Chronic obstructive pulmonary disease (COPD) is the third leading cause of death worldwide (1). The main risk factor for COPD in developed countries is smoking cigarettes. Inhaling cigarette smoke (CS) leads to different pulmonary pathologies, including emphysema, which contributes significantly to the poorly reversible airflow obstruction that is characteristic of COPD (2). The proteinase/antiproteinase hypothesis for emphysema development was formulated in the 1960s and postulates that CS increases the lung burden of proteinases to exceed the lung antiproteinase defense (2). However, in 2000, the “vascular hypothesis” emerged based on studies showing that 1) pharmacologic inhibition of VEGF (vascular endothelial growth factor) signaling via its receptors on endothelial cells (ECs), which is a crucial prosurvival pathway in these cells, leads rapidly to emphysema development in rats (3); and 2) pulmonary EC death occurs in COPD lungs associated with reduced lung levels of VEGF and its receptors (3). The vascular hypothesis proposes that components of CS that are absorbed into the circulation trigger apoptosis of pulmonary ECs (a key component of the alveolar septae), causing destruction of the alveolar walls (3).
Increased intracellular levels of ceramide (a second-messenger glycolipid molecule) were later linked to alveolar septal cell apoptosis in animal models of emphysema and human emphysematous lungs (4). Ceramide sits at a central hub that determines cell death or survival. Proapoptotic ceramide is enzymatically synthesized from serine and palmitoyl-CoA (coenzyme A) or is generated by enzymatic cleavage of sphingomyelin (a cell membrane component) or other pathways (Figure 1A). Cell survival is promoted when intracellular ceramide levels are reduced by the conversion of ceramide to sphingosine-1-phosphate or metabolites of ceramide that have been glycosylated (glycosphingolipids [GSLs], including glucosylceramide [GlcCer]) by the actions of GCS (glucosylceramide synthase) (5, 6) (Figure 1A). The GCS–GlcCer pathway has not been robustly evaluated in the pathogenesis of emphysema, but it has the potential to contribute, as Gcs−/− mice die before birth from massive apoptosis (7), and GlcCer mediates the prosurvival effects of VEGF on ECs partly by inhibiting autophagy-mediated cell death (8).
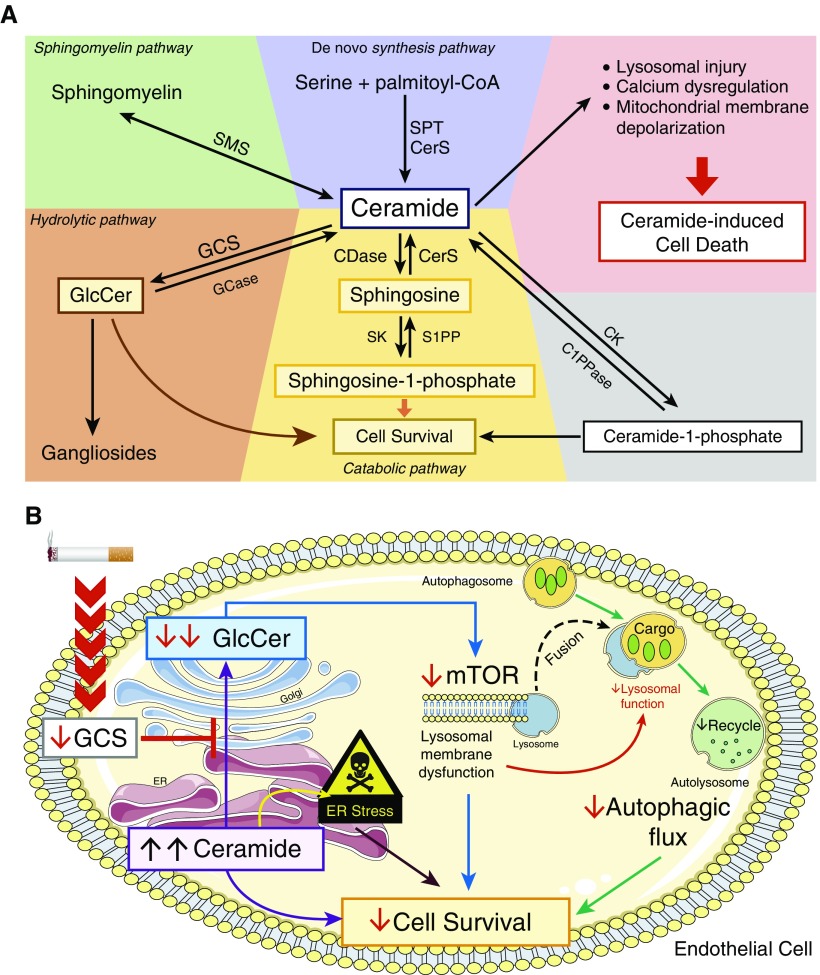
Figure 1.
(A) Overview of ceramide metabolism. Ceramide is an intracellular hub that controls cell death and survival in endothelial cells. Ceramide accumulation in the cell leads to ceramide-induced cell death. Ceramide is enzymatically synthesized de novo from serine and palmitoyl-CoA (coenzyme A) via the actions of SPT (serine palmitoyl-CoA transferase) and CerS (ceramide synthase) (the de novo synthesis pathway [blue panel]). Ceramide is also generated by other metabolic pathways, including from 1) sphingomyelin by SMS (sphingomyelinases; the sphingomyelin pathway [green panel]); 2) glucosylceramide (GlcCer) via the actions of GCase (β-glucosidase; the salvage pathway [orange panel]); 3) ceramide-1-phosphate phosphatase via the actions of C1PPase (ceramide-1-phosphate phosphatase; alternate pathway [gray panel]); and 4) sphingosine via the actions of CerS (the catabolic pathway [yellow panel]). However, ceramide is enzymatically converted to metabolites that promote cell survival, including 1) GlcCer, which is synthesized by GCS (glucosylceramide synthase); 2) sphingosine-1-phosphate, which is synthesized by SK (sphingosine kinase); and 3) ceramide-1-phosphate, which is synthesized by CK (ceramide kinase). In their study, Koike and colleagues explored the hydrolytic pathway, in which proapoptotic ceramide is converted to GlcCer (which is a glycosylated prosurvival signaling molecule) by the enzyme GCS. (B) Mechanism by which cigarette smoke (CS) induces pulmonary endothelial cell death by reducing the conversion of ceramide to GlcCer (glycosylceramide) to induce autophagic cell death. CS reduces the levels and/or activity of GCS in pulmonary endothelial cells, which leads to 1) increased intracellular levels of proapoptotic ceramide, and 2) reduced generation of GlcCer (a prosurvival mediator) in pulmonary endothelial cells. Reduced intracellular GlcCer levels lead to decreased activation of the mTOR pathway, which causes lysosomal membrane dysfunction (possibly via impaired fusion of lysosomes with autophagosomes) and failure to adequately acidify autolysosomes, which impairs autophagic flux and induces autophagic cell death. Accumulation of ceramide in the endoplasmic reticulum (ER) leads to ER stress, resulting in apoptosis. Thus, CS-induced reductions in intracellular GlcCer levels reduce cell survival by impairing autophagic flux to trigger autophagic cell death and promoting apoptosis via ceramide-induced ER stress. CDase = ceramidase; S1PP = sphingosine-1-phosphate phosphatase.
Autophagy is a normal homeostatic process by which organelles, proteins, and other cellular components (cargo) are recycled in several steps (autophagic flux; Figure 1B). Autophagosomes are assembled from invaginations of cellular membranes into which cargo is loaded from the cytosol. Loaded autophagosomes fuse with lysosomes, leading to acidification of the resulting autolysosomes and this permits degradation of the cargo by acidic proteinases such as cathepsin B (9). mTOR (mammalian target of rapamycin) is a negative regulator of autophagy (9), whereas endoplasmic reticulum (ER) stress promotes autophagy by inhibiting mTOR activation. However, when autophagic flux is excessive or impaired, this results in autophagic cell death (10). CS exposure triggers autophagy in various cells, and excessive autophagy-mediated cell death is linked to emphysema development (11).
Whether the GCS–GlcCer pathway contributes to the pathogenesis of emphysema by inhibiting autophagic cell death via effects on the mTOR pathway and/or lysosomal function is not known. In a study reported in this issue of the Journal, Koike and colleagues (pp. 1113–1125) addressed these issues by testing a GSC inhibitor, and using GCS silencing and overexpression approaches on ECs to test the hypotheses that CS exposure reduces GCS and GlcCer levels in ECs, and that reduced GCS and GlcCer levels lead to reduced mTOR signaling, impaired lysosomal function, and autophagic flux, thereby increasing EC death (12). They also investigated whether increasing GlcCer levels in cells ameliorates these processes to enhance cell survival in CS-exposed ECs.
Using liquid chromatography with tandem mass spectrometry, the authors showed that lung GlcCer levels were decreased in mice with elastase- and CS-induced emphysema, and correlated indirectly with alveolar airspace size. GCS levels were lower in the lungs of patients with COPD than in never-smoker control subjects. Using a pharmacologic inhibitor of GCS (Genz-123346), or silencing GCS expression using siRNA techniques in primary human lung microvascular ECs (HLMVECs), they showed that reducing intracellular GCS levels increased intracellular ceramide levels and led to increased ER stress levels (which triggers apoptosis of ECs). This reduction in intracellular GCS levels also decreased mTOR activation, impaired acidification of autolysosomes, reduced cathepsin B activity in the autolysosomal, impaired cleavage of cargo, and caused autophagy-induced death of HLMVECs (Figure 1B). Exposing HLMVECs to CS recapitulated the effects of GCS inhibition, including impaired autophagic flux. Overexpression of GCS or addition of exogenous GlcCer to HLMVECs reduced autophagic cell death. The authors conclude that CS causes emphysema, in part, by reducing activation of the GCS–GlcCer pathway in ECs. This leads to reduced activation of mTOR, which induces impaired lysosomal degradation of cargo (possibly by impairing lysosomal fusion with autophagosomes), leading to autophagy-mediated death of ECs.
The study has several strengths. The authors used complementary animal models of emphysema, studies of human lungs, human EC culture systems, and multiple approaches (overexpression, siRNA techniques, and pharmacological inhibition) to interrogate the GCS–GlcCer pathway. They linked the GCS–GlcCer pathway to mTOR signaling, ER stress, and autophagy for the first time. The results are consistent with previous findings that plasma GSL levels are indirectly related to emphysema severity (13). A limitation of the study is that the molecular mechanisms by which CS exposure reduces GCS and GlcCer lung levels to induce ER stress (to trigger apoptosis of ECs), and also to reduce mTOR signaling, impair lysosomal function, and induce autophagy-mediated death of ECs, were not identified. Lung GCS levels could be reduced by epigenetic silencing of GCS or oxidative stress–induced loss of GCS activity in COPD lungs (14). The human cohort that was studied was small, smokers were not included as controls, the effects of current smoking on GCS and GlcCer levels were not measured, and GCS and GlcCer levels were not related to COPD severity or activity. The GCS–GlcCer pathway was not evaluated in lung samples from patients with COPD and phenotypes other than emphysema. The study also focused exclusively on the GCS–GlcCer pathway in ECs, but the death/survival of epithelial cells, leukocytes, and fibroblasts in the small airways of patients with COPD also contributes to the pathogenesis of this disease (15).
Overall, this novel study highlights potential protective activities for GCS and its “sugarcoated” lipid product, GlcCer, in the CS-exposed lung. The results suggest that augmenting lung levels of GCS or GlcCer has potential as a novel therapeutic approach for patients with emphysema. Studies of CS-exposed mice in which GCS is overexpressed in ECs are warranted to determine whether this approach prevents or limits disease progression. Studies of the GCS–GlcCer pathway in large human COPD cohorts are needed to validate this pathway as a therapeutic target and/or biomarker for emphysema.
Supplementary Material
Footnotes
Originally Published in Press as DOI: 10.1164/rccm.201906-1254ED on July 12, 2019
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Mannino DM, Buist AS. Global burden of COPD: risk factors, prevalence, and future trends. Lancet. 2007;370:765–773. doi: 10.1016/S0140-6736(07)61380-4. [DOI] [PubMed] [Google Scholar]
- 2.Owen CA. Roles for proteinases in the pathogenesis of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2008;3:253–268. doi: 10.2147/copd.s2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kasahara Y, Tuder RM, Taraseviciene-Stewart L, Le Cras TD, Abman S, Hirth PK, et al. Inhibition of VEGF receptors causes lung cell apoptosis and emphysema. J Clin Invest. 2000;106:1311–1319. doi: 10.1172/JCI10259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Petrache I, Kamocki K, Poirier C, Pewzner-Jung Y, Laviad EL, Schweitzer KS, et al. Ceramide synthases expression and role of ceramide synthase-2 in the lung: insight from human lung cells and mouse models. PLoS One. 2013;8:e62968. doi: 10.1371/journal.pone.0062968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ogretmen B. Sphingolipid metabolism in cancer signalling and therapy. Nat Rev Cancer. 2018;18:33–50. doi: 10.1038/nrc.2017.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cuvillier O, Pirianov G, Kleuser B, Vanek PG, Coso OA, Gutkind S, et al. Suppression of ceramide-mediated programmed cell death by sphingosine-1-phosphate. Nature. 1996;381:800–803. doi: 10.1038/381800a0. [DOI] [PubMed] [Google Scholar]
- 7.Yamashita T, Wada R, Sasaki T, Deng C, Bierfreund U, Sandhoff K, et al. A vital role for glycosphingolipid synthesis during development and differentiation. Proc Natl Acad Sci USA. 1999;96:9142–9147. doi: 10.1073/pnas.96.16.9142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Giussani P, Bassi R, Anelli V, Brioschi L, De Zen F, Riccitelli E, et al. Glucosylceramide synthase protects glioblastoma cells against autophagic and apoptotic death induced by temozolomide and Paclitaxel. Cancer Invest. 2012;30:27–37. doi: 10.3109/07357907.2011.629379. [DOI] [PubMed] [Google Scholar]
- 9.Mizumura K, Choi AM, Ryter SW. Emerging role of selective autophagy in human diseases. Front Pharmacol. 2014;5:244. doi: 10.3389/fphar.2014.00244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Festa BP, Chen Z, Berquez M, Debaix H, Tokonami N, Prange JA, et al. Impaired autophagy bridges lysosomal storage disease and epithelial dysfunction in the kidney. Nat Commun. 2018;9:161. doi: 10.1038/s41467-017-02536-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vij N, Chandramani-Shivalingappa P, Van Westphal C, Hole R, Bodas M. Cigarette smoke-induced autophagy impairment accelerates lung aging, COPD-emphysema exacerbations and pathogenesis. Am J Physiol Cell Physiol. 2018;314:C73–C87. doi: 10.1152/ajpcell.00110.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Koike K, Berdyshev EV, Mikosz AM, Bronova IA, Bronoff AS, Jung JP, et al. Role of glucosylceramide in lung endothelial cell fate and emphysema. Am J Respir Crit Care Med. 2019;200:1113–1125. doi: 10.1164/rccm.201812-2311OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bowler RP, Jacobson S, Cruickshank C, Hughes GJ, Siska C, Ory DS, et al. Plasma sphingolipids associated with chronic obstructive pulmonary disease phenotypes. Am J Respir Crit Care Med. 2015;191:275–284. doi: 10.1164/rccm.201410-1771OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee KW, Pausova Z. Cigarette smoking and DNA methylation. Front Genet. 2013;4:132. doi: 10.3389/fgene.2013.00132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Domej W, Oettl K, Renner W. Oxidative stress and free radicals in COPD--implications and relevance for treatment. Int J Chron Obstruct Pulmon Dis. 2014;9:1207–1224. doi: 10.2147/COPD.S51226. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.