Abstract
Background
Recent advances in MRI technology makes it increasingly more competitive to CT also in the field of interventions. Multi-parametric MRI offers a significant amount of data relevant for characterization of human cerebral thrombi.
Patients and methods
Cerebral thrombi of 17 patients diagnosed with acute stroke were acquired by mechanical thrombectomy. The thrombi were subsequently scanned using a high spatial-resolution 3D T1-weighted MRI to obtain morphological characteristics of the thrombi and also by apparent diffusion coefficient (ADC) and transversal nuclear magnetic resonance (NMR) relaxation time (T2) mapping. The MRI results were analysed for possible correlations between thrombectomy procedure parameters (recanalization time and number of passes) and MR-measurable parameters (sample-mean ADC and T2, within-sample coefficient of variation of ADC and T2, and thrombus length).
Results
Both MRI mapping techniques enabled a good discrimination among thrombi regions of different water mobility and compaction. Within-sample coefficient of variation of ADC was found most sensitive for discrimination between the thrombi where thrombectomy procedure was performed in a single pass and those where is was performed in two or more passes (p = 0.03). Interestingly, negative correlation was found between the recanalization time and thrombus length (ρ = -0.22).
Conclusions
Preliminary results of presented study shows that pretreatment MRI assessment of thrombi in stroke patients could potentially ease stroke treatment planning. In this study it is shown that within-sample coefficient of variation of ADC could serve for prediction of possible complications during thrombectomy procedures.
Key words: stroke, mechanical thrombectomy, MR microscopy, ADC mapping, T2 mapping
Introduction
Rates of death attributable to cardiovascular disease have declined, yet the burden of disease remains high.1 The decline is among other factors also due to a significant improvement in treatment of large vessel occlusions in acute ischemic stroke. Early treatment of the stroke is usually based on recanalization approaches, namely, administration of thrombolytic agents2 and/or mechanical removal of thrombi.3 Occluding thrombi may stem from the heart or atherosclerotic lesions within or proximal to the affected vessel. Histological analysis alone is insufficient for determining the origin of occlusive thrombus since both thrombi etiologies, cardioembolic and arteriopathic, lead to a formation of thrombi with complex histological patterns that overlap.4 MRI was shown to have clinical potentials in thrombi localization and determination of its structure and composition.5 Therefore, MRI can represent an interesting in vivo alternative to histology.
Microscopically, thrombi are composed of platelets and red blood cells (RBCs) interspersed within the fibrin meshwork. A fibrin meshwork by itself is a permeable porous structure; however, its permeability may be strongly influenced by the presence of platelets as well as of fibrin cross-linking which could considerably thicken the meshwork.6, 7 Entrapped RBCs in thrombi8 reduce pore sizes within the meshwork and therefore also reduce the permeability of the thrombi.9 Furthermore, distribution and proportion of the entrapped blood cells in thrombi (RBCs and platelets) influence their mechanical properties as well.10 Therefore, overall susceptibility of thrombi to thrombolytic treatment (thrombolysis or mechanical thrombectomy) is strongly influenced by thrombi permeability and their mechanical properties.11, 12 An accurate assessment of the thrombi structure and composition could be helpful in treatment planning and prognosis of recanalization.13, 14
Magnetic resonance imaging (MRI) is a sensitive tool for diagnosing ischemic stroke based on detecting water mobility in different tissue compartments. The detection is enabled by diffusion-weighted imaging (DWI) followed by a calculation of the apparent diffusion constant (ADC) map.15 Another MRI mapping technique, namely the transversal nuclear magnetic resonance (NMR) relaxation time (T2) mapping, can be used for characterization of thrombi structure and composition. NMR relaxation times are sensitive to molecular reorientation or tumbling and are therefore also associated with tissue structure and composition.16 Differences in water mobility and NMR relaxation parameters between RBC-rich and platelet-rich regions were already found as an efficient discriminating factor for characterization of venous thrombi and assessment of their lytic outcome.17 Specifically, ADC and T2 mapping followed by the 2D histogram analysis confirmed large structural diversity of venous thrombi that differ by their origin and age.18
The aim of this study is fist to quantitatively characterize human cerebral thrombi using multi-parametric MRI and second to find possible correlations between thrombectomy procedure parameters (recanalization time and number of passes) and MR-measurable parameters (sample-mean ADC and T2, within-sample coefficient of variation of ADC and T2, and thrombus length). Preliminary results of ongoing research are presented.
Patients and methods
Patients selected for the study (n = 17, mean age = 72 ± 12 years, 10 males and 7 females) were diagnosed with acute ischemic stroke due to occluded middle cerebral artery (MCA; M1 segment). The study design followed standard steps of acute ischemic stroke management in our tertiary centre. Urgent clinical evaluation by a neurologist was followed by an urgent multimodal CT imaging (native CT, CT perfusion imaging and CT agiography) to confirm the diagnosis of an ischemic stroke. The patients were then treated with the standard full dose of rt-PA (0.9 mg/kg, maximum 90 mg) and later, due to still presented clinical signs of large cerebral artery occlusion, underwent mechanical thrombectomy. All procedures were performed by the same skilled interventional neuroradiologist, using the same standard mechanical recanalization procedure with the same thrombectomy device (Trevo® stent retriever, 4 x 20 mm, Stryker Neurovascular, Kalamazoo, MI). For each thrombectomy, the recanalization time was registered as the time between the first contact of the thrombectomy device with the thrombus and the successful recanalization through the occluded artery with complete removal of the thrombus. In addition, for each procedure, number of passes with the thrombectomy device was registered as well.
The protocol of the study was approved by the Institutional Review Board and the Ethical Committee of the National Ministry of Health of the Republic of Slovenia, approval No. 118/04/14. The study was performed in agreement with the informed-consent policy.
MR imaging of the retrieved thrombi was performed within 24 hours on an experimental MRI scanner consisting of a 2.35 T horizontal-bore superconducting magnet (Oxford Instruments, Abingdon, United Kingdom) NMR/MRI spectrometer (Tecmag, Houston, TX, USA) and accessories for MR microscopy (Bruker, Ettlingen, Germany). Each thrombus sample was rinsed with isotonic saline of 0.9% w/v of NaCl, pH 7.4 and closed in Teflon tubes to prevent tissue desiccation during the MR scanning. The tube with the sample was then inserted into a 10 mm micro-imaging probe and then analyzed by a multi-parametric MRI protocol consisting of the 3D T1-weighted MRI, followed by diffusion-weighted imaging (DWI) for ADC mapping19 and the multi-spin-echo imaging sequence for T2 mapping.20 The imaging parameters of the sequences are given in Table 1. During MRI scanning, the samples were held at a constant room temperature of 22°C.
Table 1.
MRI sequence parameters
| Sequence parameter | MRI sequence |
||
|---|---|---|---|
| 3D spin-echo | 3D PGSE DWI | 3D multi-echo | |
| Field of view [mm3] | 20 x 10 x 10 | 20 x 10 x 10 | 20 x 10 x 10 |
| Imaging matrix | 128 x 64 x 64 | 128 x 64 x 16 | 128 x 64 x 16 |
| Spatial resolution [μm3] | 156 x 156 x 156 | 156 x 156 x 625 | 156 x 156 x 625 |
| Echo/inter-echo time [ms] | 5 | 34 | 16 |
| Repetition time [ms] | 100 | 1035 | 1930 |
| Signal averages | 10 | 2 | 2 |
| Number of echoes | 1 | 1 | 8 |
| b-values [s/mm2] | / | 0, 260, 620, 1250 | / |
| Scan time [h] | 1 | 4.7 | 2.3 |
To obtain five independent MR-measurable parameters of the thrombi (sample-mean ADC and T2 values, their corresponding within-sample coefficients of variation CVADC and CVT2 and thrombus length) MR images were processed by using in-house written image-analysis software that was developed within the Matlab programming environment (MathWorks, Inc., Natick MA, USA). In the MR image processing software, ADC and T2 values were calculated pixelwise from the masked diffusion-weighted and multi-spin-echo images of all slices through the entire thrombi volumes by fitting the corresponding exponential functions. For each thrombus in the study its sample-mean ADC and T2 values and the corresponding within-sample coefficients of variation CVADC and CVT2 representing thrombus heterogeneity were calculated by analyzing the ADC and T2 maps in all slices across the thrombus volume. Similarly, thrombus length (Length) of the thrombi were determined from the corresponding T1-weighted images as the maximum length along the longitudinal direction.
MR-measurable parameters (ADC, T2, CVADC, CVT2, Length) of the thrombi in the study were then statistically analyzed for their correlations with the corresponding recanalization time (RT). Possible correlations were studied also between length of the thrombi and ADC, T2, CVADC, CVT2. In addition, the retrieved thrombi were also classified between a group with single pass retrieval, which was considered as thrombectomy non-problematic, and a group with multi-pass retrieval where the thrombectomy procedure was more difficult to perform. The two groups were subsequently statistically analyzed for significance of differences for different MR-measurable parameters using Student’s t-test.
Results
Figure 1 depicts T1-weighted images with the corresponding ADC and T2 maps of two representative cerebral thrombi having a different structure and morphology. Comparison between ADC and T2 maps of both samples in color-coded images shows a significant variability of ADC values across the thrombi for both samples, while their
Figure 1.
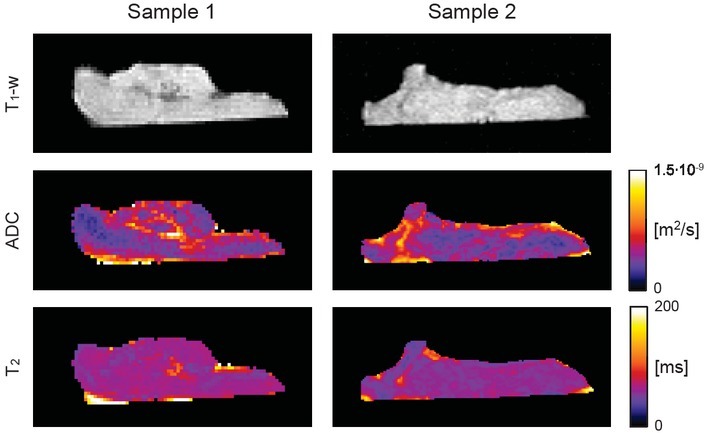
Two representative cerebral thrombi presented by central-slice T1- weighetd images, ADC and T2 maps. Low ADC and T2 values, shown in dark blue, correspond to regions of higher compaction while brighter regions are less compact regions that are more susceptible to thrombolytic therapy. From the images can also be seen that variability of ADC values across the thrombi is considerably higher than that of T2 values.
T2 maps are more uniform. In ADC maps regions with high water mobility (yellow to white regions) can be well discriminated from compacted regions (blue to violet) where water mobility is much lower. T2 maps can still discriminate between regions of different compactness/water (serum) content, however, not to the same extent.
Figure 2 shows graphs of correlation between the recanalization time and different MR-measurable thrombi parameters (ADC, T2, CVADC, CVT2, Length) as well as with number of passes with the thrombectomy device. Similarly, Figure 3 shows graphs of correlation between the thrombus length and structural and compositional parameters obtained from MR maps (ADC, T2, CVADC, CVT2). In
Figure 2.
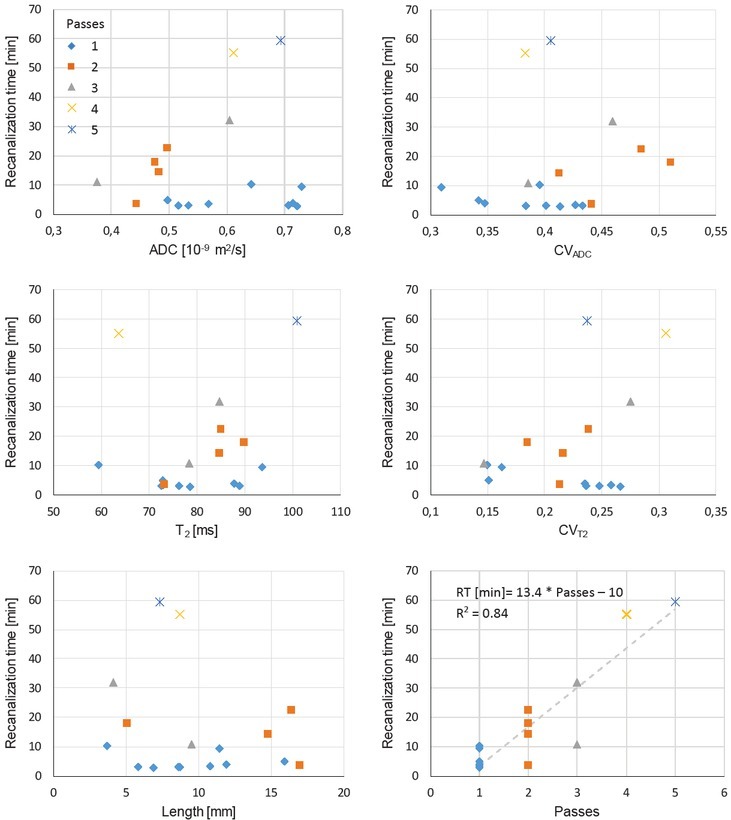
Graphs of correlation between the recanalization time (RT) of mechanical thrombectomy and different thrombi parameters: sample-mean apparent diffusion coefficient (ADC), within-sample coefficient of variation of apparent diffusion coefficient (CVADC), sample-mean spin-spin relaxation time (T2), within-sample coefficient of variation of spin-spin relaxation time (CVT2), thrombus length (Length), and number of thrombectomy procedure passes (Passes). Colour (symbol type) of experimental points indicates number of thrombectomy passes.
Figure 3.

Graphs of correlation between the thrombus length (Length) and: sample-mean apparent diffusion coefficient (ADC), within-sample coefficient of variation of apparent diffusion coefficient (CVADC), sample-mean spin-spin relaxation time (T2), and within-sample coefficient of variation of spin-spin relaxation time (CVT2). Color (symbol type) of experimental points indicates number of thrombectomy passes as defined in Figure 2.
the graphs, experimental points of different number of thrombectomy passes are colored differently and use different symbols. Correlation between different pairs of parameters given by the Pearson correlation coefficient is presented in Table 2. From the table it can be seen that correlation is the best between the recanalization time and the number of thrombectomy passes (ϱ = 0.92), which is expected. Each additional pass in average prolongs the recanalization time for 13 minutes. Other parameters do not correlate that well with the recanalization time. Among these, correlation was the best with CVT2 (ϱ = 0.38) and then with thrombus length (ϱ = -0.22), which is less than expected. Interesting is also that thrombus length correlates relatively well with ADC (ϱ = -0.41).
Table 2.
Pearson correlation coefficient for different pairs of thrombi parameters. Correlation coefficients are sorted by their decreasing absolute values
| Pair | ρ | Pair | ρ | Pair | ρ |
|---|---|---|---|---|---|
| Passes-RT | 0.92 | T2-Passes | 0.24 | CVADC-Length | -0.14 |
| ADC-CVADC | -0.43 | Length-RT | -0.22 | Length-Passes | -0.13 |
| ADC-Length | -0.41 | T2-RT | 0.21 | ADC-T2 | 0.13 |
| CVT2-RT | 0.38 | CVADC-Passes | 0.19 | CVADC-T2 | 0.12 |
| ADC-CVT2 | 0.32 | CVT2-Length | -0.16 | ADC-Passes | -0.09 |
| CVADC-CVT2 | 0.28 | ADC-RT | 0.15 | T2-Length | 0.06 |
| CVT2-Passes | 0.27 | CVADC-RT | 0.14 | T2-CVT2 | -0.01 |
ADC = apparent diffusion coefficient; CVADC = sample coefficient of variation CVADC; CVT2 = sample coefficient of variation CVT2; Length = thrombus length; ρ = Pearson correlation coefficient; RT = recanalization time
A closer inspection of correlation graphs in Figure 2 shows that distributions are significantly different for thrombi retrieved by a single pass of the thrombectomy device than for thrombi for which retrieval two or more passes were needed. In case of the single-pass thrombi group ADC and CVADC values are almost uniformly distributed over a wider range, while for the thrombi groups corresponding to multiple passes the values are more localized and are in average shifted to higher values for CVADC and to lower values for ADC. This observation is confirmed also with the t-test analysis of differences between the single-pass and multi-pass thrombi groups shown in Table 3. The test confirmed significance of difference between the groups for parameters ADC (p = 0.05) and CVADC (p = 0.03), while for other parameters (T2, CVT2, Length) the differences were not found significant.
Table 3.
Significance of differences between the single-pass (n = 9) and multi-pass (n = 8) thrombectomy groups for different parameters analysed by group-average values and by the t-test
| Thrombectomy group / t-test |
ADC_g
[10-9 m2/s] |
CVADC_g |
T2_g
[ms] |
CVT2_g | Length_g [mm] |
RT_g
[min] |
|---|---|---|---|---|---|---|
| Single-pass | 0.63 ± 0.1 | 0.38 ± 0.04 | 78 ± 10 | 0.22 ± 0.05 | 9.3 ± 3.7 | 4.8 ± 2.8 |
| Multi-pass | 0.52 ± 0.1 | 0.44 ± 0.05 | 82 ± 11 | 0.23 ± 0.05 | 10.4 ± 5.1 | 27 ± 20 |
| p-value | 0.05 | 0.03 | 0.41 | 0.64 | 0.62 | 0.006 |
ADC = apparent diffusion coefficient; ADC_g = group-average of sample-mean ADC; CVADC_g = group-average of within sample coefficient of variation CVADC; CVT2_g = group-average of within sample coefficient of variation CVT2; Length_g = group-average of thrombus length; p-value = result of the t-test analysis between single- and multi-pass thrombectomy groups; RT_g = group-average of recanalization time; T2_g = group-average of sample-mean T2
Discussion
In the study it was shown that MR-measurable parameters of a thrombus and parameters of its retrieval by mechanical thrombectomy are correlated. While no significant correlation was found between the recanalization time and MR-measurable parameters, the parameter coefficient of variance of ADC was found efficient for discrimination between thrombi retrieved in a single pass of the thrombectomy device and those retrieved by multiple passes. This finding is supported by our previous observations of thrombi susceptibility to thrombolytic treatment.17 In the study ADC was found more sensitive to structural and compositional changes in thrombi than T2 and could therefore potentially serve for prediction of thrombolytic treatment outcome. Correlation between the recanalization time and thrombus length of ϱ = -0.22, that was found in the present study, is somewhat surprising. Negative correlation between the two variables is a surprise, as one would expect that recanalization of longer thrombi should take longer21 and that several passes of the thrombectomy device are need for retrieval of these. However, in our study, the thrombi with the longest recanalization time and with most passes of the thrombectomy device are of short to medium length (from 5 to 10 mm). This result indicates a strong influence of thrombi composition and compactness on the recanalization time. The recanalization time depends also on thrombus-vessel wall interactions.22
While thrombus length can be considered as a morphological parameter, the other four MR-measurable parameters are structural-dependent. ADC is a direct measure for water mobility in different tissue compartments of thrombi. It decreases due to an increased tortuosity and decreased porosity.23 Relaxation time T2 also depends on the thrombus microstructure. It is especially sensitive to changes in surface-to-volume ratio that when increased, can lead to an increased T2 relaxation.16 Specifically, in Figure 1, low ADC/T2 values can be explained by the reduced mobility of water molecules due to an increased RBC compaction resulting in a significant reduction of the extracellular space. Another possible mechanism of the ADC/T2 reduction is a high platelet-to-fibrin content that locally contracts fibrin meshwork resulting in a pore size reduction and extracellular serum expulsion.7 From Figure 1 it can also be seen that ADC is more sensitive to structural changes in thrombi than relaxation time T2. ADC maps have higher variability than T2 maps, namely, they can better distinguish among regions of different compaction/water mobility than T2 maps.
In the study thrombi were sorted into two groups, a single- and a multi-pass group, depending on the number of thrombectomy device passes performed for the retrieval of the thrombus. Classification of thrombi between the two groups enabled statistical analysis of differences among them using t-test of which results are shown in Table 3. The differences between the groups were found statistically significant (p ≤ 0.05) for parameters CVADC and ADC, while they were not significant for CVT2, T2 and Length. Lack of significance for these three parameters could be due to too small number of samples analyzed (n = 17). With more samples included in the ongoing study the results of the study would be exacter. However, the current number of samples in the study, though low, was sufficient for demonstration of the expected trend between the MR-measurable and thrombectomy procedure parameters.
Another limitation of the study was its ex vivo design. Because of that some of the measured parameters differed from the corresponding values in vivo. This is especially true for diffusion-related parameters ADC and CVADC. Diffusion is slower at room temperature than at body temperature, while the relaxation time T2 depends much less on temperature. Therefore, it is expected that T2 values ex vivo are very similar to the corresponding in vivo values.24 A special care was also taken to preserve tissue and structural integrity of the retrieved thrombi and therefore prevent possible mechanical sources for ADC and T2 alternations. This was done during the thrombectomy procedure by minimizing possible thrombi fragmentation and after it by preventing tissue desiccation.
Ultimate goal of this research would be to perform MRI of thrombi in vivo on patients with acute ischemic stroke. Currently, MRI is too slow and does not provide sufficient resolution to compete with CT. Therefore, this application of MRI is ethically questionable. However, if thrombi would be analyzed in vivo only spectroscopically or imaged with very low resolution the scan time would be much shorter and information on the sample-mean ADC and sample-mean T2 could still be obtained. Information on the thrombus length can in principle be obtained also from high-resolution CT images. Therefore, practically all the thrombus data of this study (apart from CVADC and CVT2) could be obtained also in vivo. This information could ease decision making in stroke treatment. A proper selection of patients eligible for a treatment with mechanical recanalization is becoming extremely important since the window of thrombectomy for ischemic stroke patients was recently extended up to 24 hours.25 Therefore, more patients are potential candidates for the procedure. A possibility of the procedure duration time prediction based on prior MR imaging of thrombi could significantly influence the decision about the thrombectomy in selected patients where an extended procedure duration time could present an increased risk of reperfusion related hemorrhage. Information about the composition of the thrombus could also in the future shift the treatment decision toward mechanical recanalization without pretreatment with the thrombolysis (prediction of failed thrombolysis). Finally, the information about the thrombus composition can also affect the decision making about the thrombectomy technique (choosing between stent retriever and aspiration device). Preliminary data from our study could also have an impact on further stent retriever design improvements.
Namely, the design of the device could be tailored to different lengths of thrombi. Short thrombi may need a dedicated device, shorter in length but with much higher axial force. Alternatively, the aspiration technique may be used as a first modality of choice.
Conclusions
Preliminary results of our study shows that MRI can provide several parameters that are relevant for morphological and structural assessment of thrombi. With improvements of MRI technology, it can be expected that MRI scanning of patients with stroke could complement or even replace standard CT scanning in future. Therefore, MR-measurable parameters of thrombi, such as those presented in this study and perhaps some new ones, could in future, in combination with clinical and CT data help improving stroke treatment planning and easing treatment decisions. Results from this ongoing study my potentially influence further evolution of thrombectomy devices.
Acknowledgment
This study was financially supported by the Slovenian Research Agency grant J3-9288.
Disclosure
No potential conflicts of interest were disclosed.
References
- 1.Roger VL, Go AS, Lloyd-Jones DM, Benjamin EJ, Berry JD, Borden WB. Executive summary: heart disease and stroke statistics-2012 update: a report from the American Heart Association. Circulation. 2012;125:188–97. doi: 10.1161/CIR.0b013e31823ac046. et al. [DOI] [PubMed] [Google Scholar]
- 2.Berkhemer OA, Fransen PSS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ. A randomized trial of intraarterial treatment for acute ischemic stroke. New Engl J Med. 2015;372:11–20. doi: 10.1056/NEJMoa1411587. et al. [DOI] [PubMed] [Google Scholar]
- 3.Xavier AR, Farkas J. Catheter-based recanalization techniques for acute ischemic stroke. Neuroimag Clin N Am. 2005;15:441–53. doi: 10.1016/j.nic.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 4.Marder VJ, Chute DJ, Starkman S, Abolian AM, Kidwell C, Liebeskind D. Analysis of thrombi retrieved from cerebral arteries of patients with acute ischemic stroke. Stroke. 2006;37:2086–93. doi: 10.1161/01.STR.0000230307.03438.94. et al. [DOI] [PubMed] [Google Scholar]
- 5.Gasparian GG, Sanossian N, Shiroishi MS, Liebeskind DS. Imaging of occlusive thrombi in acute ischemic stroke. Int J Stroke. 2015;10:298–305. doi: 10.1111/ijs.12435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bajd F, Vidmar J, Fabjan A, Blinc A, Kralj E, Bizjak N. Impact of altered venous hemodynamic conditions on the formation of platelet layers in thromboemboli. Thromb Res. 2012;129:158–63. doi: 10.1016/j.thromres.2011.09.007. et al. [DOI] [PubMed] [Google Scholar]
- 7.Weisel JW. Structure of fibrin: impact on clot stability. J Thromb Haemost. 2007;5:116–24. doi: 10.1111/j.1538-7836.2007.02504.x. [DOI] [PubMed] [Google Scholar]
- 8.Lipinski B, Pretorius E, Oberholzer HM, van der Spuy WJ. Interaction of fibrin with red blood cells: the role of iron. Ultrastruct Pathol. 2012;36:79–84. doi: 10.3109/01913123.2011.627491. [DOI] [PubMed] [Google Scholar]
- 9.Fang J, Tsui PH. Evaluation of thrombolysis by using ultrasonic imaging: an in vitro study. Sci Rep. 2015;5:11669. doi: 10.1038/srep11669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gennisson JL, Lerouge S, Cloutier G. Assessment by transient elastography of the viscoelastic properties of blood during clotting. Ultrasound Med Biol. 2006;32:1529–37. doi: 10.1016/j.ultrasmedbio.2006.06.008. [DOI] [PubMed] [Google Scholar]
- 11.Varin R, Mirshahi S, Mirshahi P, Klein C, Jamshedov J, Chidiac J. Whole blood clots are more resistant to lysis than plasma clots - greater efficacy of rivaroxaban. Thromb Res. 2013;131:E100–9. doi: 10.1016/j.thromres.2012.11.029. et al. [DOI] [PubMed] [Google Scholar]
- 12.Wohner N, Sotonyi P, Machovich R, Szabo L, Tenekedjiev K, Silva MM. Lytic resistance of fibrin containing red blood cells. Arterioscl Throm Vasc Biol. 2011;31:2306–13. doi: 10.1161/ATVBAHA.111.229088. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yuki I, Kan I, Vinters HV, Kim RH, Golshan A, Vinuela FA. The impact of thromboemboli histology on the performance of a mechanical thrombectomy device. Am J Neuroradiol. 2012;33:643–8. doi: 10.3174/ajnr.A2842. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mokin M, Morr S, Natarajan SK, Lin N, Snyder KV, Hopkins LN. Thrombus density predicts successful recanalization with Solitaire stent retriever thrombectomy in acute ischemic stroke. J Neurointerv Surg. 2015;7:104–7. doi: 10.1136/neurintsurg-2013-011017. et al. [DOI] [PubMed] [Google Scholar]
- 15.van Everdingen KJ, van der Grond J, Kappelle LJ, Ramos LM, Mali WP. Diffusion-weighted magnetic resonance imaging in acute stroke. Stroke. 1998;29:1783–90. doi: 10.1161/01.str.29.9.1783. [DOI] [PubMed] [Google Scholar]
- 16.Brownstein KR, Tarr CE. Spin-lattice relaxation in a system governed by diffusion. J Magn Reson (1969) 1977;26:17–24. doi: 10.1016/0022-2364(77)90230-X. [DOI] [Google Scholar]
- 17.Vidmar J, Blinc A, Sersa I. A comparison of the ADC and T-2 mapping in an assessment of blood-clot lysability. Nmr Biomed. 2010;23:34–40. doi: 10.1002/nbm.1422. [DOI] [PubMed] [Google Scholar]
- 18.Vidmar J, Kralj E, Bajd F, Sersa I. Multiparametric MRI in characterizing venous thrombi and pulmonary thromboemboli acquired from patients with pulmonary embolism. J Magn Reson Imaging. 2015;42:354–61. doi: 10.1002/jmri.24816. [DOI] [PubMed] [Google Scholar]
- 19.Stejskal EO, Tanner JE. Spin diffusion measurements: spin echoes in the presence of a time-dependent field gradient. J Chem Phys. 1965;42:288–92. doi: 10.1063/1.1695690. [DOI] [Google Scholar]
- 20.Carr HY, Purcell EM. Effects of diffusion on free precession in nuclear magnetic resonance experiments. Phys Rev. 1954;94:630–8. doi: 10.1103/PhysRev.94.630. [DOI] [Google Scholar]
- 21.Jindal G, Miller T, Shivashankar R, Mitchell J, Stern BJ, Yarbrough K. Relationship of thrombus length to number of stent retrievals, revascularization, and outcomes in acute ischemic stroke. J Vasc Interv Radiol. 2014;25:1549–57. doi: 10.1016/j.jvir.2014.05.014. et al. [DOI] [PubMed] [Google Scholar]
- 22.Yoo AJ, Andersson T. Thrombectomy in acute ischemic stroke: challenges to procedural success. J Stroke. 2017;19:121–30. doi: 10.5853/jos.2017.00752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pisani L. Simple expression for the tortuosity of porous media. Transport Porous Med. 2011;88:193–203. doi: 10.1007/s11242-011-9734-9. [DOI] [Google Scholar]
- 24.Fan X, Macleod K, Mustafi D, Conzen SD, Markiewicz E, Zamora M. Correlation of in vivo and ex vivo ADC and T2 of in situ and invasive murine mammary cancers. Plos One. 2015;10:e0129212. doi: 10.1371/journal.pone.0129212. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K. 2018 Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2018;49:e46–e110. doi: 10.1161/STR.0000000000000158. et al. [DOI] [PubMed] [Google Scholar]


