Abstract
Subcutaneous adipose tissue (scAT) and peripheral blood mononuclear cells (PBMCs) play a significant role in obesity-associated systemic low-grade inflammation. High-fat diet (HFD) is known to induce inflammatory changes in both scAT and PBMCs. However, the time course of the effect of HFD on these systems is still unknown. The aim of the current study was to determine the time course of the effect of high fat diet (HFD) on PBMCs and scAT. New Zealand white rabbits were fed HFD for 5 or 10 weeks (i.e., HFD-5 and HFD-10), or regular chow (i.e., CNT-5 and CNT-10). Thereafter, metabolic and inflammatory parameters of PBMCs and scAT were quantitated. HFD induced hyperfattyacidemia in HFD-5 and HFD-10 groups, with the development of insulin resistance (IR) in HFD-10, while no changes were observed in scAT lipid metabolism and inflammatory status. HFD activated the inflammatory pathways in PBMCs of HFD-5 group, and induced modified autophagy in that of HFD-10. The rate of fat oxidation in PBMCs was directly associated with the expression of inflammatory markers; and tended to inversely associate with autophagosome formation markers in PBMCs. HFD affected systemic substrate metabolism, and the metabolic, inflammatory, and autophagy pathways in PBMCs in the absence of metabolic and inflammatory changes in scAT. Dietary approaches or interventions to avert HFD-induced changes in PBMCs could be essential in prevention of metabolic and inflammatory complications of obesity, and promote healthier living.
Keywords: Obesogenic diet, weight gain, low-grade inflammation, autophagy, Peripheral blood mononuclear cells, subcutaneous adipose tissue
INTRODUCTION
Obesity results in systemic and chronic low-grade inflammation with an increased number of circulating immune cells and inflammatory cytokines (1–3). This inflammation is associated with worsening metabolic health and the development of obesity-associated chronic diseases. Thus, understanding the mechanisms whereby systemic low-grade inflammation develops and determining approaches to prevent its progression should help promote or prolong healthier living. Currently, subcutaneous and visceral adipose tissue (scAT and VAT, respectively) inflammation is suggested as the main cause for the development of systemic low-grade inflammation (4–6). However, Tan et al., (7) demonstrated that 28-day overfeeding led to the development of systemic insulin resistance (IR) and increased the concentration of circulating C-reactive protein and monocyte chemoattractant protein-1 (MCP-1). Interestingly, these effects were observed in the absence of changes in scAT adipocyte size or the proportion of macrophages and T cell populations. Moreover, overfeeding-associated expansion of VAT directly correlated with inability of scAT to accumulate lipids (8). These results suggest that, in the early stages of overfeeding, the systemic inflammation may associate with activation of inflammatory mechanisms in cells other than adipocytes.
Circulating immune cells are also known to be involved in obesity-associated systemic low-grade inflammation. The effect of nutrition on peripheral blood mononuclear cells (PBMCs) has been a subject of increased interest. Food intake has been shown to transiently induce oxidative stress and pro-inflammatory changes in the PBMCs of healthy individuals (9). The plasma concentration of free fatty acids (FFAs) is known to significantly increase the expression of interleukin (IL)-6 and tumor necrosis factor alpha (TNF-α) mRNA in PBMCs (1). These data show that PBMCs have a dynamic inflammatory response to nutrient intake. However, not much is known about the long-term effect of diet. This information is critically important, as currently most of humans spend a considerable amount of time in a non-fasting, postprandial state with hyperlipidemia (10,11). This state may promote chronic activation of inflammatory pathways in PBMCs which may precede and/or drive the inflammatory changes in scAT. Moreover, in vitro studies have demonstrated that the common dietary saturated fatty acid palmitate can activate autophagy pathways (12) in PBMCs, which also play a significant role in monocyte-macrophage differentiation (13) and systemic low-grade inflammation. However, no information is available if high fat diet (HFD) affects autophagy pathways in circulating immune cells in vivo. Therefore, we aimed to test our hypothesis that increased dietary fat intake activates inflammatory and autophagy pathways in vivo in PBMCs prior to metabolic and inflammatory changes in scAT. We also aimed to determine the underlying mechanisms and the time-course effect of HFD (i.e., 5- and 10-week dietary interventions) on activation of these pathways.
METHODS
Animals and dietary interventions:
Male New Zealand white rabbits (Crossroads Rabbitry, Heflin, AL) were used for this study. We chose this model because, firstly, previous studies have shown that the lipid metabolism in this animal model of diet-induced obesity resembles that of humans with obesity (14). Secondly, the size of this animal model allows us to conduct stable isotope tracer infusion studies, and to obtain the required samples from one animal to achieve the objectives of our studies (14,15). All animals were acclimatized for 1-2 weeks before being randomly assigned to study groups. Twelve rabbits were assigned into 5- and 10-week dietary intervention with HFD (i.e., HFD-5 and HFD-10 groups, respectively). Upon completion of the intervention, all animals underwent a metabolic in vivo study as described below. Twelve control animals, age-matching for HFD-5 and HFD-10 groups, and denoted as CNT-5 and CNT-10 groups, underwent the same metabolic in vivo study. Thus, the 4 groups of animals included HFD-5 (n=6), HFD-10 (n=6), CNT-5 (n=6), and CNT-10 (n=6). Animals in the CNT groups were fed Laboratory Rabbit High Fiber diet (Cat# 5326, Labdiet®, St. Louis, MO) ad libitum. Animals in the HFD groups were fed the same diet plus additional 10% corn oil and 8% lard, as previously described (15). The compositions of diets are presented in supplemental files. All animals were individually housed with light/dark cycle, and social interaction. The welfare-related assessments were performed throughout the study duration by the veterinary personnel of the Animal Research Center, The University of Texas Medical Branch (UTMB) at Galveston. The study protocol complied with the Public Health Service Policy on Humane Care and Use of Laboratory Animals, incorporated in the Institute for Laboratory Animal Research Guide for Care and Use of Laboratory Animals, and was approved by the Animal Care and Use Committee of UTMB.
The metabolic in-vivo study:
Following the dietary intervention and after an overnight fast (~12 hr), all animals were subjected to a metabolic study. In the morning of the study, animals were anesthetized with ketamine and xylazine, and fur shaved on the chest, neck and one ear. Jugular and marginal ear veins and the carotid artery were cannulated along with intubation of the trachea or placement of a laryngeal airway mask (LMA) and ventilated (Rate 12 bpm, Volume 100 mL pressure not to exceed 20 mmHg). Venous blood (~20 ml) in EDTA vacutainer tubes, and abdominal scAT and skeletal muscle samples from vastus lateralis were collected. Thereafter, a 3-hr primed continuous infusion of U-[13C16]-palmitate (99% enriched, Cambridge Isotope Laboratories, Inc., Tewksbury, MA) in 5% albumin (priming dose [PD]: 1.0 μmol•kg-1, infusion rate [IR]: 0.1 μmol•kg-1•min-1) was started (15,16). About 3 ml of arterial blood was obtained at 30, 60, 90, 120, 150, 160, 170, and 180 min of infusion to determine the rate of appearance of palmitate [Ra] as a measure of the lipolysis rate. Thereafter, the animals were sacrificed by I.V. injection of 5 ml of Euthasol solution under general anesthesia of ketamine and xylazine. Death was confirmed by open chest observation. At this time, a liver sample was obtained.
Ex vivo studies:
Ex vivo studies were designed to determine the rate of incorporation of U-[13C16]-palmitate into palmitoyl-carnitine under a basal condition as a marker of mitochondrial FAO (16). About 4 ml of baseline blood sample collected in EDTA-containing tubes was used. After obtaining samples to measure the background parameters (i.e., the enrichment of U-[13C16]-palmitate and the total concentrations of FFAs and acyl-carnitines), the remaining samples (3.6 ml) were mixed with 1 μL•mL−1 of 2 mM U-[13C16]-palmitate dissolved in 5% human albumin. The samples were then incubated in a 37°C water bath with periodic mixing, and (0.4 ml) aliquots were collected at 5, 10, 20, 40 and 60 min after the start of incubation. All aliquots were immediately frozen in liquid nitrogen to arrest all biochemical reactions.
Sample analyses:
Glucose and Insulin measurements:
Blood glucose levels were measured using an Ascensia glucometer (Bayer). Plasma insulin concentrations were measured using ELISA kits (Mercodia, Winston Salem, NC).
Plasma FFAs:
Plasma lipids were extracted using a heptane-propanol extraction buffer and free fatty acids (FFAs) were separated using thin-layer chromatography plates (TLC; Partisil LK5D, Silica Gel 150 Å, Schleicher & Schuell, Maidstone, England). After the samples were methyl-esterified, the tracer-to-tracee ratio of U-[13C16]-palmitate in plasma FFAs was measured using GC-MS (MSD system, Agilent, Santa Clara, CA) monitoring the mass-to-charge ratios of 270, 285 and 286 for methyl palmitate. Eight fatty acids (FAs) in plasma FFAs were measured using a GC system with flame ionization detection (GC-FID 6890, Agilent, Santa Clara, CA); the total FFA concentrations were measured by means of internal standards using a GC-FID system (15,16).
Analyses of blood lipids and acyl-CNTs from ex vivo experiments:
Blood FFAs were isolated and analyzed as described above. Blood acyl-carnitines were isolated using a 1 ml of an acetonitrile-methanol mixture (3:1) with 50 μL of 1M KH2PO4 (14,15). To measure the concentrations of specific and total acyl-carnitine species, D3-C16:0-carnitine was added as an internal standard (45 μM). Samples were then analyzed to determine the concentrations of myristoyl-, palmitoyl-, and total acyl-carnitines using a Liquid Chromatograph Mass Spectrometer (LC-MS 6130 Quadruple, Agilent, Santa Clara, CA), as previously described (15,16). The enrichment of U-[13C16]-palmitoyl-carnitine was measured to determine the tracer-to-tracee ratio of the label incorporated into palmitoyl-carnitine.
Muscle and liver lipid analyses:
The lipids were extracted from 30-50 mg of tissue powder overnight at 4°C in a 1:2 (v/v) methanol: chloroform solution containing 0.05 mg/ml butylated hydroxytoluene. The samples were subjected to a TLC plate to separate TGs and FFAs, as previously described (15–17). The TGs and FFAs profiles were determined using GC-FID, and the contents were calculated using internal standard approach, as previously described (15,18).
Western blot analyses:
The PBMCs were isolated from baseline samples using Ficoll gradient separation as previously described (18). scAT proteins were isolated as previously described (17). The protein extracts of PBMC cytosolic fraction, and of scAT, (15 μg) were subjected to 4-15% SDS-PAGE (Criterion TGX, Bio-Rad Laboratories, CA), and electrophoretically transferred to polyvinylidene difluoride membranes (lmmobilon®-P, Millipore, Bedford, MA). The following monoclonal primary antibodies were used: a) t-p38 (#9212S; 1:1000; 38 kDa), p-p38 (#4511; 1:1000; 38 kDa), t-ERK1/2 (#9102; 1:100; 42/44 kDa), p-ERK1/2 (#9910T; 1:100; 42/44 kDa), LC3 (#4445; 1:1000; 14/16 kDa), p62 (#4445; 1:500; 62 kDa), t-Perilipin (#9349; 1:1000; 57 kDa) from Cell Signaling, MA; b) IL-1β (sc32294; 1:200; 17 kDa), IL-10 (sc8438; 1:200; 20 kDa), TLR4 (sc-293072; 1:1000; 95 kDa) from Santa Cruz, CA; c) p-PerilipinSer522 (#4856; 1:5000; 57 kDa; ValaSciences, CA); and d) adiponectin (ab22554; 1:1000; 30 kDa, Abeam, MA). As a secondary antibody either goat anti-mouse IgG-HRP (#1030-05, 1:5000-10000) or anti-rabbit IgG-HRP (#4050-05; 1:5000-10000; Southern Biotech, Birmingham, AL) were used. The expression levels were evaluated by quantification of the relative density of each band normalized to the β-actin band density (#A1978; 1:50000; 42 kDa Sigma-Aldrich, MO) using NIH ImageJ software v1.46r (NIH, Bethesda, MD).
Calculations:
IR (HOMA-IR) and beta cell function (HOMA-β%) were estimated from fasting glucose and insulin values using the approximated equation of Matthews et al (19), and the Disposition Index, estimated as HOMA-β%/HOMA-IR (20).
The expression of IL-1β was divided by the expression of IL-10 (arbitrary units) to calculate the ratio of IL-1β to IL-10 as a marker of distribution of activated (M1-type) vs. classical (M2-type) immune cells (21).
The [Ra] of palmitate, a measure of in vivo lipolysis rate, was calculated by dividing the enrichment of U-[13C16]-palmitate in plasma FFAs by rate of infusion of U-[13C16]-palmitate (16). For ex vivo studies, to calculate the kinetic parameters the tracer-to-tracee ratios of labeled palmitic acid and palmitoyl-carnitine were converted into molar percent excess (MPE). The fractional synthesis rate (FSR) of palmitoyl-carnitine was calculated by dividing the slope of incorporation of the label into palmitoyl-carnitine by the area under the curve (AUC) of labeled palmitate (MPE) over a 1 -hr ex vivo experimental period, and is expressed in %•hr−1. The absolute synthesis rate (ASR) of palm-carnitine was calculated by multiplying the FSR by the concentration of palmitoyl-carnitine and expressed as nMol•L−1•m−1 (16).
Statistical analyses and data presentations:
Data are presented as means ± standard errors (SE). The sample size was determined by the feasibility of minimally detectable effect sizes. The differences in parameters between the groups were evaluated using a two-way ANOVA with the factors diet (i.e., CNT or HFD) and time (i.e., 5 or 10 weeks) with post hoc Tukey correction. The approximate normality of model residuals was verified using normal quantile plots. Pairwise correlations between palmitoyl-carnitine FSR select outcomes of interest were determined using linear regression. p<0.05 was considered statistically significant.
RESULTS
Systemic and tissue metabolism:
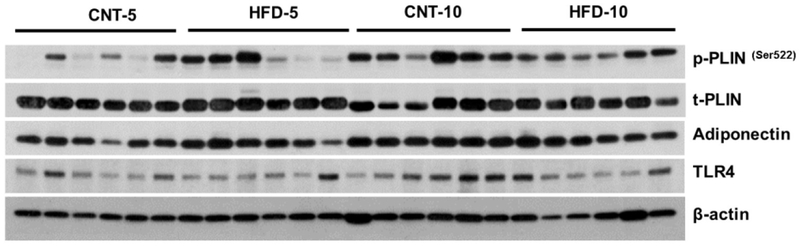
All results are presented in Table 1. Five- and 10-weeks of HFD resulted in gain of 0.65±0.16 and 1.19±0.83 kg of body weight from the baseline, respectively. However, no significant differences in body weight were observed between the CNT and HFD groups at either time point. HFD significantly increased the plasma concentration of FFAs at both time points; while the lipolysis rate [Ra] only trended to decline over time. Western blot analyses of scAT samples confirmed these data demonstrating that although the expression of tPLIN was significantly lower in HFD-10, the p/tPLIN was not altered (Fig. 1, Table 1). The expression of adiponectin and TLR4 as markers of inflammation in scAT did not differ between the HFD and CNT groups (Fig. 1, Table 1). Furthermore, liver and muscle lipid contents were not significantly altered by HFD.
Table 1.
Demographic and metabolic characteristics of rabbits.
|
p values |
||||||||
|---|---|---|---|---|---|---|---|---|
| Parameters | CNT-5 (n=6) | HFD-5 (n=6) | p value | CNT-10 (n=6) | HFD-10 (n=6) | p value | CNT-5 vs CNT-10 | HFD-5 vs HFD-10 |
| Age, days | 125±5 | 131±2 | 0.287 | 149±2 | 153±4 | 0.446 | <0.001 | <0.001 |
| Body weight, kg | 3.2±0.2 | 3.5±0.1 | 0.151 | 3.2±0.1 | 3.4±0.2 | 0.351 | 0.986 | 0.608 |
| Plasma FFA, μMol•ml−1 | 0.36±0.03 | 0.50±0.02 | 0.012 | 0.40±0.05 | 0.59±0.06 | 0.002 | 0.492 | 0.112 |
| Ra of palmitate, μMol•kg−1 •min−1 | 4.8±0.1 | 4.1±0.3 | 0.367 | 4.4±0.8 | 3.7±0.3 | 0.372 | 0.548 | 0.547 |
| Fasting glucose, mg•dL−1 | 110±7 | 136±4 | 0.086 | 118±14 | 165±12 | 0.005 | 0.533 | 0.058 |
| Serum insulin, μU•L−1 | 2.1±0.5 | 3.8±0.8 | 0.204 | 2.2±1.1 | 5.4±1.2 | 0.027 | 0.925 | 0.258 |
| HOMA-IR | 0.51±0.15 | 1.28±0.28 | 0.199 | 0.80±0.46 | 2.55±0.60 | 0.008 | 0.621 | 0.042 |
| HOMA-β | 14±4 | 19±4 | 0.401 | 11±4 | 22±4 | 0.074 | 0.534 | 0.693 |
| Disposition Index | 0.04±0.01 | 0.07±0.01 | 0.139 | 0.05±0.02 | 0.12±0.02 | 0.004 | 0.468 | 0.023 |
| Liver triglycerides, μMol•g−1 | 17±2 | 19±1 | 0.408 | 13±2 | 13±1 | 0.837 | 0.058 | 0.006 |
| Liver FFAs, μMol•g−1 | 1.15±0.55 | 0.35±0.04 | 0.086 | 0.74±0.19 | 1.31±0.23 | 0.212 | 0.361 | 0.043 |
| Muscle triglycerides, μMol•g−1 | 1.7±0.6 | 8.7±4.9 | 0.100 | 1.9±0.6 | 7.3±3.2 | 0.217 | 0.975 | 0.736 |
| Muscle FFAs, μMol•g−1 | 0.15±0.03 | 0.11±0.02 | 0.542 | 0.15±0.05 | 0.22±0.06 | 0.268 | 0.939 | 0.081 |
| Adipose tissue protein expression (arbitrary unit) | ||||||||
| t-PLIN | 3.09±0.13 | 3.49±0.09 | 0.215 | 3.20±0.34 | 2.80±0.24 | 0.225 | 0.736 | 0.040 |
| p/t-PLIN | 0.52±0.08 | 0.68±0.13 | 0.321 | 0.98±0.08 | 0.92±0.14 | 0.711 | 0.009 | 0.153 |
| Adiponectin | 0.91±0.10 | 1.02±0.13 | 0.610 | 1.12±0.05 | 1.29±0.27 | 0.469 | 0.353 | 0.255 |
| TLR4 expression | 0.27±0.04 | 0.29±0.04 | 0.650 | 0.20±0.02 | 0.27±0.03 | 0.177 | 0.125 | 0.515 |
The data are presented as Means±SE. The differences were determined using Two-way ANOVA with time and diet interaction with post hoc Tukey adjustment. The p < 0.05 was considered as statistically significant.
Figure 1.
Western Blot images of subcutaneous adipose tissue samples. CNT, control; HFD, high fat diet; p-PLIN, phosphorylated perilipin; t-PLIN, total perilipin; adiponectin, TLR4, toll-like receptor 4.
Although not reaching statistical significance, HFD-5 averaged 24% and 81% higher plasma glucose and insulin levels, respectively, than CNT-5. The HFD-10 group had significantly higher levels of glucose (p=0.005), insulin (p=0.027), HOMA-IR (p=0.008), and disposition index (p=0.004) when compared to CNT-10. HOMA-IR (p=0.042) and disposition index (p=0.023) were also higher in HFD-10 when compared to the HFD-5 group; no differences in HOMA-β due to the dietary intervention were observed at either time point (Table 1).
Inflammatory pathways in PBMCs:
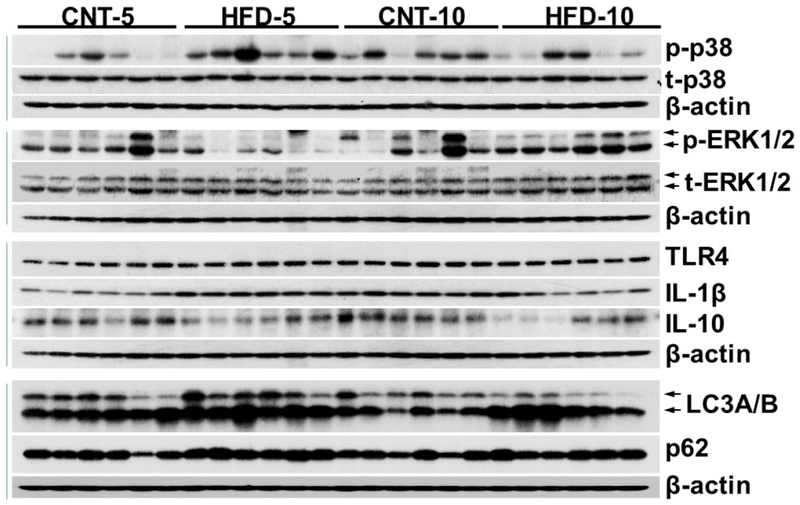
All data are presented in Fig. 2 and Table 2. HFD did not affect the expression of t-p38, while the expression of t-ERK1/2 was significantly increased in HFD-10 when compared to CNT-10 (p=0.002). Compared to age-matching groups (i.e., CNT-5 and CNT-10, respectively), the activation of p38 (i.e., p/t-p38) was significantly increased in HFD-5 group (p<0.001), but decreased in HFD-10 group (p=0.028). The expression of p-ERK1/2 was significantly decreased in the HFD-5 group (p=0.008).
Figure 2.
Western Blot images of PBMC samples. CNT, control; HFD, high fat diet; p-p38, phosphorylated p-38; t-p38, total p-38; p-ERK1/2, phosphorylated ERK1/2; t-ERK1/2, total ERK1/2; TLR4, toll-like receptor 4; IL-1β, interleukin 1β; IL-10, interleukin 10; LC3A/B, microtubule-associated proteins 1A/1B light chain 3.
Table 2.
Quantification of Western Blot analyses of PBMCs.
|
p value |
||||||||
|---|---|---|---|---|---|---|---|---|
| Parameters (arbitrary units) | CNT-5 (n=6) | HFD-5 (n=6) | p value | CNT-10 (n=6) | HFD-10 (n=6) | p value | CNT-5 vs CNT-10 | HFD-5 vs HFD-10 |
| t-p38 | 0.56±0.03 | 0.63±0.04 | 0.143 | 0.56±0.03 | 0.60±0.03 | 0.411 | 0.985 | 0.514 |
| p/t-p38 | 0.82±0.23 | 2.33±0.28 | < 0.001 | 2.10±0.33 | 1.21±0.21 | 0.028 | 0.003 | 0.008 |
| t-ERK1/2 | 0.63±0.07 | 0.70±0.07 | 0.403 | 0.56±0.04 | 0.87±0.05 | 0.002 | 0.463 | 0.065 |
| p/t-ERK1/2 | 3.53±0.42 | 1.21±0.23 | 0.008 | 3.44±0.94 | 2.90±0.34 | 0.499 | 0.915 | 0.044 |
| TLR-4 | 0.43±0.02 | 0.49±0.03 | 0.143 | 0.42±0.04 | 0.44±0.02 | 0.601 | 0.866 | 0.258 |
| IL-1β | 0.35±0.04 | 0.55±0.01 | 0.007 | 0.40±0.07 | 0.33±0.06 | 0.298 | 0.401 | 0.004 |
| IL-10 | 1.10±0.14 | 0.74±0.11 | 0.074 | 1.10±0.11 | 0.57±0.16 | 0.011 | 0.987 | 0.374 |
| IL-1β/IL-10 | 0.33±0.05 | 0.83±0.13 | 0.060 | 0.38±0.07 | 0.95±0.32 | 0.011 | 0.868 | 0.636 |
| LC3A/B-I | 0.38±0.05 | 0.50±0.04 | 0.054 | 0.38±0.04 | 0.25±0.04 | 0.029 | 0.913 | < 0.001 |
| LC3A/B-II | 0.67±0.06 | 0.80±0.02 | 0.054 | 0.58±0.04 | 0.74±0.04 | 0.022 | 0.155 | 0.314 |
| LC3A/B-II/I | 1.87±0.30 | 1.68±0.16 | 0.572 | 1.55±0.08 | 3.21±0.33 | < 0.001 | 0.357 | < 0.001 |
| p62 | 0.60±0.06 | 0.78±0.03 | 0.006 | 0.62±0.04 | 0.69±0.03 | 0.265 | 0.726 | 0.124 |
The data are presented as Means±SE. The differences were determined using Two-way ANOVA with time and diet interaction with post hoc Tukey adjustment. The p < 0.05 was considered as statistically significant.
The expression of TLR4 did not differ between the groups at any time point, while the expression of IL-1β was significantly increased in HFD-5 (p=0.007). The expression of IL-10 was significantly decreased in the HFD-10 group when compared CNT-10 group (p=0.011). The IL-1β/ IL-10 was more than doubled in both HFD groups when compared to the corresponding CNT groups; however, the difference reached statistical significance at 10-week time point (p=0.011; Table 2)
Autophagy pathways in PBMCs:
All data are presented in Fig. 2 and Table 2. The expression of LC3A/B-I was significantly decreased (p=0.029), and the expression of LC3A/B-II was significantly increased (p=0.022), in the HFD-10 group when compared to the CNT-10. As a result, the ratio of LC3A/B-II/I was significantly increased in the HFD-10 group when compared to the CNT-10 group as well as the HFD-5 (p<0.001). The expression of p62 was increased in the HFD-5 group (p=0.006) but unchanged in the HFD-10 group (p=0.265) when compared to age-matching CNT groups.
Acylcarnitine metabolism in PBMCs:
All data are presented in Table 3. The ex vivo FSR of palmitoyl-carnitine was significantly higher in the HFD-5 group when compared to the CNT-5 group (p<0.001) due to significantly higher slope of incorporation of the tracer into palmitoyl-carnitine (i.e., the enrichment of U-[13C16]-palmitate-carnitine; p<0.001). While in HFD-10 group the FSR of palmitoyl-carnitine was significantly lower when compared to CNT-10 group (p=0.002). The ex vivo ASR of palmitoyl-carnitine tended to be higher in the HFD-5 group (p=0.060) when compared to the CNT-5 group, but significantly lower in the HFD-10 group compared to CNT-10 (p=0.048) and HFD-5 (p=0.020) groups.
Table 3.
Acylcarnitine metabolism in PBMCs.
|
p value |
||||||||
|---|---|---|---|---|---|---|---|---|
| Parameter | CNT-5 (n=6) | HFD-5 (n=6) | p value | CNT-10 (n=6) | HFD-10 (n=6) | p value | CNT-5 vs CNT-10 | HFD-5 vs HFD-10 |
| Blood U-[13C16]-palmitate enrichment (MPE) | 8±2 | 14±2 | 0.064 | 12±1 | 15±2 | 0.335 | 0.216 | 0.744 |
| Slope U-[13C16]-palmitoyl-carnitine | 0.016±0.001 | 0.050±0.001 | < 0.001 | 0.038±0.001 | 0.032±0.001 | 0.505 | 0.021 | 0.055 |
| FSR palmitoyl-carnitine (MPE•m−1) | 0.20±0.02 | 0.35±0.01 | < 0.001 | 0.32±0.01 | 0.22±0.01 | 0.002 | < 0.001 | < 0.001 |
| ASR palmitoyl-carnitine (nMol•L−1•m−1) | 0.047±0.003 | 0.059±0.004 | 0.060 | 0.056±0.006 | 0.043±0.002 | 0.048 | 0.138 | 0.020 |
| Myristoyl-carnitine (nMol•L−1) | 2.21±0.29 | 1.50±0.27 | 0.054 | 1.85±0.16 | 1.57±0.26 | 0.453 | 0.309 | 0.845 |
| Palmitoyl-carnitine (μMol•L−1) | 0.03±0.01 | 0.02±0.01 | 0.006 | 0.02±0.01 | 0.02±0.01 | 0.334 | 0.010 | 0.232 |
| Total Acyl-carnitine (μMol•L−1) | 0.12±0.01 | 0.11±0.01 | 0.264 | 0.09±0.01 | 0.12±0.01 | 0.086 | 0.050 | 0.373 |
The data are presented as Means±SE. The differences were determined using Two-way ANOVA with time and diet interaction with post hoc Tukey adjustment. The p < 0.05 was considered as statistically significant.
Linear regression analyses:
The FSR of palmitoyl-carnitine was directly, although moderately, associated with the expression of p/t-p38 (r=0.468, p=0.024), and IL-1β (r=0.498, p=0.015).
DISCUSSION
Systemic low-grade inflammation is a main determinant of metabolic complications leading to the development of obesity-related chronic diseases (22–24). Thus, understanding the pathogenesis of this inflammation is critical in developing interventions to promote healthy living. Adipose tissue inflammation and the activation of circulating inflammatory cells are components of systemic low-grade inflammation (24). However, the sequence of inflammatory events in the early stages of weight gain are still not well understood. Specifically, it is still unknown whether activation of inflammatory pathways in scAT precedes that of circulating immune cells (i.e., PBMCs). Our results demonstrate that HFD induces an impairment in systemic substrate metabolism and activation of inflammatory and autophagy pathways in PBMCs before metabolic and inflammatory changes occur in scAT.
HFD-induced impaired systemic metabolism precedes changes in adipose tissue:
HFD is well-known to cause the development of metabolic abnormalities. Some, but not all, overfeeding studies have demonstrated that comparable periods of HFD resulted in significant increase in body weight, and impaired tissue lipid metabolism (25–28). In the current study, HFD did not significantly affect body weight, adipose tissue metabolism (i.e., [Ra] and activation of PLIN), inflammatory status (i.e., TLR4), or the deposition of lipids in liver and muscle (Table 1). This can potentially be explained by the differences in diet and fat composition (26). The total and saturated fat in our study diet were ~21% and 10%, respectively, amounts comparable to the average American daily intake (29), but less than the 35% total and 12% saturated fats in a typical experimental Western-style diet (26). Nevertheless, the HFD caused severe hyperfattyacidemia in both 5- and 10-week studies (Table 1). These data suggest that the scAT was in a state of adaptive expansion to accumulate the excess calorie intake, and that, under our study conditions, a 5-week or 10-week HFD feeding did not result in impaired tissue lipid metabolism with ectopic fat depositions. HFD led to worsening glycemic control over time with the development of IR in HFD-10 group (Table 1), similarly to other studies (7,25,30). In summary, a 5-week HFD impaired systemic lipid metabolism with increased levels of circulating FFAs, and a 10-week HFD impaired glycemic control; however, these changes preceded the changes in muscle, liver and adipose tissue lipid metabolism as well as scAT inflammatory status.
HFD-induced activation of inflammatory pathways in PBMCs:
The main aim of this study was to determine the effect of HFD on activation of inflammatory pathways in PBMCs. Previous studies showed activation of MAPK (i.e., p38 and ERK1/2) pathways in lymphocytes and monocytes of patients with T2D (31–33). In experimental models, specific inhibition of MAPK pathways in macrophages suppressed pro-inflammatory responses with delayed onset of diabetes (34).
Therefore, the MAPK pathway is important in obesity-related inflammation. Indeed, after 5 weeks of HFD, a significant activation of p38 (i.e., p/t-p38) and increased expression of IL-1β were observed (Fig. 2, Table 2), although the ERK1/2 was either decreased in HFD-5 or unaffected in HFD-10. Both p38 and ERK1/2 are involved in the MAPK pathway, and p38 is strongly associated with inflammation but ERK1/2 with cell differentiation pathways (35). To determine the mechanisms of MAPK pathway activation, we measured the expression of TLR4 and IL-1β. TLR4, a member of the pattern-recognition receptor family TLR, is a well-known receptor involved in saturated FA-induced activation of inflammatory pathways including MAPK pathways (36,37). The expression of TLR4 did not differ between groups at any time (p>0.05, Fig. 2, Table 2). IL-1β is also known to activate the MAPK pathways (38). Indeed, the expression of I L-1β followed the pattern of changes in expression of p/t-p38 and was significantly increased only in HFD-5 (Fig. 2, Table 2). These data may suggest that HFD caused inflammatory changes in the PBMCs of rabbits via IL-1β but not TLR4-involved mechanisms. However, these changes occurred after short-term (i.e., 5-week) HFD only, because the expression of both p-p38 and IL-1β returned to CNT-5 levels in the HFD-10 group (Fig. 2, Table 2). This may suggest that either adaptive mechanisms combating the inflammatory changes are being activated or other changes, yet unknown, are occurring that result in the “quiescence” of inflammatory response at 10-weeks of HFD. The expression of IL-10, a potent anti-inflammatory cytokine (39), was 32% lower in the HFD-5 group than in CNT-5 (p=0.074), and 48% lower in the HFD-10 group than in CNT-10 group (p=0.011, Fig. 2, Table 2). These data may suggest that IL-10 may not be involved in suppression of inflammation in PBMCs in the HFD-10 group. However, IL-10 is the classical M2 or immune regulatory cytokine while IL-1β is the classical M1 or inflammatory type (21,39). Thus, HFD may lead to the loss of M2 type immune cells. As a result, although IL-1β did not differ between the HFD-10 and CNT-10 groups, the higher IL-1β/IL-l0 ratio suggest that HFD-10 animals were immune compromised. In summary, short-term HFD (i.e., HFD-5 group) induced activation of inflammatory pathways in PBMCs in vivo in rabbits, and ultimately immune deficiency in the HFD-10 group.
HFD-modified autophagy pathways in PBMCs:
Autophagy plays a housekeeping role in clearing misfolded or aggregated proteins and damaged organelles, and has also been shown to suppress excessive inflammation (40). Activation of microtubule-associated proteins 1A/1B light chain 3 (LC3), a central protein in the autophagy pathway (41), is the main step in monocyte-macrophage pro-inflammatory differentiation (13). Upon autophagic signal, the LC3A/B-I isoform undergoes lipidation to form the LC3A/B-II isoform, which shows faster electrophoretic mobility in SDS-PAGE gels, and thus shows up at 14-16 kDa, in comparison to 16-18 kDa for LC3A/B-I. The increased ratio of LC3-II to LC3-I is a marker for autophagosomes formation. LC3A/B-II/I was significantly increased in PBMCs of the HFD-10 group (Fig. 2, Table 2) suggesting the formation of autophagosomes. Thus, these data may suggest that autophagosomes were formed as a response to activation of inflammatory pathways. However, the autophagy process is comprised of autophagosome formation, fusion of autophagosomes with lysosomes into autophagolysosomes, and the maturation of this complex for degradation or sequestration. p62 is an autophagy substrate that delivers ubiquitinated cargoes for autophagic degradation and is used as a reporter of autophagy activity. Active degradation of autophagolysosomes reduces the expression of p62 expression, while unchanged or increased p62 expression, along with increased LC3A/B-II/I, suggest modification of the autophagy pathway (41). Here, p62 expression was increased in the HFD-5 group but unchanged in the HFD-10 group (Fig. 2, Table 2). These data demonstrate that HFD induced a modified autophagy in PBMCs in vivo. Increased expression of both LC3A/B-II and p62 is critical in GM-CSF-induced monocyte-macrophage differentiation, suggesting that this modified autophagy pathway is crucial in this process (13). In obese mice, impaired macrophage autophagy enhanced pro-inflammatory monocyte-macrophage polarization (42). RostamiRad et al. also showed that treating PBMCs with palmitate significantly increases LC3A/B and p62 expressions (12). In summary, our data suggest that a 10-week HFD induced modified autophagy in PBMCs in vivo in rabbits.
Acylcarnitine metabolism in PBMCs:
The activation of inflammatory pathways can be caused by intermediate lipid metabolites (i.e., acylcarnitines), which we previously reported were produced in PBMCs utilizing FFAs (16). We also demonstrated that the ASR of palmitoyl-carnitine, estimated during hyperinsulinemic-euglycemic clamp, inversely correlates with the degree of systemic insulin sensitivity in healthy overweight women. Therefore, we measured the ex vivo FSR and ASR of palmitoyl-carnitine by blood cells (16). The ex vivo FSR of palmitoyl-carnitine was significantly higher in the HFD-5 group than in the CNT-5 group, but significantly lower in the HFD-10 group than the CNT-10 group. The mechanism of adaptation to a HFD or high calorie intake is an increase in fat mass and/or in FAO until a new equilibrium is reached to accommodate the intake (43). However, in an IR or metabolically unhealthy state the FAO is lower and cannot accommodate the excess calorie intake (44). Thus, our data on palmitoyl-carnitine synthesis in PBMCs follows this pattern of adaptive FAO with HFD intake. Interestingly, the linear regression analyses demonstrated a direct, albeit moderate, correlation of acylcarnitine metabolism with p/t-p38 and IL-1β expressions. Therefore, p38 activation and increased IL-1β expression may result from HFD-induced enhanced FAO as estimated by the ex vivo FSR of palmitoyl-carnitine. Vice versa, the lower FSR in the HFD-10 group may explain the “quiescence” of inflammatory pathways or the switch inducing the modified autophagy.
Study limitations:
1) For the HFD group, a chow diet was supplemented with 18% fat. Thus, the proportion of nutrients, not only fats, was different between the diet (supplemental file). In our future studies we may consider using dietary approaches that differ in composition of macronutrients but not of other nutrients. In general, rabbits’ food of choice contains low fats. However, rabbits fed HFD or high cholesterol diet develop impaired fat metabolism like that of humans with obesity (14) or typical characteristics of atherosclerosis (45), respectively. Thus, we think that the HFD used in the current study is acceptable to allow the current conclusions to be drawn. 2) We did not study VAT in this study. the currently available data suggest that scAT ability to accumulate lipids is instrumental in averting the expansion of VAT in early stages of weight gain, as well as the development of obesity-associated metabolic abnormalities (8,17,25,46,47). However, we plan to study VAT in future. 3) Our study lacks interventions to determine if averting the changes in FAO in PBMCs prevents the activation of inflammatory and autophagy pathways, which will be addressed in future studies.
Conclusion:
Our data confirm that HFD caused hyperfattyacidemia, peripheral IR, enhanced FAO, and activation of inflammatory and autophagy pathways in PBMCs, without changes in scAT metabolism and inflammation. These outcomes are similar to those observed in humans with obesity. The time course of these changes was not straightforward, with some occurring after 5 weeks and others after 10 weeks of HFD. The adaptive metabolic and anti-inflammatory response to the diet may be the main explanations for our findings. If a) excessive FAO causes a domino effect, activating inflammatory and autophagy pathways in PBMCs and subsequent systemic low-grade adipose tissue inflammation; and b) dietary antioxidants correlate with a better health outcome, our findings may explain why, within a group with comparable body mass index, some are metabolically healthier than others (48,49). Although the detailed mechanism is not yet characterized, we hypothesize that dietary hyperfattyacidemia causes enhanced FAO-induced activation of p38 in PBMCs, which may induce the activation of autophagy pathways. Alternatively, IR or an impaired glycemic control-associated decline in FAO (HFD-10) activated the autophagy pathways. Because a) impaired activation of autophagy pathways plays a crucial role in monocyte-macrophagy differentiation; and b) the Western Lifestyle is characterized by a continuous postprandial state, PMBCs undergoing cycles of activation of inflammatory/autophagy pathways may be the metabolic mechanism linking HFD to systemic low-grade inflammation. Thus, interventions to avert HFD-induced early activation of inflammatory and modified autophagy pathways in PBMCs could help to prevent metabolic and inflammatory complications of obesity, and promote healthier living.
Supplementary Material
ACKNOWLEDGMENT
This study was supported by the 1KL2TR001441 NIH Training grant, the Institute for Translational Sciences at the UTMB, and in part by a Clinical and Translational Science Award (#UL1 TR001439) from the National Center for Advancing Translational Sciences, National Institutes of Health, and the Shriners Grant #84090, Metabolism Unit, Shriners Hospitals for Children.
We thank Christopher Danesi, Kathleen Randolph, and Anahi D. Delgadillo for help with sample analyses. We thank Dr. Sarah Toombs-Smith for the critical editing of this work.
Footnotes
The authors declare no competing financial interests.
Reference:
- 1.Ghanim H, Aljada A, Hofmeyer D, Syed T, Mohanty P, Dandona P. Circulating mononuclear cells in the obese are in a proinflammatory state. Circulation 110(12): 1564–71,2004. [DOI] [PubMed] [Google Scholar]
- 2.Viardot A, Heilbronn LK, Samocha-Bonet D, Mackay F, Campbell LV, Samaras K. Obesity is associated with activated and insulin resistant immune cells. Diabetes Metab Res Rev. 28(5):447–54, 2012. [DOI] [PubMed] [Google Scholar]
- 3.Bañuls C, Rovira-Llopis S, Lopez-Domenech S, Diaz-Morales N, Blas-Garcia A, Veses S, Morillas C, Victor VM, Rocha M, Hernandez-Mijares A. Oxidative and endoplasmic reticulum stress is impaired in leukocytes from metabolically unhealthy vs healthy obese individuals. Int J Obes (Lond). 41 (10): 1556–1563, 2017. [DOI] [PubMed] [Google Scholar]
- 4.Wensveen FM, Valentić S, Šestan M, Turk Wensveen T, Polić B. The “Big Bang” in obese fat: Events initiating obesity-induced adipose tissue inflammation. Eur J Immunol 45(9):2446–56, 2015. [DOI] [PubMed] [Google Scholar]
- 5.Pahlavani M, Ramalho T, Koboziev I, LeMieux MJ, Jayarathne S, Ramalingam L, Filgueiras LR, Moustaid-Moussa N. Adipose tissue inflammation in insulin resistance: review of mechanisms mediating anti-inflammatory effects of omega-3 polyunsaturated fatty acids. J Investig Med. 65(7): 1021–1027, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Permana PA, Menge C, Reaven PD. Macrophage-secreted factors induce adipocyte inflammation and insulin resistance. Biochem Biophys Res Commun 10;341(2):507–14, 2006. [DOI] [PubMed] [Google Scholar]
- 7.Tam CS, Viardot A, Clément K, Tordjman J, Tonks K, Greenfield JR, Campbell LV, Samocha-Bonet D, Heilbronn LK. Short-term overfeeding may induce peripheral insulin resistance without altering subcutaneous adipose tissue macrophages in humans. Diabetes. 59(9):2164–70, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Alligier M, Gabert L, Meugnier E, Lambert-Porcheron S, Chanseaume E, Pilleul F, Debard C, Sauvinet V, Morio B, Vidal-Puig A, Vidal H, Laville M. Visceral fat accumulation during lipid overfeeding is related to subcutaneous adipose tissue characteristics in healthy men. J Clin Endocrinol Metab. 98(2):802–10, 2013. [DOI] [PubMed] [Google Scholar]
- 9.Ehlers K, Brand T, Bangert A, Hauner H, Laumen H. Postprandial activation of metabolic and inflammatory signaling pathways in human peripheral mononuclear cells. Br J Nutr. 111(12):2167–75, 2014. [DOI] [PubMed] [Google Scholar]
- 10.Gill JM, Hardman AE. Exercise and postprandial lipid metabolism: an update on potential mechanisms and interactions with high-carbohydrate diets (review) J Nutr Biochem. 14:122–132, 2003. [DOI] [PubMed] [Google Scholar]
- 11.Henson J, Dunstan DW, Davies MJ, Yates T. Sedentary behavior as a new behavioral target in the prevention and treatment of type 2 diabetes. Diabetes Metab Res Rev. 32 Suppl 1:213–20, 2016. [DOI] [PubMed] [Google Scholar]
- 12.RostamiRad A, Ebrahimi SSS, Sadeghi A, Taghikhani M, Meshkani R. Palmitate-induced impairment of autophagy turnover leads to increased apoptosis and inflammation in peripheral blood mononuclear cells. Immunobiology. 2018. March;223(3):269–278. [DOI] [PubMed] [Google Scholar]
- 13.Zhang Y, Morgan MJ, Chen K, Choksi S, Liu ZG. Induction of autophagy is essential for monocyte-macrophage differentiation. Blood. 2012. March 22;119(12):2895–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang XJ, Chinkes DL, Aarsland A, Herndon DN, Wolfe RR. Lipid metabolism in diet-induced obese rabbits is similar to that of obese humans. J Nutr. 2008. March;138(3):515–8. [DOI] [PubMed] [Google Scholar]
- 15.Zhang XJ, Wang L, Tuvdendorj D, Wu Z, Rodriguez NA, Herndon DN, Wolfe RR. Acute hyperinsulinemia and reduced plasma free fatty acid levels decrease intramuscular triglyceride synthesis. Metabolism. 2013. January;62(1):44–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chondronikola M, Asghar R, Zhang X, Dillon EL, Durham WJ, Wu Z, Porter C, Camacho-Hughes M, Zhao Y, Brasier AR, Volpi E, Sheffield-Moore M, Abate N, Sidossis L, Tuvdendorj D. Palmitoyl-carnitine production by blood cells associates with the concentration of circulating acyl-carnitines in healthy overweight women. Clin Nutr. 2017;36(5):1310–1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pan W, Ciociola E, Saraf M, Tumurbaatar B, Tuvdendorj D, Prasad S, Chandalia M, Abate N. Metabolic consequences of ENPP1 overexpression in adipose tissue. Am J Physiol Endocrinol Metab. 2011;301(5):E901–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gonzales CM, Williams CB, Calderon VE, Huante MB, Moen ST, Popov VL, Baze WB, Peterson JW, Endsley JJ. Antibacterial role for natural killer cells in host defense to Bacillus anthracis. Infection and immunity. 2015; 80(1), 234–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9. [DOI] [PubMed] [Google Scholar]
- 20.Matsuda M. Measuring and estimating insulin resistance in clinical and research settings. Nutr Metab Cardiovasc Dis. 2010;20:79–86. [DOI] [PubMed] [Google Scholar]
- 21.Jamil B, Shahid F, Hasan Z, Nasir N, Razzaki T, Dawood G, Hussain R. Interferon gamma/IL10 ratio defines the disease severity in pulmonary and extra pulmonary tuberculosis. Tuberculosis (Edinb). 2007;87(4):279–87. [DOI] [PubMed] [Google Scholar]
- 22.Shoelson SE, Herrero L, Naaz A. Obesity, inflammation, and insulin resistance. Gastroenterology 2007;132(6):2169–80. [DOI] [PubMed] [Google Scholar]
- 23.Xu H, Barnes GT, Yang Q, Tan G, Yang D, Chou CJ, Sole J, Nichols A, Ross JS, Tartaglia LA, Chen H. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest 2003;112(12):1821–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hotamisligil GS. Inflammation, metaflammation and immunometabolic disorders. Nature 2017;542(7640):177–185. [DOI] [PubMed] [Google Scholar]
- 25.Cuthbertson DJ, Steele T, Wilding JP, Halford JC, Harrold JA, Hamer M4, Karpe F. What have human experimental overfeeding studies taught us about adipose tissue expansion and susceptibility to obesity and metabolic complications? Int J Obes (Lond). 2017;41(6):853–865. [DOI] [PubMed] [Google Scholar]
- 26.Lionetti L, Mollica MP, Sica R, Donizzetti I, Gifuni G, Pignalosa A, Cavaliere G, Putti R. Differential effects of high-fish oil and high-lard diets on cells and cytokines involved in the inflammatory process in rat insulin-sensitive tissues. Int J Mol Sci. 2014. ;15(2):3040–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sevastianova K, Santos A, Kotronen A, Hakkarainen A, Makkonen J, Silander K, Peltonen M, Romeo S, Lundbom J, Lundbom N, Olkkonen VM, Gylling H, Fielding BA, Rissanen A, Yki-Järvinen H. Effect of short-term carbohydrate overfeeding and long-term weight loss on liver fat in overweight humans. Am J Clin Nutr 2012; 96: 727–734. [DOI] [PubMed] [Google Scholar]
- 28.von Frankenberg AD, Marina A, Song X, Callahan HS, Kratz M, Utzschneider KM. A high-fat, high-saturated fat diet decreases insulin sensitivity without changing intra-abdominal fat in weight-stable overweight and obese adults. Eur J Nutr. 2017;56(1):431–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. https://www.cdc.gov/nchs/data/nhanes/databriefs/calories.pdf.
- 30.Koopman KE, Caan MW, Nederveen AJ, Pels A, Ackermans MT, Fliers E, la Fleur SE, Serlie MJ. Hypercaloric diets with increased meal frequency, but not meal size, increase intrahepatic triglycerides: a randomized controlled trial. Hepatology 2014; 60:545–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tchaikovski V, Olieslagers S, Böhmer FD, Waltenberger J. Diabetes mellitus activates signal transduction pathways resulting in vascular endothelial growth factor resistance of human monocytes. Circulation. 2009;120(2):150–9. [DOI] [PubMed] [Google Scholar]
- 32.He L, Wong CK, Cheung KK, Yau HC, Fu A, Zhao HL, Leung KM, Kong AP, Wong GW, Chan PK, Xu G, Chan JC. Anti-inflammatory effects of exendin-4, a glucagon-like peptide-1 analog, on human peripheral lymphocytes in patients with type 2 diabetes. J Diabetes Investig. 2013;4(4):382–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Olieslagers S, Pardali E, Tchaikovski V, ten Dijke P, Waltenberger J. TGFβ1/ALK5-induced monocyte migration involves PI3K and p38 pathways and is not negatively affected by diabetes mellitus. Cardiovasc Res. 2011. ;91 (3):510–8. [DOI] [PubMed] [Google Scholar]
- 34.Jeong HW, Hsu KC, Lee JW, Ham M, Huh JY, Shin HJ, Kim WS, Kim JB. Berberine suppresses proinflammatory responses through AMPK activation in macrophages. Am J Physiol Endocrinol Metab. 2009;296(4):E955–64. [DOI] [PubMed] [Google Scholar]
- 35.Johnson GL, Lapadat R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science. 2002;298(5600):1911–2. [DOI] [PubMed] [Google Scholar]
- 36.Jialal I, Kaur H, Devaraj S. Toll-like receptor status in obesity and metabolic syndrome: a translational perspective. J Clin Endocrinol Metab. 2014;99(1):39–48. [DOI] [PubMed] [Google Scholar]
- 37.Kochumon S, Wilson A, Chandy B, Shenouda S, Tuomilehto J, Sindhu S, Ahmad R. Palmitate Activates CCL4 Expression in Human Monocytic Cells via TLR4/MyD88 Dependent Activation of NF-κB/MAPK/ PI3K Signaling Systems. Cell Physiol Biochem. 2018;46(3):953–964. [DOI] [PubMed] [Google Scholar]
- 38.Kulawik A, Engesser R, Ehlting C, Raue A, Albrecht U, Hahn B, Lehmann WD, Gaestel M, Klingmüller U, Häussinger D, Timmer J, Bode JG. IL-1β-induced and p38MAPK-dependent activation of the mitogen-activated protein kinase-activated protein kinase 2 (MK2) in hepatocytes: Signal transduction with robust and concentration-independent signal amplification. J Biol Chem. 2017. April 14;292(15):6291–6302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bazzoni F, Tamassia N, Rossato M, Cassatella MA. Understanding the molecular mechanisms of the multifaceted IL-10-mediated anti-inflammatory response: lessons from neutrophils. Eur J Immunol. 2010;40(9):2360–8. [DOI] [PubMed] [Google Scholar]
- 40.Zhong Z, Umemura A, Sanchez-Lopez E, Liang S, Shalapour S, Wong J, He F, Boassa D, Perkins G, Ali SR, McGeough MD, Ellisman MH, Seki E, Gustafsson AB, Hoffman HM, Diaz-Meco MT, Moscat J, Karin M. NF-κB Restricts Inflammasome Activation via Elimination of Damaged Mitochondria. Cell. 2016. February 25;164(5):896–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Klionsky DJ et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 2016;12(1):1–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu K, Zhao E, Ilyas G, Lalazar G, Lin Y, Haseeb M, Tanaka KE, Czaja MJ. Impaired macrophage autophagy increases the immune response in obese mice by promoting proinflammatory macrophage polarization. Autophagy. 2015; 11(2): 271–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Koves TR, Ussher JR, Noland RC, Slentz D, Mosedale M, Ilkayeva O, Bain J, Stevens R, Dyck JR, Newgard CB, Lopaschuk GD, Muoio DM. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008;7(1):45–56. [DOI] [PubMed] [Google Scholar]
- 44.Kelley DE, Goodpaster B, Wing RR, Simoneau JA. Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am J Physiol. 1999;277(6):E1130–41. [DOI] [PubMed] [Google Scholar]
- 45.Taylor E, Huang N, Bodde J, Ellison A, Killiany R, Bachschmid MM, Hamilton J. MRI of atherosclerosis and fatty liver disease in cholesterol fed rabbits. J Transl Med. 16(1):215, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tuvdendorj D, Chandalia M, Batbayar T, Saraf M, Beysen C, Murphy EJ, Abate N. Altered subcutaneous abdominal adipose tissue lipid synthesis in obese, insulin-resistant humans. Am J Physiol Endocrinol Metab. 305(8):E999–E1006, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Abate N, Chandalia M. Risk of Obesity-Related Cardiometabolic Complications in Special Populations: A Crisis in Asians. Gastroenterology. 152(7):1647–1655, 2017. [DOI] [PubMed] [Google Scholar]
- 48.Mongraw-Chaffin M, Foster MC, Anderson CAM, Burke GL, Haq N, Kalyani RR, Ouyang P, Sibley CT, Tracy R, Woodward M, Vaidya D. Metabolically Healthy Obesity, Transition to Metabolic Syndrome, and Cardiovascular Risk. J Am Coll Cardiol. 2018;71(17):1857–1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Guo F, Garvey WT. Cardiometabolic disease risk in metabolically healthy and unhealthy obesity: Stability of metabolic health status in adults. Obesity (Silver Spring). 2016;24(2):516–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.