Abstract
There are several differentiation methods for mesenchymal stem cells (MSCs) into hepatocyte-like cell. Investigators reported various hepatic differentiation protocols such as modifying culturing conditions or using various growth factors/cytokines. In this literature review, we compared different MSCs extraction and isolation protocols from Wharton’s jelly (WJ) and explored various MSCs differentiation methods.
Various protocols have been recommended for MSCs isolated from WJ, such as enzymatic, enzymatic-explant, and explant methods. In the explant method, valuable time is wasted, but the cost and biological contaminations are reduced and the number of isolated cells is high. However, other features, such as immune phenotype and multiline-age differentiation capacity, do not differ from other methods. There are also several differentiation methods for hepatocyte-like cell including the induction of MSC by cytokines and growth factors, and the differentiation of MSC in 2- and 3-dimensional matrix (2D and 3D). Among several cytokines, hepatocyte growth factor (HGF) and fibroblast growth factor (FGF) are essential. In the early stage of the differentiation, 2D culture is useful, and in the development stage, 3D culture system with HGF and FGF cytokines are more effective in the process of differentiation. Some studies have used 3D culture system in biocompatible scaffolds, such as alginate, collagen, gelatin, and peptide-Gly-Leu-amide (PGLA).
In conclusion, Wharton’s jelly-Mesenchymal stem cells (WJ-MSCs) can be considered as an appropriate source for hepatocyte differentiation. Moreover, we introduced the explant method as the most effective protocol. This review attempted to highlight factors in hepatocyte differentiation, but the most effective protocol is not still unknown.
Keywords: Cell differentiation , Mesenchymal stromal cells , Umbilical cord , Wharton jelly , hepatocytes
What’s Known
Wharton’s jelly-Mesenchymal stem cells (WJ-MSCs) might be a suitable candidate for stem cell therapy. They have high proliferation rates, wide multipotency, and hypo-immunogenicity. There are several differentiation methods into hepatocyte-like cells, such as induction by cytokines and growth factors, and differentiation of mesenchymal stem cells (MSCs) in 2- and 3-dimensional matrix.
What’s New
In this review, we introduced the explant method as the most effective isolation protocol for Wharton‘s Jelly (WJ) as well as summarizing and discussing current hepatocyte differentiation protocol; however, the best and most effective protocol is still unknown.
Introduction
Chronic liver failure, such as cirrhosis, can be stimulated by viral hepatitis, metabolic diseases, alcohol, drugs, and autoimmune processes. Liver transplantation is the most popular procedure for chronic liver disease.1 However, there are some problems such as lack of donor, surgical complications, immunological rejection, and high medical costs. Consequently, stem cell therapies can be a new approach to treat end-stage liver diseases.2
Several studies have tried to find the best stem cell source for hepatocyte transplantation.3,4 Stem cells are isolated from various sources such as preimplantation embryonic, fetuses, and adult organs. These sources have advantages and disadvantages. Human embryonic stem cells (h-ESCs) are pluripotent, but several problems such as insufficient cell numbers, possible teratoma formation or immune rejection after transplantation can hinder their clinical applications.
MSCs can be extracted from several different sources and are plastic-adherent cells that have the capacity to self-renew. Cells defined by the international society for cellular therapy have a specific surface phenotype and can be differentiated into various lineages including bone, cartilage, and adipose.5,6
MSCs extracted from the bone marrow and adipose tissue have limitations such as being invasive and having a painful procedure while the high degree of viral infection associated with MSCs removed from the bone marrow may lead to a restriction in their usage.7 Also, the obtained MSCs from older individuals is hard, since marrow cavity is filled with yellow fat due to aging process.8
Umbilical cord (UC) mesenchymal stem cells (MSCs) with similar immune phenotype and multilineage differentiation have higher expansion potential in comparison with bone marrow MSCs (BM-MSCs) and adipose-derived MSCs (ADMSCs).9 Umbilical cords are considered to be a medical waste; hence their clinical application in research and cell therapy is of no ethical concern. Furthermore, cells isolated from UCs proliferate rapidly in culture and they have the potential for differentiation.10 UC-MSCs are capable of suppressing the immune response in vitro, which is similar to BM-MSC properties. Many researchers have investigated MSCs extracted from human UCs tissue, which is an acceptable source.11-16 In some studies, MSCs were isolated from different parts of the umbilical cord, such as WJ matrix, perivascular regions,17 and sub-amnion membrane with various protocols.18-21
It is unclear whether human WJ-MSCs can behave as h-ESCs, human MSCs, or both. They have high proliferation rates, wide multipotency, and hypo-immunogenicity, which do not induce teratomas. Human WJ-MSCs (hWJ-MSCs) express low levels of pluripotent embryonic stem cell markers including POUF1, NANOG, SOX2, and LIN28. That explains why hWJ-MSCs do not produce teratomas. Several cytokines such as IL12A, associated with the induction of apoptosis, were significantly upregulated in hWJ-MSCs. These properties propose that hWJ-MSC is an important applicable stem cell source for cell therapy in allogeneic settings without immune rejection.22
MSCs can be isolated from WJ by different methods, such as enzymatic, enzymatic-explant, and explants.12,15,16
Extraction of MSCs is the first step of working with these cells. Recently, there has been extensive research on the transplantation of differentiated cells into damaged tissues for regenerative medicine.23
There are several differentiation methods for MSC into hepatocyte-like cell including induction of MSC by cytokines and growth factors, and differentiation of MSC in 2- and 3-dimensional matrix (2D and 3D); however, the advantages are still insufficient in clinical applications. Researchers are investigating to improve the methods of hepatic differentiation of MSCs by modifying culture conditions or adding various growth factors/cytokines. We reviewed the literature and compared different extraction and isolation protocols to extract MSCs from WJ. We also explored various MSCs differentiation methods into hepatocyte-like cells and debated the achievements and prospective development.
Human Umbilical Cord
Human UC matrix originates from extraembryonic mesoderm. The connective UC tissue is within two arteries and a vein in the amniotic wall.24 Five weeks after fertilization, the UC begins to form and gains an average of 50 cm. The different cell populations have been identified in WJ, the sub-endothelium of the umbilical vein, and UC blood. WJ is a matrix of mucous connective tissue composed of fibroblast-like stromal cells, collagen fibers, and proteoglycans, situated between the sub-amnion and the perivascular region. The perivascular region surrounding the umbilical vessels is a source of mesenchymal progenitor cells. The third source of MSCs can be found in the sub-endothelium of the umbilical vein. In addition, UC blood is a rich source of hematopoietic stem/progenitor cells but does not contain mesenchymal progenitor cells.25,26 Furthermore, MCSs harvested from UC blood have low efficiency that exhibit a variety of morphologies and differentiations.27 By investigating the umbilical cord, WJ and the sub-endothelium stem cell revealed that typical features of human MSCs could be differentiated into chondrocytes and hepatocytes.
It should be noted that the number of isolated cells extracted from UC in the vaginal delivery were more than caesarian section births.28 The efficiency and number of cells isolated from each part of umbilical cord are the same, but there is still controversy between researches.13,28,29
MSCs Isolation from WJ
MSCs Isolation by Enzymatic Method
The Culture of Total Length of UC
Some studies have described how to obtain MSCs from UC, by using the total length of the cord, which leads to an increase in stem cell yield without time-consuming producers. By using an enzymatic procedure, MSC isolation from the three recognized compartments, the perivascular, and the intra-vascular region within the WJ region, is possible. By different enzymatic isolation methods, higher cell populations can be harvested with less time. Approximately 0.95×106 cells can be isolated from the UC by using an enzymatic procedure.8,30 In this procedure, when UC is transferred into the laminar hood in complete sterile condition, the entire length of UC is used. The external surface is examined and engraved with a sterile scalpel. Next, the vessels are removed and the pieces of tissue are incubated in enzymatic cocktail containing enzymes such as hyaluronidase, collagenase, and trypsin for 45 minutes to 2 hours at 37 °C. Then, the digested suspension are collected by gravity in the 50 mL conical tube, diluted and centrifuged. Finally, the pellets are cultured.12
Culture of UC Parts
Conventionally, 5-7.5 cm-length pieces of UC from the mid-region are transferred into the sterile medium and cut into smaller 1-1.5 cm-length pieces.13
In some procedures, to isolate WJ-MSCs from these small pieces without removing the umbilical blood vessels, the matrix containing the WJ is placed face down in culture flasks or Petri dishes. The flasks containing an enzymatic solution of collagenase and hyaluronidase, in Dulbecco’s Modified Eagle’s Medium (DMEM), are incubated at 37 °C in a 5% CO2 in air atmosphere for 45 min to 2h. This allows WJ loosening and separation from the UC without complete digestion. After the incubation period, the cord pieces are transferred to a new Petri dish or culture flask containing fresh DMEM to remove any remaining enzymes. A pair of forceps is used to gently scrape off the gelatinous WJ from the inner compartments into the fresh medium. The suspension is passed through a syringe fitted with an 18G needle, centrifuged. Finally, cell pellets are re-suspended in culture media. Studies have revealed that approximately 3-4×107 fresh live WJ-MSCs could be obtained from each piece.31
Isolation of MSCs with Explants Method
In this procedure, UC is cut into smaller 2-4 cm length pieces. The obtained small pieces of the cord are washed gently, and, then, at the external surface of each piece, with a horizontal section, the outer sheath of UC and its inner surface containing the WJ are extracted into dishes.15
In the next step, vein and arteries are exposed and removed away from the inner matrix. Then, the inner matrix of WJ is divided into 3-5 mm pieces with a scalpel and washed again with a sterile dissection solution. For explant outgrowth, 6-9 pieces are transferred onto the culture dishes and left undisturbed until the jelly is attached to the dishes. Then, a culture medium such as DMEM is added. The culture dishes are left undisturbed for 3-4 days and maintained at 37 °C in a humidified atmosphere containing 5% CO2. After the attachment of Wharton jelly, the medium of dishes is changed 2-3 days. Approximately, after 7-10 days, the cells with an MSCs phenotype can be isolated.14,32
Mori and others used a stainless steel mesh to prevent tissue explant pieces from floating; therefore, in this method, the cells can be isolated very easily with high proliferation rate in comparison with the conventional methods.33
MSCs Isolation with a Mixed Enzymatic-Explant Method
In the mixed procedure, after blood removal, vessels and the outer sheath of UC, the matrix is allowed to come into direct contact with the enzymatic solution to extract WJ. Then, WJ is cut into smaller pieces. In the next step, WJ pieces are placed in a culture medium with an enzymatic cocktail such as Collagenase or Hyaluronidase, followed by trypsin-Ethylene diamine tetra acetic acid (EDTA) at 37 °C in a humidified environment containing 5% CO2 to loosen up the WJ without complete digestion. After the incubation period, the pieces are washed with PBS solution and transferred to a culture dish, where a similar procedure is repeated. In this method, the entire dish containing WJ pieces in either of the above-mentioned procedure is left undisturbed until MSCs migrate from WJ. By using mixed enzymatic-explant digestion, MSCs are isolated after 24-48 hours.16
Beeravolu and others suggested that MSCs isolated from cord-placenta junction (CPJ) have high proliferation potential and self-renew in vitro, 29 They also showed that the partially-digested perinatal tissue explants protocols are better because of less damage and higher amounts of homogeneous MSCs populations.29
Robert Smith used a new closed system that decreased contaminant in mixed mechanical and enzymatic digestion. This protocol eliminates xenogenic supplement as well as the MSCs viability up to 90% after cryopreservation.28
Comparison between Different MSCs Isolation Methods
Researchers have compared different MSCs isolation and proliferation methods. Studies have shown that cell isolation from the UC matrix using the 10-mm size tissue explant method causes shorter primary culture time, higher numbers of cells, and higher proliferation rates in comparison with the enzyme methods. In the explant method, much valuable time is wasted, but the price and biological contaminations are reduced and the number of isolated cells are high.33
However, other characteristics such as immune phenotype and multilineage differentiation capacity did not differ significantly.34 In the explanted method, the MSCs are isolated by migration. Due to MSC migratory and plastic adhesive properties, the isolation took approximately 5 days. Other studies had developed an optimized enzymatic isolation protocol within 3 hrs. Results showed that the population doubling time of the UC-MSC-Enzyme was estimated to be 2.76±0.60 days.35
The explant method has several advantages in comparison with the enzymatic isolation. These advantages include putting no proteolytic stress on cells, high yield isolation, and reduced cost and risk of biological contaminations.33 Therefore, it has been shown that the explant method is very useful and effective amongst other methods (Table 1).
Table 1.
Comparison between different MSCs isolation methods
| MSCs isolation methods | Advantage | Disadvantage |
|---|---|---|
| Enzymatic procedure | -Can use the total length of the cord and MSC isolation from the three recognized compartments or can use some parts of UC8,13,30 | -Proteolytic stress on cells |
| -Leads to an increase in stem cell yield without time-consuming producers8,30 | -Increased price and | |
| -Isolation protocol within 3 hrs.35 | -High risk of biological contaminations33 | |
| Explants procedure | -High proliferation rate | -Explant pieces floating33 |
| -Higher numbers of homogeneous cell | -The time need to isolate cells is loner35 | |
| -The price and biological contaminations are reduced33 | ||
| -no proteolytic stress on cells | ||
| Mixed enzymatic-explant digestion | -Shorter time need to isolate MSCs from tissue | -There are proteolytic stress on cells but is less than an enzymatic method |
| -High amounts of homogeneous cell.29 | -Price for enzymatic digestion also exists | |
| -MSCs viability up to 90 % after cryopreservation.28 | -High risk of biological contaminations33 |
Hormones and Cytokines Induce MSC Differentiation into Mature Hepatocytes in Vitro
In order to induce MSC differentiation into mature hepatocytes in vitro, there are methods that provide adequate stimuli to maintain cellular function such as growth hormones, cytokines, extracellular matrix or co-culture with other cell types.36 Several cytokines have an effect on cell growth and cell differentiation in to hepatocytes in vitro including hepatocyte growth factor (HGF), basic fibroblast growth factor (bFGF), Oncostatin M (OSM), epidermal growth factor (EGF), transforming growth factor (TGF), insulin, insulin-like growth factor (IGF), leukemia inhibitory factor (LIF), etc. Furthermore, the roles of chemical compounds such as norepinephrine, dexamethasone (Dex), nicotinamide (NTA), retinoic acid, sodium butyrate, and dimethylsulfoxide in promoting hepatic differentiation have been explored. Growth factors such as FGF and HGF secreted from mesoderm in the embryonic period are added to differentiated hepatocyte phenotype. Hence, several protocols have been proposed to facilitate MSC differentiation into hepatocytes with different degrees of differentiation.36 During embryonic development, endodermal specification leads to the production of HGF and FGFs.37
HGF is produced by stromal cells that promote motility, proliferation, invasion, morphogenesis, and survival hepatocytes. It is a potent mitogen and pleiotropic cytokine with a mesenchymal origin and plays a pivotal role in organ formation during embryogenesis and tissue homeostasis as well as liver regeneration in adults.38,39 Combining HGF and FGF initiates the related gene expression to different hepatocyte from MSCs. FGF cluster has a direct effect on cell growth, embryonic development, tissue restoration, and morphogenesis.36 A study showed that by pre-exposing cells to FGF4 before treating with IGF-1 and HGF, liver-specific markers increased.40 Albumin was expressed more in the control cultures, but cytokeratin 18 was expressed more in FGF4-treated cells.41
Another cytokine that promotes liver cell growth is OSM. It is a member of the interleukin 6 family cytokines. In spite of its failure to induce hepatocyte-like phenotype, OSM acts as a significant paracrine regulator in the hepatocyte development toward maturation.42
Dex, a synthetic glucocorticoid, is a potent glucocorticoid receptor (GR) activator, which is a member of the nuclear hormone receptor superfamily. GR regulates genes controlling, metabolism, and immune response. It is binding to specific response elements and that leads to the expression of anti-inflammatory and immunosuppressant reactions manifested clearly through the repression of the NFκB and AP-1 pathways.43 Dex stimulates the hepatocyte to form a cuboidal phenotypic architecture in vitro while accommodating the expression of liver-enriched transcription factors such as C/EBPα, HFN-4α, and RXRα.44 Furthermore, Dex suppresses the hepatocyte proliferative state otherwise stimulated by growth factors such as EGF.45 Dex exerts depends on the results of hepatocyte culture and differentiation, hence high doses of it lead to proliferation,46 but low concentrations of it induce hepatic lineage differentiation.47-49
Hepatic Differentiation Methods
In regenerative medicine and liver tissue engineering, one of the most important issues is hepatic differentiation. As a result, different protocols have been examined that can be categorized into two groups of cocktail and sequential.50 Several sequential methods have been examined for hepatic differentiation. Some studies have reported hepatic differentiation protocols in four, three, and two steps.51-53 For example, the following method was explained by Yoon and others (2010). Mesenchymal stem cells were differentiated by a four-step protocol:
step1: DMEM low glucose (LG)+10 ng/ml FGF-4 for 2 days
step2: DMEM (LG)+1%ITS+20 ng/ml HGF for 2 days
step3: DMEM (LG)+100nMDexamethasone+1%ITS+100 nM Glucagon+10ng/ml OSM for 2 days
step4: DMEM (LG)+100 nM Dexamethasone +1%ITS+100 nM Glucagon+10 ng/ml+ 1%DMSO or100 nM TSA for 8 days. The medium was exchanged every two days.52
The secreted molecules from human MSCs as well as growth factors in the medium affected hepatocyte-like cells differentiation.54
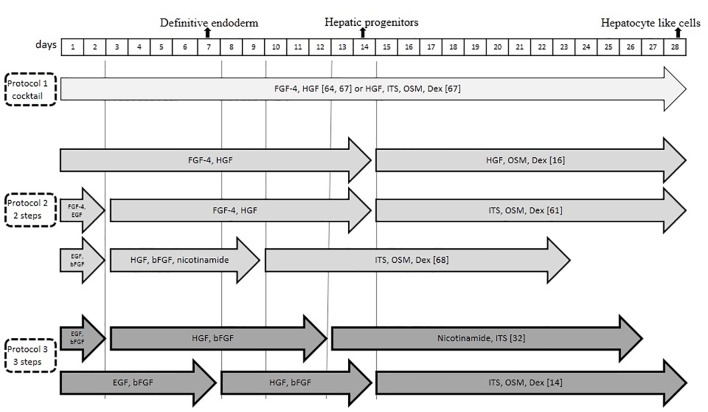
A recent study showed that stem cells with certain microRNAs (miRNAs) could be differentiated into functional hepatocyte-like cells; however, such investigations should be approved in the future studies.55 Takagi and others have introduced and optimized five-miRNA combinations to differentiate MSC-derived hepatocyte-like cells during 7 days.56Table 2 and figure 2 include various parts, such as stem cells isolation protocols, various differentiation protocols (2-4 steps or cocktail), different culturing methods (2D, 3D), and results.
Table 2.
In vitro protocols of MSCs isolation and differentiation in- to-hepatocyte-like cells
| Isolation of MSCs | Induction growth factors | Methods of hepatic differentiation | Culture (2D/3D) | References |
|---|---|---|---|---|
| Enzymatic | Animal model In vivo differentiation | Animal model In vivo differentiation | 3D In-vivo | Chen et al. 201612 |
| Result :Transplanted hUCMSCs into hepatectomized rats differentiated into hepatocyte-like cells | ||||
| Enzymatic | EGF and bFGF, HGF, bFGF and nicotinamide, Dex, ITS premix and OSM | 3 steps protocol | 2D | Lee, et al. 201213 |
| Result :Potential for hepatogenic differentiation of placenta-derived stem cells (PDSCs) containing WJ | ||||
| Explant | HGF,FGF-4,OSM, Dex 2 | 2 steps protocol | 2D | Esmaeli 201415 |
| Result: Shift in the fatty acid profile during the hepatocyte differentiation | ||||
| Enzymatic | EGF, bFGF (2 days) HGF, bFGF (10 days) nicotinamide, ITS (10 days) | Sequential 3 steps | 2D | Campard, et al. 200831 |
| Result: Human MSCs can be considered as a source with hepatogenic potential | ||||
| Explant | bFGF, EGF, | Cocktail | 3D | Su et al. 201432 |
| Result: Poly (3-hydroxybutyrate-co-3-hydroxyvalerate-co-3-hydroxyhexanoate) scaffolds loaded with UC-MSCs or differentiated UC-MSCs promoted the recovery of injured livers | ||||
| Enzymatic | DMEM/F-12 medium with HGF, EGF, ITS, Dex, OSM | 2 steps protocol | 2D | Zhou et al. 201457 |
| Result: Stimulation of host hepatocyte regeneration via hUCMSCs | ||||
| Explant | 40 ng/mL HGF And 10 ng/mL FGF-4 | Cocktail | 2D | Zhang et al. 200958 |
| Result: UC-MSC can differentiate into functional hepatocyte-like cells and have advantages over BM-MSC | ||||
| Explant | EGF and FGF4, HGF, FGF4, Dex, ITS and OSM | 2 steps protocol | 3D | Chitrangi et al. 201759 |
| Result: gelatin-vinyl acetate scaffold enhanced differentiation of UC-MSCs to hepatocyte-like cells | ||||
| Ex-vivo | - | Ex vivo | - | Gomez-Aristizabl and Davies 201260 |
| Result: Human UC perivascular Cells (hucpvcs) can act as stromal cells for rat hepatocytes, | ||||
| Explant | 10 ng/mL HGF, 10 ng/mL FGF4 | Cocktail | 2D | Ewida, et al. 201661 |
| Result: hMSCs can differentiate in vitro into functional hLCs (hepatocyte-like stem cells) in a liver fibrosis rat model | ||||
| Explant | Liver homogenate supernatants (LHS) | Cocktail | 2D | Xue, et al. 201662 |
| Result: Liver tissue microenvironment may contribute to the differentiation of hUCMSCs into hepatocytes both in vitro and in vivo | ||||
| Enzymatic | Dex, EGF, HGF, ITS and OSM | 2-step (for 2 weeks) | 2D | Liang, et al. 201263 |
| Result: Extending hUCMSCs replicative lifespan without influencing hepatogenic differentiation potential via telomerization of hUCMSCs by human telomerase reverse transcriptase (HTERT) | ||||
| Explant | FGF-4 and HGF(14 days) HGF, ITS, oncostatin and Dex. (14 days) | Sequential 2 steps | 3D | Raut, and Khanna 201664 |
| Result: improved expression of hepatocyte-specific miRNAs, miR-23b cluster (miR-27b-3p, miR-24-1-5p and miR-23b-3p), miR-30a-5p, miR-26a-5p, miR-148a-3p, miR-192-5p, miR-122-5p due to VPA pre-treatment | ||||
| Explant | ITS, HGF, OSM, Dex (15 days). | Cocktail | 2D | An, et al. 201465 |
| Result: The lineage conversion of hUCMSCs to hepatic cell fate by upregulating the expression of endodermal genes through AKT and ERK activation by valproic acid (VPA) | ||||
| Explant | EGF, bFGF, HGF, bFGF nicotinamide, OSM, ITS | 2 steps protocol | 2D | Zheng, et al. 201566 |
| Result: Oncostatin M (OSM) acts an important role in hepatogenic differentiation. | ||||
| Explant | HGF, infection with a lentivirus containing a miRNA inhibitor sequence | Cocktail | 2D | Cui, et al. 201367 |
| Result: The capability of miRNAS to converting hMSCs to a hepatocyte phenotype in vitro | ||||
| Explant | IGF-I, HGF and Dex OSM up to 23 days | 2 steps | 3D cell aggregate | Talaei-Khozani, et al. 201568 |
| Result: Facilitating hepatocyte differentiation may be by the UCMSCs aggregate formation before administration of the differentiation protocols | ||||
HGF: hepatocyte growth factor; bFGF: basic fibroblast growth factor; OSM: Oncostatin M; Dex: dexamethasone; EGF: epidermal growth factor; ITS: insulin-transferrin-selenium; IGF: insulin-like growth factor; TGF: transforming growth factor; DMEM: Dulbecco’s Modified Eagle’s Medium
Figure1.
The figure depicts the in vitro protocols of MSCs differentiation into hepatocyte-like cells
Hepatocyte Differentiation in 3-Dimensional Culture
In recent years, several studies have been conducted on 3-dimensional (3-D) differentiation of mesenchymal stem cell, using biocompatible scaffolds such as alginate, collagen, gelatin, and peptide-Gly-Leu-amide (PGLA). Scaffold encapsulation system in vitro culture has been described in both the induction of adult stem cell differentiation and increased function of mature hepatocytes, that can enhance intercellular contacts, improve cell migration, and mimic an in vivo environment condition. 3-D cell culture condition improves the cell stability as well as influencing gene expression pattern.
Several studies have indicated that 3-D co-culture of hepatocytes with different cell types mimics the in vivo, which improves hepatocyte viability and function in vitro.69 The 3D scaffold system did not change the expression level of the cytoskeletal elements CK-18 and 19, but the expression of claudine as a tight junction marker increased significantly in 3D scaffold system.70
Another study showed that the gelatin cryogel scaffold could improve hADMSCs differentiation capability into hepatocyte-like cells in comparison with 2D tissue culture polystyrene. In this scaffold cell number, morphology and viability increased. The gene expression of hepatocytes pacific genes (ALB, AFP, CK-18, and CK-19), urea production, and glycogen storage were reported to have improved.71
In addition, liver expression markers and HNF4α showed an improvement in 3-D culture in comparison with 2-D.72
Discussion
Stem cells represent a unique source of self-renewing organism. Stem cell therapy has the potential to become an alternative therapy for liver transplantation. Several important factors, such as stem cell source, cytokine, hormone, differentiation protocols, and culture system, should be considered for hepatic differentiation of stem cells into hepatocyte-like cells. Hepatic differentiation is often linked to the selection of stem cell sources. Hence, UC-MSCs have been introduced as a suitable stem cell source. Several studies have shown that UC-MSCs with similar immune phenotype and multilineage differentiation have higher potential to expand in comparison with other sources such as BM-MSCs and ADMSCs. 9,10,25,26
Selecting the best isolation method is critical since it saves time, reduces costs, and increases the cell expansion and proliferation. There are still ongoing studies on these methods. Most of these studies have reported that the explant method could be considered as a suitable alternative. A recent review showed that using explant method could lead to shorter primary culture time, higher numbers of cells, and higher proliferation rates in comparison with enzyme methods. Additionally, the price and biological contaminations were reduced as well.33
In this review, various differentiation protocols of stem cells into hepatocyte-like cells are summarized in Table 2.
One of the most important factors in hepatic differentiation is to determine an appropriate induction method. Several hormones and cytokines including HGF, FGF, OSM, EGF, TGF, IGF, LIF, and … have known to be involved in the regeneration of liver after hepatic injury and during organogenesis. Combining these cytokines initiates the related genes expression, to differentiate MSCs into hepatocyte form. During organogenesis, liver development is a sequential array of distinct biological events. Therefore, several studies have been conducted on the use of cell cytokines, such as HGF, FGF, and oncostatin M, in a sequential manner in 3 or 4 stages.
The culture system method (2D and 3D) is very important. It has been shown that 2D culture of stem cell in the first stage of differentiation is critical, and, in the second stage, 3D culture system can enhance stem cell differentiation into hepatocyte. The 3D culture system can mimic in vivo condition. Moreover, the differentiated hepatocyte has the best quality and function. Several studies investigated hepatic differentiation with cytokines in 2D and 3D culture, and the results revealed more advantages for 3D culture conditions than 2D culture conditions.69,70
Conclusion
In this review, we conclude that WJ-MSC can be considered as a suitable source for differentiation into hepatocyte-like cells since it has no ethical concern but high proliferation and low immunogenicity. WJ-MSC isolation from the umbilical cord is very efficient through explant culture method in comparison with enzymatic methods. In the early stage of differentiation, 2D culture is more useful, but in the development stage, 3D culture system with HGF and FGF cytokines can be more effective in the differentiation process. In this review, we attempted to highlight all factors in hepatocyte differentiation, but the best and most effective protocol for hepatocyte differentiation is still unknown.
Acknowledgement
The authors wish to thank the staff at Cellular and Molecular Research Center of Ahvaz Jundishapur University of Medical Sciences for their support. The authors also wish to thank Mr. H. Argasi at the Research Consultation Center (RCC) of Shiraz University of Medical Sciences for his invaluable assistance in editing this manuscript.
Conflict of Interest:None declared.
References
- 1.Sun K, Xie X, Xie J, Jiao S, Chen X, Zhao X, et al. Cell-based therapy for acute and chronic liver failures: distinct diseases, different choices. Sci Rep. 2014;4:6494. doi: 10.1038/srep06494. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Russo FP, Parola M. Stem cells in liver failure. Best Pract Res Clin Gastroenterol. 2012;26:35–45. doi: 10.1016/j.bpg.2012.01.001. [DOI] [PubMed] [Google Scholar]
- 3.Schwartz RE, Reyes M, Koodie L, Jiang Y, Blackstad M, Lund T, et al. Multipotent adult progenitor cells from bone marrow differentiate into functional hepatocyte-like cells. J Clin Invest. 2002;109:1291–302. doi: 10.1172/JCI15182. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Seo MJ, Suh SY, Bae YC, Jung JS. Differentiation of human adipose stromal cells into hepatic lineage in vitro and in vivo. Biochem Biophys Res Commun. 2005;328:258–64. doi: 10.1016/j.bbrc.2004.12.158. [DOI] [PubMed] [Google Scholar]
- 5.Sidhu KS. Frontiers in Pluripotent Stem Cells Research and Therapeutic Potentials: Bench-to-bedside. Sharjah: Bentham Science Publishers; 2012. [Google Scholar]
- 6.Kolios G, Moodley Y. Introduction to stem cells and regenerative medicine. Respiration. 2013;85:3–10. doi: 10.1159/000345615. [DOI] [PubMed] [Google Scholar]
- 7.Romanov YA, Svintsitskaya VA, Smirnov VN. Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. Stem Cells. 2003;21:105–10. doi: 10.1634/stemcells.21-1-105. [DOI] [PubMed] [Google Scholar]
- 8.Seshareddy K, Troyer D, Weiss ML. Method to isolate mesenchymal-like cells from Wharton’s Jelly of umbilical cord. Methods Cell Biol. 2008;86:101–19. doi: 10.1016/S0091-679X(08)00006-X. [DOI] [PubMed] [Google Scholar]
- 9.Simoes IN, Boura JS, dos Santos F, Andrade PZ, Cardoso CM, Gimble JM, et al. Human mesenchymal stem cells from the umbilical cord matrix: successful isolation and ex vivo expansion using serum-/xeno-free culture media. Biotechnol J. 2013;8:448–58. doi: 10.1002/biot.201200340. [DOI] [PubMed] [Google Scholar]
- 10.Cheng H, Qiu L, Ma J, Zhang H, Cheng M, Li W, et al. Replicative senescence of human bone marrow and umbilical cord derived mesenchymal stem cells and their differentiation to adipocytes and osteoblasts. Mol Biol Rep. 2011;38:5161–8. doi: 10.1007/s11033-010-0665-2. [DOI] [PubMed] [Google Scholar]
- 11.Deuse T, Stubbendorff M, Tang-Quan K, Phillips N, Kay MA, Eiermann T, et al. Immunogenicity and immunomodulatory properties of umbilical cord lining mesenchymal stem cells. Cell Transplant. 2011;20:655–67. doi: 10.3727/096368910X536473. [DOI] [PubMed] [Google Scholar]
- 12.Chen Z, Kuang Q, Lao XJ, Yang J, Huang W, Zhou D. Differentiation of UC-MSCs into hepatocyte-like cells in partially hepatectomized model rats. Exp Ther Med. 2016;12:1775–9. doi: 10.3892/etm.2016.3543. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee HJ, Jung J, Cho KJ, Lee CK, Hwang SG, Kim GJ. Comparison of in vitro hepatogenic differentiation potential between various placenta-derived stem cells and other adult stem cells as an alternative source of functional hepatocytes. Differentiation. 2012;84:223–31. doi: 10.1016/j.diff.2012.05.007. [DOI] [PubMed] [Google Scholar]
- 14.Hashemitabar M, Allahbakhshi E, Tabande MR, Orazizadeh M, Dehbashi FN, Azandeh S, et al. Isolation and Characterization of Human Umbilical Cord Mesenchymal Stem Cells and Their Differentiation into Pdx-1+ Cells. J Biomed Sci Eng. 2015;8:780–8. doi: 10.4236/jbise.2015.811074. [DOI] [Google Scholar]
- 15.Esmaeli S, Allameh A, Soleimani M, Rahbarizadeh F, Frouzandeh-Moghadam M. The role of albumin and PPAR-alpha in differentiation-dependent change of fatty acid profile during differentiation of mesenchymal stem cells to hepatocyte-like cells. Cell Biochem Funct. 2014;32:410–9. doi: 10.1002/cbf.3031. [DOI] [PubMed] [Google Scholar]
- 16.Azandeh S, Orazizadeh M, Hashemitabar M, Khodadadi A, Shayesteh AA, Nejad DB, et al. Mixed enzymatic-explant protocol for isolation of mesenchymal stem cells from Wharton’s jelly and encapsulation in 3D culture system. Journal of Biomedical Science and Engineering. 2012;5:580. doi: 10.4236/jbise.2012.510071. [DOI] [Google Scholar]
- 17.Sarugaser R, Lickorish D, Baksh D, Hosseini MM, Davies JE. Human umbilical cord perivascular (HUCPV) cells: a source of mesenchymal progenitors. Stem Cells. 2005;23:220–9. doi: 10.1634/stemcells.2004-0166. [DOI] [PubMed] [Google Scholar]
- 18.Ruetze M, Gallinat S, Lim IJ, Chow E, Phan TT, Staeb F, et al. Common features of umbilical cord epithelial cells and epidermal keratinocytes. J Dermatol Sci. 2008;50:227–31. doi: 10.1016/j.jdermsci.2007.12.006. [DOI] [PubMed] [Google Scholar]
- 19.Ilancheran S, Michalska A, Peh G, Wallace EM, Pera M, Manuelpillai U. Stem cells derived from human fetal membranes display multilineage differentiation potential. Biol Reprod. 2007;77:577–88. doi: 10.1095/biolreprod.106.055244. [DOI] [PubMed] [Google Scholar]
- 20.Weiss ML, Medicetty S, Bledsoe AR, Rachakatla RS, Choi M, Merchav S, et al. Human umbilical cord matrix stem cells: preliminary characterization and effect of transplantation in a rodent model of Parkinson’s disease. Stem Cells. 2006;24:781–92. doi: 10.1634/stemcells.2005-0330. [DOI] [PubMed] [Google Scholar]
- 21.Fong CY, Richards M, Manasi N, Biswas A, Bongso A. Comparative growth behaviour and characterization of stem cells from human Wharton’s jelly. Reprod Biomed Online. 2007;15:708–18. doi: 10.1016/s1472-6483(10)60539-1. [DOI] [PubMed] [Google Scholar]
- 22.Fong CY, Chak LL, Biswas A, Tan JH, Gauthaman K, Chan WK, et al. Human Wharton’s jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011;7:1–16. doi: 10.1007/s12015-010-9166-x. [DOI] [PubMed] [Google Scholar]
- 23.Lee BH, Li B, Guelcher SA. Gel microstructure regulates proliferation and differentiation of MC3T3-E1 cells encapsulated in alginate beads. Acta Biomater. 2012;8:1693–702. doi: 10.1016/j.actbio.2012.01.012. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fong CY, Subramanian A, Biswas A, Gauthaman K, Srikanth P, Hande MP, et al. Derivation efficiency, cell proliferation, freeze-thaw survival, stem-cell properties and differentiation of human Wharton’s jelly stem cells. Reprod Biomed Online. 2010;21:391–401. doi: 10.1016/j.rbmo.2010.04.010. [DOI] [PubMed] [Google Scholar]
- 25.Rao MS, Mattson MP. Stem cells and aging: expanding the possibilities. Mech Ageing Dev. 2001;122:713–34. doi: 10.1016/s0047-6374(01)00224-x. [DOI] [PubMed] [Google Scholar]
- 26.Huss R. Isolation of primary and immortalized CD34-hematopoietic and mesenchymal stem cells from various sources. Stem Cells. 2000;18:1–9. doi: 10.1634/stemcells.18-1-1. [DOI] [PubMed] [Google Scholar]
- 27.Manca MF, Zwart I, Beo J, Palasingham R, Jen LS, Navarrete R, et al. Characterization of mesenchymal stromal cells derived from full-term umbilical cord blood. Cytotherapy. 2008;10:54–68. doi: 10.1080/14653240701732763. [DOI] [PubMed] [Google Scholar]
- 28.Smith JR, Cromer A, Weiss ML. Human Umbilical Cord Mesenchymal Stromal Cell Isolation, Expansion, Cryopreservation, and Characterization. Curr Protoc Stem Cell Biol. 2017;41:1F 18 1–1F 23. doi: 10.1002/cpsc.24. [DOI] [PubMed] [Google Scholar]
- 29.Beeravolu N, McKee C, Alamri A, Mikhael S, Brown C, Perez-Cruet M, et al. Isolation and Characterization of Mesenchymal Stromal Cells from Human Umbilical Cord and Fetal Placenta. J Vis Exp. 2017;122:e55224. doi: 10.3791/55224. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tsagias N, Koliakos I, Karagiannis V, Eleftheriadou M, Koliakos GG. Isolation of mesenchymal stem cells using the total length of umbilical cord for transplantation purposes. Transfus Med. 2011;21:253–61. doi: 10.1111/j.1365-3148.2011.01076.x. [DOI] [PubMed] [Google Scholar]
- 31.Campard D, Lysy PA, Najimi M, Sokal EM. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology. 2008;134:833–48. doi: 10.1053/j.gastro.2007.12.024. [DOI] [PubMed] [Google Scholar]
- 32.Su Z, Li P, Wu B, Ma H, Wang Y, Liu G, et al. PHBVHHx scaffolds loaded with umbilical cord-derived mesenchymal stem cells or hepatocyte-like cells differentiated from these cells for liver tissue engineering. Mater Sci Eng C Mater Biol Appl. 2014;45:374–82. doi: 10.1016/j.msec.2014.09.022. [DOI] [PubMed] [Google Scholar]
- 33.Hendijani F. Explant culture: An advantageous method for isolation of mesenchymal stem cells from human tissues. Cell Prolif. 2017;50:e12334. doi: 10.1111/cpr.12334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hua J, Gong J, Meng H, Xu B, Yao L, Qian M, et al. Comparison of different methods for the isolation of mesenchymal stem cells from umbilical cord matrix: proliferation and multilineage differentiation as compared to mesenchymal stem cells from umbilical cord blood and bone marrow. Cell Biol Int. 2013;38:198, 210. doi: 10.1002/cbin.10188. [DOI] [PubMed] [Google Scholar]
- 35.Buyl K, Vanhaecke T, Desmae T, Lagneaux L, Rogiers V, Najar M, et al. Evaluation of a new standardized enzymatic isolation protocol for human umbilical cord-derived stem cells. Toxicol In Vitro. 2015;29:1254–62. doi: 10.1016/j.tiv.2014.12.008. [DOI] [PubMed] [Google Scholar]
- 36.Wu XB, Tao R. Hepatocyte differentiation of mesenchymal stem cells. Hepatobiliary Pancreat Dis Int. 2012;11:360–71. doi: 10.1016/S1499-3872(12)60193-3. [DOI] [PubMed] [Google Scholar]
- 37.Jung J, Zheng M, Goldfarb M, Zaret KS. Initiation of mammalian liver development from endoderm by fibroblast growth factors. Science. 1999;284:1998–2003. doi: 10.1126/science.284.5422.1998. [DOI] [PubMed] [Google Scholar]
- 38.Santos OF, Barros EJ, Yang XM, Matsumoto K, Nakamura T, Park M, et al. Involvement of hepatocyte growth factor in kidney development. Dev Biol. 1994;163:525–9. doi: 10.1006/dbio.1994.1169. [DOI] [PubMed] [Google Scholar]
- 39.Uehara Y, Minowa O, Mori C, Shiota K, Kuno J, Noda T, et al. Placental defect and embryonic lethality in mice lacking hepatocyte growth factor/scatter factor. Nature. 1995;373:702–5. doi: 10.1038/373702a0. [DOI] [PubMed] [Google Scholar]
- 40.Talaei-Khozani T. Improved Differentiation of Mesenchymal Stem Cells into Hepatocyte-like Cells using FGF4 and IGF-1 in 3D culture. Int J Stem Cell Res Transplant. 2015;3:109–17. doi: 10.19070/2328-3548-1500018. [DOI] [Google Scholar]
- 41.Vojdani Z, Khodabandeh Z, Jaberipour M, Hosseini A, Bahmanpour S, Talaei-Khozani T. The influence of fibroblast growth factor 4 on hepatogenic capacity of Wharton’s jelly mesenchymal stromal cells. Rom J Morphol Embryol. 2015;56:1043–50. [PubMed] [Google Scholar]
- 42.Kinoshita T, Sekiguchi T, Xu MJ, Ito Y, Kamiya A, Tsuji K, et al. Hepatic differentiation induced by oncostatin M attenuates fetal liver hematopoiesis. Proc Natl Acad Sci U S A. 1999;96:7265–70. doi: 10.1073/pnas.96.13.7265. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nelson G, Wilde GJ, Spiller DG, Kennedy SM, Ray DW, Sullivan E, et al. NF-kappaB signalling is inhibited by glucocorticoid receptor and STAT6 via distinct mechanisms. J Cell Sci. 2003;116:2495–503. doi: 10.1242/jcs.00461. [DOI] [PubMed] [Google Scholar]
- 44.Dasgupta A, Hughey R, Lancin P, Larue L, Moghe PV. E-cadherin synergistically induces hepatospecific phenotype and maturation of embryonic stem cells in conjunction with hepatotrophic factors. Biotechnol Bioeng. 2005;92:257–66. doi: 10.1002/bit.20676. [DOI] [PubMed] [Google Scholar]
- 45.Scheving LA, Stevenson MC, Zhang X, Russell WE. Cultured rat hepatocytes upregulate Akt and ERK in an ErbB-2-dependent manner. Am J Physiol Gastrointest Liver Physiol. 2008;295:G322–31. doi: 10.1152/ajpgi.00597.2007. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Scheving LA, Buchanan R, Krause MA, Zhang X, Stevenson MC, Russell WE. Dexamethasone modulates ErbB tyrosine kinase expression and signaling through multiple and redundant mechanisms in cultured rat hepatocytes. Am J Physiol Gastrointest Liver Physiol. 2007;293:G552–9. doi: 10.1152/ajpgi.00140.2007. [DOI] [PubMed] [Google Scholar]
- 47.Baharvand H, Hashemi SM, Shahsavani M. Differentiation of human embryonic stem cells into functional hepatocyte-like cells in a serum-free adherent culture condition. Differentiation. 2008;76:465–77. doi: 10.1111/j.1432-0436.2007.00252.x. [DOI] [PubMed] [Google Scholar]
- 48.Saito K, Yoshikawa M, Ouji Y, Moriya K, Nishiofuku M, Ueda S, et al. Promoted differentiation of cynomolgus monkey ES cells into hepatocyte-like cells by co-culture with mouse fetal liver-derived cells. World J Gastroenterol. 2006;12:6818–27. doi: 10.3748/wjg.v12.i42.6818. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shirahashi H, Wu J, Yamamoto N, Catana A, Wege H, Wager B, et al. Differentiation of human and mouse embryonic stem cells along a hepatocyte lineage. Cell Transplant. 2004;13:197–211. doi: 10.3727/000000004783984016. [DOI] [PubMed] [Google Scholar]
- 50.Banas A, Teratani T, Yamamoto Y, Tokuhara M, Takeshita F, Quinn G, et al. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology. 2007;46:219–28. doi: 10.1002/hep.21704. [DOI] [PubMed] [Google Scholar]
- 51.Snykers S, De Kock J, Tamara V, Rogiers V. Hepatic differentiation of mesenchymal stem cells: in vitro strategies. Methods Mol Biol. 2011;698:305–14. doi: 10.1007/978-1-60761-999-4_23. [DOI] [PubMed] [Google Scholar]
- 52.Yoon HH, Jung BY, Seo YK, Song KY, Park JK. In vitro hepatic differentiation of umbilical cord-derived mesenchymal stem cell. Process Biochemistry. 2010;45:1857–64. doi: 10.1016/j.procbio.2010.06.009. [DOI] [Google Scholar]
- 53.Lee KD, Kuo TKC, Whang‐Peng J, Chung YF, Lin CT, Chou SH, et al. In vitro hepatic differentiation of human mesenchymal stem cells. Hepatology. 2004;40:1275–84. doi: 10.1002/hep.20469. [DOI] [PubMed] [Google Scholar]
- 54.Takagi C, Yagi H, Hieda M, Tajima K, Hibi T, Abe Y, et al. Mesenchymal Stem Cells Contribute to Hepatic Maturation of Human Induced Pluripotent Stem Cells. Eur Surg Res. 2017;58:27–39. doi: 10.1159/000448516. [DOI] [PubMed] [Google Scholar]
- 55.Davoodian N, Lotfi AS, Soleimani M, Ghaneialvar H. The combination of miR-122 overexpression and Let-7f silencing induces hepatic differentiation of adipose tissue-derived stem cells. Cell Biol Int. 2017;41:1083–92. doi: 10.1002/cbin.10836. [DOI] [PubMed] [Google Scholar]
- 56.Zhou X, Cui L, Zhou X, Yang Q, Wang L, Guo G, et al. Induction of hepatocyte-like cells from human umbilical cord-derived mesenchymal stem cells by defined microRNAs. J Cell Mol Med. 2017;21:881–93. doi: 10.1111/jcmm.13027. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhou R, Li Z, He C, Li R, Xia H, Li C, et al. Human umbilical cord mesenchymal stem cells and derived hepatocyte-like cells exhibit similar therapeutic effects on an acute liver failure mouse model. PLoS One. 2014;9:e104392. doi: 10.1371/journal.pone.0104392. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang YN, Lie PC, Wei X. Differentiation of mesenchymal stromal cells derived from umbilical cord Wharton’s jelly into hepatocyte-like cells. Cytotherapy. 2009;11:548–58. doi: 10.1080/14653240903051533. [DOI] [PubMed] [Google Scholar]
- 59.Chitrangi S, Nair P, Khanna A. Three-dimensional polymer scaffolds for enhanced differentiation of human mesenchymal stem cells to hepatocyte-like cells: a comparative study. J Tissue Eng Regen Med. 2017;11:2359–72. doi: 10.1002/term.2136. [DOI] [PubMed] [Google Scholar]
- 60.Gomez-Aristizabal A, Davies JE. Human umbilical cord perivascular cells improve rat hepatocyte function ex vivo. Tissue Eng Part A. 2012;18:2487–96. doi: 10.1089/ten.TEA.2011.0669. [DOI] [PubMed] [Google Scholar]
- 61.Ewida SF, Abdou AG, El-Rasol Elhosary AA, El-Ghane Metawe SA. Hepatocyte-like Versus Mesenchymal Stem Cells in CCl4-induced Liver Fibrosis. Appl Immunohistochem Mol Morphol. 2017;25:736–45. doi: 10.1097/PAI.0000000000000373. [DOI] [PubMed] [Google Scholar]
- 62.Xue G, Han X, Ma X, Wu H, Qin Y, Liu J, et al. Effect of Microenvironment on Differentiation of Human Umbilical Cord Mesenchymal Stem Cells into Hepatocytes In Vitro and In Vivo. Biomed Res Int. 2016;2016:8916534. doi: 10.1155/2016/8916534. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liang XJ, Chen XJ, Yang DH, Huang SM, Sun GD, Chen YP. Differentiation of human umbilical cord mesenchymal stem cells into hepatocyte-like cells by hTERT gene transfection in vitro. Cell Biol Int. 2012;36:215–21. doi: 10.1042/CBI20110350. [DOI] [PubMed] [Google Scholar]
- 64.Raut A, Khanna A. Enhanced expression of hepatocyte-specific microRNAs in valproic acid mediated hepatic trans-differentiation of human umbilical cord derived mesenchymal stem cells. Exp Cell Res. 2016;343:237–47. doi: 10.1016/j.yexcr.2016.03.015. [DOI] [PubMed] [Google Scholar]
- 65.An SY, Han J, Lim HJ, Park SY, Kim JH, Do BR, et al. Valproic acid promotes differentiation of hepatocyte-like cells from whole human umbilical cord-derived mesenchymal stem cells. Tissue Cell. 2014;46:127–35. doi: 10.1016/j.tice.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 66.Zheng G, Liu Y, Jing Q, Zhang L. Differentiation of human umbilical cord-derived mesenchymal stem cells into hepatocytes in vitro. Biomed Mater Eng. 2015;25:145–57. doi: 10.3233/BME-141249. [DOI] [PubMed] [Google Scholar]
- 67.Cui L, Shi Y, Zhou X, Wang X, Wang J, Lan Y, et al. A set of microRNAs mediate direct conversion of human umbilical cord lining-derived mesenchymal stem cells into hepatocytes. Cell Death Dis. 2013;4:e918. doi: 10.1038/cddis.2013.429. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Talaei-Khozani T, Borhani-Haghighi M, Ayatollahi M, Vojdani Z. An in vitro model for hepatocyte-like cell differentiation from Wharton’s jelly derived-mesenchymal stem cells by cell-base aggregates. Gastroenterol Hepatol Bed Bench. 2015;8:188–99. [ PMC Free Article] [PMC free article] [PubMed] [Google Scholar]
- 69.Azandeh S, Mohammad Gharravi A, Orazizadeh M, Khodadi A, Hashemi Tabar M. Improvement of mesenchymal stem cell differentiation into the endoderm lineage by four step sequential method in biocompatible biomaterial. Bioimpacts. 2016;6:9–13. doi: 10.15171/bi.2016.02. [ PMC Free Article] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Khodabandeh Z, Vojdani Z, Talaei-Khozani T, Jaberipour M, Hosseini A, Bahmanpour S. Comparison of the Expression of Hepatic Genes by Human Wharton’s Jelly Mesenchymal Stem Cells Cultured in 2D and 3D Collagen Culture Systems. Iran J Med Sci. 2016;41:28–36. [ PMC Free Article] [PMC free article] [PubMed] [Google Scholar]
- 71.Ghaderi Gandomani M, Sahebghadam Lotfi A, Kordi Tamandani D, Arjmand S, Alizadeh S. The enhancement of differentiating adipose derived mesenchymal stem cells toward hepatocyte like cells using gelatin cryogel scaffold. Biochem Biophys Res Commun. 2017;491:1000–6. doi: 10.1016/j.bbrc.2017.07.167. [DOI] [PubMed] [Google Scholar]
- 72.Talaei-Khozani T, Khodabandeh Z, Jaberipour M, Hosseini A, Bahmanpour S, Vojdani Z. Comparison of hepatic nuclear factor-4 expression in two- and three-dimensional culture of Wharton’s jelly-derived cells exposed to hepatogenic medium. Rom J Morphol Embryol. 2015;56:1365–70. [PubMed] [Google Scholar]