimethylsilyl chloride (Me3SiCl 16) was also employed in stoichiometric amounts to serve as a trap for the fluoride generated as a byproduct of oxidation. The reaction shows good functional group tolerance, proceeding smoothly in the presence of neighboring nucleophilic groups that would often be expected to participate in anchimeric assistance (12b, d), and leaving electron-poor olefins untouched (12c). Diastereoselectivity was excellent with respect to stereocenters already present on the alkene substrate (12f).
Scheme 5.
Stereospecific syn-dichlorination of olefins: representative scope.
The mechanistic hypothesis for the catalytic cycle is founded on previous knowledge of the elementary steps [36–40]. Namely, that diphenyl diselenide is oxidized to phenylselenyl trichloride with three equivalents of chlorine (or other electrophilic chlorine source), that areneselenenyl trichlorides react with olefins to give chloroselenylated adducts, and that chloroalkyl areneseleninium(IV) dichlorides can be displaced by chloride to afford syn-vicinal dichlorides (Scheme 6).
Scheme 6.
Stereospecific syn-dichlorination of olefins: mechanistic hypothesis. Ox = [BnNEt3]+[Cl]− (14) + [PyF]+[BF4]− (15) + Me3SiCl (16).
At reaction onset, diphenyl diselenide is oxidized by two equivalents of phenylselenium(II) chloride 17 with one equivalent each of chloride source 14, oxidant 15, and fluoride scavenger 16. Typically, alkene is not added until the other reaction components have been stirred for 10 min at room temperature, so this oxidation presumably occurs once more to afford phenylselenium(IV) trichloride 18. Subsequent ionization is assumed necessary to afford the charge-separated complex 19, which has the requisite open valence available for alkene coordination. Reaction with alkene produces the Se(IV) seleniranium ion 20, which can be opened antarafacially by chloride at either of the constitutionally heterotopic carbons, affording a mixture of isomers 21a and 21b. Ionization to 22 followed by stereospecific, SN2 displacement of the cationic phenylselenium(IV) chloride by chloride anion results in a convergence of these constitutional isomers to a single vicinal syn-dichloride product 12 and regenerates phenylselenium(II) chloride 17.
The β-chloroalkyl phenylselenium(IV) dichlorides 21 are postulated to be the catalytic resting state based on their presence in the 1H NMR spectra of incomplete reaction mixtures. Thus, the turnover-limiting step is likely the ionization of 21 to 22 and nucleophilic displacement by chloride to afford the product syn-dichloride 12.
Further catalytic turnover need not, however, proceed again through oxidation to 18, but could instead result from reaction of 17 with alkene to form Se(II) seleniranium ion 23. Antarafacial nucleophilic ring opening at the constitutionally heterotopic carbons by chloride affords β-chloroalkyl phenyl selenides 24a and 24b. This mixture of aryl-alkyl selenides is then further oxidized by 14/15/16 to return to the previous cycle at 21a and 21b. These two paths are indistinguishable by NMR analysis of an incomplete reaction mixture, as the resting state of either pathway could be at their point of convergence, i.e. 21. Indeed, the relative rates of oxidation of 17 versus its reaction with alkene may vary with the concentration of alkene or oxidant, both of which decrease as the reaction progresses.
Nevertheless, phenylselenyl chloride 17 was demonstrated to be an effective catalyst for the stereospecific vicinal syn-dichlorination of alkenes, allowing for the opportunity to develop an enantioselective method using a chiral, enantioenriched arylselenium catalyst. Diastereoselective selenofunctionalizations have been performed with a variety of chiral, enantioenriched electrophilic selenium reagents in the last several decades [41]. It was thus hypothesized that application of one of these reagents to syn-dichlorination could potentially result in good enantioselectivity, provided that the initial seleniranium ion formation proceeded with high enantiotopic face selectivity. Moreover, the structural features that lead to high selectivity in diastereoselective selenofunctionalizations have been identified, allowing for the formulation of selectivity models that can be used to improve the performance of these reagents [41,42]. Such chiral, enantioenriched organoselenium reagents typically feature an aryl selenide substituted at the 2-position by a stereogenic substituent bearing a coordinating heteroatom (O, N, S) (Fig. 3).
The coordination of the heteroatom to the electrophilic selenium atom (i.e. chalcogen bonding, Lewis base activation of Lewis acid) [43,44], rigidifies the structure and enhances electrophilicity. In addition to the use of these chiral, enantioenriched organoselenium reagents to stoichiometric, diastereoselective processes, several applications to catalytic enantioselective transformations have also been reported, namely in inter- and intramolecular selenofunctionalization-eliminations (Scheme 7) [45–50].
Scheme 7.
Catalytic, enantioselective allylic etherification with chiral, enantioenriched electrophilic selenium catalysts.
Previous studies in organoselenium catalyzed suprafacial dichlorination show that anti-dichlorination can be observed, likely owing to chloride oxidation to chlorine, when catalyst turnover becomes sluggish. Thus, the catalyst design was guided by the optimization of two key parameters: sufficient room-temperature turnover to avoid anti-dichloride formation, and excellent enantiotopic face selectivity.
3. Results and discussion
3.1. Synthesis and evaluation of chiral, enantioenriched diselenides
3.1.1. Diselenides bearing ether coordinating groups
Orienting experiments for enantioselective dichlorination began with the most synthetically accessible chiral diselenides that also displayed optimal diastereoselectivity in selenofunctionalization reactions. Thus, diether 34 was selected as the first target as it was accessible by well-described synthetic transformations in six steps from 2-bromoisophthalic acid [51,52]. These syn-dichlorination reactions were performed with conditions identical to those used in the racemic reaction catalyzed by diphenyl diselenide (Scheme 5). E-4-Hexen-1-yl benzyl ether 30 and benzyl E-4-hexenoate 31 were selected as substrates for their synthetic accessibility and lack of any strong electronic bias. Although the diselenide precatalyst effected dichlorination much more slowly than diphenyl diselenide, and a significant portion of anti-dichloride was obtained from background oxidation of chloride, the enantiomeric ratio of 61:39 for the syn-dichloride was an encouraging first result suggesting that enantioselection was achievable with chiral diselenides (Table 1, entry 1).
Table 1.
Syn-dichlorination selectivities of catalysts bearing ether coordinating Groups.
 | |||||
|---|---|---|---|---|---|
| entry | catalyst | product; d.r. (syn/anti)a | e.r.c | configurationb | |
| 1 |  |
34 | 32; 30:70 | 61:39 | R,R |
| 2 |  |
35 |
32; >95:5 33; >95:5 |
54:46 54:46 |
S,S S,S |
| 3 |  |
36 | 32; 90:10 | 55:45 | S,S |
| 4 |  |
37 | 32; 70:30 | 58:42 | R,R |
| 5 |  |
38 | 32; 90:10 | 50:50 | - |
The d.r. was determined by 1H NMR integration of diagnostic signals at HC(4) and HC(5) (see Experimental).
The absolute configuration of the major enantiomer was determined by elution order compared to dichloride of known configuration (for Y = O) or by analogy to the ester (for Y = H2) (see Supporting Information).
The e.r. was determined by chiral stationary phase HPLC (see Supporting Information).
This initial result suggested that the steric crowding about the selenium atom was contributing to the significant attenuation of the rate of the catalytic process and motivated the investigation of singly 2-substituted catalyst structures. Thus, the methyl ether 35 [53,54], (entry 2) gave substantially improved reactivity, but with a decrease in enantioselectivity. Exchanging the methyl ether for the benzyl ether in 36 (entry 3) resulted in similar selectivity, while the acetate 37 [55] (entry 4) improved enantioselectivity with concomitant decrease in diastereoselectivity. Exchange for a methoxymethyl acetal in 38 [56] (entry 5) gave nearly racemic product.
3.1.2. Diselenides bearing carbonyl coordinating groups
It was readily apparent that the oxygen substituent imparted relatively little influence on enantioselectivity. Finding a more selective catalyst would require examining different coordinating groups and catalyst geometries. Urea 39 [57] was therefore selected for its alternative binding mode in which the carbonyl oxygen serves as an alternative coordinating group, forming what is expected to be a 7-membered ring. The dichloride products obtained with urea 39 and with the novel pivalamide 40 (Table 2, entries 1 and 2) had enantiomeric ratios exceeding any previously obtained with absolute configuration matching what was obtained with acetate 37.
Table 2.
Syn-dichlorination selectivities of catalysts bearing carbonyl coordinating Groups.
 | ||||||
|---|---|---|---|---|---|---|
| entry | catalyst | 33 d.r. (syn/anti)a | e.r.c | e.s. | configurationb | |
| 1 |  |
39 | 70:30 | 65:35 | 65:35 | R,R |
| 2 |  |
40 | 90:10 | 65:35 | 65:35 | R,R |
| 3 |  |
41 | 83:17 | 62:38 | 62:38 | S,S |
| 4 |  |
42d | 95:5 | 58:42 | 59:41 | R,R |
| 5 |  |
43e | 80:20 | 61:39 | 64:36 | R,R |
| 6 |  |
44f,g | 80:20 | 73:27 | 74:26 | R,R |
The d.r. was determined by 1H NMR integration of diagnostic signals at HC(4) and HC(5) (see Experimental).
The absolute configuration of the major enantiomer was determined by elution order compared to dichloride of known configuration (see Supporting Information).
The e.r. was determined by chiral stationary phase HPLC (see Supporting Information).
The e.r. of the catalyst was 95:5.
The e.r. of the catalyst was 90:10.
The e.r. of the catalyst was 97:3.
Reaction with (Z)-31 afforded predominately the anti-dichloride with a d.r. of 70:30 and an e.r. of 55:45.
Further examination involved structural modifications to the pivalamide catalyst 40. Exchange of the methyl group adjacent to the stereocenter for an isopropyl group on 41 resulted in a decrease in both enantioselectivity and diastereoselectivity (entry 3). Substitution of the aromatic ring at the 6-position with a methoxy group or a benzene ring fusion (42 and 43) also resulted in a decrease in enantioselectivity (entries 4 and 5). Only placement of a methyl group at the 6-position in 44 resulted in an increase in enantioselectivity, albeit with an attendant decrease in diastereoselectivity (entry 6), likely due to increased steric crowding about the reactive center, thus attenuating the catalytic rate.
3.1.3. Diselenides bearing oxazoline coordinating groups
A reasonable hypothesis posited that the relatively weak binding of the ether and carbonyl coordinating groups were contributing to the low selectivities. Under this premise stronger Lewis basic donors beginning with readily accessible oxazolines were tested. Although oxazoline coordinating groups had previously been employed in diastereoselective selenofunctionalizations [58], diselenide 45 did not impart any appreciable selectivity in dichlorination (Table 3, entry 1). Catalysts 46–49 were prepared to examine the effect of 6-membered ring coordination structures. Systematic variation of the substituent adjacent to the coordinating nitrogen atom had very little effect on enantioselectivity (entries 2–5). The most significant effect of the stronger coordinating group can be observed in the decreased diastereoselectivity across all oxazoline catalysts, that is, the stronger coordinating group resulted in slower catalytic dichlorination and greater predominance of the background anti-dichlorination. Introduction of an adjacent, bulky group led to further deterioration of the diastereomeric ratio.
Table 3.
Syn-dichlorination selectivities of catalysts bearing oxazoline coordinating Groups.
 | |||||
|---|---|---|---|---|---|
| entry | catalyst | 33 d.r. (syn/anti)a | e.r.c | configurationb | |
| 1 |  |
45 | 60:40 | 50:50 | - |
| 2 |  |
46 | 90:10 | 54:46 | R,R |
| 3 |  |
47 | 90:10 | 53:47 | S,S |
| 4 |  |
48 | 83:17 | 52:48 | S,S |
| 5 |  |
49 | 70:30 | 50:50 | - |
The d.r. was determined by 1H NMR integration of diagnostic signals at HC(4) and HC(5) (see Experimental).
The absolute configuration of the major enantiomer was determined by elution order compared to dichloride of known configuration (see Supporting Information).
The e.r. was determined by chiral stationary phase HPLC (see Supporting Information).
3.1.4. Bicyclic diselenides
At this stage greater variation in catalyst backbone was sought, with a return to more weakly coordinating neighboring heteroatoms to improve diastereoselectivity. Rigid tetralin-derived organoselenium reagents had been previously described and appeared to be suitable candidates [54,56]. Additionally, related electrophilic selenium reagents had recently been employed catalytically to afford excellent enantioselectivities in oxidative lactonization [59]. Silyl ether 50 provided the dichloride in a d.r. of >95:5 and an e.r. of 74:26 (Table 4, entry 1). Exchanging the silyl ether for a benzoate, pivalate, or methoxymethyl acetal (51–53) resulted in a decrease in enantioselectivity (entries 2–4), whereas replacing the tert-butyldimethylsilyl ether with a triisopropylsilyl ether 54 resulted in only a slight improvement in enantioselectivity (entry 5). The ring-contracted indane catalyst 55 gave poorer selectivity, as did the ring-expanded benzosuberan catalyst 56 (entries 6 and 7). Notably, all of the catalysts derived from tetralin 50 led to substantially faster reactions than the more flexible enantioenriched diselenides employed above. For example, the dichlorination catalyzed by 50 was complete in 5 – e faster even than diphenyl diselenide.
Table 4.
Syn-dichlorination selectivities of rigid bicyclic Catalysts.
 | |||||
|---|---|---|---|---|---|
| entry | catalyst | 33 d.r. (syn/anti)a | e.r.c | configurationb | |
| 1 |  |
50 | >95:5 | 74:26 | R,R |
| 2 |  |
51 | >95:5 | 65:35 | R,R |
| 3 |  |
52 | >95:5 | 66:34 | R,R |
| 4 |  |
53 | >95:5 | 70:30 | R,R |
| 5 |  |
54 | >95:5 | 76:24 | R,R |
| 6 |  |
55 | >95:5 | 63:37 | R,R |
| 7 |  |
56 | >95:5 | 55:45 | R,R |
The d.r. was determined by 1H NMR integration of diagnostic signals at HC(4) and HC(5) (Experimental).
The absolute configuration of the major enantiomer was determined by elution order compared to dichloride of known configuration (see Supporting Information).
The e.r. was determined by chiral stationary phase HPLC (see Supporting Information).
3.2. Mechanistic investigations
Despite a wide variety of diselenide structures surveyed, many of which afforded excellent enantioselectivities in other transformations, the enantioselectivity had plateaued at 75:25. To better understand the mechanistic features that could be leading to reduced enantioselectivity, three potential sources were identified for consideration and evaluation: (1) low intrinsic selectivity in formation of the seleniranium ion; (2) competing pathways of Se(II) and Se(IV) addition to the olefin, each pathway imparting different selectivity; and (3) epimerization of catalytic intermediates owing to reversible addition prior to catalyst turnover.
3.2.1. Determination of selenium oxidation state at seleniranium ion formation
Before intrinsic selectivity could be considered, it was necessary to establish whether the transformation was proceeding through Se(II) or Se(IV) addition to the alkene. On the basis of X-ray crystal structural data [60] for intramolecularly coordinated arylselenium(II) and (IV) chlorides, the two have very different geometries. Coordinated arylselenium(II) chlorides assume a T-shape geometry, with chlorine atom opposite the coordinating group, whereas arylselenium(IV) trichlorides are pseudo-pyramidal. It follows that alkene approach to the electrophilic selenium atom at either oxidation state could be subject to entirely different steric environments. Furthermore, both pathways could potentially be operative over the course of the reaction, as relative rates of oxidation and seleniranium ion formation could be differentially dependent on oxidant and olefin concentrations, respectively.
It was hypothesized that with a low concentration of oxidant relative to olefin, seleniranium ion formation would proceed (at least predominately) at the Se(II) oxidation state. To maintain low oxidant concentration for the duration of the dichlorination, a solution of oxidant 15 was added by syringe pump over 8 h (the typical time to full conversion) to a solution of precatalyst 53, chloride source 14, fluoride scavenger 16, and alkene 31 (Scheme 8). The dichloride product obtained had the same e.r. (70:30) as when all reagents were combined at the start of the reaction. Likewise, it was hypothesized that with a low concentration of alkene relative to the oxidant, seleniranium ion formation would proceed at the Se(IV) oxidation state. Thus, slow addition of a solution 31 to a solution of 14, 15, 16 and 53 also afforded the same e.r. (70:30). Therefore, either the same pathway was operative in all three circumstances, or the Se(II) and Se(IV) seleniranium ion formation pathways impart nearly identical selectivities on the overall transformation.
Scheme 8.
Syringe pump addition experiments limiting selenium oxidation state.
A more informative experiment was then performed by measurement of the enantiomeric ratio of aliquots at 10% and 100% conversion. In a typical dichlorination reaction, all of the reagents save for the alkene substrate were combined for 10 min [24] to allow for oxidation of the diselenide precatalyst to the arylselenium trichloride species. Therefore, at 10% conversion (i.e. one catalytic turnover), the vast majority of dichloride product should arise from Se(IV) seleniranium ion formation. At a 1 mmol scale, the amount of syn-dichloride product obtained at 10% conversion was insufficient to achieve UV detection on HPLC. Thus, new olefin substrates were prepared to allow for detection at lower concentrations, while the reaction with alkene 31 was run on 5 mmol scale. For all three ester substrates there appeared to be no significant change (Table 5). Thus, the reaction most likely proceeds through Se(IV) seleniranium ion formation from initiation through to full conversion or both oxidations states afford similar selectivity.
Table 5.
Measurement of Dichloride e.r. at 10% and 100% Conversion.
 | |||
|---|---|---|---|
| entry | Alkene | e.r. at 10% conversiona | e.r. at 100% conversion |
| 1 | 31 | 76:24 | 75:25 |
| 2 | 57 | 79:21 | 76:24 |
| 3 | 58 | 77:23 | 75:25 |
e.r. was determined by chiral stationary phase HPLC (see Experimental).
Unfortunately little is known about the enantiotopic face selectivity of Se(IV) addition to olefins, as the vast majority of stoichiometric reactions have been performed using reagents at the Se(II) oxidation state. Attempts to measure diastereomeric ratio of the Se(IV) chloroalkyl adduct by combination of stoichiometric amounts of precatalyst 50 with either sulfuryl chloride or N-F oxidant/chloride and olefin led to 1H NMR spectra too complex to make any conclusions about the d.r. It was therefore difficult to rule out low facial selectivity as a contributor to the overall low selectivity. There was, however, one especially compelling piece of evidence that pointed to a mechanistic origin for low enantioselectivity.
3.2.2. Reversibility of ArSe(IV) addition to alkene
Early 1H NMR studies wherein the combination of the β-chloroalkylarylselenium(IV) dichloride prepared from E-octene 60 (formed in situ by treatment of a trifluoroacetate precursor 59 with ethereal HCl) [61] and E-1,4-diphenyl-2-butene 61 led to exchange between the olefins to afford mixture of alkenes and chloroselenylated adducts (Fig. 4). At t = 0 before the addition of hydrochloric acid, trifluoroacetate 59 and diphenylbutene are visible. However, 20 h after the addition of hydrochloric acid, there was a clear increase in spectral complexity as multiple chloroselenylated species began to appear. These new signals changed over the subsequent 45 h until equilibrium was reached. Comparison of the new component, 63, to an independently prepared chloroselenylated 1,4-diphenylbutene adduct confirmed its identity. The reverse reaction in which E-octene 62 was introduced to the β-chloroalkylarylselenium(IV) dichloride prepared from E-1,4-diphenyl-2-butene 63 likewise led to a similar mixture in the same amount of time, ruling out any thermodynamic bias.
The reversibility of seleniranium ion formation and opening indicated that, although the initial seleniranium ion formation may have been selective, it was not likely to be the enantiodetermining step. The mechanism of the transformation may be far more complicated, as diastereomeric ring-opened chloroalkylarylselenium(IV) dichloride adducts could now equilibrate prior to irreversible chloride displacement. Furthermore, the diastereomeric and constitutionally isomeric chloroselenylated adducts comprising this mixture have the potential to undergo SN2 displacement by chloride at different rates, funneling the equilibrium to the intermediate that undergoes fastest displacement by chloride, leading to a dynamic kinetic asymmetric transformation (DyKAT) (Scheme 9) [62].
Scheme 9.
Dynamic kinetic asymmetric transformation of equilibrating chloroselenylated adducts.
3.2.3. Intramolecular trapping of the seleniranium intermediate
To provide support for this new mechanistic hypothesis and to make seleniranium ion formation the enantiodetermining step, we sought to preclude reversibility by rapid capture using a tethered nucleophile. Adapting the method to chlorolactonization indeed proved simple. Exchanging benzyl E-hexenoate for 7-phenyl-4-heptenoic acid 64 under identical reaction conditions afforded a mixture of syn-γ-lactone 66, syn-δ-lactone 67, syn-dichlorinated carboxylic acid 68 [63], and various elimination products (Scheme 10). When catalyst 50 was employed with similar conditions, substituting 15 for 1-fluoro-2,4,6-trimethylpyridinium tetrafluoroborate 65 to improve reaction homogeneity, γ-lactone 66 was formed in a 78:9:13 ratio with 67 and 68, respectively, and with an e.r. of 81:19, marginally higher than the e.r. of syn-dichloride obtained from benzyl hexenoate with the same catalyst (vide supra). The constitutional isomer 67 was obtained with a lower e.r. of 60:40.
Scheme 10.
Chlorolactonization of E-7-phenylhept-4-enoic acid 64.
Modifying oxidant stoichiometry, chloride stoichiometry, and solvent all resulted in small changes to both the product ratio and, to a lesser degree, enantioselectivity. Unfortunately, the presence of the syn-dichloride regardless of the modified conditions indicated that reversibility was still operative owing to the unexpected intermolecular chloride capture outcompeting an intramolecular capture by carboxylate. It is possible that the equivalent of acid produced as a reaction byproduct was capable of activating the lactone towards displacement by the adjacent selenium to reform the seleniranium ion (Scheme 11).
Scheme 11.
Chlorolactonization of E-7-phenylhept-4-enoic acid: mechanistic hypothesis for reversibility.
4. Conclusions
In this study we have demonstrated that chiral, enantioenriched arylselenium(IV) chlorides can be employed as redox catalysts for the suprafacial dichlorination of alkenes to provide vicinal, syn-dichloride products with modest enantioselectivity. Twenty-three diaryl diselenides across four general classes were evaluated for their selectivity in the dichlorination, with a maximum e.r. of 76:24 obtained with triisopropylsilyl ether 54.
Higher product enantioenrichment may not be attainable through improvement of catalyst enantiofacial selectivity toward the alkene alone, as the free alkene and the catalytic resting state β-chloroalkylarylselenium(IV) dichloride appear to be in equilibrium. Furthermore, it does not appear that acceleration of the seleniranium ion opening by a tethered carboxylate results in any appreciable reduction in the reversibility of the overall transformation. There is, however, potential to improve selectivity by accelerating the second displacement to outcompete any reversibility by change of substrate to allow for intramolecular capture of the resting state intermediate. Likewise, there is potential to leverage the equilibrium for a DyKAT process with a second chiral catalyst to deliver chloride selectively to one isomer of the resting state intermediate.
Research employing chiral, enantioenriched diselenides as redox catalysts for suprafacial alkene difunctionalization, wherein the second displacement is accelerated, is ongoing.
5. Experimental
5.1. General experimental
All reactions were performed in 120 °C oven- or flame-dried glassware equipped with a Teflon-coated magnetic stir bar under inert nitrogen atmosphere unless otherwise noted. Catalytic chlorination reactions were performed in flame-dried dram vials equipped with magnetic stir bars and septa under nitrogen atmosphere.
Benzyltriethylammonium chloride was purchased from Oakwood Chemical and moved into the glove box without purification. N-fluoropyridinium tetrafluoroborate and N-fluoro-2,4,6-trimethylpyridinium tetrafluoroborate were purchased from TCI America and moved into the glove box without purification. Trimethylsilyl chloride (Aldrich, 97+%) was distilled from calcium hydride at atmospheric pressure. Reaction solvent acetonitrile (Fisher, HPLC grade) was distilled from calcium hydride. Reaction solvents hexane, tetrahydrofuran, diethyl ether, toluene, N,N-dimethylformamide and dichloromethane were dried by percolation through two columns packed with neutral alumina under a positive pressure of argon. Reaction solvent ethyl acetate was degassed by sparging with argon for 2 h and stored over molecular sieves. Reaction solvent pentane was distilled from sodium. Sodium hydride suspension in mineral oil was washed several times with hexanes and stored in the glove box. Solvents for chromatography were: hexanes (Fisher, ACS Grade), ethyl acetate (Fisher, ACS Grade), diethyl ether (Fisher, ACS Grade), pentane (J. T. Baker, reagent grade), petroleum ether (J. T. Baker, reagent grade), dichloromethane (Aldrich, ACS Grade), and tert-butyl methyl ether (Acros, reagent grade). Brine refers to a saturated solution of sodium chloride in water.
1H NMR, and 13C NMR were recorded on Varian Unity-400 (400 MHz, 1H; 100 MHz, 13C) and Varian Inova-500 (500 MHz, 1H; 126 MHz, 13C) spectrometers. Spectra were referenced to residual chloroform (7.26 ppm, 1H; 77.00 ppm, 13C). Chemical shifts are reported in parts per million (ppm), multiplicities are indicated by s (singlet), d (doublet), t (triplet), q (quartet), quint (quintet), m (multiplet) and br (broad). Coupling constants, J, are reported in Hertz with integration provided and assignments indicated. Electron Impact (EI) mass spectra were performed on a 70-VSE spectrometer with methane reagent gas. Electrospray Ionization (ESI) mass spectra were performed on a Micromass Quattro spectrometer. Data are reported in the form of (M/Z) (intensity relative to the base peak = 100 where applicable).
Analytical thin-layer chromatography was performed on Merck silica gel plates with QF-254 indicator. Visualization was accomplished with UV (254 nm), and ceric ammonium molybdate staining solution or p-anisaldehyde solution. Flash column chromatography was performed using Silacycle silica gel P60 230–400 mesh (60–63 μm) or Woelm high porosity silica gel. Preparative, radial, centrifugally accelerated thin-layer chromatography was performed on a chromatotron using glass-backed, circular TLC plates prepared with silica gel (high-purity grade, pore size 60 Å, 2–25 μm particle size, without binder, with fluorescent indicator, pore volume 0.75 cm3/g) and calcium sulfate hemihydrate (≥99%). Analytical supercritical fluid chromatography (SFC) was performed with supercritical CO2 adapter for supercritical fluid chromatography and a UV detector (220 nm or 254 nm) using Daicel Chiralcel OD, OJ, and OB, Daicel Chiralpak AD and AS, and Regis (R,R)-Whelk-O1 columns.
5.2. Representative procedure for catalytic syn-dichlorination of alkenes
To a 15 × 45 mm dram vial was added the diselenide catalyst 50 (7.42 mg, 0.010 mmol, 0.050 equiv) The vial was then transferred into the glove box and [BnNEt3]+[Cl]− (14) (131 mg, 0.6 mmol, 3.0 equiv) and [PyrF]+[BF4]− (15) (48 mg, 0.26 mmol, 1.3 equiv) were added. The vial was capped with a rubber septum and removed from the glove box. Acetonitrile (1 mL) and TMSCl (16) (51 μL, 0.4 mmol, 2.0 equiv) were added and the reaction was stirred for 10 min at room temperature. Benzyl hex-4-enoate (41 mg, 0.2 mmol) was added dropwise and the mixture was stirred at room temperature. The reaction was allowed to proceed until the olefin was completely consumed, with periodic monitoring by TLC (pentane/Et2O, 19:1; 31 Rf = 0.42, 33. Rf = 0.25). Once complete, the reaction was quenched by the slow addition of saturated sodium bicarbonate solution (1 mL). After dilution with deionized water (2 mL), the organic layer was separated and the aqueous layer was extracted with Et2O (3 × 5 mL). The combined organic layers were dried over Na2SO4 (2 g), filtered, and concentrated under reduced pressure by rotary evaporation (25 °C, 4 mm Hg). The crude mixture was re-dissolved in Et2O (3 mL) and filtered through a short silica plug. The crude product was characterized qualitatively by 1H NMR spectroscopy to determine the diastereomeric ratio of 95:5. Diagnostic peaks were: syn-33: 4.25 (qd; J 6.7, 2.9 Hz, HC(10)); 4.10 (dt; J = 10.8, 3.0 Hz, HC(9)); anti-33: 4.10 (quint; J = 6.6 Hz, HC(10)); 4.01 (ddd; J = 10.2, 6.5, 2.6 Hz, HC(9)). Purification by silica gel column chromatography (1.5 cm ø × 20 cm column) eluting with hexanes/TBME, 95:5 gave syn-33 (39 mg, 71%) as a clear, colorless oil and anti-33 (1.5 mg, 3%) as a colorless residue.
Data for syn-33:
An authentic sample of anti-33 was obtained from an identical reaction without diphenyl diselenide catalyst.
| 1H NMR: | (500 MHz, CDCI3) δ 7.41–7.31 (m, 5 H, HC(aryl)), 5.14 (d, J = 3.1 Hz, 2H, HC(7)), 4.25 (qd, J = 6.7, 2.9 Hz, 1 H, HC(5)), 4.10 (dt, J = 10.8, 3.0 Hz, 1 H, HC(4)), 2.67 (ddd, J = 16.9, 7.8, 5.6 Hz, 1 H, HC(2)), 2.57 (dt, J = 16.8, 7.7 Hz, 1 H, HC(2)), 2.30 (dtd, J = 14.4, 7.8, 2.9 Hz, 1 H, HC(3)), 2.06 (dddd, J = 14.4, 10.8, 7.7, 5.5Hz, 1 H, HC(3)), 1.59 (d, J = 6.6Hz, 3H, HC(6)). |
| 13C NMR: | (126 MHz, CDCl3) δ 172.6 (C(1)), 135.9 (C(8)), 128.8 (C(10)), 128.5 (C(11)), 128.4 (C(9)), 66.7 (C(7)), 65.2 (C(4)), 59.9 (C(5)), 31.3 (c(2)), 29.4 (C(3)), 21.0 (c(6)). |
| HRMS: | (ES+) Found: 297.0427; Calc. for C13H16O2NaCl2: 297.0425 |
| HPLC: | (S, S)-33, tR 14.9 min (26.2%), (R,R)-33, tR 18.6 min (73.7%) (Daicel Chiralpak OJ-H; hexanes/i-PrOH, 9:1; 0.5 mL/min, 210 nm). |
Data for anti-33:
| 1H NMR: | (500 MHz, CDCl3) 7.45–7.30 (m, 5 H, HC(aryl)), 5.14 (d, J = 2.6 Hz, 2 H, HC(7)), 4.10 (p, J = 6.6 Hz, 1 H, HC(5)), 4.01 (ddd, J = 10.2, 6.5, 2.6 Hz, 1 H, HC(4)), 2.68 (ddd, J = 16.8, 8.4, 5.3 Hz, 1 H, HC(2)), 2.58 (ddd, J = 16.8, 8.1, 7.4 Hz, 1 H, HC(2)), 2.43 (dddd, J = 14.5, 8.4, 7.4, 2.6 Hz, 1 H, HC(3)), 2.01 (dddd, J = 14.6, 10.2, 8.1,5.3 Hz, 1 H, HC(3)), 1.63 (d, J = 6.5 Hz, 3 H, HC(6)). |
| HRMS: | (ES+) Found: 297.0427; Calc. for C13H16O2NaCl2: 297.0425 |
| HPLC: | tRl 34.5 min (50%), tR2 37.0 min (50%) (Daicel Chiralpak OJ-H; hexanes/i-PrOH, 19:1; 0.5mL/min; 210 nm) |

Supplementary Material
Scheme 1.
Symmetry analysis of stereochemical transformations in enantioselective antarafacial-vicinal dihalogenation. Enantiodetermining steps are highlighted with bold arrows. Cat* = chiral catalyst, X = halogen.
Scheme 2.
Enantioselective dihalogenation of allylic alcohols developed by Burns et al.
Scheme 3.
Symmetry analysis of stereochemical transformations in enantioselective suprafacial-vicinal dihalogenation. Enantiodetermining step is highlighted with a bold arrow. Cat* = chiral catalyst (or ligand), X = halogen.
Scheme 4.
Net transformation for suprafacial dihalogenation and redox catalytic cycle. Y = transition metal or main group element; oxidation states are relative, not absolute.
Acknowledgements
The authors acknowledge the National Science Foundation (NSF CHE1664376) for financial support. We also thank the UIUC SCS support facilities (microanalysis, mass spectrometry, and NMR spectroscopy) for their assistance.
Footnotes
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.tet.2019.05.054.
References
- [1].Roberts I, Kimball GE, J. Am. Chem. Soc 59 (1937) 947–948. [Google Scholar]
- [2].Bartlett PD, Tarbell DS, J. Am. Chem. Soc 58 (1936) 466–474. [Google Scholar]
- [3].Olah GA, Bollinger JM, J. Am. Chem. Soc 89 (1967) 4744–4752. [DOI] [PubMed] [Google Scholar]
- [4].Brown RS, Nagorski RW, Bennet AJ, McClung RED, Aarts GHM, Klobukowski M, McDonald R, Santarsiero BD, J. Am. Chem. Soc 116 (1994) 2448–2456. [Google Scholar]
- [5].Pfenninger A, Synthesis (1986) 89–116. [Google Scholar]
- [6].Hill JG, Sharpless KB, Exon CM, Regenye R, Org. Synth 63 (1985) 66. [Google Scholar]
- [7].Katsuki T, Sharpless KB, J. Am. Chem. Soc 102 (1980) 5974–5976. [Google Scholar]
- [8].Kolb HC, VanNieuwenhze MS, Sharpless KB, Chem. Rev 94 (1994) 2483–2547. [Google Scholar]
- [9].Gonzalez J, Aurigemma C, Truesdale L, Org. Synth 79 (2002) 93. [Google Scholar]
- [10].Jacobsen EN, Marko I, Mungall WS, Schroeder G, Sharpless KB, J. Am. Chem. Soc 110 (1988) 1968–1970. [Google Scholar]
- [11].Akabori S, Sakurai S, Izumi Y, Fuji Y, Nature 178 (1956) 323–324.13358737 [Google Scholar]
- [12].Knowles WS, Angew. Chem. Int. Ed 41 (2002) 1998–2007. [Google Scholar]
- [13].Noyori R, Adv. Synth. Catal 345 (2003) 15–32. [Google Scholar]
- [14].French AN, Bissmire S, Wirth T, Chem. Soc. Rev 33 (2004) 354–362. [DOI] [PubMed] [Google Scholar]
- [15].Ranganathan S, Muraleedharan KM, Narenda KV, Jayaraman N, Tetrahedron 60 (2004) 5273–5308. [Google Scholar]
- [16].Cardillo G, Orena M, Tetrahedron 46 (1990) 3321–3408. [Google Scholar]
- [17].Gribble GW, Naturally Occurring Organohalogen Compounds - A Comprehensive Update, Springer-Verlag Wien, Vienna, 2010. [Google Scholar]
- [18].Nilewski C, Geisser RW, Carreira EM, Nature 457 (2009) 573–577. [DOI] [PubMed] [Google Scholar]
- [19].Yoshimitsu T, Fukumoto N, Nakatani R, Kojima N, Tanaka T, J. Org. Chem 75 (2010) 5425–5437. [DOI] [PubMed] [Google Scholar]
- [20].Chung W-J, Carlson JS, Bedke DK, Vanderwal CD, Angew. Chem. Int. Ed 52 (2013) 10052–10055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Bedke DK, Shibuya GM, Pereira AR, Gerwick WH, Vanderwal CD, J. Am. Chem. Soc 132 (2010) 2542–2543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Chung W-J, Vanderwal CD, Angew. Chem. Int. Ed 55 (2016) 4396–4434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Cresswell AJ, Eey ST-C, Denmark SE, Angew. Chem. Int. Ed 54 (2015) 15642–15682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Cresswell AJ, Eey ST-C, Denmark SE, Nat. Chem 7 (2015) 146–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Reversibility of haliranium ion gormation can be made instrumental to obtaining high enantioselectivity through a DyKAT approach, see Ref. 23.
- [26].Nicolaou KC, Simmons NL, Ying Y, Heretsch PM, Chen JS, J. Am. Chem. Soc 133 (2011) 8134–8137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Soltanzadeh B, Jaganathan A, Yi Y, Yi H, Staples RJ, Borhan B, J. Am. Chem. Soc 139 (2017) 2132–2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Wedek V; Van Lommel R; Daniliuc CG; De Proft F; Hennecke U Angew. Chem. Int. Ed 10.1002/anie201901777. [DOI] [PubMed] [Google Scholar]
- [29].Hu DX, Seidl FJ, Bucher C, Burns NZ, J. Am. Chem. Soc 137 (2015) 3795–3798. [DOI] [PubMed] [Google Scholar]
- [30].Landry ML, Hu DX, McKenna GM, Burns NZ, J. Am. Chem. Soc 138 (2016) 5150–5158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Uemura S, Sasaki O, Okano M, J. Chem. Soc., Chem. Commun (1971) 1064–1065. [Google Scholar]
- [32].Uemura S, Onoe A, Okano M, Bull. Chem. Soc. Jpn 47 (1974) 692–697. [Google Scholar]
- [33].Uemura S, Onoe A, Okano M, Bull. Chem. Soc. Jpn 47 (1974) 3121–3124. [Google Scholar]
- [34].San Filippo JJ, Sowinski AF, Romano LJ, J. Am. Chem. Soc 97 (1975) 1599–1600. [Google Scholar]
- [35].Walsh PJ, Kozlowski MC, Fundamentals of Asymmetric Catalysis, University Science Books, Sausalito, 2009. [Google Scholar]
- [36].Garratt DG, Schmid GH, Can. J. Chem 52 (1974) 3599–3606. [Google Scholar]
- [37].Engman L, J. Org. Chem 52 (1987) 4086–4094. [Google Scholar]
- [38].Paulmier C, Phosphorus sulfur silicon, Relat. Elements 172 (2001) 25–54. [Google Scholar]
- [39].Morella AM, Ward DA, Tetrahedron Lett. 25 (1984) 1197–1200. [Google Scholar]
- [40].Morella AM, Ward DA, Tetrahedron Lett. 26 (1985) 2899–2900. [Google Scholar]
- [41].Santi C, Santoro S, Electrophilic selenium, in: Wirth T (Ed.), Organoselenium Chemistry: Synthesis and Reactions, Wiley-VCH, Weinheim, 2012, pp. 1–51. [Google Scholar]
- [42].Singh FV, Wirth T, Selenium compounds as ligands and catalysts, in: Wirth T (Ed.), Organoselenium Chemistry: Synthesis and Reactions, Wiley-VCH, Weinheim, 2012, pp. 321–360. [Google Scholar]
- [43].Mukherjee AJ, Zade SS, Singh HB, Sunoj RB, Chem. Rev 110 (2010) 4357–4416. [DOI] [PubMed] [Google Scholar]
- [44].Denmark SE, Beutner GL, Angew. Chem. Int. Ed 47 (2008) 1560–1638. [DOI] [PubMed] [Google Scholar]
- [45].Fujita K-I, Iwaoka M, Tomoda S, Chem. Lett 23 (1994) 923–926. [Google Scholar]
- [46].Fukuzawa S-I, Takahashi K, Kato H, Yamazaki H, J. Org. Chem 62 (1997) 7711–7716. [Google Scholar]
- [47].Wirth T, Häuptli S, Leuenberger M, Tetrahedron: Asymmetry 9 (1998) 547–550. [Google Scholar]
- [48].Tiecco M, Testaferri L, Santi C, Tomassini C, Marini F, Bagnoli L, Temperini A, Tetrahedron: Asymmetry 11 (2000) 4645–4650. [Google Scholar]
- [49].Tiecco M, Testaferri L, Santi C, Tomassini C, Marini F, Bagnoli L, Temperini A, Chem. Eur. J 8 (2002) 1118–1124. [DOI] [PubMed] [Google Scholar]
- [50].Browne DM, Niyomura O, Wirth T, Org. Lett 9 (2007) 3169–3171. [DOI] [PubMed] [Google Scholar]
- [51].Déziel R, Goulet S, Grenier L, Bordeleau J, Bernier J, J. Org. Chem 58 (1993) 3619–3621. [Google Scholar]
- [52].Déziel R, Malenfant E, Bélanger G, J. Org. Chem 61 (1996) 1875–1876. [DOI] [PubMed] [Google Scholar]
- [53].Wirth T, Angew. Chem. Int. Ed 34 (1995) 1726–1728. [Google Scholar]
- [54].Wirth T, Fragale G, Chem. Eur. J 3 (1997) 1894–1902. [Google Scholar]
- [55].Omori ÁT, Assis LF, Andrade LH, Comasseto JV, Porto ALM, Tetrahedron: Asymmetry 18 (2007) 1048–1053. [Google Scholar]
- [56].Uehlin L, Fragale G, Wirth T, Chem. Eur. J 8 (2002) 1125–1133. [DOI] [PubMed] [Google Scholar]
- [57].Wirth T, Kulicke KJ, Fragale G, Helv. Chim. Acta 79 (1996) 1957–1966. [Google Scholar]
- [58].Braga AL, Silva SJN, Ludtke DS, Drekener RL, Silveira CC, Rocha JBT, Wessjohann LA, Tetrahedron Lett. 43 (2002) 7329–7331. [Google Scholar]
- [59].Kawamata Y, Hashimoto T, Maruoka K, J. Am. Chem. Soc 138 (2016) 5206–5209. [DOI] [PubMed] [Google Scholar]
- [60].Kulcsar M, Silvestru A, Silvestru C, Drake JE, Macdonald CLB, Hursthouse ME, Light ME, J. Organomet. Chem 690 (2005) 3217–3228. [Google Scholar]
- [61].The β-trifluoroacetoxy selenium(IV) dichloride intermediate 59 was prepared by the opening of trans-4-octene oxide with sodium benzeneselenolate (generated from diphenyl diselenide and sodium borohydride), trifluoroacetylation of the resulting alcohol, and oxidation at selenium with sulfuryl chloride (see Supporting Information).
- [62].Steinreiber J, Faber K, Griengl H, Chem. Eur. J 14 (2008) 8060–8072. [DOI] [PubMed] [Google Scholar]
- [63].Relative configuration was confirmed by comparison to authentic anti-chlorolactone prepared by an alternative method, see:; Denmark SE, Ryabchuk P, Burk MT, Gilbert BB J. Org. Chem 81 (2016) 10411–10423. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.