Abstract
An important safety concern on cell-based gene therapy is that few methods have been available to control the proliferation and functioning of therapeutic protein-expressing cells after transplantation. We previously reported that the proliferation and functioning of the cells transfected with herpes simplex virus thymidine kinase (HSVtk) gene, a suicide gene, can be controlled by administration of ganciclovir. In this study, we tried to control the amount of murine interferon-γ (IFN-γ) secreted from transplanted murine mesenchymal stem cell line C3H10T1/2 cells to achieve safe cell-based IFN-γ gene therapy for cancer. C3H10T1/2 cells were transfected with HSVtk- and murine IFN-γ-expressing plasmid vectors to obtain C3H10T1/2/HSVtk/IFN-γ cells. C3H10T1/2/HSVtk/IFN-γ cells released IFN-γ and were sensitive to ganciclovir. C3H10T1/2/HSVtk/IFN-γ cells significantly suppressed the proliferation of murine adenocarcinoma cell line colon26 cells both in vitro and in vivo. Moreover, subcutaneous administration of ganciclovir to mice transplanted with NanoLuc luciferase-expressing C3H10T1/2/HSVtk cells for three consecutive days reduced the luminescence signals from the transplanted cells. These results indicate that the cell regulation system using HSVtk gene and ganciclovir can be useful for safe and efficient cell-based IFN-γ gene therapy for cancer.
Subject terms: Cancer therapy, Mesenchymal stem cells, Gene delivery, Genetic engineering
Introduction
Gene therapy for cancer is designed to transfer a gene encoding an antitumor protein or to transplant cells expressing such a gene to the patients1,2. This is an attractive therapeutic method as it can sustainably supply an antitumor protein for a long period with single gene transfer or cell transplantation. Unlike repeated administration in protein replacement therapy, this method can provide the stable and sustainable protein supply in the body. The first in vivo gene therapy was applied against the adenosine deaminase deficient patients in 1990. Since then, the therapeutic effects of in vivo gene therapy have been demonstrated for various diseases, e.g., Parkinson’s disease, Alzheimer’s disease, and cancers until now3–6. However, in vivo gene therapy has some crucial disadvantages to be solved. One of the most serious disadvantages is an unregulated protein synthesis because of gene overexpression, which can cause adverse effects. However, it is difficult to regulate the gene expression and protein synthesis in the body after gene transfer. Another disadvantage is serious immune responses, such as anaphylactic shock against the administered genes7,8.
Cell-based gene therapy, which is a therapeutic method to transplant genetically modified cells into patients, is another way that can sustainably supply a specific protein by single transplantation9. Studeny et al. showed that transplantation of human mesenchymal stem cells transduced with interferon-β gene into tumor-bearing mice resulted in a high plasma concentration of interferon-β and the excellent antitumor effects, compared to the injection of recombinant interferon-β protein10. Moreover, cell-based gene therapy does not need to directly transduce the gene into the body, and this reduces the risk of anaphylactic shock. However, even in cell-based gene therapy, it is difficult to regulate the amount of proteins secreted from the transplanted cells, as few methods have been developed to regulate the proliferation or functioning of the cells after transplantation11. Because the unregulated protein release from the transplanted cells may cause adverse effects, it is necessary to regulate the proliferation and/or functioning of the transplanted cells to achieve a safe cell-based gene therapy.
We previously developed a cell regulation system using cell suicide, which is the programmed cell death by apoptosis inducer, to control the proliferation and functioning of transplanted cells12. Cell suicide can cause the apoptosis of suicide gene-expressing cells in response to specific molecules, and we demonstrated that the proliferation and functioning of cells transfected with the herpes simplex thymidine kinase (HSVtk) gene, one of the suicide genes, could be regulated by administration of ganciclovir (GCV), a prodrug converted by HSVtk.
In this study, we tried to develop a safe cell-based interferon-γ (IFN-γ) gene therapy for cancer, by controlling the proliferation and functioning of the transplanted cells using the cell regulation system consisting of HSVtk gene and GCV. It has been demonstrated that IFN-γ has high antitumor activity in clinical use, and that the much higher therapeutic effects could be achieved by transplantation of IFN-γ-expressing cells into tumor-bearing mice, in some preclinical studies13. We first transfected the murine mesenchymal stem cell line C3H10T1/2 cells with HSVtk and IFN-γ genes to obtain the C3H10T1/2/HSVtk/IFN-γ cells. Then, we evaluated whether C3H10T1/2/HSVtk/IFN-γ cells could demonstrate antitumor effects against cultured cancer cells and in tumor-bearing mice. Furthermore, we also examined whether the proliferation of transplanted C3H10T1/2/HSVtk/IFN-γ cells could be regulated in mice by GCV administration, using C3H10T1/2/HSVtk cells expressing a reporter protein, NanoLuc luciferase (Nluc), for in vivo detection of the cells.
Results
Characteristics of C3H10T1/2/HSVtk/IFN-γ cells
C3H10T1/2 cells transfected with pCMV-HSVtk plasmid were selected with G418 and cloned. The cells with the highest GCV sensitivity were used for further experiments. C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells were established by transfection of C3H10T1/2 or C3H10T1/2/HSVtk cells with pEBM-IFN-γ plasmid, followed by the selection with hygromycin. Figure 1 shows the characteristics of the established C3H10T1/2/HSVtk/IFN-γ cells. To confirm the HSVtk gene specific DNA in C3H10T1/2/HSVtk cells, cDNA from cells was amplified by PCR using HSVtk specific primers. Bands of HSVtk-specific PCR products were detected in the pCMV-HSVtk plasmid (Fig. 1A, lane b) and C3H10T1/2/HSVtk cells (Fig. 1A, lane c), but not in the C3H10T1/2 cells (Fig. 1A, lane d). To confirm the expression of IFN-γ gene in these cells, the concentration of IFN-γ in the culture media of C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells was measured. C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells released a large amount of IFN-γ (Fig. 1B). When C3H10T1/2, C3H10T1/2/HSVtk, C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells were cultured with medium containing GCV for 4 days, the viability of C3H10T1/2/HSVtk and C3H10T1/2/HSVtk/IFN-γ cells decreased with an increasing concentration of GCV, while that of C3H10T1/2 and C3H10T1/2/IFN-γ cells did not change (Fig. 1C).
Figure 1.
Characteristics of C3H10T1/2/HSVtk/IFN-γ cells. (A) The HSVtk-specific bands of PCR products on agarose gel after electrophoresis. The 100 bp DNA ladder (lane a), pCMV-HSVtk plasmid (lane b), C3H10T1/2/HSVtk cells (lane c), and C3H10T1/2 cells (lane d) are shown. (B) IFN-γ secretion of C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells. Cells were cultured for 24 h and the supernatants were collected. The concentration of IFN-γ in the supernatant was measured by ELISA. Results are expressed as the mean ± SD of four samples. A representative of four independent experiments with similar results is shown. (C) The viability of C3H10T1/2/HSVtk or C3H10T1/2/HSVtk/IFN-γ cells cultured with GCV at various concentrations. These cells were cultured in medium containing various concentration of GCV for four days. C3H10T1/2 cells (white circle), C3H10T1/2/IFN-γ cells (white square), C3H10T1/2/HSVtk cells (black circle), and C3H10T1/2/HSVtk/IFN-γ cells (black square) are indicated. Results are expressed as the mean ± SD of three to four samples. *p < 0.05; statistically significant differences observed in comparison with C3H10T1/2 cells group.
Effects of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells on the proliferation of colon26/luc cells
To evaluate the effects of cell-derived IFN-γ on the viability of cancer cells, C3H10T1/2, C3H10T1/2/HSVtk, C3H10T1/2/IFN-γ, or C3H10T1/2/HSVtk/IFN-γ cells were cultured for 48 h, and the supernatants were collected as conditioned medium. These conditioned media were added to the firefly luciferase gene-expressing murine adenocarcinoma cell line colon26/luc cells and cultured for 48 h. The conditioned medium of C3H10T1/2 or C3H10T1/2/HSVtk cells hardly affected the viability of colon26/luc cells (Fig. 2A). On the other hand, the conditioned medium of C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells significantly reduced the viability of colon26/luc cells. Furthermore, C3H10T1/2, C3H10T1/2/HSVtk, C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells were co-cultured with colon26/luc cells for 48 h. It was observed that C3H10T1/2 or C3H10T1/2/HSVtk cells hardly affected the viability of colon26/luc cells, while C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells significantly reduced the viability of these cells (Fig. 2B).
Figure 2.
Effect of C3H10T1/2/IFN-γ cells or C3H10T1/2/HSVtk/IFN-γ cells on the proliferation of colon26/luc cells. (A) Viability of colon26/luc cells on incubation with the conditioned medium of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells. The conditioned media of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells were collected after 48 h incubation. The medium of colon26/luc cells was replaced with the conditioned media and the cells were cultured for 48 h. The viability of colon26/luc cells was calculated using CCK-8. Results are expressed as the mean ± SD of four samples. A representative of four independent experiments with similar results is shown. *p < 0.05; statistically significant differences observed in comparison with no treatment group. (B) Viability of colon26/luc cells after co-culture of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells with colon26/luc cells. C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells and colon26/luc cells were co-cultured for 48 h. The viability of colon26/luc cells was calculated by detecting the luminescence of colon26/luc cells. Results are expressed as the mean ± SD of four samples. A representative of ten independent experiments with similar results is shown. *p < 0.05; statistically significant differences observed in comparison with no treatment group.
Effect of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells on colon26/luc-tumor growth in mice
To confirm the effect of IFN-γ-expressing cells on tumor growth in mice, colon26/luc cells were mixed with C3H10T1/2, C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells, and transplanted into the back of mice. Figure 3 shows the time course of the tumor volume in mice after transplantation of colon26/luc cells. Co-administration of C3H10T1/2 cells significantly increased the colon26/luc tumor volume in mice. On the other hand, co-administration of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells almost completely suppressed the tumor growth. In addition, C3H10T1/2/HSVtk/IFN-γ cells significantly suppressed the tumor growth in mice with metastatic lung cancer of B16-Bl6/Nluc cells compared with C3H10T1/2 cells (Supplementary Fig. 1).
Figure 3.
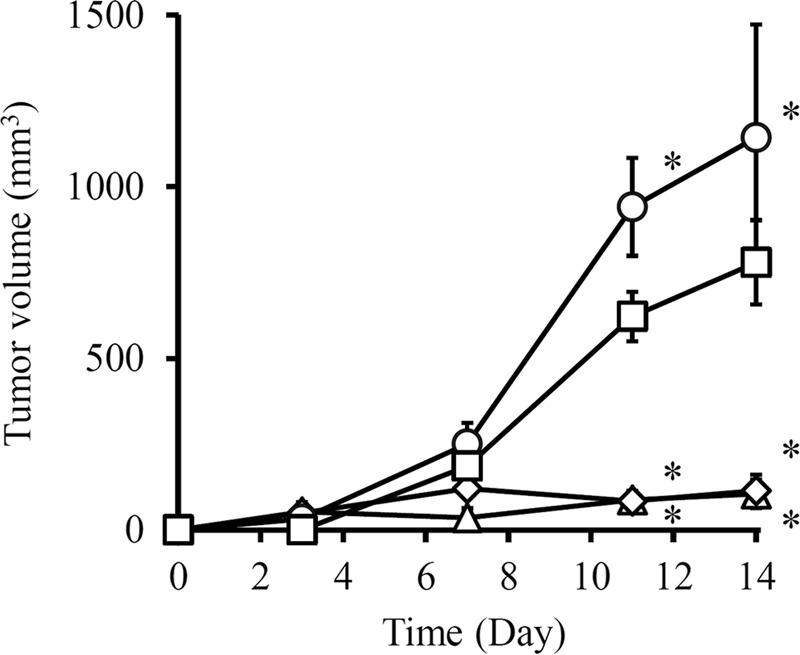
Effect of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells on tumor growth. Colon26/luc cells were mixed with C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells and transplanted into the back of BALB/c Slc-nu/nu mice. The tumor volume was measured by twice a week using a caliper. Colon26/luc cells (white square), colon26/luc cells and C3H10T1/2 cells (white circle), colon26/luc cells and C3H10T1/2/IFN-γ cells (white diamond), and colon26/luc cells and C3H10T1/2/HSVtk/IFN-γ cells (white triangle) are indicated. Results are expressed as the mean ± SD of five mice. A representative of two independent experiments with similar results is shown. *p < 0.05; statistically significant differences observed in comparison with colon26/luc cells group.
Effect of GCV on the proliferation of C3H10T1/2/HSVtk or C3H10T1/2/HSVtk/IFN-γ cells
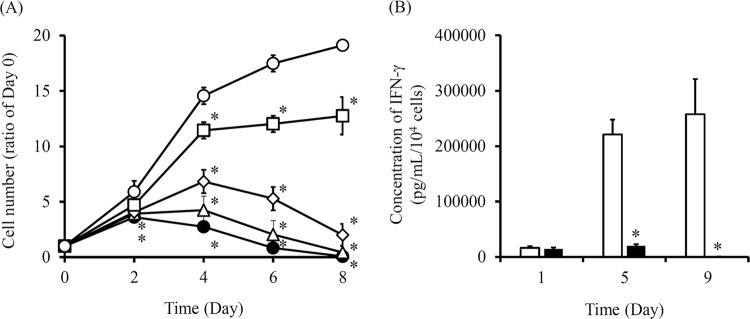
To examine the change in the proliferation and functioning of HSVtk/IFN-γ expressing cells by GCV, C3H10T1/2/HSVtk cells were cultured in media containing various concentrations of GCV, and the number of cells was measured every two days (Fig. 4A). The cell number ratio was calculated by setting the cell number of Day 0 as 1. The number of C3H10T1/2/HSVtk cells cultured in normal medium without GCV increased with time. On the other hands, the number of C3H10T1/2/HSVtk cells cultured in GCV containing medium decreased in a GCV concentration-dependent manner. Similarly, C3H10T1/2/HSVtk/IFN-γ cells were cultured in normal medium or media containing various concentration of GCV, and the IFN-γ concentration in the supernatant was measured (Fig. 4B). The IFN-γ concentration of C3H10T1/2/HSVtk/IFN-γ cells in normal medium increased with time. Conversely, IFN-γ concentration of C3H10T1/2/HSVtk/IFN-γ cells cultured in 0.25 μg/mL GCV containing medium was not detected by Day 8.
Figure 4.
Effect of GCV on the proliferation of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells. (A) The number of C3H10T1/2/HSVtk cells treated with GCV. C3H10T1/2/HSVtk cells were cultured in media containing 0.03125, 0.0625, 0.125 or 0.25 μg/mL GCV. No treatment (white circle), 0.03125 μg/mL GCV (white square), 0.0625 μg/mL GCV (white diamond), 0.125 μg/mL GCV (white triangle), and 0.25 μg/mL GCV (black circle) are indicated. Results are expressed as the mean ± SD of four samples. A representative of two independent experiments with similar results is shown. *p < 0.05; statistically significant differences observed in comparison with no treatment group. (B) The IFN-γ concentration in the supernatant of C3H10T1/2/HSVtk/IFN-γ cells cultured in normal medium or GCV containing medium. C3H10T1/2/HSVtk/IFN-γ cells were cultured in medium containing 0.25 μg/mL GCV. The medium was replaced with fresh medium containing various concentrations of GCV, and it was changed every 24 h and collected at Day 5 and Day 9. The IFN-γ concentration in medium was measured by ELISA. No treatment (white bar) and 0.25 μg/mL GCV containing medium (black bar) are indicated. Results are expressed as the mean ± SD of four samples. A representative of two independent experiments with similar results is shown. *p < 0.05; statistically significant differences observed in comparison with no treatment group.
Elimination of C3H10T1/2/HSVtk/Nluc cells from mice by GCV
Figure 5 shows the luminescent images of mice transplanted with C3H10T1/2/HSVtk/Nluc cells before and after GCV administration. First, we confirmed that the transplanted C3H10T1/2/HSVtk/Nluc cells into mice survived over four weeks (Supplementary Fig. 2). Then, C3H10T1/2/HSVtk/Nluc cells were transplanted into the back of mice, and 50 mg/kg GCV was subcutaneously administered for three consecutive days, from Day 7 after cell transplantation. In a previous report, we showed that the survival of cells without HSVtk gene was hardly affected by GCV administration12. Without GCV administration, the luminescent signal derived from C3H10T1/2/HSVtk/Nluc cells was detected in mice until Day 10 after transplantation. On the other hand, daily administration of 50 mg/kg GCV from Day 7 to Day 10 extinguished the luminescent signal from the cells. Furthermore, we also evaluated the IFN-γ amount in the skin of C3H10T1/2/HSVtk/IFN-γ cells-transplanted mice and luciferase activity in the skin of C3H10T1/2/Nluc/HSVtk cells-transplanted mice with or without GCV administration (Supplementary Fig. 3). The IFN-γ concentration in the skin of the C3H10T1/2/HSVtk/IFN-γ cell-transplanted, GCV-treated mice was significantly lower than that of the C3H10T1/2/HSVtk/IFN-γ cell-transplanted, GCV-untreated mice. The IFN-γ concentration in the skin of the C3H10T1/2/HSVtk/IFN-γ cell-transplanted mice was almost the same as that of the no treatment mice (42 ± 33 pg/mL). Similarly, the luciferase activity in the skin of the C3H10T1/2/Nluc/HSVtk cell-transplanted, GCV-treated mice was significantly lower than that of the C3H10T1/2/Nluc/HSVtk cell-transplanted, GCV-untreated mice.
Figure 5.
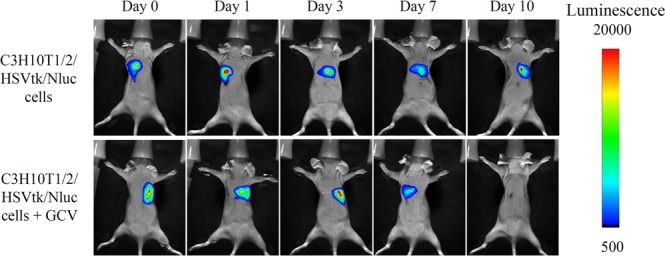
Elimination of C3H10T1/2/HSVtk/Nluc cells from mice by GCV. C3H10T1/2/HSVtk/Nluc cells were subcutaneously transplanted into the back of BALB/c Slc-nu/nu mice. GCV (50 mg/kg) was subcutaneously administered into mice for three consecutive days from Day 7 after cell transplantation. The luminescence of cells transplanted in mice was detected in an In-Vivo Xtreme Imaging System.
Evaluation of the level of creatinine, BUN, AST and ALT in plasma and body weight of mice after GCV administration
Repeated dosing of GCV (50 mg/kg, twice a day) for 10 days to C3H10T1/2/Nluc/HSVtk cells-transplanted mice hardly affected the plasma levels of creatinine, BUN, AST and ALT. In addition, the body weight of the mice was hardly changed by GCV administration (Supplementary Fig. 4).
Discussion
Many protein pharmaceutical products including IFN are clinically used in the treatment of various diseases and their effectiveness has also been confirmed14. However, the administration of these products by injection can be painful to the patients, and some products may require repeated administration because of their short in vivo half-life15. Thus, the patient’s quality of life is generally low. To reduce the dose frequency, pegylated proteins such as peginterferon and pegfilgrastim have been developed16,17. However, the activities of proteins may decrease in some cases, because PEG modification may reduce the binding affinity of proteins with receptors or may change the structure of proteins18. Hence, we thought that gene therapy, especially cell-based gene therapy, could solve the problem of protein pharmaceutical products. In this study, we first succeeded in the regulation of functioning of cell-based gene therapy using cell regulation system consisting of HSVtk gene and GCV12. When the adverse effects occur in patients because of the excessive protein secretion in cell-based gene therapy, the concentration of the therapeutic protein can be adequately regulated by the administration of GCV. Therefore, this cell regulation system is a mandatory step for the safe cell-based gene therapy.
Cell-based gene therapy for cancer has demonstrated an excellent antitumor effect in many reports. For example, TRAIL, interleukin (IL)-12, IL-27, cytosine deaminase, and oncolytic adenovirus are transduced into MSC to treat cancer. Therefore, the cell-based gene therapy can be applied to various types of cancers after selecting suitable anti-tumor cytokines13,19–25. We selected cancer and IFN-γ as the disease and the therapeutic molecule to demonstrate the usefulness of cell regulation system in cell-based gene therapy in this study. In addition, we used mesenchymal stem cell line C3H10T1/2 cells, because mesenchymal stem cells have been clinically used for many diseases in recent years26–29. Moreover, mesenchymal stem cells, among the other stem cells, are reported to rarely cause tumorigenesis30,31. Therefore, mesenchymal stem cells are safe for transplantation, and can be appropriate cells for cell-based gene therapy.
In Fig. 2A, colon26/luc cells showed reduced viability when cultured in the conditioned medium of C3H10T1/2/IFN-γ, compared to the conditioned medium of C3H10T1/2/HSVtk/IFN-γ cells. This is because the IFN-γ secretion from C3H10T1/2/IFN-γ cells was higher than that of C3H10T1/2/HSVtk/IFN-γ cells, suggesting that the cell viability is dependent on the concentration of IFN-γ. On the other hand, in co-culture experiments, the viability of colon26/luc cells was almost the same for C3H10T1/2/IFN-γ and C3H10T1/2/HSVtk/IFN-γ cells (Fig. 2B). This may be because co-culture of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells with colon26/luc cells had the higher antitumor effect than the conditioned medium. Probably because of effective delivery of IFN-γ in short distances between the cells, the different concentration of IFN-γ may have little effects on the viability of colon26/luc cells under the experimental conditions. Similar results were obtained in the in vivo antitumor experiment (Fig. 3).
Mesenchymal stem cells have been reported to show tumor supportive effects or tumor suppressive effects in some reports32. The tumor supportive effects of mesenchymal stem cells are caused by the suppression of immune response by secretion of immunosuppressive cytokines33–35. In contrast, the proliferation of malignant glioma cell lines was significantly inhibited by the treatment with human umbilical cord blood mesenchymal stem cells in vitro and in vivo. This is because the mesenchymal stem cells induced apoptosis in glioma cells by inhibition of the inhibitor of apoptosis protein36. Our results showed that co-injection of C3H10T1/2 cells increased the growth of colon26/luc tumor, suggesting that C3H10T1/2 cells may have tumor supportive effects. Further usefulness of C3H10T1/2/HSVtk/IFN-γ cells needs to be examined in tumor-bearing mice in future studies.
The apoptosis induction of HSVtk-expressing cells by GCV needs a few days, because the inhibition of DNA synthesis by phosphorylated GCV requires some time to induce cell apoptosis as previously reported37,38. Our result showed 0.25 μg/mL GCV significantly suppressed the cell proliferation on Day 4 compared to untreated cells, and completely eliminated the cells on Day 8 (Fig. 4A). The results were consistent with the IFN-γ secretion in C3H10T1/2/HSVtk/IFN-γ cells (Fig. 4B). Similarly, our previous study showed that HSVtk-expressing mouse pancreatic beta cell line MIN6 (MIN6/HSVtk) cells also needed a few days to completely undergo apoptotic cell death by GCV12. It is reported that the required time of apoptosis induction in cell suicide varies depending on the type of suicide genes39. Because the number of transplanted cells in cell-based gene therapy directly correlates with the therapeutic protein level in the systemic circulation, the type of suicide gene, i.e., the required time of cell apoptosis, should be selected depending on the disease. In addition, the amount of GCV for cell apoptosis induction is important to regulate the cell proliferation. Figure 5 shows that transplanted C3H10T1/2/HSVtk/Nluc cells in mice disappeared by administration of 50 mg/kg GCV for three consecutive days. Our previous study showed that 100 mg/kg GCV administration completely removed the MIN6/HSVtk/Nluc cells from mice, whereas the cell number remained constant after daily 50 mg/kg GCV administration12. From these results, the required amount of GCV for apoptosis induction might depend on the expression level of HSVtk gene or the type of cells. Further studies are needed to estimate the required amount of GCV for the in vivo cell regulation.
In conclusion, cell-based IFN-γ gene therapy for cancer with the cell regulation system using HSVtk gene and GCV modulated the amount of IFN-γ secreted from genetically modified cells. Furthermore, the transplanted cells could be removed from the mice by GCV administration, indicating that this cell regulation system is essential for the safe and efficient cell-based IFN-γ gene therapy for cancer.
Materials and Methods
Animals
Male BALB/c Slc-nu/nu mice (5 weeks old) were purchased from Japan SLC, Inc. (Shizuoka, Japan) and maintained under specific pathogen-free conditions. The protocols for experiments involving animals were approved by the Institutional Animal Experimentation Committee of Kyoto Pharmaceutical University and Tokyo University of Science. All experiments involving animals were conducted in accordance with the procedures outlined in the National Instituted of Health Guide for the Care and Use of Laboratory Animals.
Materials
The pSelect-zeo-HSV1tk plasmid, G418, Hygromycin B Gold, LB Broth Base, and LB Agar were purchased from InvivoGen Co. (San Diego, CA, USA). Lipofectamine 3000 was purchased from Invitrogen Co. (Carlsbad, CA, USA). pLVSIN-CMV-Neo plasmid, and Lentiviral High Titer Packaging Mix were purchased from Takara Bio Inc. (Shiga, Japan). KOD-Plus-Neo was purchased from TOYOBO Co., Ltd. (Osaka, Japan). Competent NEB 10-beta E. coli (High efficiency), SOC medium, KpnI-HF, EcoRI-HF, and NotI-HF were purchased from New England Biolabs Inc. (Ipswich, MA, USA). Ganciclovir hydrate was purchased from Tokyo Kasei Kogyo Co., Ltd. (Tokyo, Japan). Hank’s Balanced Salt solution was purchased from Sigma-Aldrich (St. Louis, MO, USA). Chloroform, isopropyl alcohol, ethanol, NaHCO3, D-(+)-glucose, 0.4 w/v% trypan blue solution, and pEBMulti-Hyg plasmid were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Fetal bovine serum was purchased from Biosera (East Sussex, UK) or Thermo Fisher Scientific (Waltham, MA, USA). Dulbecco’s Modified Eagle’s medium (DMEM), antibiotic-antimycotic mixed stock solution, and penicillin-streptomycin-glutamine mixed solution were purchased from Nacalai Tesque, Inc. (Kyoto, Japan) or Nissui Seiyaku (Tokyo, Japan). All the other chemicals used were of the highest grade available commercially.
Cell culture
Dr. Hiroki Kagawa (Department of Cell Biology, Kyoto Pharmaceutical University, Kyoto, Japan) kindly provided the murine mesenchymal stem cell line C3H10T1/2 cells. C3H10T1/2 cells were cultured in DMEM supplemented with 15% heat-inactivated fetal bovine serum and antibiotic-antimycotic mixed stock solution or penicillin-streptomycin-glutamine mixed solution at 37 °C in humidified air containing 5% CO2. Colon26 cells (colon26/luc cells) were cultured in RPMI supplemented with 10% heat-inactivated fetal bovine serum and antibiotic-antimycotic mixed stock solution or penicillin-streptomycin-glutamine mixed solution at 37 °C in humidified air containing 5% CO2. Lenti-X 293 T cells were purchased from Takara Bio Inc. (Shiga, Japan) and cultured in DMEM supplemented with 10% heat-inactivated FBS, penicillin-streptomycin-glutamine mixed solution, and 1 mM Sodium Pyruvate Solution (100×) at 37 °C in humidified air containing 5% CO2. NanoLuc Luciferase-expressing mouse melanoma B16-BL6 cells (B16-Bl6/Nluc cells) were cultured in DMEM supplemented with 10% heat-inactivated fetal bovine serum and penicillin-streptomycin-glutamine mixed solution at 37 °C in humidified air containing 5% CO2.
Construction of plasmids
To construct a plasmid coding IFN-γ gene (pEBMulti-IFN-γ), the fragment of IFN-γ gene was amplified by polymerase chain reaction (PCR) using primers (forward: 5′-CGGGGTACCATGAACGCTACACACT-3′ and reverse: 5′- TAAAGCGGCCGCTCAGCAGCGACT-3′). Subsequently, the amplified PCR product and pEBMulti-hyg vector were digested using KpnI-HF and NotI-HF restriction enzymes overnight at 37 °C. Ligation and cloning in E. coli was performed as previously reported12. Separately, to construct an HSVtk-encoding lentiviral plasmid, pLVSIN-HSVtk, the cDNA fragment of the HSVtk gene was amplified by PCR using primers (forward: 5′-CCGGAATTCATGGCTTCTTACCCTG-3′ and reverse: 5′-TAAAGCGGCCGCTTAGTTGGCCTCT-3′) from the pSelect-zeo-HSV1tk plasmid. Subsequently, the amplified PCR product and pLVSIN-CMV-Neo plasmid were digested using EcoRI-HF and NotI-HF restriction enzymes overnight at 37 °C. Ligation and cloning were performed as described above. pCMV-HSVtk and pEBMulti-NanoLuc luciferase (Nluc) plasmids were prepared in our previous study12.
Establishment of the gene-expressing C3H10T1/2 cells
HSVtk-expressing C3H10T1/2 cells were established as previously reported12. Briefly, C3H10T1/2 cells (1 × 105 cells) were seeded in a 24-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2. C3H10T1/2 cells were transfected with pCMV-HSVtk plasmid using Lipofectamine 3000 in Opti-MEM. After 24 h, the medium was replaced and cultured in a normal culture medium for a day. Then, these cells were cultured in medium containing 1500 μg/mL G418 for 5 days, and cloned and incubated until they were confluent. Cloned cells, which showed the induction of apoptosis by GCV were selected as the C3H10T1/2/HSVtk cells. To establish IFN-γ or Nluc-expressing C3H10T1/2 (C3H10T1/2/IFN-γ or C3H10T1/2/Nluc) cells and IFN-γ or Nluc-expressing C3H10T1/2/HSVtk (C3H10T1/2/HSVtk/IFN-γ or C3H10T1/2/HSVtk/Nluc) cells, C3H10T1/2 cells or C3H10T1/2/HSVtk cells (1 × 105 cells) were seeded in a 24-well culture plate and transfected with pEBMutli-IFN-γ plasmid or pEBMulti-Nluc plasmid using Lipofectamine 3000 as described above. After these cells were cultured in medium for a day, pEBMulti-IFN-γ or pEBMulti-Nluc-transfected C3H10T1/2 cells were cultured in medium containing 500 μg/mL hygromycin and pEBMulti-IFN-γ- or pEBMulti-Nluc-transfected C3H10T1/2/HSVtk cells were cultured in medium containing 300 μg/mL hygromycin. The expression of IFN-γ in cells was confirmed by measuring IFN-γ in the cell culture medium using an enzyme-linked immunosorbent assay (ELISA) kit (BioLegend, San Diego, CA, USA). The expression of Nluc in cells was confirmed by detecting the luminescence using a Nano-Glo assay reagent (Promega Co., Tokyo, Japan).
Lenti-X 293 T cells (1 × 107 cells) were seeded in a 10 cm culture dish and cultured overnight at 37 °C in humidified air containing 5% CO2. Lenti-X 293 T cells were co-transfected with pLVSIN-HSVtk plasmid and Lentiviral High Titer Packaging Mix by the calcium phosphate method. After 24 h, the medium was replaced with culture medium containing 10 μM forskolin, and cells were cultured for an additional 48 h. Then, the supernatant was collected and centrifuged at 900 g for 5 min. Furthermore, the supernatant was filtered, mixed with PEG solution (32 w/v% polyethylene glycol #6000, 400 mM NaCl, and 40 mM HEPES), and centrifuged at 2,500 g for 40 min. The precipitated viruses were collected and suspended by Opti-MEM. Then, C3H10T1/2/Nluc cells (1 × 105 cells) were seeded in a 12-well culture plate and transduced by the suspended virus. The medium was replaced after 24 h, and the cells were cultured in normal culture medium for three days, followed by 5-day culture in medium containing 1500 μg/mL G418. These cells were cloned and selected as C3H10T1/2/Nluc/HSVtk cells, which underwent apoptosis by GCV.
Confirmation of the HSVtk gene-specific DNA
Detection of the HSVtk gene-specific DNA in cells was performed using the protocol of our previous research12. Briefly, RNA was extracted from C3H10T1/2 or C3H10T1/2/HSVtk cells using Sepasol RNA I Super G (Nacalai Tesque, Inc.). The obtained RNA was converted to cDNA using ReverTra Ace qPCR RT Master Mix with gDNA Remover (TOYOBO Co., Ltd.). The HSVtk-specific PCR fragment in each cDNA was amplified by PCR using HSVtk specific primers (forward: 5′-AACATCTACACCACCCAGCAC-3′ and reverse: 5′-GAACAGCATCAGTCACAGCATAG-3′). The PCR products were separated on a 3% agarose gel and stained with ethidium bromide. The stained bands on the agarose gel were detected using a LAS 4000 mini imaging system (Fujifilm Co., Ltd., Tokyo, Japan).
IFN-γ secretion
C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ (1 × 105 cells) cells were seeded in a 12-well culture plate and cultured for 24 h at 37 °C in humidified air containing 5% CO2. The culture media were collected and centrifuged at 800 × g for 3 min. The concentration of IFN-γ secreted from C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells in the supernatant was measured using an IFN-γ ELISA kit.
Sensitivity of HSVtk-expressing C3H10T1/2 cells to GCV
C3H10T1/2, C3H10T1/2/HSVtk, C3H10T1/2/IFN-γ, and C3H10T1/2/HSVtk/IFN-γ cells (5 × 103 cells) were seeded in a 12-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2. Next day, the medium was replaced with fresh medium containing various concentrations of GCV. After four days, the number of viable cells was counted by using trypan blue exclusion test.
Effects of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells on the proliferation of colon26/luc cells
C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells (1 × 105 cells) were seeded in a 12-well culture plate and cultured for 48 h at 37 °C in humidified air containing 5% CO2. The supernatants were collected and centrifuged at 800 × g for 3 min (conditioned medium). Colon26/luc cells (5 × 103 cells) were seeded in a 96-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2. Next day, the culture medium of colon26/luc cells was replaced with the conditioned medium of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ. The cells were cultured for 48 h and the number of cells was measured by the Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan).
Additionally, colon26/luc cells (2.5 × 104 cells) were seeded in a 12-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2. Next day, C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells (2 × 105 cells) were co-cultured with colon26/luc cells for 48 h at 37 °C in humidified air containing 5% CO2. These cells were collected and lysed using lysis buffer (20 mM Tris-HCl, 200 mM NaCl, 2.5 mM MgCl2, 0.05 w/v% NP-40, pH 7.4). Total 20 μL of these cell lysates were mixed with 10 μL of Picagene kit (Toyo Inki, Tokyo, Japan) and the luminescence was measured with EnVision multi-label plate reader (Perkin-Elmer, Wellesley, MA, USA). The viability of colon26/luc cells was calculated as 100% of the luminescence of the colon26/luc cells.
Effect of C3H10T1/2/IFN-γ or C3H10T1/2/HSVtk/IFN-γ cells on tumor growth
Colon26/luc cells (2.5 × 105 cells) were mixed with or without C3H10T1/2 (2 × 106 cells), C3H10T1/2/IFN-γ (2 × 106 cells) or C3H10T1/2/HSVtk/IFN-γ cells (5 × 106 cells) and transplanted into the back of BALB/c Slc-nu/nu mice. The tumor diameters in mice were measured by twice a week using a caliper. The tumor volumes were calculated from the following formula: (large diameter) × (small diameter)2 × 0.5236. B16-Bl6/Nluc cells (2.5 × 105 cells) were intravenously inoculated, and C3H10T1/2 or C3H10T1/2/HSVtk/IFN-γ cells (5 × 106 cells) were intravenously administrated 2 h after B16-Bl6/Nluc cell inoculation. After 7 days, the lungs were removed from mice and homogenized in a lysis buffer using a homogenizer. Then, the homogenate was centrifuged at 10,000 × gfor 10 min at 4 °C. The luciferase activity in the supernatant was evaluated using a Nano-Glo assay reagent.
Effect of GCV on the proliferation of C3H10T1/2/HSVtk cells
C3H10T1/2/HSVtk cells (1 × 103 cells) were seeded in a 96-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2, and the number of cells was measured by Cell Counting Kit-8 (CCK-8, Dojindo Laboratories). The cells were next cultured in medium containing various concentrations of GCV and the number of cells was repeatedly measured every 2 days by using CCK-8.
Effect of GCV on the proliferation of C3H10T1/2/HSVtk/IFN-γ cells
C3H10T1/2/HSVtk/IFN-γ cells (5 × 104 cells) were seeded in a 24-well culture plate and cultured overnight at 37 °C in humidified air containing 5% CO2, and the medium was collected and centrifuged at 800 × g for 3 min. The medium was replaced with fresh medium containing various concentrations of GCV, which was replaced every 24 h and collected at Day 5 and Day 9. The concentration of IFN-γ in the supernatant was measured using an IFN-γ ELISA kit.
Evaluation of the survival of transplanted C3H10T1/2/HSVtk/Nluc cells in mice
C3H10T1/2/HSVtk/Nluc cells (1 × 106 cells) were mixed with Matrigel and transplanted into the back of BALB/c Slc-nu/nu mice. The blood was collected from the facial vein with a heparinized tube and Goldnord animal lancet (MEDIpoint, Inc., Mineola, NY, USA) two or three times a week for four weeks.
Transplantation of C3H10T1/2/HSVtk/Nluc cells and their elimination by GCV
C3H10T1/2/HSVtk/Nluc cells (1 × 106 cells) were mixed with Matrigel (BD Biosciences, San Diego, CA, USA) to improve the survival of cells in transplantation. The cells were transplanted into the back of BALB/c Slc-nu/nu mice. Seven days after cell transplantation, 50 mg/kg GCV was subcutaneously administered to mice for three consecutive days. The luminescence of cells in mice was detected using an In-Vivo Xtreme Imaging System (Bruker, Billerica, MA, USA) after injection of Nano-Glo assay reagent into the site of cell transplantation at a dose of 50 μL/mouse.
Measurement of IFN-γ and luciferase after inoculation of C3H10T1/2/HSVtk/IFN-γ cells and C3H10T1/2/Nluc/HSVtk cells, respectively, in mice skin
C3H10T1/2/HSVtk/IFN-γ cells (5 × 106 cells) or C3H10T1/2/Nluc/HSVtk cells (1 × 106 cells) were transplanted into the back of BALB/c Slc-nu/nu mice. GCV was subcutaneously administered at a dose of 50 mg/kg soon after transplantation and every 12 h subsequently for ten consecutive days. The skin tissue around the transplantation site of C3H10T1/2/HSVtk/IFN-γ cells was cut off 12 h after the last GCV administration and homogenized in a lysis buffer using a homogenizer (Microtec, Chiba, Japan). Then, the homogenate was centrifuged at 10,000 g for 10 min at 4 °C. The IFN-γ concentration in the supernatant was evaluated using an IFN-γ ELISA kit. Likely, the skin tissue around the cell transplantation site of C3H10T1/2/Nluc/HSVtk cells was cut off 36 h after the last GCV administration and homogenized as above. Then, the homogenate was centrifuged at 10,000 g for 10 min at 4 °C. The luciferase activity in the supernatant was evaluated using Nano-Glo assay reagent (Promega).
Evaluation of the level of creatinine, BUN, AST and ALT in plasma, and body weight of mice after GCV administration
After 10 consecutive days of GCV administration (50 mg/kg, twice a day) to C3H10T1/2/Nluc/HSVtk cells-transplanted mice, blood was collected from the postcaval vein. The plasma was collected from the blood by centrifugation for 5 min at 13,000 × g at 4 °C. Creatinine, blood urea nitrogen (BUN), aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities in plasma were measured using Labassay Creatinine (FUJIFILM, Wako Pure Chemical Industries, Ltd., Osaka, Japan), QuantiChrom Urea Assay Kit (Hayward, CA, USA) and transaminase CII test Wako (FUJIFILM, Wako Pure Chemical Industries, Ltd., Osaka, Japan). In addition, the body weight of mice was measured every other day.
Statistical analysis
Statistical differences were evaluated by one-way analysis of variance (ANOVA) followed by the Dunnett’s test for multiple comparisons or the Student’s t-test for two groups. p-values that were < 0.05 were considered statistically significant.
Supplementary information
Acknowledgements
The authors are grateful to Dr. Yuki Takahashi (Kyoto University, Kyoto, Japan) and Dr. Mitsuru Ando (Kyoto University, Kyoto, Japan) for their advice regarding establishment of HSVtk gene-expressing cells. This work was supported in part by a Grant-in-Aid for Young Scientists B (No. 17K15437) from the Japan Society for the Promotion of Science (JSPS) and by a grant from the Uehara Memorial Foundation.
Author contributions
M.T., K.K. and M.N. conceived and designed the experiments, analyzed the data, and contributed to writing and editing the manuscript. H.K., T.S. and A.Y. analyzed the data and contributed to reviewing and editing the manuscript.
Data availability
All data used for this research are publicly available.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-019-55269-6.
References
- 1.Dunbar CE, et al. Gene therapy comes of age. Science. 2017;359:6372. doi: 10.1126/science.aan4672. [DOI] [PubMed] [Google Scholar]
- 2.Naldini L. Gene therapy returns to centre stage. Nature. 2015;526:351–360. doi: 10.1038/nature15818. [DOI] [PubMed] [Google Scholar]
- 3.Blaese RM, et al. T lymphocyte-directed gene therapy for ADA- SCID: initial trial results after 4 years. Science. 1995;270:475–480. doi: 10.1126/science.270.5235.475. [DOI] [PubMed] [Google Scholar]
- 4.Bartus RT, Weinberg MS, Samulski RJ. Parkinson’s disease gene therapy: success by design meets failure by efficacy. Mol Ther. 2014;22:487–497. doi: 10.1038/mt.2013.281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li Y, Wang J, Zhang S, Liu Z. Neprilysin gene transfer: A promising therapeutic approach for Alzheimer’s disease. J Neurosci Res. 2015;93:1325–1329. doi: 10.1002/jnr.23564. [DOI] [PubMed] [Google Scholar]
- 6.Chiocca EA, et al. A phase I trial of Ad.hIFN-beta gene therapy for glioma. Mol Ther. 2008;16:618–626. doi: 10.1038/sj.mt.6300396. [DOI] [PubMed] [Google Scholar]
- 7.Vannucci L, et al. Viral vectors: a look back and ahead on gene transfer technology. New Microbiol. 2013;36:1–22. [PubMed] [Google Scholar]
- 8.Okada T, Takeda S. Current challenges and future directions in recombinant AAV-mediated gene therapy of Duchenne muscular dystrophy. Pharmaceuticals (Basel). 2013;6:813–836. doi: 10.3390/ph6070813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Orive G, et al. Long-term expression of erythropoietin from myoblasts immobilized in biocompatible and neovascularized microcapsules. Mol Ther. 2015;12:283–289. doi: 10.1016/j.ymthe.2005.04.002. [DOI] [PubMed] [Google Scholar]
- 10.Studeny M, et al. Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J Natl Cancer Inst. 2004;96:1593–1603. doi: 10.1093/jnci/djh299. [DOI] [PubMed] [Google Scholar]
- 11.Omer A, et al. Exercise induces hypoglycemia in rats with islet transplantation. Diabetes. 2004;53:360–365. doi: 10.2337/diabetes.53.2.360. [DOI] [PubMed] [Google Scholar]
- 12.Tsujimura M, et al. Regulation of proliferation and functioning of transplanted cells by using herpes simplex virus thymidine kinase gene in mice. J Control Release. 2018;275:78–84. doi: 10.1016/j.jconrel.2018.02.018. [DOI] [PubMed] [Google Scholar]
- 13.Sivanathan KN, et al. Interferon-gamma modification of mesenchymal stem cells: implications of autologous and allogeneic mesenchymal stem cell therapy in allotransplantation. Stem Cell Rev. 2014;10:351–375. doi: 10.1007/s12015-014-9495-2. [DOI] [PubMed] [Google Scholar]
- 14.George PM, et al. Pharmacology and therapeutic potential of interferons. Pharmacol Ther. 2012;135:44–53. doi: 10.1016/j.pharmthera.2012.03.006. [DOI] [PubMed] [Google Scholar]
- 15.Foon KA, et al. A phase I trial of recombinant gamma interferon in patients with cancer. Cancer Immunol Immunother. 1985;20:193–197. doi: 10.1007/BF00205575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zeuzem S, Welsch C, Herrmann E. Pharmacokinetics of peginterferons. Semin Liver Dis. 2003;23:23–28. doi: 10.1055/s-2003-41631. [DOI] [PubMed] [Google Scholar]
- 17.Cooper KL, et al. Granulocyte colony-stimulating factors for febrile neutropenia prophylaxis following chemotherapy: systematic review and meta-analysis. BMC Cancer. 2011;23:404. doi: 10.1186/1471-2407-11-404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pasut G, Veronese FM. Polymer–drug conjugation, recent achievements and general strategies. Prog Polym Sci. 2007;32:991–1007. doi: 10.1016/j.progpolymsci.2007.05.008. [DOI] [Google Scholar]
- 19.Uchibori R, Tsukahara T, Ohmine K, Ozawa K. Cancer gene therapy using mesenchymal stem cells. Int J Hematol. 2014;99:377–382. doi: 10.1007/s12185-014-1537-7. [DOI] [PubMed] [Google Scholar]
- 20.Li Z, Fan D, Xiong D. Mesenchymal stem cells as delivery vectors for anti-tumor therapy. Stem Cell Investig. 2015;26:2–6. doi: 10.3978/j.issn.2306-9759.2015.03.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ren C, et al. Cancer gene therapy using mesenchymal stem cells expressing interferon-beta in a mouse prostate cancer lung metastasis model. Gene Ther. 2008;15:1446–1453. doi: 10.1038/gt.2008.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hisada M, et al. Potent antitumor activity of interleukin-27. Cancer Res. 2004;64:1152–1156. doi: 10.1158/0008-5472.CAN-03-2084. [DOI] [PubMed] [Google Scholar]
- 23.Sasportas LS, et al. Assessment of therapeutic efficacy and fate of engineered human mesenchymal stem cells for cancer therapy. Proc Natl Acad Sci USA. 2009;106:4822–4827. doi: 10.1073/pnas.0806647106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Loebinger MR, et al. Mesenchymal stem cell delivery of TRAIL can eliminate metastatic cancer. Cancer Res. 2009;69:4134–42. doi: 10.1158/0008-5472.CAN-08-4698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jeong KY, et al. Irradiation-induced localization of IL-12-expressing mesenchymal stem cells to enhance the curative effect in murine metastatic hepatoma. Int J Cancer. 2015;137:721–730. doi: 10.1002/ijc.29428. [DOI] [PubMed] [Google Scholar]
- 26.Kebriaei P, et al. Adult human mesenchymal stem cells added to corticosteroid therapy for the treatment of acute graft-versus-host disease. Biol. Blood Marrow Transplant. 2009;15:804–811. doi: 10.1016/j.bbmt.2008.03.012. [DOI] [PubMed] [Google Scholar]
- 27.Sun L, et al. Umbilical cord mesenchymal stem cell transplantation in severe and refractory systemic lupus erythematosus. Arthritis Rheum. 2010;62:2467–2475. doi: 10.1002/art.27548. [DOI] [PubMed] [Google Scholar]
- 28.Jiang R, et al. Transplantation of placenta-derived mesenchymal stem cells in type 2 diabetes: a pilot study. Front Med. 2011;5:94–100. doi: 10.1007/s11684-011-0116-z. [DOI] [PubMed] [Google Scholar]
- 29.Kharaziha P, et al. Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: a phase I-II clinical trial. Eur J Gastroenterol Hepatol. 2009;21:1199–1205. doi: 10.1097/MEG.0b013e32832a1f6c. [DOI] [PubMed] [Google Scholar]
- 30.Prockop DJ, et al. Defining the risks of mesenchymal stromal cell therapy. Cytotherapy. 2010;12:576–578. doi: 10.3109/14653249.2010.507330. [DOI] [PubMed] [Google Scholar]
- 31.Lalu MM, et al. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoS One. 2012;7:e47559. doi: 10.1371/journal.pone.0047559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rhee KJ, Lee JI, Eom YW. Mesenchymal stem cell-mediated effects of tumor support or suppression. Int J Mol Sci. 2015;16:30015–30033. doi: 10.3390/ijms161226215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ren G, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2:141–150. doi: 10.1016/j.stem.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 34.Sato K, et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood. 2007;109:228–234. doi: 10.1182/blood-2006-02-002246. [DOI] [PubMed] [Google Scholar]
- 35.Jiang XX, et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood. 2005;105:4120–4126. doi: 10.1182/blood-2004-02-0586. [DOI] [PubMed] [Google Scholar]
- 36.Dasari VR, et al. Cord blood stem cell-mediated induction of apoptosis in glioma downregulates X-linked inhibitor of apoptosis protein (XIAP) PLoS One. 2010;5:e11813. doi: 10.1371/journal.pone.0011813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Duarte S, et al. Suicide gene therapy in cancer: where do we stand now? Cancer Lett. 2012;324:160–170. doi: 10.1016/j.canlet.2012.05.023. [DOI] [PubMed] [Google Scholar]
- 38.Aghi M, Hochberg F, Breakefield XO. Prodrug activation enzymes in cancer gene therapy. J Gene Med. 2000;2:148–164. doi: 10.1002/(SICI)1521-2254(200005/06)2:3<148::AID-JGM105>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 39.Marin V, et al. Comparison of different suicide-gene strategies for the safety improvement of genetically manipulated T cells. Hum Gene Ther Methods. 2012;23:376–386. doi: 10.1089/hgtb.2012.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data used for this research are publicly available.