Abstract
Background
Breast cancer is a leading cause of cancer-related death for women in the USA. Thus, there is an increasing need to investigate novel prognostic markers and therapeutic methods. Inflammation raises challenges in treating and preventing the spread of breast cancer. Specifically, the nuclear factor kappa b (NFκB) pathway contributes to cancer progression by stimulating proliferation and preventing apoptosis. One target gene of this pathway is PTGS2, which encodes for cyclooxygenase 2 (COX-2) and is upregulated in 40% of human breast carcinomas. COX-2 is an enzyme involved in the production of prostaglandins, which mediate inflammation. Here, we investigate the effect of Singleminded-2s (SIM2s), a transcriptional tumor suppressor that is implicated in inhibition of tumor growth and metastasis, in regulating NFκB signaling and COX-2.
Methods
For in vitro experiments, reporter luciferase assays were utilized in MCF7 cells to investigate promoter activity of NFκB and SIM2. Real-time PCR, immunoblotting, immunohistochemistry, and chromatin immunoprecipitation assays were performed in SUM159 and MCF7 cells. For in vivo experiments, MCF10DCIS.COM cells stably expressing SIM2s-FLAG or shPTGS2 were injected into SCID mice and subsequent tumors harvested for immunostaining and analysis.
Results
Our results reveal that SIM2 attenuates the activation of NFκB as measured using NFκB-luciferase reporter assay. Furthermore, immunostaining of lysates from breast cancer cells overexpressing SIM2s showed reduction in various NFκB signaling proteins, as well as pAkt, whereas knockdown of SIM2 revealed increases in NFκB signaling proteins and pAkt. Additionally, we show that NFκB signaling can act in a reciprocal manner to decrease expression of SIM2s. Likewise, suppressing NFκB translocation in DCIS.COM cells increased SIM2s expression. We also found that NFκB/p65 represses SIM2 in a dose-dependent manner, and when NFκB is suppressed, the effect on the SIM2 is negated. Additionally, our ChIP analysis confirms that NFκB/p65 binds directly to SIM2 promoter site and that the NFκB sites in the SIM2 promoter are required for NFκB-mediated suppression of SIM2s. Finally, overexpression of SIM2s decreases PTGS2 in vitro, and COX-2 staining in vivo while decreasing PTGS2 and/or COX-2 activity results in re-expression of SIM2.
Conclusion
Our findings identify a novel role for SIM2s in NFκB signaling and COX-2 expression.
Keywords: SIM2, COX-2, NFκB, Inflammation, Breast cancer, Prostaglandin E2
Background
Ductal carcinoma in situ (DCIS) is a heterogeneous disease characterized by tumor cells that are confined to the ductal system of the breast [1]. DCIS progresses to invasive ductal carcinoma (IDC) through events such as epithelial mesenchymal transition (EMT), basement membrane degradation, inflammatory signaling, and other pathways associated with a wound-healing milieu [2–4]. It is estimated that ~ 20% of mammography-detected breast cancers are DCIS [5] and ~ 65,000 cases of DCIS are diagnosed per year [6]. Provided that DCIS is removed surgically, as is the standard of care, a woman diagnosed with DCIS without recurrence is more likely to die of other causes than of breast cancer [7]. However, it is estimated that ~ 15–20% of DCIS patients develop invasive disease within a decade [8, 9]. Recently identified risk factors for DCIS recurrence include age < 40 at diagnosis, African American ethnicity, hormone receptor negativity, and HER2 positivity [7]. However, these high-risk groups only account for 20% of the DCIS patient population [9]. Therefore, identifying additional risk factors for, or markers that will predict, DCIS aggressiveness is an extremely important goal for preventing invasive cancer in DCIS patients.
There is increasing evidence that inflammation plays a key role in breast cancer progression [10]. One such specific inflammatory pathway is nuclear factor kappa b (NFκB). The NFκB signaling pathway includes five members: NFκB1 (p105/p50), NFκB2 (p100/p52), RelA (p65), RelB, and c-Rel. Dimers of the aforementioned proteins are held in the cytoplasm by inhibitor kappaB kinase (IκB) proteins, primarily IκBα. The mechanism of activation of NFκB requires phosphorylation of IκBα by inhibitor of kappaB kinase (most commonly IKKα and IKKβ), which leads to degradation of IκBα. Upon degradation of the IκBα, NFκB heterodimers, specifically the canonical heterodimer p50/p65, translocate to the nucleus and bind to promoters of target genes, leading to activation of their transcription [11, 12]. Known transcriptional targets of NFκB include mediators of inflammation, such as cytokines/chemokines and immunoreceptors, as well as proteins involved in antigen presentation, cell adhesion, stress response, apoptosis, growth factor receptor signaling, and transcription [13]. Two NFκB consensus sites are located in the promoter region of the human PTGS2 gene, which encodes for pro-inflammatory enzyme cyclooxygenase 2 (COX-2) [14]. These NFκB consensus sites contribute not only to cancer progression by preventing apoptosis but also to the activation of COX-2-mediated signaling [15]. COX-2 is the inducible form of cyclooxygenase, which is the key enzyme involved in the biosynthesis of the pro-inflammatory prostaglandins [16–21]. Importantly, COX-2 has been implicated in DCIS progression through promotion of proliferation, migration, invasion, and metastatic spread in pre-clinical models [22–24]. Additionally, expression of COX-2 is frequently observed in patients with invasive disease and is associated with DCIS recurrence. Furthermore, the therapeutic benefit of inhibiting COX-2 has been observed in the colon, esophagus, lung, bladder, breast, and prostate cancers [18, 19, 25–35]. Thus, it is logical to expect that inhibition of COX-2 signaling in breast cancer patients could enhance overall prognosis.
Previously, we have shown that Singleminded-2s (SIM2s; expressed from SIM2), a member of the bHLH/PAS family of transcription factors, is a tumor suppressor that is expressed in breast epithelial cells and downregulated in the transition from DCIS to IDC [36–39]. Specifically, using the MCF10-DCIS.COM progression model, we demonstrated that re-expression of SIM2s inhibits growth, invasive phenotypes, and progression to metastasis. Furthermore, overexpression of SIM2s in breast cancer cells promotes a more luminal-like phenotype, whereas downregulation of SIM2s leads to increased invasive potential [39]. Consistent with the role for SIM2s in cancer progression, we have also shown that the NFκB signaling pathway is negatively regulated by SIM2s in normal mammary tissues during postpartum mammary involution [40], which has been identified as a driver of tumor progression and metastasis. In this study, we demonstrate a relationship between SIM2s, the NFκB signaling pathway, and COX-2 in breast cancer cells. We suggest that re-expression of SIM2s could be mediated by inhibition of COX-2 signaling, which may serve to reduce breast cancer progression.
Methods
Cell culture
MCF7 and SUM159 cells were purchased from American Type Culture Collection (ATCC) and were maintained in accordance to their guidelines. MCF10A-DCIS.COM (DCIS.COM) cells were generously donated by Dr. Dan Medina (Baylor College of Medicine, Houston, TX, USA). Cells were plated into 6-well plates for RNA isolation experiments according the guidelines from ThermoFisher Scientific. Celecoxib experiments were performed as follows: cells were first dosed with 10 μM celecoxib for 24 h, then media was changed and treatment was performed at 20 μM celecoxib for 24 h, and then cells were harvested for analysis. DHA and PGE2 experiments on cell lines were performed as follows: cells were dosed with 50 μM DHA or 100 μM for 24 h and then harvested for analysis.
Generation of cell lines
Point mutations in the SIM2 gene were generated via long cDNA synthesis (Invitrogen). Plasmids were amplified using Subcloning Efficiency DH5a competent cells (Life Technologies). Plasmid DNA was isolated using the HiPure Plasmid Maxiprep Kit (Life Technologies) or the ZymoPURE Plasmid DNA Isolation Kit (Zymo Research). Viral transduction was performed as previously described [38]. Puromycin selection (2 μg/mL) was started the following day and maintained for a week.
Animal models
Two hundred thousand DCIS.COM cells stably expressing anti-COX-2 shRNAs (a generous gift from Kornelia Polyak and Andriy Marusyk) were orthotopically injected and tumors harvested as previously described [22, 23].
RNA isolation and real-time PCR
RNA isolation and real-time PCR were performed as previously described [39]. Primers can be found in Additional file 1: Table S2.
Immunoblotting
Cells were washed with cold PBS and lysed in high-salt lysis buffer (50 mM HEPES, 500 mM NaCl, 1.5 mM MgCl2, 1 mM ethylenediaminetetraacetic acid (EDTA), 10% glycerol, 1% Triton X-100, pH 7.5) supplemented with 1 mM Na3VO4 (Sigma) and 1 mM complete ULTRA tablets mini EDTA-free Easy pack (Roche). Protein concentration was determined using the DC Protein Assay (Bio-Rad) with bovine serum albumin as a standard. Immunoblotting and zymography were then conducted as previously described [38]. Antibodies can be found in Additional file 1: Table S1. Blots were imaged on a ChemiDoc MP (Bio-Rad) after incubating in ProSignal Pico ECL Spray (Genesee Scientific) for 3 min. Quantification was performed using ImageJ.
Immunohistochemistry
Immunohistochemistry (IHC) for COX-2 was performed as previously described [22]. Analysis for positive nuclei was performed as previously described [24]. Antibodies can be found in Additional file 1: Table S1.
Transient transfection
MCF7 or 293T cells were used for transfections for luciferase activity. One hundred nanograms (0.1 μg) of plasmid containing transcription factor was mixed with up to 1 μg (amount varies per construct) of plasmid containing promoter construct. Three microliters of Genejuice (Novagen) was used per microgram of DNA. DNA and Genejuice were mixed in Opti-MEM media (Invitrogen). Protein was harvested 2 days after transfection, using Reporter Lysis Buffer (Promega). Luciferase activity and total protein were measured as described previously [37]. Luciferase activities were normalized to total protein values and are represented as the means ± SE for three wells per condition.
Chromatin immunoprecipitation
For chromatin immunoprecipitation (ChIP) assays, 2 μg of plasmid containing repressor and 2 μg of plasmid containing the SIM2 promoter construct were transfected into 293T cells in a 10-cm plate. Chromatin was harvested 2 days after transfection.
Oncomine analysis
Analysis of SIM2 in primary breast cancer versus metastasis was performed using the Oncomine software (oncomine.org). The TCGA dataset was analyzed for SIM2 with the threshold p value set at 0.05 and the threshold fold change set at 2. Additionally, using Oncomine gene expression signatures, we evaluated the breast cancer metastasis concept, setting the odds ratio threshold at 2 and the p value at 0.01.
Statistical analysis
To address scientific rigor, all experiments in cell lines and xenografts were conducted at a minimum in biological triplicates and technical duplicates and were repeated three times. Normal distribution was confirmed before conducting unpaired t test. ANOVA analysis was performed using JMP Pro 14 to asses that means are in fact different, and then the post hoc Student’s t test was performed. Correlation analysis was performed using Prism7; Pearson’s r and a two-tailed test were performed to examine significance. Significance was considered at p < 0.05 unless otherwise noted.
Results
SIM2s downregulates NFκB signaling
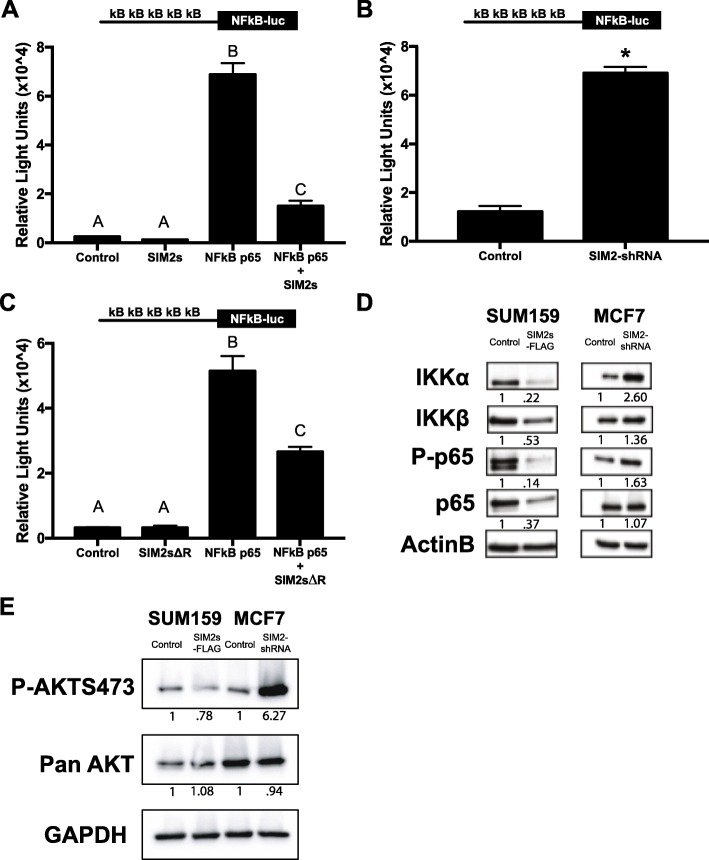
To test the hypothesis that SIM2 negatively regulates NFκB/p65-mediated transcription in breast cancer cells, we co-transfected a reporter plasmid encoding a NFκB binding site upstream of the luciferase gene (5x NFκB-luc) with the p65 subunit along with SIM2s in MCF7 cells and measured relative light units as a readout for NFκB activity. As expected, we observed that p65 strongly activated the reporter construct, but this response was blocked by co-transfection of SIM2s (Fig. 1a). Furthermore, we repeated the 5x NFκB-luc transfection in MCF7 cells with stable transduction of a SIM2 sh-RNA or control plasmid (Additional file 1: Figure S1). In the shSIM2 cells, the 5x NFκB-luc was significantly increased over the controls (Fig. 1b). To determine whether inhibition of the 5x NFκB-luc reporter by SIM2s was dependent on the transcriptional repressor activity of SIM2s, the transfection was repeated with a SIM2s expression construct missing the Pro/Ala transcriptional repression domain (SIM2sΔR). Interestingly, this construct also significantly attenuated the activation of the 5x NFκB-luc construct by NFκB/p65, demonstrating that the repression domain of SIM2s is not required for inhibition of NFκB signaling (Fig. 1c; Additional file 1: Figure S2A,B). As an alternative, we performed immunoblot analysis of key players in the NFκB signaling pathway to determine whether SIM2 modulates expression levels of key mediators of NFκB/p65 signaling in our breast cancer cell lines that could downregulate signaling in a posttranscriptional/posttranslational manner. We found that IKKα, IKKβ, phosphorylated-p65, and p65 protein expression, all of which mediate NFκB activation, are decreased in SIM2s overexpressing SUM159 cells (Fig. 1d). Similarly, we found that these same NFκB pathway protein levels are increased in SIM2s knockdown MCF7 cells (Fig. 1d). These results suggest that SIM2s may affect NFκB-mediated transcription via modulation of protein expression and/or phosphorylation of key mediators of NFκB signaling. Akt is a known activator of NFκB signaling through its ability to phosphorylate and activate IKKα/IKKβ, which leads to nuclear translocation [41, 42]. Thus, we analyzed whether activation/phosphorylation of Akt was modulated by SIM2 expression. Indeed, we observed that overexpression of SIM2s results in a modest decrease in pAkt, while SIM2s knockdown strongly restored pAkt. Together, our results suggest a SIM2s-mediated negative regulation of NFκB/p65 involves de-activation of Akt signaling.
Fig. 1.
a Luciferase activity in MCF7 cells co-transfected with 5x kB binding sites upstream of the luciferase gene (5x NFκB-luc) and NFkB p65 and/or SIM2s. (Diagram of promoter construct is shown above for reference.) b Luciferase activity in MCF7 control or MCF7 SIM2-shRNA cells with 5x NFκB-luc. c Luciferase activity in MCF7 cells co-transfected with 5x NFκB-luc and NFkB p65 and/or SIM2s with its repression domain deleted (SIM2sΔR). d SUM159 plpcx emp (control), SUM159 plpcx SIM2s-FLAG (overexpression), MCF7 psil SCR (control), and MCF7 psil SIM2-shRNA (knockdown) were analyzed by western blot for levels of IKKα, IKKβ, phospho-p65, p65, and beta actin as a loading control. e SUM159 plpcx emp (control), SUM159 plpcx SIM2s-FLAG (overexpression), MCF7 psil SCR (control), and MCF7 psil SIM2-shRNA (knockdown) were analyzed by western blot for levels of phospho-AKTs473, pan AKT, and GAPDH as loading control. ANOVA and Student’s t test were performed to test significance. a, b, c All significant at p < 0.05, *p < 0.05. Analysis was performed via ImageJ for comparison of protein expression
NFκB signaling downregulates SIM2s expression
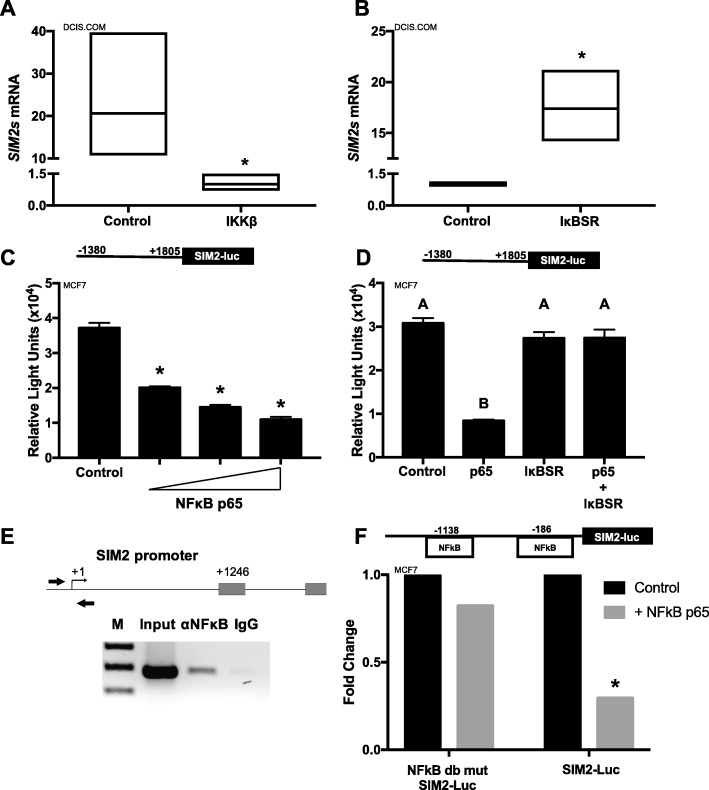
Unexpectedly, we also observed that stable overexpression of inhibitor kappa kinase beta (IKKβ), which normally functions to phosphorylate IκBα in the cytoplasm, allowing for activation of NFκB-mediated signaling, significantly decreases SIM2s gene expression in the DCIS.COM cells suggesting a reciprocal relationship between NFκB and SIM2s (Fig. 2a). Conversely, when we suppressed NFκB activation via stable transduction of the inhibitor kappaB alpha super repressor (IκB-SR), which maintains the NFκB heterodimer (p50/p65) in the cytosol, SIM2s expression was increased (Fig. 2b). To confirm that activation of NFκB downregulates SIM2 expression in breast cancer cells, we cloned a 2-kb portion of the SIM2 promoter upstream of the luciferase gene and co-transfected with increasing amounts of p65 in MCF7 cells. We observed dose-dependent repression of SIM2s promoter activity (Fig. 2c; Additional file 1: Figure S2C). Importantly, co-transfection with IκB-SR, as well as IκB-SR with NFκB p65, successfully reversed the downregulation of SIM2 promoter activity (Fig. 2d; Additional file 1: Figure S2D), suggesting that this response was not a dominant negative effect. Analysis of the SIM2 promoter identified two consensus NFκB binding sites near the transcriptional start site for SIM2. Utilizing ChIP analysis, we showed that p65 directly binds to the SIM2 promoter around the transcriptional start site (Fig. 2e). To determine whether binding of p65 to the NFκB binding sites is necessary for downregulation of SIM2s expression, we mutated the two NFκB sites in the SIM2 promoter reporter construct and performed additional co-transfection experiments with p65. The NFκB double mutant site promoter failed to inhibit SIM2 promoter activity when compared to the wild-type promoter (Fig. 2f; Additional file 1: Figure S2E), implicating a direct interaction of NFκB/p65 on the SIM2 promoter to repress SIM2s transcription. These results suggest that NFκB-mediated transcriptional repression of SIM2s may serve to reverse SIM2-mediated downregulation of NFκB signaling, allowing for its activation and promotion of transcription of known pro-inflammatory target genes such as PTGS2.
Fig. 2.
a SIM2s expression in DCIS.COM control cells and cells overexpressing IKKβ by qPCR as fold change. b SIM2s expression in DCIS.COM control cells and cells overexpressing IκB-SR by qPCR as fold change. c SIM2s promoter activity in MCF7 cells co-transfected with SIM2 promoter upstream of the luciferase gene and increasing amounts of NFκB p65 (50 ng, 100 ng, and 150 ng). d SIM2s promoter activity in MCF7 cells after co-transfection of promoter with control vector (pcDNA3), NFκB p65, and/or IκB-SR. e ChIP assay for NFκB p65 binding after transient transfection of SIM2 promoter with NFκB p65 in HEK293T cells. f SIM2s promoter activity in MCF7 cells co-transfected with SIM2s promoter upstream of the luciferase gene and 150 ng NFκB p65 compared with the SIM2s promoter activity in MCF7 cells co-transfected with NFκB double mutant SIM2s promoter upstream of the luciferase gene. ANOVA and Student’s t test were performed to test significance. a, b All significant at p < 0.05, unpaired t test: *p < 0.05
SIM2s expression downregulates COX2
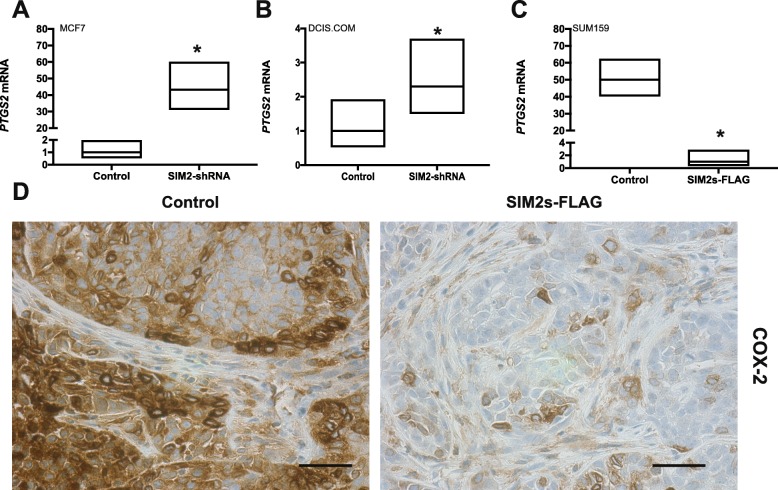
To explore the relationship between SIM2s and PTGS2 expression in breast cancer, we analyzed three different breast cancer cell lines including MCF7, DCIS.COM, and SUM159 cells. The non-invasive MCF7 cell line and the highly invasive triple-negative SUM159 cell line were utilized to examine the differential expression of SIM2s, and subsequent regulation of PTGS2, as it relates to invasion. DCIS.COM cells (also triple negative) were used for their unique ability to mimic basal-like DCIS in vivo and their ability to progress to invasive disease upon acquisition of COX-2 protein expression [22, 43]. We have previously shown that the invasive competent DCIS.COM cells have more SIM2s expression when compared with the non-invasive MCF7 [37, 38]. Confirming and extending this observation, qPCR analysis reveals the lowest PTGS2 expression in MCF7 cells, which was increased 130-fold in DCIS.COM cells and the highest in the SUM159 cells (Additional file 1: Figure S3). To determine whether reduction of SIM2s in the non-invasive cells could increase expression of PTGS2, we analyzed control and shRNA-SIM2s DCIS.COM and MCF7 cells by qPCR. Our results revealed that downregulation of SIM2s led to a significant increase in PTGS2 gene expression in both cell lines (Fig. 3a, b). Moreover, we found that overexpression of SIM2s in highly invasive SUM159 cells significantly inhibited PTGS2 expression (Fig. 3c). In previous studies, we showed that overexpression of SIM2s in DCIS.COM cells blocked invasion in vivo, whereas loss of SIM2s or overexpression of the protein product of PTGS2, COX-2, resulted in increased invasion and metastasis [22, 39]. To determine the relationship between SIM2s and COX-2 protein expression in vivo, we performed IHC analysis for COX-2 in tumors derived from control and SIM2s DCIS.COM xenografts to reveal that COX-2 levels were decreased with overexpression of SIM2s (Fig. 3d). Taken together, our results suggest that SIM2s may repress invasion in the DCIS.COM model by promoting downregulation of COX-2.
Fig. 3.
a PTGS2 expression in MCF7 control cells and cells overexpressing SIM2s by real-time qPCR as fold change. b PTGS2 expression in DCIS.COM control cells and cells with SIM2-shRNA by real-time qPCR as fold change. c PTGS2 expression in SUM159 control cells and cells overexpressing SIM2s by real-time qPCR as fold change. d Immunohistochemistry for COX-2 in DCIS.COM cells stable transduced with control vector or SIM2s-FLAG (overexpression). Unpaired t test: *p < 0.05
COX-2 downregulation restores SIM2s
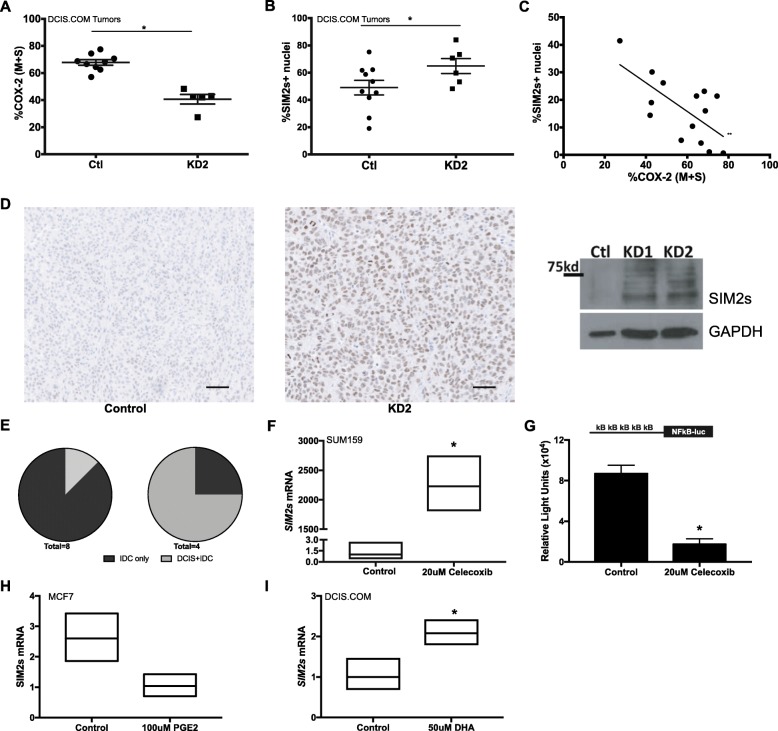
Since the invasive potential in DCIS.COM positively correlates with, and depends upon, expression and activity of COX-2 [22, 44], we tested the hypothesis that the loss of invasive phenotype observed with blocking of COX-2 expression was due, in part, to re-expression of SIM2s. Thus, we measured SIM2 protein levels by IHC analysis of tumors generated from control and shPTGS2 DCIS.COM cells, which are less invasive [22]. Our results reveal an increase in positive nuclear staining for SIM2 with PTGS2 knockdown (Fig. 4a, b; Additional file 1: Figure S4A). We also observed a significant negative correlation between expression of SIM2 and COX-2 and confirmed increased SIM2 in shPTGS2 DCIS.COM and control cells via immunoblot (Fig. 4c, d; Additional file 1: Figure S4B). Additionally, in this study, 87.5% of tumors in the control group had progressed to fully invasive disease compared with 25% in the shPTGS2 group (Fig. 4e). To determine whether COX-2 activity drives the inverse relationship between SIM2 and COX-2 and cell invasion, we treated the highly invasive SUM159 cells with a dose of the selective COX-2 inhibitor celecoxib that had previously been shown to decrease invasion of COX-2-expressing cells [22]. We observed a significant increase in SIM2s expression (Fig. 4f). Additionally, celecoxib also inhibited activation of the 5x NFκB-luc reporter MCF7 cells, suggesting that COX-2 activity can also feedback to promote NFκB-mediated suppression of SIM2s (Fig. 4g). Indeed, treatment of MCF7 cells with PGE2, the product of COX-2 activity, inhibited SIM2s gene expression (Fig. 4g). Furthermore, we extend our findings to show that docosahexaenoic (DHA), a n-3 polyunsaturated fatty acid (PUFA) that can also result in a shift to a more anti-inflammatory gene expression profile [45] and can reduce COX-2 expression [46–49], significantly increases SIM2s expression (Fig. 4i). Thus, our driving hypothesis is that reduction of inflammatory pathways via inhibition of activity and/or decreased COX-2 expression results in re-expression of SIM2s and may be one mechanism for preventing progression of DCIS to invasive or metastatic breast cancer [23]. Consistent with this hypothesis, Oncomine analysis reveals that SIM2s is in the top 5–10% of under-expressed genes in a breast cancer metastasis concept signature and in the top 10% of copy number loss genes (Table 1). Further, overall expression is significantly lower in a small number of metastasis in this dataset when compared to the expression in the primaries (Additional file 1: Figure S5), supporting our previous studies showing loss of SIM2s with DCIS progression to IDC [39, 50]. Thus, our results may be relevant for preventing metastasis in women with breast cancer.
Fig. 4.
a IHC analysis for COX-2 positive nuclei in tumors generated from control (Ctl) and shPTGS2 (KD2) DCIS.COM cells. Prism7 was utilized for statistical significance. Unpaired t test, *p value < 0.02. b IHC analysis for SIM2s positive nuclei in tumors generated from control (Ctl) and shPTGS2 (KD2) DCIS.com cells. Prism7 was utilized for statistical significance. Unpaired t test, *p value < 0.0001. c Correlation data for SIM2s and COX-2 positive nuclei in tumors generated from control and shPTGS2 DCIS.com cells. Prism7 was utilized for statistical significance. Unpaired t test, **p value < 0.01. d Images of IHC analysis for SIM2s in tumors generated from control and shPTGS2 DCIS.COM cells (left); DCIS.COM control, shPTGS2 (KD1), and shPTGS2 (KD2) were analyzed by western blot for SIM2s and GAPDH as loading control (right). e Pie Chart to show percent tumor progression DCIS+IDC or IDC only in the control group (n = 8) and shPTGS2 (n = 4). f SIM2s expression in SUM159 control cells and cells dosed with 20 μM celecoxib by qPCR as fold change. g SIM2s expression in DCIS.COM control cells and cells dosed with 50 μM DHA by qPCR as fold change. h SIM2s expression in MCF7 cells dosed with vehicle or 100 μM PGE2 for 24 h by qPCR, unpaired t test: p < 0.08. i SIM2s expression in DCIS.COM cells treated with vehicle (control) or 50 μM DHA by qPCR. Unpaired t test: *p < 0.05
Table 1 .
Oncomine analysis
| Concept type | Concept name | Size | Dataset | Expression | Top % |
|---|---|---|---|---|---|
| Oncomine gene expression signatures | Breast cancer - metastasis - top 5% under-expressed (TCGA breast) | 1019 | TCGA breast | Under-expressed | 5 |
| Oncomine gene expression signatures | Breast cancer - metastasis - top 10% copy number loss (TCGA breast 2) | 1881 | TCGA breast 2 | Copy number loss | 10 |
| Oncomine gene expression signatures | Breast cancer - metastasis - top 10% under-expressed (TCGA breast) | 2039 | TCGA breast | Under-expressed | 10 |
Discussion
Through transgenic mouse models and in vitro studies, SIM2s has been identified as a novel player in several key aspects of mammary gland development. Specifically, genetic ablation of SIM2s in mammary epithelial cells revealed that SIM2s is required for ductal morphogenesis and differentiation of luminal cells for milk production during lactation. Furthermore, mammary-specific overexpression of SIM2s resulted in a delay in post-lactational mammary gland involution through suppression of Stat3 and NFκB signaling, as well as maintenance of markers of epithelial cell differentiation normally observed only during lactation. These results suggest that SIM2s has tumor-suppressive activities in the mammary gland through maintenance of epithelial cell differentiation. Consistent with this, loss of SIM2s expression in the mammary epithelium results in EMT events, such as loss of E-cadherin and increases in matrix metalloprotease activity, and similar results are also observed in breast cancer cell lines. SIM2s is also downregulated in breast cancer patient samples, further validating its potential role in tumor suppression [39]. In this study, we demonstrate a novel role for SIM2s as a negative regulator of tumorigenesis via downregulation of the NFκB pathway, which normally results in transcriptional activation and expression of the pro-inflammatory/pro-tumorigenic enzyme COX-2, which in turn promotes DCIS invasion. Interestingly, we also identify a novel link between SIM2s and preventing signaling of the pro-tumor/pro-survival kinase Akt, which has been shown to promote tumorigenesis in part through NFκB-mediated COX-2 expression [51]. Additionally, we also show that SIM2s is directly targeted for suppression by NFκB signaling, suggesting a regulatory pro-tumorigenic feedback loop. Consistent with a role for SIM2s preventing this pro-tumorigenic cycle, loss of SIM2s also drastically increases COX-2 expression, while loss of COX-2 activity and expression results in re-expression of SIM2s and downregulation of tumor cell invasion. Thus, we have identified a reciprocal relationship between a molecule with known tumor-suppressive activities, SIM2s, and a well-established tumor promotional pathway that involves pro-survival and pro-invasive signaling mediated by Akt, NFκB, and COX-2 (Fig. 5).
Fig. 5.
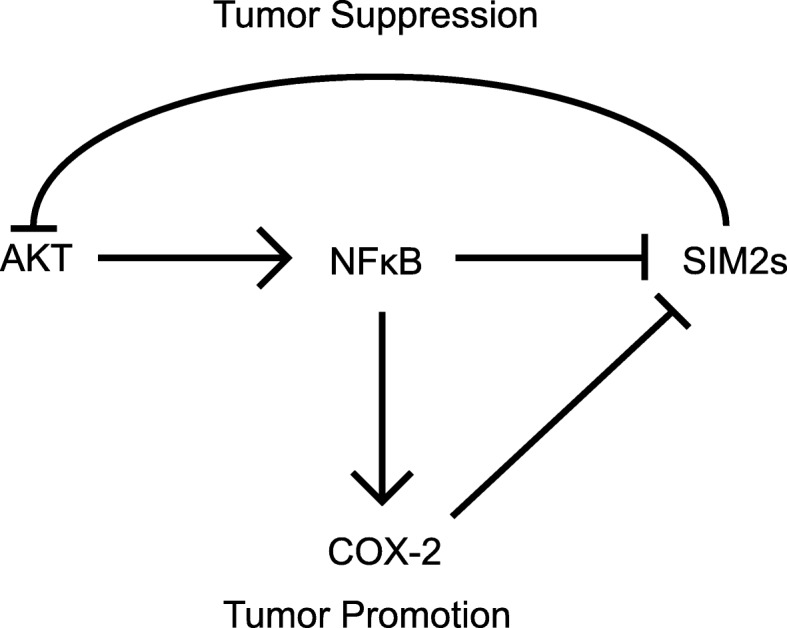
A model depicting SIM2s and NFκB cross-talk regulated to COX-2
Based on our previous results reporting a role for COX-2 in promotion of DCIS invasion [22], and results showing that SIM2 is lost in IDC compared with DCIS in patient samples [39, 50], we predict that loss of SIM2s may be important for progression of in situ lesions to invasive disease via upregulation of COX-2 expression and activity. Consistent with this hypothesis, in the DCIS.COM model, loss of SIM2s is associated with increased invasiveness and enhanced tumor aggressiveness and progression, all of which are also observed with gain of COX-2 [22, 23, 37–40, 50]. Specifically, upon loss of SIM2s in tumors, increased co-localization of keratin 5 and vimentin has been observed [39], which is indicative of mesenchymal and invasive phenotypes; furthermore, gain of COX-2 results in increased collagen deposition in the tumor microenvironment, which tumor cells utilize to invade the surrounding tissue and access the vasculature to form metastasis [22, 52, 53]. Matrix metalloproteinases (MMPs), which are associated with basement membrane degradation during mammary gland development and cancer, are also significantly increased with loss of SIM2s [54–56]. These changes likely promote an increased potential for progression of DCIS to IDC and furthermore for tumor cell escape from the primary site. Interestingly, COX-2 has been shown to promote angiogenesis [57] and to inhibit anoikis via activation of Akt [58], suggesting that this pathway can also promote dissemination and survival in circulation. Furthermore, increased COX-2 and increased proliferation are associated with subsequent recurrence of DCIS in patients [21]. Here, we show that cells with low invasive potential exhibit increased expression of COX-2 upon knockdown of SIM2s and endogenously express moderate levels of SIM2s compared with the low level of SIM2s observed in the more invasive cells [37]. Likewise, overexpression of SIM2s in invasive cells decreases COX-2 expression. Coincidently, SIM2s overexpression also significantly decreased COX-2 staining in tumor sections and all point toward a role for SIM2 in preventing metastasis. This is consistent with data from the TCGA showing downregulation of SIM2s in a metastasis gene signature. Since it is well known in the literature that COX-2 inhibition is associated with better prognosis for breast cancer patients [59, 60], further studies on strategies for re-expression of SIM2s may be beneficial in improving prognosis of breast cancer patients. Furthermore, an additional implication is that SIM2s could be utilized as a marker to identify DCIS patients that are of low risk for acquisition of COX-2 expression and progression to IDC and/or metastatic disease. However, relevance for this mechanism beyond local invasion, such as in metastatic spread, remains an unanswered question that we will address with future studies.
Conclusions
These findings support a role for SIM2s in the prevention of breast cancer progression through its ability to repress PTGS2 expression via modulating the NFκB signaling pathway. It has long been established that NFκB regulates genes involved in cell proliferation and cell survival. Specifically, blocking NFκB in tumor cells can lead to susceptibility to anti-cancer agents. However, due to the complexity of the tumor microenvironment, NFκB signaling also has been found to have anti-cancer effects in various cancer cells. Thus, it is important to investigate a mechanism, specifically in mammary tissue, in which the targeted pathways are highly involved with cell proliferation, survival, migration, and invasion. Due to elevated COX-2 expression correlating with poor prognosis, it is imperative to investigate reducing COX-2/PTGS2 expression. In the data provided here, we have demonstrated an integral role for SIM2s involvement in mediating NFκB signaling to decrease expression of COX-2/PTGS2, which could lead to an improved prognosis for breast cancer patients.
Supplementary information
Additional file 1: Figure S1: Verification of effective transduction knockdown of SIM2 via sh-RNA. Infection of MCF7 cells with the SIM2-shRNA construct results in decreased SIM2 protein levels in comparison to a nonspecific scrambled shRNA construct. Figure S2: Verification of NFkB promoter luciferase and SIM2 promoter luciferase assays in the HEK293 cell line. A. Luciferase activity in HEK293T cells co-transfected with 5x kB binding sites upstream of the luciferase gene (5x NFkB-luc) and NFkB p65 and/or SIM2s. (Diagram of promoter construct is shown above for reference.) B. Luciferase activity in HEK293T cells co-transfected with 5x NFkB-luc and NFkB p65 and/or SIM2s with its repression domain deleted (SIM2sΔR). C. SIM2 promoter activity in HEK293T cells co-transfected with SIM2 promoter upstream of the luciferase gene and increasing amounts of NFκB p65 (50ng,100ng, and 150ng). D. SIM2 promoter activity in HEK293T cells after co-transfection of promoter with control vector (pcDNA3), NFκB p65, and/or IκB-SR. E. SIM2 promoter activity in HEK293T cells co-transfected with SIM2 promoter upstream of the luciferase gene and 150ng NFκB p65 compared with the SIM2 promoter activity in HEK293T cells co-transfected with NFκB double mutant SIM2 promoter upstream of the luciferase gene. ANOVA and Student’s t-test was performed to test significance. A, B, C all significant at p<0.05, *p<0.05. Figure S3: Verification of PTGS2 in various breast cancer cell lines. PTGS2 expression in MCF7, DCIS.COM, and SUM159 parental breast cancer cell lines. ANOVA and Student’s t-test was performed to test significance. A, B, C all significant at p<0.05. Figure S4: No correlation between Tumor size and SIM2s or COX-2 gene expression. A. %SIM2s positive nuclei compared to tumor size (cm2). B. %COX-2 (M+S) compared to tumor size (cm2). Prism7 was utilized for statistical significance analysis. Two-tailed t-test was performed to test significance. Figure S5: SIM2 expression in TCGA Breast primary site and metastasis. Oncomine analysis of SIM2 expression in TCGA Breast primary site (n=529) compared to metastasis (n=3). t-Test performed to test significance, p-value=0.006. The Oncomine™ Platform (Thermo Fisher, Ann Arbor, MI) was used for analysis and visualization. For further information, refer to the terms of use. Table S1: Antibodies used in study. Table S2: Primer sequences used in study.
Acknowledgements
We gratefully acknowledge Kornelia Polyak and Andriy Marusyk for providing the shCOX-2 DCIS cell lines and Pepper Schedin for providing the shCOX2 tumors and controls from the in vivo studies. Sarah Tarullo is acknowledged for independent/blinded confirmation of the invasion analysis.
Abbreviations
- ChIP
Chromatin immunoprecipitation
- COX-2
Cyclooxygenase 2
- DCIS
Ductal carcinoma in situ
- DHA
Docosahexaenoic acid
- EMT
Epithelial mesenchymal transition
- IDC
Invasive ductal carcinoma
- IKKα
Inhibitor kappa b kinase alpha
- IKKβ
Inhibitor kappa b kinase beta
- IκB
Inhibitor kappa b
- IκBα
Inhibitor kappa b alpha
- IκB-SR
Inhibitor kappa b alpha super repressor
- NFκB
Nuclear factor kappa b
- PUFA
Polyunsaturated fatty acids
- SIM2s
Singleminded-2s
Authors’ contributions
GW performed the in vitro experiments, formal data analysis, interpretation of the data, production of figures, writing of the original draft, and reviewing and editing the final draft of the manuscript. CY and LSC were involved in animal husbandry, tumor immunostaining, and immunoblot analysis. VW was involved in designing and performing immunostaining of the xenograft studies. CMM and SWW performed treated cells, harvested proteins, harvested RNA, and assisted in the experimental design and western blots. TG was involved in animal husbandry, in vivo experiments, and ChIP analysis. YF and RC provided the polyunsaturated fatty acid resources. WP and TL oversaw all the experiments, experimental design, and interpretation of the data; provided the resources; and contributed to the writing as well as reviewing and editing of the manuscript. All authors read and approved the final manuscript.
Funding
This research is supported by an NRSA F31CA236140 (LSC/TL), the University of Colorado Department of Medicine Outstanding Early Career Scholars Program (TL), and the National Cancer Institute R21CA197896 (WP), R01HD083952 (CO-PI WP, MR), R21CA185226 (TL), and R01CA211696 (TL).
Availability of data and materials
Not applicable
Ethics approval and consent to participate
Animal studies were approved by the Institution’s laboratory care committee.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Weston W. Porter, Email: wporter@cvm.tamu.edu
Traci R. Lyons, Email: traci.lyons@ucdenver.edu
Supplementary information
Supplementary information accompanies this paper at 10.1186/s13058-019-1224-y.
References
- 1.Cowell CF, et al. Progression from ductal carcinoma in situ to invasive breast cancer: revisited. Mol Oncol. 2013;7(5):859–869. doi: 10.1016/j.molonc.2013.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schedin P, et al. Microenvironment of the involuting mammary gland mediates mammary cancer progression. J Mammary Gland Biol Neoplasia. 2007;12(1):71–82. doi: 10.1007/s10911-007-9039-3. [DOI] [PubMed] [Google Scholar]
- 3.Pensa S, Watson CJ, Poli V. Stat3 and the inflammation/acute phase response in involution and breast cancer. J Mammary Gland Biol Neoplasia. 2009;14(2):121–129. doi: 10.1007/s10911-009-9124-x. [DOI] [PubMed] [Google Scholar]
- 4.Clarkson RW, Watson CJ. Microarray analysis of the involution switch. J Mammary Gland Biol Neoplasia. 2003;8(3):309–319. doi: 10.1023/B:JOMG.0000010031.53310.92. [DOI] [PubMed] [Google Scholar]
- 5.Ernster VL, et al. Detection of ductal carcinoma in situ in women undergoing screening mammography. J Natl Cancer Inst. 2002;94(20):1546–1554. doi: 10.1093/jnci/94.20.1546. [DOI] [PubMed] [Google Scholar]
- 6.ACS. American Cancer Society: Cancer Facts and Figures 2013. American Cancer Society 2013 January 6, 2012; Available from: http://www.cancer.org/research/cancerfactsfigures/cancerfactsfigures/cancer-facts-figures-2012. Accessed Aug 2018.
- 7.Narod SA, et al. Breast cancer mortality after a diagnosis of ductal carcinoma in situ. JAMA Oncol. 2015;1(7):888–896. doi: 10.1001/jamaoncol.2015.2510. [DOI] [PubMed] [Google Scholar]
- 8.Kerlikowske K, et al. Characteristics associated with recurrence among women with ductal carcinoma in situ treated by lumpectomy. J Natl Cancer Inst. 2003;95(22):1692–1702. doi: 10.1093/jnci/djg097. [DOI] [PubMed] [Google Scholar]
- 9.Esserman L, Yau C. Rethinking the standard for ductal carcinoma in situ treatment. JAMA Oncol. 2015;1(7):881–883. doi: 10.1001/jamaoncol.2015.2607. [DOI] [PubMed] [Google Scholar]
- 10.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 11.Fitzpatrick SF, et al. An intact canonical NF-kappaB pathway is required for inflammatory gene expression in response to hypoxia. J Immunol. 2011;186(2):1091–1096. doi: 10.4049/jimmunol.1002256. [DOI] [PubMed] [Google Scholar]
- 12.Schmedtje JF, Jr, et al. Hypoxia induces cyclooxygenase-2 via the NF-kappaB p65 transcription factor in human vascular endothelial cells. J Biol Chem. 1997;272(1):601–608. doi: 10.1074/jbc.272.1.601. [DOI] [PubMed] [Google Scholar]
- 13.Pahl HL. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene. 1999;18(49):6853–6866. doi: 10.1038/sj.onc.1203239. [DOI] [PubMed] [Google Scholar]
- 14.Newton R, et al. Evidence for involvement of NF-kappaB in the transcriptional control of COX-2 gene expression by IL-1beta. Biochem Biophys Res Commun. 1997;237(1):28–32. doi: 10.1006/bbrc.1997.7064. [DOI] [PubMed] [Google Scholar]
- 15.Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008;132(3):344–362. doi: 10.1016/j.cell.2008.01.020. [DOI] [PubMed] [Google Scholar]
- 16.Hugo HJ, et al. New insights on COX-2 in chronic inflammation driving breast cancer growth and metastasis. J Mammary Gland Biol Neoplasia. 2015;20(3):109–119. doi: 10.1007/s10911-015-9333-4. [DOI] [PubMed] [Google Scholar]
- 17.Griswold DE, Adams JL. Constitutive cyclooxygenase (COX-1) and inducible cyclooxygenase (COX-2): rationale for selective inhibition and progress to date. Med Res Rev. 1996;16(2):181–206. doi: 10.1002/(SICI)1098-1128(199603)16:2<181::AID-MED3>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 18.Ristimaki A, et al. Prognostic significance of elevated cyclooxygenase-2 expression in breast cancer. Cancer Res. 2002;62(3):632–635. [PubMed] [Google Scholar]
- 19.Denkert C, et al. Elevated expression of cyclooxygenase-2 is a negative prognostic factor for disease free survival and overall survival in patients with breast carcinoma. Cancer. 2003;97(12):2978–2987. doi: 10.1002/cncr.11437. [DOI] [PubMed] [Google Scholar]
- 20.Spizzo G, et al. Correlation of COX-2 and Ep-CAM overexpression in human invasive breast cancer and its impact on survival. Br J Cancer. 2003;88(4):574–578. doi: 10.1038/sj.bjc.6600741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gauthier ML, et al. Abrogated response to cellular stress identifies DCIS associated with subsequent tumor events and defines basal-like breast tumors. Cancer Cell. 2007;12(5):479–491. doi: 10.1016/j.ccr.2007.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lyons TR, et al. Postpartum mammary gland involution drives progression of ductal carcinoma in situ through collagen and COX-2. Nat Med. 2011;17(9):1109–1115. doi: 10.1038/nm.2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lyons TR, et al. Cyclooxygenase-2-dependent lymphangiogenesis promotes nodal metastasis of postpartum breast cancer. J Clin Invest. 2014;124(9):3901–3912. doi: 10.1172/JCI73777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Black SA, et al. Semaphorin 7a exerts pleiotropic effects to promote breast tumor progression. Oncogene. 2016;35(39):5170–5178. doi: 10.1038/onc.2016.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Costa C, et al. Cyclo-oxygenase 2 expression is associated with angiogenesis and lymph node metastasis in human breast cancer. J Clin Pathol. 2002;55(6):429–434. doi: 10.1136/jcp.55.6.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Esbona K, et al. COX-2 modulates mammary tumor progression in response to collagen density. Breast Cancer Res. 2016;18(1):35. doi: 10.1186/s13058-016-0695-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Harris RE, Casto BC, Harris ZM. Cyclooxygenase-2 and the inflammogenesis of breast cancer. World J Clin Oncol. 2014;5(4):677–692. doi: 10.5306/wjco.v5.i4.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mann JR, Backlund MG, DuBois RN. Mechanisms of disease: inflammatory mediators and cancer prevention. Nat Clin Pract Oncol. 2005;2(4):202–210. doi: 10.1038/ncponc0140. [DOI] [PubMed] [Google Scholar]
- 29.Sheehan KM, et al. The relationship between cyclooxygenase-2 expression and colorectal cancer. Jama. 1999;282(13):1254–1257. doi: 10.1001/jama.282.13.1254. [DOI] [PubMed] [Google Scholar]
- 30.Visscher DW, et al. Association between cyclooxygenase-2 expression in atypical hyperplasia and risk of breast cancer. J Natl Cancer Inst. 2008;100(6):421–427. doi: 10.1093/jnci/djn036. [DOI] [PubMed] [Google Scholar]
- 31.Bourn J, Cekanova M. Cyclooxygenase inhibitors potentiate receptor tyrosine kinase therapies in bladder cancer cells in vitro. Drug Des Devel Ther. 2018;12:1727–1742. doi: 10.2147/DDDT.S158518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Buttar NS, et al. Chemoprevention of esophageal adenocarcinoma by COX-2 inhibitors in an animal model of Barrett’s esophagus. Gastroenterology. 2002;122(4):1101–1112. doi: 10.1053/gast.2002.32371. [DOI] [PubMed] [Google Scholar]
- 33.Wang W, Wang J. Toll-like receptor 4 (TLR4)/cyclooxygenase-2 (COX-2) regulates prostate cancer cell proliferation, migration, and invasion by NF-kappaB activation. Med Sci Monit. 2018;24:5588–5597. doi: 10.12659/MSM.906857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wu C, et al. IL-1beta-mediated up-regulation of WT1D via miR-144-3p and their synergistic effect with NF-kappaB/COX-2/HIF-1alpha pathway on cell proliferation in LUAD. Cell Physiol Biochem. 2018;48(6):2493–2502. doi: 10.1159/000492687. [DOI] [PubMed] [Google Scholar]
- 35.Zeng X, Yi S. Cyclooxygenase inhibitors in epithelial ovarian Cancer treatment. Int J Gynecol Cancer. 2018;28(6):1085–1089. doi: 10.1097/IGC.0000000000001269. [DOI] [PubMed] [Google Scholar]
- 36.Gustafson T L, Wellberg E, Laffin B, Schilling L, Metz R P, Zahnow C A, Porter W W. Ha-Ras transformation of MCF10A cells leads to repression of Singleminded-2s through NOTCH and C/EBPβ. Oncogene. 2009;28(12):1561–1568. doi: 10.1038/onc.2008.497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kwak HI, et al. Inhibition of breast cancer growth and invasion by single-minded 2s. Carcinogenesis. 2007;28(2):259–266. doi: 10.1093/carcin/bgl122. [DOI] [PubMed] [Google Scholar]
- 38.Laffin B, et al. Loss of Singleminded-2s in the mouse mammary gland induces an epithelial-mesenchymal transition associated with up-regulation of slug and matrix metalloprotease 2. Mol Cell Biol. 2008;28(6):1936–1946. doi: 10.1128/MCB.01701-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Scribner KC, Behbod F, Porter WW. Regulation of DCIS to invasive breast cancer progression by Singleminded-2s (SIM2s) Oncogene. 2013;32(21):2631–2639. doi: 10.1038/onc.2012.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Scribner KC, et al. Singleminded-2s (Sim2s) promotes delayed involution of the mouse mammary gland through suppression of Stat3 and NFkappaB. Mol Endocrinol. 2011;25(4):635–644. doi: 10.1210/me.2010-0423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kane LP, et al. Induction of NF-kappaB by the Akt/PKB kinase. Curr Biol. 1999;9(11):601–604. doi: 10.1016/S0960-9822(99)80265-6. [DOI] [PubMed] [Google Scholar]
- 42.Romashkova JA, Makarov SS. NF-kappaB is a target of AKT in anti-apoptotic PDGF signalling. Nature. 1999;401(6748):86–90. doi: 10.1038/43474. [DOI] [PubMed] [Google Scholar]
- 43.Miller FR, et al. MCF10DCIS.com xenograft model of human comedo ductal carcinoma in situ. J Natl Cancer Inst. 2000;92(14):1185–1186. doi: 10.1093/jnci/92.14.1185a. [DOI] [PubMed] [Google Scholar]
- 44.Hu M, et al. Role of COX-2 in epithelial-stromal cell interactions and progression of ductal carcinoma in situ of the breast. Proc Natl Acad Sci U S A. 2009;106(9):3372–3377. doi: 10.1073/pnas.0813306106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bouwens M, et al. Fish-oil supplementation induces antiinflammatory gene expression profiles in human blood mononuclear cells. Am J Clin Nutr. 2009;90(2):415–424. doi: 10.3945/ajcn.2009.27680. [DOI] [PubMed] [Google Scholar]
- 46.Calviello G, et al. n-3 PUFAs reduce VEGF expression in human colon cancer cells modulating the COX-2/PGE2 induced ERK-1 and -2 and HIF-1alpha induction pathway. Carcinogenesis. 2004;25(12):2303–2310. doi: 10.1093/carcin/bgh265. [DOI] [PubMed] [Google Scholar]
- 47.Lee SA, et al. DHA and EPA down-regulate COX-2 expression through suppression of NF-kappaB activity in LPS-treated human umbilical vein endothelial cells. Korean J Physiol Pharmacol. 2009;13(4):301–307. doi: 10.4196/kjpp.2009.13.4.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Al-Jawadi A, et al. Protective properties of n-3 fatty acids and implications in obesity-associated breast cancer. J Nutr Biochem. 2018;53:1–8. doi: 10.1016/j.jnutbio.2017.09.018. [DOI] [PubMed] [Google Scholar]
- 49.Straka S, et al. Incorporation of eicosapentaenioic and docosahexaenoic acids into breast adipose tissue of women at high risk of breast cancer: a randomized clinical trial of dietary fish and n-3 fatty acid capsules. Mol Nutr Food Res. 2015;59(9):1780–1790. doi: 10.1002/mnfr.201500161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pearson SJ, et al. ATM-dependent activation of SIM2s regulates homologous recombination and epithelial-mesenchymal transition. Oncogene. 2019;38(14):2611–2626. doi: 10.1038/s41388-018-0622-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.St-Germain ME, et al. Regulation of COX-2 protein expression by Akt in endometrial cancer cells is mediated through NF-kappaB/IkappaB pathway. Mol Cancer. 2004;3:7. doi: 10.1186/1476-4598-3-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Provenzano PP, et al. Collagen density promotes mammary tumor initiation and progression. BMC Med. 2008;6:11. doi: 10.1186/1741-7015-6-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Provenzano PP, et al. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006;4(1):38. doi: 10.1186/1741-7015-4-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liu D, et al. Association between polymorphisms in the promoter regions of matrix metalloproteinases (MMPs) and risk of cancer metastasis: a meta-analysis. PLoS One. 2012;7(2):e31251. doi: 10.1371/journal.pone.0031251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mendes O, Kim HT, Stoica G. Expression of MMP2, MMP9 and MMP3 in breast cancer brain metastasis in a rat model. Clin Exp Metastasis. 2005;22(3):237–246. doi: 10.1007/s10585-005-8115-6. [DOI] [PubMed] [Google Scholar]
- 56.Barajas-Castaneda LM, et al. Overexpression of MMP-3 and uPA with diminished PAI-1 related to metastasis in ductal breast Cancer patients attending a public hospital in Mexico City. J Immunol Res. 2016;2016:8519648. doi: 10.1155/2016/8519648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gately S, Li WW. Multiple roles of COX-2 in tumor angiogenesis: a target for antiangiogenic therapy. Semin Oncol. 2004;31(2 Suppl 7):2–11. doi: 10.1053/j.seminoncol.2004.03.040. [DOI] [PubMed] [Google Scholar]
- 58.Choi EM, et al. COX-2 inhibits anoikis by activation of the PI-3K/Akt pathway in human bladder cancer cells. Exp Mol Med. 2005;37(3):199–203. doi: 10.1038/emm.2005.27. [DOI] [PubMed] [Google Scholar]
- 59.Kwan ML, et al. NSAIDs and breast cancer recurrence in a prospective cohort study. Cancer Causes Control. 2007;18(6):613–620. doi: 10.1007/s10552-007-9003-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li J, et al. Celecoxib in breast cancer prevention and therapy. Cancer Manag Res. 2018;10:4653–4667. doi: 10.2147/CMAR.S178567. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1: Verification of effective transduction knockdown of SIM2 via sh-RNA. Infection of MCF7 cells with the SIM2-shRNA construct results in decreased SIM2 protein levels in comparison to a nonspecific scrambled shRNA construct. Figure S2: Verification of NFkB promoter luciferase and SIM2 promoter luciferase assays in the HEK293 cell line. A. Luciferase activity in HEK293T cells co-transfected with 5x kB binding sites upstream of the luciferase gene (5x NFkB-luc) and NFkB p65 and/or SIM2s. (Diagram of promoter construct is shown above for reference.) B. Luciferase activity in HEK293T cells co-transfected with 5x NFkB-luc and NFkB p65 and/or SIM2s with its repression domain deleted (SIM2sΔR). C. SIM2 promoter activity in HEK293T cells co-transfected with SIM2 promoter upstream of the luciferase gene and increasing amounts of NFκB p65 (50ng,100ng, and 150ng). D. SIM2 promoter activity in HEK293T cells after co-transfection of promoter with control vector (pcDNA3), NFκB p65, and/or IκB-SR. E. SIM2 promoter activity in HEK293T cells co-transfected with SIM2 promoter upstream of the luciferase gene and 150ng NFκB p65 compared with the SIM2 promoter activity in HEK293T cells co-transfected with NFκB double mutant SIM2 promoter upstream of the luciferase gene. ANOVA and Student’s t-test was performed to test significance. A, B, C all significant at p<0.05, *p<0.05. Figure S3: Verification of PTGS2 in various breast cancer cell lines. PTGS2 expression in MCF7, DCIS.COM, and SUM159 parental breast cancer cell lines. ANOVA and Student’s t-test was performed to test significance. A, B, C all significant at p<0.05. Figure S4: No correlation between Tumor size and SIM2s or COX-2 gene expression. A. %SIM2s positive nuclei compared to tumor size (cm2). B. %COX-2 (M+S) compared to tumor size (cm2). Prism7 was utilized for statistical significance analysis. Two-tailed t-test was performed to test significance. Figure S5: SIM2 expression in TCGA Breast primary site and metastasis. Oncomine analysis of SIM2 expression in TCGA Breast primary site (n=529) compared to metastasis (n=3). t-Test performed to test significance, p-value=0.006. The Oncomine™ Platform (Thermo Fisher, Ann Arbor, MI) was used for analysis and visualization. For further information, refer to the terms of use. Table S1: Antibodies used in study. Table S2: Primer sequences used in study.
Data Availability Statement
Not applicable