Abstract
Background
Anastomotic leak remains a significant cause of morbidity and mortality after colorectal surgery. Among multiple risk factors considered, hypoxia–ischaemia is considered to be a primary cause of intestinal anastomotic leakage. The aim of this experimental study was to assess safety, usability for surgical tasks, and efficacy of a newly developed oxygen‐producing suture material in the healing of colonic anastomoses under critical conditions.
Methods
An oxygen‐producing suture material was produced that is capable of releasing oxygen directly into the surrounding tissue. Off‐the‐shelf sutures loaded with calcium peroxide nano‐crystals and covered with poly(d,l‐lactide‐co‐glycolide) were assessed in vitro and in a rat model of hypoxic colonic anastomosis.
Results
In vitro assessment showed that these sutures can increase oxygen levels in a hypoxic environment. Potential oxygen byproducts did not seem to have a negative impact on the viability of intestinal cells. The use of oxygen‐producing sutures in vivo resulted in increased tissue oxygen saturation, measured by visible light spectroscopy, and increased mechanical stability of the anastomosis.
Conclusion
Oxygen‐producing suture material increased tissue oxygen saturation and mechanical stability of colonic anastomosis in a rat model.
Surgical relevance.
Leakage of anastomoses remains a significant problem after colorectal surgery.
An oxygen‐producing suture material was produced that was shown to be safe in vitro and significantly improved several aspects of healing of colonic anastomoses in an animal model.
Oxygen‐producing suture material or stapler devices might help to reduce the risk of anastomotic leak of intestinal anastomoses under physiological and critical conditions such as hypoxia.
Anastomotic leak remains a significant problem after colorectal surgery, and administration of supplemental perioperative oxygen has been shown to improve healing of colorectal anastomoses. The manufacture of an oxygen‐producing suture material is described; experiments showed that it improved some aspects of the healing of bowel anastomoses. Oxygen‐releasing suture (or stapling) material might be a future potential application to reduce the risk of anastomotic leak under critical conditions, such as hypoxia.

Increased mechanical stability by oxygen‐producing sutures.
Antecedentes
Las fugas anastomóticas siguen siendo una causa importante de morbilidad y mortalidad después de la cirugía colorrectal. Entre los múltiples factores de riesgo, se considera que la hipoxia/isquemia es una de las causas principales de la fuga anastomótica intestinal. El objetivo de este estudio experimental fue evaluar, en condiciones críticas, la seguridad, la facilidad de uso en los procedimientos quirúrgicos y la eficacia en la cicatrización de la anastomosis de colon de un material de sutura productor de oxígeno recientemente desarrollado.
Métodos
Hemos producido un material de sutura productor de oxígeno que es capaz de liberar oxígeno directamente en el tejido circundante. Las suturas disponibles en el mercado cargadas con nanocristales de peróxido de calcio (calcium peroxide, CPO) y cubiertas con ácido poliláctico coglicólico (PLGA) se evaluaron in vitro y en un modelo de rata de anastomosis hipóxica de colon.
Resultados
La evaluación in vitro mostró que estas suturas pueden aumentar los niveles de oxígeno en un ambiente hipóxico, y que los posibles subproductos de oxígeno no parecen tener un impacto negativo en la viabilidad de las células intestinales. El uso de suturas productoras de oxígeno in vivo causó una elevada saturación de oxígeno en el tejido medida por espectroscopia de luz visible, así como un aumento en la estabilidad mecánica de las anastomosis.
Conclusión
El material de sutura productor de oxígeno aumenta la saturación de oxígeno en los tejidos y la estabilidad mecánica de la anastomosis de colon en un modelo de rata.
Introduction
Despite much investigational effort, anastomotic leakage after colorectal surgery remains a significant problem associated with considerable morbidity and mortality1, 2, 3. Hypoxia–ischaemia has been identified as one of the primary reasons for anastomotic breakdown in both experimental4, 5 and clinical6, 7 studies. However, the application of supplemental systemic oxygen has been shown to increase tissue oxygen tension in perianastomotic and normal colonic tissue8, 9, and to improve healing of both normal and ischaemic colonic anastomoses10, 11. The effect of local oxygen administration on bowel anastomoses does not appear to have been evaluated to date.
Oxygen‐producing biomaterials have been developed for tissue engineering purposes. Components such as calcium peroxide (CPO) nano‐crystals incorporated into films of poly(d,l‐lactide‐co‐glycolide) (PLGA), as well as methylated pyridine‐derived endoperoxides, which start to release oxygen into the surrounding environment after contact with water, have been shown to improve cell viability significantly under hypoxic and normoxic conditions12, 13. In an attempt to develop a material for surgery that enables application of oxygen directly upon the anastomosis, the authors combined off‐the‐shelf Vicryl™ (Ethicon, Neuchâtel, Switzerland) sutures with the CPO–PLGA compound. After having established the manufacturing process, the effect of this oxygen‐producing suture material on the healing of colorectal anastomoses was assessed in an experimental model in vivo.
Methods
Manufacture of the oxygen‐releasing suture material
PLGA (Lakeshore Biomaterials, Short Hills, New Jersey, USA) 15 per cent w/v (85 : 15) was dissolved for 3 h in dimethyl sulphoxide (DMSO) (Fisher Scientific, Pittsburgh, Pennsylvania, USA). Calcium peroxide (Sigma‐Aldrich, Burlington, Massachusetts, USA) 10 per cent w/w was added to the PLGA solution. The mixture was stirred for 2 h. Once all components had dissolved, the solution was transferred to a 15‐ml conical tube and stored at −80°C until use.
Vicryl™ 6/0 coated sutures with a P‐1 needle (Ethicon) were wrapped around 1‐cm glass rods and secured with tape. The glass rods were suspended into the oxygenated polymer solution for 5 min. To disperse the CPO particles evenly, samples were vortexed every minute. Sutures were placed on glass trays and allowed to dry overnight. They were then dipped into a 100 per cent ethanol solution to remove any residual DMSO. The samples were air‐dried on plastic trays for a further hour. Sutures were repackaged into the original containers and sterilized using γ radiation (1 mrad, 1 h).
A methylthiazolyldiphenyl‐tetrazolium bromide (Invitrogen™; Fisher Scientific) colorimetric assay, used to evaluate compatibility of human BJ (3T3; American Type Culture Collection (ATCC), Wesel, Germany) fibroblasts with the different suture materials, showed no toxicity (data not shown).
In vitro evaluation of oxygen‐releasing capacity of the suture material
Oxygen generation was assessed using a water displacement method. Sutures previously coated with the oxygen‐generating material were placed inside a 5‐ml syringe. Some 2 ml water was added to start the oxygen generation. A 23‐G‐1 needle was attached to the end of the 5‐ml syringe. The needle was inserted into an inverted pipette (sealed at one end) that had been filled with water previously. As oxygen was generated, it collected in the pipette and displaced the water. The volume of water displaced, which corresponds to the volume of oxygen generated, was measured over a 72‐h period at room temperature and ambient conditions. Negative controls made of PLGA‐coated sutures were also prepared and evaluated.
Contractility assay
To prepare the collagen, 2·2‐mg/ml type I collagen was dissolved in 0·1 per cent acetic acid. Some 0·5 ml 10 × Eagle's Minimum Essential Medium (MEM) (Sigma Aldrich) and 0·5 ml 10 × reconstitution buffer was added under magnetic stirring in an ice bath. The pH was set to neutral by addition of 0·1 M sodium hydroxide to set the gel. Some 1 × 106 intestinal epithelial cells (IEC‐6; ATCC, Manassas, Virginia, USA), suspended in a solution composed of 89 per cent Dulbecco's Modified Eagle Medium (DMEM) high glucose (Sigma Aldrich), 10 per cent fetal bovine serum (Sigma‐Aldrich), 1 per cent penicillin–streptomycin solution (Sigma Aldrich) and 0·1 unit/ml bovine insulin, were added to each gel. Gels were cast in 48‐well plates with 250 μl per well. The different treatment groups were placed in an incubator under 1 per cent (which approximates the oxygen tensions found in the intestinal region) or 0·1 per cent oxygen (simulating hypoxia) and 5 per cent carbon dioxide conditions for 12 or 24 h14. After either 12 or 24 h in culture, three separate diameter measurements were made; the mean diameter and the area of each gel were calculated. The area of the final gel was subtracted from the original gel area, and the percentage decrease during contraction calculated. Results were then normalized to the untreated 1 per cent hypoxia group.
Preliminary animal studies
All animal studies were performed following the ARRIVE guidelines15 and according to protocols approved by the Cantonal Veterinary Office of Zurich, Switzerland, and conducted in strict accordance with the Guide for Care and Use of Laboratory Animals, University of Zurich, Switzerland, and the 3R principles were respected when calculating the sample size16.
Thirty‐three male Lewis rats from Charles River (Sulzfeld, Germany) were fed a standard laboratory diet and water ad libitum. After induction of ischaemia in the targeted colonic segment, anastomoses were performed using PLGA–CPO (oxygen‐producing), Vicryl™ (untreated) or PLGA (coated with vehicle solution only) according to the assigned treatment group (data not shown). Ischaemia was obtained by completely denuding the mesentery from the corresponding colon 1 cm on each side of the presumed anastomosis. Applying this technique, insufficient ischaemia was observed, and therefore the procedure was adapted to achieve a more pronounced tissue hypoxia in subsequent experiments.
Study design
Thirty‐six male Lewis rats (Charles River), of median weight 302 (range 244–375) g, were used in the main study; they were fed a standard laboratory diet and water ad libitum. The rats were divided into three groups (n = 12 each) according to the suture material used for the anastomoses (PLGA–CPO, Vicryl™ or PLGA sutures). In each group, the 12 animals were killed at the assigned time points (4 animals per day per group, 1, 3 and 7 days after surgery), and pathophysiological, biomechanical, histomorphological and immunohistochemical measurements of the perianastomotic tissue were made. To assess the physicochemical properties (tensile strength) of the sutures under in vivo conditions, three 5‐cm pieces of the respective sutures were implanted subcutaneously in the back of three of four animals per group.
Induction of ischaemia, surgical procedure and postoperative care
For perioperative pain relief, all animals received subcutaneous injections of buprenorphine (Temgesic®; Reckitt Benckiser, Zurich, Switzerland) 0·01–0·05 mg per kg bodyweight twice daily for 48 h, the first dose being given 30 min before the start of the operation. All animals had anaesthesia with 2 per cent isoflurane in a standard manner. After midline laparotomy, the distal end of the colon was defined, and tissue oxygen saturation was measured non‐invasively using visible light spectroscopy (O2C; LEA Medizintechnik, Giessen, Germany).
Pronounced ischaemia in the targeted colonic segment was achieved by adaption of the technique described by Hamzaoğlu and colleagues17, as follows: the nourishing vessels, including the marginal arch and respective mesentery, were ligated over a distance of 3 cm on each side of the presumed anastomosis, and the devascularized mesentery was then denuded completely from the corresponding colon.
After cutting the ischaemic bowel segment along its central part, anastomoses were performed by a single surgeon, as previously described18, using the respective suture material in an inverted and interrupted fashion. The surgeon was not blinded to the assignment of the treatment groups. After closure of the laparotomy, three of four animals per group underwent subcutaneous implantation of the respective suture material used for the anastomoses in the same animal before. At the end of the operation, 10 ml physiological sodium chloride was given to all animals by subcutaneous injection, to compensate perioperative fluid loss (Fig. 1).
Figure 1.
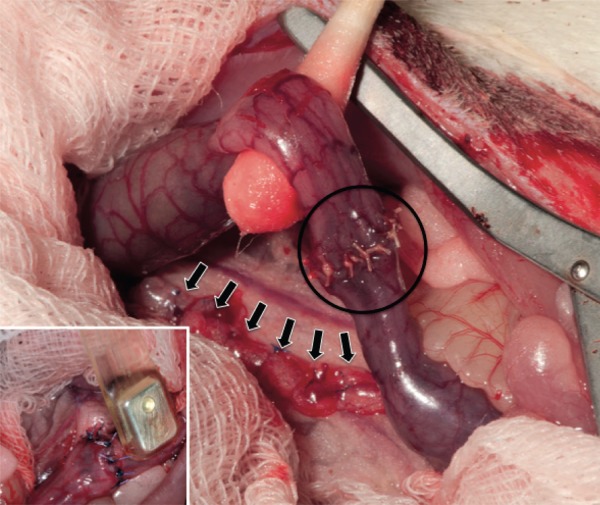
Operation site The bluish aspect of the bowel serosa indicates the surgically induced tissue hypoxia. The nourishing vessels are divided and ligated (arrows), and the respective bowel segment is denuded completely from its vascular supply over a distance of 30 mm on each side of the presumed anastomosis. The anastomosis was done in an interrupted fashion using the assigned suture material per group (circle). Insert: Tissue oxygen saturation was measured non‐invasively using a handheld probe placed at each side of the (presumed) anastomosis.
Non‐invasive measurement of perianastomotic tissue oxygen saturation
Perianastomotic tissue oxygen saturation (Sto 2 ) was measured using the O2C device. Visible light spectroscopy allowed non‐invasive measurement of perianastomotic Sto 2. The measurements were performed first at the intended site of anastomosis, then immediately after completion of the anastomosis and at death (animals under anaesthesia) using a handheld probe (Lx2; LEA Medizintechnik) placed directly on to the colonic serosa within the first 5 mm proximally and distally to the (presumed) site of the anastomosis.
Histological assessment and morphometric analysis
Transverse sections of the embedded tissue were stained with haematoxylin and eosin, and histomorphological assessment was performed. Colonic crypt depth (CCD), corresponding to the mucosal thickness of the colonic wall, which is generally considered to be a surrogate parameter of mucosal healing, was recorded by measuring the depth of the glands of Lieberkühn. Increased crypt depth indicates more mucosal proliferation and better healing19. Ten random measurements per animal were performed within the viable mucosa, where glands were perpendicular to the underlying muscularis.
Bursting pressure technique
Mechanical stability of the anastomosis is a clinically relevant parameter to evaluate its capacity not to leak out. Bursting pressure (BP) is a commonly used indicator to determine mechanical stability of the anastomosis. BPs were evaluated as described previously20. After dissection of the anastomotic site and submersion in a saline bath, BP was measured using a sphygmomanometer with an in‐line pressure transducer by increasing the intraluminal pressure in increments of 10 mmHg over 10 s at intervals of 10 s. BP was determined by noticing leakage of air or gross rupture of the anastomosis or any other part of the segment. Using untreated suture material, the BPs of normal and ischaemic colonic anastomoses in rats were shown previously21 to be approximately 128 and 109 mmHg respectively, on postoperative day 5. This corresponds to a 15 per cent difference in BP in favour of non‐ischaemic bowel anastomoses. Ideally, oxygen‐producing sutures could overcome this difference and yield at least 15 per cent better BP values compared with untreated Vicryl™ or PLGA threads in ischaemic anastomoses.
Assessment of tensile strength of the sutures
At death, the sutures implanted in the animal's back were harvested and stored in phosphate‐buffered saline at −80°C until analysis, in order to prevent possible degradation of the material through hydrolysis. Three sutures of each condition were analysed using the Instron® 5864 electromechanical testing system (Instron, Darmstadt, Germany), supported by Bluehill V2.12 software (Instron). The cross‐sectional area of the suture was defined as 12·27 μm2, corresponding to a diameter of 125 μm. To prevent slipping of the sutures within the pneumatic clamps of the tensile strength analyser, the ends of the sutures were taped between a folded adhesive foil that was inserted between the clamps. The clamps were operated at 5 bar. According to Ethicon, Johnson & Johnson (Johnson & Johnson Medical, Medical Affairs, Norderstedt, Germany), the manufacturer of the undyed 6/0 thread used in this experiment, normal tensile strength is specified with 1041 N/mm2 at day 0. There are no available data on the expected decrease of this value under particular conditions (postimplant degradation, postharvesting manipulation such as deep‐freezing of the specimen) and along the time axis. However, available manufacturer data showed that, for other types of Vicryl™ suture, a decline in tensile strength of approximately 20–50 per cent can be expected within the first 7 days after application (by courtesy of Ethicon, Johnson & Johnson; data not shown).
Statistical analysis
All results are expressed as mean(s.d.) values. Unless described otherwise, a minimum of three measurements was performed for each parameter evaluated. Significance of differences was assessed by one‐way ANOVA. To minimize the family‐wise error rate, results were adjusted by post hoc Holm–Šídák correction. Where these results remained significant, Student's t test contrasts were performed. P < 0·050 was considered statistically significant. SigmaStat version 3.5 (Systat Software, San Jose, California, USA) was used for these calculations.
Results
Generation of oxygen
Oxygen generation begins when the PLGA–CPO sutures are in contact with water, and can be witnessed by the gas bubbles emanating from the thread (Fig. 2 a,b). Oxygen generated by the sutures was collected through a water displacement method. Some 2 mg PLGA–CPO coating yielded approximately 90 μl oxygen over a 72‐h period. A typical suture was calculated to contain 2 mg of the PLGA–CPO coating. Thus, one suture generated approximately 90 µl oxygen (Fig. 2 c). The bulk of the oxygen was generated within the first 24 h of exposure to water. This experiment may have overestimated the rate of oxygen generation, as an excess of water was present.
Figure 2.
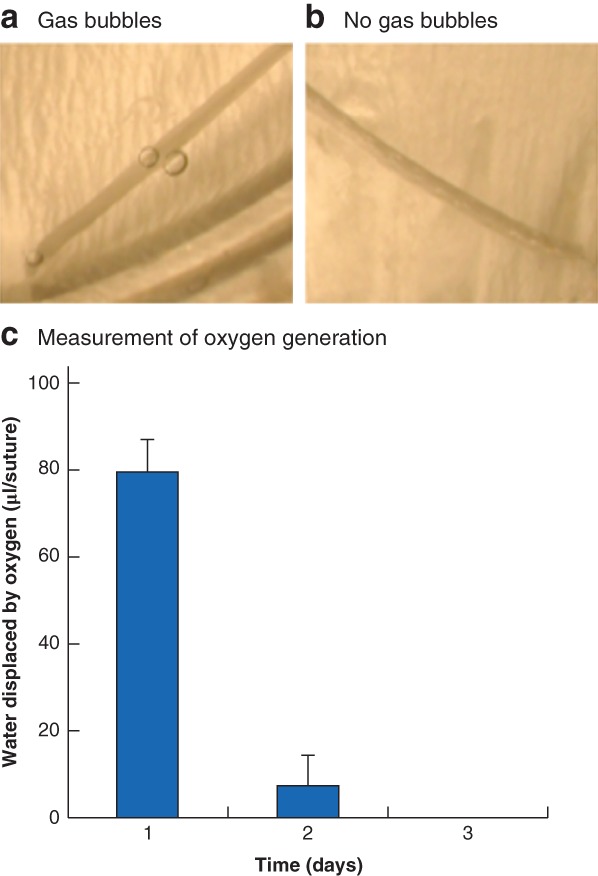
Generation of oxygen a Shortly after putting the manufactured suture containing calcium peroxide (CPO) nano‐crystals into normal tap water, gas bubbles start to emerge from the suture. b No gas bubbles emanate from the untreated control suture. c Oxygen release was measured using a water displacement method. Some 2 mg poly(d,l‐lactide‐co‐glycolide) (PLGA)–CPO‐coated suture was added to a syringe and measured over a 72‐h period. Values are mean(s.d.).
Contractility assay
A decrease in the contractility response of the IEC‐6 cells with hypoxia was observed. No impact of the non‐oxygenating sutures was observed. Addition of the oxygen‐generating sutures resulted in a statistically significant increase in the contractility response compared with that in the control group when cultured under 1 and 0·1 per cent oxygen (data not shown).
Tissue oxygen saturation of the perianastomotic zone
Pooled data for all 69 operated animals yielded a mean(s.d.) baseline Sto 2 on the target colonic segment of 81(9) per cent. This value changed to 77(10) per cent in the 33 animals in the preliminary study, corresponding to a mean drop in Sto 2 levels of only 5 per cent.
After adequate induction of ischaemia in a further 36 animals, Sto 2 levels of 51(8) per cent were achieved, equating to a mean decline in baseline perianastomotic Sto 2 of 37 per cent. Consequently, for further evaluation, only the 36 animals in which the more severe hypoxia model was applied were included.
There was no statistically significant difference between the different treatment groups with respect to baseline or immediate postoperative values. Sto 2 was significantly higher for PLGA–CPO than for Vicryl™ or PLGA anastomoses on day 1 (both P < 0·001), day 3 (P = 0·011 and P = 0·001 respectively) and day 7 (P = 0·038 and P < 0·001) (Fig. 3). Values were not significantly different between Vicryl™ and PLGA anastomoses, except on day 7 (P = 0·044) (Fig. 3). Mean(s.d.) perianastomotic Sto 2 values were 74(5), 52(8) and 54(11) per cent on day 1, 77(12), 60(12) and 55(11) per cent on day 3, and 83(5), 74(10) and 64(7) per cent on day 7 in animals where PLGA–CPO, Vicryl™ and PLGA were used respectively.
Figure 3.
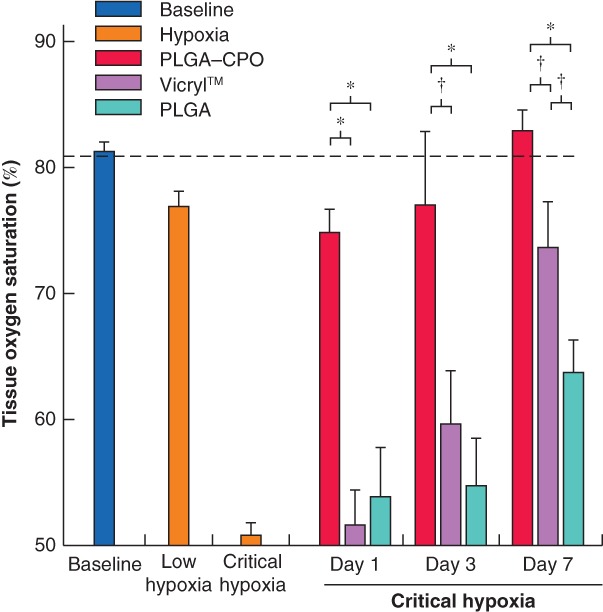
Increased tissue oxygen saturation in sutured anastomoses under critical hypoxia Ineffective induction of hypoxia (low hypoxia) resulted in little effect on perianastomotic tissue oxygen saturation (Sto 2) in anastomoses sutured with poly(d,l‐lactide‐co‐glycolide) (PLGA)–calcium peroxide (CPO), Vicryl™ or PLGA at all assessed time points. Under critical hypoxia, perianastomotic Sto 2 was significantly higher in anastomoses performed using oxygen‐releasing sutures compared with controls (Vicryl™ or PLGA) at all measured time points. Oxygen‐producing sutures were able to maintain Sto 2 close to the baseline level (dashed line). Values are mean(s.d.). Baseline, n = 69; low hypoxia, n = 33; critical hypoxia, n = 36; PLGA–CPO, Vicryl™ and PLGA, n = 4 each, per postoperative day. *P ≤ 0·001, †P < 0·050 (one‐way ANOVA and Student's t‐test contrasts).
Macroscopic and microscopic pathological assessment
At necropsy, no difference with respect to the extent of intra‐abdominal adhesions was observed between the treatment groups. Neither apparent leaks nor signs of peritonitis were noted in any animal in the study.
The CCD was increased significantly in animals where the anastomosis was performed using PLGA–CPO compared with Vicryl™ sutures on day 1 (P = 0·024), day 3 (P = 0·020) and day 7 (P = 0·006). No significant difference in CCD was found between PLGA–CPO and PLGA, or between Vicryl™ and PLGA anastomoses at any assessed postoperative time point. Mean(s.d.) CCD was 324(14), 283(24) and 296(40) μm on day 1, 396(10), 338(28) and 378(25) μm on day 3, and 437(16), 378(24) and 395(30) μm on day 7 for PLGA–CPO, Vicryl™ and PLGA respectively (Fig. 4).
Figure 4.
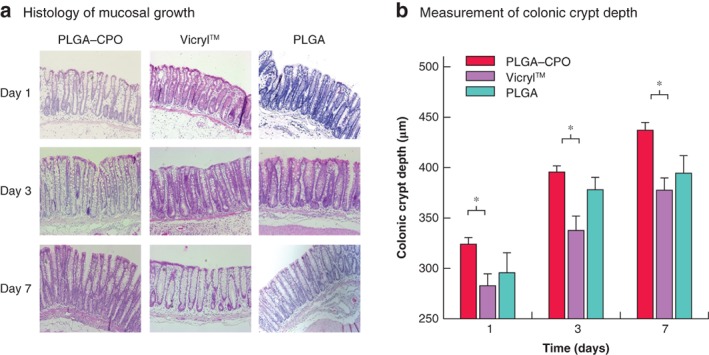
Colonic crypt depth a Histological examination demonstrated significantly more perianastomotic mucosal growth when using oxygen‐releasing compared with Vicryl™ sutures at all assessed postoperative time points. b Bar graph shows that there was no significant difference in mucosal thickness between poly(d,l‐lactide‐co‐glycolide) (PLGA)–calcium peroxide (CPO) and PLGA, or between PLGA and Vicryl™ anastomoses. Values are mean(s.d.). PLGA–CPO, Vicryl™ and PLGA, n = 4 each, per postoperative day. *P < 0·050 (one‐way ANOVA and Student's t‐test contrasts).
Mechanical stability of the anastomoses
BP evaluation revealed that bursting occurred in all animals at or near the anastomotic site. BP was significantly higher when anastomoses were performed using PLGA–CPO compared with Vicryl™ and PLGA sutures (day 1: P = 0·037 and P = 0·008; day 3: P = 0·025 and P = 0·010 respectively). On day 7, the BPs for PLGA–CPO anastomoses were similar to those for Vicryl™ and PLGA, with no significant difference. The BPs for Vicryl™ and PLGA anastomoses was not significantly different at any assessed postoperative time point. Mean(s.d.) BP was 91·3(10·3), 72·5(9·6) and 63·8(9·5) mmHg on day 1, 106·3(21·4), 67·5(15·0) and 61·3(11·1) mmHg on day 3, and 171·3(18·4), 165·0(9·1) and 158·8(6·3) mmHg on day 7 in PLGA–CPO, Vicryl™ and PLGA groups respectively (Fig. 5).
Figure 5.
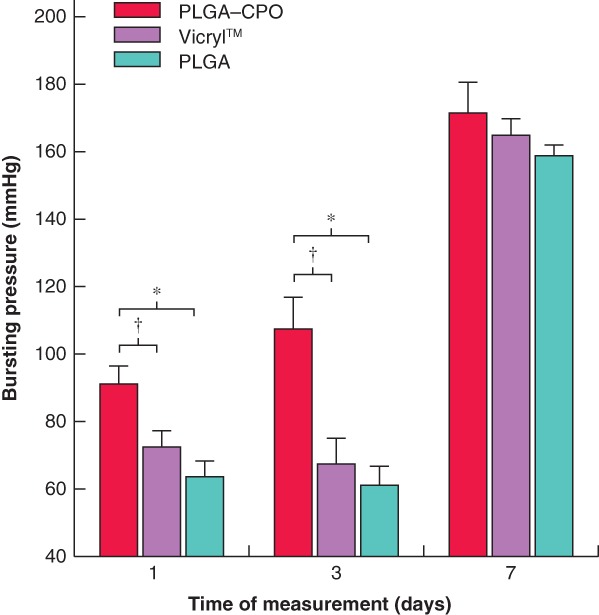
Mechanical stability of anastomoses Anastomoses performed using oxygen‐releasing suture material (poly(d,l‐lactide‐co‐glycolide) (PLGA)–calcium peroxide (CPO)) sustained significantly higher intraluminal air pressure before leakage than non‐oxygen‐releasing controls (Vicryl™ and PLGA) on days 1 and 3 after surgery. Values are mean(s.d.). PLGA–CPO, Vicryl™ and PLGA, n = 4 each, per postoperative day. *P ≤ 0·010, †P < 0·050 (one‐way ANOVA and Student's t‐test contrasts).
Tensile strength of the sutures in vivo
The tensile strength of the different suture materials used in this study was not significantly different on day 1. PLGA sutures appeared to sustain significantly more tensile stress than both PLGA–CPO (P = 0·019) and Vicryl™ (P < 0·001) sutures on day 3. On postoperative day 7, PLGA again showed significantly greater tensile strength than Vicryl™ (P < 0·001) but not PLGA–CPO (P = 0·057) sutures. Mean(s.d.) tensile strength was 561·5(48·8), 489·9(67·6) and 545·0(37·4) N/mm2 on day 1, 493·9(34·9), 445·9(34·0) and 555·6(31·9) N/mm2 on day 3, and 466·5(20·5), 412·4(47·7) and 530·6(61·2) N/mm2 on day 7 for PLGA–CPO, Vicryl™ and PLGA materials respectively.
Discussion
Anastomotic leakage after colorectal surgery remains a major concern of the surgical community. Sufficient oxygen supply to the restoring tissue is generally considered as one of the key factors of anastomotic healing. It therefore seems reasonable that suture material releasing supplemental oxygen directly into the wound bed might considerably improve the healing of anastomoses. With regard to oxygen and wound healing, there are concerns on the generation of potentially cytotoxic oxygen byproducts, referred to as reactive oxygen species (ROS). This study has shown that survival in vitro of 3 T3 fibroblast cultures was not impaired, and that IEC cells incubated with oxygen‐producing sutures, and cultured under hypoxic conditions, had contractility responses comparable to those of cells cultured under normoxia. These results confirm the findings of an earlier study22 that demonstrated a proliferative effect of hyperbaric oxygen (HBO) on murine 3 T3 fibroblasts, in spite of enhanced ROS production. However, ROS were demonstrated to play a beneficial key role in intestinal wound healing by stimulating cell attraction, migration and adhesion, and immune cell activation, effects that appear to be potentiated by the presence of commensal bacteria23, 24.
In the present in vivo studies, non‐invasively measured perianastomotic Sto 2 was significantly higher where oxygen‐producing suture material was used. While remaining significant, the difference became less notable over time. Given the maintenance of a high level of perianastomotic Sto 2 in animals receiving PLGA–CPO from the first postoperative day, the authors assume that the well known angiogenic effect of acute hypoxia25 might consequently be attenuated in these animals compared with that in the other two treatment groups. Furthermore, gradually increasing oxygen supply due to hypoxia‐driven neovascularization may explain the commensurate scaling down of the difference between the Sto 2 of Vicryl™ and PLGA–CPO anastomoses over 7 days.
Besides collagen formation, mucosal growth is a key factor in early healing of colonic anastomoses. The correlation between mucosal growth and mechanical stability of the anastomoses has been demonstrated in pertinent publications where anastomotic healing was assessed under different conditions18, 20. The present results show that mucosal thickness of the target segment of the colon was increased significantly in animals where the anastomosis was performed using oxygen‐producing suture material compared with the untreated off‐the‐shelf thread. However, there was no significant difference between PLGA–CPO and PLGA alone. The more traumatic properties of the irregular and somewhat spiky shaping of the coated PLGA–CPO and PLGA threads may have contributed to a more dramatic wound‐healing reaction, and hence mucosal proliferation, than observed with Vicryl™ anastomoses. Yet, as oxygen‐producing sutures were consistently associated with the most pronounced mucosal proliferation (expressed as the CCD) in all animals at every time point in these experiments, it can be assumed that oxygen also has an impact on mucosal proliferation, even though the present results do not allow precise quantification of this effect. In addition, no significant difference in mucosal thickness could be seen between the two non‐oxygen‐releasing suture materials. The exact mechanisms by which oxygen promotes mucosal growth are unknown. However, HBO has been shown to promote healing of oral mucosal flaps by enhancing wound vascular regeneration26, and is effective in the treatment of inflammatory bowel disease27, at least in part via stimulation of colonic stem cells28.
A commonly used quantitative method for studying experimental anastomotic repair is the measurement of BP, defined as the intraluminal pressure at which the anastomosis or adjacent colon disrupts, and which is sensitive to early changes in anastomotic healing29. In the present study, the mechanical stability of PLGA–CPO anastomoses was significantly better than that of control anastomoses (Vicryl™ or PLGA) at day 1 and 3. These results support the positive effect of supplemental oxygen administration on the healing and mechanical stability of colonic anastomoses reported by others30, 31.
Effects of oxygen on wound healing are manifold and may be of particular importance in a highly contaminated environment such as a colonic anastomosis. Oxygen enhances fibroblast migration and replication32, increases the rate of collagen production and tensile strength of collagen fibres33, and promotes macrophage chemotaxis34. Furthermore, oxygen is considered to enhance antibacterial activities of leucocytes, including phagocytic function35, which may promote the removal of cell debris and thereby promote cleaning of the wound bed. Physiological wound healing (following a surgical trauma) includes the same processes36 without the boost of supplemental oxygen in the early healing phase. This might explain why mechanical stability is still higher for PLGA–CPO anastomoses compared with controls, but no longer significant at day 7.
Differences in resistance of the anastomoses to mechanical stress cannot be explained by the physical properties of the diverse suture materials themselves, thus emphasizing the positive effect of supplemental oxygen on enhanced healing and increased mechanical stability. Although the tensile strength of subcutaneously implanted PLGA sutures was maintained at a constant level over 7 days, substantial degradation of PLGA–CPO and Vicryl™ sutures can be observed in vivo within 1 week. PLGA coating seems to render the suture material more resistant, resulting in significantly greater tensile strength compared with unprepared Vicryl™ and with PLGA–CPO sutures. However, the production of oxygen and its byproducts by PLGA–CPO threads may alter their chemical and/or physical properties, and subsequently entail faster degradation compared with that of the PLGA‐only coated product.
The results of the present study demonstrate the potential impact of locally administered oxygen on healing of experimental colonic anastomoses. However, the lack of blinding of the investigator who gathered the results to the suture used per animal is a limitation of the study. Furthermore, corresponding to the 3R (replace, reduce, refine) principles, the authors tried to reduce the number of animals used in this experiment to a minimum. Accordingly, only four animals per group per day were included. This low sample size may increase the risk of a statistical type II error, and thus of false‐negative results. Despite the low number of animals used, a significant positive effect of oxygen was shown for several aspects of anastomotic healing. Even though clinical outcome measures, such as signs of anastomotic leakage and presence of abdominal adhesions, were assessed at the time of death, they were not recorded and analysed systematically. This can rightly be regarded as a flaw of the study. The authors did not include these data in the statistical analysis, as from previous experience18, 20 a substantial difference in the clinically apparent leakage rate would not be expected between the different treatment groups in the experimental model used. Moreover, adhesions are not supposed to develop within the first 7 days after surgery, unless there is contained leakage or intra‐abdominal sepsis. The authors' expectations were confirmed in this study. Another limitation of the study is the relatively short follow‐up of 7 days, which reflects, but does not encompass, the critical time frame for the occurrence of clinically apparent leakage (normally between postoperative day 5 and 10). However, even though a leak might not yet be clinically evident at postoperative day 7, it can be assumed that the leak would have developed and, therefore, should be visible at death. Most, if not all, experimental studies on healing of intestinal anastomoses schedule the follow‐up within the first postoperative week18, 30, 37.
Oesophageal and colorectal anastomoses are considered to be the most critical ones in visceral surgery, and are commonly performed using stapler devices rather than sutures. Loading of the oxygen‐producing nano‐crystals on suture instead of stapler material was chosen for the simple reason of availability of threads usable in an animal model, whereas no established model of such small‐scale stapled anastomoses is available in rats. A next step in developing a tool that might find its way into clinical practice would be to establish staplers with oxygen‐producing capacity.
The manufacture of oxygen‐producing suture material amenable to surgical use is feasible. Possible oxygen byproducts do not significantly affect cell survival and proliferation in vitro or wound healing in vivo. Under hypoxic conditions, oxygen‐producing sutures yield significantly higher perianastomotic Sto 2, a tendency towards better mucosal healing, and enhanced mechanical stability of the anastomoses compared with non‐oxygen‐producing controls. Oxygen‐producing materials might be a way to reduce the risk of anastomotic leakage under critical conditions, such as tissue hypoxia.
Acknowledgements
The authors thank K. Feldman, Department of Materials, Swiss Federal Institute of Technology, Zürich (ETHZ), for his help with the tensile strength analysis. They also thank D. Balmer for critical review of the manuscript. This study was funded partly by the Insula Foundation, Bern, Switzerland.
Disclosure: The authors declare no conflict of interest.
Presented in part to the Third TERMIS World Congress 2012, Vienna, Austria, September 2012
Funding information
Insula Stiftung
References
- 1. Kang CY, Halabi WJ, Chaudhry OO, Nguyen V, Pigazzi A, Carmichael JC et al Risk factors for anastomotic leakage after anterior resection for rectal cancer. JAMA Surg 2013; 148: 65–71. [DOI] [PubMed] [Google Scholar]
- 2. Krarup PM, Nordholm‐Carstensen A, Jorgensen LN, Harling H. Association of comorbidity with anastomotic leak, 30‐day mortality, and length of stay in elective surgery for colonic cancer: a nationwide cohort study. Dis Colon Rectum 2015; 58: 668–676. [DOI] [PubMed] [Google Scholar]
- 3. Bakker IS, Grossmann I, Henneman D, Havenga K, Wiggers T. Risk factors for anastomotic leakage and leak‐related mortality after colonic cancer surgery in a nationwide audit. Br J Surg 2014; 101: 424–432. [DOI] [PubMed] [Google Scholar]
- 4. Posma LA, Bleichrodt RP, van Goor H, Hendriks T. Transient profound mesenteric ischemia strongly affects the strength of intestinal anastomoses in the rat. Dis Colon Rectum 2007; 50: 1070–1079. [DOI] [PubMed] [Google Scholar]
- 5. Attard JA, Raval MJ, Martin GR, Kolb J, Afrouzian M, Buie WD et al The effects of systemic hypoxia on colon anastomotic healing: an animal model. Dis Colon Rectum 2005; 48: 1460–1470. [DOI] [PubMed] [Google Scholar]
- 6. Kruschewski M, Rieger H, Pohlen U, Hotz HG, Buhr HJ. Risk factors for clinical anastomotic leakage and postoperative mortality in elective surgery for rectal cancer. Int J Colorectal Dis 2007; 22: 919–927. [DOI] [PubMed] [Google Scholar]
- 7. Vignali A, Gianotti L, Braga M, Radaelli G, Malvezzi L, Di Carlo V. Altered microperfusion at the rectal stump is predictive for rectal anastomotic leak. Dis Colon Rectum 2000; 43: 76–82. [DOI] [PubMed] [Google Scholar]
- 8. Emir S, Gurdal SO, Sozen S, Bali I, Yesildag E, Celik A et al Does hyperbaric oxygen therapy reduce the effects of ischemia on colonic anastomosis in laparoscopic colon resection? Ann Ital Chir 2016; 87: 83–91. [PubMed] [Google Scholar]
- 9. Kimberger O, Fleischmann E, Brandt S, Kugener A, Kabon B, Hiltebrand L et al Supplemental oxygen, but not supplemental crystalloid fluid, increases tissue oxygen tension in healthy and anastomotic colon in pigs. Anesth Analg 2007; 105: 773–779. [DOI] [PubMed] [Google Scholar]
- 10. Kemik O, Adas G, Arikan S, Gurluler E, Dogan Y, Toklu AS et al Evaluation of the effects of hyperbaric oxygen treatment and enoxaparin on left colon anastomosis. An experimental study. Eur Rev Med Pharmacol Sci 2013; 17: 2286–2292. [PubMed] [Google Scholar]
- 11. Schietroma M, Carlei F, Cecilia EM, Piccione F, Bianchi Z, Amicucci G. Colorectal Infraperitoneal anastomosis: the effects of perioperative supplemental oxygen administration on the anastomotic dehiscence. J Gastrointest Surg 2012; 16: 427–434. [DOI] [PubMed] [Google Scholar]
- 12. Oh SH, Ward CL, Atala A, Yoo JJ, Harrison BS. Oxygen generating scaffolds for enhancing engineered tissue survival. Biomaterials 2009; 30: 757–762. [DOI] [PubMed] [Google Scholar]
- 13. Benz S, Nötzli S, Siegel JS, Eberli D, Jessen HJ. Controlled oxygen release from pyridone endoperoxides promotes cell survival under anoxic conditions. J Med Chem 2013; 56: 10 171–10 182. [DOI] [PubMed] [Google Scholar]
- 14. Zheng L, Kelly CJ, Colgan SP. Physiologic hypoxia and oxygen homeostasis in the healthy intestine. A review in the theme: cellular responses to hypoxia. Am J Physiol Cell Physiol 2015; 309: C350–C360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol 2010; 8: e1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Charan J, Kantharia ND. How to calculate sample size in animal studies? J Pharmacol Pharmacother 2013; 4: 303–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Hamzaoğlu I, Karahasanoğlu T, Aydin S, Sahin DA, Carkman S, Sariyar M et al The effects of hyperbaric oxygen on normal and ischemic colon anastomoses. Am J Surg 1998; 176: 458–461. [DOI] [PubMed] [Google Scholar]
- 18. Inglin RA, Baumann G, Wagner OJ, Candinas D, Egger B. Insulin‐like growth factor I improves aspects of mycophenolate mofetil‐impaired anastomotic healing in an experimental model. Br J Surg 2008; 95: 793–798. [DOI] [PubMed] [Google Scholar]
- 19. Dekaney CM, Fong JJ, Rigby RJ, Lund PK, Henning SJ, Helmrath MA. Expansion of intestinal stem cells associated with long‐term adaptation following ileocecal resection in mice. Am J Physiol Gastrointest Liver Physiol 2007; 293: G1013–G1022. [DOI] [PubMed] [Google Scholar]
- 20. Egger B, Inglin R, Zeeh J, Dirsch O, Huang Y, Büchler MW. Insulin‐like growth factor I and truncated keratinocyte growth factor accelerate healing of left‐sided colonic anastomoses. Br J Surg 2001; 88: 90–98. [DOI] [PubMed] [Google Scholar]
- 21. Farías‐Llamas OA, Orozco‐Mosqueda A, Portilla‐del Buen E, Leal‐Cortés CA, Ruiz‐Chávez IE, González‐Ojeda A. [Bursting pressure in normal and ischemic colonic anastomoses in rats; using biological and synthetic adhesives.] Cir Cir 2005; 73: 31–42. [PubMed] [Google Scholar]
- 22. Conconi MT, Baiguera S, Guidolin D, Furlan C, Menti AM, Vigolo S et al Effects of hyperbaric oxygen on proliferative and apoptotic activities and reactive oxygen species generation in mouse fibroblast 3T3/J2 cell line. J Investig Med 2003; 51: 227–232. [DOI] [PubMed] [Google Scholar]
- 23. Cordeiro JV, Jacinto A. The role of transcription‐independent damage signals in the initiation of epithelial wound healing. Nat Rev Mol Cell Biol 2013; 14: 249–262. [DOI] [PubMed] [Google Scholar]
- 24. Swanson PA II, Kumar A, Samarin S, Vijay‐Kumar M, Kundu K, Murthy N et al Enteric commensal bacteria potentiate epithelial restitution via reactive oxygen species‐mediated inactivation of focal adhesion kinase phosphatases. Proc Natl Acad Sci U S A 2011; 108: 8803–8808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Howard MA, Asmis R, Evans KK, Mustoe TA. Oxygen and wound care: a review of current therapeutic modalities and future direction. Wound Repair Regen 2013; 21: 503–511. [DOI] [PubMed] [Google Scholar]
- 26. Helmers R, Milstein DM, van Hulst RA, de Lange J. Hyperbaric oxygen therapy accelerates vascularization in keratinized oral mucosal surgical flaps. Head Neck 2014; 36: 1241–1247. [DOI] [PubMed] [Google Scholar]
- 27. Dulai PS, Gleeson MW, Taylor D, Holubar SD, Buckey JC, Siegel CA. Systematic review: the safety and efficacy of hyperbaric oxygen therapy for inflammatory bowel disease. Aliment Pharmacol Ther 2014; 39: 1266–1275. [DOI] [PubMed] [Google Scholar]
- 28. Bekheit M, Baddour N, Katri K, Taher Y, El Tobgy K, Mousa E. Hyperbaric oxygen therapy stimulates colonic stem cells and induces mucosal healing in patients with refractory ulcerative colitis: a prospective case series. BMJ Open Gastroenterol 2016; 3: e000082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Månsson P, Zhang XW, Jeppsson B, Thorlacius H. Anastomotic healing in the rat colon: comparison between a radiological method, breaking strength and bursting pressure. Int J Colorectal Dis 2002; 17: 420–425. [DOI] [PubMed] [Google Scholar]
- 30. Boersema GS, Wu Z, Kroese LF, Vennix S, Bastiaansen‐Jenniskens YM, van Neck JW et al Hyperbaric oxygen therapy improves colorectal anastomotic healing. Int J Colorectal Dis 2016; 31: 1031–1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Poyrazoglu Y, Topal T, Yuksel R, Bircan FS, Simsek K, Gocgeldi E et al Effects of hyperbaric oxygen and preconditioning on wound healing in colonic anastomoses. J Invest Surg 2015; 28: 188–195. [DOI] [PubMed] [Google Scholar]
- 32. Knighton DR, Silver IA, Hunt TK. Regulation of wound‐healing angiogenesis‐effect of oxygen gradients and inspired oxygen concentration. Surgery 1981; 90: 262–270. [PubMed] [Google Scholar]
- 33. Gailit J, Clark RA. Wound repair in the context of extracellular matrix. Curr Opin Cell Biol 1994; 6: 717–725. [DOI] [PubMed] [Google Scholar]
- 34. Bosco MC, Delfino S, Ferlito F, Puppo M, Gregorio A, Gambini C et al The hypoxic synovial environment regulates expression of vascular endothelial growth factor and osteopontin in juvenile idiopathic arthritis. J Rheumatol 2009; 36: 1318–1329. [DOI] [PubMed] [Google Scholar]
- 35. Hohn DC, MacKay RD, Halliday B, Hunt TK. Effect of O2 tension on microbicidal function of leukocytes in wounds and in vitro. Surg Forum 1976; 27: 18–20. [PubMed] [Google Scholar]
- 36. Broughton G II, Janis JE, Attinger CE. Wound healing: an overview. Plast Reconstr Surg 2006; 117(Suppl): 1e‐S–32e‐S. [DOI] [PubMed] [Google Scholar]
- 37. Pommergaard HC, Achiam MP, Burcharth J, Rosenberg J. Impaired blood supply in the colonic anastomosis in mice compromises healing. Int Surg 2015; 100: 70–76. [DOI] [PMC free article] [PubMed] [Google Scholar]


