Idiopathic pulmonary fibrosis (IPF) and non–small cell lung cancer (NSCLC) share important risk factors, including advanced age and cigarette smoking. Moreover, evidence of the efficacy of nintedanib, a small-molecule tyrosine kinase inhibitor, for slowing the progression of IPF (1) as well as for treating NSCLC has fueled hopes that similar epithelial “transformation” profiles in cancer and fibrosis might support the repurposing of other cancer therapies for treatment of IPF (2, 3). However, the potential usefulness of nintedanib for treating IPF and NSCLC belies critical differences in the pathobiology of these two disease processes. Although the development of NSCLC is characterized by the accumulation of characteristic somatic genetic alterations in lung epithelial cells, including in TP53, KRAS, and EGFR, leading to the expansion of malignant clones and ultimately metastasis (4), there is no evidence that somatic mutations, clonal epithelial cell expansion, or metastasis are relevant to the biology of IPF. Despite these differences, the growing array of small molecules available for the treatment of cancer is attractive as a potential source of therapies for IPF; however, how to best determine which ones might offer the most promise remains an open question.
In this issue of the Journal, Ulke and colleagues (pp. 713–726) tackle this question by performing a gene set enrichment analysis of publically available NSCLC and IPF data sets (5). Starting with genes that were upregulated in patients with NSCLC compared with control subjects, the authors identified a set of 92 genes that were shared with IPF. These genes were more strongly enriched in alveolar epithelial type 2 (AT2) cells isolated from mouse lungs injured with bleomycin or from patients with IPF than fibroblasts isolated from the same patients, perhaps consistent with the epithelial origins of NSCLC.
Using standard gene set analysis databases (Gene Ontology, Kyoto Encyclopedia of Genes and Genomes, and Reactome), the authors found that the upregulated genes that were most shared by NSCLC and IPF were largely associated with mitosis/cell-cycle control. ECT2 (epithelial cell transformation-2), a guanine nucleotide exchange factor (i.e., activator) for Rho-family GTPases, was chosen for further analysis because of its previously described oncogenic activity and established roles in cytokinesis and ERK (extracellular-signal–regulated kinase) signaling (6). The authors nicely verified upregulation of ECT2 protein in the alveolar epithelium of patients with IPF, in what appeared to be large hyperplastic AT2 cells coexpressing the proliferation marker PCNA (proliferating cell nuclear antigen). They also found that primary AT2 cells (isolated from bleomycin-injured mice and manifesting elevated ECT2 expression) showed enhanced DNA content by flow analysis, as well as synthesis (S-phase cyclin D1 expression). Conversely, transient knockdown of ETC2 in these same cells reduced DNA synthesis. These data certainly raise the possibility that ECT2 may contribute to the proliferative capacity of NSCLCs and IPF-associated AT2 cells. But is there another way to view these data, perhaps also accommodating the abundant literature demonstrating epithelial senescence in IPF (7, 8)?
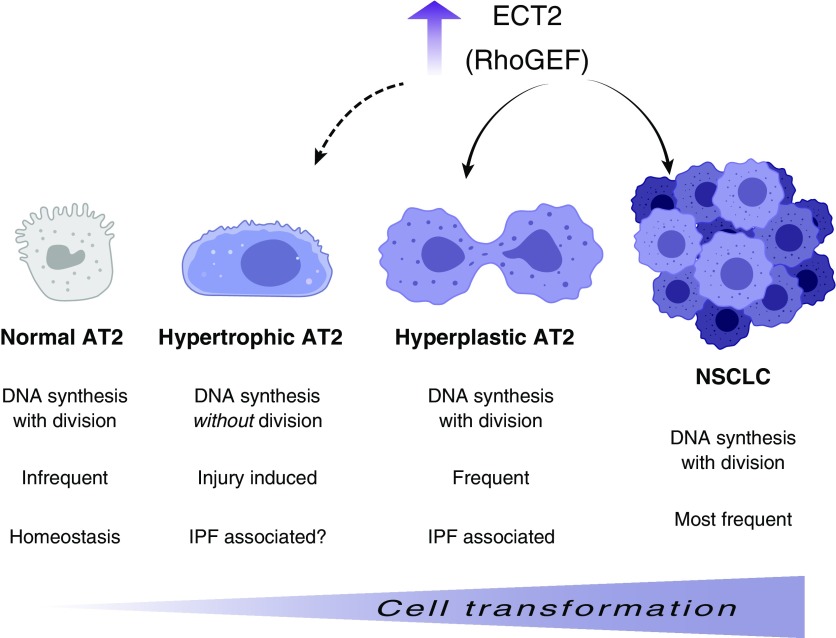
Although the upregulated genes shared by NSCLC and IPF are enriched in those that control the “mitosis/cell cycle,” it is important to recognize that such identifiers do not necessarily mean that the cells are proliferating. Indeed, an emerging field of research reveals that tissue injury can lead to cells doubling their DNA content but not dividing. This process, known as endoreplication, enables cells to become polyploid, which leads to increased cell size and genetic variability, features that favor adaptation to chronic stress and tissue damage (9, 10). In this context, the findings of Ulke and colleagues clearly show that ECT2 contributes to DNA synthesis (e.g., 5-ethynyl-2′-deoxyuridine incorporation, DNA content by propidium iodide staining, and colabeling with the S-phase marker PCNA), but it remains an open question whether this upregulation uniformly leads to a greater proliferative capacity of IPF-associated AT2 cells. One way to assess whether cells in tissues are truly in mitosis is to stain for the serine pHH3 (10-phosphorylated histone H3), in addition to other markers for DNA replication, such as PCNA and Ki67. Indeed, one recent study found no evidence of pHH3 staining in the alveolar epithelial component of IPF tissue, and the few positive cells were apparently nonepithelial (11). One wonders, therefore, about the degree to which AT2 cells in IPF are hyperplastic (i.e., dividing) versus hypertrophic (i.e., undergoing S-phase but not dividing). This distinction matters because polyploid cells are typically end-stage, inefficiently giving rise to new cells. Polyploid cells that do manage to segregate their chromosomes often make mistakes, leading to chromosome rearrangements that can drive carcinogenesis (12). In this way, polyploid AT2 cells might also contribute to the higher rates of lung cancer associated with IPF (Figure 1).
Figure 1.
Hypothetical model for ECT2 (epithelial cell transformation-2) upregulation and alveolar epithelial type 2 (AT2) cell transformation. Ulke and colleagues show that the ECT2 Rho guanine nucleotide exchange factor (RhoGEF) is upregulated in both idiopathic pulmonary fibrosis (IPF)-associated hyperplastic AT2 cells and non–small cell lung cancer (NSCLC; solid arrows), suggesting a common proliferative mechanism that may be targeted for future therapy. Evidence that ECT2 positively contributes to DNA synthesis raises the possibility that it may also contribute to the formation of IPF-associated hypertrophic AT2 cells, which endoreplicate their DNA but do not divide (dashed arrow). Polyploid, hypertrophic AT2 cells that ultimately manage to divide may segregate their DNA imperfectly, leading to AT2 cell transformation and cancer. Graphics courtesy of BioRender.
It is worth noting that Spek and Duitman recently undertook a similar analysis of differentially expressed genes in IPF and lung cancer, but also examined the downregulated genes as well as those oppositely regulated in IPF versus NSCLC (13). This broader analysis supports the view that IPF and NSCLC are largely transcriptomically divergent, although a similarly small group of upregulated genes were shared by IPF and NSCLC (n = 123). Although the gene set analysis by Spek and Duitman highlighted collagen organization, catabolism, and adhesion processes as commonly upregulated in IPF and NSCLCs, the study by Ulke and colleagues also identified a number of upregulated matrix metalloproteinases. Given longstanding evidence that integrin-based matrix adhesions are key upstream regulators of cell-cycle progression (14), it seems likely that the small number of upregulated genes shared by IPF and cancer will shed important light on the similarities and differences between these two diseases.
Ulke and colleagues used their gene set analysis of common differentially regulated genes between IPF and NSCLC to identify a molecular target for developing novel therapies for fibrosis and cancer. Their linkage of exploratory genomic analyses to mechanistic studies nicely demonstrates how to extend systems biology approaches beyond mere hypothesis generation, and proves that even in the case of divergent diseases like IPF and NSCLC, focusing on their commonalities may be fruitful.
Supplementary Material
Footnotes
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Richeldi L, du Bois RM, Raghu G, Azuma A, Brown KK, Costabel U, et al. INPULSIS Trial Investigators. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370:2071–2082. doi: 10.1056/NEJMoa1402584. [DOI] [PubMed] [Google Scholar]
- 2.Vancheri C. Idiopathic pulmonary fibrosis and cancer: do they really look similar? BMC Med. 2015;13:220. doi: 10.1186/s12916-015-0478-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yoon JH, Nouraie M, Chen X, Zou RH, Sellares J, Veraldi KL, et al. Characteristics of lung cancer among patients with idiopathic pulmonary fibrosis and interstitial lung disease: analysis of institutional and population data. Respir Res. 2018;19:195. doi: 10.1186/s12931-018-0899-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–454. doi: 10.1038/nature25183. [DOI] [PubMed] [Google Scholar]
- 5.Ulke HM, Mutze K, Lehmann M, Wagner DE, Heinzelmann K, Günther A, et al. The oncogene ECT2 contributes to a hyperplastic, proliferative lung epithelial cell phenotype in idiopathic pulmonary fibrosis. Am J Respir Cell Mol Biol. 2019;61:713–726. doi: 10.1165/rcmb.2019-0047OC. [DOI] [PubMed] [Google Scholar]
- 6.Fields AP, Justilien V. The guanine nucleotide exchange factor (GEF) Ect2 is an oncogene in human cancer. Adv Enzyme Regul. 2010;50:190–200. doi: 10.1016/j.advenzreg.2009.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Disayabutr S, Kim EK, Cha SI, Green G, Naikawadi RP, Jones KD, et al. miR-34 miRNAs regulate cellular senescence in type II alveolar epithelial cells of patients with idiopathic pulmonary fibrosis. PLoS One. 2016;11:e0158367. doi: 10.1371/journal.pone.0158367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lehmann M, Korfei M, Mutze K, Klee S, Skronska-Wasek W, Alsafadi HN, et al. Senolytic drugs target alveolar epithelial cell function and attenuate experimental lung fibrosis ex vivo. Eur Respir J. 2017;50:1602367. doi: 10.1183/13993003.02367-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schoenfelder KP, Fox DT. The expanding implications of polyploidy. J Cell Biol. 2015;209:485–491. doi: 10.1083/jcb.201502016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Miettinen TP, Caldez MJ, Kaldis P, Björklund M. Cell size control: a mechanism for maintaining fitness and function. BioEssays. 2017;39:1700058. doi: 10.1002/bies.201700058. [DOI] [PubMed] [Google Scholar]
- 11.Xu Y, Mizuno T, Sridharan A, Du Y, Guo M, Tang J, et al. Single-cell RNA sequencing identifies diverse roles of epithelial cells in idiopathic pulmonary fibrosis. JCI Insight. 2016;1:e90558. doi: 10.1172/jci.insight.90558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zack TI, Schumacher SE, Carter SL, Cherniack AD, Saksena G, Tabak B, et al. Pan-cancer patterns of somatic copy number alteration. Nat Genet. 2013;45:1134–1140. doi: 10.1038/ng.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Spek CA, Duitman J. Is idiopathic pulmonary fibrosis a cancer-like disease? Transcriptome analysis to fuel the debate. ERJ Open Res. 2019;5:00157–2018. doi: 10.1183/23120541.00157-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moreno-Layseca P, Streuli CH. Signalling pathways linking integrins with cell cycle progression. Matrix Biol. 2014;34:144–153. doi: 10.1016/j.matbio.2013.10.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.