Abstract
Chronic wounds exhibit persistent inflammation with markedly delayed healing. The significant burden of chronic wounds, which are often resistant to standard therapy, prompts further research on novel therapies. Since the interleukin-17 family has been implicated as a group of proinflammatory cytokines in immune-mediated diseases in the gut and connective tissue, as well as inflammatory skin conditions, we consider here if it may contribute to the pathogenesis of chronic wounds. In this review, we discuss the interleukin-17 family's signaling pathways and role in tissue repair. A PubMed review of the English literature on interleukin-17, wound healing, chronic wounds, and inflammatory skin conditions was conducted. Interleukin-17 family signaling is reviewed in the context of tissue repair, and preclinical and clinical studies examining its role in the skin and other organ systems are critically reviewed. The published work supports a pathologic role for interleukin-17 family members in chronic wounds, though this needs to be more conclusively proven. Clinical studies using monoclonal interleukin-17 antibodies to improve healing of chronic skin wounds have not yet been performed, and only a few studies have examined interleukin-17 family expression in chronic skin wounds. Furthermore, different interleukin-17 family members could be playing selective roles in the repair process. These studies suggest a therapeutic role for targeting interleukin-17A to promote wound healing; therefore, interleukin-17A may be a target worthy of pursuing in the near future.
1. Introduction
More than 9 million people in the United States are diagnosed with chronic wounds, and the incidence rate is expected to grow rapidly with the aging and increasingly diabetic and obese population [1]. Treatment for chronic wounds costs approximately $28-31 billion each year [1]. The public health concern and excessive financial burden warrant further efforts to effectively manage chronic wounds. “Chronic” wounds are defined by the US Centers for Medicare & Medicaid Services (CMS) as “wounds that do not heal completely after receiving standard medical treatment for 30 days” [2] and are characterized by delayed reepithelialization with persistent elevation of inflammatory markers [3–5]. While inflammation is a necessary component of the early wound healing process, excessive, prolonged, and dysregulated inflammation is associated with impaired wound healing [3–5]. The lack of response to standard therapies prompts ongoing studies for pathogenic biomarkers and trials for novel therapies. Therefore, in this review, we discuss the potential pathologic role of the interleukin-17 (IL-17) family in the wound repair process in order to evaluate it as a possible target for therapy.
The IL-17 family consists of a group of proinflammatory cytokines with an active role in host defense, yet implicated in the pathogenesis of a wide range of immune-mediated diseases, including psoriasis, psoriatic arthritis, rheumatoid arthritis, ankylosing spondylitis, and others such as hidradenitis suppurativa [6–9]. Members of the IL-17 family that are now recognized as critical cytokines altering skin function in psoriasis and psoriatic arthritis are IL-17A, IL-17C, and IL-17F. These cytokines act on keratinocytes to induce the expression of several chemokines leading to the recruitment and accumulation of neutrophils, T cells, and dendritic cells, causing epidermal and vascular hyperplasia as seen in psoriasis [6, 10]. Studies have revealed increased expression of IL-17A, IL-17C, and IL-17F in psoriatic skin as compared to nonlesional skin with the upregulation of IL-17A showing a positive association with disease severity [11, 12]. Monoclonal antibodies targeting IL-17A (i.e., secukinumab and ixekizumab) or the IL-17 receptor subunit IL-17RA (i.e., brodalumab) have already demonstrated dramatic therapeutic results in patients with psoriasis, psoriatic arthritis, and ankylosing spondylitis, thus confirming the pathogenic relevance of IL-17 family members in mediating inflammation in psoriasis, psoriatic arthritis, and ankylosing spondylitis [13–16].
Recent studies have also demonstrated the IL-17 family to be a possible mediator of inflammation in hidradenitis suppurativa. Hidradenitis suppurativa (HS) has shown a T-helper 17 cell- (TH17-) skewed cytokine profile in inflamed HS skin, based on intracellular cytokine staining, with the ratio of TH17 to regulatory T cells dysregulated in favor of TH17 cells (identified by flow cytometry using antibody to IL-17A or IL-17F). Lesional skin from antitumor necrosis factor- (TNF-) treated HS patients revealed a reduction in the frequency of TH17 cells and normalization of the TH17 to regulatory T cell ratio [17]. This clustering of TH1/TH17-associated cytokines around the lesional inflammation has also been demonstrated in other studies on HS [18].
Six members of the IL-17 family have been identified (IL-I7A, IL-17B, IL-17C, IL-17D, IL-17E/IL-25, and IL-17F) [9] by their conserved C-terminal region but differing N-terminal segments [19]. Different members of the IL-17 family have opposing signaling pathways and biological outcomes; however, not all pathways have been comprehensively elucidated. Thus, we have focused on the signaling pathways most relevant to the role of the IL-17 family members in wound pathogenesis. Furthermore, the role of the IL-17 family in chronic cutaneous wounds has not been fully determined; however, the cumulative preclinical and clinical studies in other organ systems suggest that further investigation is warranted to determine its potential as a therapeutic target for chronic wound patients.
2. IL-17 Pathway
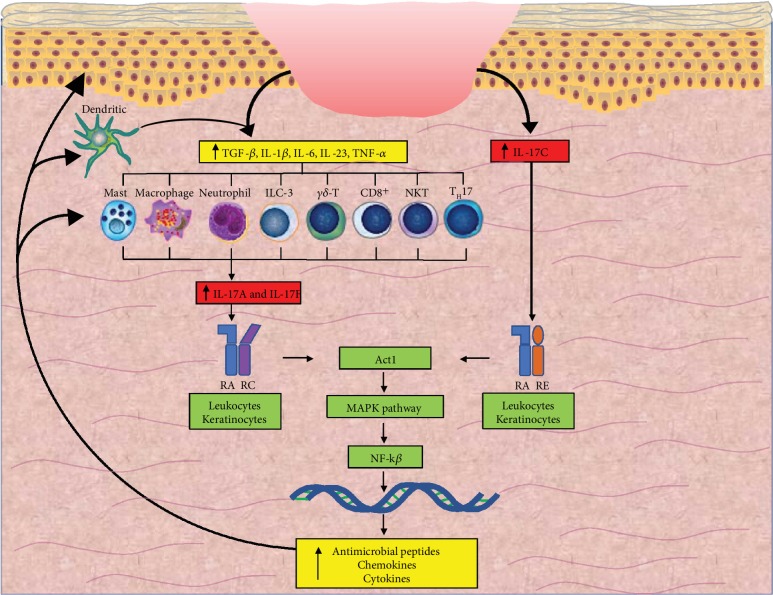
The members of the IL-17 family are released from cells of both the innate and adaptive arms of immunity, as well as a wide range of nonhematopoietic cells such as epithelial cells, depending on the cytokine subtype (Table 1) [9]. Transforming growth factor-β (TGF-β) and proinflammatory cytokines IL-1β, IL-6, and IL-23 stimulate IL-17A and IL-17F release from T-helper 17 (TH17) cells, CD8-positive T cells, ɣδ-T cells, natural killer T (NKT) cells, and Type 3 innate lymphoid cells (ILC3), while IL-1β and TNF-α activate IL-17C release from epithelial cells (Figure 1) [20–32]. Additional studies have suggested the release of IL-17A from macrophages, neutrophils, mast cells, and dendritic cells and IL-17C from mononuclear cells (Figure 1) [28, 33–38]. Interestingly, IL-17E has been shown to attenuate the TH17 response in a model of experimental autoimmune encephalomyelitis by stimulating the production of the T-helper 2 (TH2) cytokine IL-13, which inhibits the production of TH17-promoting cytokines IL-1β, IL-6, and IL-23 from dendritic cells [39]. This suggests the possibility of IL-17E as a potential downregulator of IL-17A and IL-17F.
Table 1.
IL-17 family.
| Subtype | Reported biological sources | Heterodimer receptor subunits |
|---|---|---|
| A | Mast cells, macrophages, neutrophils, ILC3, ɣδ-T, NKT, CD8+, TH17 [9] | RA/RC [9] |
| B | Gastrointestinal tract, pancreas, gonadal tissue, chondrocytes, synovial membrane, neurons, lymphocytes [40, 60] | RA/RB [40] |
| C | Epithelial cells in the colon, trachea, and skin [32] | RA/RE [32] |
| D | Pancreas, adipose, skeletal muscle, brain, heart, lung [61] | Unidentified |
| E | Mast cells, macrophages, eosinophils, basophils, respiratory epithelium, and mucosa [9] | RA/RB [9] |
| F | Mast cells, macrophages, neutrophils, ILC3, ɣδ-T, NKT, CD8+, TH17 [9] | RA/RC [9] |
Figure 1.
Skin wounding induces the release of IL-17A, IL-17F, and IL-17C from keratinocytes and leukocytes. These ligands bind to their respective heterodimeric receptors (IL-17A and IL-17F at IL-17RA/RC; IL-17C at IL-17RA/RE) on keratinocytes and leukocytes to release antimicrobial peptides, chemokines, and cytokines—contributing to a self-perpetuating cycle of inflammation. TGF-β: transforming growth factor-β; IL: interleukin; TNF-α: tumor necrosis factor-α; ILC3: Type 3 innate lymphoid cells; NKT: natural killer T; TH17: T-helper 17; RA: receptor A subunit; RC: receptor C subunit; RE: receptor E subunit; MAPK: mitogen-activated protein kinase; NF-κB: nuclear factor-kB.
Upon release, IL-17 family ligands bind heterodimeric transmembrane receptors consisting of a common IL-17 receptor A (IL-17RA) subunit and a unique second subunit specific for cytokine subtype: IL-17A and IL-17F bind IL-17RA/RC; IL-17C binds IL-17RA/RE; IL-17B and IL-17E bind IL-17RA/RB (Table 1) [32, 40–42]. The receptor moiety specific for IL-17D has yet to be identified. All five IL-17 receptors share a fibronectin type III (FnIII) extracellular domain and, contrary to other cytokine receptors but similar to toll-like receptors, a cytoplasmic SEF-IL-17R domain [43]. Upon binding to their receptors, IL-17A, IL-17C, and IL-17F induce a conformational change in their receptors that allows heterodimeric complex formation between the specific receptor subunits (subunits RA and RC heterodimerize upon binding ligands IL-17A or IL-17F; subunits RA and RE heterodimerize upon binding ligand IL-17C) and subsequently activates Act1, which is an adaptor protein that initiates the mitogen-activated protein kinase (MAPK) and nuclear factor-kB (NF-κB) signaling cascades (Figure 1) [43–49]. These signaling pathways lead to the expression of antimicrobial peptides, chemokines, and cytokines such as TNF-α, IL-1β, IL-6, and IL-23, thereby triggering recruitment of additional proinflammatory immune cells including neutrophils and TH17 cells, further perpetuating the inflammation cycle (Figure 1) [50–53]. Specifically in keratinocytes, IL-17A activates a signaling cascade via the IL-17R–Act1–TRAF4–MEKK3–ERK5 pathway, which stimulates keratinocyte proliferation [54]. In addition to proliferation, primary keratinocytes stimulated with IL-17 alone transcribe several neutrophil chemokines and antimicrobial peptides, creating a proinflammatory skewed environment [55].
It is also important to note that the IL-17 family exhibits synergistic effects with other cytokines such as TNF-α, IL-1, and IL-6 [52, 56–59]. In contrast with other IL-17 subtypes, IL-17E has been implicated to induce the release of TH2-related cytokines, instigating eosinophilic recruitment in allergic and parasitic conditions [8, 9, 48]. Further studies are necessary to clarify the signaling pathway of IL-17B and IL-17D.
3. IL-17 Modulates Tissue Repair
3.1. In Skin
In homeostasis, genetic deletion of IL-17RA on nonhematopoietic cells in mice resulted in increased filaggrin monomer expression, decreased barrier integrity, increased inflammation, and skin microbiome dysbiosis [62]. This study demonstrated the regulatory role that IL-17RA signaling plays in maintaining the barrier function of uninjured skin and managing the delicate relationship between the skin microbiome and host inflammatory response. Upon tissue injury, IL-17 family members are released in the early phase of inflammation in multiple tissue types [7, 63], including the skin and joints [64, 65].
While the IL-17 family members may initially play a beneficial role in acute wound healing by promoting keratinocyte proliferation and production of antimicrobial peptides [7, 38, 54, 55], unregulated IL-17 family signaling may lead to prolonged inflammation and delayed wound healing. Tanno and colleagues found that deletion of invariant natural killer T cells (iNKTs) delayed healing via increased macrophage inflammatory protein-2 (MIP-2), keratinocyte chemoattractant, and IL-17A production, which increased neutrophil infiltration and decreased neutrophil apoptosis, suggesting that iNKTs act to limit neutrophil recruitment and dwelling to allow proper resolution of the inflammatory phase of healing [66]. In addition to proinflammatory macrophages, dermal Vγ4 T cells produce IL-17A that acts in conjunction with IL-1β on dendritic epidermal T cells to inhibit IGF-1 production, resulting in delayed healing [67]. Therefore, these studies demonstrate that it is imperative to regulate the expression of IL-17A in the healing process.
Persistently elevated levels of IL-17 family members may be associated with wound chronicity. Indeed, studies have shown that levels of the IL-17 family in wound fluid from venous ulcers are elevated relative to the patients' contemporaneously sampled venous blood levels, and the IL-17 family is likewise elevated in lesional tissue samples from patients with pyoderma gangrenosum relative to normal skin [68, 69]. However, neither study examined specific subtypes of the IL-17 family. Another study demonstrated that IL-17C and its receptor are expressed by human keratinocytes from recurrent oral aphthous ulcers, whereas IL-17A and its receptor were not [70]. This finding may indicate the site-specific functional role of different IL-17 subtypes. Interestingly, a recent study found decreased IL-17A in wound fluid from chronic diabetic foot ulcers when compared to acute surgical wounds [71]. However, it is important to consider the difference not only in the timing of the wounds, since IL-17 family expression is increased in the early phase of healing as expected in an acute wound, but also the etiological differences between diabetic and surgical wounds, which may have a confounding influence when comparing these two separate cohorts. Furthermore, other IL-17 subtypes that may have exhibited different patterns were not included in the study.
Animal models have also suggested the pathologic role of the IL-17 family in wound healing (Table 2). The effects may be mediated by cellular responses, specifically that of macrophages and neutrophils. Rodero et al. found that genetic deletions in IL-17A or administration of IL-17A antibody accelerated wound healing in association with increased expression of a prohealing macrophage population [72]. Conversely, recombinant IL-17A (rIL-17A) administration to wild-type wounds produced delayed healing due to increased inflammatory cell infiltration [53]. Consistent with these findings, diabetic mice with IL-23 or IL-17 family gene deletions or blocking antibodies also demonstrated accelerated wound healing in association with alternatively activated prohealing macrophage expression [73]. Yet another study showed that injection of rIL-17, subtype not specified, into excisional wounds increased macrophage infiltration and monocyte chemotactic protein (MCP) production [74], while treatment with the IL-17 production inhibitor Y320 accelerated wound healing at earlier timepoints [74].
Table 2.
Studies suggesting antireparative effects of the IL-17 family.
| Disease model | IL-17 related finding | Ref. |
|---|---|---|
| IL-17A KO mice on C57BL/6 background full-thickness excisional wound healing, males and females | KO mice showed increased myofibroblast numbers and mature collagen with decreased neutrophil infiltration. Delayed healing generated by rIL-17A administration was improved with the addition of neutrophil elastase inhibitor. | [53] |
| Ob/Ob wound healing, males and females | Higher proportion of proinflammatory macrophages to proreparative macrophages in Ob/Ob wounds compared to WT results in delayed wound healing. IL-17A production by proinflammatory macrophages contributed to the delay and was ameliorated with anti-IL-17 antibody. | [72] |
| Ob/Ob wound healing, males and females | Blocking the IL-17A pathway improved wound reepithelialization in Ob/Ob impaired healing model. Further, obese mice with genetic IL-17 family knockout showed reduced proinflammatory macrophages and iNOS but kept proreparative macrophages that express CD206 and LYVE1. | [73] |
| BALB/c full-thickness excisional wound healing, males | Subcutaneous injection of recombinant mouse IL-17 family resulted in increased low-Ly6C macrophage infiltration via proinflammatory mediator levels including MCPs, which produced more type I collagen and delayed wound closure. | [74] |
| BALB/c bone healing, females | Drill injury to the femur in normal and OVX mice showed inhibited healing with anti-IL-17 treatment on day 3 post injury. However, this effect was reversed on days 10 and 21, when groups treated with anti-IL-17 had better healing, especially OVX mice on day 21 post injury. The impact of the IL-17 family on healing was mediated by decreased osteogenic protein expression and increased oxidative stress at the injury site. | [82] |
| BRONJ | BRONJ lesions showed increased IL-17+ cells and IL-17 family in C57BL/6 mice and humans. Lesions also showed increased M1 macrophages, which was attenuated with anti-IL-17A treatment. Exogenous IL-17, subtype not specified, enhanced M1 phenotype and suppressed M2 signaling in murine and human monocytes cultured under polarizing conditions. | [83] |
KO: knockout; rIL-17: recombinant interleukin-17; Ob/Ob: obese diabetic; WT: wild type; iNOS: inducible nitric oxide synthase; MCP: monocytic chemotactic protein; OVX: osteopenic; BRONJ: bisphosphonate-related osteonecrosis of the jaw.
IL-17A may also modulate collagen formation in the wound. One study demonstrated that IL-17A knockout mice displayed enhanced wound closure and collagen deposition as well as decreased neutrophilic infiltrate when compared to wild-type mice [53]. Interestingly, adding a neutrophil elastase inhibitor to rIL-17A administration ameliorated the detrimental effects of rIL-17A, resulting in wound healing similar to wild-type mice [53]. Therefore, amplified IL-17A expression in the wound environment may promote collagen degradation by stimulating proteases secreted from neutrophils. Matrix metalloproteinase induction by the IL-17 family, alone or synergistically with IL-1α, IL-6, and TNF-α, may also contribute to extracellular matrix degradation in nonhealing wounds [44, 59, 75–78].
Furthermore, wounds in IL-23-deficient mice displayed reduced expression of the IL-17 family, which was subsequently reversed by injecting IL-23 directly into the wound bed [73]. These results highlight the relationship between the IL-17 family and IL-23, implicating the IL-17 family as a downstream molecule of IL-23.
Overall, studies investigating the expression of the IL-17 family in cutaneous wounds are scarce; however, findings of related proinflammatory cytokines that induce or synergize with the IL-17 family, such as TNF-α, IL-1, IL-6, and IL-23, in chronic wounds of different etiologies wounds may implicate the IL-17 family in the pathogenesis of these wounds [68, 69, 79–81].
3.2. In Connective Tissue
In addition to cutaneous wounds, the IL-17 family has been implicated in other types of degenerative tissue pathology. The IL-17 family was markedly expressed in synovial fluid from idiopathic juvenile arthritis patients and was positively correlated with disease severity [78]. Similarly, IL-17A and IL-17F were detected in synovial tissue from rheumatoid arthritis patients [58]. IL-17A induced a stronger response than IL-17F in downstream cytokine production in the synovium, additionally displaying synergism with TNF-α [58]. The stronger response from IL-17A suggests greater ligand affinity and/or efficacy than IL-17F upon binding its receptor and a potentially more effective therapeutic target than IL-17F. In a murine cortical bone defect model, blocking IL-17A improved healing even better than blocking the known bone growth regulator, the receptor activator of nuclear factor kappa-B ligand (RANKL) (Table 2) [82]. This study importantly highlights the time-dependent effects of IL-17A on tissue repair, as blocking IL-17A initially reduced bone volume three days post injury yet was associated with enhanced bone healing by days 10 and 21. Another study demonstrated increased IL-17A in the oral mucosa at tooth extraction sites of nonhealing osteonecrotic tissue and in serum of humans and mice associated with proinflammatory M1 macrophage phenotypic shift and the protective effects of blocking the IL-17 family associated with prohealing M2 macrophage phenotype in mice (Table 2) [83].
3.3. In Intestinal Tissue
Additionally, the IL-17 family appears to play a protective role in intestinal epithelium tissue repair [84–86]. Indeed, the increased incidence of inflammatory bowel disease (IBD) has been reported following treatment with secukinumab, ixekizumab, and brodalumab in psoriasis and ankylosing spondylitis patients, although causation has not been confirmed [87, 88]. Therefore, therapeutic agents targeting the IL-17 family in chronic wounds may not be appropriate in patients with IBD. The protective effects of the IL-17 family in the gut may parallel its protective effects in the skin by mediating the microbiome and modulating inflammation [89]. Inhibiting these similarly protective effects in the skin with anti-IL17 monoclonal antibody alone may increase the risk of delayed wound healing due to skin dysbiosis and an increased risk for wound infection. Thus, it may be prudent to use topical antimicrobial agents adjunctively with anti-IL-17 monoclonal antibody for chronic wounds that have failed to heal with standard of care alone. Clinical trials would be necessary to determine the appropriate management.
3.4. Proreparative Effects
Contrary to the reported antireparative effects of the IL-17 family, proreparative effects have been suggested in two studies (Table 3). Indeed, anti-IL-17 antibody administration in rats with medial collateral ligament injury did not improve healing but rather resulted in proinflammatory cytokine production, increased T lymphocytes, and decreased prohealing macrophage phenotype expression [90]. The experimental model consisted of male rats at an unspecified age range, not accounting for the effects of aging and sexual dimorphism on wound healing and TH17/IL-17 expression or function [91–99]. Furthermore, the data was collected at only one timepoint, seven days post injury, limiting examination of the kinetics of specific IL-17 subtypes. Additionally, the effects of the IL-17 family in ligamentous injury may differ from cutaneous wounds due to histological differences. Proreparative effects of IL-17A in cutaneous wounds were suggested when Macleod et al. demonstrated that pharmacological blocking or genetic deletions of IL-17A resulted in delayed wound closure in mice, in contrast with findings reported by other aforementioned animal studies on cutaneous wounds [38]. Administering rIL-17A improved wound healing in IL-17A−/− mice, highlighting the beneficial role of IL-17A in the early stages of healing [38]. The inconsistent findings may indicate different roles of IL-17A in different age groups or genders, as Macleod et al. did not account for either. Time of administration relative to the stage of healing may also skew results. As age and gender may influence wound healing and TH17/IL-17 expression or function, it is imperative for studies to match subjects by these confounding variables and assess differences among groups.
Table 3.
Studies suggesting proreparative effects of the IL-17 family.
| Disease model | IL-17 related finding | Ref. |
|---|---|---|
| C57BL/6, Tcrd−/−, IL-17A−/− full-thickness excisional wound healing | Found an IL-17A producing subset of dendritic epidermal T cells that activate after skin injury. Further showed in vitro that exogenous IL-17A induced keratinocytes to produce antimicrobial peptides important for wound healing. | [38] |
| Rat ligament healing, males | IL-17, subtype not specified, neutralizing antibody treatment decreased M2 macrophage numbers and increased T cell-activating cytokines (IL-2, IL-6, and IL-12) as well as T cell numbers. Type I collagen levels were decreased but no changes were found in the wound area or length. | [90] |
IL: interleukin.
4. Adverse Events of Targeting the IL-17 Family
If clinical trials targeting the IL-17 family were to be performed for chronic wound management, then it would be prudent to consider previously reported adverse events. In addition to the increased incidence of IBD that we have discussed, other adverse events that have been reported at relatively low incidence with targeting the IL-17 family include neutropenia, noncutaneous infection, and depression with or without suicidality, although causality of the latter has been recently disproven [100–102]. Since chronic skin wounds often display increased relative abundance of pathogenic bacteria Staphylococcus aureus and Pseudomonas aeruginosa in wound tissue, neutropenia and potential dissemination of infection are of particular concern [103–105]. Thus, monitoring for neutropenia in wound patients receiving monoclonal IL-17 antibody therapy may be warranted.
5. Conclusion
The search for chronic wound biomarkers and therapeutic targets is critical due to the growing numbers of patients suffering from this ailment. The role of the IL-17 family members in cutaneous disease indicated its potential involvement in the pathogenesis of chronic wounds. In our review of the literature, we found animal studies demonstrating a proreparative role for IL-17A in cutaneous wound healing in tissue homeostasis and in response to acute wound healing. However, the IL-17 family appears to play a role in the pathogenesis of chronic wounds (summarized in Figure 1). Studies in animal wound models with age-matched and gender-matched groups suggest a therapeutic role for targeting IL-17A to promote wound healing. The limited number of human studies prompts the need for assessing IL-17 family expression in a greater number of subjects with wounds of various etiological types (i.e., chronic venous ulcer, chronic diabetic ulcer, nonhealing surgical wound, and chronic burn injury). Clinical trials of monoclonal IL-17 antibody have yet to be initiated and the possibility of disseminated infection in the context of an increased microbial load from chronic wounds considered. Furthermore, expanded information on the signaling activities and biological function of each IL-17 subtype could pave the road to therapeutic interventions selectively targeting specific subtypes in order to optimize wound healing.
Acknowledgments
We thank Daniel Hsu for assisting with the preparation of Figure 1.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Sen C. K. Human wounds and its burden: an updated compendium of estimates. Advances in Wound Care. 2019;8(2):39–48. doi: 10.1089/wound.2019.0946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Agency for Healthcare Resaerch and Quality. Usual care in the management of chronic wounds: a reive of the recent literature. 2005. December 2018, https://www.cms.gov/Medicare/Coverage/DeterminationProcess/downloads/id37TA.pdf. [PubMed]
- 3.Morton L. M., Phillips T. J. Wound healing and treating wounds: differential diagnosis and evaluation of chronic wounds. Journal of the American Academy of Dermatology. 2016;74(4):589–605. doi: 10.1016/j.jaad.2015.08.068. [DOI] [PubMed] [Google Scholar]
- 4.Raja K. S., Garcia M. S., Isseroff R. R. Wound re-epithelialization: modulating kerationcyte migration in wound healing. Frontiers in Bioscience. 2007;12:2849–2868. doi: 10.2741/2277. [DOI] [PubMed] [Google Scholar]
- 5.Rousselle P., Braye F., Dayan G. Re-epithelialization of adult skin wounds: cellular mechanisms and therapeutic strategies. Advanced Drug Delivery Reviews. 2019;146:344–365. doi: 10.1016/j.addr.2018.06.019. [DOI] [PubMed] [Google Scholar]
- 6.Beringer A., Noack M., Miossec P. IL-17 in chronic inflammation: from discovery to targeting. Trends in Molecular Medicine. 2016;22(3):230–241. doi: 10.1016/j.molmed.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 7.Isailovic N., Daigo K., Mantovani A., Selmi C. Interleukin-17 and innate immunity in infections and chronic inflammation. Journal of Autoimmunity. 2015;60:1–11. doi: 10.1016/j.jaut.2015.04.006. [DOI] [PubMed] [Google Scholar]
- 8.Speeckaert R., Lambert J., Grine L., van Gele M., de Schepper S., van Geel N. The many faces of interleukin‐17 in inflammatory skin diseases. British Journal of Dermatology. 2016;175(5):892–901. doi: 10.1111/bjd.14703. [DOI] [PubMed] [Google Scholar]
- 9.Monin L., Gaffen S. L. Interleukin 17 family cytokines: signaling mechanisms, biological activities, and therapeutic implications. Cold Spring Harbor Perspectives in Biology. 2018;10(4, article a028522) doi: 10.1101/cshperspect.a028522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lowes M. A., Russell C. B., Martin D. A., Towne J. E., Krueger J. G. The IL-23/T17 pathogenic axis in psoriasis is amplified by keratinocyte responses. Trends in Immunology. 2013;34(4):174–181. doi: 10.1016/j.it.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lowes M. A., Kikuchi T., Fuentes-Duculan J., et al. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. The Journal of Investigative Dermatology. 2008;128(5):1207–1211. doi: 10.1038/sj.jid.5701213. [DOI] [PubMed] [Google Scholar]
- 12.Johansen C., Usher P. A., Kjellerup R. B., Lundsgaard D., Iversen L., Kragballe K. Characterization of the interleukin‐17 isoforms and receptors in lesional psoriatic skin. The British Journal of Dermatology. 2009;160(2):319–324. doi: 10.1111/j.1365-2133.2008.08902.x. [DOI] [PubMed] [Google Scholar]
- 13.Spuls P. I., Hooft L. Brodalumab and ixekizumab, anti‐interleukin‐17‐receptor antibodies for psoriasis: a critical appraisal. British Journal of Dermatology. 2012;167(4):710–713. doi: 10.1111/bjd.12025. [DOI] [PubMed] [Google Scholar]
- 14.Mease P. J., Genovese M. C., Greenwald M. W., et al. Brodalumab, an anti-IL17RA monoclonal antibody, in psoriatic arthritis. The New England Journal of Medicine. 2014;370(24):2295–2306. doi: 10.1056/NEJMoa1315231. [DOI] [PubMed] [Google Scholar]
- 15.Marzo-Ortega H., Sieper J., Kivitz A., et al. Secukinumab provides sustained improvements in the signs and symptoms of active ankylosing spondylitis with high retention rate: 3-year results from the phase III trial, MEASURE 2. RMD Open. 2017;3(2, article e000592) doi: 10.1136/rmdopen-2017-000592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chiricozzi A., Krueger J. G. IL-17 targeted therapies for psoriasis. Expert Opinion on Investigational Drugs. 2013;22(8):993–1005. doi: 10.1517/13543784.2013.806483. [DOI] [PubMed] [Google Scholar]
- 17.Moran B., Sweeney C. M., Hughes R., et al. Hidradenitis suppurativa is characterized by dysregulation of the Th17:Treg cell axis, which is corrected by anti-TNF therapy. The Journal of Investigative Dermatology. 2017;137(11):2389–2395. doi: 10.1016/j.jid.2017.05.033. [DOI] [PubMed] [Google Scholar]
- 18.Thomi R., Cazzaniga S., Seyed Jafari S. M., Schlapbach C., Hunger R. E. Association of hidradenitis suppurativa with T helper 1/T helper 17 phenotypes: a semantic map analysis. JAMA Dermatology. 2018;154(5):592–595. doi: 10.1001/jamadermatol.2018.0141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hymowitz S. G., Filvaroff E. H., Yin J. P., et al. IL-17s adopt a cystine knot fold: structure and activity of a novel cytokine, IL-17F, and implications for receptor binding. The EMBO Journal. 2001;20(19):5332–5341. doi: 10.1093/emboj/20.19.5332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Veldhoen M., Hocking R. J., Atkins C. J., Locksley R. M., Stockinger B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity. 2006;24(2):179–189. doi: 10.1016/j.immuni.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 21.Bettelli E., Carrier Y., Gao W., et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441(7090):235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 22.Zhou L., Ivanov I. I., Spolski R., et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nature Immunology. 2007;8(9):967–974. doi: 10.1038/ni1488. [DOI] [PubMed] [Google Scholar]
- 23.Awasthi A., Riol-Blanco L., Jäger A., et al. Cutting edge: IL-23 receptor gfp reporter mice reveal distinct populations of IL-17-producing cells. Journal of Immunology. 2009;182(10):5904–5908. doi: 10.4049/jimmunol.0900732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mangan P. R., Harrington L. E., O'Quinn D. B., et al. Transforming growth factor-β induces development of the TH17 lineage. Nature. 2006;441(7090):231–234. doi: 10.1038/nature04754. [DOI] [PubMed] [Google Scholar]
- 25.Huber M., Heink S., Pagenstecher A., et al. IL-17A secretion by CD8+ T cells supports Th17-mediated autoimmune encephalomyelitis. The Journal of Clinical Investigation. 2013;123(1):247–260. doi: 10.1172/JCI63681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McGeachy M. J., Chen Y., Tato C. M., et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nature Immunology. 2009;10(3):314–324. doi: 10.1038/ni.1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.He D., Wu L., Kim H. K., Li H., Elmets C. A., Xu H. CD8+ IL-17-producing T cells are important in effector functions for the elicitation of contact hypersensitivity responses. Journal of Immunology. 2006;177(10):6852–6858. doi: 10.4049/jimmunol.177.10.6852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cua D. J., Tato C. M. Innate IL-17-producing cells: the sentinels of the immune system. Nature Reviews Immunology. 2010;10(7):479–489. doi: 10.1038/nri2800. [DOI] [PubMed] [Google Scholar]
- 29.Villanova F., Flutter B., Tosi I., et al. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44+ ILC3 in psoriasis. The Journal of Investigative Dermatology. 2014;134(4):984–991. doi: 10.1038/jid.2013.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Marks B. R., Nowyhed H. N., Choi J. Y., et al. Thymic self-reactivity selects natural interleukin 17-producing T cells that can regulate peripheral inflammation. Nature Immunology. 2009;10(10):1125–1132. doi: 10.1038/ni.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kronenberg M. Toward an understanding of NKT cell biology: progress and paradoxes. Annual Review of Immunology. 2005;23:877–900. doi: 10.1146/annurev.immunol.23.021704.115742. [DOI] [PubMed] [Google Scholar]
- 32.Ramirez-Carrozzi V., Sambandam A., Luis E., et al. IL-17C regulates the innate immune function of epithelial cells in an autocrine manner. Nature Immunology. 2011;12(12):1159–1166. doi: 10.1038/ni.2156. [DOI] [PubMed] [Google Scholar]
- 33.Li L., Huang L., Vergis A. L., et al. IL-17 produced by neutrophils regulates IFN-γ-mediated neutrophil migration in mouse kidney ischemia-reperfusion injury. The Journal of Clinical Investigation. 2010;120(1):331–342. doi: 10.1172/JCI38702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hoshino A., Nagao T., Nagi-Miura N., et al. MPO-ANCA induces IL-17 production by activated neutrophils in vitro via its Fc region- and complement-dependent manner. Journal of Autoimmunity. 2008;31(1):79–89. doi: 10.1016/j.jaut.2008.03.006. [DOI] [PubMed] [Google Scholar]
- 35.Noordenbos T., Blijdorp I., Chen S., et al. Human mast cells capture, store, and release bioactive, exogenous IL‐17A. Journal of Leukocyte Biology. 2016;100(3):453–462. doi: 10.1189/jlb.3HI1215-542R. [DOI] [PubMed] [Google Scholar]
- 36.Lin A. M., Rubin C. J., Khandpur R., et al. Mast cells and neutrophils release IL-17 through extracellular trap formation in psoriasis. Journal of Immunology. 2011;187(1):490–500. doi: 10.4049/jimmunol.1100123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hwang S. Y., Kim H. Y. Expression of IL-17 homologs and their receptors in the synovial cells of rheumatoid arthritis patients. Molecules and Cells. 2005;19(2):180–184. [PubMed] [Google Scholar]
- 38.MacLeod A. S., Hemmers S., Garijo O., et al. Dendritic epidermal T cells regulate skin antimicrobial barrier function. The Journal of Clinical Investigation. 2013;123(10):4364–4374. doi: 10.1172/JCI70064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kleinschek M. A., Owyang A. M., Joyce-Shaikh B., et al. IL-25 regulates Th17 function in autoimmune inflammation. The Journal of Experimental Medicine. 2007;204(1):161–170. doi: 10.1084/jem.20061738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ramirez-Carrozzi V., Ota N., Sambandam A., et al. Cutting edge: IL-17B uses IL-17RA and IL-17RB to induce type 2 inflammation from human lymphocytes. Journal of Immunology. 2019;202(7):1935–1941. doi: 10.4049/jimmunol.1800696. [DOI] [PubMed] [Google Scholar]
- 41.Toy D., Kugler D., Wolfson M., et al. Cutting edge: interleukin 17 signals through a heteromeric receptor complex. The Journal of Immunology. 2006;177(1):36–39. doi: 10.4049/jimmunol.177.1.36. [DOI] [PubMed] [Google Scholar]
- 42.Rickel E. A., Siegel L. A., Yoon B. R., et al. Identification of functional roles for both IL-17RB and IL-17RA in mediating IL-25-induced activities. The Journal of Immunology. 2008;181(6):4299–4310. doi: 10.4049/jimmunol.181.6.4299. [DOI] [PubMed] [Google Scholar]
- 43.Ely L. K., Fischer S., Garcia K. C. Structural basis of receptor sharing by interleukin 17 cytokines. Nature Immunology. 2009;10(12):1245–1251. doi: 10.1038/ni.1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yao Z., Nie L., Zhao Y., et al. Salubrinal suppresses IL-17-induced upregulation of MMP-13 and extracellular matrix degradation through the NF-kB pathway in human nucleus pulposus cells. Inflammation. 2016;39(6):1997–2007. doi: 10.1007/s10753-016-0435-y. [DOI] [PubMed] [Google Scholar]
- 45.Qian Y., Liu C., Hartupee J., et al. The adaptor Act1 is required for interleukin 17-dependent signaling associated with autoimmune and inflammatory disease. Nature Immunology. 2007;8(3):247–256. doi: 10.1038/ni1439. [DOI] [PubMed] [Google Scholar]
- 46.Schwandner R., Yamaguchi K., Cao Z. Requirement of tumor necrosis factor receptor-associated factor (TRAF)6 in interleukin 17 signal transduction. The Journal of Experimental Medicine. 2000;191(7):1233–1240. doi: 10.1084/jem.191.7.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ruddy M. J., Wong G. C., Liu X. K., et al. Functional cooperation between interleukin-17 and tumor necrosis Factor-α is mediated by CCAAT/enhancer-binding protein family members. The Journal of Biological Chemistry. 2004;279(4):2559–2567. doi: 10.1074/jbc.M308809200. [DOI] [PubMed] [Google Scholar]
- 48.Song X., He X., Li X., Qian Y. The roles and functional mechanisms of interleukin-17 family cytokines in mucosal immunity. Cellular & Molecular Immunology. 2016;13(4):418–431. doi: 10.1038/cmi.2015.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Song X., Zhu S., Shi P., et al. IL-17RE is the functional receptor for IL-17C and mediates mucosal immunity to infection with intestinal pathogens. Nature Immunology. 2011;12(12):1151–1158. doi: 10.1038/ni.2155. [DOI] [PubMed] [Google Scholar]
- 50.Onishi R. M., Gaffen S. L. Interleukin‐17 and its target genes: mechanisms of interleukin‐17 function in disease. Immunology. 2010;129(3):311–321. doi: 10.1111/j.1365-2567.2009.03240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jovanovic D. V., Di Battista J. A., Martel-Pelletier J., et al. IL-17 stimulates the production and expression of proinflammatory cytokines, IL-β and TNF-α, by human macrophages. Journal of Immunology. 1998;160(7):3513–3521. [PubMed] [Google Scholar]
- 52.Goldberg M., Nadiv O., Luknar-Gabor N., Agar G., Beer Y., Katz Y. Synergism between tumor necrosis factor alpha and interleukin-17 to induce IL-23 p19 expression in fibroblast-like synoviocytes. Molecular Immunology. 2009;46(8-9):1854–1859. doi: 10.1016/j.molimm.2009.01.004. [DOI] [PubMed] [Google Scholar]
- 53.Takagi N., Kawakami K., Kanno E., et al. IL‐17A promotes neutrophilic inflammation and disturbs acute wound healing in skin. Experimental Dermatology. 2017;26(2):137–144. doi: 10.1111/exd.13115. [DOI] [PubMed] [Google Scholar]
- 54.Wu L., Chen X., Zhao J., et al. A novel IL-17 signaling pathway controlling keratinocyte proliferation and tumorigenesis via the TRAF4-ERK5 axis. The Journal of Experimental Medicine. 2015;212(10):1571–1587. doi: 10.1084/jem.20150204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nograles K. E., Zaba L. C., Guttman-Yassky E., et al. Th17 cytokines interleukin (IL)‐17 and IL‐22 modulate distinct inflammatory and keratinocyte‐response pathways. The British Journal of Dermatology. 2008;159(5):1092–1102. doi: 10.1111/j.1365-2133.2008.08769.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shen F., Ruddy M. J., Plamondon P., Gaffen S. L. Cytokines link osteoblasts and inflammation: microarray analysis of interleukin‐17‐ and TNF‐α‐induced genes in bone cells. Journal of Leukocyte Biology. 2005;77(3):388–399. doi: 10.1189/jlb.0904490. [DOI] [PubMed] [Google Scholar]
- 57.Hartupee J., Liu C., Novotny M., Li X., Hamilton T. IL-17 enhances chemokine gene expression through mRNA stabilization. The Journal of Immunology. 2007;179(6):4135–4141. doi: 10.4049/jimmunol.179.6.4135. [DOI] [PubMed] [Google Scholar]
- 58.Zrioual S., Ecochard R., Tournadre A., Lenief V., Cazalis M. A., Miossec P. Genome-wide comparison between IL-17A- and IL-17F-induced effects in human rheumatoid arthritis synoviocytes. The Journal of Immunology. 2009;182(5):3112–3120. doi: 10.4049/jimmunol.0801967. [DOI] [PubMed] [Google Scholar]
- 59.Koshy P. J., Henderson N., Logan C., Life P. F., Cawston T. E., Rowan A. D. Interleukin 17 induces cartilage collagen breakdown: novel synergistic effects in combination with proinflammatory cytokines. Annals of the Rheumatic Diseases. 2002;61(8):704–713. doi: 10.1136/ard.61.8.704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bie Q., Jin C., Zhang B., Dong H. IL-17B: a new area of study in the IL-17 family. Molecular Immunology. 2017;90:50–56. doi: 10.1016/j.molimm.2017.07.004. [DOI] [PubMed] [Google Scholar]
- 61.Starnes T., Broxmeyer H. E., Robertson M. J., Hromas R. Cutting edge: IL-17D, a novel member of the IL-17 family, stimulates cytokine production and inhibits hemopoiesis. Journal of Immunology. 2002;169(2):642–646. doi: 10.4049/jimmunol.169.2.642. [DOI] [PubMed] [Google Scholar]
- 62.Floudas A., Saunders S. P., Moran T., et al. IL-17 receptor a maintains and protects the skin barrier to prevent allergic skin inflammation. Journal of Immunology. 2017;199(2):707–717. doi: 10.4049/jimmunol.1602185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.MacLeod A. S., Mansbridge J. N. The innate immune system in acute and chronic wounds. Advances in Wound Care. 2016;5(2):65–78. doi: 10.1089/wound.2014.0608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Godoy-Santos A. L., Ranzoni L., Teodoro W. R., et al. Increased cytokine levels and histological changes in cartilage, synovial cells and synovial fluid after malleolar fractures. Injury. 2017;48:S27–S33. doi: 10.1016/S0020-1383(17)30772-6. [DOI] [PubMed] [Google Scholar]
- 65.Cui W., Yang L. F., Wei W. H., et al. Interleukin-17 expression in murine pressure ulcer tissues. Experimental and Therapeutic Medicine. 2013;5(3):803–806. doi: 10.3892/etm.2013.912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tanno H., Kawakami K., Kanno E., et al. Invariant NKT cells promote skin wound healing by preventing a prolonged neutrophilic inflammatory response. Wound Repair and Regeneration. 2017;25(5):805–815. doi: 10.1111/wrr.12588. [DOI] [PubMed] [Google Scholar]
- 67.Li Y., Wang Y., Zhou L., et al. Vγ4 T cells inhibit the pro-healing functions of dendritic epidermal T cells to delay skin wound closure through IL-17A. Frontiers in Immunology. 2018;9:p. 240. doi: 10.3389/fimmu.2018.00240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ligi D., Mosti G., Croce L., Raffetto J. D., Mannello F. Chronic venous disease - part I: inflammatory biomarkers in wound healing. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. 2016;1862(10):1964–1974. doi: 10.1016/j.bbadis.2016.07.018. [DOI] [PubMed] [Google Scholar]
- 69.Marzano A. V., Damiani G., Ceccherini I., Berti E., Gattorno M., Cugno M. Autoinflammation in pyoderma gangrenosum and its syndromic form (pyoderma gangrenosum, acne and suppurative hidradenitis) The British Journal of Dermatology. 2017;176(6):1588–1598. doi: 10.1111/bjd.15226. [DOI] [PubMed] [Google Scholar]
- 70.Al‐Samadi A., Kouri V.‐. P., Salem A., et al. IL‐17C and its receptor IL‐17RA/IL‐17RE identify human oral epithelial cell as an inflammatory cell in recurrent aphthous ulcer. Journal of Oral Pathology & Medicine. 2014;43(2):117–124. doi: 10.1111/jop.12095. [DOI] [PubMed] [Google Scholar]
- 71.Bekeschus S., Schmidt A., Napp M., et al. Distinct cytokine and chemokine patterns in chronic diabetic ulcers and acute wounds. Experimental Dermatology. 2017;26(2):145–147. doi: 10.1111/exd.13215. [DOI] [PubMed] [Google Scholar]
- 72.Rodero M. P., Hodgson S. S., Hollier B., Combadiere C., Khosrotehrani K. Reduced Il17a expression distinguishes a Ly6cloMHCIIhi macrophage population promoting wound healing. The Journal of Investigative Dermatology. 2013;133(3):783–792. doi: 10.1038/jid.2012.368. [DOI] [PubMed] [Google Scholar]
- 73.Lee J., Rodero M. P., Patel J., Moi D., Mazzieri R., Khosrotehrani K. Interleukin-23 regulates interleukin-17 expression in wounds, and its inhibition accelerates diabetic wound healing through the alteration of macrophage polarization. The FASEB Journal. 2018;32(4):2086–2094. doi: 10.1096/fj.201700773R. [DOI] [PubMed] [Google Scholar]
- 74.Zhang J., Qiao Q., Liu M., et al. IL-17 promotes scar formation by inducing macrophage infiltration. The American Journal of Pathology. 2018;188(7):1693–1702. doi: 10.1016/j.ajpath.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 75.Schminke B., Trautmann S., Mai B., Miosge N., Blaschke S. Interleukin 17 inhibits progenitor cells in rheumatoid arthritis cartilage. European Journal of Immunology. 2016;46(2):440–445. doi: 10.1002/eji.201545910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wu Y., Zhu L., Wei H., Peng B. Regulation of matrix metalloproteinases, tissue inhibitor of matrix metalloproteinase-1, and extracellular metalloproteinase inducer by interleukin-17 in human periodontal ligament fibroblasts. Journal of Endodontia. 2013;39(1):62–67. doi: 10.1016/j.joen.2012.09.025. [DOI] [PubMed] [Google Scholar]
- 77.Shibata M., Shintaku Y., Matsuzaki K., Uematsu S. The effect of IL‐17 on the production of proinflammatory cytokines and matrix metalloproteinase‐1 by human periodontal ligament fibroblasts. Orthodontics & Craniofacial Research. 2014;17(1):60–68. doi: 10.1111/ocr.12033. [DOI] [PubMed] [Google Scholar]
- 78.Agarwal S., Misra R., Aggarwal A. Interleukin 17 levels are increased in juvenile idiopathic arthritis synovial fluid and induce synovial fibroblasts to produce proinflammatory cytokines and matrix metalloproteinases. The Journal of Rheumatology. 2008;35(3):515–519. [PubMed] [Google Scholar]
- 79.Guenova E., Teske A., Fehrenbacher B., et al. Interleukin 23 expression in pyoderma gangrenosum and targeted therapy with ustekinumab. Archives of Dermatology. 2011;147(10):1203–1205. doi: 10.1001/archdermatol.2011.168. [DOI] [PubMed] [Google Scholar]
- 80.Nagaraja S., Wallqvist A., Reifman J., Mitrophanov A. Y. Computational approach to characterize causative factors and molecular indicators of chronic wound inflammation. Journal of Immunology. 2014;192(4):1824–1834. doi: 10.4049/jimmunol.1302481. [DOI] [PubMed] [Google Scholar]
- 81.Liu T., Yang F., Li Z., Yi C., Bai X. A prospective pilot study to evaluate wound outcomes and levels of serum C-reactive protein and interleukin-6 in the wound fluid of patients with trauma-related chronic wounds. Ostomy/Wound Management. 2014;60(6):30–37. [PubMed] [Google Scholar]
- 82.Dixit M., Singh K. B., Prakash R., Singh D. Functional block of IL-17 cytokine promotes bone healing by augmenting FOXO1 and ATF4 activity in cortical bone defect model. Osteoporosis International. 2017;28(7):2207–2220. doi: 10.1007/s00198-017-4012-5. [DOI] [PubMed] [Google Scholar]
- 83.Zhang Q., Atsuta I., Liu S., et al. IL-17-mediated M1/M2 macrophage alteration contributes to pathogenesis of bisphosphonate-related osteonecrosis of the jaws. Clinical Cancer Research. 2013;19(12):3176–3188. doi: 10.1158/1078-0432.CCR-13-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nishikawa K., Seo N., Torii M., et al. Interleukin-17 induces an atypical M2-like macrophage subpopulation that regulates intestinal inflammation. PLoS One. 2014;9(9, article e108494) doi: 10.1371/journal.pone.0108494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zepp J. A., Zhao J., Liu C., et al. IL-17A-induced PLET1 expression contributes to tissue repair and colon tumorigenesis. Journal of Immunology. 2017;199(11):3849–3857. doi: 10.4049/jimmunol.1601540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Song X., Dai D., He X., et al. Growth factor FGF2 cooperates with interleukin-17 to repair intestinal epithelial damage. Immunity. 2015;43(3):488–501. doi: 10.1016/j.immuni.2015.06.024. [DOI] [PubMed] [Google Scholar]
- 87.Whitlock S. M., Enos C. W., Armstrong A. W., et al. Management of psoriasis in patients with inflammatory bowel disease: from the Medical Board of the National Psoriasis Foundation. Journal of the American Academy of Dermatology. 2018;78(2):383–394. doi: 10.1016/j.jaad.2017.06.043. [DOI] [PubMed] [Google Scholar]
- 88.Lozano M. J. F., Giménez R. S., Fernández M. C. Emergence of inflammatory bowel disease during treatment with secukinumab. Journal of Crohn's and Colitis. 2018;12 doi: 10.1093/ecco-jcc/jjy063. [DOI] [PubMed] [Google Scholar]
- 89.Kumar P., Monin L., Castillo P., et al. Intestinal interleukin-17 receptor signaling mediates reciprocal control of the gut microbiota and autoimmune inflammation. Immunity. 2016;44(3):659–671. doi: 10.1016/j.immuni.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Clements A. E., Chamberlain C. S., Leiferman E. M., Murphy W. L., Vanderby R. Impacts of interleukin-17 neutralization on the inflammatory response in a healing ligament. Journal of Cytokine Biology. 2017;2(1) doi: 10.4172/2576-3881.1000113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kim D. J., Mustoe T., Clark R. A. Cutaneous wound healing in aging small mammals: a systematic review. Wound Repair and Regeneration. 2015;23(3):318–339. doi: 10.1111/wrr.12290. [DOI] [PubMed] [Google Scholar]
- 92.Gilliver S. C., Ruckshanthi J. P., Hardman M. J., Nakayama T., Ashcroft G. S. Sex dimorphism in wound healing: the roles of sex steroids and macrophage migration inhibitory factor. Endocrinology. 2008;149(11):5747–5757. doi: 10.1210/en.2008-0355. [DOI] [PubMed] [Google Scholar]
- 93.Ashcroft G. S., Mills S. J., Lei K., et al. Estrogen modulates cutaneous wound healing by downregulating macrophage migration inhibitory factor. The Journal of Clinical Investigation. 2003;111(9):1309–1318. doi: 10.1172/JCI16288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Guo S., Dipietro L. A. Factors affecting wound healing. Journal of Dental Research. 2010;89(3):219–229. doi: 10.1177/0022034509359125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Li Z., Yue Y., Xiong S. Distinct Th17 inductions contribute to the gender bias in CVB3-induced myocarditis. Cardiovascular Pathology. 2013;22(5):373–382. doi: 10.1016/j.carpath.2013.02.004. [DOI] [PubMed] [Google Scholar]
- 96.Yu J. J., Ruddy M. J., Conti H. R., Boonanantanasarn K., Gaffen S. L. The interleukin-17 receptor plays a gender-dependent role in host protection against Porphyromonas gingivalis-induced periodontal bone loss. Infection and Immunity. 2008;76(9):4206–4213. doi: 10.1128/IAI.01209-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Gracey E., Yao Y. C., Green B., et al. Sexual dimorphism in the Th17 signature of ankylosing spondylitis. Arthritis & Rhematology. 2016;68(3):679–689. doi: 10.1002/art.39464. [DOI] [PubMed] [Google Scholar]
- 98.Hoh B. L., Rojas K., Lin L., et al. Estrogen deficiency promotes cerebral aneurysm rupture by upregulation of Th17 cells and interleukin-17A which downregulates E-cadherin. Journal of the American Heart Association. 2018;7(8) doi: 10.1161/JAHA.118.008863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Molnar I., Bohaty I., Somogyine-Vari E. High prevalence of increased interleukin-17A serum levels in postmenopausal estrogen deficiency. Menopause. 2014;21(7):749–752. doi: 10.1097/GME.0000000000000125. [DOI] [PubMed] [Google Scholar]
- 100.Papp K. A., Bachelez H., Blauvelt A., et al. Infections from seven clinical trials of ixekizumab, an anti‐interleukin‐17A monoclonal antibody, in patients with moderate‐to‐severe psoriasis. The British Journal of Dermatology. 2017;177(6):1537–1551. doi: 10.1111/bjd.15723. [DOI] [PubMed] [Google Scholar]
- 101.Pavelka K., Kivitz A., Dokoupilova E., et al. Efficacy, safety, and tolerability of secukinumab in patients with active ankylosing spondylitis: a randomized, double-blind phase 3 study, MEASURE 3. Arthritis Research & Therapy. 2017;19(1):p. 285. doi: 10.1186/s13075-017-1490-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lebwohl M. G., Papp K. A., Marangell L. B., et al. Psychiatric adverse events during treatment with brodalumab: analysis of psoriasis clinical trials. Journal of the American Academy of Dermatology. 2018;78(1):81–89.e5. doi: 10.1016/j.jaad.2017.08.024. [DOI] [PubMed] [Google Scholar]
- 103.Choi Y., Banerjee A., McNish S., et al. Co-occurrence of anaerobes in human chronic wounds. Microbial Ecology. 2019;77(3):808–820. doi: 10.1007/s00248-018-1231-z. [DOI] [PubMed] [Google Scholar]
- 104.James G. A., Swogger E., Wolcott R., et al. Biofilms in chronic wounds. Wound Repair and Regeneration. 2008;16(1):37–44. doi: 10.1111/j.1524-475X.2007.00321.x. [DOI] [PubMed] [Google Scholar]
- 105.Wolcott R. D., Hanson J. D., Rees E. J., et al. Analysis of the chronic wound microbiota of 2,963 patients by 16S rDNA pyrosequencing. Wound Repair and Regeneration. 2016;24(1):163–174. doi: 10.1111/wrr.12370. [DOI] [PubMed] [Google Scholar]