Abstract
Diabetes mellitus is one of the most concerning non-communicable diseases worldwide. The prevalence of diabetes increased rapidly by the influence of socioeconomic interactions. The thrifty hypothesis postulates that certain genes that are involved in positive selection promote efficient fat deposition and storage. This is beneficial for the survival of mankind in adverse conditions. However, in this modern society, these genes have become disadvantageous as people are significantly less likely to experience famines and nutrition shortages. The socioeconomic development that has occurred during the 20th century induced abundance of food supplies in almost all regions of the world. This has led to a rapid rise in the prevalence of obesity, and type 2 diabetes as a consequence. Boom of diabetic pandemic in newly developed countries compare with others those who developed gradually can be explain by thrifty hypothesis, as a result of the difference in the exposure to environmental factors and famine by the ancestors leads. The globalization, urbanization, lack of physical activity, intake of high calorie food and migration is major cause of pandemic emergence of diabetes in high as well as middle and low-income countries.
Keywords: Diabetes, Thrifty, Phenotype, Drifty genotype, Epigenotype, Intra-uterine, Urbanization, Globalization
INTRODUCTION
Diabetes is a metabolic disorder characterized by hyper-glycemia resulting from the interaction of genetic and environmental factors. Type 2 Diabetes (T2D) is characterized by insufficient secretion of insulin from the β-cells of the pancreatic islets, coupled with insulin resistance. Globally, 415 million people are affected by diabetes and by 2040; this will rise to 642 million. The average prevalence of diabetes in the age profile (20–79 years) in high-, middle- and low-income economies countries are 9.37%, 9.56% and 4.68% respectively [1]. In past 3.5 decades, the prevalence of diabetes remarkably increased in middle income countries as well as high income countries with rapid economic growth (Table 1).
Table 1.
The age adjusted diabetes prevalence (%) in high-, middle- and low-income countries in 1980 [30] and 2017 [1]
| Countries | Prevalence of diabetes (%),1980* | Prevalence of diabetes (%), 2017 | Fold increase |
|---|---|---|---|
| High income countries | |||
| Steady economic growth | |||
| Austria | 3.92 | 6.4 | 1.63 |
| Netherlands | 3.77 | 5.3 | 1.40 |
| United Kingdom | 4.37 | 4.3 | 0.98 |
| Rapid economic growth | |||
| China | 4.28 | 9.7 | 2.26 |
| Qatar | 11.19 | 16.5 | 1.47 |
| Saudi Arabia | 8.20 | 17.7 | 2.15 |
| UAE | 9.98 | 17.3 | 1.73 |
| Kuwait | 12.41 | 15.8 | 1.27 |
| Middle income countries | |||
| Bangladesh | 3.80 | 8.4 | 2.21 |
| India | 4.13 | 10.4 | 2.51 |
| Pakistan | 5.40 | 8.3 | 1.53 |
| Ukraine | 5.57 | 7.1 | 1.27 |
| Low income countries | |||
| Afghanistan | 4.92 | 9.6 | 1.95 |
| Nepal | 3.80 | 7.3 | 1.92 |
| Madagascar | 2.52 | 3.9 | 1.54 |
| Guinea | 2.4 | 2.4 | 1.00 |
Diabetes prevalence (%) in 1980 were calculated by averaging the prevalence of men and women.
The human species underwent various changes during evolutionary history from the hunting and gathering period, the discovery of fire, to present times, mainly after the implementation of agriculture [2]. Humans are thought to have struggled to find food in sufficient amounts and quality to ensure survival for reproduction [3]. These changes exerted a direct influence on feeding habits and which impact on human health. Data on modern time hunter-gatherers as well as hunter-gatherer agriculturalists who consumed ancient diets indicate that such societies are broadly free of diseases (eg. obesity, coronary heart disease, and diabetes) during civilization [4].
To consider this, the prevalence of diabetes increased rapidly in last 30–40 years period and shown an enormous difference in the diabetes prevalence among population living in same economic status. This rapid increase in prevalence of diabetes cannot be explained by genetics only. The studies on different countries reported that, the factors that have been proposed as major reasons for rapid increase in diabetes worldwide are environmental factors (early life factors including intrauterine condition, low birth weight, childhood growth, age and time of exposure to famine), unhealthy diet (overconsumption of calorie rich diet), rapid economic development of the countries in short period of time (urbanization, globalization) and lack of physical activity contribute to increasing prevalence of obesity and in turn T2D [5–11]. Hence, we assume that thrifty hypothesis and its supporting factors have a major role in the increased prevalence of diabetes in various populations. This review explains the increased prevalence of diabetes in different population based on recent thrifty hypothesis with supporting data. There are several proposed hypothesis for the increased prevalence of diabetes in regards with thrifty concept, these include thrifty gene hypothesis, thrifty pheneotype hypothesis, drifty gene hypothesis, thrifty epigenotypes hypothesis.
HYPOTHESIS
1. Thrifty gene hypothesis
First time in 1962, James Neel, a professor of Human genetics proposed the thrifty gene hypothesis (TGH), addressing the etiology of diabetes and obesity from the perspective of evolutionary and genetic cause [12]. TGH explains that ancestor’s genes undergo multiple natural selection procedure and served energy stores can be used in the time of famine or fall. In TGH, individuals predisposed to develop diabetes might appear because of genetic variations. Human genetic variation might be beneficial in certain environmental conditions, or nonbeneficial in different environment and may cause disease. In recent times, when physical activity has decreased, and calories are in constant supply the “thrifty genotypes rendered by detrimental by progress” and support the development of obesity and diabetes [13]. The TGH fails to identify major thrifty genes. According to this hypothesis, population, are genetically adapted to an environment during which calories are deficient. The TGH explains that genes promote efficient fat deposition and the storage of fat was beneficial for ancestors for survival. In modern community, such genes are disadvantageous because not everybody who lives a modernized life is obese.
2. Thrifty phenotype hypothesis
The thrifty phenotype hypothesis (TPH), introduce by Hales and Barker in 1992. TPH propose etiological model for T2D. In this model, undernutrition acts not as a selection pressure acting over many generations to alter the genetic makeup of the population, but rather as an early environmental factor acting in early life. According to this hypothesis maladaptive responses occur as a result of environmentally induced alteration of physiology in the early life [14].
3. Drifty genotype hypothesis
Drifty genotype hypothesis (DGH) by John Speakman in a year 2008 defines that there will be a genetic drift in the genes rather than positive selection. The DGH state that the prevalence of thrifty genes cannot be under natural positive selection for energy storage genes. It is due to genetic drift resulting from the removal of predative natural selection pressures [15]. Even though drifty genotype hypotheses fail to explain the emerging diabetes susceptibility and related risk in modern developed world. But widely accepted thrifty phenotype and thrifty epigenotype hypothesis win to explain the role of environmental factors over the genes, impact of globalization and changes in lifestyle on diabetes susceptibility over modern industrialized countries.
4. Thrifty epigenotypes
Thrifty epigenotype hypothesis (TEH) was proposed by Reinhard Stoger in 2008. Thrifty epigenotypes hypothesis addresses the limitations of thrifty genotype and thrifty phenotype and unifies merits of both hypotheses [16]. A growing body of evidences supports the idea that epigenetic changes such as DNA methylation and histone protein modification, both involve chromatin remodeling and contributing to epigenetic reprogramming of the fetus. Fetal adaptation to impaired nutritional environment settings presumes profound changes in gene expression. This involves regulation of tissue-specific motifs of methylated cytosine residues, modulation of the histone acetylation and deacetylation alterations, cell differentiation, and stem cell pluripotency. Intrauterine growth restriction is mostly associated with the induction of continuous changes in tissue structure and its functionality. Similarly, a maternal obesogenic environment is most probably associated with epigenetic reprogramming of glucose and lipid metabolism [17]. Epigenotype hypothesis suggest that an individual acquire metabolic thrift established during a time of nutrient oversupplied or malnutrition. The phenotypes upon confronting different environmental conditions from those existed during the programming period, established phenotype fails to adapt, which rises disease risk. Moreover, epigenotypes can be inherited across generations and predisposed to obesity and T2D (Table 2) [16–18].
Table 2.
Thrifty hypothesis and drawbacks
| Hypothesis | Explanation | Draw backs |
|---|---|---|
| Thrifty gene hypothesis | Genes undergo multiple natural selection procedure and serve energy stores can be used in the time of famine or fall. | Unable to explain the heterogeneity of diabetes and obesity between and within populations |
| Thrifty phenotype | The early environmental factors acting in early life acts as a selection pressure over many generations to alter the genetic makeup of the population. | Fails to explain the role of current living environmental conditions |
| Drifty gene hypothesis | The prevalence of thrifty genes or fat storage gene is due to genetic drift in the genes rather than positive selection. | Fails to explain the emerging diabetes susceptibility and related risk in modern developed world. |
| Epigenotype | An individual acquires metabolic thrift established during a time of nutrient oversupplied or malnutrition. The phenotypes upon confronting different environmental conditions from those existed during the programming period, established phenotype fails to adapt, which rises disease risk. |
THRIFTY HYPOTHESIS AND SUPPORTING EVIDENCE
At different times, different hypothesis was given to sudden rise in prevalence of diabetes and obesity. As we all know that the genetic selection takes many decades to show any effect in gene changes. Therefore, evidence-based factors supporting thrifty hypothesis mainly includes environmental factors such as early life factors including intra-uterine condition, low birth weight, childhood growth, age and time exposure to famine, unhealthy diet by overconsumption of calorie rich diet, sudden economic development of the countries, lack of physical activity, migration and epigenetic mechanisms.
1. Environmental factors
According to Hales and Barker, poor nutrition during the fetal period or infancy, followed by good or over nutrition is major cause of T2D [14]. In support of this hypothesis, altered glucose–insulin metabolism was found in the findings from experiments in rodents and other animals having maternal energy deficit during pregnancy, especially when combined with overfeeding in the postnatal period [19]. Lumey et al. assess the risk of T2D in Ukrainian adults whose mothers were pregnant during Ukraine famine (Holodomar) of 1932–1933 [20]. This study reported that the T2D increases 1.5 times in individuals who were born in 1934 and 1.3 times increased T2D prevalence in the adults who born in the region affected severe famine [20]. These findings extent to other studies reporting diabetes related outcomes after prenatal famine in the high-income countries such as Netherlands, China and Austria [21]. The evidence from the brief duration Dutch famine provides information about the effects of fetal undernutrition at different stages of gestation confirmed that famine exposure in early gestation led to disturbance of lipid metabolism while in mid and late gestation it led to disturbance of glucose-insulin metabolism [8].
Birth weight is an indicator of intrauterine nutrition. A study with four-year-old Indian migrant children supports that children with reduced intra-uterine growth or low birth weight (< 2.4 kg) have reduced glucose homeostasis after a glucose challenge [22]. Another study, low-birth-weight Pimas are thinner at ages 5–29 years, yet they are more insulin resistant relative to their body size than those of normal birth weight and leads to increased risk for T2D [23]. However, an evidence from a low-income country Gambia, reveals that small babies, including those born during a nutritionally-debilitating hungry season, maintain excellent cardiovascular health into adulthood, with a complete absence of the metabolic syndrome, as long as they retain their ‘lean, fit and frugal’ lifestyle in the rural areas [24]. These findings confirmed TPH that maternal and childhood undernutrition and later exposure to western diet and life styles through migration to urban area or improved economic conditions may promote increase prevalence of obesity and in turn T2D. The Great Leap Forward Famine (GLFF) in China (1959–1961) explains the adverse effect and selection effect vary among famine exposure at different age. Prenatal/infant exposure to GLFF leads to reduced risk of having chronic disease in later lives and puberty exposure to the GLFF results in increased risk of having chronic diseases [10].
2. Unhealthy diet
The Pacific Island of Nauru gives an insight into how improving nutrition may be associated with a rise in T2D followed by a famine period [9]. The traditional lifestyle of Nauruan was based on agriculture and fishing and involved frequent episodes of scarcity of food because of droughts and poor quality of soil. The Nauruan were chronically mal-nourished until the end of the Second World War. After the Second World War, new wealth from phosphate mines leads increased sugar consumption (average of a pound per day) and increased use of store-bought food by Nauruan. The Nauruan people had “irregular eating habits” consisting of three high-caloric meals per day, supplemented with frequent in-between meal snacking. Few vegetables were eaten. The average caloric intake for males 20–39 exceeded 8,700 calories [25]. The immediate consequence of this was a great increase in obesity and the emergence of an ‘epidemic’ of T2D [9].
The rise in chronic diseases is link to dietary pattern of high income Middle Eastern gulf countries. The per capita cereal consumption in Saudi Arabia is decreased by 0.1% and meat consumption is increased on average by 2.2% per year between 1993 and 2003. The over consumption of western diet also has an impact on increased diabetes prevalence in above Middle Eastern countries [26]. Other studies with high income countries reported that individuals exposed to marked food shortage during gestation prefer high-fat diet and have higher energy intake in late middle age, increasing the risk for obesity and diabetes over time [27–29]. This also confirmed the thrifty epigenotype hypothesis that phenotypes upon confronting different environmental conditions from those existed during the programming period, established phenotype fails to adapt, which rises disease risk.
3. Rapid economic development of the countries in short period of time
The high-income countries in Middle Eastern region are Saudi Arabia, UAE, Kuwait and Qatar. The Arabians whose traditional way of life was mostly dependent on oasis-dwelling farming and herding, fishing, crafts work and long-distance trading. The oil and gas exploitation has led to a sudden and exceptional economic growth of the above countries and lead to the richest economies in the world. The sudden changes in lifestyle along with the economic growth they have experienced contributes to a marked rise in obesity and chronic disease prevalence [26]. Data collected during the last 35 years reveals that there is a three-fold increase in diabetes prevalence from 4% to 11% reported in Middle Eastern countries. Similarly, the traditional lifestyle of Nauruan was based on agriculture and fishing and involved frequent episodes of scarcity of food because of droughts and poor quality of soil. The Nauruan were chronically mal-nourished until the end of the Second World War. The flourishing phosphate mines built up after the war drastically changed the economic and nutritional welfare of the population leading to a great increase in T2D [9].
Even though Europeans have a high economic status, the increase of diabetes prevalence during the last 3 decades is from 5.45% to 6.2% only [30]. The Europeans are the people who were less exposed to famine traditionally. Europe’s famines were ended by following factors, by efficient distribution of surplus grain to famine areas; increasingly efficient food transport by land and sea; emergence of diversified agriculture after AD 1492 by broadening the agriculture of crops such as potatoes and corn (maize) which were imported from outside and thereby reducing the risk of starvation from failure of a single crop; and finally the ‘irrigation agriculture’ which reduced the risk of a crop failure as well as expense of food transport within Europe. This confirmed that those stayed in same environmental condition would have undergone little selection of thrifty genotype hence less prevalence of diabetes [25]. The above evidence confirms the thrifty epigenotype hypothesis that an individual acquires metabolic thrift established during a time of nutrient oversupply or malnutrition.
Globalization and economic development has made food products, such as edible oil and sugar, more accessible and relatively cheaper than in the past which leads to spurred nutrition transitions in many developing nations [31]. This nutritional transition typically involves more frequent intake of fast foods, increased consumption of energy dense foods, intake of animal fat and decreased fiber. Notable changes that have occurred in Asia in the last several decades include a large shift from consumption of coarse grains to polished rice and refined wheat. The high/middle income or urban population among India and China starts reduced intake of cereals and consumption of low nutrition food and higher energy intake among the poor. In both groups increased consumption of fat and high intake of meat, edible oil in China and increased consumption of dairy products, especially highly saturated ghee (clarified butter), and added sugar in India [32]. The popularity of fast food in China, exemplifies the influence of food marketing, fueled by globalization in the urban areas and also nutrition transition leads to a rapid increase of overweight/obese people in last few decades [33]. So, before the globalization period there developed a metabolic thrift on individuals and sudden establishment of globalization leads phenotype fails to adapt, which rises the diabetes prevalence.
Evidence from the urbanization plays a critical role in rapid rise of diabetes prevalence among the population of same ethnic background. The Africans lived in Cape Town, faced a rapid development of new townships in 1983. The studies held after a decade clearly established the role of urbanization in South Africa, an increased prevalence of diabetes from 3.6% to 8% in two decades [34]. The studies held in India with two populations having considerable socioeconomic differences but belonging to the same ethnic background, having high literacy and same food habits confirmed that the high prevalence of T2D in the urban southern Indian population when compare with rural areas [35]. As a result of urbanization diabetes increased in rural Bangladesh and Pakistan and suburban and rural areas in Nepal [36–38]. In high income country like Saudi Arabia the rapid economic development leads to increased urbanization. An article published in 2004 reported that the overall prevalence of Diabetes mellitus (DM) in adults in Saudi Arabia was 23.7% and also DM was more prevalent among Saudis living in urban areas of 25.5% compared to rural Saudis of 19.5% [39].
4. Lack of physical activity
The lack of physical activity and increased prevalence of diabetes can be explained by the evidence from high income population U.S Pima Indians, and middle-income population Mexican Pimas, who have similar genetic background but adapted to contrasting lifestyles. But the situations in high income middle eastern countries like Saudi Arabia, Kuwait and UAE who have a diabetes prevalence of more than 20% is quite different. Here the decreased physical activity not only due to the greater availability of cars and mechanic appliances, cheap migrant workers and influence of computers and electronic devices but also may have been reinforced by cultural impediments to physical exercise and sports. According to the National Oceanic Atmospheric Administration (NOAA) heat index in these countries are at dangerous level [26]. So, thrift established during the time of low economic conditions fails to adapt the programmed phenotype during sudden changes in lifestyle including sedentary life style leads to the sudden increase in prevalence of diabetes in a very short period. So here confirmed thrifty epigenotype hypothesis.
5. Migration
Another supporting factor for TPH is migration from low income countries to high income countries. The people who migrate from their country of origin to a new host country are commonly found to have a higher diabetes prevalence compared to the population in the new host country. The relation between migration status and disease pattern is complex and it may depend on the conditions of their country of origin, their new host country, and also conditions during migration process itself [40–43]. In a 20 year longitudinal follow-up study with first generation migrants of Indian Asian (n = 839) and African Caribbean (n = 335) residing in the UK compared with Europeans (n = 1354). The results from the above study shows that Indian Asian population had an almost 3 fold excess incidence of T2D and African Caribbeans had more than 2 fold excess incidences than native Europeans [44].
In Middle Eastern countries whose overall prevalence of diabetes in 2014 was reported as 11.4% and age standardized prevalence is 13.6% and is considered the third largest prevalence among region specific prevalence. In 2003 studies on high and middle-income Arab- community who immigrated to Michigan reported that 41% of the population 20–75 years of age and >70% of those over 60 years of age are affected by abnormal glucose tolerance [45]. But age standardized prevalence of diabetes in Middle Eastern countries in 2003 reported as 9.9% only [30].
Among high income groups the European immigrants of United States and Australia who have British and German ancestry has much higher prevalence of T2D than the people who are living in Europe under similar conditions. From the evidence of history-based study. Zimmet et al. claiming that the Europeans who stayed at home country tended to be richer than those who emigrated [46]. The predisposed gene for T2D in native group may already go to multiple selection procedure in time of availability of abundant food, whereas those who emigrated may have been the starvation-prone poor who still carried the thrifty genotype [46]. So, this may cause of higher prevalence of diabetes in migratory group of Europeans. To clarify the above statements, they compare the Europeans still living in Europe with migrated group and found that overseas descendants of Irish emigrants in the 1840s are more susceptible to diabetes than Ireland’s natives [25]. These examples give a clear idea that the factors affecting the difference in diabetes prevalence in a population is not only by the population of origin, but also factors related to migration itself, to social and lifestyle factors in the new host countries. From the above evidences it proved the thrifty phenotype hypothesis that undernutrition acts not as a selection pressure acting over many generations to alter the genetic makeup of the population, but rather as an early environmental factor acting in early life and also the maladaptive responses occur as a result of environmentally induced alteration of physiology in the early life.
6. Epigenotypes
The “thrifty genes” favors fat storage during a period of famine and leads to obesity and diabetes during a time of plenty. Here thrifty gene can better be explained by the loss of uricase which leads to increased uric acid and later obesity and diabetes. The evolution based genetic studies reported that reduced availability of fruits and climatic changes results in the loss of uricase in human ancestors in mid–Miocene period leads to the consequence of increasing susceptibility to become obese [47–51]. The increasing susceptibility of fat in liver cells is mediated by the action of fructose and thus it seems that uricase act as a ‘thrifty gene’ which increases the chance to get diabetes [52].
Among the three major ancestral groups of Pacific people (Polynesians, Micronesians, and Melanesians), Micronesians (such as those living in Nauru) and the Polynesians (including the New Zealand Māori) are the population who were more susceptible to obesity and having high prevalence of diabetes [53]. Genetic studies suggest the Micronesians and Polynesians were originated from aborigines in Taiwan. Interestingly, the serum uric acid levels in Taiwanese aborigines, Micronesians and Polynesians appear to be higher than the other ethnic group [54,55]. The Taiwanese aboriginal population also suffers from higher rates of obesity and diabetes than the Han Chinese living in Taiwan [56]. Johnson et al. propose that the epigenetic modifications together with polymorphism in Polynesian and Micronesian peoples may have an effect on higher endogenous uric acid level [57].
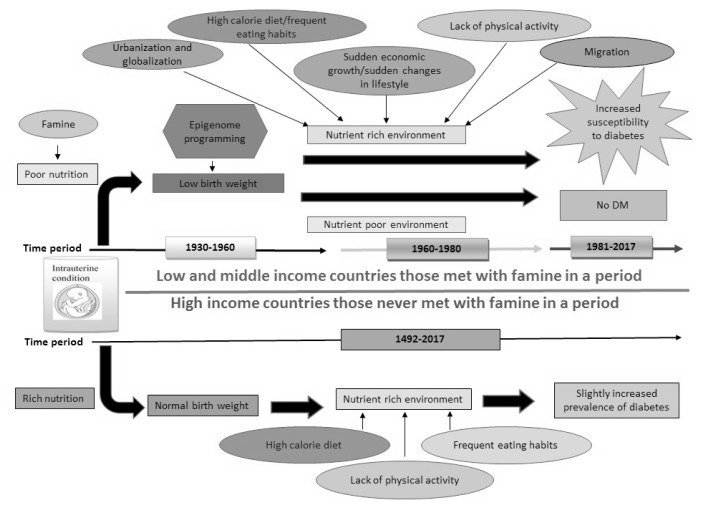
The evidence of the epigenetics for obesity and T2D always points towards somatic tissues in the fetus and intra-uterine environment conditions which appear to be a considerable determinant of the body fat mass of an individual later in life. It was reported that the future function of hypothalamic centers regulating food intake is influenced by the number of calories available at crucial times of development [58]. The effect of all above supporting factors of thrifty hypothesis over a period of time on low, middle and high-income countries were briefly illustrated in Fig. 1.
Fig. 1.
Different supporting factors of thrifty hypothesis on the increased prevalence of diabetes mellitus in low and middle-income countries those that experienced a famine period. Low- and middle-income countries those met with famine, undergoing undernutrition in fetal stage resulting in low birth weight children. These children undergo epigenetic programming and those who met a nutrient rich environment and went through some certain conditions such as lack of physical activity, intake of high calorie diet, frequent eating habits, sudden economic growth, sudden changes in lifestyle, urbanization, globalization, and immigration to high income country in their later life were more susceptible to diabetes. While, those who stayed in poor nutrient environment in their later life were less susceptible to diabetes. Unlike low income countries, high-income countries that never experienced (or managed their famine period), were born with a normal weight, and then were also subjected to nutrient rich environment with lack of physical activity, intake of high calorie diet, frequent eating habits in their later life, will either not develop diabetes or will have a lower risk.
CONCLUSION
Now a day there are a lot of studies that support thrifty hypothesis to explain sudden increment of diabetes in different populations. These evidence-based studies can give a better understanding on thrifty hypothesis. The increased prevalence of diabetes is not only affecting the high-income countries but also low and middle-income countries. The current thrifty hypotheses added that the selection pressures faced by the ancestors of all inhabitants of developed countries today were different which leads to the difference in the prevalence of diabetes. Studies on effect of change in environmental condition and inherited traits has gone stronger and opens the door to acceptable hypothesis such as thrifty phenotype and epigenotype. Apart from this the evidence from changes in eating habits, intake of high calorie foods, globalization, and urbanization, lack of physical activity, migration and interaction of environment on genes supports the pandemic increase of diabetes in developed world.
ACKNOWLEDGEMENT
The authors would like to acknowledge the members of biochemistry department, Strategic Center for Diabetes Research for supporting, assisting and editing the manuscript.
Footnotes
CONFLICTS OF INTERESTS
None to declare.
REFERENCES
- 1.International Diabetes Federation. IDF Diabetes Atlas. 7th ed. International Diabetes Federation; Brussel (Belgium): 2017. Available from: https://idf.org/e-library/epidemiology-research/diabetes-atlas/13-diabetes-atlas-seventh-edition.html. [Google Scholar]
- 2.Fernando SZ. Evolution of the human feeding behavior. Psychol Neurosci. 2011;4:131–41. doi: 10.3922/j.psns.2011.1.015. [DOI] [Google Scholar]
- 3.Anette PG, Oluf P. Omics driven discoveries in preventing and treatment of type 2 diabetes. Eur J clin Invest. 2012;42:579–88. doi: 10.1111/j.1365-2362.2012.02678.x. [DOI] [PubMed] [Google Scholar]
- 4.Milton K. Hunter-gatherer diets—a different. Am J Clin Nutr. 2000;71:665–7. doi: 10.1093/ajcn/71.3.665. [DOI] [PubMed] [Google Scholar]
- 5.Hu FB. Globalization of diabetes: The role of diet, lifestyle, and genes. Diabetes Care. 2011;34:1249–57. doi: 10.2337/dc11-0442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nanditha A, Ma RC, Ramachandran A, Snehalatha C, Chan JC, Chia KS, Shaw JE, Zimmet PZ. Diabetes in Asia and the Pacific: Implications for the global epidemic. Diabetes Care. 2016;39:472–85. doi: 10.2337/dc15-1536. [DOI] [PubMed] [Google Scholar]
- 7.Crowther NJ, Cameron N, Trusler J, Gray IP. Association between poor glucose tolerance and rapid post-natal weight gain in seven-year-old children. Diabetologia. 1998;41:1163–7. doi: 10.1007/s001250051046. [DOI] [PubMed] [Google Scholar]
- 8.Hales CN, Barker DJ. The thrifty phenotype hypothesis. Br Med Bull. 2001;60:5–20. doi: 10.1093/bmb/60.1.5. [DOI] [PubMed] [Google Scholar]
- 9.Lindsay RS, Dabelea D, Roumain J, Hanson RL, Bennett PH, Knowler WC. Type 2 diabetes and low birth weight: The role of paternal inheritance in the association of low birth weight and diabetes. Diabetes. 2000;49:445–9. doi: 10.2337/diabetes.49.3.445. [DOI] [PubMed] [Google Scholar]
- 10.Hu X, Liu GG, Fan M. Long term effects of famine on chronic diseases: Evidence from China’s great leap forward famine [Internet] 2015 doi: 10.2139/ssrn.2621044. [updated 2015 Jun 20]. Available from: [DOI] [PubMed]
- 11.Chakravarthy MV, Booth FW. Eating, exercise, and “thrifty” genotypes: connecting the dots toward an evolutionary understanding of modern chronic diseases. J Appl Physiol. 2004;96:3–10. doi: 10.1152/japplphysiol.00757.2003. [DOI] [PubMed] [Google Scholar]
- 12.Neel JV. Diabetes mellitus: a “thrifty” genotype rendered detrimental by “progress”? Am J Hum Genet. 1962;14:353–62. [PMC free article] [PubMed] [Google Scholar]
- 13.Reaven GM. Muscle insulin resistance is the (“not-so”) thrifty genotype. Diabetologia. 1998;41:482–4. doi: 10.1007/s001250050933. [DOI] [PubMed] [Google Scholar]
- 14.Hales CN, Barker DJ. Type 2 (non-insulin-dependent) diabetes mellitus: The thrifty phenotype hypothesis. Diabetologia. 1992;35:595–601. doi: 10.1007/BF00400248. [DOI] [PubMed] [Google Scholar]
- 15.Speakman JR. Thrifty genes for obesity, an attractive but flawed idea, and an alternative perspective: the ‘drifty gene’ hypothesis. Int J Obes (Lond) 2008;32:1611–7. doi: 10.1038/ijo.2008.161. [DOI] [PubMed] [Google Scholar]
- 16.Stöger R. The thrifty epigenotype: Acquired and herit-able predisposition for obesity and diabetes? Bioessays. 2008;30:156–66. doi: 10.1002/bies.20700. [DOI] [PubMed] [Google Scholar]
- 17.Sookoian S, Gianotti TF, Burgueño AL, Pirola CJ. Fetal metabolic programming and epigenetic modifications: A systems biology approach. Pediatr Res. 2013;73:531–42. doi: 10.1038/pr.2013.2. [DOI] [PubMed] [Google Scholar]
- 18.Tomar AS, Tallapragada DS, Nongmaithem SS, Shrestha S, Yajnik CS, Chandak GR. Curr Obes Rep. 2015;44:418–28. doi: 10.1007/s13679-015-0175-6. [DOI] [PubMed] [Google Scholar]
- 19.Nielsen JH, Haase TN, Jaksch C, Nalla A, Søstrup B, Nalla AA, Larsen L, Rasmussen M, Dalgaard LT, Gaarn LW, Thams P, Kofod H, Billestrup N. Impact of fetal and neonatal environment on beta cell function and development of diabetes. Acta Obstet Gynecol Scand. 2014;93:1109–22. doi: 10.1111/aogs.12504. [DOI] [PubMed] [Google Scholar]
- 20.Lumey LH, Khalangot MD, Vaiserman AM. Association between type 2 diabetes and prenatal exposure to the Ukraine famine of 1932–33: A retrospective cohort study. Lancet Diabetes Endocrinol. 2015;3:787–94. doi: 10.1016/S2213-8587(15)00279-X. [DOI] [PubMed] [Google Scholar]
- 21.deRooij SR, Roseboom TJ, Painter RC. Famines in the last 100 years: Implications for diabetes. Curr Diab Rep. 2014;14:536. doi: 10.1007/s11892-014-0536-7. [DOI] [PubMed] [Google Scholar]
- 22.Yajnik CS, Fall CH, Vaidya U, Pandit AN, Bavdekar A, Bhat DS, Osmond C, Hales CN, Barker DJ. Fetal growth and glucose and insulin metabolism in four-year-old Indian children. Diabet Med. 1995;12:330–6. doi: 10.1111/j.1464-5491.1995.tb00487.x. [DOI] [PubMed] [Google Scholar]
- 23.Dabelea D, Pettitt DJ, Hanson RL, Imperatore G, Bennett PH, Knowler WC. Birth weight, type 2 diabetes, and insulin resistance in Pima Indian children and young adults. Diabetes Care. 1999;22:944–50. doi: 10.2337/diacare.22.6.944. [DOI] [PubMed] [Google Scholar]
- 24.Moore SE, Halsall I, Howarth D, Poskitt EME, Prentice AM. Glucose, insulin and lipid metabolism in rural Gambians exposed to early malnutrition. Diabet Med. 2001;18:646–53. doi: 10.1046/j.1464-5491.2001.00565.x. [DOI] [PubMed] [Google Scholar]
- 25.Diamond J. The double puzzle of diabetes. Nature. 2003;423:599–602. doi: 10.1038/423599a. [DOI] [PubMed] [Google Scholar]
- 26.Klautzer L, Becker J, Mattke S. The curse of wealth -Middle Eastern countries need to address the rapidly rising burden of diabetes. IJHPM. 2014;2:109–14. doi: 10.15171/ijhpm.2014.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lussana F, Painter RC, Ocke MC, Buller HR, Bossuyt PM, Roseboom TJ. Prenatal exposure to the Dutch famine is associated with a preference for fatty foods and a more atherogenic lipid profile. Am J Clin Nutr. 2008;88:1648–52. doi: 10.3945/ajcn.2008.26140. [DOI] [PubMed] [Google Scholar]
- 28.Stein AD, Rundle A, Wada N, Goldbohm RA, Lumey LH. Associations of gestational exposure to famine with energy balance and macronutrient density of the diet at age 58 years differ according to the reference population used. J Nutr. 2009;139:1555–61. doi: 10.3945/jn.109.105536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gross LS, Li L, Ford ES, Liu S. Increased consumption of refined carbohydrates and the epidemic of type 2 diabetes in the United States: An ecologic assessment. Am J Clin Nutr. 2004;79:774–9. doi: 10.1093/ajcn/79.5.774. [DOI] [PubMed] [Google Scholar]
- 30.NCD Risk Factor Collaboration. Diabetes: Evolution of diabetes over time [Internet] 2016 Available from: http://www.ncdrisc.org/d-diabetes.html.
- 31.Popkin BM. The nutrition transition and obesity in the developing world. J Nutr. 2001;131:871S–873S. doi: 10.1093/jn/131.3.871S. [DOI] [PubMed] [Google Scholar]
- 32.Popkin BM, Horton S, Kim S, Mahal A, Shuigao J. Trends in diet, nutritional status, and diet-related non-communicable diseases in China and India: The economic costs of the nutrition transition. Nutr Rev. 2001;59:379–90. doi: 10.1111/j.1753-4887.2001.tb06967.x. [DOI] [PubMed] [Google Scholar]
- 33.Bell AC, Ge K, Popkin BM. Weight gain and its predictors in Chinese adults. Int J Obes Relat Metab Disord. 2001;257:1079–86. doi: 10.1038/sj.ijo.0801651. [DOI] [PubMed] [Google Scholar]
- 34.Levitt NS, Katzenellenbogen JM, Bradshaw D, Hoffman MN, Bonnici F. The Prevalence and identification of risk factors for NIDDM in urban Africans in Cape Town, South Africa. Diabetes Care. 1993;16:601–7. doi: 10.2337/diacare.16.4.601. [DOI] [PubMed] [Google Scholar]
- 35.Ramachandran A, Snehalatha C, Dharmaraj C, Viswanathan M. Prevalence of glucose intolerance in Asian Indians. Urban-rural difference and significance of upper body adiposity. Diabetes Care. 1992;15:1348–55. doi: 10.2337/diacare.15.10.1348. [DOI] [PubMed] [Google Scholar]
- 36.Rahim MA, Hussain A, Azad Khan AK, Sayeed MA, Keramat Ali SM, Vaaler S. Rising prevalence of type 2 diabetes in rural Bangladesh: a population-based study. Diabetes Res Clin Pract. 2007;77:300–5. doi: 10.1016/j.diabres.2006.11.010. [DOI] [PubMed] [Google Scholar]
- 37.Shera AS, Jawad F, Maqsood A. Prevalence of diabetes in Pakistan. Diabetes Res Clin Pract. 2007;76:219–22. doi: 10.1016/j.diabres.2006.08.011. [DOI] [PubMed] [Google Scholar]
- 38.Sasaki H, Kawasaki T, Ogaki T, Kobayashi S, Itoh K, Yoshimizu Y, et al. The prevalence of diabetes mellitus and impaired fasting glucose/glycemia (IFG) in suburban and rural Nepal—the communities-based cross-sectional study during the democratic movements in 1990. Diabetes Res Clin Prac. 2005;67:167–74. doi: 10.1016/j.diabres.2004.06.012. [DOI] [PubMed] [Google Scholar]
- 39.Al-Nozha MM, Al-Maatouq MA, Al-Mazrou YY, Al-Harthi SS, Arafah MR, Khalil MZ, et al. Diabetes mellitus in Saudi Arabia. Saudi Med J. 2004;25:1603–10. [PubMed] [Google Scholar]
- 40.Kristensen JK, Bak JF, Wittrup I, Lauritzen T. Diabetes prevalence and quality of diabetes care among Lebanese or Turkish immigrants compared to a native Danish population. Prim Care Diabetes. 2007;1:159–65. doi: 10.1016/j.pcd.2007.07.007. [DOI] [PubMed] [Google Scholar]
- 41.Ujcic-Voortman JK, Schram MT, Jacobs-van der Bruggen MA, Verhoeff AP, Baan CA. Diabetes prevalence and risk factors among ethnic minorities. Eur J Public Health. 2009;19:511–5. doi: 10.1093/eurpub/ckp096. [DOI] [PubMed] [Google Scholar]
- 42.Jenum AK, Diep LM, Holmboe-Ottesen G, Holme IM, Kumar BN, Birkeland KI. Diabetes susceptibility in ethnic minority groups from Turkey, Vietnam, Sri Lanka and Pakistan compared with Norwegians - the association with adiposity is strongest for ethnic minority women. BMC Public Health. 2012;12:150. doi: 10.1186/1471-2458-12-150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Misra A, Ganda OP. Migration and its impact on adiposity and type 2 diabetes. Nutr. 2007;23:696–708. doi: 10.1016/j.nut.2007.06.008. [DOI] [PubMed] [Google Scholar]
- 44.Tillin T, Hughes AD, Godsland IF, Whincup P, Forouhi NG, Welsh P, Sattar N, McKeigue PM, Chaturvedi N. Insulin resistance and truncal obesity as important determinants of the greater incidence of diabetes in Indian Asians and African Caribbeans compared with Europeans: the Southall And Brent REvisited (SABRE) cohort. Diabetes Care. 2013;36:383–93. doi: 10.2337/dc12-0544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jaber LA, Brown MB, Hammad A, et al. Epidemiology of diabetes among Arab Americans. Diabetes Care. 2003;26:308–13. doi: 10.2337/diacare.26.2.308. [DOI] [PubMed] [Google Scholar]
- 46.Zimmet P. The challenge of diabetes - diagnosis, classification, ‘cocacolonization’, and the diabetes epidemic. In: Fischer EP, Moller G, editors. The medical challenge: Complex traits. Piper; Munich (Germany): 1997. pp. 55–110. [Google Scholar]
- 47.Johnson RJ, Lanaspa MA, Gaucher EA. Uric acid: A danger signal from the RNA world that may have a role in the epidemic of obesity, metabolic syndrome, and cardiorenal disease: evolutionary considerations. Semin Nephrol. 2011;31:394–9. doi: 10.1016/j.semnephrol.2011.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Watanabe S, Kang DH, Feng L, Nakagawa T, Kanellis J, Lan H, Mazzali M, Johnson RJ. Uric acid, hominoid evolution, and the pathogenesis of salt-sensitivity. Hypertension. 2002;40:355–60. doi: 10.1161/01.HYP.0000028589.66335.AA. [DOI] [PubMed] [Google Scholar]
- 49.Johnson RJ, Andrews P. Fructose, uricase, and the back-to-Africa hypothesis. Evol Anthropol. 2010;19:250–7. doi: 10.1002/evan.20266. [DOI] [Google Scholar]
- 50.Oda M, Satta Y, Takenaka O, Takahata N. Loss of urate oxidase activity in hominoids and its evolutionary implications. Mol Biol Evol. 2002;19:640–53. doi: 10.1093/oxfordjournals.molbev.a004123. [DOI] [PubMed] [Google Scholar]
- 51.Keebaugh AC, Thomas JW. The evolutionary fate of the genes encoding the purine catabolic enzymes in hominoids, birds, and reptiles. Mol Biol Evol. 2010;27:1359–69. doi: 10.1093/molbev/msq022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kratzer JT, Lanaspa MA, Murphy MN, Cicerchi C, Graves CL, Tipton PA, Ortlund EA, Johnson RJ, Gaucher EA. Evolutionary history and metabolic insights of ancient mammalian uricases. Proc Natl Acad Sci U S A. 2014;111:3763–8. doi: 10.1073/pnas.1320393111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zimmet P. Epidemiology of diabetes and its macro-vascular manifestations in Pacific populations: the medical effects of social progress. Diabetes Care. 1979;2:144–53. doi: 10.2337/diacare.2.2.144. [DOI] [PubMed] [Google Scholar]
- 54.Lennane GA, Rose BS, Isdale IC. Gout in the Maori. Ann Rheum Dis. 1960;19:120–5. doi: 10.1136/ard.19.2.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cheng LS, Chiang SL, Tu HP, Chang SJ, Wang TN, Ko AM, et al. Genome wide scan for gout in taiwanese aborigines reveals linkage to chromosome 4q25. Am J Hum Genet. 2004;75:498–503. doi: 10.1086/423429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ho CS, Tsai AC. Prevalence of overweight and obesity and its associated factors in aboriginal Taiwanese: findings from the 2001 National Health Interview Survey in Taiwan. Asia Pac J Clin Nutr. 2007;16:572–9. [PubMed] [Google Scholar]
- 57.Johnson RJ, Lanaspa M, Sanchez-Lozada L, Rivard C, Bjornstad, Merriman T, Sundborn G. Fat storage syndrome in Pacific peoples: A combination of environment and genetics? Pac Health Dialog. 2014;20:11–6. [PubMed] [Google Scholar]
- 58.Stöger R. The thrifty epigenotype: An acquired and heritable predisposition for obesity and diabetes? BioEssays. 2008;30:156–66. doi: 10.1002/bies.20700. [DOI] [PubMed] [Google Scholar]