Abstract
It has been demonstrated that the K+-dependent Na+/Ca2+ exchanger, NCKX2, is a new promising stroke neuroprotective target. However, because no pharmacological activator of NCKX2 is still available, microRNA (miRNA) may represent an alternative method to modulate NCKX2 expression. In particular, by bioinformatics analysis, miR-223-5p emerged as a possible modulator of NCKX2 expression. In the light of these premises, the aims of the present study were: (1) to evaluate miR-223-5p and NCKX2 expression in the temporoparietal cortex and striatum of rats subjected to transient middle cerebral artery occlusion; (2) to evaluate whether miR-223-5p targets the 3′ UTR of the NCKX2 transcript; and (3) to evaluate the effect of miR-223-5p modulation on brain ischemic volume and neurological deficits. Our results showed that miR-223-5p expression increased in a time-dependent manner in the striatum of ischemic rats in parallel with NCKX2 downregulation, and that the transfection of cortical neurons with miR-223-5p induced a reduction of NCKX2 expression. Moreover, a luciferase assay showed that miR-223-5p specifically interacts with the NCKX2 3′ UTR subregion (+7037 to +8697), thus repressing NCKX2 translation. More interestingly, intracerebroventricular infusion of anti-miR-223-5p prevented NCKX2 downregulation after ischemia, thus promoting neuroprotection. The present findings support the idea that blocking miR-223-5p by antimiRNA is a reasonable strategy to reduce the neurodetrimental effect induced by NCKX2 downregulation during brain ischemia.
Introduction
MicroRNAs (miRNAs) are non-coding short RNA molecules of 18–25 nt present in eukaryotic cells that target mRNAs and favor their degradation or prevent their translation.1,2 miRNA-based strategies, using miRNA mimics or antagomirs, have recently emerged as a promising therapeutic approach for several diseases.3 Indeed, owing to their small size, relative ease of delivery, sequence specificity in recognizing their targets, and multitarget properties, miRNAs represent promising therapeutic options for several CNS disorders such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, and cerebral ischemia.4 During ischemia, a deregulation of sodium and calcium homeostasis occurs. Indeed, transporters involved in the control of ionic homeostasis exert a pivotal role in the progression of the ischemic damage.5,6 Among them, we have previously demonstrated that the member 2 of the K+-dependent Na+/Ca2+ exchanger family, NCKX2, is a new potential target to be investigated in the study of the molecular mechanisms involved in the progression of cerebral ischemic damage.7 In fact, disruption of the NCKX2 gene by genetic manipulation renders neurons more susceptible to the ischemic insult, and both knocking down and knocking out NCKX2 expression increase the extent of the ischemic lesion in rats and mice subjected to permanent and temporal middle cerebral artery occlusion (pMCAO and tMCAO, respectively).7
Until now, no pharmacological activator of NCKX2 is available. miRNA may represent an alternative method to modulate NCKX2 expression in the attempt to ameliorate ischemic damage progression. In particular, by bioinformatics approaches, miR-223-5p emerged as a possible candidate modulator of NCKX2 expression. In the light of these premises, the specific aims of the present study were (1) to evaluate miR-223-5p expression in the temporoparietal cortex and striatum of ischemic rats at different time intervals from stroke induction; (2) to correlate miR-223-5p expression with NCKX2 protein expression in the same brain regions; (3) to evaluate whether miR-223-5p targets the 3′ UTR of the NCKX2 transcript; and (4) to evaluate the effect of miR-223-5p modulation through the selective anti-miR-223-5p on brain ischemic volume and neurological functions.
Results
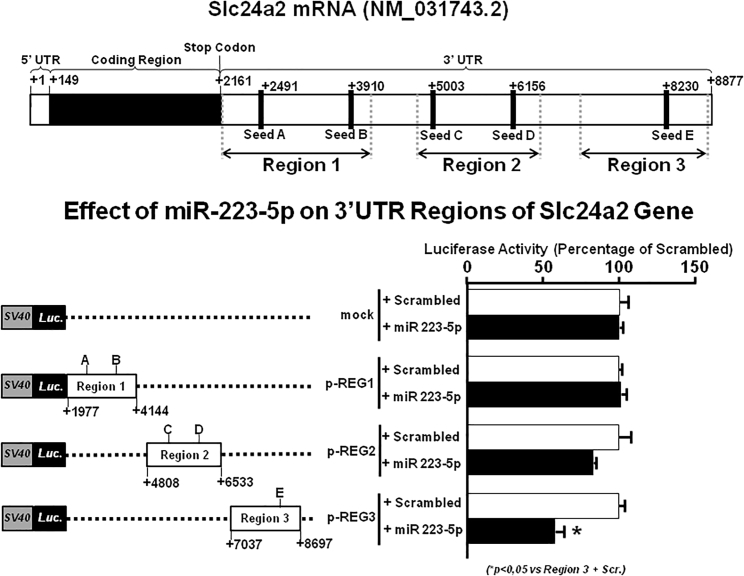
miR-223-5p Interacts with the 3′ UTR of NCKX2 RNA Messenger
To demonstrate that miR-223-5p directly binds to the 3′ UTR of NCKX2 RNA messenger and downregulates NCKX2 expression, a luciferase gene reporter assay was used. Three different subregions of the 3′ UTR of NCKX2 messenger, Slc24a2, were cloned downstream of the firefly luciferase gene and constructs were transfected in PC12 cells as described in the Materials and Methods section. Three different miRNA reporter constructs containing regions 1, 2, or 3 of the NCKX2 3′ UTR have been obtained, as indicated in Figure 1. Luciferase activity significantly decreased in PC12 cells cotransfected with miR-223-5p and the construct containing region 3 of the NCKX2 3′ UTR compared to scrambled-treated cells. In contrast, cells transfected with constructs containing region 1 or 2 of the NCKX2 3′ UTR did not change luciferase activity after miRNA transfection (Figure 1).
Figure 1.
Analysis of Luciferase Activity of NCKX2 3′ UTR in the Presence of miR-223-5p
Luciferase activity assay was evaluated in PC12 cells by co-expressing the vector containing 3′ UTRs of NCKX2 and candidate miRNA. The effect of 3′ UTR activity on the reporter gene was calculated as a ratio of firefly luciferase to renilla luciferase and is expressed as the percentage of scrambled-treated control as previously shown. This experiment was performed in triplicate and is representative of three independent experiments. Data are expressed as mean ± SEM. *p < 0.05.
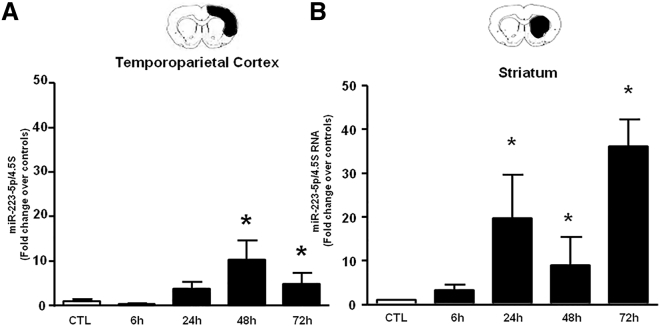
In the Cortex and Striatum of Ischemic Rats, the Expression of miR-223-5p Is Upregulated whereas Its Target NCKX2 Is Downregulated
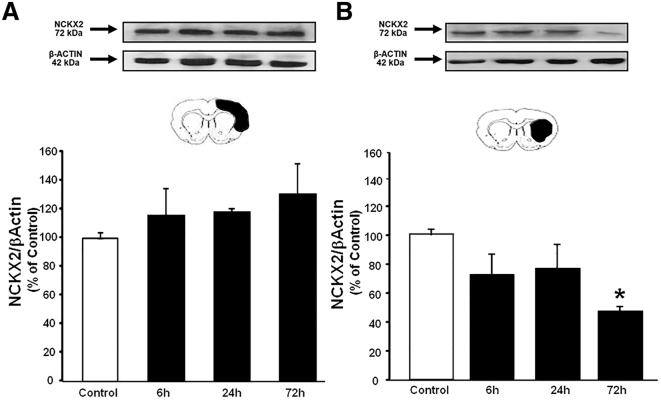
Analysis by RT-PCR revealed a significant increase in miR-223-5p levels in both the ipsilateral temporoparietal cortex and the striatum obtained from ischemic rats at different time intervals from ischemia induction. In particular, while in the cortex there was a 10-fold increase starting 48 h after ischemia (Figure 2A), in the striatum the miR-223-5p increase was significantly higher (20- to 30-fold) and occurred 24 h after the induction of ischemia (Figure 2B). In parallel with the drastic increase of miR-223-5p in the striatum, the analysis by western blot showed a significant decrease in NCKX2 protein levels 72 h after tMCAO (Figure 3B).
Figure 2.
Time Course of miR-223-5p Expression in Temporoparietal Cortex and Striatum after tMCAO in Rats
(A and B) Time course of miR-223-5p expression levels in temporoparietal cortex (A) and striatum (B) samples harvested from rats subjected to 100 min of tMCAO and sacrificed at 6, 24, 48, and 72 h. Results are expressed as fold changes over sham-operated control. n = 3–4 animals per group. *p < 0.05 versus sham-operated group. Data are expressed as mean ± SEM.
Figure 3.
Analysis of NCKX2 Expression in Temporoparietal Cortex and Striatum at Different Intervals after tMCAO in Rats
(A and B) Time course of NCKX2 protein expression levels in temporoparietal cortex (A) and striatum (B) samples harvested from rats subjected to 100 min of tMCAO and sacrificed at 6, 24, and 72 h. Results are normalized for β-actin and expressed as percentage of controls. n = 3–4 animals per group. *p < 0.05 versus control animals. Data are expressed as mean ± SEM.
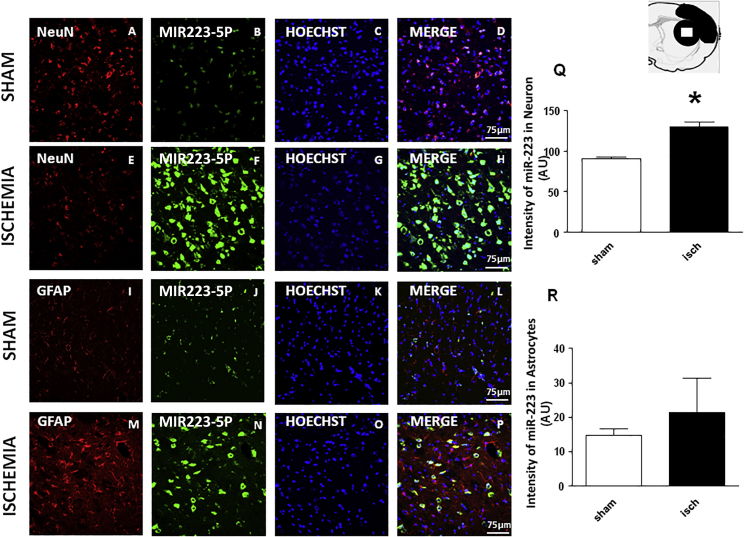
tMCAO Increases miR-223-5p Expression in NeuN-Positive Cells of the Ischemic Core
miR-223-5p expression was evaluated also by double fluorescence in situ hybridization with the neuronal marker NeuN and the astrocytic marker glial fibrillary acidic protein (GFAP) in tissue slices from ipsilesional striatum of both ischemic rats subjected to 100 min of tMCAO followed by 72 h of reperfusion and from sham-operated rats. Double fluorescence immunostaining revealed that, in ischemic rats, an increased miR-223-5p immunoreactivity was localized mainly in the cytosol and in the nuclei of NeuN-positive neurons (Figures 4E–4H). In particular, the expression of miR-223-5p was more abundant in striatal neurons of rats subjected to 100 min of tMCAO than in sham-operated rats (Figures 4A–4H). Furthermore, miR-223-5p immunoreactivity was present in the nuclei of GFAP-positive cells while it was totally absent in astrocyte branching (Figures 4I–4P). Quantification of the fluorescence intensity of miR-223-5p in NeuN-positive cells showed that miR-223-5p expression significantly increased in rats subjected to 100 min of tMCAO compared to sham-operated rats. Instead, the expression of miR-225-5p in GFAP-positive cells did not increase significantly in tMCAO rats, demonstrating that in ischemic rats the expression of miR-223-5-p was much more in neurons than in astrocytes
Figure 4.
Effect of 100 min of Transient Brain Ischemia (tMCAO) on miR-223-5p Expression
(A–P) Confocal microscopic images displaying NeuN (A and E) and GFAP (I and M) in red, miR-223-5p (B, F, J, and N) in green, Hoechst (C, G, K, and O) in blue, and merge (D, H, L, and P) in yellow in the brain ischemic regions of rats subjected to sham and tMCAO followed by 72 h of reperfusion. A representative brain slice cartoon indicating the area of interest is on the top of the figure. Scale bars, 75 μm. (Q and R) Quantification of miR-223-5p immunoreactivity in neurons (Q) and astrocytes (R). Image analysis of NeuN and GFAP was performed by NIH imaging software by measuring the intensity of fluorescent miR-223-5p immunolabeling in 80 NeuN-positive neurons for each group and 60 GFAP-positive astrocytes for each group. The intensity of miR-223-5p immunoreactivity is expressed in arbitrary units.
miR-223-5p Induces NCKX2 Downregulation in Rat Cortical Neurons
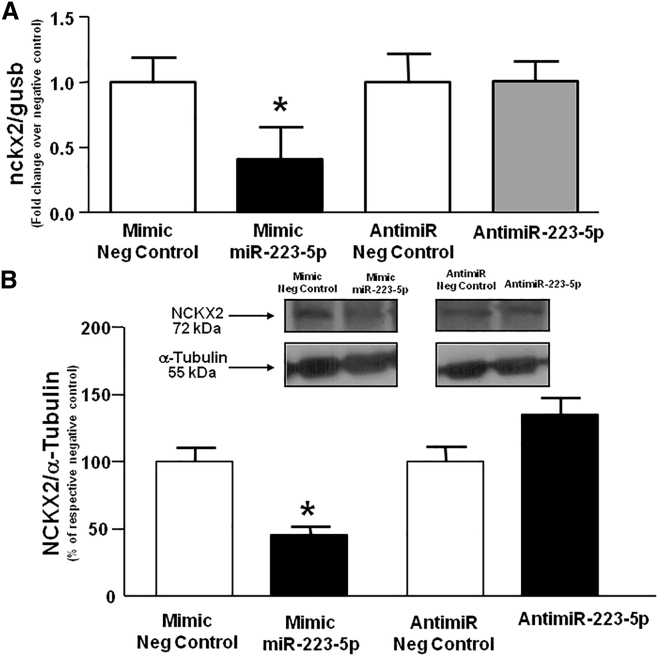
In order to verify the inverse correlation between miR-223-5p and NCKX2 levels, rat primary cortical neurons were transfected with miR-223-5p mimic or with the corresponding negative control and, 24 h after transfection, both transcripts and NCKX2 protein levels were analyzed. The results showed that the mimic of miR-223-5p caused a 50% reduction in both transcript and protein levels (Figure 5).
Figure 5.
Analysis of NCKX2 Transcript (A) and Protein (B) Expression in Cortical Neurons 24 h after Transfection of miR223-5p mimic
NCKX2 expression in cortical neurons transfected with miR-223-5p or anti-miR-223-5p. Values are normalized with respect to α-tubulin. *p < 0.05 versus negative controls. Data are expressed as mean ± SEM. *p < 0.05.
No change in NCKX2 protein levels was observed after transfection of the anti-miR-223-5p. This result might be explained by the fact that miR-223-5p levels are already very low in cortical neurons in physiological conditions and therefore the transfection of antimiRNA could not affect the expression of the target protein NCKX2 at all (Figure 5).
Anti-miR-223-5p Reduces Ischemic Damage and Improves Neurological Deficits
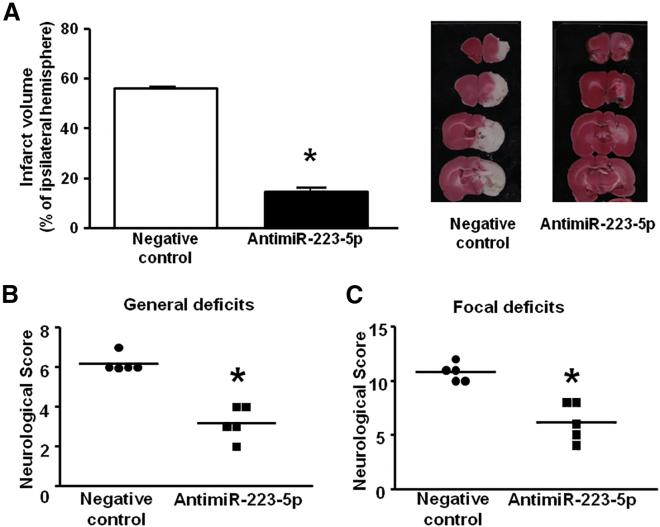
To evaluate the effects of anti-miR-223-5p on ischemic brain damage, it was administered by an osmotic pump starting 6 h before ischemia induction and up to 72 h later. The analysis of the ischemic volume by 2,3,5-triphenyltetrazolium hydrochloride (TTC) staining showed a significant reduction (65%) in the ischemic volume of animals treated with antimiRNA compared to the animals treated with the corresponding negative control (Figure 6A). The decrease in ischemic volume was also accompanied by a parallel improvement of both general and focal neurological deficits (Figures 6B and 6C).
Figure 6.
Evaluation of Ischemic Damage and Neurological Deficit 72 h after tMCAO in Rats Treated with miR-223-5p Inhibitor
(A) Evaluation of ischemic damage 72 h after tMCAO in rats treated with anti-miR-223-5p. *p < 0.05 versus ischemic rats treated with negative control. n = 5–6 animals per group. Each column represents the mean ± SEM. (B and C) General (B) and focal (C) neurological deficits 72 h after tMCAO in rats treated with anti-miR-223-5p (horizontal line indicates the mean neurological scores).
Anti-miR-223-5p Prevents NCKX2 Downregulation after Brain Ischemia
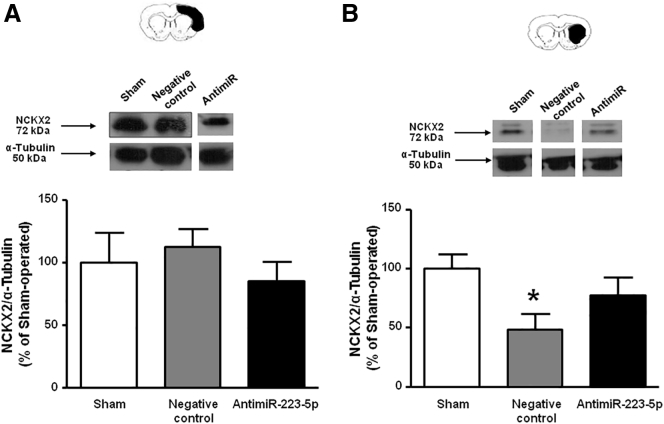
In order to verify whether the reduction of the ischemic volume and the improvement of neurological deficits observed after the administration of the anti-miR-223-5p were associated with a variation of the expression levels of NCKX2, its expression was analyzed in the striatum and temporoparietal cortex of ischemic rats treated with anti-miR-223-5p (Figure 7). The results obtained showed that the anti-miR-223-5p completely prevented the reduction in NCKX2 protein levels observed in the striatum 72 h after ischemia (Figure 7B).
Figure 7.
Evaluation of the Expression of NCKX2 Protein Levels in Ischemic Rats after Anti-miR-223-5p Treatment
(A and B) Evaluation of NCKX2 protein levels 72 h after tMCAO in rats treated with Negative control antimiRNA or with anti-miR-223-5p in cortex (A) and in striatum (B). Results are normalized for α-tubulin and expressed as percentage of sham-operated animals. n = 5 animals per group. *p < 0.05 versus sham-operated group. Data are expressed as mean ± SEM.
Discussion
The results shown in the present study demonstrate for the first time that NCKX2, the neuronal member of the K+-dependent Na+/Ca2+ exchangers family, is regulated by miR-223-5p during ischemia and that the modulation of miR-223-5p, using the corresponding antagomir, may prevent the decrease of NCKX2 protein levels, thus promoting neuroprotection against ischemic damage.
Interestingly, we demonstrated that the expression of NCKX2 is reduced 72 h after ischemia in ipsilateral striatum and that this decrease reflects the upregulation of miR-223-5p in the same area. In contrast, the increase in miR-223-5p levels in the temporoparietal cortex is not accompanied by an alteration of the expression of NCKX2. In this regard, it is necessary to highlight that the percentage of miR-223-5p increase in the cortex is much lower than that observed in the striatum, and therefore it is possible that the most important effect of this miRNA in the cortex occurs mainly through the modulation of other target proteins, different from NCKX2. Furthermore, the miR-223-5p increase in cortex was delayed compared to the striatum, thus suggesting a higher importance of this miRNA in the striatum area. The correlation between the expression of NCKX2 and miR-223-5p was also confirmed by our in vitro results, demonstrating that primary neuronal cultures transfected with miR-223-5p showed a significant reduction in the expression of NCKX2. The fact that no change in protein levels of NCKX2 was observed after transfection of the anti-miR-223-5p might be explained by the observation that miRNA levels are already very low in cortical neurons under physiological conditions, and therefore the transfection of antimiRNA is not able to induce any further effect on the expression of the target protein NCKX2.
The validation of a direct interaction between miR-223-5p and the 3′ UTR of NCKX2 transcript resides on the results of a luciferase assay, showing a reduction in luciferase activity when the construct containing region 3 of 3′ UTR was transfected with the miR-223-5p mimic.
Interestingly, the experiments show that antimiRNA treatment ameliorates brain ischemia and improves both general and focal neurological deficits by preserving NCKX2 protein levels in the striatum. This finding supports the idea that NCKX2 plays a key role in the regulation of sodium and calcium homeostasis in this area during ischemic conditions.7 Indeed, thanks to its electrophysiological properties, the role of NCKX2 in striatum is fundamental. Unlike the other NCX isoforms, this antiporter may be still operative under the ionic conditions occurring in striatum after ischemia. In particular, its KD for the potassium ions of about 40 mM allows NCKX2 to operate only when the extracellular concentrations of K+ overcome the physiological levels.8,9 Interestingly, in the ischemic core, K+ extracellular concentrations reach levels higher than those corresponding to the Kd of the external sites of K+ for NCKX2.10 Therefore, a transporter with the features of NCKX2 may effectively counterbalance the altered ionic homeostasis occurring after stroke and responsible for ischemic damage propagation from striatum to the cortex, thus supporting our findings showing that antimiRNA treatment is effective in protecting the ischemic cortex, although no variation in NCKX2 expression was recorded after ischemia in this region. Furthermore, it has to be emphasized that double immunofluorescence experiments showed that the increase in miR-223-5p expression mainly occurs in neuronal cells of striatum and not in GFAP-positive cells. In fact, NCKX2 is a selectively neuronal-expressed transporter.
Alternatively, the neuroprotective role of anti-miR-223-5p occurring also through the prevention of stroke-induced NCKX2 downregulation is in line with our previous study on the role of NCKX2 in cerebral ischemia, demonstrating that primary cortical neurons from NCKX2−/− mice were more susceptible to hypoxic conditions and that both NCKX2 knockdown and knockout impaired brain damage and neurological function.7
In this context, given the absence of molecules able to activate NCKX2, the antimiRNA strategy, successfully used in other contexts,11 may represent a valid and translatable opportunity to modulate the expression of a target protein. Alternatively, it cannot be excluded that the neuroprotective effect in stroke of antimiRNA-223-5p may be attributable also to other targets, different from NCKX2. In fact, this miRNA may modulate other relevant proteins, potentially able to interfere with the progression of cerebral ischemic damage. In particular, it is interesting to note that the two cell death receptors, tumor necrosis factor receptor 1 (TNFR1) and DR6, were identified as targets of miR-223 in a model of cardiac ischemia and reperfusion.12 Furthermore, in diabetic rats subjected to cerebral ischemia, the administration of antimiR-223 increased the expression of CD31 and vascular endothelial growth factor (VEGF), indicating that this miRNA plays an additional role on other factors involved in cerebral ischemic damage, besides its role in modulating NCKX2 expression, demonstrated in the present paper.13
Collectively, the results of the present work show for the first time that the miR-223-5p inhibition through antimiRNA strategy may pave the way for an innovative therapeutic intervention based on the identification of a new druggable target.
Materials and Methods
Identification of miR-223-5p as an NCKX2 Modulator
TargetScan (http://www.targetscan.org/vert_72/) has been used in order to identify the most promising miRNA candidate able to control NCKX2 expression. In fact, TargetScan is a computational method to predict the targets of conserved vertebrate miRNAs, integrating the model of interaction miRNA-mRNA on the basis of thermodynamics and sequence alignment analysis between miRNA binding sites among different species.14 In this way, many false positives are filtered from the beginning of the prediction process, thereby resulting in estimates in a range between 22% and 31%. In this way, in the present study we predicted the existence of a strict complementarity between NCKX2 and miR-223-5p. Such a prediction was based on the Watson-Crick annealing between the “seed sequence” of the miRNA of interest and the target sequence on NCKX2 mRNA.
Drugs and Chemicals
AntimiRNA for miR-223-5p (mmu-miR-223-5p miRCURY LNA miRNA inhibitor, ID: YI04100703), antimiRNA control (negative control A miRCURY LNA miRNA inhibitor control, ID: YI00199006), miR-223-5p mimic (mmu-miR-223-5p miRCURY LNA miRNA, ID: YM00472737), and the corresponding negative control (negative control miRCURY LNA miRNA mimic, ID: YM00479902) were purchased from QIAGEN (Denmark). For double fluorescence in situ hybridization and immunohistochemistry the probe-detected miR-223-5p (probe sequence: 5′-DiGN/CAACTCAGCTTGTCAAATACAC/3′-DiGN/) was purchased from Exiqon.
Luciferase Activity Assay
Three different subregions (+1977 to +4144, +4808 to +6533, +7037 to +8697) of Slc24a2 3′ UTR (ENSRNOG00000008169.6) were cloned downstream of the firefly luciferase gene in pmirGLO Dual-Luciferase empty vector (Promega, Italy). All plasmid sequences were verified by DNA sequencing (Microgem, Naples, Italy). One hundred nanograms of luciferase Slc24a2 construct plus 50 nM miR-223-5p mimic, or negative scrambled control, was transfected with Lipofectamine 2000 (Thermo Fisher, Italy) in PC12 cells according to the manufacturer’s protocol. Forty-eight hours after transfection, luciferase activity was determined using the Dual-Luciferase assay kit (Promega) with a GloMax 20/20 luminometer (Promega). The effect of 3′ UTR activity on the reporter gene was calculated as previously shown11,15 as a ratio of firefly luciferase to renilla luciferase and is expressed as a percentage of scrambled-treated control.
Rat Primary Cortical Neurons
Rat primary cortical neurons were prepared from 17-day-old Wistar rat embryos (Charles River).16 Rats were first anesthetized and then decapitated to minimize animal pain and distress. Dissection and dissociation were performed in Ca2+/Mg2+-free PBS containing glucose (30 mM). Tissues were incubated with papain for 10 min at 37°C and dissociated by trituration in Earle’s balanced salt solution (EBSS) containing DNase (0.16 U/mL), BSA (10 mg/mL), and ovomucoid (10 mg/mL). Neurons were plated in plastic Petri dishes (Falcon/Becton Dickinson) precoated with poly-d-lysine (20 μg/mL) and were grown in DMEM/F12 (Life Technologies) containing glucose, 5% of deactivated fetal bovine serum (FBS), and 5% of horse serum (HS, Life Technologies), glutamine (2 mM), penicillin (50 U/mL), and streptomycin (50 μg/mL) (Invitrogen). Within 48 h of plating, cytosine arabinoside (arabinoside-C) (10 μM) was added to prevent non-neuronal cell growth. Neurons were cultured at 37°C in a humidified 5% CO2 atmosphere and used after 7–10 days of culture. Cell density was 5 × 106 cells/well of 60 mm for analysis of qRT-PCR and 15 × 106 cells/well of 100 mm for western blot analysis.
Transfection of Rat Cortical Neurons
Primary rat cortical neurons were transfected with 50 nM miR-223-5p mimic or miR-223-5p antimiRNA and respective negative controls. Lipofectamine LTX (Thermo Fisher) was used as the transfection agent, according to the manufacturer’s protocol. After an incubation period of 24 h, the medium was replaced and cells were harvested and used for western blot or PCR analysis.17
In Vivo Experimental Groups
Seventy-two Sprague-Dawley male rats (Charles River Laboratories, Calco, Varese, Italy) weighing 200–250 g were housed under diurnal lighting conditions (12-h light/12-h dark).
It has been calculated that about 20% of the animals used were excluded from the experimental groups due to the absence of ischemic lesions or to mortality related to the experimental procedure.
Experiments were performed according to international guidelines for animal research. The experimental protocol was approved by the Animal Care Committee of the “Federico II” University of Naples.
Transient Focal Ischemia and Anti-miRNA Administration
Transient focal ischemia was induced, as previously described,18 by individuals who did not implant osmotic pumps in animals. In brief, occlusion of the middle cerebral artery (MCA) was performed in male rats anesthetized using a mixture of oxygen and sevoflurane at 3.5% (medical oxygen concentrator LFY-I-5A). A 5-0 surgical monofilament nylon suture (Doccol, Sharon, MA, USA) was inserted from the right external carotid artery into the right internal carotid artery and advanced into the circle of Willis up to the branching point of the MCA, thereby occluding the MCA. Achievement of ischemia was confirmed by monitoring regional cerebral blood flow in the area of the right MCA. Cerebral blood flow was monitored through a disposable microtip fiber optic probe (diameter, 0.5 mm) connected through a master probe to a laser Doppler computerized main unit (PF5001; Perimed, Järfälla, Sweden) and analyzed using PSW Perisoft 2.5.19 Animals not showing a cerebral blood flow reduction of at least 70% were excluded from the experimental group, as were animals that died after ischemia induction. Rectal temperature was maintained at 37 ± 0.5°C with a thermostatically controlled heating pad and lamp. All surgical procedures were performed under an operating stereomicroscope.
For the evaluation of NCKX2 and miR-223-5p expression, rats were divided into five experimental groups: (1) control animals, (2) ischemic rats sacrificed after 6 h of reperfusion, (3) ischemic rats sacrificed after 24 h of reperfusion, (4) ischemic rats sacrificed after 48 h of reperfusion, and (5) ischemic rats sacrificed after 72 h of reperfusion. For evaluation of the antimiRNA effect on ischemic damage, rats were divided into three experimental groups: (1) sham-operated rats receiving intracerebroventricular (i.c.v.) infusion of vehicle, (2) ischemic rats receiving i.c.v. infusion of anti-miR-223-5p, and (3) ischemic rats receiving i.c.v. infusion of negative control antimiR. These animals were euthanized 72 h after the 100-min tMCAO by an overdose of sevoflurane to evaluate neurological scores, infarct volume, or NCKX2 protein expression.
The continuous release of anti-miR-223-5p into brain lateral ventricle was achieved by using osmotic pumps (Alzet, Palo Alto, CA, USA). The osmotic pumps were prefilled with anti-miR-223-5p or negative control antimiRNA in a blinded manner by individuals who did not perform tMCAO surgery on animals. Implantation of the osmotic pump frame was carried out in rats positioned on a stereotaxic apparatus 6 h before the induction of transient ischemia. The osmotic pump was connected to a brain infusion kit (Alzet, no. 0004760) made of a stainless steel cannula that was implanted into the right lateral ventricle using the stereotaxic coordinates from the bregma: 0.4 mm caudal, 2 mm lateral, and 2 mm below the dura and secured to the skull with dental cement.20 The pump was placed in the skin fold on the neck of the rat. Anti-miR-223-5p and the negative control antimiRNA were diluted to the final concentration in a previously filtered saline solution (0.9% NaCl g/L). The antimiRNA was i.c.v. administered at the concentration 8 μmol/L (9 μg/kg body weight), and the release of antimiRNA by the osmotic pump within the rat cerebral ventricle was set up at a speed of 1 μL/h. Rectal temperature was maintained at 37 ± 0.5°C with a thermostatically controlled heating pad during the whole surgical procedure. In the animals, a catheter was inserted into the femoral artery to measure arterial blood gases before and after ischemia (Rapid Laboratory 860; Chiron Diagnostics, Emeryville, CA, USA). All surgical procedures were performed under an operating stereomicroscope in a blinded manner.
Evaluation of the Infarct Volume and of Neurological Deficit Scores
For evaluation of ischemic volume, animals were killed with sevoflurane overdose 72 h after ischemia. Brains were quickly removed, sectioned coronally at 1-mm intervals, and stained by immersion in the vital dye (2% in PBS) TTC. The infarct volume was calculated by summing the infarction areas of each section and by multiplying the total by slice thickness. To avoid that edema could affect the infarct volume value, infarct volume was expressed as the percentage of ischemic damage by dividing the total infarct volume by the total ipsilateral hemispheric volume.21 In this way any potential interference due to increased brain volume caused by water content increase is eliminated.
Neurological scores were evaluated before euthanization according to the following two scales: a general neurological scale and a focal neurological scale. In the general score, the following six general neurological functions were evaluated: (a) hair conditions (0–2), (b) position of ears (0–2), (c) eye conditions (0–4), (d) posture (0–4), (e) spontaneous activity (0–4), and (f) epileptic behavior (0–12). For each of the six general functions measured, animals received a score depending on the severity of the symptoms: the higher the score, the worse is the rat condition. The scores of investigated items were then summed to provide a total general score ranging from 0 to 28. In the focal score, the following seven areas were assessed: (a) body symmetry, (b) gait, (c) climbing, (d) circling behavior, (e) front limb symmetry, (f) compulsory circling, and (g) whisker response. For each of these items, animals were rated between 0 and 4 depending on the severity. The seven items were then summed to give a total focal score ranging between 0 and 28.20 Infarct volumes and neurological scores were evaluated in a blinded manner by individuals who did not perform the surgical procedures.
Western Blot Analysis
Samples from cortical neurons and rat ischemic brain regions were homogenized in a lysis buffer (50 mmol/L Tris-HCl [pH 7.5], 100 mmol/L NaCl, 1% Triton X-100) containing protease and phosphatase inhibitors. After centrifugation at 12,000 × g at 4°C for 15 min, the supernatants were collected. Protein concentration was estimated using the Bradford method, by means of a spectrophotometer (Eppendorf). Then, 80–100 μg of protein lysate was mixed with a Laemmli sample buffer and boiled at 95°C for 5 min. The samples were resolved by SDS-PAGE and transferred to nitrocellulose membranes.21 Blots were probed with antibodies for NCKX2 (1:500, Vinci Biochem), β-actin (1:10,000, Sigma), and α-tubulin (1:10,000, Abcam) diluted in Tris-buffered saline (TBS) with Tween 20 (TBST)/1% BSA overnight (4°C). Then, they were detected using horseradish peroxidase-conjugated secondary antibody (1:2,000, mouse and rabbit; Cell Signaling; 60 min at room temperature in 5% non-fat milk) and an enhanced luminescence kit (Amersham Pharmacia Biotech, NJ, USA).22,23
Real-Time PCR
Total RNA from neuronal cultures or brain tissues was extracted by using TRIzol following the supplier’s instructions (TRI Reagent, Sigma). For miRNA analysis, 5 ng of RNA were retrotranscribed in cDNA, using a high-capacity cDNA reverse transcription kit (Applied Biosystems) and TaqMan probes, following the TaqMan Small RNA Assays protocol (16°C for 30 min, 42°C for 30 min, and 85°C for 5 min); in this way, only the RNA target was retrotranscribed in cDNA. TaqMan technology (Thermo Fisher Scientific) consists of a pair of probes: one for retrotranscription and one for PCR. Probes used are the following: miRNA assay mmu-miR-223-5p (ID: 007896) and miRNA control assay 4.5S RNA (ID 001716). Quantitative real-time PCR was performed with TaqMan Universal PCR Master Mix II (Applied Biosystems) in a 7500 fast real-time PCR system (Applied Biosystems). cDNA samples were amplified simultaneously in triplicate in one assay run, following the protocol for TaqMan assays: 50°C for 2 min, 95°C for 10, 40 cycles of amplification of 95°C for 15 s, and 60°C for 1 minute. All reactions were run in triplicate. Results were analyzed and exported with 7500 Fast System SDS Software.24
For NCKX2 mRNA, 1 μg of RNA was retrotranscribed with a high-capacity cDNA reverse transcription kit (Applied Biosystems), but following the manufacturer’s protocol: 25°C for 10 min, 37°C for 120 min, and 85°C for 5 min. Real-time PCR was performed as described above, with GAPDH as endogenous control RNA. Probes used are the following: gene expression assay Slc24a2 (ID: Rn00582020_m1); gene expression assay Gapdh (ID: Rn01462661_g1).
Double Fluorescence In Situ Hybridization and Immunohistochemistry
For in situ hybridization, all procedures were performed in autoclaved solutions and RNAse-free conditions. Rats were perfused with 1× PBS and 4% paraformaldehyde solution in PBS. The brain tissues from ischemic and control rats were fixed in 4% paraformaldehyde solution in PBS overnight at 4°C and subsequently cryoprotected in 30% sucrose in PBS overnight at 4°C and cryosectioned at 10-μm thickness.
Frozen tissue sections were prepared following the description of miRNA protocol for in situ hybridization on frozen sections (Exiqon, Denmark).
Briefly, brain sections from ischemic and control animals 72 h after reperfusion were submerged in neutral buffered formalin (10%) for 15 min and then washed in PBS three times for 5 min. Sections were incubated in proteinase K buffer containing 1 M Tris-HCl (pH 7.4), 0.5 M EDTA, 5 M NaCl, and proteinase K (15 μg/mL) in RNase-free water for 10 min at 37°C, and then the sections were washed three times for 3 min in PBS. Sections were then incubated in 3% H2O2 for 5 min to inhibit endogenous peroxidase activity and then washed in PBS three times for 3 min. Sections were sequentially hybridized for 1 h at 55°C and 54°C. The final concentration of the probe was 20 nM. The sections were hybridized in hybridization buffer containing 50% deionized formamide, 0.3 M NaCl, 20 mM Tris-HCl (pH 8.0), 5 mM EDTA, 10 mM NaPO4 (pH 8.0), 10% dextran sulfate, 1× Denhardt’s solution, 0.5 mg/mL yeast RNA, and probe. Post-hybridization washes were performed sequentially twice for 5 min at hybridization temperature in 5× saline sodium citrate (SSC) buffer, three times for 5 min at hybridization temperature in 1× SSC buffer, twice for 5 min at hybridization temperature in 0.2× SSC, and once for 5 min at room temperature in 0.2× SSC buffer. Following the stringent washing, the sections were incubated in blocking solution containing 2% sheep serum and 1% BSA in PBS with 0.1% Tween 20 for 15 min at room temperature. Then, the sections were incubated for 60 min with anti-digoxigenin-peroxidase (POD), antigen-binding fragments (Fabs; Roche Diagnostics, Germany) diluted 1:400 in 1% sheep serum/1% BSA, and PBS with 0.05% Tween 20. Then, the sections were washed in PBS three times for 5 min and incubated for 5 min in Cy2-conjugated tyramide (tyramide signal amplification [TSA] Plus Fluorescein kit, PerkinElmer, USA) by diluting TSA stock solution 1:50 in 1× amplification diluent. After washing three times for 10 min with TBS, sections were incubated for 30 min in 3% H2O2 in TBS to quench peroxidase activity from the initial TSA reaction. After washing, sections were incubated with the following primary antibodies in blocking solution: anti-NeuN 1:200 (Elabscience, USA) and anti-GFAP 1:200 (Millipore, USA) overnight. Sections were sequentially washed three times for 10 min with PBS and then incubated for 2 h in Alexa Fluor 594-conjugated donkey anti-mouse/rabbit antiserum diluted 1:300 in blocking solution. Following washing three times for 10 min with PBS, sections were incubated with Hoechst for 20 min and mounted onto slides using Fluoromount aqueous mounting medium (Sigma, Germany), air-dried, and stored in a dark room.25 As controls, the sections were incubated without the anti-digoxigenin-POD or without the TSA Plus Fluorescein kit or primary antibodies, and the immunoreactivity was completely abolished (data not shown).
To obtain an indirect measure of the amount of miR-223-5p in neurons and astrocytes, image analysis of NeuN and GFAP was performed by NIH image software by measuring the intensity of fluorescent miR-223-5p immunolabeling in 80 NeuN-positive neurons for each group and 60 GFAP-positive astrocytes for each group. The intensity of miR-223-5p immunoreactivity was expressed in arbitrary units. Digital images were taken with a ×40 objective, and identical laser power settings and exposure times were applied to all images from each experimental set. Images from the same areas of each brain region were compared.26
Statistical Analysis
Values are expressed as means ± SEM. In particular, real-time PCR results are expressed as fold change (2−ΔΔCt) compared to the control group set to 1. Briefly, the difference between Ct values of a gene of interest and internal control (ΔCt) is calculated for both the control sample and target sample. Then, the difference between ΔCt of the target sample and control sample (ΔΔCt) is calculated. Fold change of gene expression of target samples compared to control sample is calculated as 2−ΔΔCt. For Western blot analysis, results are expressed as percentage of variation of target protein (already normalized for internal control) of the test sample compared to control sample. Statistical analysis was performed with GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA), using ANOVA followed by Newman-Keuls test or Bonferroni post hoc test for more than two groups. To compare two groups, unpaired t test was used. Statistical significance was accepted at the 95% confidence level (p < 0.05).
Author Contributions
O.C. and A.V. conducted in vivo experiments. C.P., R.S, V.V., N.G., and A.S. performed in vitro experiments. S.A. and P.B. performed immunohistochemistry experiments. O.C., L.F., and P.M. analyzed data. O.C., L.A., and G.P. designed the experiments and wrote the paper.
Acknowledgments
We thank Dr. Lucia d’Esposito for invaluable support in all in vivo studies, and Daniela Di Donato for technical support for ex vivo studies. This work was supported by grants from Programma Operativo Nazionale (PON_01602, PON03PE_00146_1, and PON PERMEDNET ArSol-1226) from the Italian Ministry of Research, MIUR, to L.A.; PRIN 2015, no. 2015783N45-CUP: E62F1600136001 from the Italian Ministry of Research, MIUR, to G.P.; Nanomirnictus F/050139/01,02/X32 to L.A. from the Italian Ministry of Economic Development, MISE; and SIR 2014, no. RBSI14QECG to A.V. from the Italian Ministry of Research, MIUR.
Contributor Information
Lucio Annunziato, Email: lannunzi@unina.it.
Giuseppe Pignataro, Email: giuseppe.pignataro@unina.it.
References
- 1.Bartel D.P. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 2.Bartel D.P. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adams L. Non-coding RNA: pri-miRNA processing: structure is key. Nat. Rev. Genet. 2017;18:145. doi: 10.1038/nrg.2017.6. [DOI] [PubMed] [Google Scholar]
- 4.Karnati H.K., Panigrahi M.K., Gutti R.K., Greig N.H., Tamargo I.A. miRNAs: key players in neurodegenerative disorders and epilepsy. J. Alzheimers Dis. 2015;48:563–580. doi: 10.3233/JAD-150395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cuomo O., Vinciguerra A., Cerullo P., Anzilotti S., Brancaccio P., Bilo L., Scorziello A., Molinaro P., Di Renzo G., Pignataro G. Ionic homeostasis in brain conditioning. Front. Neurosci. 2015;9:277. doi: 10.3389/fnins.2015.00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Annunziato L., Pignataro G., Boscia F., Sirabella R., Formisano L., Saggese M., Cuomo O., Gala R., Secondo A., Viggiano D. ncx1, ncx2, and ncx3 gene product expression and function in neuronal anoxia and brain ischemia. Ann. N Y Acad. Sci. 2007;1099:413–426. doi: 10.1196/annals.1387.050. [DOI] [PubMed] [Google Scholar]
- 7.Cuomo O., Gala R., Pignataro G., Boscia F., Secondo A., Scorziello A., Pannaccione A., Viggiano D., Adornetto A., Molinaro P. A critical role for the potassium-dependent sodium-calcium exchanger NCKX2 in protection against focal ischemic brain damage. J. Neurosci. 2008;28:2053–2063. doi: 10.1523/JNEUROSCI.4912-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee S.H., Kim M.H., Park K.H., Earm Y.E., Ho W.K. K+-dependent Na+/Ca2+ exchange is a major Ca2+ clearance mechanism in axon terminals of rat neurohypophysis. J. Neurosci. 2002;22:6891–6899. doi: 10.1523/JNEUROSCI.22-16-06891.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Visser F., Valsecchi V., Annunziato L., Lytton J. Exchangers NCKX2, NCKX3, and NCKX4: identification of Thr-551 as a key residue in defining the apparent K+ affinity of NCKX2. J. Biol. Chem. 2007;282:4453–4462. doi: 10.1074/jbc.M610582200. [DOI] [PubMed] [Google Scholar]
- 10.Gidö G., Kristián T., Siesjö B.K. Extracellular potassium in a neocortical core area after transient focal ischemia. Stroke. 1997;28:206–210. doi: 10.1161/01.str.28.1.206. [DOI] [PubMed] [Google Scholar]
- 11.Vinciguerra A., Formisano L., Cerullo P., Guida N., Cuomo O., Esposito A., Di Renzo G., Annunziato L., Pignataro G. MicroRNA-103-1 selectively downregulates brain NCX1 and its inhibition by anti-miRNA ameliorates stroke damage and neurological deficits. Mol. Ther. 2014;22:1829–1838. doi: 10.1038/mt.2014.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Qin D., Wang X., Li Y., Yang L., Wang R., Peng J., Essandoh K., Mu X., Peng T., Han Q. MicroRNA-223-5p and -3p cooperatively suppress necroptosis in ischemic/reperfused hearts. J. Biol. Chem. 2016;291:20247–20259. doi: 10.1074/jbc.M116.732735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhu L., Wang X., Wu J., Yang J., Sun X., Fan X., Zhang M., Zhou S. Effects of miR-223 on cerebral ischemic injury and angiogenesis in diabetic rats. Int. J. Clin. Exp. Med. 2016;9:13972–13978. [Google Scholar]
- 14.Anantharam A., Lewis A., Panaghie G., Gordon E., McCrossan Z.A., Lerner D.J., Abbott G.W. RNA interference reveals that endogenous Xenopus MinK-related peptides govern mammalian K+ channel function in oocyte expression studies. J. Biol. Chem. 2003;278:11739–11745. doi: 10.1074/jbc.M212751200. [DOI] [PubMed] [Google Scholar]
- 15.Valsecchi V., Pignataro G., Sirabella R., Matrone C., Boscia F., Scorziello A., Sisalli M.J., Esposito E., Zambrano N., Cataldi M. Transcriptional regulation of ncx1 gene in the brain. Adv. Exp. Med. Biol. 2013;961:137–145. doi: 10.1007/978-1-4614-4756-6_12. [DOI] [PubMed] [Google Scholar]
- 16.Scorziello A., Santillo M., Adornetto A., Dell’aversano C., Sirabella R., Damiano S., Canzoniero L.M., Renzo G.F., Annunziato L. NO-induced neuroprotection in ischemic preconditioning stimulates mitochondrial Mn-SOD activity and expression via Ras/ERK1/2 pathway. J. Neurochem. 2007;103:1472–1480. doi: 10.1111/j.1471-4159.2007.04845.x. [DOI] [PubMed] [Google Scholar]
- 17.Guida N., Laudati G., Serani A., Mascolo L., Molinaro P., Montuori P., Di Renzo G., Canzoniero L.M.T., Formisano L. The neurotoxicant PCB-95 by increasing the neuronal transcriptional repressor REST down-regulates caspase-8 and increases Ripk1, Ripk3 and MLKL expression determining necroptotic neuronal death. Biochem. Pharmacol. 2017;142:229–241. doi: 10.1016/j.bcp.2017.06.135. [DOI] [PubMed] [Google Scholar]
- 18.Formisano L., Guida N., Valsecchi V., Cantile M., Cuomo O., Vinciguerra A., Laudati G., Pignataro G., Sirabella R., Di Renzo G., Annunziato L. Sp3/REST/HDAC1/HDAC2 complex represses and Sp1/HIF-1/p300 complex activates ncx1 gene transcription, in brain ischemia and in ischemic brain preconditioning, by epigenetic mechanism. J. Neurosci. 2015;35:7332–7348. doi: 10.1523/JNEUROSCI.2174-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cantarella G., Pignataro G., Di Benedetto G., Anzilotti S., Vinciguerra A., Cuomo O., Di Renzo G.F., Parenti C., Annunziato L., Bernardini R. Ischemic tolerance modulates TRAIL expression and its receptors and generates a neuroprotected phenotype. Cell Death Dis. 2014;5:e1331. doi: 10.1038/cddis.2014.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cuomo O., Pignataro G., Gala R., Scorziello A., Gravino E., Piazza O., Tufano R., Di Renzo G., Annunziato L. Antithrombin reduces ischemic volume, ameliorates neurologic deficits, and prolongs animal survival in both transient and permanent focal ischemia. Stroke. 2007;38:3272–3279. doi: 10.1161/STROKEAHA.107.488486. [DOI] [PubMed] [Google Scholar]
- 21.Cuomo O., Pignataro G., Sirabella R., Molinaro P., Anzilotti S., Scorziello A., Sisalli M.J., Di Renzo G., Annunziato L. Sumoylation of LYS590 of NCX3 f-Loop by SUMO1 participates in brain neuroprotection induced by ischemic preconditioning. Stroke. 2016;47:1085–1093. doi: 10.1161/STROKEAHA.115.012514. [DOI] [PubMed] [Google Scholar]
- 22.Cuomo O., Pignataro G., Gala R., Boscia F., Tortiglione A., Molinaro P., Di Renzo G., Lytton J., Annunziato L. Involvement of the potassium-dependent sodium/calcium exchanger gene product NCKX2 in the brain insult induced by permanent focal cerebral ischemia. Ann. N Y Acad. Sci. 2007;1099:486–489. doi: 10.1196/annals.1387.051. [DOI] [PubMed] [Google Scholar]
- 23.Molinaro P., Cantile M., Cuomo O., Secondo A., Pannaccione A., Ambrosino P., Pignataro G., Fiorino F., Severino B., Gatta E. Neurounina-1, a novel compound that increases Na+/Ca2+ exchanger activity, effectively protects against stroke damage. Mol. Pharmacol. 2013;83:142–156. doi: 10.1124/mol.112.080986. [DOI] [PubMed] [Google Scholar]
- 24.Anzilotti S., Brancaccio P., Simeone G., Valsecchi V., Vinciguerra A., Secondo A., Petrozziello T., Guida N., Sirabella R., Cuomo O. Preconditioning, induced by sub-toxic dose of the neurotoxin L-BMAA, delays ALS progression in mice and prevents Na+/Ca2+ exchanger 3 downregulation. Cell Death Dis. 2018;9:206. doi: 10.1038/s41419-017-0227-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gargiulo S., Anzilotti S., Coda A.R., Gramanzini M., Greco A., Panico M., Vinciguerra A., Zannetti A., Vicidomini C., Dollé F. Imaging of brain TSPO expression in a mouse model of amyotrophic lateral sclerosis with 18F-DPA-714 and micro-PET/CT. Eur. J. Nucl. Med. Mol. Imaging. 2016;43:1348–1359. doi: 10.1007/s00259-016-3311-y. [DOI] [PubMed] [Google Scholar]
- 26.Anzilotti S., Tornincasa M., Gerlini R., Conte A., Brancaccio P., Cuomo O., Bianco G., Fusco A., Annunziato L., Pignataro G., Pierantoni G.M. Genetic ablation of homeodomain-interacting protein kinase 2 selectively induces apoptosis of cerebellar Purkinje cells during adulthood and generates an ataxic-like phenotype. Cell Death Dis. 2015;6:e2004. doi: 10.1038/cddis.2015.298. [DOI] [PMC free article] [PubMed] [Google Scholar]