Abstract
Objective
To summarize information on the effects of opioid use in pregnancy on subsequent pediatric development and behaviour.
Data sources
Searches were performed in EMBASE, MEDLINE, and PsycINFO for peer-reviewed, English articles, including a manual search of their references, that were published between January 1, 2000, and May 1, 2018.
Study selection
Of the 543 articles reviewed, 19 relevant articles that focused on developmental effects of opioid exposure in utero were identified. Most of the studies provided level II evidence. One level I meta-analysis and 1 level III expert committee report were included.
Synthesis
The literature was divided between documenting some level of impairment or normalization of early development deficits over time. Often no opioid effect was found once researchers controlled for socioenvironmental factors. The degree to which environmental factors, opioid exposure, or both affect pediatric development remains to be determined.
Conclusion
The effect of maternal opioid use on pediatric development is unclear and the evidence is inconsistent. However, opioid exposure in pregnancy does define these children as a population at risk. They might experience developmental delays compared with their peers, yet remain within population norms in cognition, fine-motor skills, hand-eye coordination, executive function, and attention and impulsivity levels.
Résumé
Objectif
Résumer l’information concernant les effets de l’utilisation d’opioïdes durant la grossesse sur le développement et les comportements pédiatriques subséquents.
Sources des données
Des recherches documentaires ont été effectuées dans EMBASE, MEDLINE et PsycINFO en quête d’articles en anglais révisés par des pairs, de même qu’une recension manuelle dans leurs références, dont les dates de publication se situaient entre le 1er janvier 2000 et le 1er mai 2018.
Sélection des études
Parmi les 543 articles passés en revue, nous avons cerné 19 articles pertinents qui portaient sur les effets d’une exposition in utero aux opioïdes. La plupart des études ont présenté des données probantes de niveau II. Nous avons aussi inclus 1 méta-analyse de niveau I et 1 rapport d’un comité d’experts de niveau III.
Synthèse
Les conclusions des ouvrages scientifiques étaient divisées entre la documentation d’un certain degré de déficience ou la normalisation des déficits du développement précoces avec le temps. Souvent, aucun effet des opioïdes n’a été constaté après rajustements par les chercheurs pour tenir compte des facteurs socio-environnementaux. Il reste à déterminer la mesure dans laquelle les facteurs environnementaux, l’exposition aux opioïdes ou les 2 nuisent au développement pédiatrique.
Conclusion
Les effets de l’utilisation d’opioïdes par la mère sur le développement pédiatrique demeurent incertains, et les données factuelles ne sont pas cohérentes. Par contre, les enfants exposés aux opioïdes pendant la grossesse de leur mère sont effectivement désignés comme une population à risque. Ils pourraient connaître des retards du développement par rapport à leurs pairs, mais demeurent quand même dans la norme populationnelle sur les plans de la cognition, de la motricité fine, de la coordination œil-main, de la fonction exécutive, ainsi que des degrés d’attention et d’impulsivité.
Opioid use in Canada poses an increasing health risk. Canadian hospitalizations from opioid overdoses increased by 53% between 2007 and 2017.1 Opioid-related deaths increased in 1 year by 40% in 2017.2
This opioid crisis extends to pregnancy: 6.7% of Canadian mothers reported using street drugs in the 3 months before conception, with 1% to 4% continuing to use throughout the pregnancy.3,4 Opioid use in pregnancy is more prevalent in certain regions of the country, particularly in the remote First Nations communities of northwestern Ontario, where up to 30% of pregnancies include opioid exposure.5 Neonates are also affected; the national incidence of neonatal abstinence syndrome (NAS) increased 17% from 4.3 to 5.2 per 1000 live births from 2013 to 2017.6
The effect of illicit or prescribed opioid use on child development is unclear. Maintenance with opioid agonist therapy (OAT) is commonly instituted in pregnancy to stabilize drug intake and encourage a healthier pregnancy.7,8 While NAS is well documented, opioid effects beyond the newborn period are unknown and might affect the child’s and family’s primary care needs. This systematic review considers the evidence on cognitive and behavioural development in children older than age 2 following in utero opioid exposure.
DATA SOURCES
Searches were performed in EMBASE, MEDLINE, and PsycINFO for peer-reviewed, English articles, including a manual search of their references, that were published between January 1, 2000, and May 1, 2018. We chose studies published since 2000 to cover a time frame with more recent types of drug use and pediatric assessment strategies.
Study selection
Search terms combined opioids and opiates and opioid and opiate agonists with pregnancy and maternal exposure, child development, and pediatrics, and with a variety of child development characteristics (cognition, memory, learning, language, intellectual, fine-motor skills, and behaviour). We excluded studies if they included subjects who had widespread concurrent alcohol use (if among > 25% of mothers) or polysubstance use (if > 50% of mothers used a nonopioid substance). Evidence was categorized using the Canadian Task Force on Preventive Health Care guidelines.
SYNTHESIS
Literature search results
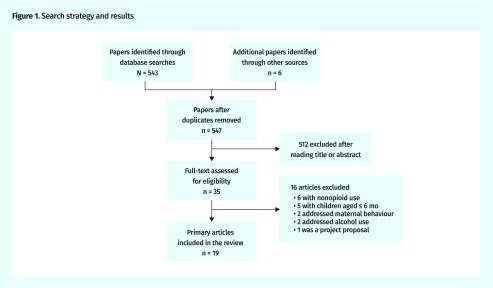
We identified 543 unique papers whose abstracts were screened for children older than age 2 and who were born to mothers using opioids or taking OAT with measured development and behavioural outcomes. Nineteen research studies9–27 met these criteria (Figure 1). Twelve of these took place in Scandinavian countries where the primary opioid exposure was heroin or methadone.9–18,20,24 A set of 6 Norwegian studies initially followed 78 participants over the course of their childhood, some into adulthood.9–14 Studies were small; 5 of them had fewer than 35 subjects.16,18,20,23,24 One study was population-based with 892 subjects.15
Figure 1.
Search strategy and results
Seventeen studies were prospective, of which 15 included multivariate analysis.9–14,16–24 Methodology was mainly case-control studies,9–14,16–23 accompanied by a case series24 and a 2014 meta-analysis of 4 studies that provided the only level 1 evidence.25 Most evidence was level II. One well-informed expert committee report (level III evidence) was also included.26
The 19 studies reviewed here examine the effect of opioid exposure in pregnancy on pediatric development, including cognition, motor skills, language, and attention and behaviour outcomes (Table 1).9–27 The studies had limitations and variable findings. The studies are broadly distributed in their scope and findings.
Table 1.
Summary of studies on childhood development following prenatal opioid exposure
| STUDY | NO. OF PARTICIPANTS | AGE AT EVALUATION | MAIN EXPOSURE | FINDINGS | LEVEL OF EVIDENCE |
|---|---|---|---|---|---|
| Most recent | |||||
| • Kaltenbach et al,27 2018 | 96 | Birth to 3 y | Methadone or buprenorphine | Normal physical, cognitive, language, sensory, and behavioural development | III |
| Population-based | |||||
| • Skovlund et al,15 2017 | Cohort of 892 | 3 y | Opioids | Trend toward effect of analgesic opioid use during pregnancy on childhood language development and communication | II |
| Longitudinal case series* | 45 to 78 | Opioids (mainly heroin) | |||
| • Nygaard et al,12 2017 | 17–21 y | Cognitive and fine-motor functions below those of peers | II | ||
| • Nygaard et al,11 2015 | 1–8.5 y | Cognition below peers | II | ||
| • Walhovd et al,14 2007 | 11.3 y | Lower neuroanatomical volumes than those of peers | II | ||
| • Slinning, 200413 | 1–4.5 y | Cognitive and motor deficits compared with peers; impulsivity and attention-related problems | II | ||
| • Moe, 200210 | 1–4.5 y | Development assessments scored below peers; specific weakness in visual-motor abilities | II | ||
| • Moe and Slinning,9 2001 | 1–3 y | Development assessments scored below peers | II | ||
| Case-control studies | |||||
| • Konijnenberg and Melinder,17 2015 | 35 | 4 y | OMT | Short-term memory, inhibition, and executive function below peers | II |
| • Konijnenberg and Melinder,16 2013 | 15 | 4 y | OMT | Difficulty using proactive goal-directed eye movements | II |
| • Melinder et al,18 2013 | 26 | 4 y | OMT | Behavioural and visual-motor deficits compared with peers | II |
| • Wahlsten and Sarman,24 2013 | 25 | 6 y | Opioids | Problems with motor skills, memory, hyperactivity, impulsivity, and attention | III |
| • Salo et al,20 2009 | 21 | Birth to 3 y | Buprenorphine | Developmental deficits compared with peers | II |
| • Hunt et al,22 2008 | 133 | 18 mo and 3 y | Methadone | Delays in mental, motor, and social development compared with peers | II |
| • Messinger et al,21 2004 | 50 | 3 y | Opioids | No cognitive delay at 3 y | II |
| • Hans and Jeremy,23 2001 | 33 | Birth to 2 y | Opioids (mainly methadone) | Delay in mental development and motor functioning compared with peers | II |
| • Ornoy et al,19 2001 | 65 | 5 to 12 y | Heroin, OMT | Higher levels of inattention, hyperactivity, and behaviour problems than peers | II |
| Meta-analysis | |||||
| • Baldacchino et al,25 2014 | NA | Birth to 12 y | Opioids | Impairments in cognitive, psychomotor, and behavioural development compared with peers | I |
| Expert panel | |||||
| • Reddy et al,26 2017 | NA | From birth throughout childhood | Opioids | No consensus on the long-term effects of in utero opioid exposure on development; evidence is inconsistent | III |
NA—not applicable, OMT—opioid maintenance therapy.
These 6 studies comprise a longitudinal case series following the same cohort from birth to 17–21 years of age.
A 2017 American expert consensus workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development and several pediatric and obstetric societies concluded that there is a lack of evidence regarding the long-term effects of prenatal opioid exposure. The executive summary noted that existing study results were inconsistent, were of small sample size, were conducted before the current opioid epidemic, and had multiple confounders (level III evidence).26
A 2014 systematic review and meta-analysis identified 4 of 200 studies (3 of which are included in our review10,22,23; 1 predates our time frame) that quantitatively reported on neurobehavioural development after maternal opioid use in pregnancy (level I evidence).25
The largest study (N = 892) examined only language and communication skills in 892 Norwegian children exposed to opioids and found no difference from the general pediatric population.15 The most recent 2018 study (N = 96) found no deleterious effects on physical and mental development by 3 years of age and concluded that opioid use in pregnancy was safe (level II evidence).27
Studies that documented specific developmental delay in exposed children acknowledged the difficulty in distinguishing specific opioid exposure effects from the influence of growing up in a home environment where addiction was present.16–20,22,23 This socioenvironmental setting might include unstable relationships and bonding, poverty, poor nutrition, and increased stress (level II evidence).
These factors limit the ability to ascribe any documented delays specifically to the effects of opioid exposure. The likelihood of undocumented maternal polysubstance use also compromises accurate ascribing of any developmental concerns specifically to opioid exposure. Other influences not controlled for in these studies are gestational age and low birth weight; factors that carry their own development considerations and are not uncommon in opioid-exposed pregnancy.
Findings from the studies
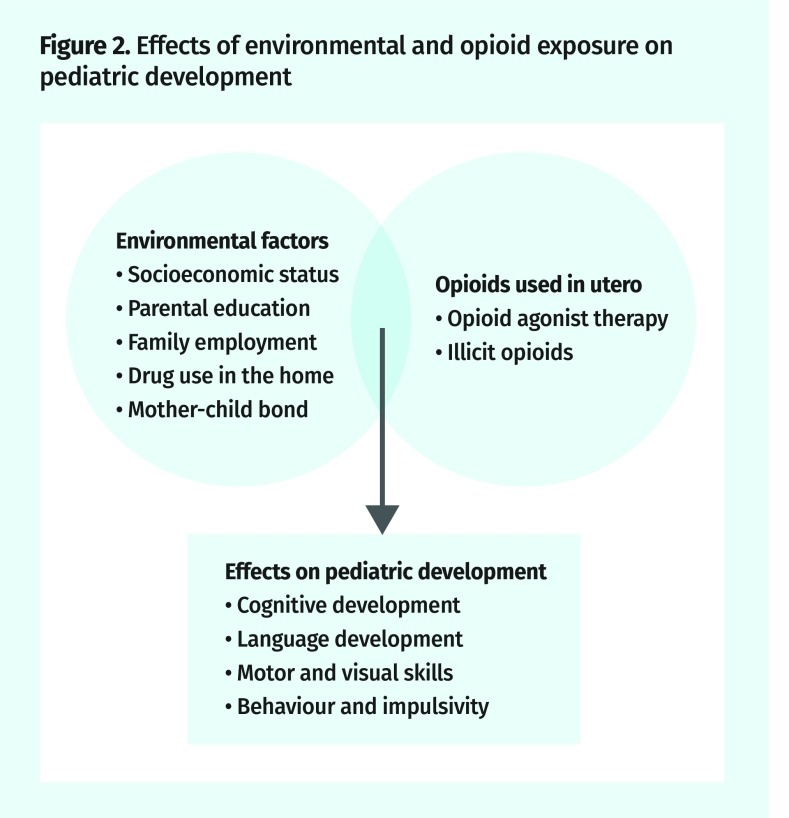
The effect of in utero opioid exposure on subsequent pediatric development is unclear and the evidence inconsistent. While no definitive opioid effect has been established, these children can be considered a population at risk, with fetal exposure to illicit drugs as a marker of socioenvironmental risk (Figure 2).
Figure 2.
Effects of environmental and opioid exposure on pediatric development
Cognitive development.
Nine studies assessed cognitive development in the preschool years, one of which followed the exposed cohort into adolescence.9,10,13,17,20–23,27 Observed cognitive deficits were more strongly associated with socioenvironmental influences than with drug use. In a series of Norwegian studies of 78 children at 1, 3, 4.5, and 8.5 years, general cognitive development and perceptual abilities were lower than nonexposed children but remained within population norms.9–11,13 In these studies, cognitive deficits built throughout childhood until 8 years of age, after which deficits remained stable with exposed children at a slight deficit compared with their peers.11,12 Forty-five of the original 78 children were followed into adulthood; among them, small cognitive deficits persisted at ages 17 to 21, despite having been adopted before the age of 1.12 The study found that by age 4.5, parental socioeconomic status substantially affected perceptual performance (level II evidence).10
An environmental effect on cognitive development was also supported by a 2001 study of 33 American opioid-exposed children; it found that initial differences in mental development were no longer significant once environmental risk was controlled for (level II evidence).23
Studies from Norway10 and Sweden24 that considered parents’ socioeconomic levels found memory deficits at ages 4.5 to 6, while 2 other unrelated studies found no deficits when controlling for socioeconomic status19 or maternal education17 (level II evidence).
Language.
Five studies that measured language development found that children with prenatal opioid exposure experience a delay in language development that improves over time.19,20,22,24,27 Two of these studies controlled for home environment19,20 and again identified an environmental, with a strongly positive “adoption-related,” effect on language development (N = 86) (level II evidence).
An international randomized controlled study comparing treatment with methadone versus buprenorphine followed 37 exposed children from birth to age 3 as a subset of the MOTHER (Maternal Opioid Treatment: Human Experimental Research) study. The study found similar outcomes (level I evidence).27 Effects include early deficits in language, physical growth, cognitive development, sensory processing, and temperament, which normalized by age 3; the authors concluded that prenatal opioid exposure was not deleterious to normal development (level III evidence).27 This contrasts with the findings of 3 other studies that showed ongoing impaired language and verbal skills at ages 3 and 6 (level II evidence).20,22,24
Motor and visual skills.
Visual and fine-motor skills were often assessed simultaneously. A range of motor skills are variably affected in young children with in utero opioid exposure. Three studies showed an early delay in motor skills in infancy that reached population norms by ages 2 to 3.9,23,27 Moe and Slinning’s 2001 study found a delay in psychomotor development (general coordination, finer manipulation of the hands, and recognizing objects by sense of touch) in exposed children during their first year of age that had reached the level of their peers by ages 2 to 3 (level II evidence).9
Kaltenbach and colleagues’ MOTHER study findings included early delays in motor skills; these delays improved by the age of 3 to being within population norms, but exposed children remained below unexposed peers matched for similar socioeconomic status.22,27 In contrast, Wahlsten and Sarman’s 2013 study of 25 children’s motor development (coordination, imitating actions, and drawing specific objects) remained below normal at ages 5 to 6 (level II evidence).24
Four-year-olds who were exposed to opioid maintenance therapy had specific fine-motor issues in the Konijnenberg et al and the Melinder et al studies.16,18 In the first study the deficit disappeared when controlling for maternal education,16 but it remained in the latter study.18 Both studies found impaired goal-directed eye movement,16,18 suggesting issues with visually tracking objects (level II evidence)18 that contributed to fine-motor control and coordination performance.16,18 When participants were followed into adulthood, Nygaard and colleagues found fine-motor skills had not caught up to the skills of peers by age 17 to 21 (n = 45) and proposed a contributing effect of impaired executive function (level II evidence).12
Behaviour and attention deficit hyperactivity disorder (ADHD).
A longitudinal Norwegian study included assessments of behaviour and impulsivity; behaviour typically measured in the normal population range but below the level of the study control group. The range of behaviour issues examined included attention, impulsivity deficits, ADHD diagnoses, and executive function.
The first series of measurements at age 2 and 4 indicated increased impulsivity and attention problems in heroin-exposed children.13 The authors found attention deficits and hyperactivity presented as early as age 2 and worsened by age 4.5 (n = 42) (level II evidence).13 These deficits were associated with perceptual performance and motor abilities. Those children with more behaviour issues also tended to score lower in intelligence, memory, and motor tests.13 In school-aged Swedish children (aged 5 to 6), teacher questionnaires indicated ADHD in all the subset of 16 children tested, as well as issues with hyperactivity and conduct problems (level II evidence).24 Behavioural deficits were present regardless of whether children had been adopted at an early age.24 In contrast, the participants in these studies performed best in neuropsychological testing if they had been placed in an adoptive family at an early age12,24 and were born to a mother who used the lowest drug dosage.12 By adulthood, executive function was now within the population norm but still below that of the control group. Again, those who performed the best were born to mothers who used the lowest opioid dosage and were placed with foster or adoptive parents at an early age (level II evidence).12
Parental ADHD might contribute to the attention problems in the children,19,24 and parents with opioid use disorder have a high prevalence of ADHD.19 In Ornoy and colleagues’ 2001 study, an increased prevalence of ADHD in exposed children was associated with concurrent maternal ADHD (level II evidence).19 Attention deficit hyperactivity disorder was also implicated among parents and family members in a Swedish study of pregnancies exposed to opiate maintenance treatment with buprenorphine.24 Wahlsten and Sarman studied 25 buprenorphine-OAT pregnancies and performed development and behavioural testing in children aged 5 to 6. Exposed children showed signs of problems with motor skills and hyperactivity, and a high frequency of ADHD was seen in parents and other family members (level II evidence).24
Socioeconomic status also affects behaviour and attention. Kaltenbach and colleagues’ 2018 study found children of low socioeconomic status differed in attention regardless of prenatal opioid exposure.27 Boys seemed to be more adversely affected. Two studies found they performed worse on tasks related to development, cognition, memory, motor skills, and attention (level II and III evidence).9,10,13,24
Deficits in one area might have broad effects. Konijnenberg and Melinder suggest that opioid exposure in pregnancy might change neurotransmitter levels in the developing brain, affecting social, motor, and cognitive development.16 The affected processes are interrelated. Visual deficits affect cognitive scores, hand-eye coordination, and executive function.12,16,18 Behaviour issues are coincident with cognition and memory problems.13 Attention issues affect cognitive and motor performance, and children with impaired cognition, memory, and motor skills had more difficulties with attention in Slinning’s 2004 Norwegian study (level II evidence).13
Environmental factors and development.
Numerous studies recognized the role of socioeconomic status, maternal education, employment, drug use in the household, and emotional attachment in pediatric development.16,17,19,20 The bond between mother and child is particularly important for development in drug-exposed children.28 The Salo et al and Kaltenbach et al studies documented low self-esteem and self-assessed maternal ability in mothers with opioid use disorder.20,27 In Salo and colleagues’ study of 21 buprenorphine-exposed Finnish children, childhood emotional responsiveness at 3 years of age mirrored measured maternal emotional capacity and was lower than controls.20 Mothers included in most studies had lower educational levels than the general population9,16–19,21 and nearly 50% unemployment rates (level II evidence).9,16–18
Some studies controlled for environment effects by comparing exposed children who were adopted or fostered to those remaining in the care of their birth mothers.13,19,20 Norwegian young adults (n = 45) who had been placed in permanent foster or adoptive homes at a young age still experienced slight deficits in cognition, memory, fine-motor skills, and executive function that remained into adulthood.12 In exposed Finnish (n = 14) and Israeli children (n = 34) aged 5 to 12, adoption mitigated cognitive differences relative to peers (level II evidence).19,20
Adoptive status had a substantial effect on intellectual performance in the Ornoy et al study of 160 Israeli children exposed to prenatal heroin and methadone replacement therapy.19 The 34 adopted, exposed children had better intellectual performance than those who remained with birth parents.19 Ornoy and colleagues commented that Israeli adoptions are often to affluent families,19 and Nygaard and colleagues similarly noted the socioeconomic status of adoptive Norwegian families was relatively high.12
Children in families with opioid exposure experience more upheaval in living circumstances. In Hunt and colleagues’ study of children of 133 mothers using methadone maintenance therapy, 28% had been adopted or placed in foster care at 18 months, rising to 32% by age 3; 10 mothers had died from opioid-related causes as well as 3 fathers.22 The unexposed comparison group (n = 103), matched for socioeconomic status, were all living with their birth parents.22
In Moe’s study of exposed children aged 1 to 4.5, both maternal opioid use and socioeconomic status made independent contributions to observed deficits in cognitive development (n = 64).10 Similarly, in Salo and colleagues’ study, no cognitive deficits were found when subjects were compared with peers of similar socioeconomic status.20 Moe and Slinning also found adoption status to be protective. The 66% (of 78) of adopted, exposed children experienced a “catch up” in cognitive function by age 3 (level II evidence).9
The dose of maternal opioid use was not predictive of any developmental delay in 4 separate studies; nor was the presence or severity of NAS at birth (level II evidence).16,17,24,27
Detection and follow-up
These studies note the importance of monitoring of childhood cognitive, motor, visual, language, and behavioural development.13,17,24 Family physicians should be aware of early intervention pediatric services and allied health professionals’ services available to these at-risk children.
Postpartum support for mothers should include consistent follow-up, breastfeeding support, education, and mental health care for parents, as well as social services assessments of care to the newborns and other children.26 Interventions at the environmental level can affect development to some degree; however, there is evidence that the children might not be able to fully “catch up” to their peers (level II evidence).12
The Parent-Child Assistance Program developed in Washington State in 1991 led to the institution of Alberta’s Parent Child Assistance Program in 2006. These programs provide comprehensive family services to families dealing with addiction. Postpartum case management and home visits for a 3-year period have demonstrated improved health for at-risk mothers and their children, with a decrease in subsequent affected pregnancies and child protective custody referrals.29,30 Programs targeting caregiver stability and mother-child bonding are difficult to provide.17,19,20 More focused group and one-on-one psychotherapy and psychosocial support targeting mother-child interaction and practical counseling are even more resource intense but succeed in improving the relationship between child and mother.31 Mothers with opioid use disorder have also been shown to benefit from long-term confidential relationships with a small number of clinicians,32 a role suited for the family physician (level II evidence).
Limitations
The main limitation in this review is the heterogeneity of both the study participants and the variety of assessment tools used. It was challenging to isolate relevant clinical categories from a set of studies that did not share the same developmental focus.
Conclusion
The effect of maternal opioid use on pediatric development is unclear. However, studies on opioid exposure in pregnancy define these children as a population at risk. These children might experience developmental delays compared with their peers, yet remain within population norms in cognition, fine-motor skills, hand-eye coordination, executive function, and attention and impulsivity levels. The literature was divided between documenting some level of impairment or normalization of early deficits over time. Often no opioid effect was found once researchers controlled for socioenvironmental factors. The degree to which environmental factors, opioid exposure, or both affect pediatric development remains to be determined.
Editor’s key points
▸ The effect of illicit or prescribed opioid use on child development is unclear. Maintenance with opioid agonist therapy is commonly instituted in pregnancy to stabilize drug intake and encourage a healthier pregnancy. While neonatal abstinence syndrome is well documented, opioid effects beyond the newborn period are unknown and might affect the child’s and family’s primary care needs. This systematic review considers the evidence on cognitive and behavioural development in children older than age 2 following in utero opioid exposure.
▸ The effect of in utero opioid exposure on subsequent pediatric development is unclear and the evidence inconsistent. Socioenvironmental factors might have more influence than opioid exposure does. Following the systematic review of the evidence, the authors note the importance of monitoring of childhood cognitive, motor, visual, language, and behavioural development. Family physicians should be aware of early intervention pediatric services, as well as allied health professional services, available to these at-risk children.
Points de repère du rédacteur
▸ Les effets de l’utilisation d’opioïdes illicites ou prescrits sur le développement des enfants demeurent nébuleux. Durant la grossesse, il est courant d’amorcer une thérapie de maintien aux agonistes opioïdes pour stabiliser la consommation de drogues et favoriser une grossesse plus saine. Même si le syndrome d’abstinence néonatale est bien documenté, les effets des opioïdes au-delà de la période néonatale sont inconnus et pourraient avoir une influence sur les besoins en soins primaires de l’enfant et de la famille. Cette revue systématique examine les données probantes concernant le développement cognitif et comportemental chez les enfants de plus de 2 ans à la suite d’une exposition in utero aux opioïdes.
▸ Les répercussions d’une exposition in utero aux opioïdes sur le développement pédiatrique subséquent ne sont pas bien connues, et les données factuelles manquent de cohérence. Les facteurs socio-environnementaux pourraient avoir une plus grande influence que l’exposition aux opioïdes. À la suite de la revue systématique des données probantes, les auteurs ont fait remarquer l’importance de surveiller le développement cognitif, moteur, visuel, langagier et comportemental durant l’enfance. Les médecins de famille devraient connaître les services pédiatriques d’intervention précoce, de même que les services offerts par d’autres professionnels de la santé qui sont à la disposition de ces enfants à risque.
Footnotes
Contributors
All authors contributed to the concept and design of the study; data gathering, analysis, and interpretation; and preparing the manuscript for submission.
Competing interests
None declared
This article has been peer reviewed.
Cet article a fait l’objet d’une révision par des pairs.
References
- 1.Canadian Institute for Health Information . Opioid-related harms in Canada. Ottawa, ON: Canadian Institute for Health Information; 2017. Available from: https://secure.cihi.ca/free_products/opioid-harms-chart-book-en.pdf. Accessed 2019 Nov 8. [Google Scholar]
- 2.Public Health Agency of Canada [website] National report: apparent opioid-related deaths in Canada. Ottawa, ON: Public Health Agency of Canada; 2018. Available from: www.canada.ca/en/public-health/services/publications/healthy-living/national-report-apparent-opioid-related-deaths-released-june-2018.html#a2.1. Accessed 2019 Nov 8. [Google Scholar]
- 3.Public Health Agency of Canada . What mothers say: the Canadian Maternity Experiences Survey. Ottawa, ON: Public Health Agency of Canada; 2009. Available from: www.phac-aspc.gc.ca/rhs-ssg/pdf/survey-eng.pdf. Accessed 2019 Nov 8. [Google Scholar]
- 4.Falk J, Dahl M, Raymond CB, Chateau D, Katz A, Leong C, et al. Opioid use during pregnancy: a population-based cohort study. CMAJ Open. 2017;5(2):E517–23. doi: 10.9778/cmajo.20160102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nishnawbe Aski Nation . Resolution: 09/92. Prescription drug abuse state of emergency. Thunder Bay, ON: Nishnawbe Aski Nation; 2009. [Google Scholar]
- 6.Canadian Institute for Health Information . Opioid-related harms in Canada. Ottawa, ON: Canadian Institute for Health Information; 2018. Available from: https://www.cihi.ca/en/opioids-in-canada/2018/opioid-related-harms-in-canada. Accessed 2019 Nov 20. [Google Scholar]
- 7.Fischer G, Johnson RE, Eder H, Jagsch R, Peternell A, Weninger M, et al. Treatment of opioid-dependent pregnant women with buprenorphine. Addiction. 2000;95(2):239–44. doi: 10.1046/j.1360-0443.2000.95223910.x. [DOI] [PubMed] [Google Scholar]
- 8.Svikis D, Lee J, Haug N, Stitzer M. Attendance incentives for outpatient treatment: effects in methadone- and nonmethadone-maintained pregnant drug dependent women. Drug Alcohol Depend. 1997;48(1):33–41. doi: 10.1016/s0376-8716(97)00101-4. [DOI] [PubMed] [Google Scholar]
- 9.Moe V, Slinning K. Children prenatally exposed to substances: gender-related differences in outcome from infancy to 3 years of age. Infant Ment Health J. 2001;22(3):334–50. [Google Scholar]
- 10.Moe V. Foster-placed and adopted children exposed in utero to opiates and other substances: prediction and outcome at four and a half years. J Dev Behav Pediatr. 2002;23(5):330–9. doi: 10.1097/00004703-200210000-00006. [DOI] [PubMed] [Google Scholar]
- 11.Nygaard E, Moe V, Slinning K, Walhovd KB. Longitudinal cognitive development of children born to mothers with opioid and polysubstance use. Pediatr Res. 2015;78(3):330–5. doi: 10.1038/pr.2015.95. Epub 2015 May 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nygaard E, Slinning K, Moe V, Walhovd KB. Cognitive function of youths born to mothers with opioid and poly-substance abuse problems during pregnancy. Child Neuropsychol. 2017;23(2):159–87. doi: 10.1080/09297049.2015.1092509. Epub 2015 Oct 16. [DOI] [PubMed] [Google Scholar]
- 13.Slinning K. Foster placed children prenatally exposed to poly-substances. Eur Child Adolesc Psychiatry. 2004;13(1):19–27. doi: 10.1007/s00787-004-0350-x. [DOI] [PubMed] [Google Scholar]
- 14.Walhovd KB, Moe V, Slinning K, Due-Tønnessen P, Bjørnerud A, Dale AM, et al. Volumetric cerebral characteristics of children exposed to opiates and other substances in utero. Neuroimage. 2007;36(4):1331–44. doi: 10.1016/j.neuroimage.2007.03.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Skovlund E, Handal M, Selmer R, Brandlistuen RE, Skurtveit S. Language competence and communication skills in 3-year-old children after prenatal exposure to analgesic opioids. Pharmacoepidemiol Drug Saf. 2017;26(6):625–34. doi: 10.1002/pds.4170. [DOI] [PubMed] [Google Scholar]
- 16.Konijnenberg C, Melinder A. Neurodevelopmental investigation of the mirror neurone system in children of women receiving opioid maintenance therapy during pregnancy. Addiction. 2013;108(1):154–60. doi: 10.1111/j.1360-0443.2012.04006.x. Epub 2012 Oct 5. [DOI] [PubMed] [Google Scholar]
- 17.Konijnenberg C, Melinder A. Executive function in preschool children prenatally exposed to methadone or buprenorphine. Child Neuropsychol. 2015;21(5):570–85. doi: 10.1080/09297049.2014.967201. [DOI] [PubMed] [Google Scholar]
- 18.Melinder A, Konijnenberg C, Sarfi M. Deviant smooth pursuit in preschool children exposed prenatally to methadone or buprenorphine and tobacco affects integrative visuomotor capabilities. Addiction. 2013;108(12):2175–82. doi: 10.1111/add.12267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ornoy A, Segal J, Bar-Hamburger R, Greenbaum C. Developmental outcome of school-age children born to mothers with heroin dependency: importance of environmental factors. Dev Med Child Neurol. 2001;43(10):668–75. doi: 10.1017/s0012162201001219. [DOI] [PubMed] [Google Scholar]
- 20.Salo S, Kivistö K, Korja R, Biringen Z, Tupola S, Kahila H, et al. Emotional availability, parental self-efficacy beliefs, and child development in caregiver-child relationships with buprenorphine-exposed 3-year-olds. Parenting. 2009;9(3–4):244–59. [Google Scholar]
- 21.Messinger DS, Bauer CR, Das A, Seifer R, Lester BM, Lagasse LL, et al. The maternal lifestyle study cognitive, motor, and behavioral outcomes of cocaine exposed and opiate exposed infants through three years of age. Pediatrics. 2004;113(6):1677–85. doi: 10.1542/peds.113.6.1677. [DOI] [PubMed] [Google Scholar]
- 22.Hunt RW, Tzioumi D, Collins E, Jeffery HE. Adverse neurodevelopmental outcome of infants exposed to opiate in-utero. Early Hum Dev. 2008;84(1):29–35. doi: 10.1016/j.earlhumdev.2007.01.013. Epub 2007 Aug 28. [DOI] [PubMed] [Google Scholar]
- 23.Hans SL, Jeremy RJ. Postneonatal mental and motor development of infants exposed in utero to opioid drugs. Infant Ment Health J. 2001;22(3):300–15. [Google Scholar]
- 24.Wahlsten VS, Sarman I. Neurobehavioural development of preschool-age children born to addicted mothers given opiate maintenance treatment with buprenorphine during pregnancy. Acta Paediatr. 2013;102(5):544–9. doi: 10.1111/apa.12210. Epub 2013 Mar 20. [DOI] [PubMed] [Google Scholar]
- 25.Baldacchino A, Arbuckle K, Petrie DJ, McCowan C. Neurobehavioral consequences of chronic intrauterine opioid exposure in infants and preschool children: a systematic review and meta-analysis. BMC Psychiatry. 2014;14:104. doi: 10.1186/1471-244X-14-104. Erratum in: BMC Psychiatry 2015;15:134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Reddy U, Davis J, Ren Z, Greene M. Opioid use in pregnancy, neonatal abstinence syndrome, and childhood outcomes. Executive summary of a Joint Workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, American Congress of Obstetricians and Gynecologists, American Academy of Pediatrics, Society for Maternal-Fetal Medicine, Centers for Disease Control and Prevention, and the March of Dimes Foundation. Obstet Gynecol. 2017;130(1):10–28. doi: 10.1097/AOG.0000000000002054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kaltenbach K, O’Grady K, Heil SH, Salisbury AL, Coyle M, Fischer G, et al. Prenatal exposure to methadone or buprenorphine: early childhood developmental outcomes. Drug Alcohol Depend. 2018;185:40–9. doi: 10.1016/j.drugalcdep.2017.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brown JV, Bakeman R, Coles CD, Platzman KA, Lynch ME. Prenatal cocaine exposure: a comparison of 2-year-old children in parental and nonparental care. Child Dev. 2004;75(4):1282–95. doi: 10.1111/j.1467-8624.2004.00739.x. [DOI] [PubMed] [Google Scholar]
- 29.Grant T, Ernst C. Parent-Child Assistance Program (PCAP): prevention & intervention with high-risk mothers and their children. Washington, DC: University of Washington Alcohol and Drug Abuse Institute; 2019. Available from: http://depts.washington.edu/pcapuw/inhouse/PCAP_Summary_of_Evidence.pdf. Accessed 2019 Nov 8. [Google Scholar]
- 30.Family Services of Central Alberta [website] Parent Child Assistance Program. Red Deer, AB: Family Services of Central Alberta; Available from: https://fsca.ca/parent-and-child-assistance-program-pcap. Accessed 2019 Nov 8. [Google Scholar]
- 31.Belt RH, Flykt M, Punamäki R, Pajulo M, Posa T, Tamminen T. Psychotherapy groups and individual support to enhance mental health and early dyadic interaction among drug-abusing mothers. Infant Ment Health J. 2012;33(5):520–34. doi: 10.1002/imhj.21348. [DOI] [PubMed] [Google Scholar]
- 32.Suchman NE, DeCoste C, Castiglioni N, McMahon TJ, Rounsaville B, Mayes L. The mothers and toddlers program, an attachment-based parenting intervention for substance using women: post-treatment results from a randomized clinical pilot. Attach Hum Dev. 2010;12(5):483–50. doi: 10.1080/14616734.2010.501983. [DOI] [PMC free article] [PubMed] [Google Scholar]