Abstract
This study aimed to determine the role of magnetic resonance imaging (MRI) in diagnosing and describing the characteristics of fistula-in-ano, and the agreement between MRI and operative findings. We conducted a retrospective study in 367 patients with fistula-in-ano who were diagnosed and had an operation at the University Medical Center between January 2016 and January 2018. MRI findings were evaluated and compared with surgical findings using the kappa coefficient (k) method. 367 patients (327 male and 40 female, mean age 39.3 ± 12.4 years). A total of 411 primary fistulas were found during surgery. There was a strong agreement between MRI and surgery for classifying primary tracts (k = 0.89) and detecting secondary tracts (k = 0.94). While the sensitivity and specificity of MRI for detecting internal openings were 99% and 85.2% respectively; these rates were 100% for abscesses. Both T2-weighted turbo spin-echo (T2W TSE) and postcontrast fat-saturated T1-weighted turbo spin-echo (FS T1W TSE) sequences showed high sensitivity (96.6% and 98.4% respectively) and specificity (92.6% and 81.5% respectively) for depicting internal openings and secondary tracts. Post-contrast FS T1W TSE sequence was very effective in detecting abscesses with an accuracy of 100%. In conclusion, MRI can be considered an accurate tool for the preoperative evaluation of fistula-in-ano, which is a major determinant of the surgical outcome. Both T2W TSE and post-contrast FS T1W TSE sequences are highly accurate in depicting the features of fistula-in-ano. If there are no contraindications, contrast administration is recommended to differentiate abscesses from active inflammation.
Subject terms: Gastrointestinal diseases, Anal diseases
Introduction
Fistula-in-ano is an inflammatory disorder of anorectal region characterised by a tract between the anal canal and the perianal skin1,2. Fistula-in-ano is usually a sequela of a poorly managed perianal abscess. This condition can also be associated with tuberculosis, cancer, and radiotherapy, etc.2,3.
Fistula-in-ano is the second most common anorectal disease after haemorrhoids2. Surgery is considered the treatment of choice aiming to avoid recurrence and preserve anal sphincter function. The risk of recurrence increases to 25% if surgeons fail to recognise and remove radically a fistula and its associated elements during corrective surgery, especially internal openings and secondary tracts1,4–6. Accordingly, a precise and comprehensive preoperative assessment of fistula tract is a pivotal diagnostic strategy and contributes significantly to the success rate of surgery.
Before the era of magnetic resonance imaging (MRI), fistulography was used to evaluate fistula-in-ano. However, this technique has a low diagnostic accuracy (~16%), and inability to visualise secondary tracts, abscesses and the sphincter complex due to its suboptimal contrast opacification7. As a result, fistulograms are not able to provide information about the relationship between fistula tracts and anal sphincters. Endoanal ultrasonography is the first imaging technique that provides the anatomical details of anal canal1. It can be used for the diagnosis and management of not only abscesses and fistula-in-ano, but also anorectal and prostate tumours. Endoanal ultrasonography is particularly helpful in identifying primary fistulous tracts and internal openings with high accuracy rates8,9. However, the limited field of view is regarded as an inherent limitation of this technique, discounting its value to evaluate secondary tracts or supralevator extensions of a primary tract.
Recently, MRI has been considered the ‘gold standard’ technique for the preoperative evaluation of fistula-in-ano. An accurate and comprehensive assessment to detect primary tracks, associated ramifications and abscesses plays a crucial role in determining surgical outcomes and minimising complications, such as faecal incontinence, as well as recurrent lesions1,10–12.
The aim of this study was to evaluate the accuracy of MRI for the diagnosis and characterisation of fistula-in-ano and the agreement between preoperative MRI and surgical findings.
Materials and Methods
This retrospective study was conducted in the University Medical Center, Ho Chi Minh City. The protocol was reviewed and approved by the Human Research Ethics Committee of the University Medical Center of Ho Chi Minh City. The study was performed in accordance with the Declaration of Helsinki. All the patients, or their guardians, provided written informed consent prior to their involvement in the study.
Subjects
The images of patients who had preoperative MRI assessment and surgery for fistula-in-ano from January 1 2016 to January 31 2018 were collected. All patients also underwent a physical examination by a proctologist to document the number and location of cutaneous openings after detailed medical history had been collected.
MRI protocol
MRI examinations were performed on either 1.5 T (Magnetom Avanto, Siemens Healthcare Limited, Germany) or 3.0 T MR (Magnetom, Siemens Healthcare Limited, Germany) scanners using a phased-array surface coil with 6 channels.
No patient preparation was required and patients were placed flat on their back. Precontrast images obtained were as follows: sagittal T2-weighted turbo spin-echo (T2W TSE), oblique coronal fat-saturated (FS) T2W TSE, oblique axial T2W TSE, oblique axial FS T2W TSE, oblique axial T1-weighted turbo spin-echo (T1W TSE) and axial FS T1W TSE. After an intravenous administration of gadolinium (0.2 ml/kg), post-contrast FS T1W TSE images were acquired in three planes (sagittal, coronal and axial). Parameters of the used sequences are shown in Table 1.
Table 1.
MRI sequences for preoperative assessment of fistula-in-ano.
| Sequence | TR (ms) | TE (ms) | FOV (cm) | Matrix | Thick (mm) | Gap (mm) | NSA | |
|---|---|---|---|---|---|---|---|---|
| T2W TSE sagittal | 4570 | 86 | 23 | 320 × 256 | 3,5 | 0,35 | 1 | |
| T2W TSE axial | 5000 | 86 | 20 | 320 × 240 | 3,5 | 0,35 | 2 | |
| FS T2W TSE axial | 5160 | 86 | 20 | 320 × 240 | 3,5 | 0,35 | 2 | |
| FS T2W TSE coronal | 3220 | 74 | 25 | 320 × 240 | 3,2 | 0,32 | 1 | |
| T1W TSE axial | 544 | 10 | 20 | 320 × 224 | 3,5 | 0,35 | 1 | |
| FS T1W TSE axial | 670 | 10 | 20 | 320 × 224 | 3,5 | 0,35 | 1 | |
| Postcontrast FS T1W TSE | axial | 670 | 10 | 20 | 320 × 224 | 3,5 | 0,35 | 2 |
| coronal | 600 | 12 | 25 | 320 × 256 | 3,2 | 0,32 | 1 | |
| sagittal | 655 | 12 | 23 | 320 × 224 | 3,5 | 0,35 | 1 | |
TSE: turbo spin echo, FS: fat-saturated, TR: repetition time, TE: echo time, FOV: field of view, NSA: number of acquisitions.
Image analysis
Images were interpreted and reported independently on picture archiving and communication system (PACS) by two radiologists who had more than 5 years of experience in analysing fistula-in-ano MRI. In the cases where there were discrepancies in interpretation between the two radiologists, a senior radiologist’s evaluation was considered the final result.
The following characteristics were assessed for each fistula-in-ano: the location of primary tracts, the presence of secondary tracts and abscess formation and the site of internal and external openings. Fistulas were classified according to the Parks and St. James’s University Hospital classifications13,14. In the image interpretation, it was assumed that a fluid collection larger than 10 mm in diameter with rim enhancement on post-contrast T1W TSE images was an abscess as per the criteria of Singh et al. and Torkzad et al.15,16.
During surgery, the characteristics of each fistula-in-ano were also carefully documented and then used as a reference standard to compare to MRI findings.
Statistical analysis
For each MRI characteristic, 2 × 2 contingency table was used to calculate sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and diagnostic accuracy. Agreement between the MRI and surgical findings was assessed using the weighted kappa coefficient (k) with a 95% confidence interval. The degree of agreement was classified as follows: poor (k < 0.2), fair (0.2 ≤ k < 0.4), moderate (0.4 ≤ k < 0.6), good (0.6 ≤ k < 0.8), or very good (k ≥ 0.8). All analyses were performed using STATA version 14 (STATA Corp., Texas, USA). A P value < 0.05 was considered significant in all analyses. Data are shown as mean values ± SDs.
Results
367 patients were eligible for the study. They were 320 males and 47 females with a ratio of 9:1. Their ages ranged from 12 to 84 years with a mean of 39.3 ± 12.4 years. Of 367 patients, 6.0% were aged over 60 years, 91.6% were aged from 20 to 60 years and only 2.4% were aged less than 20 years. 27% of patients had previous surgery for anal fistula.
External opening
The external openings were identified in 353/367 patients (96.2%) with 442 external openings. 289 (81.9%) patients had one external opening. 64 (18.1%) patients had multiple external openings: 47 (73.4%) had two, 10 (15.6%) had three, 6 (9.4%) had four and 1 (1.6%) had five external openings.
The mean distance between external opening and anal verge was 2.9 ± 2 cm. Distances of different fistula types are shown in Table 2. 95% of external openings were within 5 cm from the anal verge and 60.4% of external openings were posterior.
Table 2.
Distance between external opening and anal verge.
| ≤3 cm | 3–5 cm | >5 cm | |
|---|---|---|---|
| Intersphincteric fistula |
88 (95.7%) |
4 (4.3%) |
0 (0%) |
| Low transsphincteric fistula |
150 (71.8%) |
46 (22.0%) |
13 (6.2%) |
| High transsphincteric fistula |
5 (9.3%) |
45 (83.3%) |
4 (7.4%) |
| Supra and extrasphincteric fistula |
0 (0%) |
10 (83.3%) |
2 (16.7%) |
Primary tract
411 primary tracts were detected in 367 (%) patients during surgery. 263 (64.0%) tracts were transsphincteric, 92 (22.4%) tracts were intersphincteric according to the Parks classification (Table 3). Using this classification system, surgeons also reported 7 (1.7%) superficial fistulas and 37 (9.0%) blind tracts, which were left unclassified. Up to 333 (90.7%) patients had a single primary tract and 3 (0.8%) patients had four primary tracts.
Table 3.
Agreement between MRI and surgery in the classification of primary tract according to the Parks classification.
| Surgery | Inter | Trans | Supra | Extra | Superficial | Blind tract | No primary tract | Total |
|---|---|---|---|---|---|---|---|---|
| MRI | ||||||||
| Inter | 92 | 16 | 0 | 0 | 0 | 4 | 1 | 113 |
| Trans | 0 | 245 | 0 | 0 | 0 | 0 | 0 | 245 |
| Supra | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 3 |
| Extra | 0 | 0 | 0 | 9 | 0 | 0 | 0 | 9 |
| Superficial | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 7 |
| Blind tract | 0 | 0 | 0 | 0 | 0 | 32 | 0 | 32 |
| No primary tract | 0 | 2 | 0 | 0 | 0 | 1 | 0 | 3 |
| Total | 92 | 263 | 3 | 9 | 7 | 37 | 1 | 412 |
Inter: Intersphincteric fistula, Trans: transsphincteric fistula, Supra: suprasphincteric fistula, Extra: extrasphincteric fistula, Superficial: superficial fistula.
Data are presented as numbers of fistulas. Kappa value: 0.89 (0.85–0.94), p < 0.001.
According to the St. James’s University Hospital classification, 89 (24.1%) patients had grade 1, 24 (6.5%) patients had grade 2, 157 (42.4%) patients had grade 3, 88 (23.8%) patients had grade 4, and 12 (3.2%) patients had grade 5 fistulas (Table 4).
Table 4.
MRI grading of anal fistulas according to St. James’s University Hospital classification.
| Grade | Number |
|---|---|
| Grade 1 |
89 (24.1%) |
| Grade 2 |
24 (6.5%) |
| Grade 3 |
157 (42.4%) |
| Grade 4 |
88 (23.8%) |
| Grade 5 |
12 (3.2%) |
Low transsphincteric fistulas occurred more frequently (79.5%) than high transsphincteric fistulas (20.5%). There was a strong agreement for primary tract classification between MRI and surgical findings, with k = 0.89 (Table 3).
Internal opening
385 internal openings were detected in 367 patients. 360 (93.5%) internal openings were located at the dentate line with a mean distance to the anal verge of 2.2 ± 0.2 cm and 179 (46.5%) internal openings were located at 6 o’clock position.
MRI correctly identified 381/385 (99.0%) internal openings. In one case, MRI failed to detect the presence of internal openings, and in 3 other cases, there was a mismatch between MRI and surgical findings.
Abscess
47 abscesses were detected in 41 (11.2%) patients. Abscess locations were as follow: 16 (34%) in ischioanal, 16 (34%) in intersphincteric, 10 (21.3%) in perianal, 4 (8.5%) in supralevator and 1 (2.1%) in deep postanal space. 15/41 (36.6%) patients had horseshoe abscess development.
The mean diameter of abscesses was 2.3 ± 1.1 cm. Both sensitivity and specificity of MRI in diagnosing abscesses were 100%.
Secondary tract
132 secondary tracts were detected on MRI in 101 (27.5%) patients. 78 (77.2%) patients had one secondary tract. 23 (22.8%) patients had multiple secondary tracts: 18 (17.8%) had two, 3 (3.0%) had three, 1 (1.0%) had four and 1 (1.0%) had five secondary tracts. The agreement between MRI and surgical findings in identifying the location of secondary tracts is shown in Table 5.
Table 5.
Agreement between MRI and surgery for detecting the location of secondary tracts.
| MRI | Surgery | Perianal | Inter | Ischio | Supra | Deep postanal | Sub | No secondary tract | Total |
|---|---|---|---|---|---|---|---|---|---|
| Perianal | 3 | 3 | |||||||
| Inter | 45 | 6 | 51 | ||||||
| Ischio | 56 | 3 | 59 | ||||||
| Supra | 14 | 2 | 16 | ||||||
| Deep postanal | 3 | 3 | |||||||
| Sub | 0 | 0 | |||||||
| No secondary tract | 1 | 1 | 266 | 268 | |||||
| Total | 3 | 46 | 56 | 14 | 3 | 1 | 277 | 400 | |
Perianal: perianal space, Inter: Intersphincteric space, Ischio: ischioanal space, Supra: supralevator space, Deep postanal: deep postanal space, Sub: submucosa space.
Data are presented as numbers of secondary tracts. Kappa value: 0.94 (0.90–0.97), p < 0.001.
The sensitivity, specificity, PPV, NPV and accuracy of T2W TSE and post-contrast FS T1W TSE sequences to detect internal openings, secondary tracts and abscesses are shown in Table 6.
Table 6.
Comparison between T2W TSE and post-contrast FS T1W TSE sequences in the characterisation of fistulas.
| Sequence | Sensitivity | Specificity | PPV | NPV | Accuracy | |
|---|---|---|---|---|---|---|
| Internal opening | T2W TSE | 96.6 | 92.6 | 99.5 | 65.8 | 96.4 |
| Postcontrast FS T1W TSE | 98.4 | 81.5 | 98.7 | 78.6 | 97.3 | |
| Abscess | T2W TSE | not calculated | not calculated | not calculated | not calculated | not calculated |
| Postcontrast FS T1W TSE | 100 | 100 | 100 | 100 | 100 | |
| Secondary tract | T2W TSE | 96.7 | 99.2 | 98.3 | 98.5 | 98.5 |
| Postcontrast FS T1W TSE | 98.4 | 97.7 | 95.3 | 99.2 | 97.9 |
Discussion
Our study has demonstrated that perianal fistula occurs predominantly in adult males with the male to female ratio of 9:1 and the mean age of disease is 40 years, which are consistent with previous reports3,15,17,18.
We observed a strong association between the distance from external opening to anal verge and the type of anal fistula. The majority of intersphincteric (95.7%) and low transsphincteric (71.8%) fistulas had their external openings near the anal verge (≤ 3 cm) (Fig. 1), while external openings were located more distally to the anal verge (> 3 cm) for the most of transsphincteric (90.7%), and all the suprasphincteric (100%) and extrasphincteric (100%) fistulas. However, there were some exceptional cases, especially when external openings on the posterior midline of scrotum. In these cases, external openings were distal to the anal verge, but fistulas might still be low-transsphincteric due to their long subcutaneous course.
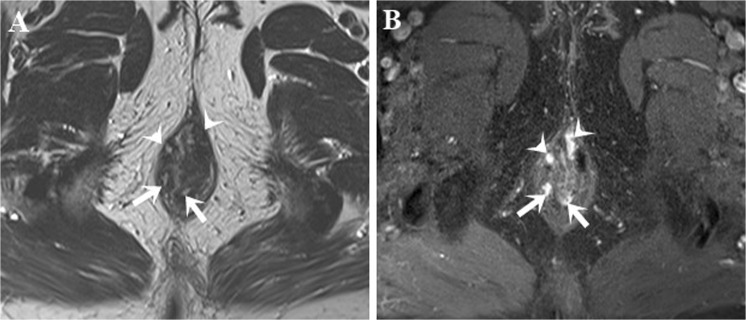
Figure 1.
Intersphincteric and transsphincteric fistulas. Axial T2W TSE (A) and post-contrast FS T1W TSE (B) images show four fistulas in one patient: two intersphincteric fistulas (arrow) and two low transsphincteric fistulas penetrating the subcutaneous part of external anal sphincter (head arrow).
MRI has been studied by many researchers and it is now recognised as an essential imaging modality in the preoperative evaluation and management of perianal fistulas. In particularly, MRI has the ability to detail accurately fistulas, associated abscesses and secondary extensions, which would otherwise be challenging for other radiology modalities. In addition, it can provide comprehensive images of the anatomical correlation between fistulas and anal sphincters, pelvic floor and levator ani muscle. This information is very important in surgical planning to remove completely infected lesions and reduce complications as well as recurrence. One previous study has demonstrated that the recurrence rate reduces substantially (~75%) following MRI navigated surgery5.
Consistent with the previous study by Buchanan et al.8, the majority of 411 primary tracts found during surgery were transsphincteric (64.0%) and intersphincteric (22.4%). These rates differ from the previous report that the most common fistulas are the intersphincteric3,13. However, this could be explained by our different study population which included only patients who had preoperative MRI prior to corrective surgery. In contrast to our study, other authors3,13 included those who had corrective surgery with or without preoperative MRI. Most of the patients in our study had a single primary tract (90.7%), which was similar to the previous study by Baik et al. (83%)19.
The strong agreement observed in our study (k = 0.89) between MRI and surgical findings in terms of the classification of primary tracts is very close to a previous study (k = 0.84)8. Similarly, Singh et al.15 and Beets-Tan et al.20 reported that MRI had a high accuracy rate (94% and 93% respectively) in detecting primary tracts. Our study, as well as previous studies, support the concept that MRI is the imaging modality of choice in the preoperative assessment of anal fistulas8,15,16. In our study, MRI was found to misclassify 10% of primary tracts compared with the reference standard. However, this misclassification rate could be attributed to a minor difference in the tract description between radiologists and surgeons. In particular, tracts crossing the most distal fibres of subcutaneous external sphincter were identified as intersphincteric fistulas on MRI, and then reassigned as transsphincteric fistulas by surgeons.
Precise evaluation of the location of internal openings is essential for a successful surgical outcome. On MRI, an internal opening is defined as the nearest point of a fistula to the anal canal, which is often seen in the intersphincteric space and it is nearly impossible to trace along a tract to its very end into the anal mucosa. Previous authors have suggested that the potential location of internal opening, in most cases, is often positioned in the most inflammatory area of intersphincteric space5,20. In our study, we used the similar strategy and noticed that the sensitivity, specificity, PPV, NPV and accuracy of MRI for depicting the internal opening were 99%, 85.2%, 99%, 85.2%, 98%, respectively. These findings are not surprising and in agreement with previous studies. Comparable high rates were reported by Beets-Tan et al.20 (sensitivity, specificity, PPV and NPV − 96%, 90%, 90% and 96% respectively) and Singh et al.15 (sensitivity, specificity and PPV − 96%, 80%, and 98% respectively). In another study18, the concordance rate for the identification of internal openings between MRI and surgery was 87%. The majority of internal openings were observed at the dentate line where most anal glands empty into the anus.
MRI has the advantages of resolution and large field of view, especially when a multichannel phased array coil is combined with a high field strength of 1.5-Tesla or 3-Tesla. In this study, we used the diameter-based criteria, proposed by Torkzad et al.16, to classify fluid collections, i.e., a roundish fluid collection ≥ 10 mm was identified as an abscess while an elongated fluid collection < 10 mm was identified as a fistula (Fig. 2). We have found that both sensitivity and specificity of MRI in differentiating abscesses from fistulas were 100%. Prior to our study, Beets-Tan et al.20 and Singh et al.15 also reported high sensitivity (96% and 87.5% respectively) and high specificity (97% and 95.2% respectively) with regards to abscess detection.
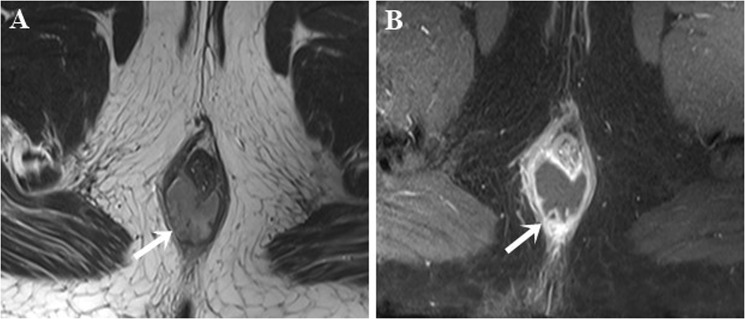
Figure 2.
Intersphincteric horseshoe abscess. Axial T2W TSE (A) and post-contrast FS T1W TSE (B) images show a horseshoe abscess (arrow) with high signal intensity on T2W TSE and rim enhancement on post-contrast FS T1W TSE.
The agreement between MRI and surgical findings in terms of identifying the location of secondary tracts was found to be good (k = 0.94) (Fig. 3), approximating to the previous study by Buchanan et al. (k = 0.88)8. There were 11 false-positive secondary tracts, identified as tiny or short fistulas on MRI. We suspect that these tracts were probably resected together with primary tracts, however, not detected by surgeons.
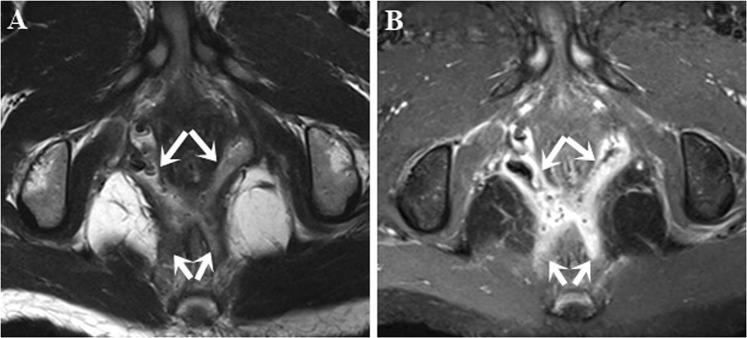
Figure 3.
Secondary tracts. Axial T2W TSE (A) and post-contrast FS T1W TSE (B) images show a high transsphincteric fistula with secondary tracts in the ischioanal space (arrows).
Both T2W TSE and post-contrast FS T1W TSE sequences demonstrated a high degree of accuracy in depicting the characteristics of fistula. In contrast to a ring-enhancing abscess, homogeneous enhancement is often seen in active inflammation on post-contrast FS T1W sequence (Fig. 4). Therefore, post-contrast FS T1W TSE sequence is suited better for distinguishing between an abscess and inflammation as they both appear hyperintense on T2W TSE sequences. However, T2W TSE sequences tended to be more optimal to position fistulas in their anatomical plane and assess their connection to anal sphincters. Torkzad et al.16 have indicated that T2W sequences must be included in the MRI protocol for the most reliable evaluation of fistula-in-ano in conjunction with post-contrast T1W sequences. Conversely, Singh et al.15 concluded that both T2W TSE and post-contrast FS T1W TSE sequences were comparable for assessing abscess and horseshoeing. However, a limitation of this study was that the study population did not include patients who had recurrent fistulas and/or previous surgery.
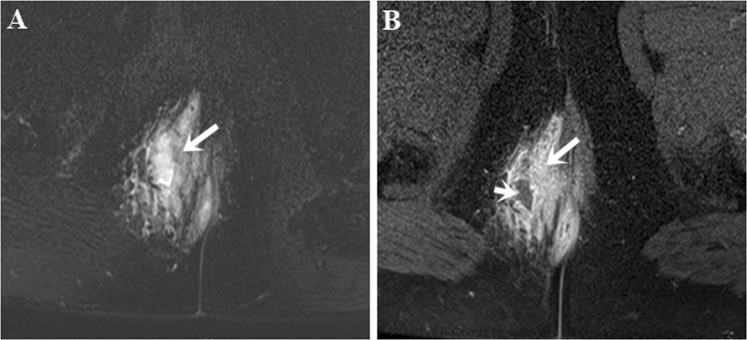
Figure 4.
Abscess and active inflammation. Axial FS T2W TSE image (A) shows a hyperintense lesion which may be an abscess and/or active inflammation. Axial post-contrast FS T1W TSE image (B) differentiates an abscess (short arrow) from adjacent active inflammation (long arrow).
In interpreting our observations, specific limitations should be acknowledged: our study was retrospective in design; and all surgeons, albeit proctologists, had no equivalent level of experience in assessing fistula-in-ano, which might compromise the quality and consistency of the reference standard.
Conclusions
Our study reinforces the importance of MRI in the characterisation and preoperative mapping of fistula-in-ano which are major contributors to the surgical prognosis. Using both T2W TSE and post-contrast FS T1W TSE sequences is a necessity for an adequate assessment of fistula-in-ano and contrast study should be included to differentiate an abscess from active inflammation.
Acknowledgements
The authors thank Dr. Thai Thanh Truc from the Department of Statistics, University Medical Centre, for assistance with the statistical analyses. This research received no specific grant from any funding agency.
Author contributions
D.V.: designed and conducted research, analysed data, wrote and reviewed paper; C.P.: conducted research and analysed data; L.N.: designed research, analysed data and reviewed paper; H.L.: conducted research and reviewed paper; T.N.: reviewed paper; H.P.: reviewed paper and has primary responsibility for the final version of manuscript. All authors read and approved the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.de Miguel Criado J, et al. MR imaging evaluation of perianal fistulas: spectrum of imaging features. Radiographics: a review publication of the Radiological Society of North America, Inc. 2012;32:175–194. doi: 10.1148/rg.321115040. [DOI] [PubMed] [Google Scholar]
- 2.Nguyen, D. H. Gastrointestinal surgical treatment (ed. Nguyen, D. H.) 191–217 (Medical Publishing House, 2007).
- 3.Abcarian, H. Anal fistula: principles and management. (ed. Abcarian, H.) 13–32 (Springer, 2014).
- 4.Do DC. The reasons of failure in surgery for anal fistula. Ho Chi Minh City Medical Journal. 2007;11:177–179. [Google Scholar]
- 5.Halligan S, Stoker J. Imaging of fistula in ano. Radiology. 2006;239:18–33. doi: 10.1148/radiol.2391041043. [DOI] [PubMed] [Google Scholar]
- 6.Lilius HG. Fistula-in-ano, an investigation of human foetal anal ducts and intramuscular glands and a clinical study of 150 patients. Acta chirurgica Scandinavica. Supplementum. 1968;383:7–88. [PubMed] [Google Scholar]
- 7.Kuijpers HC, Schulpen T. Fistulography for fistula-in-ano. Is it useful? Diseases of the colon and rectum. 1985;28:103–104. doi: 10.1007/bf02552656. [DOI] [PubMed] [Google Scholar]
- 8.Buchanan GN, et al. Clinical examination, endosonography, and MR imaging in preoperative assessment of fistula in ano: comparison with outcome-based reference standard. Radiology. 2004;233:674–681. doi: 10.1148/radiol.2333031724. [DOI] [PubMed] [Google Scholar]
- 9.Vo TD, Nguyen QTD. Endorectal ultrasonography with hydrogen peroxide in diagnosis of anal fistulas and ano-vaginal fistulas. Ho Chi Minh City Medical Journal. 2007;11:17–23. [Google Scholar]
- 10.Agha ME, Eid M, Mansy H, Matarawy K, Wally M. Preoperative MRI of perianal fistula: Is it really indispensable? Can it be deceptive? Alexandria Journal of Medicine. 2013;49:133–144. doi: 10.1016/j.ajme.2012.09.003. [DOI] [Google Scholar]
- 11.Chaudhari NH, Sinkar AD, Swoyam S. Role of magnetic resonance imaging in evaluation of perianal fistulas. International Journal of Research in Medical Sciences. 2016;4:482. doi: 10.18203/2320-6012.ijrms20160300. [DOI] [Google Scholar]
- 12.Konan A, Onur MR, Özmen MN. The contribution of preoperative MRI to the surgical management of anal fistulas. Diagnostic and Interventional Radiology. 2018;24:321. doi: 10.5152/dir.2018.18340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Parks A, Gordon PH, Hardcastle J. A classification of fistula‐in‐ano. British Journal of Surgery. 1976;63:1–12. doi: 10.1002/bjs.1800630102. [DOI] [PubMed] [Google Scholar]
- 14.Morris J, Spencer JA, Ambrose NS. MR imaging classification of perianal fistulas and its implications for patient management. Radiographics: a review publication of the Radiological Society of North America, Inc. 2000;20:623–635. doi: 10.1148/radiographics.20.3.g00mc15623. [DOI] [PubMed] [Google Scholar]
- 15.Singh K, Singh N, Thukral C, Singh KP, Bhalla V. Magnetic resonance imaging (MRI) evaluation of perianal fistulae with surgical correlation. Journal of clinical and diagnostic research: JCDR. 2014;8:RC01. doi: 10.7860/JCDR/2014/7328.4417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Torkzad MR, Ahlström H, Karlbom U. Comparison of different magnetic resonance imaging sequences for assessment of fistula-in-ano. World journal of radiology. 2014;6:203. doi: 10.4329/wjr.v6.i5.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buchanan G, et al. Effect of MRI on clinical outcome of recurrent fistula-in-ano. The Lancet. 2002;360:1661–1662. doi: 10.1016/S0140-6736(02)11605-9. [DOI] [PubMed] [Google Scholar]
- 18.Le TD, Vo TD. Initial investigation of the value of MRI in anal fistula. Ho Chi Minh City Medical Journal. 2010;14:87–91. [Google Scholar]
- 19.Baik J, Kim SH, Lee Y, Yoon J-H. Comparison of T2-weighted imaging, diffusion-weighted imaging and contrast-enhanced T1-weighted MR imaging for evaluating perianal fistulas. Clinical imaging. 2017;44:16–21. doi: 10.1016/j.clinimag.2017.03.019. [DOI] [PubMed] [Google Scholar]
- 20.Beets-Tan RG, et al. Preoperative MR imaging of anal fistulas: does it really help the surgeon? Radiology. 2001;218:75–84. doi: 10.1148/radiology.218.1.r01dc0575. [DOI] [PubMed] [Google Scholar]