Abstract
Cancer is a complex interaction among multiple signaling pathways involving a variety of target molecules. Cancer causes morbidity and mortality in millions of people worldwide, and due to its prevalence, the discovery of novel anticancer drugs is urgently required. Nature is considered an important source of the discovery of anticancer treatments, and many of the cytotoxic medicines in clinics today are derived from plants and other natural sources. Reactive oxygen species (ROS) induce a variety of human cancers, and antioxidants or scavengers are used to counteract them. The current study reports on the screening of extracts from 57 plants that are used in the galilee district as a food and/or for traditional medicine. Investigating the free radical scavenging capacity and these plants, and their cytotoxicity, may prove helpful to high-throughput screening projects that use antioxidants and cytotoxic natural products. The current study assessed the correlation between free radical scavenging and cytotoxicity. Correlational analysis is important for increasing the efficiency of the screening process. In the present study, free radical scavenging was assessed using a DPPH assay, while cytotoxicity was measured using a XTT assay. A total of 9 extracts were indicated to exhibit EC50 values <250 µg/ml, and 4 others exhibited a high antioxidant content, with EC50 values, for free radical scavenging, of <0.5 µg/ml. An in-depth analysis of the results revealed that the extracts of plants that exhibit an EC50 of free radical scavenging ≤10 µg/ml show a degree of enrichment toward increased cytotoxicity. It is recommended that future studies test the validity of the conclusions of the current study on other cancer cell-lines, and isolate and identify the bioactive agents that are found in the most cytotoxic extracts of plants.
Keywords: cytotoxicity, antioxidant, free radical scavenging, medicinal plant, edible plant
Introduction
A large variety of plant-based nutrients and phytochemicals consumed by humans has long been considered to be associated with human health, and has even proved to reduce the risk of inflammation (1–3) and illnesses such as diabetes (4), cardiovascular (5), neurodegenerative (6), microbial-related diseases (7,8), and certain types of cancer (9–13). Cancer continues to be a major health challenge, constituting the second-leading cause of death worldwide, despite intensive and extensive research that has revealed much about its biology in last few decades (14,15). In parallel, considerable progress in anti-cancer therapies has been made, allowing the cure of cancer patients and helping to prolong their survival rate. Despite such advances in its early detection, and improvements in treatment and prevention, cancer remains a major challenge in terms of morbidity and mortality. Therefore, enormous scientific and commercial endeavors have been made to discover further anti-cancer agents. In view of this, natural products that have been studied for a long time, have been found to have pharmacologic activity, and have proved to be safe with long-term exposure (16). Some of these plant-extracted products are currently available on the pharmaceutical market as antioxidants or scavengers, and are used to counteract reactive oxygen species (ROS), the triggers for various types of human cancer (17). ROS, produced either endogenously or by exogenous stimuli, can damage DNA, proteins, and lipids, which can lead to the transformation of normal cells into cancer cells through the mutation of key genes (18). Cancer initiation and progression can also occur due to an unbalanced redox equilibrium, an inherent defense system of cells that endogenously generate and scavenge ROS, leading to increased DNA damage, prevention of cell apoptosis, and consequently, to a higher rate of cell survival (19,20). Given this, the excessive production of intracellular ROS has been targeted by antioxidants as therapeutic agents to prevent or suppress the development or propagation of cancer cells (21). Many plants have been found to have significant ROS-scavenging activity (antioxidant activity), which is associated with cytotoxicity (antiproliferative activity) toward cancer cells, and thus could be used as therapeutic and preventive agents (22–24). This association is clear in the observations that curcumin, a natural polyphenol derived from the rhizome of turmeric, and quercetin, an anti-oxidant derived from fruits and vegetables, have been shown to have potent free radical-scavenging and cytotoxic activity (25,26).
As cancer cells have developed the capability to escape apoptosis through a number of mechanisms-cellular transformation, apoptosis dysregulation, proliferation, migration, angiogenesis, and metastasis. Patients with cancer have been treated and managed by conventional surgery, chemotherapy, or radiotherapy. However, researchers have begun to find encouraging clinical results pointing to the value of plant-based products in cancer treatment, and physicians have started to use these medications. They support, and even strengthen, various systems of the body that are under stress due to chemical toxicity or traumatic events. In contrast to chemotherapy, which often induces a number of undesired toxic side effects, natural therapies, such as the use of plant-derived products, may have the capability to reduce some of these toxicities (27). Plants and other natural products have for a long time been the main source of anti-tumor drug candidates. Many of the anticancer drugs used today, such as vinblastine, vincristine, Paclitaxel, and camptothecin, are based mainly on natural drugs (14,28).
ROS are unstable species that pair up their odd free electrons by attacking healthy cells, causing a loss of cell structure and/or function (29). The impaired cells are key contributors to degenerative diseases such as cancer, inflammation, immune system weakening, liver disease, brain dysfunction, cardiovascular conditions, diabetes, and renal failure (30). Therefore, antioxidants/free radical-scavenging agents are vital to controlling the damaging effect of free radicals in the human body (31). As a result, verifying the type of correlation between free radical-scavenging of plant extracts and their cytotoxicity to cancer cells is an issue of great importance. The purpose of this study is to evaluate the free radical-scavenging of dozens of plant-based extracts, as well as their cytotoxicity to the HepG2 cell line (liver cancer). To the best of our knowledge to date, the correlation between free radical-scavenging and cytotoxicity has not yet been tested on a large scale nor reported in scientific journal. To explore and possibly verify this correlation, we tested fifty-seven methanolic extracts derived from regional plants that see heavy use as food and as traditional medicines.
Finding the type of correlation between free radical-scavenging of plant extracts and their cytotoxicity could be helpful in high-throughput screening projects that search for cytotoxic natural products. If a positive correlation exists, then, it may not imply that a change in the value of one parameter will cause a change in the value of the other parameter.
Materials and methods
Materials and cells
All plants that were used in this study were purchased from Al-Alim Ltd. (Medicinal Herb Center) or from the local market (those that are labeled with a symbol a in Table I). All our research involving wild-type species are not at risk of extinction and not registered in the endangered species flora list. The gallic acid, DPPH, and the solvents were purchased from Sigma. HepG2 liver cancer cell line was purchased from the American Tissue Culture Collection (ATCC; catalog no. HB-8065; passage 05–10). Eagle's minimum essential medium (EMEM), fetal bovine serum, antibiotics, and the XTT kit were purchased from Biological Industries.
Table I.
A list of medicinal and edible plants that were used in the current study and their yield of methanolic extraction, EC50 for free radical scavenging, percentage of inhibition at a concentration of 250 µg/ml of plant extract and 4-EC50 cytotoxicity for the most active plant.
| Scientific name (part of the plant) | The extract yield (%) | EC50 of free radical scavenging (µg/ml) | % Inhibition at concentrations of 250 µg/ml of (%) | EC50 of cytotoxicity (µg/ml) |
|---|---|---|---|---|
| Vitis vinifera (leaf) | 6.32 | 4.63 | 13 | |
| Stevia rabaudiana (leaf) | 19.38 | 9.77 | 12 | |
| Rosmarinus officinalis (leaf) | 11.72 | 3.45 | 95 | 131 |
| Rubus idaeus (leaf) | 4.82 | 4.25 | 22 | |
| Punuca granatum (fruit peel) | 38.02 | 87.30 | 49 | |
| Origanum vulgare (leaf) | 10.00 | 1.67 | 90 | 180 |
| Vitex agnus-castus (seeds) | 3.60 | 166.43 | 99 | 42 |
| Thymus vulgaris (leaf) | 11.63 | 1.98 | 33 | |
| Mentha piperita (leaf) | 10.97 | 1.68 | 30 | |
| Melissa officinalis (leaf) | 9.70 | 0.28 | 36 | |
| Urtica urens/pilulifera (leaf) | 7.33 | 220.92 | 0 | |
| Orea europaea (leaf) | 25.20 | 2.98 | 30 | |
| Camelia sinensis (leaf) | 11.26 | 54.2 | 33 | |
| Cynara cardunculus (leaf) | 20.15 | 17.61 | 95 | 152 |
| Foeniculurn vulgare (seeds) | 4.06 | 26.74 | 0 | |
| Petroselinum crispum (leaf) | 18.78 | 282.70 | 0 | |
| Pelargonium spp (leaf) | 11.30 | 2.83 | 53 | |
| Lippia citriodora (leaf) | 8.33 | 4.14 | 92 | 197 |
| Ocimum basilicum (leaf) | 11.22 | 8.73 | 3 | |
| Sumac (ripe fruit) | 30.44 | <0.5 | 66 | |
| Zingiber officinale (root) | 4.32 | 81.0 | 95 | 109 |
| Cinnamomum aromaticum (bark) | 4.26 | 1.67 | 100 | 162 |
| Cuminum cyminum (seeds) | 10.14 | 23.90 | 10 | |
| Portulaca oleracea (leaf and stem) | 10.08 | 47.33 | 14 | |
| Centaurea (leaf and stem) | 12.10 | 249.97 | 18 | |
| Scolymus maculatus (leaf and stem) | 3.82 | 259.03 | 0 | |
| Cichorium intybus (leaf) | 15.08 | 83.96 | 0 | |
| Malvaa (leaf) | 14.77 | <0.5 | 0 | |
| Allium cepa (leaf) | 8.15 | 215.20 | 0 | |
| Corchorus olitorius (leaf) | 11.23 | 10.64 | 0 | |
| Gundelia tournefortiia (stem) | 10.44 | 140.79 | 0 | |
| Hyssopus (leaf) | 3.60 | 13.10 | 0 | |
| Green tea (leaf) | 14.73 | 0.38 | 0 | |
| Petroselinum (leaf and stem) | 6.96 | 35.97 | 0 | |
| Thymus capitatus (leaf) | 9.28 | 2.12 | 37 | |
| Foeniculurn vulgare (leaf and stem) | 4.76 | 20.86 | 33 | |
| Melissa officinalis (leaf) | 8.80 | 24.0 | 0 | |
| Petroselium (leaf) | 17.55 | 381.20 | 4 | |
| Laurus nobilis (leaf) | 12.74 | 8.92 | 93 | 182 |
| Salvia officinalis (leaf) | 9.22 | 267.92 | 90 | 142 |
| Cymbopogon citratus (leaf) | 11.54 | 9.80 | 24 | |
| Linum usitatissimum (seeds) | 0.40 | 4,523.5 | 0 | |
| Avena sativa (seeds) | 22.00 | 635.51 | 1 | |
| Ceratonia siliqua (ripe fruit) | 25.06 | 130.9 | 12 | |
| Origanum syriacum (leaf) | 13.53 | 1.84 | 15 | |
| Camomile (leaf and flowers) | 16.25 | 29.00 | 7 | |
| Salvia hispanica (seeds) | 0.50 | 3,736.5 | 4 | |
| Crocusa (seeds) | 0.96 | 765.2 | 31 | |
| Vitex agnus-castusa (stem + leaf) | 12.00 | 266.51 | 0 | |
| Marrubium vulgarea (leaf) | 2.56 | 51.3 | 0 | |
| Ficus religiosaa (stem) | 4.22 | 215.92 | 23 | |
| Lepidium sativuma (seeds) | 0.84 | 2,529.8 | 0 | |
| Angelica sylvestrisa (leaf) | 10.10 | 2.00 | 3 | |
| Gentianaa (leaf) | 30.76 | 396.03 | 0 | |
| Pelargonium sp.a (stem) | 3.88 | 146.5 | 3 | |
| Eryngiuma (stem) | 4.32 | 253.31 | 0 | |
| Humulus lupupusa (leaf) | 12.36 | 4.22 | 3 |
Plants purchased at the local market.
Extraction of plants
To perform the extraction, one gram of dried plant material was packed in a tube, soaked with 10 milliliters of methanol, sonicated for 75 min at 40°C, and then left in for 3 h to cool down. After complete extraction, the methanolic extract solution was filtered with Whatman paper, grade 1, dried under vacuum, weighed, and then dissolved by DMSO at a concentration of 100 mg/ml. The extract was kept at 4°C until it was used.
Free radical scavenging (FRS)
The FRS of the methanolic extracts of the various plants was measured by microdilution using the DPPH assay protocol, with slight modifications (32,33). The microdilution of DPPH was performed using two-fold serial dilution in ddH2O. The tests were carried out in 96-well, flat-bottomed plates. 100 µl of ethanolic DPPH solution (200 ppm) was added to 100 µl of the plant extract at the concentration stated in Table I. The mixture was then shaken and allowed to settle for 30 min in the dark at room temperature. The absorbance of the solution was measured at 517 nm and converted into a percentage of FRS using the following equation:
FRS%=100*{1-[(Asample-Ablank_1))/(Acontrol-Ablank_2)]}
where
Asample is the absorbance of the mixture (of plant extract and DPPH)
Ablank-1 is the absorbance of the plant extract
Acontrol is the absorbance of the ethanolic solution of DPPH, and Ablank-2 is the absorbance of ethanol.
Gallic acid at a concentration of 100 µg/ml was used as a positive control. FRS was expressed in terms of the EC50 (the amount of antioxidant necessary to decrease the initial DPPH absorbance by 50%). The EC50 value for each extract was determined by extracting the value from the equation for the linear part of the graph. We substituted 50% for the y value, while calculating the concentration value of the x-axis.
Cell culture and cytotoxicity assay
HepG2 cells were cultured in EMEM medium supplemented with 10% fetal bovine serum and 100 U/ml of penicillin streptomycin (Biological Industries). The cells were cultured at 37°C in an incubator with 5% CO2. The cytotoxic effect of the extracts on the cells was assessed using a cell proliferation kit (XTT-based). In short, 2.5×103 HepG2 cells were seeded into each well of a 96-well plate and cultured for 24 h. The cells were treated with various concentrations of plant extract (0, 10, 20, 30, 40, 50, 100, 200, 300, 400 and 500 µg/ml) for 48 h and then incubated with XTT reagents for 3 h at 37°C, and absorbance was measured at 450 nm. The mean absorbance of non-treated cells served as the reference value for calculating the percentage of cellular viability. The assay was carried out in triplicate. Culture medium without cells was used as a background control (blank) and was subtracted from the other measurements.
Model assessments
Parameters such as the Matthews correlation coefficient (MCC), accuracy, the precision enrichment factor, and the area under the ROC curve (AUC) were used to assess the quality of the cytotoxicity/free radical-scavenging correlation models.
Equation 1. Matthews correlation coefficient (MCC).
where
P, N, Pf and Nf are the numbers of true positive, true negative, false positive, and false negative predictions, respectively. A perfect prediction gives MCC=1.0, while a random performance gives MCC=0.0. MCC=−1.0 indicates a completely erroneous prediction.
Equation 2. Accuracy.
Accuracy=(P + N)/(P + N + Pf + Nf)
Equation 3. Precision.
Precision=P/(P + Pf)
Equation 4. Enrichment factor.
EF=TFRS/TRS
where
TFRS is the % of actives when using the FRS threshold criterion, and TRS is the % of actives by random selection.
Statistical analysis
All statistical analyses were conducted using Excel spreadsheet software (v16.0; Microsoft). The quality of correlation between any two parameters was evaluated using a regression analysis based on the value of the coefficient of determination (R2). Reliability decreases with a decrease in the R2 value (R2=1 means completely reliable, while R2=0 means completely unreliable).
Results and Discussion
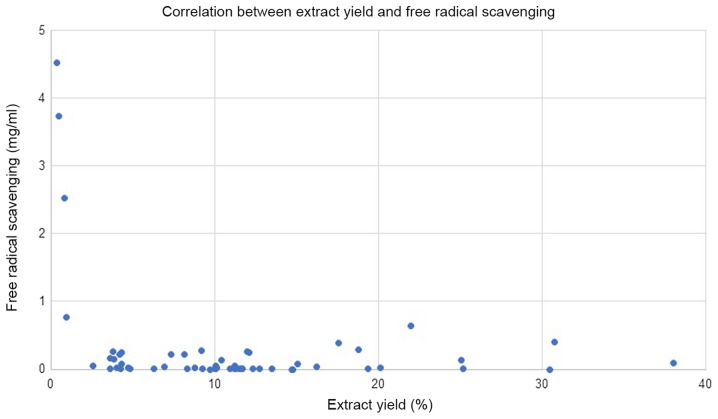
Fifty-seven edible medicinal plants were studied by measuring their free radical-scavenging (by DPPH assay) and their cytotoxic activity (by XTT assay). All results are summarized in Table I. The average yield of extraction by methanol was 11.2%, and as shown in Fig. 1, no correlation was detected between the % of extraction yield and free radical scavenging. However, the four plants that gave a % of yield of <1% (Linum usitatissimum, Salvia hispanica, Lepidium sativum and Crocus) possess the lowest free radical-scavenging activity (EC50 values, of 4,523, 3,736, 2,529 and 765 µg/ml, respectively). A review of column 3 of Table I reveals that Malva, sumac, Melissa officinalis, and green tea have the highest content of antioxidants, with EC50 values of free radical-scavenging of <0.5 µg/ml. Cytotoxicity was first verified by screening all extracts for their activity, using one concentration of 250 µg/ml; column 4 depicts the % of inhibition at this concentration. The extracts that gave a % of inhibition above 90% were tested in the second round at lower concentrations in dose response manner to extract their EC50 values. The results are summarized in column 5. Nine extracts were found to have EC50 values of <250 µg/ml (Rosmarinus officinalis, Origanum vulgare, Vitex agnus-castus, Cynara cardunculus, Lippia citriodora, Zingiber officinale, Cinnamomum aromaticum, Laurus nobilis, and Salvia officinalis). Their EC50 values are 131, 180, 42, 152, 197, 109, 162, 182 and 142 µg/ml, respectively. Three other extracts [Punuca granatum (fruit peel), Pelargonium spp (leaf), Sumac (ripe fruit)] have EC50 values close to 250 µg/ml, where treatment with 250 µg/ml inhibit viability of liver cancer cells by 49, 53 and 66%, respectively.
Figure 1.
Correlation between % of yield that was obtained by extraction with methanol and free radical scavenging.
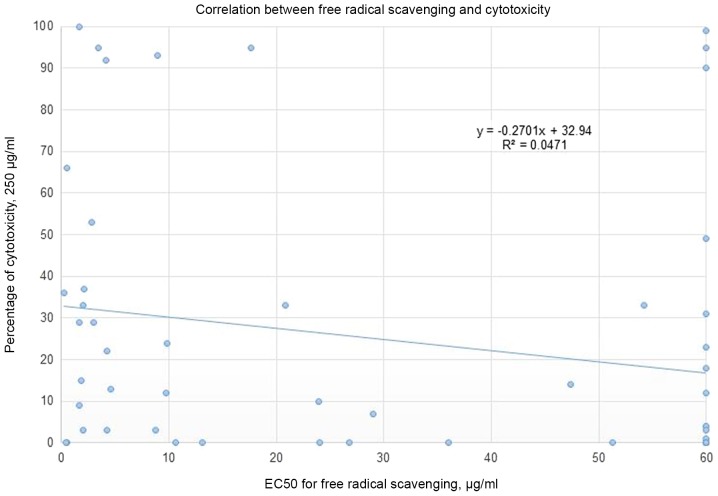
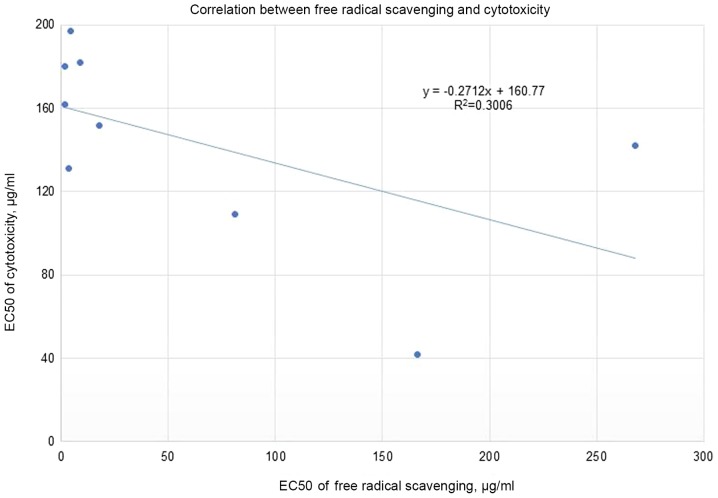
Rules-based analysis using Matthew's correlation coefficient (MCC) scores and enrichment factors as criteria for the evaluation of the models' efficiency revealed that the plant extracts whose EC50 for free radical scavenging ≤10 µg/ml showed some degree of enrichment toward more cytotoxicity (Table II). The values for the enrichment factor, the MCC, accuracy, and precision are 2.6, 0.28, 0.67 and 0.5, respectively. No correlation was detected between the % of cytotoxicity (using a concentration of 250 µg/ml of plant extract) and the EC50 for free radical scavenging (Fig. 2). Moreover, the correlation between the EC50 for cytotoxicity and the EC50 for free radical scavenging for the nine most cytotoxic plant extracts (Fig. 3) tends slightly toward the negative. The most cytotoxic plant extract (Vitex agnus-castus) showed, by several orders of magnitude, free radical-scavenging less than seven other extracts (Rosmarinus officinalis, Origanum vulgare, Cynara cardunculus, Lippia citriodora, Zingiber officinale, Cinnamomum aromaticum, and Laurus nobilis) out of the nine most cytotoxic plants. The obtained results show that differences in cytotoxic activities among the extracts are not mainly accredited to the level of antioxidants but could also be associated with the inhibitory effects via other signaling pathways.
Table II.
MCC scores and enrichment factors were utilized as criteria for evaluating the models. All calculations are based on the assumption that a % of cytotoxicity ≥30%, at a concentration of 250 µg/ml of plant extract, is considered active (a true positive); otherwise, it is considered inactive. One third of the tested plants (nineteen extracts) showed activity ≥30% cytotoxicity.
| EC50 cutoff of FRS (≤) | ||||
|---|---|---|---|---|
| Criteria | 10 µg/ml | 50 µg/ml | 250 µg/ml | No limit |
| No. active plants (true positives)a | 11 | 13 | 17 | 19 |
| No. inactive plants (false positives)b | 11 | 19 | 28 | 38 |
| No. inactive plants (true negatives)c | 27 | 19 | 10 | – |
| No. active plants (false negatives)d | 8 | 6 | 2 | – |
| Precision | 0.5 | 0.41 | 0.38 | 0.34 |
| Accuracy | 0.67 | 0.56 | 0.47 | 0.34 |
| Enrichment factor | 1.5 | 1.22 | 1.13 | 1.0 |
| MCC | 0.280 | 0.175 | 0.183 | 0.0 |
Number of plant extracts that have an EC50 of FRS less than the indicated threshold and ≥30% cytotoxicity against HepG2 cancerous cells at a concentration of 250 µg/ml plant extract
Number of plant extracts that have an EC50 of FRS less than the indicated threshold and <30% cytotoxicity against HepG2 cancerous cells at a concentration of 250 µg/ml plant extract
Number of plant extracts that have an EC50 of FRS greater than the indicated threshold and <30% cytotoxicity against HepG2 cancerous cells at a concentration of 250 µg/ml plant extract
Number of plant extracts that have an EC50 of FRS greater than the indicated threshold and ≥30% cytotoxicity against HepG2 cancerous cells at a concentration of 250 µg/ml plant extract. MCC, Matthew's correlation coefficient.
Figure 2.
Correlation between the % of cytotoxicity at a concentration of 250 µg/ml of plant extract and an EC50 for free radical scavenging expressed in units of µg/ml. The EC50 of free radical scavenging for each plant extract, which is >60 µg/ml, was set at 60 µg/ml.
Figure 3.
Correlation between the EC50 for cytotoxicity and the EC50 for free radical scavenging for the nine most cytotoxic plant extracts.
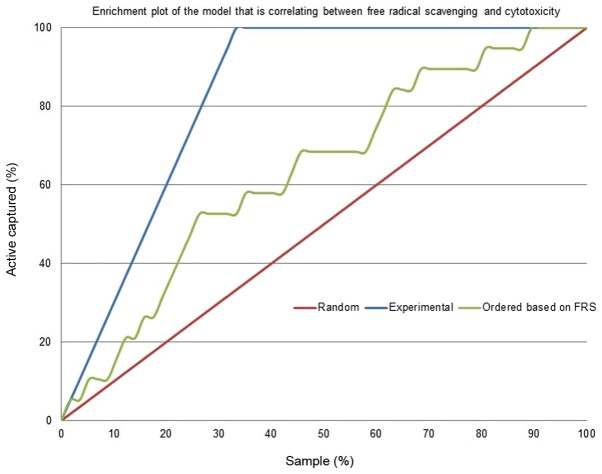
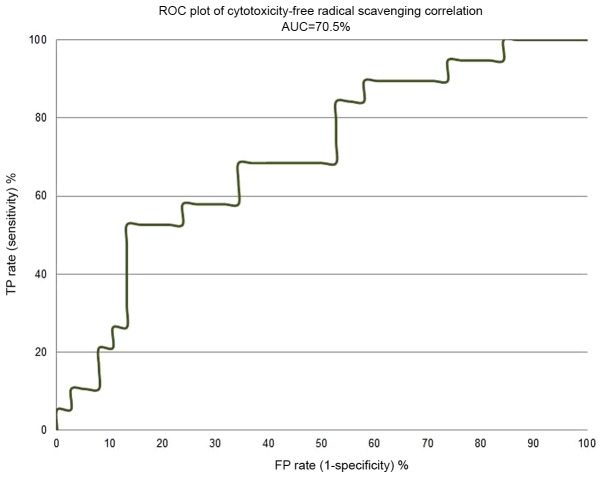
Figs. 4 and 5 depict the enrichment plot and the receiver operating characteristic (ROC) plot for the cytotoxicity/free radical-scavenging correlation model. It is worth noting that a fully random model should yield an AUC value around 0.5. The area under the curve (AUC) that was attained for the current model, as shown in Fig. 5, is 0.705, which means that the model is very poor, indicating a very weak correlation between cytotoxicity and free radical scavenging. The enrichment plot that is shown in Fig. 4 illustrates how quickly cytotoxic extracts of plants can be identified when they are sorted according to their free radical scavenging activity. If the enrichment plot of the proposed model is close to the perfect model, it indicates high prioritization power. A close look reveals that the shape of the figure fits well with the conclusions drawn from the detailed analysis of Table II, which disclosed that plant extracts with an EC50 of free radical scavenging ≤10 µg/ml display some degree of enrichment toward more cytotoxicity. At this point, the model line is closer to the experimental line than to the random line.
Figure 4.
Enrichment plot of the prediction model for the cytotoxicity of the plant extracts, based on their free radical-scavenging activity. FRS, free radical scavenging.
Figure 5.
A receiver operating characteristic curve showing the performance of the cytotoxicity/free radical-scavenging correlation model. TP, true positive; FP false positive.
In mid-October 2018, the electronic database PubMed was searched using the scientific names of the demonstrably cytotoxic plants disclosed herein and the keyword anticancer. Eight out of the nine cytotoxic plant extracts were reported to be active against cancer cell lines. As well, half of the eight were reported as active against HepG2, while the rest are reported here for the first time as showing activity against HepG2. Rosmarinus officinalis (34,35) and its components, the phenolic compound rosmarinic acid (36) and the abietane diterpenoid sageone (37), were reported to show anticancer properties, but had not been tested on HepG2. Origanum vulgare (38) and its main constituents (carvacrol, thymol, citral, and limonene) have been tested on HepG2 cell line (liver cancer) and were reported as active against cancer. Cynara cardunculus L. has evidenced anticancer potential (39) on triple-negative breast cancer (TNBC). It highlights the antiproliferative effects of lipophilic extracts from the leaves and florets of C. cardunculus L., and of their major constituents, namely cynaropicrin and taraxasteryl acetate, against MDA-MB-231 cells. A review article by Bahramsoltani (40) reported anticancer effects for Lippia citriodora against human colon cancer (HT29) cells; its extract enhances BAX (a pro-apoptotic gene) and reduces the expression level of Bcl-2 (an anti-apoptotic gene). Zingiber officinale extract significantly inhibited the proliferation of HepG2 cells and induced apoptosis (41). Cinnamomum cassia (syn. Cinnamomum aromaticum) extracts were reported to have anticancer activity (42). Laurus nobilis was reported as active against cancer cell lines (43) such as HeLa cells, but its activity on HepG2 had not been tested. Salvia officinalis extracts were confirmed to have cytotoxic effects on HepG2 cells (44). Recently, Kikuchi et al (45). demonstrated that an extract from the ripe fruit of Vitex angus-castus (Vitex), might be a promising anticancer candidate. It was the only scientific report to mention its cytotoxicity, which was tested by its effects on HL-60 cells, but not on HepG2; no phytochemicals were identified as the source of its cytotoxicity. We are currently working on isolating and identifying its bioactive chemical ingredients.
Since reactive oxygen species (ROS) are known to be triggers of various human cancers, and antioxidants or scavengers are used to counteract these dangerous species, we have raised a question regarding the correlation between free radical scavenging and the cytotoxicity of plant extracts. Free radical scavenging was assessed by DPPH assay, while cytotoxicity was measured by XTT assay. Nine extracts were found to be cytotoxic with EC50 values of <250 µg/ml, and four others had a high content of antioxidants, with EC50 values of free radical scavenging of <0.5 µg/ml. Upon looking on the results which were obtained from screening fifty-seven plants for their cytotoxic activity, we concluded, from first inspection, that there is no correlation between free radical scavenging and cytotoxicity. However, an in-depth analysis of the results reveals that the extracts of plants that had an EC50 for free radical scavenging ≤10 µg/ml exhibited a certain enrichment toward more cytotoxicity (enrichment factor of 1.5). We suggest checking further the validity of the conclusions that are drawn from the current study on other cancer cell-lines, and also by utilizing aqueous or other organic solvents to perform the extraction. The nine active extracts of plants disclosed here could be a source of anticancer hits/lead phytochemicals, worth the effort it would take to isolate and chemically identify them.
Acknowledgements
Not applicable.
Funding
This work was partially supported by the Al-Qasemi Research Foundation and the Ministry of Science, Space and Technology, Israel.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
AR conceived the study, designed the experiments, and contributed to writing and editing the manuscript. MF interpreted the data and wrote the original manuscript. BAF and IR collected the plants, performed their extraction and the run free radical-scavenging experiments. MS designed and performed the cytotoxicity experiments, interpreted the data and wrote the original draft.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Aswad M, Rayan M, Abu-Lafi S, Falah M, Raiyn J, Abdallah Z, Rayan A. Nature is the best source of anti-inflammatory drugs: Indexing natural products for their anti-inflammatory bioactivity. Inflamm Res. 2018;67:67–75. doi: 10.1007/s00011-017-1096-5. [DOI] [PubMed] [Google Scholar]
- 2.Frank A, Abu-Lafi S, Adawi A, Schwed JS, Stark H, Rayan A. From medicinal plant extracts to defined chemical compounds targeting the histamine H4 receptor: Curcuma longa in the treatment of inflammation. Inflamm Res. 2017;66:923–929. doi: 10.1007/s00011-017-1075-x. [DOI] [PubMed] [Google Scholar]
- 3.Zaid H, Raiyn J, Osman M, Falah M, Srouji S, Rayan A. In silico modeling techniques for predicting the tertiary structure of human H4 receptor. Front Biosci (Landmark Ed) 2016;21:597–619. doi: 10.2741/4409. [DOI] [PubMed] [Google Scholar]
- 4.Zeidan M, Rayan M, Zeidan N, Falah M, Rayan A. Indexing natural products for their potential anti-diabetic activity: Filtering and mapping discriminative physicochemical properties. Molecules. 2017;22:E1563. doi: 10.3390/molecules22091563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sui YB, Liu L, Tian QY, Deng XW, Zhang YQ, Li ZG. A retrospective study of traditional Chinese medicine as an adjunctive therapy for patients with chronic heart failure. Medicine (Baltimore) 2018;97:e11696. doi: 10.1097/MD.0000000000011696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maiti P, Dunbar GL. Use of curcumin, a natural polyphenol for targeting molecular pathways in treating age-related neurodegenerative diseases. Int J Mol Sci. 2018;19:E1637. doi: 10.3390/ijms19061637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Masalha M, Rayan M, Adawi A, Abdallah Z, Rayan A. Capturing antibacterial natural products with in silico techniques. Mol Med Rep. 2018;18:763–770. doi: 10.3892/mmr.2018.9027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kacergius T, Abu-Lafi S, Kirkliauskiene A, Gabe V, Adawi A, Rayan M, Qutob M, Stukas R, Utkus A, Zeidan M, Rayan A. Inhibitory capacity of Rhus coriaria L. extract and its major component methyl gallate on Streptococcus mutans biofilm formation by optical profilometry: Potential applications for oral health. Mol Med Rep. 2017;16:949–956. doi: 10.3892/mmr.2017.6674. [DOI] [PubMed] [Google Scholar]
- 9.Vallejo MJ, Salazar L, Grijalva M. Oxidative stress modulation and ROS-mediated toxicity in cancer: A review on in vitro models for plant-derived compounds. Oxid Med Cell Longev. 2017;2017:4586068. doi: 10.1155/2017/4586068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chikara S, Nagaprashantha LD, Singhal J, Horne D, Awasthi S, Singhal SS. Oxidative stress and dietary phytochemicals: Role in cancer chemoprevention and treatment. Cancer Lett. 2018;413:122–134. doi: 10.1016/j.canlet.2017.11.002. [DOI] [PubMed] [Google Scholar]
- 11.Gavamukulya Y, Wamunyokoli F, El-Shemy HA. Annona muricata: Is the natural therapy to most disease conditions including cancer growing in our backyard? A systematic review of its research history and future prospects. Asian Pac J Trop Med. 2017;10:835–848. doi: 10.1016/j.apjtm.2017.08.009. [DOI] [PubMed] [Google Scholar]
- 12.Kapinova A, Stefanicka P, Kubatka P, Zubor P, Uramova S, Kello M, Mojzis J, Blahutova D, Qaradakhi T, Zulli A, et al. Are plant-based functional foods better choice against cancer than single phytochemicals? A critical review of current breast cancer research. Biomed Pharmacother. 2017;96:1465–1477. doi: 10.1016/j.biopha.2017.11.134. [DOI] [PubMed] [Google Scholar]
- 13.Rayan A, Raiyn J, Falah M. Nature is the best source of anticancer drugs: Indexing natural products for their anticancer bioactivity. PLoS One. 2017;12:e0187925. doi: 10.1371/journal.pone.0187925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tariq A, Sadia S, Pan K, Ullah I, Mussarat S, Sun F, Abiodun OO, Batbaatar A, Li Z, Song D, et al. A systematic review on ethnomedicines of anti-cancer plants. Phytother Res. 2017;31:202–264. doi: 10.1002/ptr.5751. [DOI] [PubMed] [Google Scholar]
- 15.Aggarwal BB, Shishodia S. Molecular targets of dietary agents for prevention and therapy of cancer. Biochem Pharmacol. 2006;71:1397–1421. doi: 10.1016/j.bcp.2006.02.009. [DOI] [PubMed] [Google Scholar]
- 16.Raza A, Sood GK. Hepatocellular carcinoma review: Current treatment, and evidence-based medicine. World J Gastroenterol. 2014;20:4115–4127. doi: 10.3748/wjg.v20.i15.4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ali H, Dixit S, Ali D, Alqahtani SM, Alkahtani S, Alarifi S. Isolation and evaluation of anticancer efficacy of stigmasterol in a mouse model of DMBA-induced skin carcinoma. Drug Des Devel Ther. 2015;9:2793–2800. doi: 10.2147/DDDT.S83514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ziech D, Franco R, Georgakilas AG, Georgakila S, Malamou-Mitsi V, Schoneveld O, Pappa A, Panayiotidis MI. The role of reactive oxygen species and oxidative stress in environmental carcinogenesis and biomarker development. Chem Biol Interact. 2010;188:334–339. doi: 10.1016/j.cbi.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 19.Galaris D, Skiada V, Barbouti A. Redox signaling and cancer: The role of ‘labile’ iron. Cancer Lett. 2008;266:21–29. doi: 10.1016/j.canlet.2008.02.038. [DOI] [PubMed] [Google Scholar]
- 20.Jambunathan S, Bangarusamy D, Padma PR, Sundaravadivelu S. Cytotoxic activity of the methanolic extract of leaves and rhizomes of Curcuma amada Roxb against breast cancer cell lines. Asian Pac J Trop Med. 2014;7S1:S405–S409. doi: 10.1016/S1995-7645(14)60266-2. [DOI] [PubMed] [Google Scholar]
- 21.Fuchs-Tarlovsky V. Role of antioxidants in cancer therapy. Nutrition. 2013;29:15–21. doi: 10.1016/j.nut.2012.02.014. [DOI] [PubMed] [Google Scholar]
- 22.Chua MT, Tung YT, Chang ST. Antioxidant activities of ethanolic extracts from the twigs of Cinnamomum osmophloeum. Bioresour Technol. 2008;99:1918–1925. doi: 10.1016/j.biortech.2007.03.020. [DOI] [PubMed] [Google Scholar]
- 23.Abu-Lafi S, Rayan B, Kadan S, Abu-Lafi M, Rayan A. Anticancer activity and phytochemical composition of wild Gundelia tournefortii. Oncol Lett. 2019;17:713–717. doi: 10.3892/ol.2018.9602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hwang YJ, Lee EJ, Kim HR, Hwang KA. In vitro antioxidant and anticancer effects of solvent fractions from Prunella vulgaris var. lilacina. BMC Complement Altern Med. 2013;13:310. doi: 10.1186/1472-6882-13-310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Radomska-Leśniewska DM, Hevelke A, Skopiński P, Bałan B, Jóźwiak J, Rokicki D, Skopińska-Różewska E, Białoszewska A. Reactive oxygen species and synthetic antioxidants as angiogenesis modulators: Clinical implications. Pharmacol Rep. 2016;68:462–471. doi: 10.1016/j.pharep.2015.10.002. [DOI] [PubMed] [Google Scholar]
- 26.Boots AW, Haenen GR, Bast A. Health effects of quercetin: From antioxidant to nutraceutical. Eur J Pharmacol. 2008;585:325–337. doi: 10.1016/j.ejphar.2008.03.008. [DOI] [PubMed] [Google Scholar]
- 27.Kadan S, Rayan M, Rayan A. Anticancer Activity of Anise (Pimpinella anisum L.) Seed Extract. Open Nutraceuticals J. 2013;6:1–5. doi: 10.2174/1876396001306010001. [DOI] [Google Scholar]
- 28.Zaid H, Rayan A, Said O, Saad B. Cancer treatment by Greco-Arab and Islamic herbal medicine. Open Nutraceuticals J. 2010;3:203–213. doi: 10.2174/18763960010030100203. [DOI] [Google Scholar]
- 29.Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009;7:65–74. doi: 10.2174/157015909787602823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Perrone S, Santacroce A, Longini M, Proietti F, Bazzini F, Buonocore G. The free radical diseases of prematurity: From cellular mechanisms to bedside. Oxid Med Cell Longev. 2018;2018:7483062. doi: 10.1155/2018/7483062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44–84. doi: 10.1016/j.biocel.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 32.Abu-Lafi S, Rayan M, Masalha M, Abu-Farich B, Al-Jaas H, Abu-Lafi M, Rayan A. Phytochemical composition and biological activities of wild Scolymus maculatus L. Medicines (Basel) 2019;6:E53. doi: 10.3390/medicines6020053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Masalha M, Abu-Lafi S, Abu-Farich B, Rayan M, Issa N, Zeidan M, Rayan A. A new approach for indexing honey for its heath/medicinal benefits: Visualization of the concept by indexing based on antioxidant and antibacterial activities. Medicines (Basel) 2018;5:E135. doi: 10.3390/medicines5040135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Moore J, Yousef M, Tsiani E. Anticancer effects of rosemary (Rosmarinus officinalis L.) extract and rosemary extract polyphenols. Nutrients. 2016;8:E731. doi: 10.3390/nu8110731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gonzalez-Vallinas M, Reglero G, Ramirez de Molina A. Rosemary (Rosmarinus officinalis L.) extract as a potential complementary agent in anticancer therapy. Nutr Cancer. 2015;67:1221–1229. doi: 10.1080/01635581.2015.1082110. [DOI] [PubMed] [Google Scholar]
- 36.Swamy MK, Sinniah UR, Ghasemzadeh A. Anticancer potential of rosmarinic acid and its improved production through biotechnological interventions and functional genomics. Appl Microbiol Biotechnol. 2018;102:7775–7793. doi: 10.1007/s00253-018-9223-y. [DOI] [PubMed] [Google Scholar]
- 37.Shrestha S, Song YW, Kim H, Lee DS, Cho SK. Sageone, a diterpene from Rosmarinus officinalis, synergizes with cisplatin cytotoxicity in SNU-1 human gastric cancer cells. Phytomedicine. 2016;23:1671–1679. doi: 10.1016/j.phymed.2016.09.008. [DOI] [PubMed] [Google Scholar]
- 38.Elshafie HS, Armentano MF, Carmosino M, Bufo SA, De Feo V, Camele I. Cytotoxic activity of Origanum Vulgare L. on hepatocellular carcinoma cell line HepG2 and evaluation of its biological activity. Molecules. 2017;22:E1435. doi: 10.3390/molecules22091435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ramos PA, Guerra AR, Guerreiro O, Santos SA, Oliveira H, Freire CS, Silvestre AJ, Duarte MF. Antiproliferative Effects of Cynara cardunculus L. var. altilis (DC) lipophilic extracts. Int J Mol Sci. 2016;18:E63. doi: 10.3390/ijms18010063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bahramsoltani R, Rostamiasrabadi P, Shahpiri Z, Marques AM, Rahimi R, Farzaei MH. Aloysia citrodora Palau (Lemon verbena): A review of phytochemistry and pharmacology. J Ethnopharmacol. 2018;222:34–51. doi: 10.1016/j.jep.2018.04.021. [DOI] [PubMed] [Google Scholar]
- 41.Elkady AI, Abu-Zinadah OA, Hussein R. Crude flavonoid extract of medicinal herb Zingibar officinale inhibits proliferation and induces apoptosis in hepatocellular carcinoma cells. Oncol Res. 2017;25:897–912. doi: 10.3727/096504016X14816352324532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lin CY, Hsieh YH, Yang SF, Chu SC, Chen PN, Hsieh YS. Cinnamomum cassia extracts reverses TGF-β1-induced epithelial-mesenchymal transition in human lung adenocarcinoma cells and suppresses tumor growth in vivo. Environ Toxicol. 2017;32:1878–1887. doi: 10.1002/tox.22410. [DOI] [PubMed] [Google Scholar]
- 43.Berrington D, Lall N. Anticancer activity of certain herbs and spices on the cervical epithelial carcinoma (HeLa) cell line. Evid Based Complement Alternat Med. 2012;2012:564927. doi: 10.1155/2012/564927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jiang Y, Zhang L, Rupasinghe HP. Antiproliferative effects of extracts from Salvia officinalis L. and Saliva miltiorrhiza Bunge on hepatocellular carcinoma cells. Biomed Pharmacother. 2017;85:57–67. doi: 10.1016/j.biopha.2016.11.113. [DOI] [PubMed] [Google Scholar]
- 45.Kikuchi H, Yuan B, Nishimura Y, Imai M, Furutani R, Kamoi S, Seno M, Fukushima S, Hazama S, Hirobe C, et al. Cytotoxicity of Vitex agnus-castus fruit extract and its major component, casticin, correlates with differentiation status in leukemia cell lines. Int J Oncol. 2013;43:1976–1984. doi: 10.3892/ijo.2013.2133. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.