Abstract
Background
Reduced susceptibility of mosquito vectors to currently used insecticides hampers control interventions. Recently pyriproxyfen, an insect growth regulator has been demonstrated to effectively reduce the reproductive potential in vector mosquitoes.
Methods
Pyriproxyfen (PPF), in different concentrations (0.75%, 0.075%, 0.0075%, and 0.00075%) was applied on papers and Indian wild type Aedes aegypti female mosquitoes (N ≥ 20 for each treatment) were exposed onto it as per WHO guidelines, to study the reproductive disruption. PPF concentration on treated papers was quantitatively cross-determined using HPLC method. Reduction in fecundity, fertility and adult emergence in exposed female Ae. aegypti was determined. Abnormal development in ovary and eggs of exposed females was studied microscopically after different time intervals.
Results
Eggs laid, eggs hatched, pupae formed and adults emerged per female exposed in both before blood meal and after blood meal groups declined significantly from lowest to highest concentration of PPF (F ≥ 5.2; p < 0.02). Adult emergence inhibition in females exposed to PPF before and after blood meal groups ranged from 58.8% [OR = 0.18 (95% CI = 0.09–0.36)] to 79.2% [OR = 0.04 (95% CI = 0.02–0.10)] and 64.4% [OR = 0.12 (95% CI = 0.05–0.28)] to 77.1% [OR = 0.05 (95% CI = 0.02–0.14)] respectively in different concentrations. The probit model used suggested that FI50 (50% fertility inhibition) and EI50 (50% emergence inhibition) were 0.002% (p = 0.82) and 0.0001% (p = 0.99) for females exposed before blood meal, while 0.01% (p = 0.63) and <0.0001% (p = 0.98) for the females exposed after blood meal, respectively. The eggs laid by the females exposed to PPF-treated surface showed altered body organization, desegmentation and disoriented abdominal and cervical regions in the developing embryo. Quantification of PPF on impregnated papers showed that it was uniformly distributed throughout the matrix.
Conclusions
The present study has shown that tarsal contact to PPF-treated surface for a small time drastically influenced the fecundity, fertility and adult emergence in Indian wild Ae. aegypti mosquitoes. Results suggest that a certain minimum concentration of PPF through contact exposure can reduce the abundance of vector mosquitoes to a considerable level. The formulations based on combination of PPF and other compatible insecticides may be an impactful approach where susceptible mosquitoes are killed by the insecticide component while resistant mosquitoes are sterilised by PPF.
Author summary
Development of resistance against insecticides has challenged mosquito control programmes globally and prompted the research of alternative options that can complement insecticides. An insect growth regulator, pyriproxyfen (PPF) usage against mosquitoes can effectively reduce the vector population. PPF mainly inhibits the metamorphosis of mosquito larvae into pupae and prevent the adult emergence, therefore it is generally applied in mosquito breeding habitats. PPF has been shown to exhibit delayed residual effect that may impair the reproductive capacity by affecting the survival, fecundity and fertility of adult mosquito exposed through tarsal contact. Presently, the effectiveness of different concentrations of PPF intended to be delivered through contact have been evaluated against dengue vector Ae. aegypti. Results suggested that very low PPF concentration treated surfaces drastically reduce the fecundity, fertility and adult emergence in mosquitoes. Study suggests that control interventions based on PPF-treated surfaces could provide an additional route to target mosquito vector control by overall population density reduction.
Introduction
Synthetic pyrethroids are currently WHO recommended and widely preferred insecticides in mosquito control due to their effectiveness and strong excito-repellency against various insect vectors. Primarily, the use of Long Lasting Insecticidal Nets (LLINs), Indoor Residual Spray (IRS) and other similar measures have substantially reduced many mosquito borne diseases in the endemic countries [1, 2]. However, the reduced mosquito susceptibility to recommended insecticide groups in different endemic regions may seriously deter the impact of control interventions using these insecticides [3, 4]. Additionally, the change in vectors biting pattern where mosquitoes adapt themselves to bite at the time when host is not using LLIN and sometime shifting their resting places to areas where IRS is not done, has further raised the concern for effective interventions [5, 6].
Ae. aegypti is an important vector of dengue and known to spread majority of dengue fever cases annually. Reports suggest that, although dengue fever is one of the major concerns in entire WHO region, but about 75% of the population at risk of dengue infection lives in Asia-Pacific Region [7]. The efforts to control dengue vectors mainly rely on the use of insecticides and reducing the breeding habitats, nevertheless dengue vectors are regularly invading into the new areas. Dengue vectors primarily bite during the day time therefore protection methods such as use of LLINs may not be equally effective against these vectors. Furthermore, widespread prevalence of resistance against the commonly used larvicides and application of ineffective methods or ineffective delivery of control methods could undermine the interventions that intend to reduce dengue vectors abundance [4, 8]. Hence efforts aiming at dengue vectors control have not been able to significantly reduce the dengue fever incidences in endemic countries. Consequently, strategies including the use of alternative options that could complement the existing intervention efforts need to be developed and employed in the vector mosquito control.
Pyriproxyfen (4-phenoxyphenyl (RS)-2-(2-pyridyloxy) propyl ether) is a pyridine based juvenile hormone analogue that acts as insect growth regulator (IGR). Among insects, the hormonal activity interferes in the normal process of embryogenesis and metamorphosis to inhibit many important processes including reproduction and development. The developmental changes in insects take place through moulting, which continues through the egg, larvae and pupae stages and before ending in the adult stage. Pyriproxyfen (PPF) is mainly involved in inhibiting the emergence of adult from pupal stage, however it has also been found associated in interfering different developmental stages in the insects [7–11]. Some recent studies have shown that use of PPF against dengue vector larvae efficiently reduced the dengue incidences in the intervention population [12]. Unlike the insecticides that are commonly used to kill the insects, PPF is not recognized as an adulticide and generally used in mosquito breeding sites for the control of mosquito vectors [11, 13]. Nevertheless, many studies have highlighted its role in reducing the longevity of the exposed insects [14–16]. Ohashi et al. [14] has shown that PPF treated net exposure decreased the survival rate of exposed Anopheles mosquitoes. This study further mentions that PPF exposed mosquitoes died within 8 days after exposure.
Nevertheless PPF is not well appreciated as mosquito adulticide, yet many studies have demonstrated that PPF elicit delayed residual effect that impairs the insect reproductive capacity by affecting the fecundity and fertility of adult mosquitoes exposed through tarsal contact. This effect may depend on the dose and time of exposure with respect to the blood meal. Ohba et al. [17] showed that exposure of female Ae. albopictus to bed net treated with 350 mg/m2 and 35 mg/m2 PPF effectively reduced the fecundity and egg hatchability. The study further suggested the auto-dissemination of PPF into breeding sites by adult mosquitoes through contact with PPF treated bed nets [17]. In An. gambiae and Culex quinquefasciatus adult females exposed to 2.6 mg/m2 PPF caused significant sterilizing effect between 24 hr before and after blood meal [18]. Koama et al. [19] studied that adult females An. gambiae exposed to 1% treated PPF net before and after blood meal resulted in nearly complete inhibition of fecundity and fertility. Furthermore, PPF has been found to be effective against the mosquitoes that have developed resistance to various insecticides [14, 20–23]. Organophosphate (OP) resistant strain of Cx. quinquefasciatus exposed to PPF in insecticide paint Inesfly 5A IGR caused reduction in fecundity, fertility and adult emergence [24]. PPF is safe to the non-target organisms and categorized in WHO list of chemicals that are unlikely to produce hazardous effect in normal use [25].
Present study was undertaken to evaluate the impact of PPF adsorbed surface exposure on Indian wild type Ae. aegypti at different concentrations to determine the minimum concentration of PPF on surface that could be effective in mosquito control.
Material and methods
Mosquitoes
Indian wild type Ae. aegypti larvae were collected from natural dwelling locations in field from Gwalior, Madhya Pradesh, India and maintained in the insectary of Vector Management Division, Defence Research and Development Establishment, Gwalior, India. Larvae were kept in disposable cups (100 ml capacity) in gauge wooden cages (size: 750mmX600mmX600mm) and given commercially available dog biscuits and yeast granules as food. The mosquito culture was maintained at 27±2°C temperature, 75±5% relative humidity (RH) and 12:12 [light:dark] hr. Cotton soaked with 10% sugar solution was provided ad-libitum to the adults for nourishment and the female mosquitoes were fed on rabbits for blood meal. Females of F1 population of the collected Ae. aegypti mosquitoes were used in the present study.
Preparation of PPF treated papers and mosquito exposure
Four different concentrations of PPF (0.75%, 0.075%, 0.0075%, and 0.00075%) were prepared by diluting PPF (98.2% purity) in HPLC grade acetonitrile (Merck, India) and treatment on paper was done following standard method. The amount of PPF in 0.75% concentration was 4.95 mg which gives 275 mg/m2 concentration of active ingredient. The papers after treatment were left to air dry for 30 min at room temperature. Control papers were prepared using acetonitrile only. The impregnated papers used in the present study were prepared using 12 × 15 cms Whatman No. 1 filter papers (thickness—180 μm; pore size—11 μm). The experiments were carried out in batches of 5–7 females for each treatment (N ≥ 20). Fresh impregnated paper was used for every batch in each concentration. Female mosquitoes (4–5 days old) were exposed to impregnated and control papers in WHO bioassay test kits for 30 min and thereafter transferred to holding tubes. One cohort of mosquitoes was exposed to different concentrations 24 hr before blood meal, whereas a separate cohort was exposed 24 hr after the blood meal. Once both exposure and blood feed were complete, the females were maintained in PPF free cages containing non-chlorinated tap water filled cups lined with filter papers for oviposition. Total eggs laid by each batch in subsequent five days were recorded. The eggs that failed to hatch after seven days were treated as dead [26]. The effect of exposure was observed in terms of fecundity, fertility, pupae formation and adult emergence.
Estimation of PPF by HPLC
Isocratic elution was performed by a mixture of HPLC grade acetonitrile (Merck, India) and water (Elix-MiliQ; filtered through 0.22 μM membrane filter) as mobile phase at 80:20 (v/v) (Waters 1525 binary HPLC pump; flow rate of 1 ml/min). Chromatographic separation was performed and signals were recorded at 280 nm (Waters XTerra C18 column 4.6 x 250 mm; 5μm; 2487 UV detector). Different concentration solutions of PPF (stock as well as working standards) were prepared in acetonitrile and injected through a Rheodyne injector fitted with 10 μL loop.
The PPF in impregnated papers was extracted in acetonitrile and estimated. Briefly, each paper (either impregnated or control) of area 180 cm2 was cut into four equal parts (45 cm2) and thereafter each part was cut into small pieces and extracted with acetonitrile (2 x 5 ml) in a 25 ml conical flask under sonication for 5 min. Resulting solution containing PPF was transferred to a 10 ml volumetric flask followed by volume make up with acetonitrile. Three samples were prepared for each part of paper. Each sample was used for HPLC and PPF content was calculated by using following equation:
Where [PPF] is concentration of PPF in mg/ml; Astandard and Asample are peak areas of PPF in standard and sample solution respectively; P is the purity of standard PPF. Multiplication factor 4 was used because a quarter of each paper was used for sample preparation.
Fertility and fecundity assessment
Average number of eggs laid by single female was determined for control as well as for all the concentrations. Inhibition (%) in fecundity was calculated as reduction in number of eggs laid per female for a given concentration relative to the control using following calculation;
where Lc is average number of eggs laid per female in the control, while Lt in a given treatment.
Similarly, average number of eggs hatched per female in control and treatment was used to determine the fertility inhibition, as follows;
where Hc and Ht are average number of eggs hatched per female in the control and a given treatment respectively.
Furthermore, the percentage reduction in overall adult emergence per female in treatment relative to the control was determined using following equation;
where Ec and Et are the reproductive rates represented by the average number of adult emerged per female in control and a given treatment respectively.
Ovary development
Developmental stages of ovaries in exposed as well as control female Ae. aegypti were observed after dissecting the mosquitoes used in the study. Altogether, 20 females from control, while 30 females from each exposure concentration were dissected under microscope (Leica, EZ4). Females were anesthetized at −20°C for 8–10 min and individually dissected in a drop of distilled water by gently pulling out the last abdominal segment. The extracted ovaries were studied under different magnifications of microscope (Leica, MZ 205A).
Statistical analysis
Mean outcomes such as eggs laid, egg hatched, pupae formed and offspring emerged for control and treatments were compared using analysis of variance (ANOVA) followed by Tukey Kramer test of multiple comparison. Student’s ‘t’ test was used for comparison between the control and a given treatment. Percentage inhibition outcomes were subjected to probit analysis using log-probit method (Ldp Line software, Ehabsoft, Egypt) to find inhibitory concentrations for egg laid, egg hatched and offspring emerged. Chi square (χ2) test was used to assess the fitment of probit. The overall significance has been assessed using Bonferroni procedure. Concentration of PPF extracted from impregnated papers was presented as mean ± standard deviation.
Ethical statement
This study was approved by the institutional animal ethical committee (IAEC) of DRDE, Gwalior, India vide research protocol number VMD-06/56/DS-I. The IAEC adheres to the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Animal Welfare Division, Government of India.
Results
Reduction in fecundity, fertility and emergence in female Ae. aegypti exposed 24 hr before blood meal
Indian wild type Ae. aegypti exposed to different concentrations of PPF impregnated on paper surface displayed considerable reproductive disruption (Tables 1 & 2). Average eggs laid, eggs hatched, pupae formed and adult emerged per female exposed before blood meal showed declining trend (S1 Fig) from lowest to highest concentration of PPF (F ≥ 41.9; p < 0.0001; R2 ≥ 0.94; df = 4). The control females not exposed to PPF from before blood meal group laid 64.1±1.5 (average ± standard error of mean) number of eggs, which was significantly higher as compared to all the test concentrations (t ≥ 8.9; p ≤ 0.01) (Table 1). Similarly average number of eggs hatched (fertility) and adult emerged per female was also high in control as compare to different concentrations of PPF used in the present study (t ≥ 10.5; p ≤ 0.009 for fertility and t ≥ 11.1; p ≤ 0.008 for adult emergence). Percentage inhibition in fecundity (egg laid) ranged from 36.9% [OR = 0.41 (95% CI = 0.24–0.71)] in 0.00075% to 45.1% [OR = 0.30 (95% CI = 0.17–0.54)] in 0.75% PPF concentration. Similarly % inhibition in fertility ranged from 46.9% [OR = 0.28 (95% CI = 0.16–0.51)] in the lowest concentration to 61.8% [OR = 0.15 (95% CI = 0.08–0.29)] in the highest concentration respectively. Furthermore, the inhibition (%) observed in adult emergence in females exposed to PPF before blood meal was found to be 58.8% [OR = 0.18 (95% CI = 0.09–0.36)] and 79.2% [OR = 0.04 (95% CI = 0.02–0.10)] in 0.00075% (lowest concentration) and 0.75% (highest concentration) PPF respectively. Probit analysis suggested that FI50 (50% fertility inhibition) value was 0.002% (p = 0.82), whereas EI50 (50% emergence inhibition) was 0.0001% (p = 0.99) respectively (Table 3). Presently the log-dose probit model used to determine the EI50 and FI50 values displayed normal distribution of percent EI and FI with concentration.
Table 1. Effect on Ae. aegypti fecundity, fertility and adult emergence when exposed before blood meal.
| Effect on fecundity of Ae. aegypti females exposed 24 hr before blood meal | |||||
| Exposure |
Eggs laid (average ±SEM) |
95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 64.1±1.5 | 57.8–70.4 | 0 | ND | ND |
| 0.00075 | 40.5±1.6 | 33.5–47.4 | 36.9 | 0.41(0.24–0.71) | 0.012(8.9) |
| 0.0075 | 38.8±1.1 | 34.2–43.3 | 39.5 | 0.37(0.21–0.65) | 0.002(23.6) |
| 0.075 | 37.3±3.0 | 24.3–50.2 | 41.9 | 0.33(0.19–0.59) | 0.004(16.3) |
| 0.75 | 35.2±1.4 | 29.5–41.1 | 45.1 | 0.30(0.17–0.54) | <0.0001(257.6) |
| Effect on fertility of Ae. aegypti females exposed 24 hr before blood meal | |||||
| Exposure | Eggs hatched (average ±SEM) | 95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 61.7±1.7 | 54.5–68.8 | 0 | ND | ND |
| 0.00075 | 32.8±1.5 | 26.2–39.3 | 46.9 | 0.28(0.16–0.51) | 0.009(10.5) |
| 0.0075 | 27.3±0.8 | 23.7–30.9 | 55.7 | 0.19(0.10–0.36) | 0.002(25.5) |
| 0.075 | 26.4±2.1 | 17.6–35.3 | 57.1 | 0.18(0.09–0.34) | 0.0002(69.9) |
| 0.75 | 23.6±1.5 | 17.3–29.8 | 61.8 | 0.15(0.08–0.29) | 0.0003(54.0) |
| Effect on adult emergence of Ae. aegypti females exposed 24 hr before blood meal | |||||
| Exposure |
Emergence (average ±SEM) |
95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 50.1±2.5 | 39.5–60.8 | 0 | ND | ND |
| 0.00075 | 20.7±0.7 | 17.7–23.6 | 58.8 | 0.18 (0.09–0.36) | 0.008(11.1) |
| 0.0075 | 16.6±0.2 | 15.7–17.4 | 67.0 | 0.12 (0.05–0.25) | 0.005(13.7) |
| 0.075 | 13.5±1.3 | 8.0–19.1 | 73.0 | 0.08 (0.03–0.18) | 0.001(30.2) |
| 0.75 | 10.4±0.4 | 8.5–12.3 | 79.2 | 0.04 (0.02–0.10) | 0.002(19.3) |
ND- not determined; CI-confidence interval; OR-odds ratio; SEM-standard error of mean.
Table 2. Effect on Ae. aegypti fecundity, fertility and adult emergence when exposed after blood meal.
| Effect on fecundity of Ae. aegypti females exposed 24 hr after blood meal | |||||
| Exposure |
Eggs laid (average ±SEM) |
95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 65.1±6.0 | 39.4–90.7 | 0 | ND | ND |
| 0.00075 | 49.5±3.6 | 34.0–65.0 | 24.0 | 0.59(0.35–1.00) | 0.02(6.6) |
| 0.0075 | 45.1±0.2 | 44.1–46.2 | 30.7 | 0.48(0.28–0.82) | 0.04(3.3) |
| 0.075 | 42.8±5.7 | 18.5–67.1 | 34.3 | 0.44(0.25–0.75) | 0.001(32.1) |
| 0.75 | 44.3±0.8 | 41.1–47.5 | 32.0 | 0.46(0.27–0.79) | 0.04(3.1) |
| Effect on fertility of Ae. aegypti females exposed 24 hr after blood meal | |||||
| Exposure | Eggs hatched (average ±SEM) | 95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 55.8±5.1 | 33.7–77.7 | 0 | ND | ND |
| 0.00075 | 33.5±2.6 | 22.3–44.7 | 40.0 | 0.37(0.20–0.67) | 0.01(8.8) |
| 0.0075 | 27.7±0.2 | 26.9–28.4 | 50.4 | 0.25(0.13–0.47) | 0.03(5.4) |
| 0.075 | 25.1±1.6 | 18.1–32.2 | 55.0 | 0.20(0.10–0.39) | 0.02(8.0) |
| 0.75 | 25.5±0.4 | 23.9–27.0 | 54.3 | 0.22(0.11–0.42) | 0.03(6.0) |
| Effect on adult emergence of Ae. aegypti females exposed 24 hr after blood meal | |||||
| Exposure | Emergence (average ±SEM) | 95% CI | Inhibition (%) | OR (95%CI) | p(t) |
| Control | 43.3±4.0 | 27.4–59.2 | 0 | ND | ND |
| 0.00075 | 15.4±1.2 | 10.4–20.3 | 64.4 | 0.12(0.05–0.28) | 0.01(11.0) |
| 0.0075 | 11.5±0.6 | 8.97–14.0 | 73.4 | 0.08(0.03–0.19) | 0.02(7.6) |
| 0.075 | 9.9±1.3 | 4.38–15.5 | 77.1 | 0.05(0.02–0.14) | 0.01(13.7) |
| 0.75 | 10.7±0.5 | 8.6–12.7 | 75.3 | 0.07(0.03–0.17) | 0.01(8.8) |
ND- not determined; CI-confidence interval; OR-odds ratio; SEM-standard error of mean.
Table 3. Probit analysis of fertility and adult emergence inhibition impact of PPF exposure on Ae. aegypti mosquitoes.
| Exposure | Effect | Concentration (%) | χ2(p) | Slope | r2 | g |
|---|---|---|---|---|---|---|
| Before blood meal | FI50 | 0.002 | 0.4(0.82) | 0.12±0.06 | 0.96 | 0.90 |
| EI50 | 0.0001 | 0.02(0.99) | 0.20±0.06 | 1.0 | 0.36 | |
| After blood meal | FI50 | 0.01 | 0.2(0.63) | 0.19±0.09 | 0.98 | 0.86 |
| EI50 | <0.0001 | 0.02(0.98) | 0.19±0.09 | 0.98 | 0.98 |
FI50/EI50-fertility/adult emergence inhibition 50%; χ2 –chi square
Reproductive disruption impact on female Ae. aegypti exposed 24 hr after blood meal
Impact of PPF exposure on reproduction competence of female Ae. aegypti mosquitoes has been presented in Table 2. There was significant concentration dependent change in fecundity, fertility and adult emergence as number of eggs laid, eggs hatched and adult emerged per female were found decreasing with increasing concentration (F ≥ 5.2; p < 0.02; R2 ≥ 0.67; df = 4). It was found that average eggs laid, hatched and adult emerged for placebo control females were statistically more than females exposed to different concentrations (t ≥ 5.4; p ≤ 0.03) (Table 2 & S2 Fig). Results showed that fecundity inhibition (%) ranged from 24.0% [OR = 0.59 (95% CI = 0.35–1.00)] to 34.3% [OR = 0.44 (95% CI = 0.25–0.75)]. On the other hand, inhibition in fertility was found ranging from 40.0% [OR = 0.37 (95% CI = 0.20–0.67)] in 0.00075% to 55.0% [OR = 0.20 (95% CI = 0.10–0.39)] in 0.075% of PPF concentration respectively. Similarly, the adult Ae. aegypti emergence inhibition varied from 64.4% [OR = 0.12 (95% CI = 0.05–0.28)] to 77.1% [OR = 0.05 (95% CI = 0.02–0.14)] among the different concentrations used in the present study (Table 2). In females exposed to PPF 24 hr after blood meal, the FI50 value was 0.01% (p = 0.63), whereas EI50 was observed to be <0.0001% (p = 0.98) respectively (Table 3). Furthermore, the log dose probit model used to determine FI50 and EI50 did not deviate from the linearity (p≥0.05) (Table 3).
Results have also suggested that reduction found in egg laying, egg hatching and pupae formation was similar in both the exposure groups (24 hr before blood meal group and 24 hr after blood meal group), but differ statistically in adult emergence for all the concentrations (p≤ 0.02; t≤ 3.9), except for 0.75% (p = 0.8; t = 0.3).
Impact on ovary maturation
We could not find any visible difference between ovaries of control group females and those exposed at concentrations lower than 0.75%. In control group, the ovaries displayed normal vitellogenesis during 18 hr, 24 hr and 48 hr, and fully developed eggs post 72 hr of blood meal (Fig 1a–1f). However in 0.75% exposure group, no vitellus formation could be seen after 18 hr of blood feed (Fig 2a), whereas after 72 hr there was arrest in follicle maturation as the eggs could not attain Christopher stage V (Fig 2b–2d). Of this group, only 11 (36.7%) females showed microscopically distinguishable morphological deformities as compared to the control. Furthermore, in successfully laid eggs of exposure group females, there was altered body organization, chitinization and disoriented cervical and abdominal region as compared to control (Fig 3a and 3b). Such eggs either did not hatch or hatched improperly and died subsequently (Fig 3c and 3d).
Fig 1. Sequential development stages of ovaries in control Ae. aegypti female.
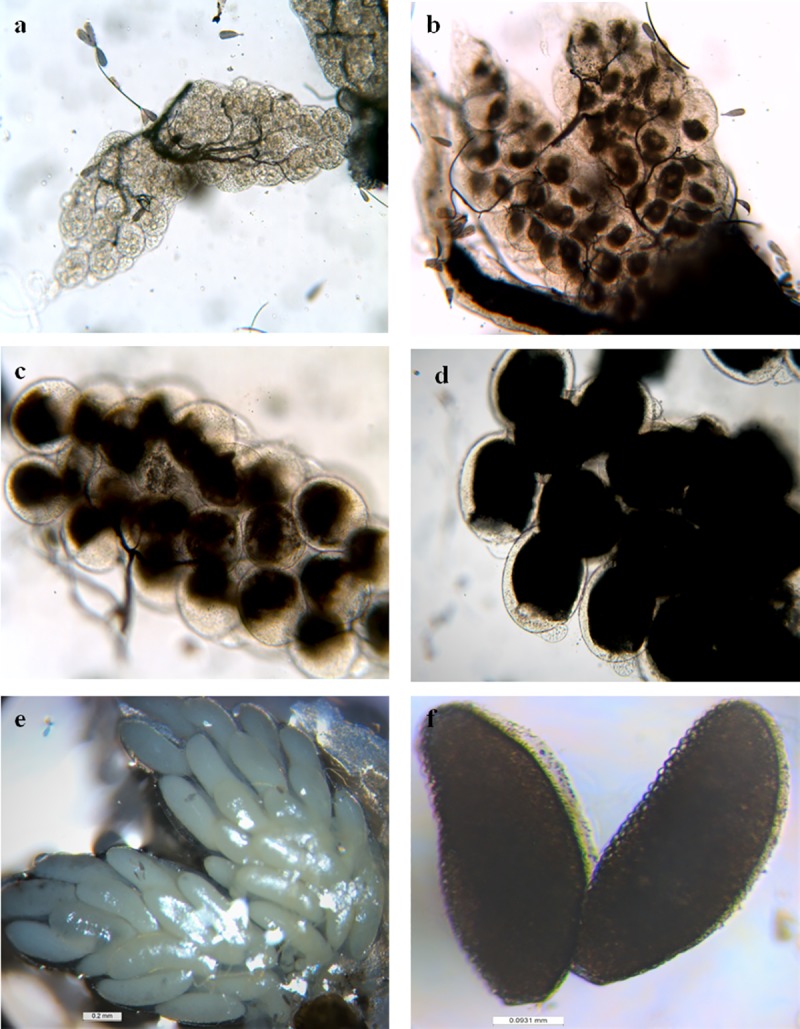
(a) Before blood meal, (b) after 18 hr of blood meal, (c) after 24 hr of blood meal, (d) after 48 hr of blood meal, (e) after 72 hr of blood meal, (f) fully mature single egg seen after 72 hr of blood meal.
Fig 2. Ovary of exposed female Ae. aegypti.
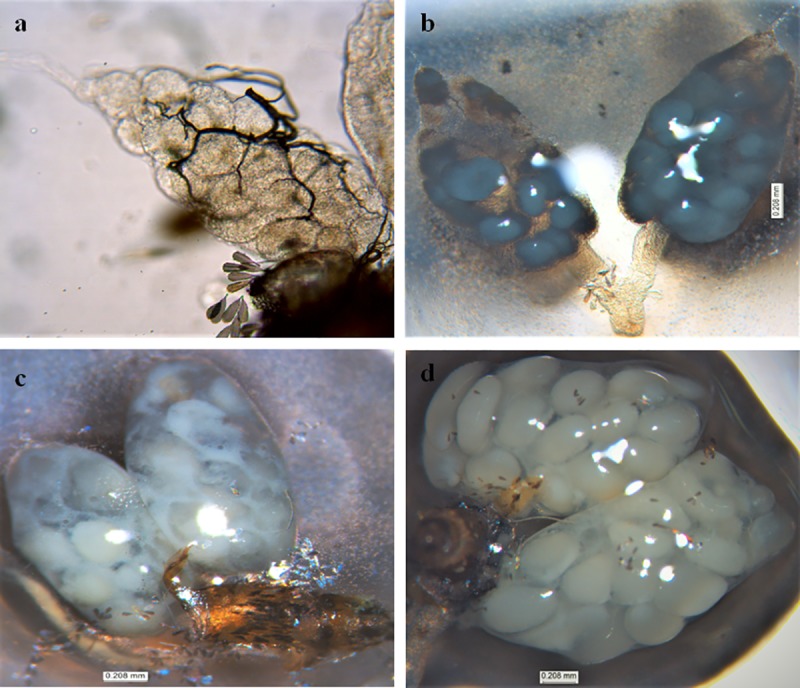
(a) Eggs showing arrested yolk formation, (b) ovarioles showing distorted eggs formation, (c) showing abnormal aborted egg (d) showing retarded growth in egg development.
Fig 3. Eggs laid by female Ae. aegypti.

(a) Control egg showing normal segmentation, (b) egg of exposed female displaying disrupted differentiation and no segmentation, (c and d) abnormal hatching of eggs of exposed female showing death of larval stage.
Concentration and uniformity of PPF on impregnated papers
Presently, the isocratic method of elution used for quantitative analysis of PPF (S3 Fig) provided an excellent correlation coefficient (0.998) between peak area and concentration of standard PPF solutions in the working range of 0.00012375 to 0.12375 mg/ml (S1 Table & S4 Fig). At present, this method has been found effective in separating the PPF from other interfering chemicals extracted from the impregnated paper (S5 Fig). It was found that in 0.00075% concentration impregnated paper, HPLC method estimated 0.00067 ± 0.00011%, whereas for 0.75%, it estimated 0.748 ± 0.059% quantitatively (S2 Table). Results further showed that the impregnation of PPF was uniform throughout the paper. Nevertheless, in all cases, the HPLC estimated less PPF than impregnated initially on the paper and the degree of estimation ranged from 0.72–0.98.
Discussion
Amidst reports of widespread insecticide resistance to different mosquito vectors, priority is to use chemicals that can improve control interventions and are safe to non-target organisms. In the recent years, PPF delivered using different methods has been shown to effectively control immature stages of vector mosquitoes in variety of habitats in endemic regions. The impact of PPF on aquatic stages of vector mosquitoes has been much studied primarily because control measures using PPF are mostly aimed at targeting developmental stages that are present in breeding habitat [10, 16, 26, 27]. However its efficacy and persistence in aquatic conditions has been inconsistent due to various reasons [16]. On the other hand its usefulness while treated on surfaces, such as nets, cloths, paints and spray formulations, which are used in human dwellings, could be more practical in achieving sustainable persistence and efficacy for a considerable time.
Current study has demonstrated that PPF impregnated on surface at very low concentration is capable of interrupting reproduction in field collected vector mosquitoes. Ae. aegypti females exposed to a range of PPF concentrations revealed reduction in egg laying, egg hatching and adult emergence. There was concentration dependent non-significant decline in egg laying in exposed female (both exposures), indicating that minimum concentration used in the study was equally capable in reducing the fecundity (p≥0.3). However in case of fertility and adult emergence reduction, there was concentration dependent, but significant effect as the impact was more in higher concentrations (p≤0.02; F≥6.2). This suggests that PPF present on a surface in lower concentration can reduce the egg laying ability among exposed female mosquitoes to the same extent as that of higher concentration. Overall the impact of PPF was found hampering egg laying, egg hatching and adult emergence when exposed to all the concentrations. The results have suggested that a concentration of 0.0001% PPF impregnated on surface could be sufficient to considerably reduce the adult emergence in dengue vector Ae. aegypti. In an earlier study, PPF treated on hard surface has been demonstrated to produce reduction in different developmental stages in An. gambiae and Cx. quinquefasciatus mosquitoes with a reduction ranging from 60 to 94% as compared to control [18]. Furthermore, the study also showed that emergence inhibition was high when females were exposed immediately before oviposition. In the present study, the reduction compared to control females never inclined above 79.2%, but the emergence inhibition impact was similar to that reported by Mbare et al [18]. Studies have also reported that higher concentrations used on hard surfaces are capable of producing complete sterility in exposed females. Koama et al. [19] has suggested that females of An. gambiae exposed to 1% PPF treated bed nets could not lay eggs even after many blood meals, indicating that higher concentration of PPF could induce persistent and longer residual activity on mosquito reproduction. In contrast, another study [21] revealed that 1% PPF alone or in combination with permethrin could reduce fertility by 7–12% relative to control, while no effect could be found in fecundity of wild resistant An. gambiae s.s. in field experiments. Therefore it is more likely that overall impact of PPF may vary depending on concentration used, mode of delivery, exposure time, size of mosquito, and other abiotic factors [10, 11, 16].
Several studies have suggested that effectiveness of PPF decreases over time as the concentration present on the treated surface and that available to the exposed insects decline with time [9, 16]. Keeping this in mind, present study has used different concentrations offering wide range of dose exposure to the tested female mosquitoes to understand whether reduced concentration expected over a period of time could sufficiently impact the mosquito development. Present results have indicated that a concentration of 0.002% and 0.0001% were enough to reduce fertility and adult emergence respectively by 50% in females exposed before blood meal. Similarly, 0.01% and <0.0001% concentrations could be able to cause 50% reduction in fertility and adult emergence in females exposed after blood meal.
Exposure to PPF-treated surface prevented the rate at which Ae. aegypti female ovaries develop through different previtellogenic stages to attain maturity. It has been reported that PPF based formulations obstruct the previtellogenic developmental process and block the yolk proteins synthesis, hence disrupting the hormonal route necessary for egg development [19]. Microscopic observation of control female ovaries showed that ovarioles followed a sequential developmental pattern showing vitellus agglomeration at the base of follicles and contained a single large oocyte as compared to the exposed ovarioles where oocytes were round and much reduced in size (Fig 1). The exposed females showed deterioration of follicular membrane and cytoplasm in the ovarioles, and reduced vitellus granules in the follicles during the initial 18 hr of blood meal (Fig 2a). In some cases, ovaries contained a few ovarioles only that were dark in colour (Fig 2b), more opaque and sometime with diffused and improperly organized eggs (Fig 2c and 2d), hence displaying clear signs of abortion. In Africa, the females of malaria vector An. gambiae exposed to PPF treated nets showed delayed development that never reached to maturity [19]. Furthermore, PPF treatment has been found associated with the abnormal development of eggs in different vector mosquito species by arresting the embryo development at different development stages [26]. There was explicit segmentation showing clear body division in the eggs that were laid by control females (Fig 3a), whereas the exposed female eggs showed distinct desegmentation in the eggs (Fig 3b). In some cases, the eggs in exposed females could not hatch properly and died subsequently. Such eggs showed undifferentiated mass of underdeveloped larvae attached to operculum as well as to the larger abopercular portion of the egg (Fig 3c and 3d). Suman et al. [26] has reported that some IGR analogues are capable of causing morphological deformities and disoriented body organisation, however they showed that PPF alter the hormonal actions during embryogenesis but may not affect the egg hatching process.
Cross estimation of PPF on impregnated papers using HPLC method was carried out to find out the concentration of free PPF available for exposed mosquitoes. Various studies have argued that chromatographic methods could be used to estimate the decline in insecticide residue over a period of time [28], but it is equally important to first estimate the initial concentration of active anti-insect chemical and then determine the decline in light of estimated concentration. Presently used HPLC method was effective to extract and estimate PPF, and displayed >0.72 degree of estimation as compared to a previous study that could estimate 60.9% of permethrin [29]. Due to excellent correlation between concentration and peak area, this method can suitably be employed to estimate the concentration and to check the uniformity of PPF in impregnated paper. The small variation in estimated PPF concentrations to treated concentrations may be due to various factors associated with impregnation process and the inherent property of the paper to hold back PPF tightly in its molecular architecture. Therefore, despite higher PPF concentration present in the treated matrix, the bioactivity depends upon the actual concentration available on the surface.
Presently, majority of the studies have demonstrated that PPF could be used effectively when applied against larval stages of vector mosquitoes, while few have suggested that PPF impregnated on nets was able to reduce the vector population in different areas. However data on efficacy of similar formulations that could be applied on surfaces such as walls, ceilings and other similar objects is scanty. Furthermore, none of the study to our knowledge has demonstrated the minimum concentration of PPF present in any surface/matrix that would be capable of reducing the mosquito population while delivered through tarsal contact. Present results provided sufficient evidence that PPF exposure caused irreversible structural damage to the ovaries of female Ae. aegypti mosquitoes, thereby making them unfit to lay normal eggs under laboratory condition. The PPF based formulations applied on different surfaces could be equally effective even after the concentration is reduced to minimum. These minimum effective concentrations can be used as cut-off limits for determining effective life of similar formulations that use PPF alone or in combination with the insecticides against mosquito vectors of public health importance. It might be possible for exposed female mosquitoes to carry PPF into the nearby breeding habitats. Since very small concentration of PPF is sufficient enough, transferring pyriproxyfen into small and cryptic mosquito breeding habitats through adults as vehicles could be an added advantage. PPF could be perfect to be used along with suitable insecticide for better control interventions in the areas where insecticide susceptibility has been compromised. Although, PPF used in formulations that offer its delivery through contact provides a promising strategy for reducing Aedes abundance, its similar effectiveness on other vector mosquitoes and role in reduction of disease incidences need to be established in field for better results in control programmes.
Conclusion
PPF impregnated on surface substantially reduced the fecundity, fertility and adult emergence in Indian wild Ae. aegypti mosquitoes. Although the study did not investigate the relation between intrinsic activity of PPF in light of the behaviour of vector mosquitoes, but suggested that a certain minimum concentration of PPF through tarsal contact can reduce the vector mosquitoes population to a considerable level. The study advocates that formulations based on combination of PPF and other compatible insecticides may be an impactful approach where susceptible mosquitoes are killed by insecticide component and remaining resistant mosquitoes are sterilised by PPF. More importantly, integrating PPF in insecticide based vector control formulations that are used indoors could be more promising and control oriented. The results of current study can also be utilised to ascertain the effectiveness of PPF based formulations applied on surface by quantitative analysis rather performing time consuming and laborious bioassay experiments.
Supporting information
(a) egg laying, (b) eggs hatching, (c) pupation and (d) adult emergence [values: median (min to max) per female].
(TIF)
(a) egg laying, (b) eggs hatching, (c) pupation and (d) adult emergence [values: median (min to max) per female].
(TIF)
HPLC chromatogram of standard PPF solution (Rt = 5.993 min) at 0.001238 mg/ml concentration.
(TIF)
Correlation between concentration (mg/ml) and corresponding peak area of PPF in HPLC analysis in the working range of 0.00012375 to 0.12375 mg/ml concentration.
(TIF)
Representative HPLC chromatogram of PPF solution extracted from impregnated paper. Peak at 5.999 min corresponds to PPF.
(TIF)
(DOCX)
(DOCX)
Acknowledgments
Authors are thankful to Director DRDE, Gwalior for his continuous encouragement during the experiments. Current manuscript was provided accession number DRDE-VMD-16-2019 in Defence R&D Establishment, Gwalior, India.
Data Availability
All data is contained within the manuscript.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Che-Mendoza A, Medina-Barreiro A, Koyoc-Cardena E, Uc-Puc V, Contreras-Perera Y, Herrera-Bojórquez J, et al. House screening with insecticide-treated netting provides sustained reductions in domestic populations of Aedes aegypti in Merida, Mexico. PLoS Negl Trop Dis. 2018; 12(3):e0006283 10.1371/journal.pntd.0006283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organisation. World Malaria Report 2017. WHO. 2018; WHO Press, 20 Avenue Apia, Geneva, Switzerland. [Google Scholar]
- 3.Seixas G, Grigoraki L, Weetman D, Vicente JL, Silva AC, Pinto J, et al. Insecticide resistance is mediated by multiple mechanisms in recently introduced Aedes aegypti from Madeira Island (Portugal). PLoS Negl Trop Dis. 2017; 11(7):e0005799 10.1371/journal.pntd.0005799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yadav K, Rabha B, Dhiman S, Veer V. Multi-insecticide susceptibility evaluation of dengue vectors Stegomyia albopicta and St. aegypti in Assam, India. Parasit Vectors. 2015; 8:143 10.1186/s13071-015-0754-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Meyers JI, Pathikonda S, Popkin-Hall ZR, Medeiros MC, Fuseini G, Matias A, et al. Increasing outdoor host-seeking in Anopheles gambiae over 6 years of vector control on Bioko Island. Malar J. 2016; 15:239 10.1186/s12936-016-1286-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ndiath MO, Mazenot C, Sokhna C, Trape JF. How the malaria vector Anopheles gambiae adapts to the use of insecticide-treated nets by African populations. PLoS One. 2014; 9(6):e97700 10.1371/journal.pone.0097700 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 7.World Health Organization. Global Strategy for Dengue Prevention and Control, 2012–2020. WHO. 2012; WHO Press, 20 Avenue Apia, Geneva, Switzerland. [Google Scholar]
- 8.Marcombe S, Chonephetsarath S, Thammavong P, Brey PT. Alternative insecticides for larval control of the dengue vector Aedes aegypti in Lao PDR: insecticide resistance and semi-field trial study. Parasit Vectors. 2018; 11:616 10.1186/s13071-018-3187-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mbare O, Lindsay SW, Fillinger U. Dose–response tests and semi-field evaluation of lethal and sub-lethal effects of slow release pyriproxyfen granules (SumilarvW0.5G) for the control of the malaria vectors Anopheles gambiae sensu lato. Malaria J. 2013; 12:94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Suman DS, Wang Y, Dong L, Gaugler R. Effects of larval habitat substrate on pyriproxyfen efficacy against Aedes albopictus (Diptera: Culicidae). J Med Entomol. 2013; 50(6):1261–6. 10.1603/me13068 [DOI] [PubMed] [Google Scholar]
- 11.Jaffer A, Protopopoff N, Mosha FW, Malone D, Rowland MW, Oxborough RM. Evaluating the sterilizing effect of pyriproxyfen treated mosquito nets against Anopheles gambiae at different blood-feeding intervals. Acta Trop. 2015; 150:131–5. 10.1016/j.actatropica.2015.07.011 [DOI] [PubMed] [Google Scholar]
- 12.Bowman LR, Donegan S, McCall PJ. Is dengue vector control deficient in effectiveness or evidence?: Systematic review and meta-analysis. PLoS Negl Trop Dis. 2016; 10(3):e0004551 10.1371/journal.pntd.0004551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yapabandara AM, Curtis CF. Control of vectors and incidence of malaria in an irrigated settlement scheme in Sri Lanka by using the insect growth regulator pyriproxyfen. J Am Mosq Control Assoc. 2004; 20:395–400. [PubMed] [Google Scholar]
- 14.Ohashi K, Nakada K, Ishiwatari T, Miyaguchi J, Shono Y, Lucas JR, et al. Efficacy of pyriproxyfen-treated nets in sterilizing and shortening the longevity of Anopheles gambiae (Diptera: Culicidae). J Med Entomol. 2012; 49:1052–8. 10.1603/me12006 [DOI] [PubMed] [Google Scholar]
- 15.Lwetoijera DW, Harris C, Kiware SS, Killeen GF, Dongus S, Devine GJ, et al. Comprehensive sterilization of malaria vectors using pyriproxyfen: A step closer to malaria elimination. Am J Trop Med Hyg. 2014; 90(5):852–855. 10.4269/ajtmh.13-0550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maoz D, Ward T, Samuel M, Mueller P, Runge-Ranzinger S, Toledo J, et al. Community effectiveness of pyriproxyfen as a dengue vector control method: A systematic review. PLoS Negl Trop Dis. 2017; 11(7):e0005651 10.1371/journal.pntd.0005651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ohba SY, Ohashi K, Pujiyati E, Higa Y, Kawada H, Mito N, et al. The effect of pyriproxyfen as a “population growth regulator” against Aedes albopictus under semi-field conditions. PLoS One. 2013; 8:e67045 10.1371/journal.pone.0067045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mbare O, Lindsay SW, Fillinger U. Pyriproxyfen for mosquito control: female sterilization or horizontal transfer to oviposition substrates by Anopheles gambiae sensu stricto and Culex quinquefasciatus. Parasit Vectors. 2014; 7:280 10.1186/1756-3305-7-280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Koama B, Naountougou M, Sanou R, Ndo S, Quattara A, Dabire RK, et al. The sterilizing effect of pyriproxyfen on the malaria vector Anopheles gambiae: physiological impact on ovaries development. Malar J. 2015; 14:101 10.1186/s12936-015-0609-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kawada H, Dida GO, Ohashi K, Kawashima E, Sonye G, Njenga SM, et al. A small-scale field trial of pyriproxyfen-impregnated bed nets against pyrethroid-resistant Anopheles gambiae s.s. in Western Kenya. PLoS One. 2014; 9(10):e111195 10.1371/journal.pone.0111195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koffi AA, Alou LPA, Djenontin A, Kabran JPK, Dosso Y, Kone A, et al. Efficacy of Olyset Duo, a permethrin and pyriproxyfen mixture net against wild pyrethroid-resistant Anopheles gambiae s.s. from Cote d'Ivoire: an experimental hut trial. Parasite. 2015; 22:28 10.1051/parasite/2015028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ngufor C, N’Guessan R, Fagbohoun J, Odjo A, Malone D, Akogbeto M, et al. Olyset Duo (a pyriproxyfen and permethrin mixture net): an experimental hut trial against pyrethroid resistant Anopheles gambiae and Culex quinquefasciatus in Southern Benin. PLoS One. 2014; 9:e93603 10.1371/journal.pone.0093603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ngufor C, N’Guessan R, Fagbohoun J, Todjinou D, Odjo A, Malone D, et al. Efficacy of the Olyset Duo net against insecticide-resistant mosquito vectors of malaria. Sci Transl Med. 2016; 8(356):356ra121. [DOI] [PubMed] [Google Scholar]
- 24.Mosqueira B, Duchon S, Chandre F, Hougard JM, Carnevale P, Coma SM. Efficacy of an insecticide paint against insecticide susceptible and resistant mosquitoes—Part 1: Laboratory evaluation. Malar J. 2010; 9:340 10.1186/1475-2875-9-340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.World Health Organisation. The WHO recommended classification of pesticides by hazard and guidelines to classification 2009. WHO. 2010; WHO Press, 20 Avenue Appia, Geneva, Switzerland. [Google Scholar]
- 26.Suman DS, Wang Yi, Bilgrami AL, Gaugler R. Ovicidal activity of three insect growth regulators against Aedes and Culex mosquitoes. Acta Trop. 2013; 128:103–9. 10.1016/j.actatropica.2013.06.025 [DOI] [PubMed] [Google Scholar]
- 27.Darriet F, Corbel V. Laboratory evaluation of pyriproxyfen and spinosad, alone and in combination, against Aedes aegypti larvae. J Med Entomol. 2006; 43(6):1190–4. 10.1603/0022-2585(2006)43[1190:leopas]2.0.co;2 [DOI] [PubMed] [Google Scholar]
- 28.Tan KR, Coleman J, Smith B, Hamainza B, Sakala CK, Kean C, et al. A longitudinal study of the durability of long-lasting insecticidal nets in Zambia. Malar J. 2016; 15:106 10.1186/s12936-016-1154-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.DeRaedt BS, Orsborne J, Gezan SA, Kaur H, Wilder-Smith A, Lindsey SW, et al. Permethrin-treated clothing as protection against the dengue vector, Aedes aegypti: Extent and duration of protection. PLoS Negl Trop Dis. 2015; 9(10): e0004109 10.1371/journal.pntd.0004109 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(a) egg laying, (b) eggs hatching, (c) pupation and (d) adult emergence [values: median (min to max) per female].
(TIF)
(a) egg laying, (b) eggs hatching, (c) pupation and (d) adult emergence [values: median (min to max) per female].
(TIF)
HPLC chromatogram of standard PPF solution (Rt = 5.993 min) at 0.001238 mg/ml concentration.
(TIF)
Correlation between concentration (mg/ml) and corresponding peak area of PPF in HPLC analysis in the working range of 0.00012375 to 0.12375 mg/ml concentration.
(TIF)
Representative HPLC chromatogram of PPF solution extracted from impregnated paper. Peak at 5.999 min corresponds to PPF.
(TIF)
(DOCX)
(DOCX)
Data Availability Statement
All data is contained within the manuscript.


