Abstract
A 58-year-old female patient presented with left breast lump. Mammography and ultrasonography were performed, which reported lesions suspicious of malignancy in the left breast and axilla. Core biopsy of the lesions revealed invasive ductal carcinoma with axillary lymph node metastasis. Staging CT scan (thorax, abdomen and pelvis) identified a coexisting neoplasm in the ascending colon. Colonoscopy was performed and the tumour in the ascending colon was biopsied. Histopathological examination revealed adenocarcinoma. In the multidisciplinary team cancer meeting, it was decided to treat the breast cancer first and then the colon cancer, followed by adjuvant chemotherapy. In the first operation, left modified radical mastectomy was performed. Two weeks after her initial operation, the patient underwent laparoscopic right hemicolectomy. Postoperatively, the patient did not develop any complications and was referred to oncology for chemotherapy.
Keywords: breast cancer, colon cancer, breast surgery, general surgery, cancer intervention
Background
Cancers developing from more than one origin are called multiple primary cancers (MPCs) and have a reported frequency of 0.73%–11%.1 MPCs can be divided into synchronous and metachronous tumours. Synchronous tumours are defined as two different tumours originating in the same patient that are detected at the same time or within 6 months of primary tumour diagnosis, whereas tumours developing 6 months after primary tumour detection are called metachronous tumours.2 There are no guidelines for the management of synchronous tumours. Because of this, development of patient-oriented treatment strategy is necessary to decide which cancer to treat first and how to stratify further treatments. This process involves multidisciplinary team (MDT) cancer meeting input from oncologists, surgeons, radiologists and pathologists to ensure a favourable patient outcome.
We present a patient who was diagnosed with synchronous breast and colon cancer. This case explores the issues of initial treatment and subsequent changes in management based on incidental finding of colonic mass on CT scan for staging of primary breast cancer. It highlights the important role of MDT cancer meeting discussion and clinical challenge in the management of synchronous cancers in order to provide the best prognosis.
Case presentation
A 58-year-old Bahraini female patient presented to the breast clinic with complaints of left breast lump. She stated that she had felt the lump in her left breast for 3 months, and for the last 2 months, she had unintentional weight loss of 6 kg. There was no history of any nipple abnormalities or skin changes. Her menstrual history included menarche at 11 years and menopause at 52 years. She is multiparous and all of her children were breastfed. There was no history of use of oral contraceptive pills and no family history of breast or other cancers. Physical examination showed a hard mass in the left lower outer quadrant and palpable left axillary lymph nodes. There were no further relevant clinical findings and systemic examination was unremarkable.
Investigations
Laboratory investigations revealed hypochromic, microcytic anaemia with haemoglobin of 108 g/L as well as elevated CA15-3 (45.8 U/mL) and carcinoembryonic antigen (CEA) (8.2 µg/L) tumour markers. On mammography (figure 1), 2×1.7 cm spiculated opacity with specks of microcalcifications, suspicious for malignancy, was detected in the lower outer quadrant of the left breast. Ultrasonography (US) reported hypoechoic lesion measuring 1.2×1×1.4 cm in size with irregular infiltrated margins in the left breast, in keeping with neoplastic lesion, and suspicious ipsilateral axillary lymph nodes. US-guided core biopsy of the lesions revealed non-specific invasive ductal carcinoma (IDC) with axillary lymph node metastasis. Immunohistochemistry (IHC) examination revealed positive oestrogen receptor (ER) and progesterone receptor (PR). Human epidermal growth factor receptor (HER2-neu) was also positive and Ki-67 ratio was high (50%). CT scan of the thorax, abdomen and pelvis for staging of breast cancer revealed a well-defined lobulated mass measuring 2×2 cm in diameter with an irregular outline in the left lower breast parenchyma (figure 2) and multiple axillary nodes with short axis measuring 10 mm. In addition, a well-defined lobulated mass, 5×5 cm in diameter, with marked thickening of the colonic wall was found in the ascending colon, consistent with malignancy (figure 3). Isotope bone scintigraphy was negative for bony metastases. Colonoscopy was done and an ulcerative, non-obstructing mass in the ascending colon and pedunculated polyp in the descending colon were seen and biopsied. Histopathological examination of the biopsies revealed adenocarcinoma of the ascending colon and tubular adenoma with low-grade dysplasia in the descending colon. On IHC, the tumour showed preserved MutS homolog 2 protein (MSH-2), MutS homolog 6 protein (MSH-6), MutL homolog 1 protein (MLH-1) and Mismatch repair endonuclease gene (PMS-2). Our pathology department also performed ER, PR and HER2-neu IHC on the colon specimen to ensure that the colon adenocarcinoma represents a primary malignancy and not metastasis from breast carcinoma. The IHC results were negative.
Figure 1.
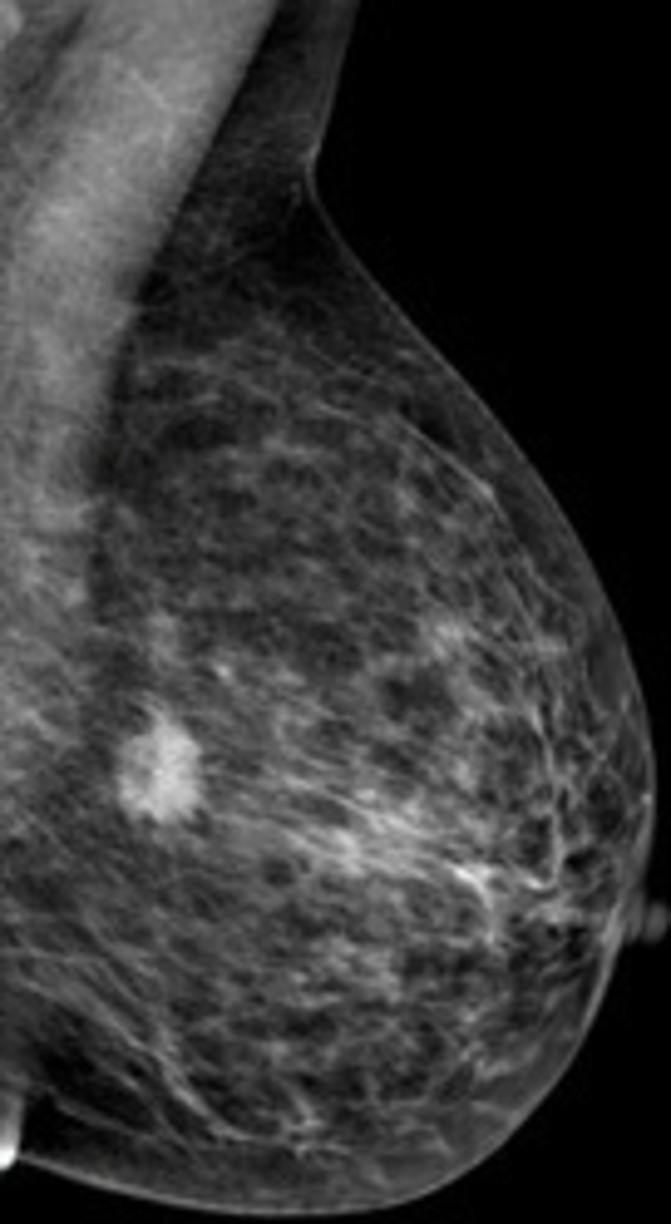
Mammogram showing suspicious spiculated opacity with specks of microcalcifications in the left breast.
Figure 2.

CT chest showing a mass in the lower parenchyma of the left breast.
Figure 3.

CT abdomen showing a mass in the ascending colon with thickening of the colonic wall.
Differential diagnosis
Following the above investigations, the diagnosis of synchronous breast and colon cancer was made.
Treatment
During an MDT tumour board, it was decided to first perform surgery for the breast cancer and then the hemicolectomy. In the first operation, the patient underwent left modified radical mastectomy (MRM). There were no complications in the early postoperative period. Two weeks after the first operation, the patient underwent laparoscopic right hemicolectomy. Histopathological examination of the breast and axilla specimens revealed grade II, 20 mm IDC with 6 out of 22 axillary lymph nodes showing metastatic carcinoma. The pathological stage was pT1N2Mx, that is, stage IIIA breast cancer. Examination of the colon segment revealed a moderately differentiated adenocarcinoma of 4×3.5×2.5 cm in size, with invasion of muscularis propria. There were 28 lymph nodes in the mesentery of the resected colonic segment, with no metastasis detected. The pathological stage was identified as pT2N0Mx (Dukes stage A), making this a stage I colon cancer.
Outcome and follow-up
After the two procedures, the patient did not develop postoperative complications and was referred to the oncology department for adjuvant therapy, as recommended by the MDT. She remains well 2 years following surgical treatment and has completed adjuvant radiation therapy and chemotherapy with ACTH (doxorubicin, cyclophosphamide, paclitaxel plus trastuzumab), without CT evidence of metastasis or disease recurrence. Endocrine therapy continues in the absence of radiological evidence of progresssion of breast cancer. The patient continues on a 6-monthly clinical and radiological follow-up in the outpatient clinic.
Discussion
The incidence of breast and colon cancer in women at the same time is 3.85%.3 We reported a case in which synchronous colon cancer was detected incidentally on CT scan for staging of breast cancer. Synchronous tumours have been reported to be encountered by CT and positron emission tomography (PET)-CT scans used in the staging and monitoring of malignancies.4 5
The clinical and pathological features of synchronous tumours of the breast and colon are not fully established and controversy exists as to the relationship between the two.6 There is correlation between family history and synchronous tumours.7 A specific genetic mutation, CHEK2*1100delC (CHEK2), has been described in patients with hereditary breast and colorectal cancer phenotype.8 It has been reported to be a low-penetrance breast cancer-predisposing gene associated with a threefold to fivefold increased risk of breast cancer.9 It has also been shown to confer a risk of colorectal cancer in patients with hereditary non-polyposis colorectal cancer (HNPCC).8 10 Its function by either conferring a high risk of one cancer type and a slightly elevated risk of the other or through a predisposition to one of the two cancers and chance occurrence of the other, may help to explain synchronous occurrence of breast and colon cancer.4 Perhaps in the future such genetic testing will become applicable to patients such as in our case. MSH-2, MSH-6, MLH-1 and PMS-2, which are associated with familial Lynch syndrome or HNPCC, were all preserved in the colonic specimen resected in our patient. However, our patient does not fulfil The Amsterdam II Criteria used to diagnose Lynch syndrome, and breast cancer is not included as one of the cancer subtypes found in HNPCC. In the absence of any family history, it is likely that the patient developed sporadic synchronous primary tumours.
Because of the lack of definitive guidelines for synchronous tumours, the management of each patient should be determined as the outcome of MDT cancer meetings.5 In our MDT cancer meeting, it was decided to proceed with MRM and then hemicolectomy, followed by adjuvant chemotherapy. The absence of colonic obstruction in our patient suggested that emergent surgery was not required for colon cancer. In fact, MRM is thought to have less morbidity and makes it possible to perform the hemicolectomy in a short time frame, such as in our case.6 The prognosis of synchronous tumours depends on each tumour stage independently. Compared with single tumours, synchronous tumours have no worse prognosis with effective management.5 It is important to consider what chemotherapy regimen should be given in the postoperative setting should both tumours require adjuvant chemotherapy, but the challenge is to find an anticancer therapy strategy that covers both cancer types without increased toxicity or relevant pharmacological interactions, and without a negative impact on the overall outcome.
In our MDT cancer meeting, it was decided to direct chemotherapy to the tumour with more advanced stage. However, in our case, only the breast cancer needed adjuvant chemotherapy in view of the lymph node status, HER2-neu status, tumour grade and possibly Ki-67 ratio, whereas the colon cancer had favourable pathological features with no lymph node metastases and warranted observation only.
Learning points.
Synchronous breast and colon cancers are rare, particularly in the absence of family history.
Synchronous tumors should be considered during the workup performed for the primary malignancy.
There are no definitive guidelines for the management of synchronous tumors. Therefore, treatment should be individualised for each patient through multidisciplinary team cancer meeting input in order to provide the best outcome.
If adjuvant therapy is required, it should be directed towards the tumor with the more advanced stage.
The prognosis of synchronous tumours depends on each tumour stage independently.
Footnotes
Contributors: HAA was involved in preparation of the manuscript. RA was the operating consultant surgical oncologist responsible for the patient’s care, who revised the manuscript and gave it final approval. AA was responsible for editing the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1. Demandante CG, Troyer DA, Miles TP. Multiple primary malignant neoplasms: case report and a comprehensive review of the literature. Am J Clin Oncol 2003;26:79–83. [DOI] [PubMed] [Google Scholar]
- 2. Bittorf B, Kessler H, Merkel S, et al. Multiple primary malignancies: an epidemiological and pedigree analysis of 57 patients with at least three tumours. Eur J Surg Oncol 2001;27:302–13. 10.1053/ejso.2001.1112 [DOI] [PubMed] [Google Scholar]
- 3. Fisher CS, Wachtel MS, Margenthaler JA. Outcomes for patients who develop both breast and colorectal cancer. Ann Surg Oncol 2012;19:242–8. 10.1245/s10434-011-1843-8 [DOI] [PubMed] [Google Scholar]
- 4. Karayiannakis AJ, Kakolyris S, Kouklakis G, et al. Synchronous breast and rectal cancers in a man. Case Rep Oncol;4:281–6. 10.1159/000328995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Yetkin G, Celayir F, Akgun IE, et al. Synchronous occurrence of primary breast carcinoma and primary colon adenocarcinoma. Case Rep Surg 2017;2017:1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Higgins L, Robertson I, Khan W, et al. Synchronous breast and colon cancer: factors determining treatment strategy. BMJ Case Rep 2013;2013 10.1136/bcr-2013-009450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kimura T, Iwagaki H, Fuchimoto S, et al. Synchronous colorectal carcinomas. Hepatogastroenterology 1994;41:409–12. [PubMed] [Google Scholar]
- 8. Meijers-Heijboer H, Wasielewski M, Wagner A, et al. The CHEK2 1100delC mutation identifies families with a hereditary breast and colorectal cancer phenotype. Am J Hum Genet 2003;72:1308–14. 10.1086/375121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Weischer M, Bojesen SE, Ellervik C, et al. CHEK2 *1100delC Genotyping for Clinical Assessment of Breast Cancer Risk: Meta-Analyses of 26,000 Patient Cases and 27,000 Controls. J Clin Oncol 2008;26:542–8. 10.1200/JCO.2007.12.5922 [DOI] [PubMed] [Google Scholar]
- 10. Wasielewski M, Vasen H, Wijnen J, et al. CHEK2 1100delC is a susceptibility allele for HNPCC-related colorectal cancer. Clin Cancer Res 2008;14:4989–94. 10.1158/1078-0432.CCR-08-0389 [DOI] [PubMed] [Google Scholar]


