Abstract
Purpose
To evaluate the biomechanical efficacy and safety of in vivo microbial transglutaminase (Tgases)-induced corneal crosslinking in a rabbit model.
Methods
A total of 34 white New Zealand rabbits were divided into two groups, a biochemistry group and a photochemistry group. The right eye of every rabbit was treated and left eyes served as negative controls. In the biochemistry group, a 1 U/mL solution of crosslinking agent microbial Tgases (Tgases CXL) was applied to the corneal surface, while in the photochemistry group, clinical ultraviolet A-riboflavin crosslinking (UVA/RF CXL) was used. Efficacy and safety evaluated on the 14th day after the procedures. Twelve pairs of corneal strips were harvested from the eyes of 12 euthanized rabbits in every group, and uniaxial tensile tests were performed to evaluate ex vivo biomechanical effects. The CXL-treated eye to its corresponding untreated eye ratio of tangent modulus were calculated. Another five pairs of corneal button were excised from euthanized animals in every group for corneal stroma and endothelium staining to evaluate changes in keratocyte distribution and endothelial cell damage.
Results
In tensile tests, tangent modulus was statistically higher in the Tgases CXL groups under 1.0 MPa (26.59 ± 4.54 vs. 21.47 ± 4.72 MPa, P = 0.04) and 1.5 MPa (29.75 ± 5.01 vs. 20.47 ± 6.63 MPa, P = 0.00). The tangent modulus ratio of Tgases group (1.72 ± 1.0 vs. 1.05 ± 0.22, P = 0.04) was significantly higher than that of UVA/RF under 1.5-MPa stress. The distribution of keratocytes in the corneal stroma and the morphologies of endothelial cells were similar in Tgases CXL-treated and untreated corneas. However, in the UVA/RF CXL group, keratocytes in the anterior half of stromal thickness were lost, and clear endothelial cell apoptosis was observed.
Conclusions
Tgases-CXL effectively stiffened the cornea and caused no damage to the endothelium and keratocytes in the cornea. This crosslinking method could be useful as a next-generation treatment for corneal ectasia and could replace CXL of photochemistry.
Translational Relevance
These findings may give a new hope to biomechanically compromised corneal disease due to mechanical forces, such as corneal ectasia and keratoconus. A next-generation treatment to these corneal diseases due to mechanical forces may be designed based on the new findings.
Keywords: transglutaminase, cornea crosslinking, biomechanics
Introduction
Corneal ectasia is a group of eye disorders characterized by bilateral thinning and distortion of the central, paracentral, or peripheral cornea. The primary forms are keratoconus and ectasia after refractive surgery.1 A conservative approach to the management of keratectasia is spectacles and rigid gas-permeable (RGP) contact lenses. Additionally, surgical interventions, such as keratoplasty and intracorneal ring segment implantation, can be necessary in patients with rapid progression or RGP intolerance, acute corneal hydrops, and central corneal scarring. However, this treatment option for corneal ectasia is aimed only to overcome refractive limitations and does not affect the underlying physiopathology.2 On the other hand, corneal crosslinking (CXL) is a management method that can possibly stiffen corneal tissue and has recently become a common method in treatment of corneal ectasia.3
The only CXL treatment currently applied in a clinical setting is ultraviolet-A and riboflavin (UVA/RF) crosslinking. This method involves the de-epithelialization of the cornea, soaking the corneal stroma with a chromophore (riboflavin) and UVA irradiation.4 The method has shown effectiveness in stopping the progression of keratoconus and arresting postsurgical corneal ectasia.4–6 It has been proposed that the chromophore absorbs UVA to act as a photosensitizer to produce free-radical (oxygen singlet)-induced collagen CXL by activating the natural lysyl oxidase pathway.3 Recently, the mRNA and protein levels of fibronectin and transglutaminases (Tgases) were found to be higher in human corneal keratocytes treated with UVA/RF, and the induction of Tgases in the cornea was proposed as a new mechanism for inducing crosslinking.7,8 A possible advantage of Tgases CXL is avoidance of the loss of keratocytes and the discomfort in patients commonly associated with UVA/RF CXL.9 In this study, we sought to test the biomechanical and histologic changes that occur in corneal tissues treated with microbial Tgases and evaluate the validity and safety of this novel method of CXL.
Materials and Methods
Animal Model
Thirty-four healthy, female, 2-month-old rabbits (3.0–3.5 kg) were divided into two equal groups to study the efficacy and safety of CXL with Tgases and standard UVA/RF. All animals were provided by and housed at Peking University First Hospital Animal Center and had ad libitum access to food and water. All animal experiments were performed in accordance with the Chinese Ministry of Science and Technology Guidelines on Human Treatment of Laboratory Animals (Vgkfcz-2006-398) and the ARVO Statement on the use of Animals in Ophthalmic and Vision Research. This study was also approved by the Ethics Committee of Peking University First Hospital.
Crosslinking Procedure
Anesthesia was induced via an auricular vein injection of 5% pentobarbital sodium (Sinopharm Chemical Reagent Co., Ltd, Shanghai, China) with 0.5 mL/kg concentration. All right eyes were treated, while the contralateral left eyes served as controls and were kept closed during the CXL procedures. The central 8 mm of the epithelium of the cornea was removed in each right eye using a stab knife (72-1501; Sharpoint, Reading, PA). In the Tgases CXL group, a Tgase solution was dripped on the cornea every 2 minutes for a total of 16 times. The Tgase solution consisted of 1 U/mL Tgase (Rubio, Germany) in phosphate-buffer solution (PBS) and was stored at 4°C until used. In the UVA/RF CXL group, the riboflavin (RF) solution consisted of 0.1% RF (Sigma-Aldrich, Darmstadt, Germany) in 20% dextran (Adamas, Shanghai, China) and was applied to the cornea as a droplet every 5 minutes for 30 minutes. Subsequently, we performed a clinical protocol, including UVA exposure (365 ± 5 nm, 3 mW/cm2, 8-mm diameter light spot) performed using a UVA lamp (Lamplic Technology, Shenzhen, China).10 Immediately after treatment, Ofloxacin gel ointment (Sinqi Pharmaceutical, Shenyang, China) was applied. Application of the ointment was repeated twice daily for 2 weeks.
Methods for Biomechanical Evaluation
All rabbits were euthanized via an intravenous injection of an overdose of pentobarbital sodium (1 mL/kg), and corneal samples were prepared for biomechanical testing (12 subjects of every group) and histologic or cytology evaluation (5 subjects of every group).
Twelve eyeballs in each group were prepared for biomechanical testing, and a central cornea strip was harvested in the superior-inferior direction within 4-hours post mortem from each eye. The strips were 16-mm long and 4-mm wide and included 2.0 to 3.0 mm of sclera tissue at each end. The ends of the strips were clamped in skid proof holders (with 320-grit sandpaper) with a gauge length of 10 mm. The thickness of each specimen was measured along the length using an ultrasound pachymeter (PachPen; Accitome, Malvern, PA) for three times and the average value was used in analysis. The strips were tested on an Instron 5848 materials testing machine (Norwood, MA) equipped with a 5-Newton capacity load cell at room temperature (Fig. 1).
Figure 1.
Biomaterial testing machine with a cornea strip between the clamps.
The specimens were first subjected to three preconditioning loading cycles to stabilize their behavior. The loading cycles were applied using a strain rate of 0.2 per minute and involved cycles of stretching to a length of 11.0 mm and relaxation down to a length of 10.1 mm. Thirty seconds were allowed between each two cycles to allow relaxation and reduce the effect of strain history on subsequent loading steps. Then, after preconditioning cycles, the specimens were loaded to failure at the same strain rate of 0.2 per minute. The output, including the specimen elongation in millimeters and the axial tension load in Newtons, were electronically recorded. Stress was then determined as the load divided by the specimen's initial cross-sectional area, and strain as the elongation divided by the initial specimen length.
As the stress-strain (σ-ε) behavior was found to have a nonlinear exponential form, the test results were fitted to the following equation:
 |
Where the values of constants A, R0, and σ0 were determined using the least squares method. The stress in Equation 1 was differentiated with respect to strain to determine the value of the tangent modulus:
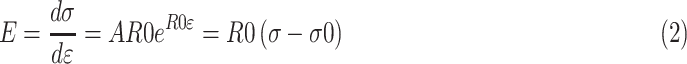 |
Origin 9 (OriginLab Corp., Northampton, MA) software was used to process the data and obtain the strain-stress and modulus-stress relationships.
Methods Used to Evaluate Safety
Endothelium Evaluation
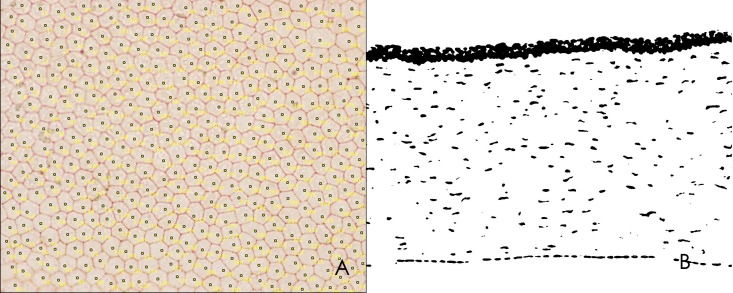
Another five corneal buttons were excised for endothelial staining within 24 hours after euthanasia. Corneal materials were obtained with a trephine (8-mm diameter) and divided equally into two parts. One half of the sample was stained with alizarin red (Sigma-Aldrich, St. Louis, MO) and trypan blue (Sigma-Aldrich). First, 2.5% trypan blue solution was dropped on the endothelium, which was incubated for 30 seconds and then washed with PBS. Then, 1% alizarin red was dropped onto the endothelium, which was incubated for 2 minutes and then washed with PBS. After observation, these tissues were used for histopathologic staining. The density of endothelial cells was analyzed using ImageJ software (National Institutes of Health, Bethesda, MD; available at http://rsb.info.nih.gov/ij/download.html). A sample field (×400 magnification with a size of 433 × 330 μm) was selected in each corneal button, and all endothelial cells in this field were counted (Fig. 2A).
Figure 2.
Image used to analyses keratocyte density in ImageJ. (A) Endothelium evaluation. (B) Keratocyte evaluation.
Keratocyte Evaluation
The remaining halves of the five corneal buttons were used to evaluate keratocyte apoptosis, which was detected using TUNEL assays. A TUNEL assay kit (Roche) was used in this study, and cryosections of cornea were subjected to analysis according to the manufacturer's instructions. The tissues were then stained with 4′,6-diamidino-2-phenylindole (DAPI; ZSbio, Beijing, China). The density of keratocytes was analyzed using ImageJ software. A sample field (×200 magnification with a size of 877 × 660 μm) was imaged in each corneal sample, and the image was converted to white and black by ImageJ software (Fig. 2B). Keratocytes are indicated by black dots of appropriate size and were automatically counted by ImageJ.
Statistical Analyses
Statistical analyses were performed with SPSS 14.0 software (SPSS Inc., Chicago, IL). Descriptive statistical results, including tangent moduli and cell densities, are described as the mean ± standard deviation (SD). The CXL-treated eye to its contralateral untreated eye ratio of tangent moduli were calculated. The tangent moduli and ratio's difference between two CXL groups was analyzed with independent t-test. The difference in Young's moduli between CXL eye and its contralateral negative controls was analyzed with paired t-tests. A P value below 0.05 was considered statistically significant.
Results
The Results of Efficacy Evaluations Analyses Based on Tensile Tests
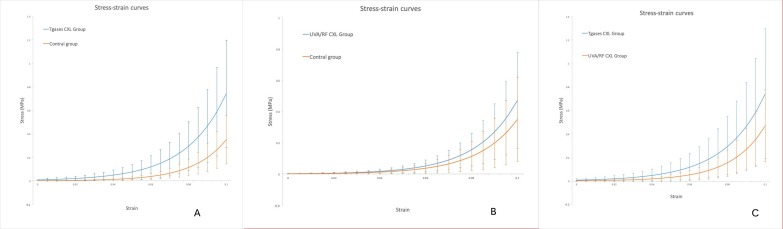
Strain-stress curves showed that the mechanical behaviors of two CXL corneal strips were both altered (Fig. 3). The slopes were much steeper in the Tgases CXL-treated curve than in those in the control groups, indicating an increase in corneal stiffness. While the slopes were slightly steeper than in those in the control groups in the UVA/RF CXL-treated curve.
Figure 3.
The stress-strain curves of the two CXL and their control groups.
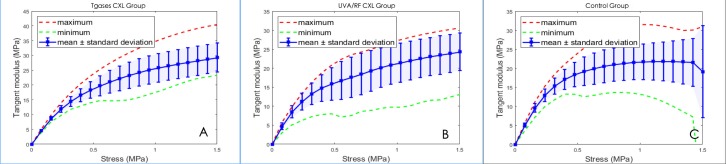
Tangent modulus was also different during tensile tests (Fig. 4). Young's modulus and its SD were calculated at stresses of 0.5, 1.0, and 1.5 MPa. A paired t-test was used to compare Young's modulus between Tgases CXL eyes and their corresponding control eyes. The results showed that there was a significant difference in tissues under heavy stress (1.0 and 1.5 MPa) (Table 1). Young's modulus ratio (CXL-treated/untreated eyes) under 0.5, 1.0, and 1.5 MPa were also calculated. An independent t-test was used to compare tangent modulus and modulus ratio between the two CXL groups, and the results showed that Young's modulus ratio was higher under heavy stress (1.5 MPa) in the Tgases CXL eyes (Fig. 4, Table 2, 3).
Figure 4.
Modulus-stress curves in three groups. (A) Tgases CXL. (B) UVA/RF CXL. (C) Control group.
Table 1.
Tangent Moduli in Tissues Under Different Stresses (Mpa) in Tgases CXL and Corresponding Control Eyes
| 0.5 MPa |
1.0 MPa |
1.5 MPa |
|
| Tgases CXL (n = 12) | 18.27 ± 2.78 | 26.59 ± 4.54 | 29.75 ± 5.01 |
| Control group (n = 12) | 18.35 ± 3.19 | 21.47 ± 4.72 | 20.47 ± 6.63 |
| t | −0.066 | 2.020 | 3.634 |
| P | 0.948 | 0.04 | 0.00 |
Table 2.
Tangent Moduli in Tissues Under Different Stresses (Mpa) in the Two CXL Group
| 0.5 MPa |
1.0 MPa |
1.5 MPa |
|
| Tgases CXL (n = 12) | 18.27 ± 2.78 | 26.59 ± 4.54 | 29.75 ± 5.01 |
| UVA/RF CXL (n = 12) | 15.86 ± 4.30 | 21.46 ± 5.14 | 24.33 ± 4.92 |
| t | 1.783 | 2.591 | 3.634 |
| P | 0.08 | 0.02 | 0.01 |
Table 3.
The Comparison of Treated/Untreated Ratio of Tangent Moduli in Tissues Under Different Stresses in the Two CXL Groups
| 0.5 MPa |
1.0 MPa |
1.5 MPa |
|
| Ratio of Tgases CXL group (n = 12) | 1.03 ± 0.86 | 1.31 ± 0.41 | 1.73 ± 1.00 |
| Ratio of UVA/RF CXL group (n = 12) | 1.18 ± 0.53 | 1.02 ± 0.25 | 1.04 ± 0.22 |
| t | 0.869 | 1.991 | 2.178 |
| P | 0.394 | 0.06 | 0.04 |
The Results of Safety Evaluations
Changes in the Endothelium of the Corneas
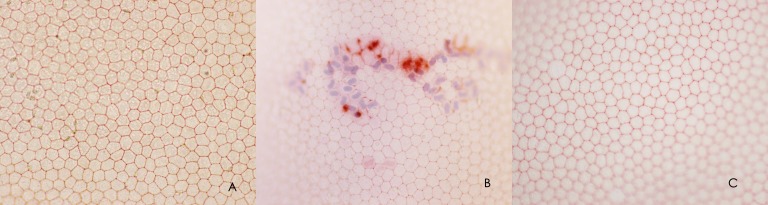
After alizarin red and trypan blue staining, normal hexagonal endothelial cells were present in the Tgases CXL groups. The borders were clear, and the density of cells was similar to the control groups (Table 4). However, in the UVA/RF CXL group, the hexagonal structure of the endothelium was obscure, and many apoptotic cells were present (Fig. 5).
Table 4.
The Density of Stroma Keratocytes and Endothelial Cells in Tgases CXL and Corresponding Control Eyes
| Stroma Keratocytes Density, Cells/mm2 |
Endothelial Cells Density, Cells/mm2 |
|
| Tgases CXL (n = 5) | 183.1 ± 14.3 | 2859.3 ± 303.2 |
| Control group (n = 5) | 194.8 ± 28.1 | 2651.8 ± 237.5 |
| t | −0.834 | 1.204 |
| P | 0.429 | 0.263 |
Figure 5.
Alizarin and trypan blue staining of the endothelium after different treatments. (A) Tgases CXL group. (B) UVA/RF CXL group. (C) Control group.
Changes in Stromal Keratocytes in the Corneas
Detailed examinations of micrographs of corneal sections revealed that there was no marked difference in cellular apoptosis between the Tgases CXL group and the control group. The density of keratocytes in the corneas treated with Tgases was similar to that observed in the control groups (Table 3), and both were different from that observe in the UVA/RF CXL group. As illustrated in Figure 6, in the UVA/RF CXL group, nearly all of the keratocytes before the CXL border had died, and the density of cells was significantly lower than that observed before the CXL border in the Tgases CXL and control groups.
Figure 6.
Illustration of cell apoptosis in cells labeled via TUNEL assays and cell density in tissues labelled with DAPI. (A) Tgases CXL group. (B) UVA/RF CXL group. (C) Control group).
Discussion
Corneal ectasia could not be effectively treated until Spoerl et al.11 proposed a new technique, UVA-RF CXL, that increased the biomechanical stiffness of the cornea. UVA-RF CXL was the first and is currently the only treatment capable of effectively arresting the clinical progress of corneal ectasia. In the standard UVA/RF CXL method, riboflavin (in dextran solution) is used as a photosensitizer, and UVA light is applied at 366 nm for photoactivation radiation. Although UVA/RF CXL produces positive results, this treatment has limitations. It has been recommended that CXL should not be performed within 400 μm of the central cornea because it can cause damage to endothelial cells.12 Corneal sensitivity is substantially decreased after the procedure.13 In addition, obvious cytotoxicity was observed in the anterior half of the cornea. Other methods of performing photochemical CXL have been reported in the literature. These include WST-D/NIR CXL14 and rose Bengal-green light CXL,15 which induce physical crosslinking and therefore have the same advantages and disadvantages as UVA/RF CXL. Another type of CXL, biochemical crosslinking, also known as enzymatic crosslinking, may be a potential method for inducing crosslinking.
Transglutaminase was first isolated from Streptoverticillium sp in 1989 and was subsequently found to be an enzyme that catalyzes the formation of isopeptide bonds between the γ-carboxamides of glutamine residues (donor) and the first-order e-amine groups of different compounds.16 This crosslinking property of Tgases is widely used in various processes in the food and manufacturing industries. Early on, commercial Tgases could only be obtained from animal tissues (e.g., guinea pig livers), and the low yields and high price prevented Tgases from being more widely applied. Recently, Tgases have been obtained from microorganisms at increased yields, and this has allowed many novel potential applications to be developed using Tgases.17 Tgases are crosslinking enzymes that mediate a biochemical reaction between glutamic acid and lysine by catalyzing the formation of isopeptide bonds, ε-(γ-glutamyl) lysine bonds.18 Although Tgases are enzymes that are widely distributed throughout the human body, they are scarce in the cornea because of its lack of blood and lymphatic vessels. The cornea consists almost exclusively of Type I collagen and is rich in glutamic acid and lysine. In theory, corneal collagen fibers could be crosslinked by Tgases, potentially resulting in more resistant mechanical properties.
In this study, we applied microbial Tgases in the ophthalmic sector and confirmed its effect on the biomechanical stability of cornea. As the cornea has been observed to behave in a viscoelastic manner,19 tensile tests performed by exposing corneal tissue to stress under controlled conditions are thought to be a noncontroversial way to analyze the biomechanical properties of the cornea. Strain-stress curves also showed that the slope was steeper in both Tgases CXL and UVA/RF CXL corneas, with the Tgases CXL group having a steeper slope than was found in UVA/RF-CXL tissues. The stress-strain curves were fitted by specific software after cornea tensile tests, and tangent modulus was calculated. In our study, tangent modulus was increased by approximately 50% in the Tgases CXL group. Because keratoconic corneas exhibit approximately 60% of the stiffness observed in healthy corneas,20 the 50% increase induced by Tgases CXL may be effective in this corneal disorder.
Because this study is the first to use Tgases CXL in corneas in vivo, we were initially concerned about the reactions of the animals. Although they were not recorded and evaluated, conjunctival congestion and chemosis were observed at lower rates in the Tgases CXL group than in the UVA/RF-treatment animals. In the cytotoxicity analysis, no cell apoptosis was found in the corneal stroma or endothelium, but UVA/RF clearly induced cell apoptosis in the anterior half of the corneal stroma and the endothelium in the UVA/RF-treatment groups. The densities of stromal keratocytes and endothelial cells were similar between the Tgases-treated groups and the control groups.
An obvious advantage of this application of Tgases is that it does not require a UVA- or near-infrared light-induced photochemical reaction to cause biochemical stiffening in the cornea. The cascade of events triggered by photosensitizers begins with the generation of reactive oxygen species (ROS), which not only induce crosslinking between collagen but also cause deeper damage in the eyeball.9,14 ROS cause oxidative stress and induce necrosis or apoptosis in ocular cells. According to Wollensak et al.,9 the dosage of a standard treatment of UVA-irradiance is 3 mW/m2, which is 6-fold higher than the irradiance dosage that induces a cytotoxic effect (0.5 mW/cm2 when combined with photosensitizer). It has also been shown that UVA can damage not only in the lens but also in the retina in animals.21,22 In our study, keratocyte loss was observed in the anterior half of the stroma in the UVA/RF-treatment group, but no obvious cell apoptosis was observed in the Tgases treatment group. Rabbit corneas are thinner than 400 μm; thus, their endothelial cells can be damaged by UVA, and cell apoptosis has been observed in Trypan blue–stained tissues.23 In contrast, endothelial cells showed a higher rate of Trypan blue staining in the Tgases groups, suggesting that Tgases CXL may have a better safety profile.
Our findings indicate that the biochemical properties induced by and the safety of Tgases are superior to those of UVA-RF. In some ways, Tgases are ideal crosslinking agents in the cornea, and they show promise for being developed in future CXL applications. A larger sample size and a longer follow-up period are required to accurately determine the efficacy and safety of Tgases-induced crosslinking.
In addition, the efficacy of and stiffness induced by the Tgases treatment were much better than those in the UVA/RF group in our study. These results may be different in humans. The corneas of rabbits are much thinner than those of humans, and this treatment may therefore cause more damage to the whole eyeball in rabbits than in humans. In the UVA/RF group, the inflammation observed in the rabbit cornea as a result of free radicals could cause edema of the collagenous fibers, and thereby decrease the biomechanical strength of the tissue. This may be the reason that corneal biomechanics were worse in the UVA/RF group compared with Tgases group.
In conclusion, Tgases seem to be a promising agent for CXL and deserve to be further explored as a treatment for keratectasia.
Acknowledgments
Supported by grants from Natural Science Foundation of Tibet Autonomous Region (NO XZ2017ZR-ZY032).
Disclosure: Y. Wu, None; W. Song, None; Y. Tang, None; A. Elsheikh, None; Y. Shao, None; X. Yan, None
References
- 1.Ziaei M, Barsam A, Shamie N, et al. Reshaping procedures for the surgical management of corneal ectasia. J Cataract Refract Surg. 2015;41:842–872. doi: 10.1016/j.jcrs.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 2.Raiskup F, Spoerl E. Corneal crosslinking with riboflavin and ultraviolet A. I. Principles. Ocul Surf. 2013;11:65–74. doi: 10.1016/j.jtos.2013.01.002. [DOI] [PubMed] [Google Scholar]
- 3.Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;137:620–627. doi: 10.1016/s0002-9394(02)02220-1. [DOI] [PubMed] [Google Scholar]
- 4.Hafezi F, Kanellopoulos J, Wiltfang R, Seiler T. Corneal collagen crosslinking with riboflavin and ultraviolet A to treat induced keratectasia after laser in situ keratomileusis. J Cataract Refract Surg. 2007;33:2035–2040. doi: 10.1016/j.jcrs.2007.07.028. [DOI] [PubMed] [Google Scholar]
- 5.Hatami-Marbini H, Jayaram SM. UVA/riboflavin collagen crosslinking stiffening effects on anterior and posterior corneal flaps. Exp Eye Res. 2018;176:53–58. doi: 10.1016/j.exer.2018.05.014. [DOI] [PubMed] [Google Scholar]
- 6.Webb JN, Su JP, Scarcelli G. Mechanical outcome of accelerated corneal crosslinking evaluated by Brillouin microscopy. J Cataract Refract Surg. 2017;43:1458–1463. doi: 10.1016/j.jcrs.2017.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kopsachilis N, Tsaousis KT, Tsinopoulos IT, Kruse FE, Welge-Luessen U. A novel mechanism of UV-A and riboflavin-mediated corneal cross-linking through induction of tissue transglutaminases. Cornea. 2013;32:1034–1039. doi: 10.1097/ICO.0b013e31828a760d. [DOI] [PubMed] [Google Scholar]
- 8.Haberman ID, Lang PZ, Broncano AF, Kim SW, Hafezi F, Randleman JB. Epithelial remodeling after corneal crosslinking using higher fluence and accelerated treatment time. J Cataract Refract Surg. 2018;44:306–312. doi: 10.1016/j.jcrs.2017.12.021. [DOI] [PubMed] [Google Scholar]
- 9.Wollensak G, Spoerl E, Reber F, Seiler T. Keratocyte cytotoxicity of riboflavin/UVA-treatment in vitro. Eye. 2004;18:718–722. doi: 10.1038/sj.eye.6700751. [DOI] [PubMed] [Google Scholar]
- 10.Wollensak G, Spoerl E, Seiler T. Stress-strain measurements of human and porcine corneas after riboflavin-ultraviolet-A-induced cross-linking. J Cataract Refract Surg. 2003;29:1780–1785. doi: 10.1016/s0886-3350(03)00407-3. [DOI] [PubMed] [Google Scholar]
- 11.Spoerl E, Huhle M, Kasper M, Seiler T. Increased rigidity of the cornea caused by intrastromal cross-linking [in German] Ophthalmologe. 1997;94:902–906. doi: 10.1007/s003470050219. [DOI] [PubMed] [Google Scholar]
- 12.Impact of crosslinking/riboflavin-UVA-photodynamic inactivation onviability, apoptosis and activation of human keratocytes in vitro. J Biomed Res. 2015;29:321–325. doi: 10.7555/JBR.29.20130173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wasilewski D, Mello GHR, Moreira H. Impact of collagen crosslinking on corneal sensitivity in keratoconus patients. Cornea. 2013;32:899–902. doi: 10.1097/ICO.0b013e31827978c8. [DOI] [PubMed] [Google Scholar]
- 14.Brekelmans J, Goz A, Dickman MM, et al. Long-term biomechanical and histologic results of WST-D/NIR corneal stiffening in rabbits, up to 8 months follow-up. Invest Ophthalmol Vis Sci. 2017;58:4089–4095. doi: 10.1167/iovs.17-22108. [DOI] [PubMed] [Google Scholar]
- 15.Bekesi N, Kochevar IE, Marcos S. Corneal biomechanical response following collagen cross-linking with Rose Bengal-green light and riboflavin-UVA. Invest Ophthalmol Vis Sci. 2016;57:992–1001. doi: 10.1167/iovs.15-18689. [DOI] [PubMed] [Google Scholar]
- 16.Kieliszek M, Misiewicz A. Microbial transglutaminase and its application in the food industry. A review. Folia Microbiol (Praha) 2014;59:241–250. doi: 10.1007/s12223-013-0287-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhu Y, Tramper J. Novel applications for microbial transglutaminase beyond food processing. Trends Biotechnol. 2008;26:559–565. doi: 10.1016/j.tibtech.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 18.Griffin M, Casadio R, Bergamini CM. Transglutaminases: nature's biological glues. Biochem J. 2002;368:377–396. doi: 10.1042/BJ20021234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lombardo M, Lombardo G, Carbone G, de Santo MP, Barberi R, Serrao S. Biomechanics of the anterior human corneal tissue investigated with atomic force microscopy. Invest Ophthalmol Vis Sci. 2012;53:1050–1057. doi: 10.1167/iovs.11-8720. [DOI] [PubMed] [Google Scholar]
- 20.Andreassen TT. Hjorth Simonsen A, Oxlund H. Biomechanical properties of keratoconus and normal corneas. Exp Eye Res. 1980;31:435–441. doi: 10.1016/s0014-4835(80)80027-3. [DOI] [PubMed] [Google Scholar]
- 21.Rogers CS, Chan LM, Sims YS, Byrd KD, Hinton DL, Twining SS. The effects of sub-solar levels of UV-A and UV-B on rabbit corneal and lens epithelial cells. Exp Eye Res. 2004;78:1007–1014. doi: 10.1016/j.exer.2003.12.011. [DOI] [PubMed] [Google Scholar]
- 22.Dong X, Löfgren S, Ayala M, Söderberg PG. Maximum tolerable dose for avoidance of cataract induced by ultraviolet radiation-B for 18 to 60 week old rats. Exp Eye Res. 2005;80:561–566. doi: 10.1016/j.exer.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 23.Spoerl E. Corneal cross-linking and safety issues. Open Ophthalmol J. 2011;5:14–16. doi: 10.2174/1874364101105010014. [DOI] [PMC free article] [PubMed] [Google Scholar]