Abstract
Essential oils (EOs) are one of the most important groups of plant metabolites responsible for their biological activities. This study was carried out to study the chemical composition and the antimicrobial effects of Artemisia herba-alba and Origanum majorana essential oils against some Gram-positive and Gram-negative bacteria, and a fungal strain isolated from spoiled butter. The plants were collected in the region Azzemour of South West Morocco and the EOs, extracted by hydrodistillation, were analyzed by GC-MS. The antimicrobial activity was determined using the agar paper disc method. The main components of A. herba-alba EO were cis-thujone, trans-thujone and vanillyl alcohol; in O. majorana EO terpinen-4-ol, isopulegol and β-phellandrene predominated. Both essential oils exhibited growth inhibiting activities in a concentration-dependent manner on several microorganism species. Our results demonstrated that O. majorana and A. herba-alba EOs could be effective natural antibacterial agents in foods.
Keywords: essential oils, antimicrobial activity, chemical characterization, Artemisia herba-alba, Origanum majorana
1. Introduction
Essential oils (EOs) are complex mixtures derived from various parts of plants with strong aromatic components such as terpenes. They are used in many fields such as medicine, cosmetic, and food industry [1,2]. The available literature reported that EOs possess, among others, significant antiseptic, antibacterial, antiviral, antioxidant, anti-parasitic, antifungal, and insecticidal activities [3].
At the moment, Morocco is considered as one of the principal suppliers and producers of some aromatic plants, such as Artemisia herba-alba Asso, Mentha pulegium L., Lavandula stoechas L., and Rosmarinus officinalis L. Moreover, these plants produce very high added value products contributing to the economic development of Morocco [4].
Artemisia herba-alba, chih in Arabic, belongs to the Asteraceae family; its essential oil is known for its antimicrobial, antioxidant, insecticidal, and antispasmodic activities. It is also used in traditional medicine as an antispasmodic and in treatment of diabetes mellitus [2,5].
Origanum majorana L. is a lamiaceous species, known for its antimicrobial, antioxidant, antidiabetic, and antitumoral activities [6]. In traditional medicine, the plant is used as an antiepileptic and a sedative drug [7].
The aim of the present study was to identify the components of A. herba-alba and O. majorana EOs from Morocco, and to evaluate their antimicrobial activity, against some Gram-positive and Gram-negative bacteria, and their antifungal efficacy.
2. Results
2.1. Essential Oil Yields and Composition
Hydrodistillation of the aerial parts of A. herba alba and of O. majorana resulted in pale yellow oils in 0.86% and 0.97% yield, on a dry mass basis, respectively. Table 1 and Table 2 report the percent composition of the essential oils; compounds are listed according to their elution on a HP-5MS column. Fifty-eight compounds were identified, 14 for A. herba-alba, and 44 for O. majorana, accounting for 97.6% and 97.8% of the total oil, respectively. In the essential oil from A. herba-alba cis-thujone (25.5%), trans-thujone (17.7%), vanillyl alcohol (11.5%), and nor-davanone (7.8%) are the main components. In the essential oil from O. majorana, terpinen-4-ol (34.1%), α-terpinene (19.2%), and terpineol (8.9%) are the main constituents.
Table 1.
Chemical composition of Artemisia herba-alba essential oil.
| Compound | % | Ki a | Ki b | Identification c |
|---|---|---|---|---|
| trans-Arbusculone | 4.5 | 1048 | 1,2 | |
| cis-Thujone | 25.5 | 1079 | 1102 | 1,2,3 |
| trans-Thujone | 17.7 | 1111 | 1114 | 1,2,3 |
| Camphor | 4.9 | 1150 | 1146 | 1,2,3 |
| nor-Davanone | 7.8 | 1200 | 1231 | 1,2 |
| cis-Chrysanthenylacetate | 4.7 | 1231 | 1265 | 1,2 |
| Undec-10-en-1-al | 1.3 | 1261 | 1296 | 1,2 |
| Cyclosativene | T | 1342 | 1368 | 1,2 |
| cis, threo-Davanafuran | 5.8 | 1386 | 1415 | 1,2 |
| Vanillyl Alcohol | 11.5 | 1424 | 1447 | 1,2 |
| n-Dodecanol | 3.1 | 1445 | 1470 | 1,2 |
| Isobornyl n-butyrate | 4.9 | 1466 | 1491 | 1,2 |
| <E>-Jasmolactone | 3.4 | 1483 | 1491 | 1,2 |
| Artedouglasia Oxide C | 2.5 | 1496 | 1523 | 1,2 |
| Total | 97.6 | |||
| Oxygenated monoterpene | 56.4 | |||
| Oxygenated sesquiterpenes | 2.5 | |||
| Other compounds | 38.7 |
a Kovats retention index on HP-5 MS column; b Kovats retention index on HP Innovax column; c Identification: 1 = Kovats retention index, 2 = mass spectrum, 3 = co-injection with pure compound; T = traces, less than 0.05%.
Table 2.
Chemical composition of Origanum majorana essential oil.
| Compound | % | KI a | KI b | Identification c |
|---|---|---|---|---|
| α-Pinene | 4.1 | 941 | 932 | 1,2,3 |
| p-Cymene | 2.6 | 950 | 1024 | 1,2,3 |
| iso-Sylvestrene | 0.6 | 952 | 1008 | 1,2,3 |
| β-Pinene | 0.2 | 975 | 974 | 1,2,3 |
| α-Phellandrene | 2.6 | 984 | 1002 | 1,2,3 |
| δ-3-Carene | 1.9 | 1008 | 1011 | 1,2 |
| α-Terpinene | 19.2 | 1021 | 1017 | 1,2,3 |
| Limonene | 0.1 | 1038 | 1029 | 1,2,3 |
| 1,8 Cineole | 3.0 | 1047 | 1031 | 1,2,3 |
| β-Ocimene | 0.1 | 1061 | 1037 | 1,2,3 |
| cis-Sabinene hydrate | 1.3 | 1070 | 1070 | 1,2 |
| Terpinen-4-ol | 34.1 | 1096 | 1149 | 1,2,3 |
| endo-Fenchyl-acetate | 9.8 | 1114 | 1220 | 1,2 |
| Pulegone | 0.7 | 1122 | 1237 | 1,2 |
| trans-Pinocarveol | 0.3 | 1143 | 1139 | 1,2 |
| Terpineol | 8.9 | 1160 | 1133 | 1,2,3 |
| cis-Limonene oxide | T | 1188 | 1136 | 1,2 |
| dihydro-Linalool | 0.1 | 1191 | 1135 | 1,2 |
| cis-Verbenol | T | 1193 | 1141 | 1,2,3 |
| Viridene | 0.1 | 1199 | 1167 | 1,2 |
| (E)-Isocitral | 0.2 | 1205 | 1180 | 1,2 |
| Thymol | 0.2 | 1211 | 1290 | 1,2,3 |
| Carvacrol | 0.3 | 1220 | 1299 | 1,2,3 |
| γ-Elemene | 0.1 | 1233 | 1338 | 1,2,3 |
| α-Terpinyl acetate | 0.8 | 1242 | 1349 | 1,2 |
| Eugenol | T | 1271 | 1359 | 1,2,3 |
| Neryl acetate | 0.2 | 1274 | 1361 | 1,2 |
| α-Copaene | T | 1278 | 1376 | 1,2,3 |
| Geranyl acetate | 0.3 | 1293 | 1381 | 1,2,3 |
| iso-Longifolene | 0.1 | 1303 | 1390 | 1,2 |
| (E)-Caryophillene | 2.1 | 1314 | 1407 | 1,2,3 |
| β-Duprezianene | T | 1321 | 1422 | 1,2 |
| β-Cedrene | T | 1324 | 1420 | 1,2,3 |
| β-Copaene | T | 1327 | 1432 | 1,2,3 |
| α-Guaiene | 0.2 | 1332 | 1439 | 1,2,3 |
| Aromadendrene | 0.3 | 1336 | 1441 | 1,2,3 |
| allo-Aromadendrene | 1.3 | 1370 | 1460 | 1,2 |
| Valencene | 0.2 | 1401 | 1496 | 1,2,3 |
| Caryophyllene oxide | T | 1436 | 1583 | 1,2,3 |
| Epiglobulol | T | 1445 | 1590 | 1,2 |
| (-)-Spathulenol | T | 1453 | 1578 | 1,2 |
| β-Atlanthol | 1.6 | 1464 | 1608 | 1,2,3 |
| Rosifoliol | 0.1 | 1485 | 1600 | 1,2 |
| Cubenol | T | 1497 | 1646 | 1,2 |
| Total | 97.8 | |||
| Monoterpene hydrocarbons | 33.1 | |||
| Oxygenated monoterpene | 57.9 | |||
| Sesquiterpene hydrocarbons | 5.1 | |||
| Oxygenated sesquiterpenes | 1.7 |
a Kovats retention index on HP-5 MS column; b Kovats retention index on HP Innovax column; c Identification: 1 = Kovats retention index, 2 = mass spectrum, 3 = co-injection with pure compound. T = traces, less than 0.05%.
2.2. Antimicrobial Activity
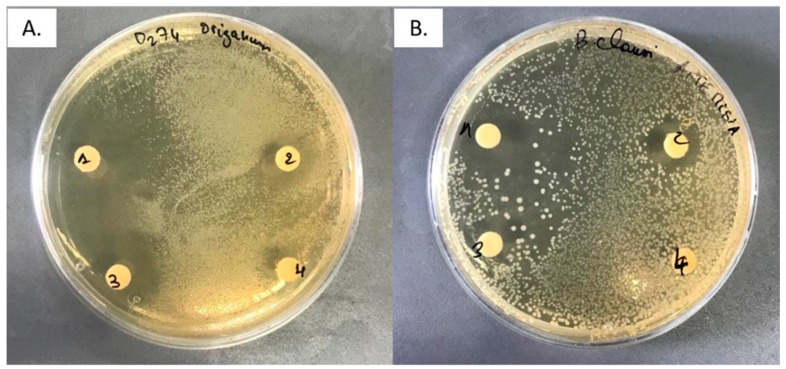
The antimicrobial activity of A. herba-alba and O. majorana essential oils was tested, at different concentrations, against 20 microorganisms, both Gram-positive and Gram-negative strains. Figure 1 shows a representative image of the antimicrobial activity. The EO of A. herba-alba showed inhibitory effects against 15 bacterial strains, the most sensitive being Brochothrix thermosphacta 7R1, Bacillus clausii 2226 and Salmonella Typhimurium; five strains resulted resistant to this EO: Hafnia alvei 53M, Carnobacterium maltaromaticum F1201, Carnobacterium maltaromaticum D1203, Enterococcus faecalis 226 and Enterococcus faecalis ES1 (Table 3). O. majorana essential oil showed a wider spectrum of activity as it was active against all microbial strains tested (Table 4).
Figure 1.
Representative antimicrobial activity of (A) Origanum majorana essential oil against Brochothrix thermosphacta D274 at the dose of 50, 40, 20, and 15 µL (from 1 to 4, respectively) and (B) Artemisia herba-alba essential oil against Bacillus clausii 2226 at the concentrations of 20, 15, 10, and 5 µL (from 1 to 4, respectively).
Table 3.
Antibacterial activity of the essential oil of A. herba-alba.
| Strain | Control | Essential Oil | ||||
|---|---|---|---|---|---|---|
| Gentamicin | Tetracyclin | 5 μL | 10 μL | 15 μL | 20 μL | |
| B. clausii 2226 | 11.0 ± 1.0 | 16.3 ± 1.5 | 10.3 ± 0.6 | 14.7 ± 0.6 | 19.7 ± 1.5 a,D | 24.0 ± 1.0 a,A |
| Br. thermosphacta 7R1 | 18.3 ± 1.5 | 19.3 ± 1.2 | na | 6.0 ± 0.0 | 6.0 ± 0.1 | 12.3 ± 0.6 |
| Br. thermosphacta D274 | 6.0 ± 0.0 | 8.7 ± 1.2 | 6.0 ± 0.0 | 11.7 ± 0.6 a,C | 14.7 ± 0.6 a,A | 17.7 ± 0.6 a,A |
| C. maltaromaticum 9P | 6.0 ± 0.0 | 24.3 ± 1.2 | 6.0 ± 0.0 | 9.0 ± 1.0 b | 11.7 ± 0.6 a | 12.3 ± 0.6 a |
| C. maltaromaticum D1203 | 6.0 ± 0.0 | 22.3 ± 0.6 | na | na | na | na |
| C. maltaromaticum F1201 | 6.0 ± 0.0 | 23.3 ± 1.5 | na | na | na | na |
| C. maltaromaticum H1201 | 10.0 ± 0.0 | na | na | na | na | 6.0 ± 0.0 A |
| E. coli 32 | 14.7 ± 0.6 | 18.7 ± 1.2 | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 |
| Ent. faecalis 226 | 6.0 ± 0.0 | 9.0 ± 1.0 | na | na | na | na |
| Ent. faecalis E21 | 6.0 ± 0.0 | 14.7 ± 0.6 | na | na | na | na |
| H. alvei 53M | 11.7 ± 1.5 | 9.7 ± 0.6 | na | na | na | na |
| L. innocua 1770 | 25.3 ± 0.6 | 20.3 ± 1.5 | na | na | na | 6.0 ± 0.0 |
| P. fragi 6P2 | 14.7 ± 0.6 | 17.0 ± 1.0 | na | 6.0 ± 0.0 | 9.3 ± 0.6 | 13.3 ± 2.1 |
| Staph. aureus | 6.0 ± 0.0 | 15.3 ± 0.6 | na | na | na | 6.0 ± 0.0 |
| S. Typhimurium | 9.7 ± 0.6 | 12.7 ± 1.2 | na | 6.0 ± 0.0 | 14.0 ± 1.7 b | 17.7 ± 0.6 a,B |
| Serr. proteamaculans 20P | 12.3 ± 0.6 | 24.3 ± 1.2 | na | 6.0 ± 0.0 | 7.7 ± 0.6 | 10.3 ± 0.6 |
| Str. salivarius | 6.0 ± 0.0 | 18.7 ± 1.2 | na | 6.0 ± 0.0 | 14.0 ± 1.0 a | 18.3 ± 1.5 a |
| Staph. saprophyticus 3S | 24.0 ± 1.0 | 29.0 ± 3.6 | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 |
| Staph. sp.ES1 | 19.3 ± 1.2 | 29.3 ± 1.2 | na | 6.0 ± 0.0 | 8.3 ± 0.6 | 10.3 ± 0.6 |
| Staph.sp.GB1 | 21.3 ± 1.2 | 27.7 ± 2.5 | 6 ± 0.0 | 11.3 ± 1.2 | 14.7 ± 0.6 | 17 ± 1.0 |
Data represent the diameter inhibition (in mm). Results are the mean of three repetitions ± standard deviation (SD) of the inhibition zone. na = not active. Dunnett’s test vs. Gentamicin (a,b,c,d) or Tetracycline (A,B,C,D): a,A p < 0.0001; b,B p < 0.001; c,C p < 0.01; d,D p < 0.05. B.: Bacillus; Br.: Brochothrix; C.: Carnobacterium; E.: Enterococcus; Staph.: Staphylococcus; L.: Listeria; E.: Escherichia; H.: Hafnia; P.: Pseudomonas; S.: Salmonella; Serr.: Serratia; Str.: Streptococcus.
Table 4.
Activity of the essential oil of O. majorana.
| Strain | Control | Essential Oil | ||||||
|---|---|---|---|---|---|---|---|---|
| Gentamicin | Tetracyclin | 5 µL | 10 µL | 15 µL | 20 µL | 40 µL | 50 µL | |
| B. clausii 2226 | 11.0 ± 1.0 | 16.3 ± 1.5 | na | 6.0 ± 0.0 | 15.3 ± 0.6b | 23.3 ± 1.5 a,B | 24.7 ± 0.6 a,B | 28.3 ± 1.5 a,B |
| Br. thermosphacta D274 | 6.0 ± 0.0 | 8.7 ± 1.2 | 10.6 ± 0.6d | 13.3 ± 1.2 a | 18.3 ± 0.6 a,B | 23.0 ± 1.7 a,B | 24.3 ± 1.2 a,B | 26.3 ± 1.2 a,B |
| Br. thermosphacta 7R1 | 18.3 ± 1.5 | 19.3 ± 1.2 | 11.3 ± 1.2 | 15.3 ± 0.6 | 18.0 ± 0.0 | 20.3 ± 0.6* | 20.0 ± 0.0 | 21.3 ± 0.6c,D |
| C. maltaromaticum 9P | 6.0 ± 0.0 | 24.3 ± 1.2 | na | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 | 9.3 ± 0.6a |
| C.maltaromaticum H1201 | 10.0 ± 0.0 | na | 6.0 ± 0.0A | 6.0 ± 0.0 A | 8.7 ± 1.2 A | 9.7 ± 0.6 A | 9.3 ± 0.6 A | 9.7 ± 0.6 A |
| C. maltaromaticum D1203 | 6.0 ± 0.0 | 22.3 ± 0.6 | na | na | na | 6.0 ± 0.0 | 9.3 ± 0.6 a | 13.3 ± 1.5 a |
| C. maltaromaticum F1201 | 6.0 ± 0.0 | 23.3 ± 1.5 | na | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 | 6.0 ± 0.0 |
| E. coli 32 | 14.7 ± 0.6 | 18.7 ± 1.2 | 9.7 ± 0.6 | 10.7 ± 1.2 | 17.7 ± 0.6 | 20.0 ± 0.0 a | 24.3 ± 1.2 a,B | 26.7 ± 0.6 a,B |
| Ent. faecalis 226 | 6.0 ± 0.0 | 9.0 ± 1.0 | na | na | na | 6.0 ± 0.0 | 6.0 ± 0.1 | 9.7 ± 0.7 |
| Ent. faecalis E21 | 6.0 ± 0.0 | 14.7 ± 0.6 | na | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 | 6.0 ± 0.0 |
| H. alvei 53M | 11.7 ± 1.5 | 9.7 ± 0.6 | 8.3 ± 0.6 | 10.3 ± 0.6 | 11.7 ± 0.6 C | 12.7 ± 0.6 A | 15.0 ± 0.0 b,A | 20.3 ± 0.6 a,B |
| L. innocua 1770 | 25.3 ± 0.6 | 20.3 ± 1.5 | na | 6.0 ± 0.0 | 9.3 ± 0.6 | 11.7 ± 0.6 | 12.3 ± 0.6 | 13.7 ± 1.5 |
| P. fragi 6P2 | 14.7 ± 0.6 | 17.0 ± 1.0 | na | na | 6.0 ± 0.0 | 6.0 ± 0.0 | 6.0 ± 0.0 | 9.3 ± 0.6 |
| Staph. aureus | 6.0 ± 0.0 | 15.3 ± 0.6 | 10.7 ± 0.6 d | 11.7 ± 1.5 c | 16.3 ± 1.5 a | 24.3 ± 2.1 a,B | 27.7 ± 0.6 a,B | 32.3 ± 2.5 a,B |
| S. Typhimurium | 9.7 ± 0.6 | 12.7 ± 1.2 | 7.7 ± 2.1 | 11.7 ± 0.6 | 14.0 ± 1.7 d | 17.3 ± 1.2 c,D | 23.3 ± 2.9 a,B | 29.7 ± 0.6 a,B |
| Serr.proteamaculans 20P | 12.3 ± 0.6 | 24.3 ± 1.2 | na | na | na | 6.0 ± 0.0 | 16.3 ± 1.5a | 19.3 ± 1.2a |
| Str. salivarius | 6.0 ± 0.0 | 18.7 ± 1.2 | 9.7 ± 0.6 c | 11.3 ± 0.6 a | 13.0 ± 1.0 a | 19.3 ± 1.2 a | 20.7 ± 1.2 a | 24.3 ± 1.2 a,B |
| Staph. saprophyticus 3S | 24.0 ± 1.0 | 29.0 ± 3.6 | 9.9 ± 1.0 | 11.7 ± 0.6 | 19.3 ± 1.2 | 20.3 ± 0.6 | 21.0 ± 1.0 | 24.3 ± 1.2 |
| Staph.sp. ES1 | 19.3 ± 1.2 | 29.3 ± 1.2 | 6.0 ± 0.0 | 6.0 ± 0.0 | 14.7 ± 7.6 | 18.7 ± 1.2 | 20.0 ± 0.0 | 21.0 ± 1.0 |
| Staph.sp.GB1 | 21.3 ± 1.2 | 27.7 ± 2.5 | na | na | 5.7 ± 0.6 | 10 ± 0.0 | 14.7 ± 0.6 | 19.3 ± 1.2 |
Data represent the diameter inhibition(in mm). Results are the mean of three repetitions ± standard deviation (SD) of the inhibition zone. na = not active. Dunnett’s test vs. Gentamicin (a,b,c,d) or Tetracycline (A,B,C,D): a,A p < 0.0001; b,B p < 0.001; c,C p < 0.01; d,D p < 0.05. B.: Bacillus; Br.: Brochothrix; C.: Carnobacterium; E.: Enterococcus; Staph.: Staphylococcus; L.: Listeria; E.: Escherichia; H.: Hafnia; P.: Pseudomonas; S.: Salmonella; Serr.: Serratia; Str.: Streptococcus.
Data analysis showed for the EO of A. herba-alba the same antimicrobial activity of tetracycline against Streptococcus salivarius, but higher than gentamicin, and exhibited stronger antimicrobial activity than both antibiotics against Br. thermosphacta D274, B. clausii 2226, and S. Typhimurium and lower antimicrobial activity than that of both antibiotics against Staphylococcus sp. GB1, Staphylococcus saprophyticus 3S, Escherichia coli 32, Br. thermosphacta 7R1, Staphylococcus sp. ES1, and Serratia proteamaculans 20P. On the other hand, the antimicrobial activity of O. majorana essential oil was stronger than both antibiotics against Str. salivarius, E. coli 32, Br. thermosphacta 7R1, H. alvei 53M, Salmonella sp. ES1, Br. thermosphacta D274, B. clausii 2226, Ente. faecalis 226, S. Typhimurium, and Staphylococcus aureus. The same antimicrobial activity as gentamicin was recorded against Staph. saprophyticus 3S, C. maltaromaticum H1201, C. maltaromaticum F1201, Ent. faecalis E21 and lower antimicrobial activity than both antibiotics against Staph. sp GB1 and Listeria innocua 1770.
2.3. Antifungal Activity
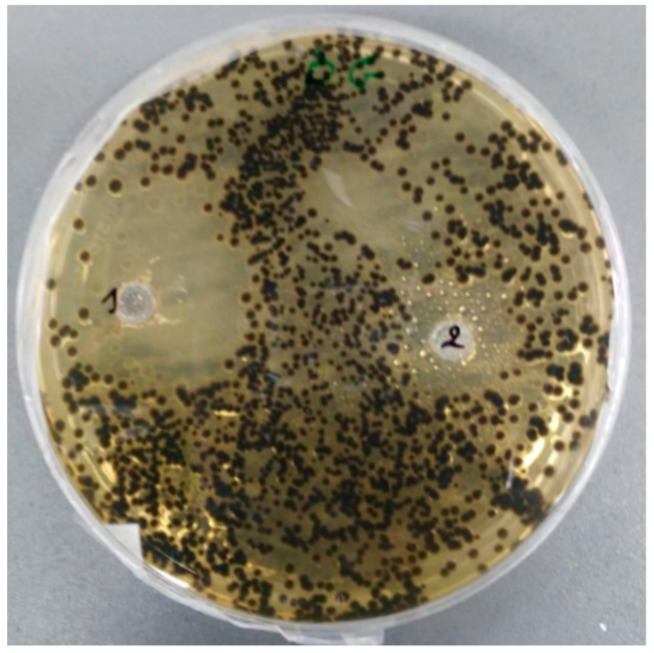
Table 5 reports the inhibition halos in (mm) at the dose of 20 µL of the two essential oils against Aspergillus niger isolated from the spoiled butter. This fungal strain was sensitive to both essential oils; the highest inhibitory activity was showed by A. herba-alba essential oil against A. niger. Figure 2 shows the antifungal activity of both EOs as determined in the same agar dish. However, diameters of inhibition halos were measured on two separated agar dishes.
Table 5.
Antifungal activity of A. herba-alba and O. majorana essential oils.
| Aspergillus niger | |
|---|---|
| Artemisia herba-alba | 23.6 ± 1.5 |
| Origanum majorana | 14.0 ± 1.0 |
Data represent the diameter inhibition (in mm). Results are the mean of three repetitions ± standard deviation (SD) of the inhibition zone.
Figure 2.
Antifungal activity of A. herba-alba (1) and O. majorana (2) essential oils against Aspergillus niger at the dose of 20 µL.
3. Discussion
In our A. herba-alba essential oil oxygenated monoterpenes (57.3%) predominated, with cis-thujone (25.5%) and trans-thujone (17.7%) as the main constituents. Vanillyl alcohol (11.5%) and nor-davanone (7.8%) were in appreciable amounts.
These results agree with literature on the essential oil from A. herba-alba from different countries that evidenced cis- and/or trans-thujone as the principal constituents [8,9,10]. On the other hand, other studies showed eucalyptol (32.8%) as the main constituent of the A. herba-alba EO from Iran, and caryophyllene acetate (10.75%) for a Jordanian EO [11,12]. These compounds are totally absent in our essential oil. Camphor is reported as principal component in essential oil from Algeria and Tunisia (ranging between 19.6% and 50.5%) [13,14], but it is present in a low percentage in our sample (4.9%).
Moreover, other studies evidenced that davanone is one of the main constituents, with a percent greater than 10% [15,16]. In our EO, davanone and its derivative, cis, threo-davanafuran, accounted for 13.6% of the oil. Instead, this is the first report on the presence of vanillyl alcohol as one of the main constituents of this EO. Other studies reported camphor as the major component of the essential oil (17.8%–50.3%) [13,15,17,18,19] that, instead, is absent in our sample or chrysanthenone, present in our EO with its derivative, iso-chrysanthenyl acetate [20,21].
Monoterpenes predominated (91.1%) in the oil of O. majorana, both hydrocarbons and oxygenated compounds; sesquiterpenes accounted for 6.8%. The main components are terpinen-4-ol (34.1%), α-terpinene (19.2%), and terpineol (8.9%). Our results are in agreement with many studies that reported terpinen-4-ol among the principal constituents of the essential oil of O. majorana [22,23,24,25,26]. Moreover, α-terpinene was present in similar percentage (ranging from 11.08% to 12.72%) also in essential oils from Tunisia and Morocco [22,27]. Instead, in the EO from the Venuezelan Andes α-terpinene is reported in lesser percentages (3.6%) [26]. trans-Sabinene hydrate was reported as one of the principal components in other studies [28,29], in our sample its isomer was present in a low quantity (1.3%). Linalool, absent in our essential oil, is the main compound in the EO of O. majorana from Turkey with a percent of 88.01% [30]. Moreover, 4-terpinene and γ-terpinene were identified as the main components in O. majorana from Taiwan and Morocco, respectively [27,31].
Most microorganisms used in this study were sensitive to both essential oils, with the dose of 20 μL of EO sufficient to stop the growth of almost all tested Gram-positive and Gram-negative strains. In particular, O. majorana EO resulted more active, showing a wide spectrum of activity. On the other hand, the EO of A. herba-alba showed inhibitory effects against 15 bacterial strains.
The available literature reports the antimicrobial activity of A. herba-alba essential oil against Staph. aureus, E. coli, and B. cereus [23,32,33]. Moreover, several studies showed a great potential of A. herba-alba EO oil as an antibacterial agent against Klebsiella pneumoniae, Listeria monocytogenes, Vibrio colerae, and S. Typhimurium [34,35,36]. Our results showed variable antimicrobial and antifungal activity of the essential oil, being the inhibition zones in the range of 10–24 mm. Gram-positive bacteria resulted more sensitive to this EO. The Gram-positive B. clausii 2226 was the most sensitive tested strain, with the strongest inhibition zone (24.00 ± 1 mm). B. clausii was used as a model of spore-forming aerobic microorganism and our findings showed that our A. herba-alba EO is suitable to control the growth of this microorganism. It is well known that spore forming bacteria (also called thermoduric) are the main problem in pasteurized foods, both from the point of view of food spoilage and human intoxication. Gram-negative strains also displayed variable degree of susceptibility to this EO. The maximum activity was showed against the pathogen strain S. Typhimurium (17.7 ± 0.6), but C. maltaromaticum F1201, C. maltaromaticum D1203, H. alvei 53M, Ent. faecalis E21, and Ent. faecalis 226 resulted resistant, since no inhibition zone was observed. Due to the involvement of S. Typhimurium in the majority of food intoxication across the world, the antimicrobial capability of this EO could be of pivotal importance in the control of this microorganism in foods.
The antimicrobial activity of O. majorana essential oil appears to be similarly effective against both Gram-positive and Gram-negative microorganisms. These results agree with literature data [21,34,35]. Data of previous research showed that O. majorana essential oil was active against a large spectrum of different bacteria strains: E. coli, Str. agalactiae, Shigella dysenteriae, Salmonella Enteritidis, Staph. aureus, Ent. faecalis, E. coli, and Klebsiella pneumoniae [26,37].
In our study, all tested strains were sensitive to this essential oil, with the Gram-positive S. aureus the most sensitive with the greatest inhibition zone (32.2 ± 2.5 mm); the more sensitive Gram-negative was S. Typhimurium, with an inhibition zone of 29.7 ± 0.6 mm.
The antimicrobial activity of both essential oils could be related to their content in oxygenated monoterpenes, which constitute about 57.3% and 53.0% of the EOs of A. herba alba and O. majorana, respectively. Similar findings have been already previously reported [38,39].
The major components of O. majorana EO, e.g., terpinen-4-ol, α-terpinol and α-pinene, have been reported for their antimicrobial and antifungal properties [21]. Additionally, the main constituents of the EO of A. herba-alba, cis- and trans-thujone and vanillyl alcohol, have been reported for their antimicrobial, anti-inflammatory, and antioxidant activities [40,41]. Oxygenated monoterpenes exhibit high antimicrobial activity on whole cell and possess antifungal effects. These compounds diffuse into and damage cell membrane structures [42].
Our results showed high antifungal activity for both essential oils, with the highest inhibitory activity shown by the EO of A. herba-alba against Aspergillus niger (inhibition zone 23.6 ± 1.5 mm). These results are consistent with data previously reported [29,43].
4. Materials and Methods
4.1. Plant Material
The aerial parts of A. herba-alba and O. majorana were collected in the Azzemour region, South West Morocco, in June 2018, in flowering stage, and dried in the shade. The plants were identified by Prof. V. De Feo. A voucher specimen of each plant is stored in Department of Agricultural Sciences, University of Naples Federico II.
4.2. Essential Oil Extraction
One kilogram of A. herba-alba and O. majorana aerial parts was subjected to hydrodistillation for 3 h, according to the standard procedure described in the European Pharmacopoeia [44]. The oils were solubilized in n-hexane, filtered over anhydrous sodium sulphate and stored under N2 at +4 °C in the dark, until tested and analyzed.
4.3. GC-FID Analysis
Analytical gas chromatography was carried out on a Perkin-Elmer Sigma-115 gas-chromatograph (Perkin Elmer, Waltham, MA, USA) equipped with an FID and a data handling processor. The separation was achieved using a HP-5 MS fused-silica capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness). Column temperature: 40 °C, with 5 min initial hold, and then to 270 °C at 2 °C/min, 270 °C (20 min); injection mode splitless (1 µL of a 1:1000 n-hexane solution). Injector and detector temperatures were 250 °C and 290 °C, respectively. Analysis was also run by using a fused silica HP Innowax polyethylene glycol capillary column (50 m × 0.20 mm i.d., 0.25 µm film thickness). In both cases, helium was used as carrier gas (1.0 mL/min).
4.4. GC/MS Analysis
Analysis was performed on an Agilent 6850 Ser. II apparatus (Agilent, Roma, Italy), fitted with a fused silica DB-5 capillary column (30 m × 0.25 mm i.d., 0.33 µm film thickness), coupled to an Agilent Mass Selective Detector MSD 5973 (Agilent); ionization energy voltage 70 eV; electron multiplier voltage energy 2000 V. Mass spectra were scanned in the range 40–500 amu, scan time 5 scans/s. Gas chromatographic conditions were as reported in the previous paragraph; transfer line temperature, 295 °C.
4.5. Identification of the Essential Oil Components
Most constituents were identified by comparison of their Kovats retention indices (Ri) (determined relative to the tR of n-alkanes (C10–C35), with either those of the literature [45,46,47] and mass spectra on both columns or those of authentic compounds available in our laboratories by means of NIST 02 and Wiley 275 libraries [48]. The components relative concentrations were obtained by peak area normalization.
4.6. Antibacterial Assay
The antibacterial activity was evaluated in vitro, by means of the agar diffusion test on the plate. The activity of the essential oils was tested on the 20 microorganisms reported in Table 6. All of them belong to the collection of the Department of Agricultural Sciences, University of Naples Federico II.
Table 6.
Source and optimal growth conditions of microorganisms.
| Gram | Microorganism | Source | Growth Conditions |
|---|---|---|---|
| Positive | B. clausii 2226 | Supplement | TSB 24h at 30 °C |
| Br. thermosphacta 7R1 | Meat | TSB 24h at 20 °C | |
| Br. thermosphacta D274 | Meat | TSB 24h at 20 °C | |
| C. maltaromaticum 9P | Meat | TSB 24h at 20 °C | |
| C. maltaromaticum D1203 | Meat | TSB 24h at 25 °C | |
| C. maltaromaticum F1201 | Meat | TSB 24h at 25 °C | |
| C. maltaromaticum H1201 | Meat | TSB 24h at 25 °C | |
| Ent. faecalis 226 | Milk | TSB 24h at 30 °C | |
| Ent. faecalis E21 | Milk | TSB 24h at 30 °C | |
| Staph. aureus | Meat | TSB 24h at 37 °C | |
| Staph. saprophyticus 3S | Fermented meat | TSB 24h at 37 °C | |
| Staph. sp. ES1 | Fermented meat | TSB 24h at 37 °C | |
| Staph. sp. GB1 | Fermented meat | TSB 24h at 37 °C | |
| L. innocua 1770 | Milk | TSB 24h at 30 °C | |
| E. coli 32 | Meat | TSB 24h at 37 °C | |
| Str. salivarius | Milk | TSB 24h at 30 °C | |
| Negative | H.alvei 53M | Meat | TSB 24h at 30 °C |
| Pseud. fragi 6P2 | Meat | TSB 24h at 20 °C | |
| S. Typhimurium | Chicken meat | TSB 24h at 30 °C | |
| Serr.proteamaculans20P | Meat | TSB 24h at 25 °C |
B.: Bacillus; Br.: Brochothrix; C.: Carnobacterium; E.: Enterococcus; Staph.: Staphylococcus; L.: Listeria; E.: Escherichia; H.: Hafnia; P.: Pseudomonas; S.: Salmonella; Serr.: Serratia; Str.: Streptococcus.
Microbial strains were previously grown in TSB tryptone soya broth for 24 h. A volume of 0.1 mL of the microbial suspensions (about 1 × 108 CFU/mL) was uniformly distributed on Nutrient agar plates in sterile conditions. Different amounts of essential oils were spotted on the inoculated plates: 50, 40, 20, 15, 10, and 5 µL for O. majorana and 20, 15, 10, and 5 µL for A. herba-alba essential oils. After 10 min, under sterile conditions, plates were then incubated at optimal growth condition culture of each strain. The antimicrobial activity was evidenced by measuring the diameter (in mm) of the zone of inhibition. Ethanol was used as the negative control; tetracycline (10 µg) and gentamycin (10 µg) were used as positive controls. Each experiment was carried out in three independent replicates and result is the average with standard deviation.
4.7. Antifungal Activity
The antifungal activity was evaluated in vitro, using the agar well diffusion method on the plates. The activity was tested against Aspergillus niger isolated from a spoiled butter sample and identified by phenotypic characteristics. The fungus was previously grown in TSA agar plates at 28 °C until spore formation. Then, 1 mL of a spore suspension in quarter strength Ringer solution, containing about 1 × 108 spores per mL, was uniformly distributed on Nutrient agar plates in sterile conditions, then a hole was punched with sterile cork and 20 µL of each EO was introduced into the well; the plates were incubated at 28 °C for 4–5 days. Ethanol was used as the negative control. The antifungal activity was evaluated by measuring diameter of the inhibition area. Each experiment was carried out in three independent replicates and result is the average with standard deviation.
4.8. Statistical Analysis
Data of each experiment were statistically analyzed using GraphPad Prism 6.0 software (GraphPad Software Inc., San Diego, CA, USA), followed by comparison of means (one-way ANOVA) using Dunnett’s multiple comparisons test, at the significance level of p < 0.05.
5. Conclusions
The composition of Artemisia herba-alba and Origanum majorana essential oils growing in Morocco was analyzed and its antibacterial and antifungal activity investigated. The results indicated an important antimicrobial activity against different microorganisms especially from O. majorana essential oil. Thus, they can maybe be applied in food industry as natural preservatives, due to their antibacterial properties. Further organoleptic features and toxicological studies are required to prove the safety of the oils.
Author Contributions
Conceptualization, T.F., G.M., and V.D.F.; formal analysis, L.C. and G.A.; investigation, G.A., A.L.S.; writing—original draft preparation, G.A. and L.C.; writing—review and editing, T.F., G.M. and V.D.F.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Samples of A. herba-alba and O. majorana essential oils are available from the authors.
References
- 1.Rassem H., Nour A. Techniques for Extraction of Essential Oils From Plants. Aust. J. Basic Appl. Sci. 2016;10:117–127. [Google Scholar]
- 2.Gonzalez-Coloma A., Reina M. Natural Product-Based Biopesticides for Insect Control. Modul. Chem. Mol. Sci. Chem. Eng. 2013;3:237–268. [Google Scholar]
- 3.Kaloustian J., Chevalier J. Étude de Six Huiles Essentielles: Composition Chimique et Activité Antibactérienne. Phytothérapie. 2008;6:160–164. doi: 10.1007/s10298-008-0307-1. [DOI] [Google Scholar]
- 4.Santana O., Andrés M.F., Sanz J., Errahmani N., Abdeslam L., González-Coloma A. Valorization of essential oils from Moroccan aromatic plants. Nat. Prod. Commun. 2014;9:1109–1114. doi: 10.1177/1934578X1400900812. [DOI] [PubMed] [Google Scholar]
- 5.Mohamed A.E.H.H., El-Sayed M., Hegazy M.E., Helaly S.E., Esmail A.M., Mohamed N.S. Chemical constituents and biological activities of Artemisia herba-alba. Rec. Nat. Prod. 2010;4:1–25. [Google Scholar]
- 6.Yazdanparast R., Shahriyary L. Comparative effects of Artemisia dracunculus, Satureja hortensis and Origanum majorana on inhibition of blood platelet adhesion, aggregation and secretion. Vasc. Pharmacol. 2008;48:32–37. doi: 10.1016/j.vph.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 7.Abdollahi Fard M., Shojaii A. Efficacy of Iranian traditional medicine in the treatment of epilepsy. BioMed Res. Int. 2013;692751:1–8. doi: 10.1155/2013/692751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sbayou H., Ababou B. Chemical Composition and Antibacterial Activity of Artemisia Herba-Albaand Mentha pulegium Essential oils. J. Life Sci. 2014;8:35–41. [Google Scholar]
- 9.Aghaie M., Alizadeh M. Chemical composition of essential oil of Artemisia Herba-Alba from west Azerbaijan, Iran. J. Environ. Agric. Food Chem. 2011;10:2413–2416. [Google Scholar]
- 10.Majdouli K., Elhazzouzi H. Chemical composition and antibacterial activity of Artemisia Herba Alba Hugeii essential oil from south of Morocco. Indon. J. Chem. Res. 2015;7:14397–14404. [Google Scholar]
- 11.Aldosary N.H., Omar D., Awang R.M., Adam N.A. Chemical Profiling and Insecticidal Activity of Artemisia Herba-Alba Essential Oil Against Papaya Mealybug Paracoccus marginatus (Hemiptera: Pseudococcidae) Res. J. Appl. Sci., Engin. Technol. 2018;15:261–269. doi: 10.19026/rjaset.15.5908. [DOI] [Google Scholar]
- 12.Qnais E.Y., Alatshan A.Z., Bseiso Y.G. Chemical composition, antinociceptive and anti-inflammatory effects of Artemisia herba-alba essential oil. J. Food Agric. Environ. 2016;14:20–27. [Google Scholar]
- 13.Bertella A., Benlahcen K. Artemisia herba-alba Asso: Essential oil antibacterial activity and acute toxicity. Ind. Crops Prod. 2018;116:137–143. doi: 10.1016/j.indcrop.2018.02.064. [DOI] [Google Scholar]
- 14.Younsi F., Mehdi S., Aissi O., Rahali N., Jaouadi R., Boussaid M., Messaoud C. Essential Oil Variability in Natural Populations of Artemisia campestris (L.) and Artemisia herba-alba (Asso) and Incidence on Antiacetylcholinesterase and Antioxidant Activities. Chem. Biodivers. 2017;14:e1700017. doi: 10.1002/cbdv.201700017. [DOI] [PubMed] [Google Scholar]
- 15.Dahmani-Hamzaoui N., Baaliouamer A. Chemical Composition of Algerian Artemisia herba-alba Essential Oils Isolated by Microwave and Hydrodistillation. J. Essent. Oil Res. 2010;22:514–517. doi: 10.1080/10412905.2010.9700386. [DOI] [Google Scholar]
- 16.Mohsen H., Ali G. Essential Oil Composition of Artemisia herba-alba from Southern Tunisia. Molecules. 2009;14:1585–1594. doi: 10.3390/molecules14041585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amri I., De Martino L., Marandino A., Lamia H., Mohsen H., Scandolera E., De Feo V., Mancini E. Chemical composition and biological activities of the essential oil from Artemisia herba-alba growing wild in Tunisia. Nat. Prod. Commun. 2013;8:407–410. doi: 10.1177/1934578X1300800333. [DOI] [PubMed] [Google Scholar]
- 18.Boutkhil S., El Idrissi M. Chemical composition of the essential oil of Artemisia herba alba (Asso) populations. J. Phys. Chem. C. 2009;47:133–137. [Google Scholar]
- 19.Aljaiyash A., Kasrati A. Effect of cultivation on chemical composition and bioactivities of essential oils from Artemisia herba-alba Asso grown in Morocco. Biochem. Syst. Ecol. 2018;81:74–79. doi: 10.1016/j.bse.2018.10.001. [DOI] [Google Scholar]
- 20.Rekkab S., Abaza I.S. Chemical composition of the essential oil of aerial parts of Artemisia herba-alba Asso from Oum El-Bouaghi (Algeria) and chemotaxonomic survey. J. Mater. Environ. Sci. 2016;7:4383–4390. [Google Scholar]
- 21.Janackovic P., Novacovic J. Composition and antimicrobial activity of essential oils of Artemisia judaica, A. herba-alba and A. arborescens from Libya. Arch. Biol. Sci. 2015;67:455–466. doi: 10.2298/ABS141203010J. [DOI] [Google Scholar]
- 22.Ben Salha G., Herrera Díaz R. Deterpenation of Origanum majorana L. essential oil by reduced pressure steam distillation. Ind. Crops Prod. 2017;109:116–122. doi: 10.1016/j.indcrop.2017.08.016. [DOI] [Google Scholar]
- 23.Della Pepa T., Elshafie H.S., Capasso R., De Feo V., Camele I., Nazzaro F., Scognamiglio M.R., Caputo L. Antimicrobial and Phytotoxic Activity of Origanum heracleoticum and O. majorana Essential Oils Growing in Cilento (Southern Italy) Molecules. 2019;24:2576. doi: 10.3390/molecules24142576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hajlaoui H., Mighri H. Chemical composition and in vitro evaluation of antioxidant, antimicrobial, cytotoxicity and anti-acetylcholinesterase properties of Tunisian Origanum majorana L. essential oil. Microb. Pathog. 2016;95:86–94. doi: 10.1016/j.micpath.2016.03.003. [DOI] [PubMed] [Google Scholar]
- 25.Jiang T., Wang Y. Volatile Oil Composition of Natural Spice, Origanum majorana L., Grown in China. J. Essent. Oil Bear. Plants. 2011;14:458–462. doi: 10.1080/0972060X.2011.10643601. [DOI] [Google Scholar]
- 26.El-Akhal F., Lalami A.E.O., Zoubi Y.E., Greche H., Guemmouh R. Chemical composition and larvicidal activity of essential oil of Origanum majorana (Lamiaceae) cultivated in Morocco against Culex pipiens (Diptera: Culicidae) Asian Pac. J. Trop. Biomed. 2014;4:746–750. doi: 10.12980/APJTB.4.2014APJTB-2014-0392. [DOI] [Google Scholar]
- 27.Nurzyñska-Wierdak R., Zawislak G. The Content and Composition of Essential Oil of Origanum majorana L. Grown in Poland Depending on Harvest Tme and Method of Raw Material Preparation. J. Essent. Oil Bear. Plants. 2015;18:1482–1489. doi: 10.1080/0972060X.2013.831569. [DOI] [Google Scholar]
- 28.Alizadeh A., Khosh-Khui M. Chemical composition of the essential oil, total phenolic content and antioxidant activity in Origanum majorana L. (Lamiaceae) cultivated in Iran. Adv. Environ. Biol. 2011;5:2326–2331. [Google Scholar]
- 29.Lakehal S., Meliani A. Essential Oil Composition and Antimicrobial Activity of Artemisia herba-alba Asso Grown in Algeria. Med. Chem. 2016;6:435–439. doi: 10.4172/2161-0444.1000382. [DOI] [Google Scholar]
- 30.Karabörklü S., Ayvaz A., Yilmaz S., Akbulut M. Chemical composition and fumigant toxicity of some essential oils against Ephestia kuehniella. J. Econom. Entomol. 2011;104:1212–1219. doi: 10.1603/EC10284. [DOI] [PubMed] [Google Scholar]
- 31.Lin P.C., Lee J.J., Chang I.J. Essential oils from Taiwan: Chemical composition and antibacterial activity against Escherichia coli. J. Food Drug Anal. 2016;24:464–470. doi: 10.1016/j.jfda.2015.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ramos S., Rojas L. Chemical Composition and Antibacterial Activity of Origanum majorana L. Essential Oil from the Venezuelan Andes. J. Essent. Oil Res. 2011;23:45–49. doi: 10.1080/10412905.2011.9700481. [DOI] [Google Scholar]
- 33.Baydar H., Sağdiç O. Antibacterial activity and composition of essential oils from Origanum, Thymbra and Satureja species with commercial importance in Turkey. Food Control. 2004;15:169–172. doi: 10.1016/S0956-7135(03)00028-8. [DOI] [Google Scholar]
- 34.Zouari S., Zouari N. Chemical composition and biological activities of a new essential oil chemotype of Tunisian Artemisia herba alba Asso. J. Med. Plants Res. 2010;4:871–880. [Google Scholar]
- 35.Goudjil M.B., Ladjel S., Bencheikh S.E., Zighmi S., Hamada D. Chemical compounds profile, antibacterial and antioxidant activities of the essential oil extracted from the Artemisia herba-alba of Southern Algeria. Int. J. Biol. Chem. 2015;9:70–78. doi: 10.3923/ijbc.2015.70.78. [DOI] [Google Scholar]
- 36.Bellili S., Jazi S., Hrira M.Y., Lamari A., Dhifi W., Diouani M.F., Mnif W. Phytochemical identification of volatile fraction, essential oil and screening of antioxidant, antibacterial, allelopathic and insecticidal potential from Artemisia herba-alba leaves. Main Group Chem. 2017;16:95–109. doi: 10.3233/MGC-170229. [DOI] [Google Scholar]
- 37.Ezzeddine N.B.H.B., Abdelkefi M.M., Aissa R.B., Chaabouni M.M. Antibacterial screening of Origanum majorana L. oil from Tunisia. J. Essent. Oil Res. 2001;13:295–297. doi: 10.1080/10412905.2001.9699698. [DOI] [Google Scholar]
- 38.Dorma H., Deans S. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000;88:308–316. doi: 10.1046/j.1365-2672.2000.00969.x. [DOI] [PubMed] [Google Scholar]
- 39.Nazzaro F., Fratianni F., De Martino L., Coppola R., De Feo V. Effect of the essential oils on pathogenic bacteria. Pharmaceuticals. 2013;6:1451–1474. doi: 10.3390/ph6121451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Natalia A., Kim S. Antioxidant and antibacterial activity of fatty acid vanillyl ester produced by Proteusvulgaris K80 lipase-mediated transesterification. J. Mol. Catal. B Enzym. 2016;133:475–481. doi: 10.1016/j.molcatb.2017.03.012. [DOI] [Google Scholar]
- 41.Mockute D., Nivinskiene G. The cis-thujone chemotype of Salvia officinalis L essential oils. Chemija. 2003;14:216–220. [Google Scholar]
- 42.Cox S., Mann C. Determining the Antimicrobial Actions of Tea Tree Oil. Molecules. 2001;6:87–91. doi: 10.3390/60100087. [DOI] [Google Scholar]
- 43.Mehni M., Segni L., Terzi V., Morcia C., Ghizzoni R., Goudgil B., Benchhikh S. Antifungal Activity of Artemisia herba-alba on Various Fusarium. Phytothérapie. 2018;16:87–90. doi: 10.3166/s10298-016-1071-2. [DOI] [Google Scholar]
- 44.Council of Europe . European Pharmacopoeia. 5th ed. Volume I. Council of Europe; Strasbourg, France: 2004. p. 217. [Google Scholar]
- 45.Jenning W., Shibamoto T. Qualitative Analysis of Flavor and Fragrance Volatiles by Glass Capillary Gas Chromatography. Academic Press; San Francisco, CA, USA: 1980. [Google Scholar]
- 46.Davies N. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicon and Carbowax 20M phases. J. Chromatogr. 1990;503:1–24. doi: 10.1016/S0021-9673(01)81487-4. [DOI] [Google Scholar]
- 47.Adams R. Identification of Essential Oil Components by Gas Chromatography/mass Spectroscopy. Allured Publishing; Carol Stream, IL, USA: 2007. [Google Scholar]
- 48.Johnston C. The Wiley/NBS Registry of Mass Spectral Data. J. Chem. Educ. 1989;66:256. [Google Scholar]