Abstract
AIM
To explore the effects and mechanisms of mechanical stress and transforming growth factor-beta2 (TGF-β2) on epithelial-mesenchymal transition (EMT) in cultured human retinal pigment epithelial (RPE) cells.
METHODS
Human RPE cells were inoculated on BioFex 6-well plates and RPE cells received 0, 1, 2, 3, or 4 mild stretch injuries delivered 3h apart after 24h of culture. The device of mechanical stress parameters were set to sine wave, frequency 1 Hz, stretch strength 20%. For treatment with TGF-β2, when the inoculated RPE cells in 6-well plates were around 60% confluent, serum was reduced to 0 for 12h and recombinant human TGF-β2 (0, 1, 5, 10 ng/mL) was added for 48h. α-SMA, Vimentin and N-Cadherin, fibronectin proteins expressions were detected by Western blotting, confocal cell immunofluorescence and quantitative real-time polymerase chain reaction (qRT-PCR). Then we detected the change of miRNA-29b and ascertained the changes of phosphatidylinositol 3-kinase-serine threonine protein kinase (PI3K/Akt) pathway after RPE cells were stretched by the device of mechanical stress and induced by TGF-β2 by Western blotting, confocal cell immunofluorescence and qRT-PCR.
RESULTS
Mechanical stress induce EMT and activate the PI3K/Akt pathway in ways that lead to the EMT process. TGF-β2 induce RPE cells EMT and in a certain range and TGF-β2 decrease the miRNA-29b expression in RPE cells, and the inhibitory effect is more obvious with the increase of TGF-β2 concentration.
CONCLUSION
Our findings are crucial steps in determining the critical roles of the PI3K/Akt signaling pathway and miRNA-29b in pathogenesis of proliferative vitreoretinopathy (PVR) which may be a potential target for preventing or treating PVR.
Keywords: mechanical stress, transforming growth factor-beta2, microRNA 29b, epithelial-mesenchymal transition, phosphatidylinositol 3-kinase-serine threonine protein kinase pathway, proliferative vitreoretinopathy
INTRODUCTION
Proliferative vitreoretinopathy (PVR) is most commonly found in long-term non-treated rhegmatogenous retinal detachment (RRD) and the retinal detachment after surgical treatment. It has long been credited with the main cause of failure of retinal detachment surgery and significant vision loss[1].
Epiretinal membranes, a main feature of PVR, is constantly contracting over the process of PVR, and leads to retinal detachment and, eventually, the loss of vision[1]. During the formation of epiretinal membranes, human retinal pigment epithelial (RPE) cells occurs epithelial mesenchymal transition (EMT) plays a decisive role[2], and cell pathways and various cytokines are involved in this EMT process, including the phosphatidylinositol 3-kinase-serine threonine protein kinase (PI3K/Akt) pathway[3].
Transforming growth factor-beta2 (TGF-β2) was found abnormal high expressed in epiretinal membranes of PVR patients[4]–[5] and is gotten labeled as an induction factor of EMT, also in RPE cells. Additionally, a growing body of studies have reported that microRNAs (miRNAs) have a strong capacity for gene regulation in physiological processes and in some pathophysiological processes[6]–[9]. This small non-coding endogenous RNA participates in the expression regulation of approximately 30%-50% of genes encoding proteins by interacts with the 3′ untranslated region (3′-UTR) of its objective genes[10]. It is reported that there were 304 miRNA expression changes during TGF-β2 induced EMT process in RPE cells, of which 185 were down-regulated and 119 were up-regulated, indicating that miRNAs might play an important role in EMT of RPE cells[11].
Published data have demonstrated that mechanical stress plays an increasingly critical role in some physiological processes, including cell proliferation, differentiation, apoptosis, gene expression, organization growth, and some pathological processes[12]. These processes are similar to the PVR processes which are caused by RPE cells abnormal proliferation. So, we can establish a more accurate and more fit model of PVR in vitro to provide important methods for further study of PVR. However, we still don't have enough information about this mechanism of the activation of retinal cells induced by excessive mechanical stress from a pathogenetic point of view[13].
So, starting with the association between PVR progression and PI3K-Akt signaling pathway and miRNA-29b, our study expounds the mechanism of PVR and provide a well-established theoretical foundation for further study of the prevention and treatment of PVR. We applied mechanical stretching on human RPE cells and induced RPE cells EMT process through the PI3K/Akt signaling pathway. In addition, we confirmed that TGF-β2 also can induce RPE cells EMT and inhibit the expression of miRNA-29b and this inhibitory effect is more pronounced with increasing concentration and time in vitro. The purpose of the present study was to investigate how human RPE cells respond and adapt to mechanical stress.
MATERIALS AND METHODS
Cell Culture Treatment
Fetal bovine serum (10%; Gibco, California, USA) and 1% penicillin/streptomycin (Gibco, California, USA) were added to the DMEM/F12 (Gibco, California, USA) to culture ARPE-19 cell line at a humidity of 5% CO2 at 37°C after they were bought from American Type Culture Collection (ATCC; Manassas, USA). Change culture fluid every three days.
Stretch by the Device of Mechanical Stress
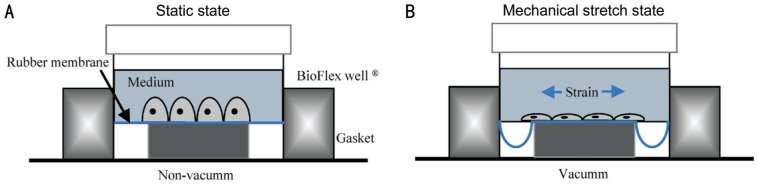
RPE cells were inoculated on BioFex 6-well plates and the RPE cell densities were 1×106 cells per well. RPE cells received 0, 1, 2, 3, or 4 mild stretch injuries delivered 3h apart after 24h of culture. The device of mechanical stress parameters were set to sine wave, frequency 1 Hz, stretch strength 20% (Figure 1). The cells in BioFex 6-well culture plate were collected and analyzed by Western blot and confocal cell immunofluorescence.
Figure 1. A diagram of the mechanical stress cell model.
A: After RPE cells were stretched by the device of mechanical stress, the shape of cells was changed and RPE cells lost epithelial morphology (A) and presented with this classical spindle-shaped appearance (B).
Treatment with Transforming Growth Factor-Beta2
When the inoculated RPE cells in 6-well plates were around 60% confluent, serum was reduced to 0 for 12h and recombinant human TGF-β2 (0, 1, 5, 10 ng/mL) (Peprotech, Rocky Hill, USA) was added for 48h. During this period, observe the changes in cell morphology with a phase contrast microscopy (Olympus, Tokyo, Japan) and finally, cells in 6-well plates were collected and analyzed for fibronectin (FN) and N-cadherin expression by Western blot analysis and quantitative real-time polymerase chain reaction (qRT-PCR). For miRNA-29b expression analysis, we used different concentrations (0, 1, 5, 10 ng/mL) of TGF-β2 to stimulate RPE cells for 24h, and also used a constant concentration (5 ng/mL) of TGF-β2 to stimulate RPE cells at 0, 3, 6, 12, 24, 48h, qRT-PCR allowed the identification of miRNA-29b expression.
Western Blotting Analysis
Firstly, RPE cells should be washed and lysed with 400 µL radioimmunoprecipitation buffer [containing 50 mmol/L Tris (pH 7.4), 1% Triton X-100, 150 mmol/L NaCl, 1% sodium deoxycholate, 0.1% SDS, 5 mmol/L sodium orthovanadate, 1 mmol/L phenylmethanesulfonyl fluoride, 5 mmol/L EDTA (Beyotime Institute of Biotechnology, Shanghai, China)] and 4 µL protease inhibitors (Jiangsu KeyGen Bio Tech Corp, Ltd, Nanjing, China) at 4°C for 5min. And then using a cell scraper and cryogenic centrifuge to gather cell lysates and centrifuge lysates at 13 000 rpm for 10min at 4°C. After determination with the BCA kit (Jiangsu KeyGen Bio Tech Corp.Ltd, Nanjing, China) for total protein concentration, the samples which contained 20 µg protein per well were denatured, subjected to 7%-10% SDS-PAGE, and moved onto PVDF (EMD Millipore, Belize, USA). And the membranes were kept first in 5% TBST for 2h at room temperature and later in primary antibodies (1:1000) at 4°C for 24h to against phosphorylated-Akt (p-Akt; Bioworld, Bloomington, USA), phosphorylated-PI3K (p-PI3K; Affinity, Cincinnati, USA), N-Cadherin (N-cad; Cell Signaling Technology, Danvers, USA) and FN (Abcam, Cambridge, USA). Again, after the membranes were rinsed three times, kept in secondary antibody (1:10 000, Boster, Pleasanton, USA) for 1h. Finally, we visualized proteins with ImageQuant LAS4000 system (GE Healthcare, USA). The relative levels of each protein were quantified with Image J software (National Institutes of Health, Bethesda, USA).
Real-time Quantitative Polymerase Chain Reaction
After the total RNA were isolated with TRIzol reagent (Invitrogen, California, USA), measured concentration and purity via spectrophotometry, giving an RNAA260/280 ratio of 1.8-2.0 (GE, USA). Reverse transcription using a Prime Script RT Master Mix kit (TaKaRa, Kusatsu, Japan), and the fluorescence of each cycle was quantified with a 7300 RT-PCR system (Applied Biosystems, California, USA) using the SYBR1 Premix Ex TaqTM kit (TaKaRa, Kusatsu, Japan). As shown in Table 1, the specific primers were used in this experiment. The time, temperature and cycle index of the reaction were set according to manufacturer's instructions. Using the 2−ΔΔCt method to analyze the relative mRNA and miRNA expression level. GAPDH and U6 primers served as the internal controls.
Table 1. Specific primers of quantitative polymerase chain reaction.
| Genes | Sequences (5′-3′) |
| FN | F: 5′-ATCACCCTCAACCTCAC-3′ |
| R: 5′-TCCCTCGGAACATCAGAAAC-3′ | |
| N-Cadherin | F: 5′-AGCTTCTCACGGCATACACC-3′ |
| R: 5′-GTGCATGAAGGACAGCCTCT-3′ | |
| GAPDH | F: 5′-CGGAGTCAACGGATTTGGTCGTAT-3′ |
| R: 5′-AGCCTTCTCCATGGTGGTGAAGAC-3′ |
F: Forward; R: Reverse; FN: Fibronectin; N-Cadherin: Nerve calcium adhesion protein; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase.
Confocal Cell Immunofluorescence
After the cells were stretched with mechanical stress for 9h, they were fixed in 4% paraformaldehyde, rinsed 3 times with PBS, kept in 0.5% Triton X-100 (Sigma, Burlington, USA) and 5% goat serum. All the above operations are performed at room temperature. And slides were kept in primary antibodies (p-PI3K, 1:50, Affinity, Cincinnati, USA; p-Akt, 1:50, Bio world, Blooming, USA) overnight at 4°C. The slips were then kept in fluorescent dye-labeled antibody in dark for 40min. Cells were finally double stained with DAPI (1:1000; Invitrogen, California, USA) for 2min and observed RPE cell using a confocal laser scanning microscope (ZEISS, Oberkochen, Germany).
Statistical Analysis
Results involved in this study were independently completed at least three times, and the means± standard deviation (SD) was used to present the quantitative data. Analyze data with SPSS 17.0 software. One-way analysis of variance (ANOVA) method was used to analyze the relationship between different groups. As long as P<0.05, the data could be regarded as a statistically significant results.
RESULTS
ARPE-19 Cells Treated With Mechanical Stress to Assess the Impact of EMT
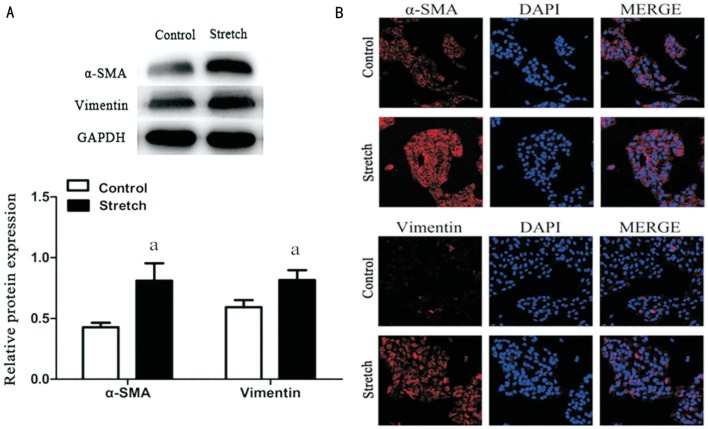
After exposure to mechanical stretch for 9h, changes in expression levels of the mesenchymal marker were measured. Western blot and confocal cell immunofluorescence showed that expression of two mesenchymal proteins, α-SMA and Vimentin, were significantly enhanced in RPE cells following stretch by the device of mechanical stress (Figure 2). Thus, these data suggested that stretch can induce EMT in RPE cells.
Figure 2. Stretch induced RPE cells EMT.
RPE cells were stretched by the device of mechanical stress for 9h before detection. A: Western blotting revealed an increase in α-SMA and Vimentin proteins levels. GAPDH was selected for internal reference (aP<0.05); B: α-SMA and Vimentin expression by confocal cell immunofluorescence. Performed all experiments in triplicate.
Increased Protein Expression of p-AKT and p-PI3K in RPE Cell
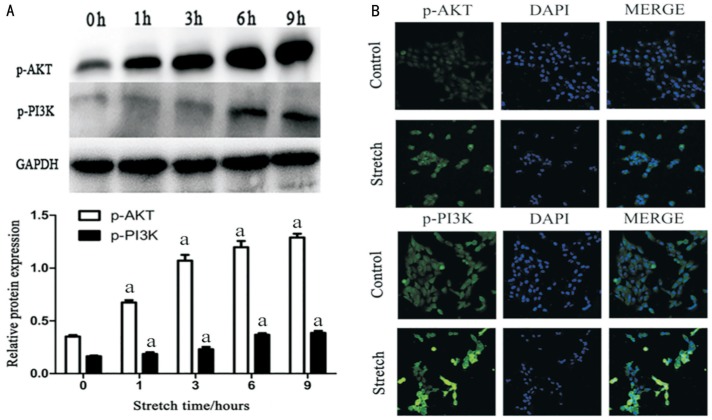
To confirm that stretching RPE cells can activate the PI3K/Akt pathway in ways that lead to EMT (RPE cells were stretched for 0, 1, 3, 6, 9h), we next detected the phosphorylation level of Akt and PI3K by Western blotting and confocal immunofluorescence. Results from quantitative immunoblotting analysis indicates that phosphorylation of PI3K and Akt increased depending on time (Figure 3A). Furthermore, the results of immunofluorescence of the co-culture stretched cells showed that in comparison with the non-stretched group, the p-Akt and p-PI3K expression were more higher following stretched. The green fluorescence could be observed in cells express p-Akt and p-PI3K, and be more pronounced in the stretched cells (Figure 3B).
Figure 3. Mechanical stress induces EMT and active PI3K/Akt signaling pathway in RPE cells.
RPE cells were stretched by the device of mechanical stress (0, 1, 3, 6, 9h). A: Protein abundance of p-Akt and p-PI3K in mechanical stress stretched RPE cell was quantified by Western blotting and increased with increasing time (aP<0.05). B: Investigate the p-Akt and p-PI3K expression by confocal cell immunofluorescence. Three independent experiments were performed.
Increased Proteins Expression of FN and N-Cadherin in TGF-β2 Induced RPE Cells
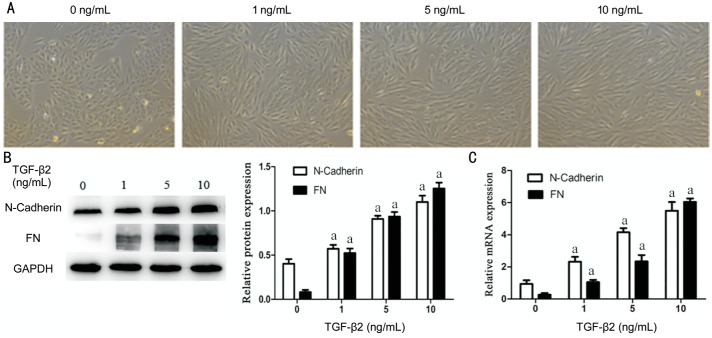
After stimulating RPE cells for 48h with various concentrations of TGF-β2 (0, 1, 5, 10 ng/mL), the shape of cells was changed and presented with this classical spindle-shaped appearance which was more obvious with increase of concentration of TGF-β2 (Figure 4A). As further proof, after stimulating RPE cells for 48h by different concentrations (0, 1, 5, 10 ng/mL) of TGF-β2, mesenchymal marker FN and N-Cadherin expression was measured by Western blotting and qRT-PCR. The results proved that TGF-β2 can induce FN and N-cadherin expression and the effect was more obvious with dose increasing (Figure 4B, 4C). Therefore, it can be shown that TGF-β2 can be used to induce EMT in RPE cells.
Figure 4. TGF-β2 induce EMT in RPE cells.
RPE cells were exposed to three different concentrations (1, 5, 10 ng/mL) of TGF-β2 for 48h and 0 ng/mL TGF-β2 is a blank control group. A: The cell morphological appearance was analyzed by a phase-contrast microscope at 100× magnification; B: Western blotting showed TGF-β2 could induce N-Cadherin and FN proteins expression with obvious dose-dependence. GAPDH was selected for internal reference (aP<0.05). C: qRT-PCR analysis showed N-Cadherin and FN mRNA expression increases with the TGF-β2 concentration. GAPDH was selected for internal reference (aP<0.05). Performed all experiments in triplicate.
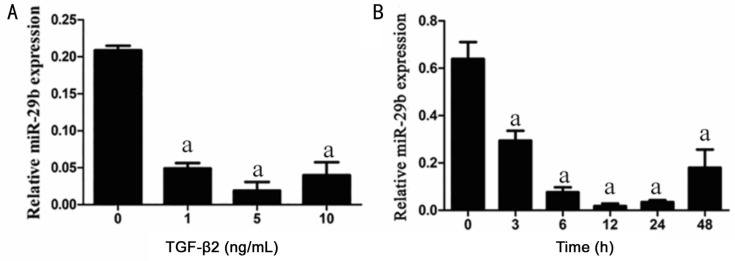
TGF-β2 Decreased the miRNA-29b Expression in RPE Cells
To examine the miRNA-29b expression, the RPE cells stimulated for 24h with different concentration (0, 1, 5, 10 ng/mL) of TGF-β2 and stimulated with 5 ng/mL TGF-β2 for 0, 3, 6, 12, 24, 48h. The result of qRT-PCR showed that, miRNA-29b expression was decreased with the increase of TGF-β2 concentration, and reached the lowest in 5 ng/mL TGF-β2 induced group (Figure 5A). The expression of miRNA-29b in RPE cells decreased with the increase of TGF-β2 induction time, and miRNA-29b expression was lowest at 12h after induction (Figure 5B).
Figure 5. The miRNA-29b expression was inhibited after induced with different concentrations and different time of TGF-β2.
A: Once stimulating with different concentrations of TGF-β2 in RPE cells, the miRNA-29b expression appeared to be significantly changed. Cultured RPE cells under different concentrations of TGF-β2 (0, 1, 5, 10 ng/mL) for 24h. qRT-PCR analysis showed that miRNA-29b regulation took on the typically negative regulation function by TGF-β2 in RPE cells. U6 was selected for internal reference (aP<0.05). B: RPE cells were stimulated with a constant concentration (5 ng/mL) of TGF-β2 at 0, 3, 6, 12, 24, 48h. qRT-PCR analysis showed that miRNA-29b expression in cells decreased with the increase of the time of TGF-β2 addition. U6 was selected for internal reference (aP<0.05).
DISCUSSION
EMT is a biological process with many regulation factors, which is accompanied with significant reduction of mesenchymal markers and increase of epithelial markers. There are series physiological and biological reactions in the course of EMT, and these reactions can seriously affect cell motility, proliferation, apoptosis, and protein expression. Pulmonary fibrosis, liver fibrosis, renal fibrosis and breast cancer metastasis are closely related to the EMT process[14]–[17]. It has been shown that RPE cells that have undergone EMT is the main cause of retinal traction and surgery failure and the initiation factor of PVR[18]. However, the mechanisms involved during PVR are remains unexplored.
Flex cell tissue mechanical culture system can provide different stress, magnitude of the force and simulate different stress time. Therefore, Flex cell device of mechanical stress stimulates in vitro to induce RPE cells to EMT could be not only more accurate but also produce persistent mechanical stretch similar to produce by fibrous proliferative membrane. Therefore, mechanical stress could be used to simulate the pathophysiological process of PVR[19]. In our study, after exposure to mechanical stretch for 9h, changes in expression levels of the mesenchymal marker were measured. So we demonstrated that mechanical stress induce EMT in RPE cells and established a PVR model in vitro.
It has been reported a number of factors are involved in cell growth, proliferation, as well as the migration and the 3′-phosphatidylinositol, which is produced by an intracellular signaling molecule, PI3K, being one of them. Akt, a downstream kinase of PI3K, is always phosphorylated at ser473 and recruited to the membrane[20]–[22]. In our study, we explored the changes of PI3K and Akt phosphorylation level caused by stretching in PRE cells using Western blotting and confocal cell immunofluorescence to confirm that PI3K/Akt signaling pathways were activated during EMT in RPE cells. The Western blotting result revealed the expressions level of phosphorylated Akt and PI3K increased with the increasing time of stretch. Similarly, the expression of p-Akt and p-PI3K in stretched RPE cells were higher in confocal cell immunofluorescence. The above results indicated that mechanical stress might induce RPE cells EMT, and this process could active Akt/PI3K pathways.
Furthermore, we noticed that after stimulating RPE cells for 48h with various concentrations of TGF-β2, the RPE cells could undergo an EMT process. And more interestingly, according to the reference of a great deal of documents, mechanical stress induces VEGF expression and promotes angiogenesis in RPE cells, and regulation of VEGF mRNA expression and protein secretion by TGF-β2 in human RPE cells[23]. This new finding is also consistent with research on other organs[24]. Therefore, we believed that TGF-β2 plays an important role in the course of mechanical stress, and so, the regulation of TGF-β2 by other gene regulators, will be an important idea in suppressing mechanical stress induced EMT and, thus, in preventing or treating the undesirable outcomes of PVR.
miRNA, as a small non-coding endogenous RNAs, can participated in gene expression regulation and importantly affect many different mRNAs through miRNA-mRNA interactions (miRNA interacts directly with 3′UTR of its corresponding mRNA target), that renders them regulate cell physiological and pathological processes. During the induction of TGF-β2, there were 304 miRNA expression changes, among which the expression of miRNA-29b was down regulated more than 80%[25]. miRNA-29b, an important mediator of TGF-β induced EMT, is down regulated in fibrotic disorders[26]–[27] and cancers[28]–[29] compared with normal tissues. Many researches have been done showing that miRNA-29 not only associated with the formation of extracellular matrix (ECM), but participates the regulation process of organ fibrosis[29]. In the cardiac tissue of C57BL/6 mice, down-regulation of the miRNA-29b expression can induced the overexpression of Col1a1, Col1a2 and Col3a1 mRNA[30]. Those researches are consistent with our findings that TGF-β2 can inhibit miRNA-29b expression in RPE cells in time-dose dependence, and that miRNA-29b is a potential and attractive therapeutic target in the treatment of fibrotic diseases.
In conclusion, our result suggests that Akt/PI3K signaling pathway was activated during EMT induced by mechanical stress in RPE cells. Furthermore, the miRNA-29b down regulated by a time-dose dependence in TGFβ2-induced EMT. These findings suggest that mechanical stress and TGF-β2 can induce RPE cells EMT and further studies could focus on inhibitors of the PI3K/Akt pathway and overexpression of miRNA-29b to prevent or treat PVR. Knowledge of these mechanisms could further understand the pathogenesis of PVR, and indicate new prevention strategies and therapeutic targets.
Acknowledgments
We are grateful to Key Laboratory for Oral Disease Research of Nanjing Medical University kindly to provide the experimental facilities and equipment for this study.
Foundations: Supported by the National Natural Science Foundation of China (No.81600754).
Conflicts of Interest: Cao Q, None; Deji QZ, None; Liu YJ, None; Ye W, None; Zhaba WD, None; Jiang Q, None; Yan F, None.
REFERENCES
- 1.Kwon OW, Song JH, Roh MI. Retinal detachment and proliferative vitreoretinopathy. Dev Ophthalmol. 2016;55:154–162. doi: 10.1159/000438972. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Y, Zhao D, Yang S, Yao H, Li M, Zhao C, Zhang J, Xu GT, Li H, Wang F. Protective effects of fucoidan on epithelial-mesenchymal transition of retinal pigment epithelial cells and progression of proliferative vitreoretinopathy. Cell Physiol Biochem. 2018;46(4):1704–1715. doi: 10.1159/000489246. [DOI] [PubMed] [Google Scholar]
- 3.Zhang C, Su L, Huang L, Song ZY. GSK3β inhibits epithelial-mesenchymal transition via the Wnt/β-catenin and PI3K/Akt pathways. Int J Ophthalmol. 2018;11(7):1120–1128. doi: 10.18240/ijo.2018.07.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pastor JC, Rojas J, Pastor-Idoate S, Di Lauro S, Gonzalez-Buendia L, Delgado-Tirado S. Proliferative vitreoretinopathy: a new concept of disease pathogenesis and practical consequences. Prog Retin Eye Res. 2016;51:125–155. doi: 10.1016/j.preteyeres.2015.07.005. [DOI] [PubMed] [Google Scholar]
- 5.Wang HF, Ma JX, Shang QL, An JB, Chen HT. Crocetin inhibits the proliferation, migration and TGF-β2-induced epithelial-mesenchymal transition of retinal pigment epithelial cells. Eur J Pharmacol. 2017;815:391–398. doi: 10.1016/j.ejphar.2017.09.041. [DOI] [PubMed] [Google Scholar]
- 6.Wienholds E, Kloosterman WP, Miska E, Alvarez-Saavedra E, Berezikov E, de Bruijn E, Horvitz HR, Kauppinen S, Plasterk RH. MicroRNA expression in zebrafish embryonic development. Science. 2005;309(5732):310–311. doi: 10.1126/science.1114519. [DOI] [PubMed] [Google Scholar]
- 7.Yi R, O'Carroll D, Pasolli HA, Zhang Z, Dietrich FS, Tarakhovsky A, Fuchs E. Morphogenesis in skin is governed by discrete sets of differentially expressed microRNAs. Nat Genet. 2006;38(3):356–362. doi: 10.1038/ng1744. [DOI] [PubMed] [Google Scholar]
- 8.Allegra A, Alonci A, Campo S, Penna G, Petrungaro A, Gerace D, Musolino C. Circulating microRNAs: new biomarkers in diagnosis, prognosis and treatment of cancer (Review) Int J Oncol. 2012;41(6):1897–1912. doi: 10.3892/ijo.2012.1647. [DOI] [PubMed] [Google Scholar]
- 9.He J, Jing Y, Li W, Qian X, Xu Q, Li FS, Liu LZ, Jiang BH, Jiang Y. Roles and mechanism of miR-199a and miR-125b in tumor angiogenesis. PLoS One. 2013;8(2):e56647. doi: 10.1371/journal.pone.0056647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen X, Ye S, Xiao W, Luo L, Liu Y. Differentially expressed microRNAs in TGFβ2-induced epithelial-mesenchymal transition in retinal pigment epithelium cells. Int J Mol Med. 2014;33(5):1195–1200. doi: 10.3892/ijmm.2014.1688. [DOI] [PubMed] [Google Scholar]
- 12.Downward J. Mechanisms and consequences of activation of protein kinase B/Akt. Curr Opin Cell Biol. 1998;10(2):262–267. doi: 10.1016/s0955-0674(98)80149-x. [DOI] [PubMed] [Google Scholar]
- 13.Kinoshita H, Suzuma K, Maki T, Maekawa Y, Matsumoto M, Kusano M, Uematsu M, Kitaoka T. Cyclic stretch and hypertension increase retinal succinate: potential mechanisms for exacerbation of ocular neovascularization by mechanical stress. Invest Ophthalmol Vis Sci. 2014;55(7):4320–4326. doi: 10.1167/iovs.13-13839. [DOI] [PubMed] [Google Scholar]
- 14.Wu Z, Yang L, Cai L, Zhang M, Cheng X, Yang X, Xu J. Detection of epithelial to mesenchymal transition in airways of a bleomycin induced pulmonary fibrosis model derived from an alpha-smooth muscle actin-Cre transgenic mouse. Respir Res. 2007;8:1. doi: 10.1186/1465-9921-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carew RM, Wang B, Kantharidis P. The role of EMT in renal fibrosis. Cell Tissue Res. 2012;347(1):103–116. doi: 10.1007/s00441-011-1227-1. [DOI] [PubMed] [Google Scholar]
- 16.David AB, Tatiana K, David S, et al. Origin of myofibroblasts in liver fibrosis. Fibrogenesis Tissue Repair. 2012;5(Suppl 1):S17. doi: 10.1186/1755-1536-5-S1-S17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Burgess DJ. Breast cancer: circulating and dynamic EMT. Nat Rev Cancer. 2013;13(3):148. doi: 10.1038/nrc3475. [DOI] [PubMed] [Google Scholar]
- 18.Saika S, Yamanaka O, Okada Y, Tanaka S, Miyamoto T, Sumioka T, Kitano A, Shirai K, Ikeda K. TGF beta in fibroproliferative diseases in the eye. Front Biosci (Schol Ed) 2009;1:376–390. doi: 10.2741/S32. [DOI] [PubMed] [Google Scholar]
- 19.Farjood F, Vargis E. Novel devices for studying acute and chronic mechanical stress in retinal pigment epithelial cells. Lab Chip. 2018;18(22):3413–3424. doi: 10.1039/c8lc00659h. [DOI] [PubMed] [Google Scholar]
- 20.Cantley LC. The phosphoinositide 3-ki18nase pathway. Science. 2002;296(5573):1655–1657. doi: 10.1126/science.296.5573.1655. [DOI] [PubMed] [Google Scholar]
- 21.Downward J. Mechanisms and consequences of activation of protein kinase B/Akt. Curr Opin Cell Biol. 1998;10(2):262–267. doi: 10.1016/s0955-0674(98)80149-x. [DOI] [PubMed] [Google Scholar]
- 22.Su CH, Wang CY, Lan KH, Li CP, Chao Y, Lin HC, Lee SD, Lee WP. Akt phosphorylation at Thr308 and Ser473 is required for CHIP-mediated ubiquitination of the kinase. Cell Signal. 2011;23(11):1824–1830. doi: 10.1016/j.cellsig.2011.06.018. [DOI] [PubMed] [Google Scholar]
- 23.Wang X, Ma W, Han S, Meng Z, Zhao L, Yin Y, Wang Y, Li J. TGF-β participates choroid neovascularization through Smad2/3-VEGF/TNF-α signaling in mice with laser-induced wet age-related macular degeneration. Sci Rep. 2017;7(1):9672. doi: 10.1038/s41598-017-10124-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Froese AR, Shimbori C, Bellaye PS, Inman M, Obex S, Fatima S, Jenkins G, Gauldie J, Ask K, Kolb M. Stretch-induced activation of transforming growth factor-β1 in pulmonary fibrosis. Am J Respir Crit Care Med. 2016;194(1):84–96. doi: 10.1164/rccm.201508-1638OC. [DOI] [PubMed] [Google Scholar]
- 25.Chen X, Ye S, Xiao W, Luo L, Liu Y. Differentially expressed microRNAs in TGFβ2-induced epithelial-mesenchymal transition in retinal pigment epithelium cells. Int J Mol Med. 2014;33(5):1195–1200. doi: 10.3892/ijmm.2014.1688. [DOI] [PubMed] [Google Scholar]
- 26.Cushing L, Kuang PP, Qian J, Shao F, Wu J, Little F, Thannickal VJ, Cardoso WV, Lü J. miR-29 is a major regulator of genes associated with pulmonary fibrosis. Am J Respir Cell Mol Biol. 2011;45(2):287–294. doi: 10.1165/rcmb.2010-0323OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roderburg C, Urban GW, Bettermann K, Vucur M, Zimmermann H, Schmidt S, Janssen J, Koppe C, Knolle P, Castoldi M, Tacke F, Trautwein C, Luedde T. Micro-RNA profiling reveals a role for miR-29 in human and murine liver fibrosis. Hepatology. 2011;53(1):209–218. doi: 10.1002/hep.23922. [DOI] [PubMed] [Google Scholar]
- 28.Wang C, Bian Z, Wei D, Zhang JG. miR-29b regulates migration of human breast cancer cells. Mol Cell Biochem. 2011;352(1-2):197–207. doi: 10.1007/s11010-011-0755-z. [DOI] [PubMed] [Google Scholar]
- 29.Zhang W, Qian JX, Yi HL, Yang ZD, Wang CF, Chen JY, Wei XZ, Fu Q, Ma H. The microRNA-29 plays a central role in osteosarcoma pathogenesis and progression. Mol Biol (Mosk) 2012;46(4):622–627. [PubMed] [Google Scholar]
- 30.van Rooij E, Sutherland LB, Thatcher JE, DiMaio JM, Naseem RH, Marshall WS, Hill JA, Olson EN. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci U S A. 2008;105(35):13027–13032. doi: 10.1073/pnas.0805038105. [DOI] [PMC free article] [PubMed] [Google Scholar]