Abstract
Caspase 7 (CASP7) is located on chromosome 10q25.3 that has been identified to be a susceptibility locus of ischaemic stroke (IS) by genome-wide association study. Elevated CASP7 was observed in IS, acting as a key apoptotic mediator in the development of IS. The aim of this study was to investigate the association between genetic polymorphisms in CASP7 and risk of IS. The CASP7 polymorphisms were genotyped using a TaqMan allelic discrimination assay. The expression levels of CASP7 mRNA were examined using quantitative polymerase chain reaction and luciferase activity was analyzed using the Dual Luciferase reporter assay. The rs12415607 in the promoter of CASP7 was associated with a reduced risk of IS (AA vs. CC: adjusted OR = 0.55, 95% CI: 0.38–0.80, P = 0.002; CA/AA vs. CC: adjusted OR = 0.70, 95% CI: 0.54–0.91, P = 0.007; AA vs. CC/CA: adjusted OR = 0.64, 95% CI: 0.46–0.90, P = 0.01; A vs. C: adjusted OR = 0.74, 95% CI: 0.62–0.89, P = 0.001). Moreover, the rs12415607 AA genotype carriers exhibited lower levels of CASP7 mRNA and the rs12415607 A allele decreased the promoter activity. These findings indicate that the rs12415607 A allele induces lower levels of transcriptional activity and CASP7 mRNA, and thus is associated with a reduced risk of IS.
Subject terms: Genetics of the nervous system, Risk factors
Introduction
Cerebral ischaemia may lead to stroke, which is a main reason for mortality and permanent disability in both developed and developing countries1,2. Among all, ischaemic stroke (IS) accounts for about 85–90% of the stroke cases3,4. Although the exact etiology is not full known, conventional risk factors have been identified, such as hypertension, diabetes mellitus, and dyslipidemia5–8. Additionally, previous genetic epidemiological studies provided substantial evidence that single nucleotide polymorphisms (SNPs) may be involved in the development of IS. For example, we previously reported that long non-coding RNA growth arrest-specific 5 (GAS5) rs145204276 ins/ins genotype and miR-181b rs322931 CT and CT/TT genotypes were associated with an increased risk of IS9,10.
Ischaemia preferentially triggers neuronal damage through an apoptotic-like process rather than necrosis11. Caspases, a family of cysteine aspartases, play a critical role in apoptotic cell death, including delayed neuronal death following IS11,12. It is evident that caspases are cleaved and activated in human brains and experimental models of stroke and neurodegenerative diseases13–15. When activated, executioner caspases (caspases 3 and 7) facilitate cell demise by targeting and degrading numerous substrate proteins11. Cell death can be attenuated by administering endogenous caspase inhibitors during and after brain ischaemic injury16–18. The neuroprotective role was also observed in caspase 3 deficient mice that were more resistant to ischaemic stress both in vivo and in vitro19. These findings suggest that drugs targeting caspase-independent programmed cell death may be an effective therapy for IS.
Since 2007, genome-wide association study (GWAS) has been used in exploring susceptibility genes of human diseases. To date, several risk loci of IS have been identified, such as chromosome 10q25, 12p13, 14q13.3, 1p13.2, 12q24.12, 10q11.21, 9p21, and 1p3220–27. Caspase 7 (CASP7), located on chromosome 10q25.3, has been reported to be upregulated in a rat model of focal cerebral ischemia28, acting as a key apoptotic mediator in the development of IS. Based on this background, we hypothesized that SNPs in CASP7 may affect the risk of IS. To test this hypothesis, we carried out a case-control study to evaluate the role of SNPs in CASP7 in the development of IS in a Chinese population. Genotype-phenotype and potential mechanism analysis was also explored.
Materials and Methods
Study population
The study protocol was approved by the Institutional Review Board of the China-Japan Union Hospital of Jilin University, and informed consent was obtained from all individual participants included in the study. The characteristics of the study population have been described previously9,10. Briefly, 505 patients with IS and 652 controls were consecutively obtained from the China-Japan Union Hospital of Jilin University between March 2014 and July 2017. IS diagnosis was confirmed based on clinical manifestations and computed tomography scans and/or magnetic resonance imaging. Patients were excluded if they had the following medical records: hemorrhagic stroke, subarachnoid hemorrhage, traumatic brain injury, malignancy, and brain inflammatory diseases. The controls were enrolled from the same hospital during the same time period if they were healthy Chinese Han subjects and did not report any family history of IS and thromboembolic diseases. The controls were frequency matched to cases based on age, gender, ethnicity, and living area. Clinical information was collected from medical records, including age, gender, hypertension, diabetes mellitus, total cholesterol (TCH), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C).
SNPs selection
We selected SNPs in CASP7 according to the following criteria: (1) TagSNPs; (2) Minor allele frequency more than 10% in Chinese Han population; and (3) Bioinformational analysis predicted to be functional. Finally, 5 SNPs were identified, including a nonsynonymous polymorphism in exon 8 of CASP7 (rs2227310), a polymorphism in the promoter region of CASP7 (rs12415607) with the C but not the A allele creating a binding site to the transcriptional factor TFII-I, and 3 polymorphisms in the 3′-untranslated region (3′-UTR) of CASP7 with different allele having different affinity to miRNAs. Detailed information of the 3 polymorphisms in the 3′-UTR of CASP7 is presented in Supplementary Table 1.
DNA extraction and genotyping
Genomic DNA was extracted from whole blood using a commercial kit from Tiangen company (Beijing, China). Genotyping was performed using a TaqMan allelic discrimination assay (Applied Biosystems, Foster City, CA, USA). Polymerase chain reaction (PCR) was run on the ABI 7500 real-time PCR System (Applied Biosystems) using the following probes for the 5 polymorphisms: C__27432681_20, C__500779_20, C__500776_10, C__12119563_10, and C__500777_10, respectively. For quality control, negative control in each run was performed using distilled water as template and accuracy of genotyping was confirmed by DNA sequencing.
RNA isolation and quantitative real-time PCR (qPCR)
Total cellular RNA was extracted from peripheral blood cells using the RNAprep pure Blood Kit (Tiangen, Beijing, China) according to the manufacturer’s instructions. A total of 1 μg RNA was reverse-transcribed using the First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. Relative expression levels of CASP7 were determined by qPCR using the following primers: CASP7 sense primer: CGTTTGTACCGTCCCTCTTC and antisense primer: GCCCAGCTTTTCAAAATTCA; GAPDH sense primer: CTCTCTGCTCCTCCTGTTCG AC and antisense primer: TGAGCGATGTGGCTCGGCT. Each 10 μL qPCR reaction contained 5 μL of 2 X SYBR Green master mix (Thermo Fisher Scientific), 10 μM each primer and 1 μL of cDNA. PCR conditions were set as follows: 95 °C for 2 min, 40 cycles of 95 °C for 15 sec and 60 °C for 1 min. The relative expression levels of CASP7 mRNA were calculated using the 2−ΔΔCt method29.
Plasmid constructs
Genomic DNA from human CASP7 promoter region was amplified by PCR using primers: 5′-TTCTCGAGAAAAGACTAGGGCAGCCACA-3′ (forward) and 5′-CAAGCTTGCCCCTCGCTCTACAAAGTT-3′ (reverse). For constructing luciferase reporter plasmids, oligonucleotides containing the rs12415607 A and C allele were cloned into pGL3-basic vector (Promega, Madison, WI, USA) after digestion with Xho I and Hind III. All PCR products were verified by DNA sequencing.
Cell culture and dual luciferase reporter assay
Human embryonic kidney (HEK293) cells were cultured in Dulbecco’s modified Eagle medium supplemented with 10% FBS (HyClone, Logan, UT, USA) and 1% penicillin-streptomycin (Life Technologies, Grand Island, NY, USA). pGL3-rs12415607A, pGL3-rs12415607C, and pGL3 empty vector (1 μg) were introduced into cells per well in a 12-well plate using Lipofectamine 3000 (Thermo Fisher Scientific) according to the manufacturer’s protocol. Renilla luciferase pRL vector (20 ng) was cotransfected as an internal control. Luciferase activities were checked at 48 h after transfection using the dual luciferase reporter assay system (Promega) and relative luciferase activities were measured using the ratio of firefly luciferase activity to renilla luciferase activity.
Statistical analysis
Continuous data were reported as mean ± standard deviation and compared using Student’s t test. Dichotomous data were reported as frequencies (percentages) and compared using χ2 test. Hardy-Weinberg equilibrium (HWE) and the association of the 5 SNPs with IS risk were evaluated using χ2 test. Odds ratios (ORs) and 95% confidence intervals (CIs) were computed after adjustment for age, gender, hypertension, and diabetes mellitus. Bonferroni corrected test was used for multiple comparisons and the corrected P value was set as 0.01 (0.05/5). Binary logistic regression was used to identify independent risk factors for IS. Haplotype analysis was performed using a SHEsis software (http://analysis.bio-x.cn/myAnalysis.php)30. Relative expression levels of CASP7 mRNA were reported as median with interquartile range and compared using Whitney U test. Statistics were performed using the SPSS software version 19.0 (SPSS, Chicago, IL, USA) and a two-sided P < 0.05 was considered significant.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of China-Japan Union Hospital of Jilin University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Results
Characteristics of study population
Table 1 shows the characteristics of the study population. The mean age of the patients was 59.9 ± 10.9 years, which was similar to that of the controls (59.0 ± 11.9 years; P = 0.16 for t-test). Of the 505 patients, 325 (64.4%) were males vs. 61.2% of controls (P = 0.27 for χ2 test). The percentage of hypertension and diabetes mellitus in IS patients was significantly higher than that in controls (P < 0.001 and = 0.01, respectively). The serum levels of TCH, TG, and LDL-C in IS patients were higher than controls (P < 0.001), while there was no difference of HDL-C levels between cases and controls (P = 0.43).
Table 1.
Characteristics of the study population.
| Variables | Controls, n = 652 | Patients with IS, n = 505 | P value |
|---|---|---|---|
| Age, mean (±SD) | 59.0 (±11.9) | 59.9 (±10.9) | 0.16 |
| Gender (%) | |||
| Male | 399 (61.2) | 325 (64.4) | 0.27 |
| Female | 253 (38.8) | 180 (35.6) | |
| Hypertension, n (%) | |||
| Yes | 132 (20.2) | 277 (54.9) | <0.001 |
| No | 520 (79.8) | 228 (45.1) | |
| Diabetes mellitus, n (%) | |||
| Yes | 70 (10.7) | 79 (15.6) | 0.01 |
| No | 582 (89.3) | 426 (84.4) | |
| TCH, mmol/L | 4.69 ± 0.79 | 5.04 ± 0.72 | <0.001 |
| TG, mmol/L | 1.11 ± 0.36 | 1.85 ± 1.10 | <0.001 |
| HDL-C, mmol/L | 1.55 ± 0.36 | 1.57 ± 0.38 | 0.43 |
| LDL-C, mmol/L | 2.26 ± 0.97 | 2.68 ± 0.98 | <0.001 |
IS, ischemic stroke; SD, standard deviation; TCH, total cholesterol; TG: triglyceride; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
Association between CASP7 polymorphisms and risk of IS
The genotype distributions of the 5 SNPs were in HWE among controls (P > 0.05). The rs12415607 in the promoter of CASP7 was associated with a reduced risk of IS in homozygote comparison, dominant comparison, recessive comparison, and allele comparison (AA vs. CC: adjusted OR = 0.55, 95% CI: 0.38–0.80, P = 0.002; CA/AA vs. CC: adjusted OR = 0.70, 95% CI: 0.54–0.91, P = 0.007; AA vs. CC/CA: adjusted OR = 0.64, 95% CI: 0.46–0.90, P = 0.01; A vs. C: adjusted OR = 0.74, 95% CI: 0.62–0.89, P = 0.001). However, the rs2227310, rs10787498, rs1127687, and rs4353229 were not associated with the risk of IS (Table 2).
Table 2.
Association between CASP7 polymorphisms and risk of IS.
| Polymorphisms | Controls, n = 652 (%) | IS, n = 505 (%) | Adjusted OR (95% CI)† | P value |
|---|---|---|---|---|
| rs12415607 | ||||
| CC | 219 (33.6) | 215 (42.6) | 1.00 | |
| CA | 300 (46.0) | 223 (44.2) | 0.76 (0.58–1.00) | 0.05 |
| AA | 133 (20.4) | 67 (13.3) | 0.55 (0.38–0.80) | 0.002 |
| Dominant model | 433 (66.4) | 290 (57.4) | 0.70 (0.54–0.91) | 0.007 |
| Recessive model | 519 (79.6) | 438 (86.7) | 0.64 (0.46–0.90) | 0.01 |
| C allele | 738 (56.6) | 653 (64.7) | 1.00 | |
| A allele | 566 (43.4) | 357 (35.3) | 0.74 (0.62–0.89) | 0.001 |
| rs2227310 | ||||
| CC | 216 (33.1) | 191 (37.8) | 1.00 | |
| CG | 334 (51.2) | 243 (48.1) | 0.84 (0.64–1.10) | 0.21 |
| GG | 102 (15.6) | 71 (14.1) | 0.85 (0.58–1.24) | 0.39 |
| Dominant model | 436 (66.9) | 314 (62.2) | 0.84 (0.65–1.10) | 0.21 |
| Recessive model | 550 (84.4) | 434 (85.9) | 0.94 (0.66–1.33) | 0.71 |
| C allele | 766 (58.7) | 625 (61.9) | 1.00 | |
| G allele | 538 (41.3) | 385 (38.1) | 0.91 (0.76–1.09) | 0.29 |
| rs10787498 | ||||
| TT | 369 (56.6) | 298 (59.0) | 1.00 | |
| TG | 253 (38.8) | 189 (37.4) | 0.89 (0.69–1.16) | 0.39 |
| GG | 30 (4.6) | 18 (3.6) | 0.74 (0.39–1.41) | 0.35 |
| Dominant model | 283 (43.4) | 207 (41.0) | 0.88 (0.68–1.13) | 0.30 |
| Recessive model | 622 (95.4) | 487 (96.4) | 0.77 (0.41–1.47) | 0.43 |
| T allele | 991 (76.0) | 785 (77.7) | 1.00 | |
| G allele | 313 (24.0) | 225 (22.3) | 0.89 (0.72–1.09) | 0.26 |
| rs1127687 | ||||
| GG | 430 (66.0) | 325 (64.4) | 1.00 | |
| AG | 198 (30.4) | 165 (32.7) | 1.11 (0.84–1.45) | 0.46 |
| AA | 24 (3.7) | 15 (3.0) | 0.73 (0.35–1.50) | 0.39 |
| Dominant model | 222 (34.0) | 180 (35.6) | 1.07 (0.82–1.38) | 0.63 |
| Recessive model | 628 (96.3) | 490 (97.0) | 0.72 (0.35–1.46) | 0.36 |
| G allele | 1058 (81.1) | 815 (80.7) | 1.00 | |
| A allele | 246 (18.9) | 195 (19.3) | 1.01 (0.81–1.27) | 0.91 |
| rs4353229 | ||||
| TT | 205 (31.4) | 155 (30.7) | 1.00 | |
| CT | 320 (49.1) | 260 (51.5) | 1.11 (0.84–1.48) | 0.45 |
| CC | 127 (19.5) | 90 (17.8) | 0.92 (0.63–1.33) | 0.65 |
| Dominant model | 447 (68.6) | 350 (69.3) | 1.07 (0.81–1.40) | 0.64 |
| Recessive model | 525 (80.5) | 415 (82.2) | 0.87 (0.63–1.20) | 0.39 |
| T allele | 730 (56.0) | 570 (56.4) | 1.00 | |
| C allele | 574 (44.0) | 440 (43.6) | 0.99 (0.82–1.18) | 0.87 |
CASP7, caspase 7; IS, ischemic stroke; OR, odds ratio; CI, confidence interval.
†Adjusted by age, gender, hypertension, and diabetes mellitus.
Haplotype analysis
Nine common haplotypes are summarized in Table 3. Compared to the CCTGT haplotype, the CCGAT and AGGAT haplotypes were associated with a reduced risk of IS (CCGAT vs. CCTGT: OR = 0.40, 95% CI: 0.24–0.67, P = 3.34E-4; AGGAT vs. CCTGT: OR = 0.38, 95% CI: 0.20–0.73, P = 0.003, respectively), whereas the CCGGC, CCGGT, and AGGGT haplotypes were associated with an increased risk of IS (CCGGC vs. CCTGT: OR = 5.86, 95% CI: 3.39–10.12, P = 4.74E-12; CCGGT vs. CCTGT: OR = 5.66, 95% CI: 3.18–10.09, P = 1.43E-10; AGGGT vs. CCTGT: OR = 2.19, 95% CI: 1.23–3.92, P = 0.007, respectively).
Table 3.
Haplotype analysis of CASP7 polymorphisms with risk of IS.
| Haplotypes† | Controls (%) | IS (%) | OR (95% CI) | P value |
|---|---|---|---|---|
| CCTGT | 309 (23.7) | 208 (20.6) | 1.00 | |
| CCTGC | 260 (19.9) | 170 (16.8) | 0.97 (0.75–1.26) | 0.83 |
| AGTGT | 233 (17.9) | 134 (13.3) | 0.85 (0.65–1.13) | 0.26 |
| AGTGC | 166 (12.7) | 94 (9.3) | 0.84 (0.62–1.15) | 0.27 |
| CCGAT | 78 (6.0) | 21 (2.1) | 0.40 (0.24–0.67) | 3.34E-4 |
| CCGGC | 18 (1.4) | 71 (7.0) | 5.86 (3.39–10.12) | 4.74E-12 |
| CCGGT | 16 (1.2) | 61 (6.0) | 5.66 (3.18–10.09) | 1.43E-10 |
| AGGAT | 47 (3.6) | 12 (1.2) | 0.38 (0.20–0.73) | 0.003 |
| AGGGT | 21 (1.6) | 31 (3.1) | 2.19 (1.23–3.92) | 0.007 |
CASP7, caspase 7; IS, ischemic stroke; OR, odds ratio; CI, confidence interval.
†Only the frequency more than 1% was presented.
Multivariate regression analysis
Multivariate logistic regression analysis was performed. As shown in Table 4, 6 independent risk factors of IS were identified, that is, hypertension (OR = 4.62, 95%CI: 3.39–6.30, P = 3.86E-22), TCH (OR = 1.24, 95%CI: 1.02–1.50, P = 0.03), TG (OR = 6.10, 95%CI: 4.55–8.20, P = 2.66E-33), LDL-C (OR = 1.95, 95%CI: 1.65–2.31, P = 8.00E-15), rs12415607 (OR = 13.67, 95%CI: 3.77–49.48, P = 6.81E-5), and rs2227310 (OR = 12.20, 95%CI: 3.34–43.48, P = 1.52E-4).
Table 4.
Logistic regression analysis for independent risk factors of IS.
| Variables | B | Walds | OR (95% CI) | P value |
|---|---|---|---|---|
| Hypertension | 1.53 | 93.60 | 4.62 (3.39–6.30) | 3.86E-22 |
| TCH | 0.21 | 4.53 | 1.24 (1.02–1.50) | 0.03 |
| TG | 1.81 | 144.57 | 6.10 (4.55–8.20) | 2.66E-33 |
| LDL-C | 0.67 | 60.33 | 1.95 (1.65–2.31) | 8.00E-15 |
| rs12415607 | 2.62 | 15.86 | 13.67 (3.77–49.48) | 6.81E-5 |
| rs2227310 | 2.50 | 14.34 | 12.20 (3.34–43.48) | 1.52E-4 |
IS, ischemic stroke; OR, odds ratio; CI, confidence interval; TCH, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol.
The rs12415607 AA genotype associated to lower levels of CASP7 mRNA
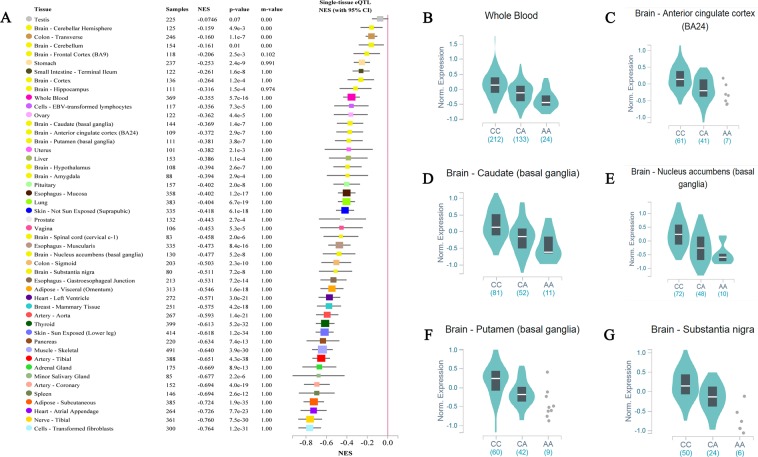
qPCR was used to examine the expression levels of CASP7 mRNA in IS patients and controls (n = 86). As shown in Fig. 1A, CASP7 mRNA levels were found to be highly elevated in IS patients compared to controls (P = 0.03). We compared CASP7 mRNA levels in individuals with different genotypes of the rs12415607, and we found that the rs12415607 AA carriers had lower levels of CASP7 mRNA both in controls (P = 0.02, Fig. 1B) and in patients with IS (P = 0.03, Fig. 1C). The results were confirmed by data from expression Quantitative Trait Loci (eQTL, https://www.gtexportal.org/home/) which shows lower levels of CASP7 mRNA in multiple human tissues except for testis (Fig. 2A). Figure 2B–G presents representative data in single tissue, including whole blood, anterior cingulate cortex, caudate (basal ganglia), nucleus accumbens (basal ganglia), putamen (basal ganglia), and substantia nigra.
Figure 1.
Relative expression of CASP7 mRNA in IS patients and controls. (A) The relative expression of CASP7 mRNA in IS patients and controls (n = 86); (B) The relative expression of CASP7 mRNA in control subjects carrying the rs12415607 CC, CA, and AA genotype; (C) The relative expression of CASP7 mRNA in IS patients carrying the rs12415607 CC, CA, and AA genotype. Aligned dot plot shows median with interquartile range (*P < 0.05).
Figure 2.
The lower levels of CASP7 mRNA in the rs12415607 AA carriers were confirmed by eQTL. (A) eQTL shows lower levels of CASP7 mRNA in multiple tissues except for testis; Representative data was presented in single tissue, such as whole blood (B), anterior cingulate cortex (C), caudate (basal ganglia) (D), nucleus accumbens (basal ganglia) (E), putamen (basal ganglia) (F), and substantia nigra (G).
The rs12415607 A allele reduced the promoter activity
Dual luciferase reporter assay was carried out to assess the effect of the rs12415607 A allele on the promoter activity. Figure 3A shows the schematic representation of CASP7 promoter containing the rs12415607 C/A into pGL3 vector. Compared to the rs12415607 C, the rs12415607 A exhibited a lower promoter activity (P = 0.004, Fig. 3B).
Figure 3.
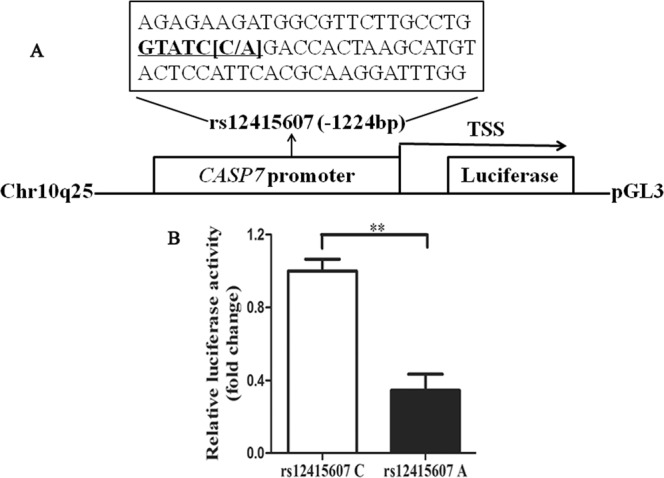
The rs12415607 A allele in the promoter region of CASP7 reduced the luciferase activity. (A) Schematic representation of CASP7 promoter containing the rs12415607 C/A into pGL3 vector. TSS, transcriptional start site. (B) The CASP7 promoter containing the rs12415607 C or A was inserted into pGL3 vector and transfected into HEK293 cells. At 48 h after transfection, the promoter activity was measured using the Dual Luciferase Reproter assay. Data are presented as mean ± standard error (n = 3, **P < 0.01).
Discussion
The present study demonstrates that the rs12415607 AA in the promoter of CASP7 was associated with a reduced risk of IS. Haplotype analysis showed that the CCGAT and AGGAT haplotypes were associated with a reduced risk of IS, whereas the CCGGC, CCGGT, and AGGGT haplotypes were associated with an increased risk of IS. To follow the aim of analyzing the possible reason for the protective effect of the rs12415607 AA on IS risk, qPCR, and dual-luciferase reporter assay were performed, and we found that the rs12415607 AA genotype associated to lower levels of CASP7 mRNA and reduced promoter activity. These findings indicate that the rs12415607 may be used as a biomarker for the etiology of IS in the Chinese population.
Over the past decades, apoptosis has been demonstrated to contribute to a significant proportion of neuron death following acute brain ischemia11,12,31–33. Several biomarkers of apoptosis have been discovered in cerebrospinal fluid and peripheral blood after IS, such as caspase proteases, notably caspase-3-mediated pathways34–36. Clinically, elevated caspase-3/7 activity was reported in both acute and late phases of stroke patients35–37, and acute caspase-3/7 activation correlated with TNF-α levels, which is an important mediator for IS progression35,37,38. Experimental study also showed that CASP7 mRNA was increased in a rat model of focal cerebral ischemia28. Additionally, microRNA-146a down-regulation correlated to neuroprotection in cerebral ischemic injury in vitro by targeting pro-apoptotic genes: CASP7 and Bcl-2-associated transcription factor 139. Blocking the activity of CASP7 using some traditional Chinese medicines such as aqueous extracts of Lianqiao (Fructus Forsythiae) and Shouwuteng (Caulis Polygoni multiflori) can reduce stroke-inflicted brain damage40. These findings provide strong evidence of causal involvement of CASP7 in stroke, suggesting that targeting physiologic and pharmacologic inhibitors of CASP7 may be a critical therapeutic strategy for IS.
CASP7 is located on chromosome 10q25.3 that has been identified to be a susceptibility locus of IS by GWAS20. We hypothesized in this study that SNPs in CASP7 may affect the individual’s susceptibility to IS. Our findings confirmed this hypothesis, and we found that the rs12415607 AA in the promoter of CASP7 was a protective factor against the occurrence of IS. To date, only three publications investigating the rs12415607 and all of them were involved in cancer. The rs12415607 A allele not only showed a positive association with the risk of cancer but also modulated survival of patients with non-small cell lung cancer treated with platinum-based chemotherapy41–43. Moreover, in this study, we provided the first evidence that the rs2227310 represents an independent risk factor of IS besides the rs12415607. The CASP7 rs2227310 was associated with a potential apoptosis effect in patients with mitochondrial diabetes but not rheumatoid arthritis44,45. The conflicting results indicate that the genetic background is different in different human diseases. Even though no significant association between the rs2227310, rs10787498, rs1127687, and rs4353229 in CASP7 and IS risk was observed in single site analysis, we found in haplotype analysis that the CCGAT and AGGAT haplotypes were associated with a reduced risk of IS, whereas the CCGGC, CCGGT, and AGGGT haplotypes were associated with an increased risk of IS. These findings further support the idea that IS is a complex disease and related to several genetic sites.
In vitro study was then performed to explore the possible reason for the rs12415607 AA decreasing IS risk. After transfection into HEK293 cells, the rs12415607 A allele exhibited a lower level of transcriptional activity compared to the rs12415607 C allele. Genotype-phenotype analysis also showed that the rs12415607 AA carriers had lower levels of CASP7 mRNA. Our findings were verified by data from eQTL, indicating our results are robust. Taken together, we may conclude that the rs12415607 A allele induces lower levels of transcriptional activity and CASP7 mRNA, and thus is associated with a reduced risk of IS.
There are limitations in the current study: i) As the study subjects comprised only Han Chinese, further studies will be required in different ethnicities. ii) Environmental factors are evident to be responsible for the development of IS5–8, gene–environment interaction analysis cannot be performed due to lack of objective data in this study. iii) The study design is hospital-based and selection bias cannot be ruled out, and thus population-based association studies are of great importance.
In conclusion, the present results suggest that the CASP7 rs12415607 on chromosome 10q25.3 may be a susceptibility locus for IS in Chinese individuals. The protective effect of the rs12415607 AA on IS risk may be explained by decreasing CASP7 mRNA levels. Determination of the CASP7 rs12415607 genotype may prove informative for assessment of the genetic risk of IS in such a population. Further studies are warranted to confirm these findings, especially in different ethnic groups.
Supplementary information
Detailed information of the polymorphisms in the 3′- untranslated region of CASP7 and predicted binding miRNAs
Acknowledgements
We would like to thank the subjects for their participation in this research study. This study was supported by the Project of International Cooperation of Jilin Province in China [20180414062GH] and Natural Science Foundation of Jilin Province in China [20180101300JC].
Author contributions
Xuemei Han designed and wrote the manuscript. Zhaoshi Zheng, Songyan Liu, and Chunhui Wang performed experiments and drafted the manuscript. Chuheng Wang and Dong Tang collected samples. Yuqing Shi and Zhaoshi Zheng performed statistical analysis.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-019-55201-y.
References
- 1.Bonita R, et al. The global stroke initiative. The Lancet. Neurology. 2004;3:391–393. doi: 10.1016/S1474-4422(04)00800-2. [DOI] [PubMed] [Google Scholar]
- 2.Weinstein JR, Koerner IP, Moller T. Microglia in ischemic brain injury. Future neurology. 2010;5:227–246. doi: 10.2217/fnl.10.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adams HP, Jr., et al. Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Stroke; a journal of cerebral circulation. 2007;38:1655–1711. doi: 10.1161/STROKEAHA.107.181486. [DOI] [PubMed] [Google Scholar]
- 4.Feigin VL, et al. Global and regional burden of stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet. 2014;383:245–254. doi: 10.1016/S0140-6736(13)61953-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yong H, et al. A systematic literature review of risk factors for stroke in China. Cardiology in review. 2013;21:77–93. doi: 10.1097/CRD.0b013e3182748d37. [DOI] [PubMed] [Google Scholar]
- 6.Bak S, Gaist D, Sindrup SH, Skytthe A, Christensen K. Genetic liability in stroke: a long-term follow-up study of Danish twins. Stroke; a journal of cerebral circulation. 2002;33:769–774. doi: 10.1161/hs0302.103619. [DOI] [PubMed] [Google Scholar]
- 7.Dichgans M. Genetics of ischaemic stroke. The Lancet. Neurology. 2007;6:149–161. doi: 10.1016/S1474-4422(07)70028-5. [DOI] [PubMed] [Google Scholar]
- 8.Schulz UG, Flossmann E, Rothwell PM. Heritability of ischemic stroke in relation to age, vascular risk factors, and subtypes of incident stroke in population-based studies. Stroke; a journal of cerebral circulation. 2004;35:819–824. doi: 10.1161/01.STR.0000121646.23955.0f. [DOI] [PubMed] [Google Scholar]
- 9.Zheng Z, Liu S, Wang C, Han X. A Functional Polymorphism rs145204276 in the Promoter of Long Noncoding RNA GAS5 Is Associated with an Increased Risk of Ischemic Stroke. Journal of stroke and cerebrovascular diseases: the official journal of National Stroke Association. 2018;27:3535–3541. doi: 10.1016/j.jstrokecerebrovasdis.2018.08.016. [DOI] [PubMed] [Google Scholar]
- 10.Han X, Zheng Z, Wang C, Wang L. Association between MEG3/miR-181b polymorphisms and risk of ischemic stroke. Lipids in health and disease. 2018;17:292. doi: 10.1186/s12944-018-0941-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lo EH, Dalkara T, Moskowitz MA. Mechanisms, challenges and opportunities in stroke. Nature reviews. Neuroscience. 2003;4:399–415. doi: 10.1038/nrn1106. [DOI] [PubMed] [Google Scholar]
- 12.Radak D, et al. Apoptosis and Acute Brain Ischemia in Ischemic Stroke. Current vascular pharmacology. 2017;15:115–122. doi: 10.2174/1570161115666161104095522. [DOI] [PubMed] [Google Scholar]
- 13.Nicotera P, Leist M, Fava E, Berliocchi L, Volbracht C. Energy requirement for caspase activation and neuronal cell death. Brain pathology. 2000;10:276–282. doi: 10.1111/j.1750-3639.2000.tb00261.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yamashima T. Implication of cysteine proteases calpain, cathepsin and caspase in ischemic neuronal death of primates. Progress in neurobiology. 2000;62:273–295. doi: 10.1016/S0301-0082(00)00006-X. [DOI] [PubMed] [Google Scholar]
- 15.Schulz JB, Weller M, Moskowitz MA. Caspases as treatment targets in stroke and neurodegenerative diseases. Annals of neurology. 1999;45:421–429. doi: 10.1002/1531-8249(199904)45:4<421::AID-ANA2>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 16.Hara H, et al. Inhibition of interleukin 1beta converting enzyme family proteases reduces ischemic and excitotoxic neuronal damage. Proceedings of the National Academy of Sciences of the United States of America. 1997;94:2007–2012. doi: 10.1073/pnas.94.5.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Han BH, et al. Selective, reversible caspase-3 inhibitor is neuroprotective and reveals distinct pathways of cell death after neonatal hypoxic-ischemic brain injury. The Journal of biological chemistry. 2002;277:30128–30136. doi: 10.1074/jbc.M202931200. [DOI] [PubMed] [Google Scholar]
- 18.Rideout HJ, Stefanis L. Caspase inhibition: a potential therapeutic strategy in neurological diseases. Histology and histopathology. 2001;16:895–908. doi: 10.14670/HH-16.895. [DOI] [PubMed] [Google Scholar]
- 19.Le DA, et al. Caspase activation and neuroprotection in caspase-3- deficient mice after in vivo cerebral ischemia and in vitro oxygen glucose deprivation. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:15188–15193. doi: 10.1073/pnas.232473399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cheng YC, et al. Genome-Wide Association Analysis of Young-Onset Stroke Identifies a Locus on Chromosome 10q25 Near HABP2. Stroke; a journal of cerebral circulation. 2016;47:307–316. doi: 10.1161/STROKEAHA.115.011328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ikram MA, et al. Genomewide association studies of stroke. The New England journal of medicine. 2009;360:1718–1728. doi: 10.1056/NEJMoa0900094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee TH, et al. Identification of PTCSC3 as a Novel Locus for Large-Vessel Ischemic Stroke: A Genome-Wide Association Study. Journal of the American Heart Association. 2016;5:e003003. doi: 10.1161/JAHA.115.003003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Network NSG. & International Stroke Genetics, C. Loci associated with ischaemic stroke and its subtypes (SiGN): a genome-wide association study. The Lancet. Neurology. 2016;15:174–184. doi: 10.1016/S1474-4422(15)00338-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kilarski LL, et al. Meta-analysis in more than 17,900 cases of ischemic stroke reveals a novel association at 12q24.12. Neurology. 2014;83:678–685. doi: 10.1212/WNL.0000000000000707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhu R, Liu X, He Z. Genetic variants on chromosome 10q11.21 are associated with ischemic stroke in the northern Chinese Han population. Journal of molecular neuroscience: MN. 2013;51:394–400. doi: 10.1007/s12031-013-0025-5. [DOI] [PubMed] [Google Scholar]
- 26.Anderson CD, et al. Chromosome 9p21 in ischemic stroke: population structure and meta-analysis. Stroke; a journal of cerebral circulation. 2010;41:1123–1131. doi: 10.1161/STROKEAHA.110.580589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu C, et al. Minor allele C of chromosome 1p32 single nucleotide polymorphism rs11206510 confers risk of ischemic stroke in the Chinese Han population. Stroke; a journal of cerebral circulation. 2010;41:1587–1592. doi: 10.1161/STROKEAHA.110.583096. [DOI] [PubMed] [Google Scholar]
- 28.Harrison DC, et al. Caspase mRNA expression in a rat model of focal cerebral ischemia. Brain research. Molecular brain research. 2001;89:133–146. doi: 10.1016/S0169-328X(01)00058-4. [DOI] [PubMed] [Google Scholar]
- 29.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262S1046-2023(01)91262-9. [DOI] [PubMed] [Google Scholar]
- 30.Shi YY, He L. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. 2005;15:97–98. doi: 10.1038/sj.cr.7290272. [DOI] [PubMed] [Google Scholar]
- 31.Unal-Cevik I, Kilinc M, Can A, Gursoy-Ozdemir Y, Dalkara T. Apoptotic and necrotic death mechanisms are concomitantly activated in the same cell after cerebral ischemia. Stroke; a journal of cerebral circulation. 2004;35:2189–2194. doi: 10.1161/01.STR.0000136149.81831.c5. [DOI] [PubMed] [Google Scholar]
- 32.Broughton BR, Reutens DC, Sobey CG. Apoptotic mechanisms after cerebral ischemia. Stroke; a journal of cerebral circulation. 2009;40:e331–339. doi: 10.1161/STROKEAHA.108.531632. [DOI] [PubMed] [Google Scholar]
- 33.Siren AL, et al. Erythropoietin prevents neuronal apoptosis after cerebral ischemia and metabolic stress. Proceedings of the National Academy of Sciences of the United States of America. 2001;98:4044–4049. doi: 10.1073/pnas.051606598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang X, et al. Proteolysis consistent with activation of caspase-7 after severe traumatic brain injury in humans. Journal of neurotrauma. 2006;23:1583–1590. doi: 10.1089/neu.2006.23.1583. [DOI] [PubMed] [Google Scholar]
- 35.Lin CH, Chen M, Sun MC. Circulating apoptotic factors in patients with acute cerebral infarction. Clinical biochemistry. 2010;43:761–763. doi: 10.1016/j.clinbiochem.2010.03.002. [DOI] [PubMed] [Google Scholar]
- 36.Rosell A, et al. Caspase-3 is related to infarct growth after human ischemic stroke. Neuroscience letters. 2008;430:1–6. doi: 10.1016/j.neulet.2007.05.006. [DOI] [PubMed] [Google Scholar]
- 37.Pascotini ET, et al. Apoptotic markers and DNA damage are related to late phase of stroke: Involvement of dyslipidemia and inflammation. Physiology & behavior. 2015;151:369–378. doi: 10.1016/j.physbeh.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 38.Chen AQ, et al. Microglia-derived TNF-alpha mediates endothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke. Cell death & disease. 2019;10:487. doi: 10.1038/s41419-019-1716-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou X, et al. MicroRNA-146a down-regulation correlates with neuroprotection and targets pro-apoptotic genes in cerebral ischemic injury in vitro. Brain research. 2016;1648:136–143. doi: 10.1016/j.brainres.2016.07.034. [DOI] [PubMed] [Google Scholar]
- 40.Fattorusso R, Frutos S, Sun X, Sucher NJ, Pellecchia M. Traditional Chinese medicines with caspase-inhibitory activity. Phytomedicine: international journal of phytotherapy and phytopharmacology. 2006;13:16–22. doi: 10.1016/j.phymed.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 41.Zhang ZY, Xuan Y, Jin XY, Tian X, Wu R. A literature-based systematic HuGE review and meta-analysis show that CASP gene family polymorphisms are associated with risk of lung cancer. Genetics and molecular research: GMR. 2013;12:3057–3069. doi: 10.4238/2013.January.4.22. [DOI] [PubMed] [Google Scholar]
- 42.Yan S, et al. HuGE systematic review and meta-analysis demonstrate association of CASP-3 and CASP-7 genetic polymorphisms with cancer risk. Genetics and molecular research: GMR. 2013;12:1561–1573. doi: 10.4238/2013.May.13.10. [DOI] [PubMed] [Google Scholar]
- 43.Qian J, et al. Association of CASP7 polymorphisms and survival of patients with non-small cell lung cancer with platinum-based chemotherapy treatment. Chest. 2012;142:680–689. doi: 10.1378/chest.11-2522. [DOI] [PubMed] [Google Scholar]
- 44.Tabebi M, et al. Association study of apoptosis gene polymorphisms in mitochondrial diabetes: A potential role in the pathogenicity of MD. Gene. 2018;639:18–26. doi: 10.1016/j.gene.2017.09.063. [DOI] [PubMed] [Google Scholar]
- 45.Garcia-Lozano JR, et al. Caspase 7 influences susceptibility to rheumatoid arthritis. Rheumatology. 2007;46:1243–1247. doi: 10.1093/rheumatology/kem096. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Detailed information of the polymorphisms in the 3′- untranslated region of CASP7 and predicted binding miRNAs