Summary
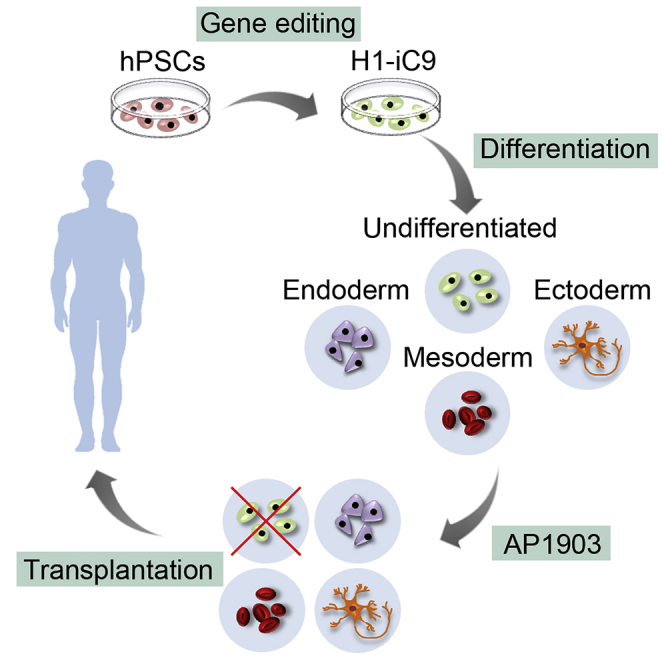
A major challenge in using human pluripotent stem cells (hPSCs) in therapy is the risk of teratoma formation due to contaminating undifferentiated stem cells. We used CRISPR-Cas9 for in-frame insertion of a suicide gene, iC9, into the endogenous SOX2 locus in human embryonic stem cell (ESC) line H1 for specific eradication of undifferentiated cells without affecting differentiated cells. This locus was chosen over NANOG and OCT4, two other well-characterized stem cell loci, due to significantly reduced off-target effect. We showed that undifferentiated H1-iC9 cells were induced to apoptosis by iC9 inducer AP1903, whereas differentiated cell lineages including hematopoietic cells, neurons, and islet beta-like cells were not affected. We also showed that AP1903 selectively removed undifferentiated H1-iC9 cells from a mixed cell population. This strategy therefore provides a layer of safety control before transplantation of a stem-cell-derived product in therapy.
Subject Areas: Cellular Therapy, Techniques in Genetics, Stem Cells Research
Graphical Abstract
Highlights
-
•
In-frame iC9 gene insertion into the SOX2 locus to target undifferentiated hESCs
-
•
The residual ESCs are selectively removed without affecting their differentiated progeny
-
•
Eradication of residual ESCs before transplantation provides an extra safety control
Cellular Therapy; Techniques in Genetics; Stem Cells Research
Introduction
One of the many promises of hPSCs is to provide a platform to support the treatment of human diseases (Thomson et al., 1998). Protocols built on knowledge gained from studying the developmental cues for a particular tissue or organ have been established to guide hPSC differentiation (Klimanskaya et al., 2004, Ng et al., 2005, Pagliuca et al., 2014, Rezania et al., 2014, Talkhabi et al., 2016, Wichterle et al., 2002). Although most hPSC-derived cells are not functionally mature as their counterparts in adult tissues, a few of them including cardiomyocytes, retina pigment epithelial cells, and pancreatic progenitor cells have entered clinical trials or are set to enter clinical trials soon (Menasche et al., 2018, Schwartz et al., 2015, Song et al., 2015). Despite these progresses, important challenges remain. One major challenge is the risk of teratoma formation due to contamination of undifferentiated hPSCs in hPSC-derived cell products. In almost all cases, stem-cell-derived cell products are comprised of a mixed cell population with the desired cell type constituting only a fraction of the entire population. If sufficient quantities of undifferentiated hPSCs remained, they could form teratomas in patients upon transplantation (Hentze et al., 2009). This risk represents a major hurdle in using hPSCs in cell-based therapy.
The use of a suicide gene to selectively eradicate undifferentiated hPSCs represents an appealing strategy to address this hurdle (Li and Xiang, 2013). It relies on the ability of the suicide gene product to convert a non-toxic and bio-inert prodrug into a cytotoxic product that triggers cell death. Selective expression of a suicide gene only in undifferentiated hPSCs would remove these cells from differentiated cell products. Previous attempts to express suicide genes to control graft-versus-host disease or to ablate transformed cells mainly relied on random insertion of the suicide gene into host chromosomes (Bonini et al., 1997, Zarogoulidis et al., 2013). Limiting suicide gene expression only in undifferentiated hPSCs with such a strategy would be difficult. Random gene insertion also increases the risk of insertional mutagenesis (Bokhoven et al., 2009). Modulating expression of a suicide gene from stem cell loci addresses these concerns. Direct control of the suicide gene from a stem cell locus ensures rapid shutdown of the suicide gene once hPSCs are committed to differentiate. Precise gene insertion also avoids the risk of insertional mutagenesis.
In-frame insertion of a fluorescence reporter into the well-characterized stem cell OCT4 locus in hPSCs for monitoring cell-fate decision in real time has been reported previously (Balboa et al., 2017, Hockemeyer et al., 2009, Krentz et al., 2014, Zhu et al., 2015). However, close examination of the sequence near the OCT4 stop codon, the target site for in-frame gene insertion, revealed multiple genomic sites with similar or identical sequences (Figure S1). It is likely that these off-target sites would be cleaved by the OCT4-specific nucleases used in those studies and generate insertion-deletion (indel) or other lesions such as chromosome translocation, although such a possibility was not investigated in those studies. Because of the potential off-target effect with the nucleases used in the OCT4 locus, we considered two other well-characterized stem cell loci, NANOG and SOX2, for suicide gene insertion. As the OCT4 locus, several genomic sites exhibit sequence homology with the target site in the NANOG locus for in-frame gene insertion (Figure S1). In contrast, only one additional genomic site exhibits such sequence homology with the target site in the SOX2 locus (Figure S1). To minimize the off-target effect, we therefore focused our effort in the current study on the insertion of a suicide gene into the SOX2 locus for selective eradication of undifferentiated hPSCs.
Two suicide strategies are widely used in cell-based therapy, including herpes simplex virus thymidine kinase (HSV-TK) and inducible caspsae-9 (iC9) (Zarogoulidis et al., 2013). HSV-TK induces cell death by converting the non-toxic prodrug ganciclovir (GCV) into a toxic form to block DNA replication (Moolten, 1986, Reardon, 1989). Multiple studies have demonstrated the effectiveness of expressing HSV-TK to kill undifferentiated hPSCs (Liang et al., 2018, Schuldiner et al., 2003). Since this system relies on cell division, it is not suitable for treating proliferating cells such as differentiated progenitor cells to remove undifferentiated hPSCs. In the current study, we focused on the iC9 suicide system for the removal of contaminating undifferentiated hPSCs from stem cell-derived products before transplantation. The iC9 suicide gene encodes a fusion protein between human Caspase 9 and FK506-binding protein (Straathof et al., 2005). Individual iC9 subunits do not induce cell apoptosis. Dimerization of the iC9 subunits can be induced by a small molecule AP1903, which is well tolerated in culture cells and in clinical studies (Clackson et al., 1998). Dimerization of iC9 activates one of the last steps in the apoptotic cascade, resulting in rapid cell death. To maintain stem cell pluripotency, levels of SOX2 need to be stringently regulated (Boer et al., 2007, Kopp et al., 2008). In-frame insertion of the iC9 gene following the coding region of the SOX2 gene minimizes the risk of disrupting normal SOX2 expression. In the current study, this site-specific gene insertion was achieved by using CRISPR-Cas9 in human embryonic stem cell (ESC) line H1. We showed that iC9 gene insertion led to the eradication of undifferentiated H1 cells without affecting the viability of multiple H1-derived cell lineages, including hematopoietic cells, neurons, and pancreatic beta-like cells. Our results demonstrate that suicide gene insertion into the SOX2 locus is an effective strategy to selectively eradicate undifferentiated hPSCs and prevent teratoma formation. This strategy therefore provides a layer of safety control to reduce the risk of using hPSC-derived cell products in therapy.
Results
Stem Cells Expressing iC9 Are Selectively Eradicated by AP1903 Treatment
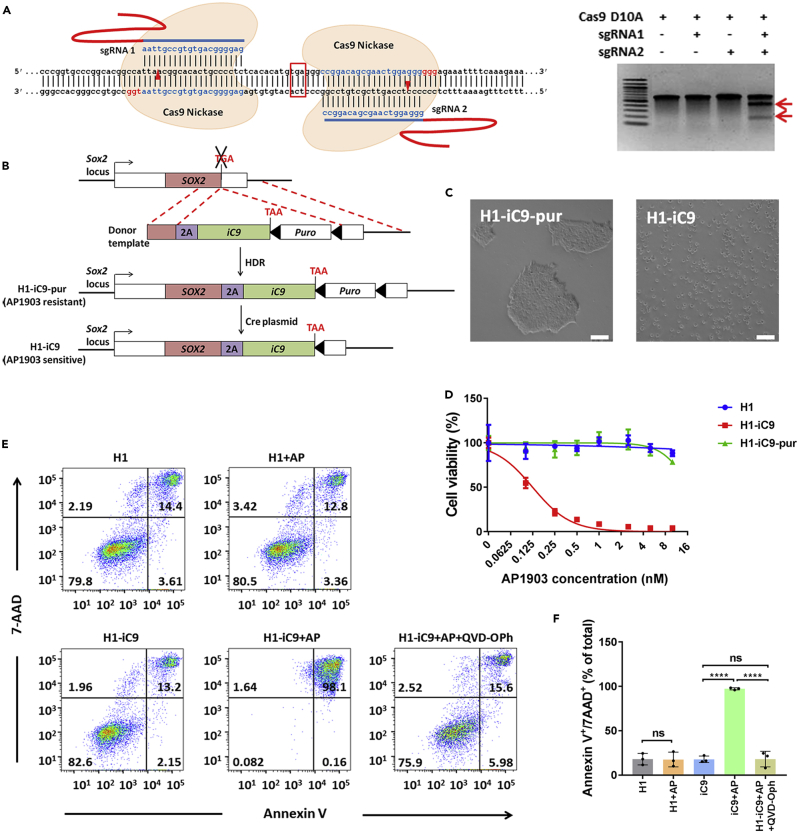
To selectively express iC9 in undifferentiated hPSCs but not in their differentiated progeny, we used CRISPR-Cas9 to insert the iC9 gene into the SOX2 locus in H1 cells. A pair of sgRNA targeting a region near the stop codon of the SOX2 locus was designed (Figure 1A, left). This pair, sgRNA1 and sgRNA2, efficiently cleaved their target at the SOX2 locus when co-expressed with Cas9 nickase (Ran et al., 2013), whereas Cas9 nickase with single sgRNA could not generate the cleavage (Figure 1A, right). Because a steady level of SOX2 is crucial for the maintenance of stem cell pluripotency and self-renewal, our strategy involves in-frame fusion of a 2A-iC9 transgene cassette into the SOX2 locus to minimize the risk of disrupting SOX2 expression (Figure 1B). Inclusion of the self-cleaving 2A peptide between the SOX2 and iC9 proteins is expected to facilitate normal production of SOX2. We used a donor template harboring the 2A-iC9 and the puromycin-resistant (Puro) gene encoding puromycin N-acetyl transferase flanked by homology arms spanning the stop codon of the SOX2 gene for homologous recombination (Figures 1B and S2A). After puromycin selection, we picked three H1-iC9-pur clones containing monoallelic insertion of the iC9 gene for further analysis (Figures S2B and S2C).
Figure 1.
CRISPR-Cas9-Mediated iC9 Gene Insertion into the SOX2 Locus Renders H1 Cells Susceptible to the Killing by AP1903
(A) Left: sequences of sgRNA1, sgRNA2, and their genomic targets in the SOX2 locus. Stop codon of the SOX2 coding region is boxed. PAM sequence is labeled in red. Cleavage site of CRISPR-Cas9 is marked by the red arrowhead. Right: Cas9 D10A nickase-mediated cleavage of the SOX2 genomic target was analyzed by the Surveyor assay in transiently transfected HEK293T cells. Two red arrows mark the expected cleavage products.
(B) Scheme to use CRISPR-Cas9 to establish H1 clones with in-frame insertion of the 2A-iC9 transgene into the endogenous SOX2 locus (H1-iC9-pur clone) and to remove the Puro gene by Cre/loxP-mediated excision (H1-iC9 clone). Broken lines specify the regions of homology between the donor template and the SOX2 locus for homologous recombination. Filled arrowheads indicate the loxP site. HDR: homology-directed recombination.
(C) AP1903 treatment of H1-iC9-pur and H1-iC9 cells. Treatment was carried out overnight in media containing 100 nM AP1903. Scale bar = 50 μm.
(D) Cell viability after overnight treatment with AP1903 at the indicated concentration determined by MTS assay for H1, H1-iC9-pur, and H1-iC9 cells. The iC50 of AP 1903 in H1-iC9 cells was 0.1242 ± 0.0083 nM calculated from the dose-response curve. Values represent mean ± s.d. (N = 4 independent experiments).
(E and F) Analysis of apoptotic cells after overnight treatment with AP1903 at a concentration of 10 nM. qVD-Oph at a concentration of 20 mM was applied simultaneously with AP1903 for overnight treatment. Representative FACS profiles (E) and the bar graph that depicts Annexin V+/7AAD+ apoptotic cells from three independent experiments (F) were shown. Bars indicate the mean (±s.d.) of individual data points. ****p < 0.0001, ns: not statistically significant.
Treatment of H1-iC9-pur cells with 100 nM AP1903 led to no observable cell death (Figure 1C). Because previous studies showed that positive selection cassettes could interfere with the expression of the neighboring gene (Davis et al., 2008, Zhu et al., 2015), we delivered Cre-expressing plasmid into H1-iC9-pur clones by nucleofection to remove the Puro gene flanked by loxP sites and established the H1-iC9 clones (Figures 1B and S3). Unlike H1-iC9-pur cells, AP1903 treatment efficiently eradicated H1-iC9 cells (Figure 1C). Karyotype analysis showed that both H1-iC9-pur and H1-iC9 are normal as parental H1 cells (Figure S4). To assay for off-target effect of the two sgRNAs used, we sequenced the potential off-target site on chromosome 8 (Figure S1) in H1-iC9 cells for indel formation and found none (data not shown). In addition, sequence analysis of the predicted top five off-target sites for each individual sgRNA exhibited no nucleotide changes (Table S1). These results are consistent with the previous report that the use of paired nickases in gene editing significantly reduced the off-target effect (Ran et al., 2013).
To assess the drug sensitivity, H1, H1-iC9-pur, and H1-iC9 cells were exposed to increasing concentrations of AP1903. A dose-dependent cytotoxicity of the drug was observed in H1-iC9 cells with an IC50 of 0.1242 nM, whereas the viability of parental H1 cells and H1-iC9-pur cells was not affected by the treatment (Figure 1D). AP1903-induced toxicity was further analyzed by flow cytometry (Figures 1E and 1F). There was no significant increase in apoptotic cells when H1 cells were exposed to 10 nM AP1903 relative to the untreated control. In contrast, the majority of H1-iC9 cells became apoptotic with the same treatment. Cell killing was mediated through the Caspase signaling pathway as demonstrated by the block of cell death with pan-Caspase inhibitor qVD-Oph (Figures 1E and 1F) (Caserta et al., 2003).
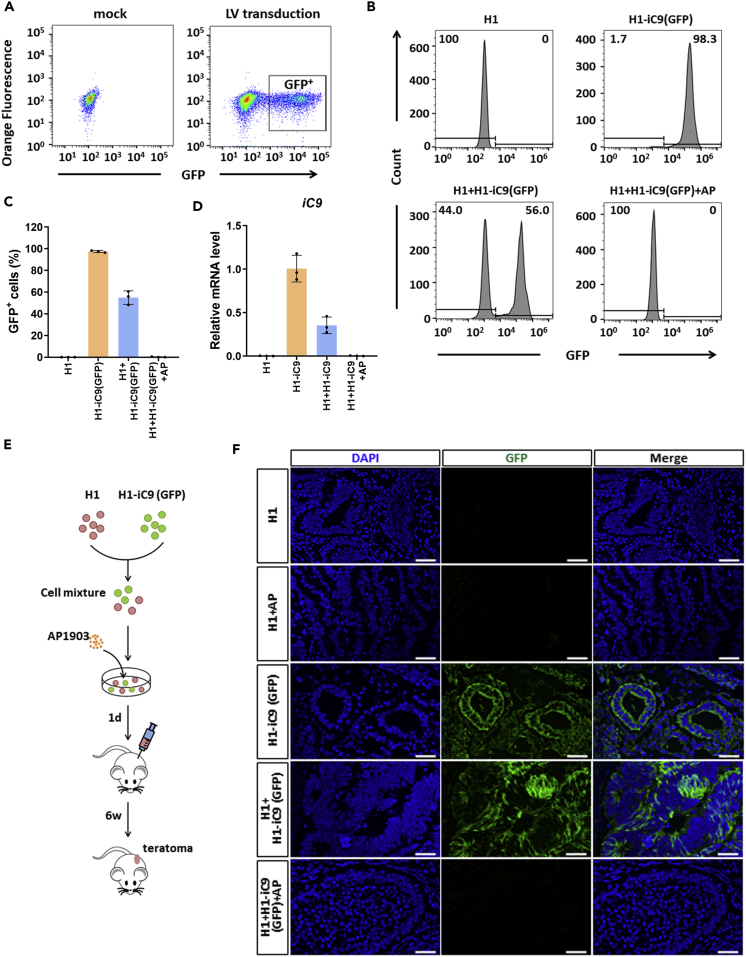
To determine whether AP1903 can selectively eradicate iC9-expressing cells, we transduced H1-iC9 cells with a GFP lentivirus (Figures S5A and 2A). The sorted GFP+ cells, H1-iC9(GFP), were mixed with H1 cells. AP1903 exposure completely eradicated the population of H1-iC9(GFP) cells (Figures 2B–2D). Killing of the GFP+ cell population was also detected by fluorescence microscopy, whereas most of the GFP− cell remained viable with the drug treatment (Figure S5B). Inoculation of H1-iC9(GFP) cells into NOD scid gamma (NSG) mice led to the formation of teratomas containing cells derived from the three germ layers (Figure S6), suggesting that iC9 expression did not affect the pluripotency of H1 cells. To validate selective eradication of H1-iC9(GFP) cells from a mixed cell population, H1 and H1-iC9(GFP) cells mixed at a ratio of 1:1 were either mock treated or treated with AP1903 followed by transplantation into NSG mice (Figure 2E). Both GFP+ and GFP− cells were present in teratomas derived from mock treatment (Figure 2F). Treatment with AP1903 before transplantation gave rise to teratomas containing no detectable GFP+ cells (Figure 2F). Taken together, these results demonstrated the susceptibility of iC9-expressing H1 cells to AP1903.
Figure 2.
iC9-Expressing Cells Are Selectively Eradicated by AP1903
(A) Marking of H1-iC9 cells with GFP. Structure of the lentiviral vector containing the GFP gene is shown. H1-iC9 cells were transduced with the GFP vector at a multiplicity of infection of two, followed by cell sorting 48 h after transduction. GFP+ cells were pooled and expanded.
(B and C) Flow cytometry analysis of H1 and H1-iC9(GFP) cells either alone or in mixtures with and without AP1903 treatment as indicated. (B) FACS plots present the percentage of GFP+ cells in each group. (C) Bar graph shows mean (±s.d.) of pooled data from three independent experiments.
(D) Expression of iC9 transgene mRNA in the indicated cells. Results of three independent experiments are shown as the mean ± s.d.
(E) Experimental scheme for the teratoma formation assay. H1 and H1-iC9(GFP) cells were mixed at an equal dose and either mock treated or treated with AP1903 overnight at a concentration of 10 nM, followed by subcutaneous injection of 2 × 106 cells into NSG mice with four mice per group. Teratomas were removed six weeks after injection.
(F) Representative immunofluorescence staining of GFP in teratoma sections derived from indicated cells. Scale bar = 50 μm.
Resistance of H1-iC9-Derived Hematopoietic Cells to AP1903 Treatment
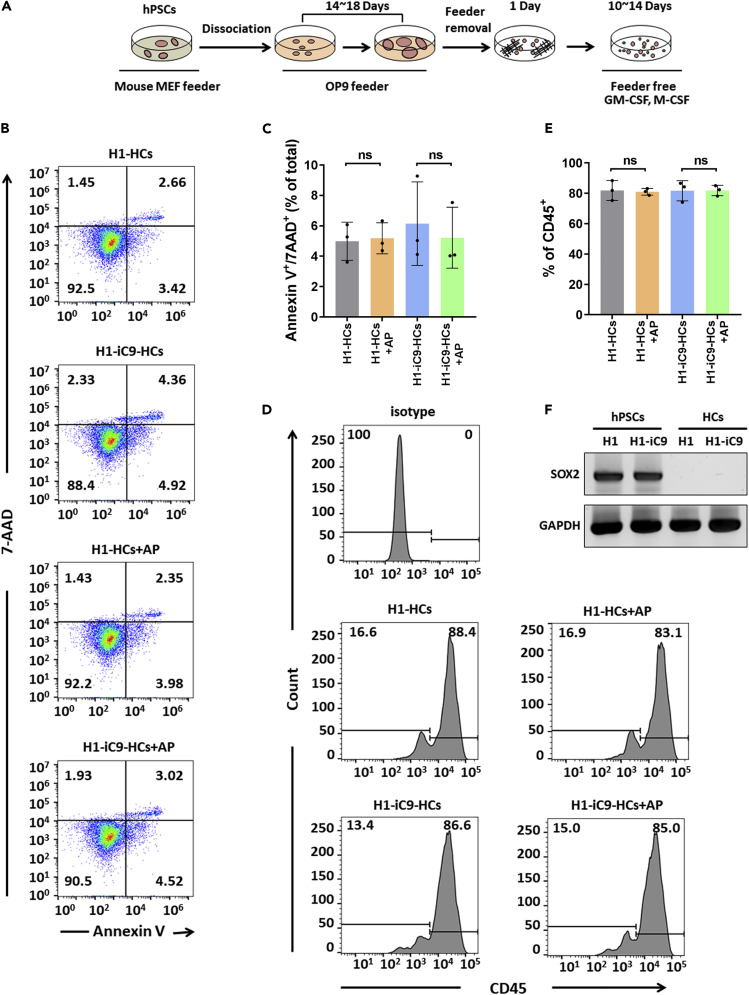
To determine the effect of iC9 on hPSC-derived cell lineages, we first differentiated H1-iC9 cells into hematopoietic cells (Figure 3A). AP1903 treatment of the hematopoietic populations derived from H1 and H1-iC9 cells showed little change in the percentage of apoptotic cells compared with mock control (Figures 3B and 3C). Flow cytometry analysis showed that the percentage of CD45+ hematopoietic cells derived from H1 and H1-iC9 cells was not altered significantly by AP1903 treatment (Figures 3D and 3E). Quantitative RT-PCR analysis showed that SOX2 transcript disappeared upon hematopoietic differentiation (Figure 3F). Thus, differentiation of H1-iC9 cells into the CD45+ hematopoietic cell lineage silenced iC9 expression, rendering these cells resistant to AP1903-induced apoptosis.
Figure 3.
Effect of AP1903 Treatment on H1-iC9-Derived Hematopoietic Cells
(A) Scheme of differentiation from hPSCs to hematopoietic cells.
(B and C) Flow cytometry analysis of apoptosis of hematopoietic cells derived from H1 and H1-iC9 cells with AP1903 treatment. Representative FACS profiles (B) and the bar graph showing percentage of apoptotic cells from three independent experiments (C) were shown and presented as the mean ± s.d. ns: p value not significant.
(D and E) The fraction of CD45+ hematopoietic cells derived from H1 and H1-iC9 differentiation was not affected by AP1903 treatment. Mock- or AP1903-treated hematopoietic cells were analyzed by flow cytometry. FACS plots (D) and bar graph that represents the frequency of CD45+ (E) were shown and presented as the mean ± s.d. (N = 3 independent experiments) ns: not statistically significant.
(F) Semi-quantitative RT-PCR analysis of the SOX2 transcript in undifferentiated H1 and H1-iC9 cells as well as in the hematopoietic populations derived from these two cell lines.
Resistance of H1-iC9-Derived Neurons to AP1903 Treatment
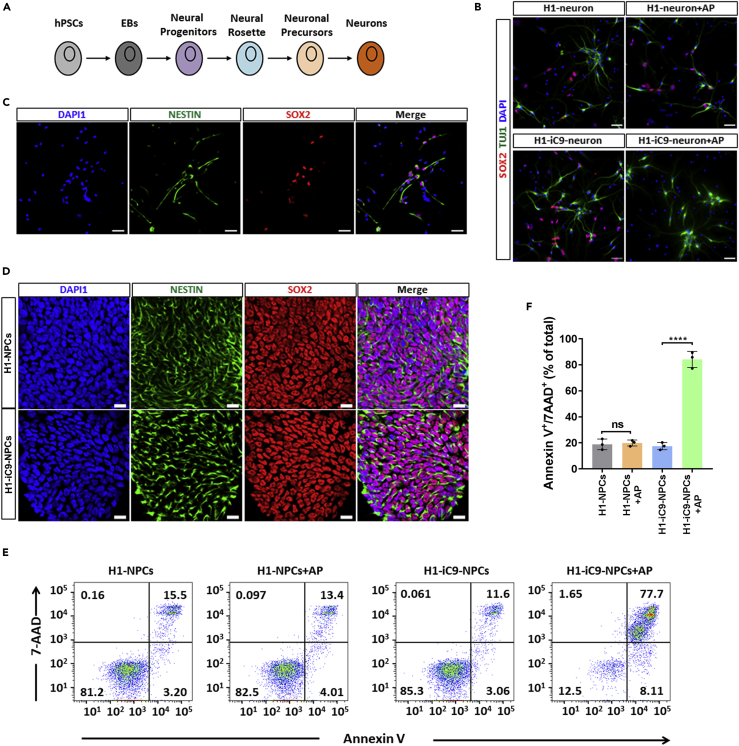
SOX2 is known to express in neural progenitor cells (NPCs) where it regulates proliferation/survival and neurogenesis (Graham et al., 2003). However, its expression is down-regulated during terminal neuronal differentiation (Graham et al., 2003). We differentiated H1 and H1-iC9 cells into neurons (Figure 4A). Many cells in the differentiated culture expressed the terminal differentiated neuronal marker class III beta-tubulin (TuJ1) (Figure 4B). Although a portion of SOX2+ cells existed in the differentiated neuronal culture derived from H1 and H1-iC9, the majority of TuJ1+ neurons lacked SOX2 expression (Figure 4B). These SOX2+ cells in the H1-iC9-derived culture were eradicated by AP1903 treatment, whereas most of the TuJ1+ cells were not affected (Figure 4B). By contrast, AP1903 treatment did not seem to affect the viability of the SOX2+ cells in the H1-derived culture. Immunofluorescence study confirmed that the SOX2+ cells in the H1-iC9-derived culture were NPCs, which co-expressed the progenitor marker Nestin (Figure 4C). In support of this conclusion, we differentiated H1 and H1-iC9 cells into NPCs, and immunofluorescence staining demonstrated that the majority of the cells stained positive for both Nestin and SOX2 (Figure 4D). Quantitative real-time PCR showed that expression of NANOG and OCT4 was undetectable in the NPC culture in contrast to readily detectable SOX2, indicating highly efficient differentiation (Figure S7). AP1903 treatment led to a significant loss of these cells derived from H1-iC9 cells but not those from H1 cells (Figures 4E and 4F). Thus, this suicide system permits selective removal of SOX2+ cells without affecting terminal differentiated neurons in the same culture.
Figure 4.
Effect of AP1903 Treatment on H1-iC9-Derived Neuronal Cells
(A) Scheme of neuron differentiation from hPSCs.
(B and C) Representative immunofluorescence staining of SOX2/TUJ1 (B) and SOX2/NESTIN (C) in mock- and AP1903-treated neuronal cultures derived from H1 and H1-iC9 cells. Scale bar = 50 μm.
(D) Representative immunofluorescence staining of Nestin and SOX2 in NPCs derived from H1 and H1-iC9 cells. Scale bar = 50 μm.
(E) Representative of flow cytometry analysis of cell apoptosis after overnight treatment of H1 and H1-iC9-derived NPCs with AP1903.
(F) Quantification of the apoptotic cell fraction from three independent experiments. Bars indicate the mean ± s.d., ****p < 0.0001, ns: not statistically significant.
Resistance of H1-iC9-Derived Beta-like Cells to AP1903 Treatment
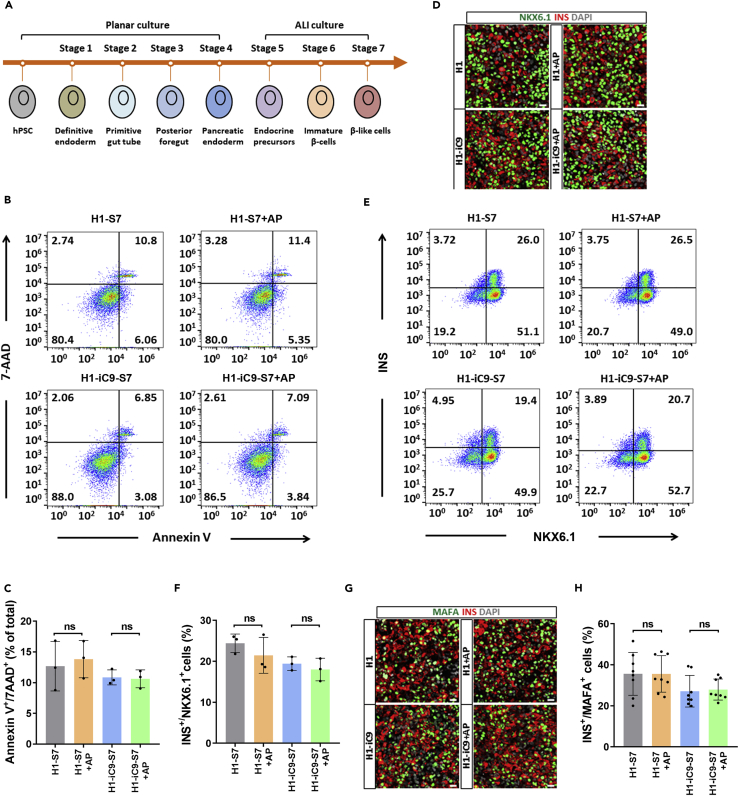
To evaluate the effect of iC9 on hPSC-derived endocrine lineages, we adopted a seven-stage differentiation protocol to generate pancreatic beta-like cells from H1 and H1-iC9 cells (Figure 5A) (Rezania et al., 2014). AP1903 treatment did not noticeably alter the viability of differentiated cells derived from either H1 or H1-iC9 in stage 4 (S4) or S7 (Figures 5B, 5C, and S8A). Examination of AP1903-treated S4 cells revealed no major change in the fraction of pancreatic progenitor cells marked by PDX1 or NKX6.1 expression (Figure S8B). More importantly, the fractions of NKX6.1+/Insulin+ cells and MAFA+/Insulin+ cells considered to be more mature beta cells in S7 were not affected by AP1903 treatment (Figures 5D–5H). Thus, H1- and H1-iC9-derived pancreatic progenitor cells and beta-like cells are resistant to AP1903 treatment, presumably due to down-regulation of SOX2 expression.
Figure 5.
H1-iC9-Derived Beta-like Cells Are Resistant to AP1903 Treatment
(A) Seven-stage differentiation of hPSCs into beta-like cells. Cells were grown in monolayer from stage 1 to stage 4 and in an air-liquid (ALI) interphase from stage 5 to stage 7.
(B) Flow cytometry analysis of the apoptotic cells in the S7 population derived from H1 and H1-iC9 cells after overnight AP1903 treatment.
(C) Quantification of the fraction of Annexin V+/7AAD+ apoptotic cells from three independent experiments. Data present the mean frequency ±s.d., ns: not statistically significant.
(D) Representative immunofluorescence staining of NKX6.1+/INS+ cells in the S7 population derived from H1 and H1-iC9 cells with and without AP1903 treatment.
(E) Flow cytometry analysis of NKX6.1+ and INS+ cells in H1 or H1-iC9-derived S7 cells with and without AP1903 treatment.
(F) Quantification of NKX6.1+/INS+ cell proportion from three independent experiments. Data indicate the mean ± s.d., ns: not statistically significant.
(G) Representative immunofluorescence staining of MAFA+ and INS+ cells.
(H) Quantification of the fraction of MAFA+/INS+ beta-like cells. Bars represent the mean ± s.d. from three independent experiments, ns: not statistically significant. MAFA+/INS+ beta-like cells were counted in eight randomly picked fields. Scale bar = 20 μm.
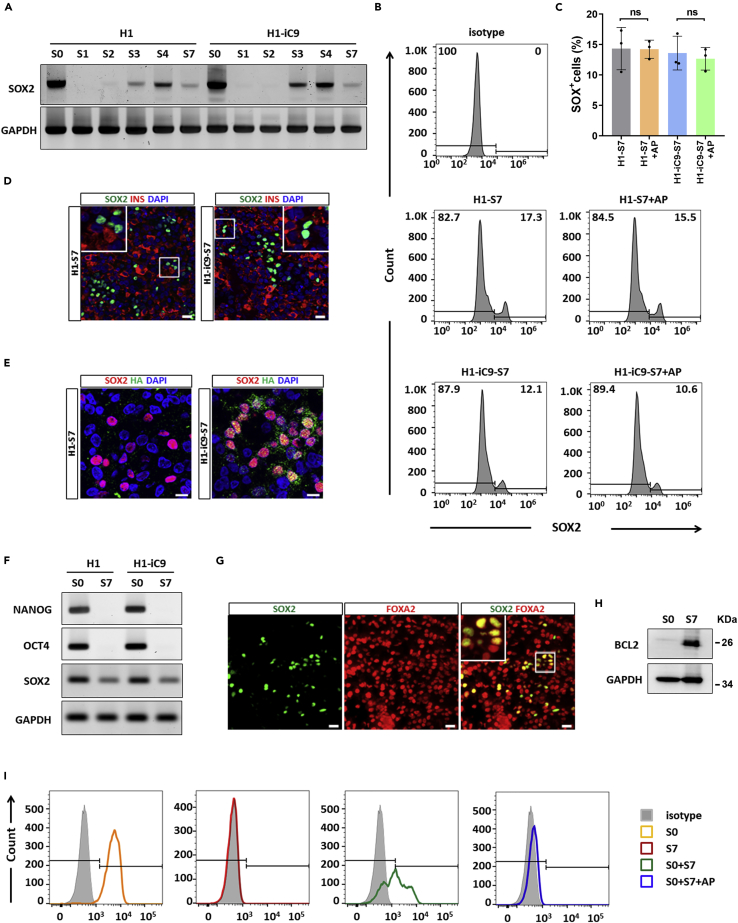
However, real-time PCR of the SOX2 transcript showed that although SOX2 expression was significantly down-regulated in S1 and S2 during pancreatic differentiation of H1 and H1-iC9 cells, it was reactivated in S3, peaked in S4, and persisted at lower levels in S7 (Figure 6A). Flow cytometry analysis of S7 cells showed SOX2+ cells in both H1- and H1-iC9-derived cultures, and AP1903 treatment did not lead to the elimination of the SOX2+ cells (Figures 6B and 6C). Immunofluorescence staining of insulin and SOX2 confirmed that these SOX2+ cells were not beta-like cells (Figure 6D). The iC9 transcript displayed a similar pattern of changes during differentiation relative to the SOX2 transcript (Figure S9). In consistent with the transcript, iC9 protein was detected in SOX2+ cells at S7, indicating that transgene was not shutdown (Figure 6E). To ensure that AP1903 resistance of these SOX2+ cells is not due to iC9 mutation, we sequenced the iC9 transcript in the S7 culture and found no changes in its sequence (data not shown). To determine whether these SOX2+ cells are contaminating undifferentiated cells, we performed real-time PCR of S7 cells and found no trace of OCT4 and NANOG transcripts (Figure 6F). Immunofluorescence staining and FACS analysis confirmed that SOX2+ cells in the S7 population failed to co-express OCT4 or NANOG but co-expressed definitive endoderm marker FOXA2 (Figures 6G and S10). Moreover, mRNA level of FOXA2 was dramatically increased in S4 and S7 cells compared with undifferentiated cells, indicating the generation of definitive endoderm cells upon differentiation (Figure S11). It is well known that the pluripotency marker SOX2 reemerges as an anterior foregut marker (Green et al., 2011). We thus analyzed transcripts of other anterior foregut markers including HOXA1, HOXA2, and PAX9. In contrast to SOX2 gene, which was highly expressed in undifferentiated cells, expression of all these markers was undetectable in both H1 and H1-iC9 cells. PAX9 expression remained undetectable upon differentiation (data not shown). By contrast, HOXA1 and HOXA2 expression was markedly up-regulated in S4 cells and down-regulated in S7 cells (Figure S11). Collectively, these data suggested the possibility that SOX2+ cells reappeared during beta-like cell differentiation were endoderm-derived anterior foregut cells.
Figure 6.
SOX2+ Cells in the S7 Population Remain Viable after AP1903 Treatment Are Not Undifferentiated Stem Cells
(A) Reactivation of SOX2 expression during H1 and H1-iC9 cell differentiation into beta-like cells. Semi-quantitative RT-PCR was carried out using total RNAs isolated from the indicated differentiation stages as defined by Rezania et al. (Rezania et al., 2014).
(B and C) Resistance to AP1903 treatment of SOX2+ cells in the S7 population. Flow cytometry analysis of the SOX2+ expression in mock- and AP1903-treated S7 populations derived from H1 and H1-iC9 cells and quantification of SOX2+ proportion from B. Bars present the mean ± s.d. of three independent experiments. ns: not statistically significant.
(D) Representative immunofluorescence staining of SOX2 and insulin in S7 cells. Scale bar = 20 μm.
(E) Co-staining of SOX2 and the HA tag in iC9 in the S7 population derived from H1 and H1-iC9 cells. Cell nuclei were counterstained with DAPI. Scale bar = 10 μm.
(F) Semi-quantitative RT-PCR analysis of NANOG, OCT4, and SOX2 transcripts in undifferentiated (S0) and the Stage 7 (S7) populations derived from H1 and H1-iC9 cells.
(G) Representative immunofluorescence staining of SOX2 and FOXA2 in the H1-iC9-derived S7 population. Scale bar = 20 μm.
(H) Western blot analysis of BCL2 expression in S0 and S7 populations derived from H1-iC9 cells.
(I) Selective eradication of undifferentiated H1-iC9 cells spiked into H1-iC9-derived S7 population. Undifferentiated H1-iC9 cells spiked into H1-iC9-derived S7 population were treated with 10 nM AP1903 overnight. The resulting cell mixture was stained for OCT4 and analyzed by flow cytometry.
Previous studies showed that the Caspase 9 signaling cascade plays a role in amplifying mitochondrial disruption through cleavage of anti-apoptotic BCL-2 family proteins, and expression of a cleavage-resistant BCL-2 mutant or overexpression of wild-type BCL-2 inhibited Caspase-9-mediated apoptosis (Chen et al., 2007, Yang et al., 1997). Since BCL-2 expression was shown to be up-regulated upon differentiation of mouse ESCs and hPSCs (LeBlanc et al., 2018, Lee et al., 2013), we carried out Western blot analysis to detect BCL-2 expression in H1-iC9 and H1-iC9-derived S7 cells. As shown in Figure 6H, the level of BCL-2 in the S7 population was strongly up-regulated compared with that in undifferentiated cells. This result raises the possibility that up-regulation of BCL-2 may play a role in conferring the resistance of SOX2+ cells in S7 to AP1903 treatment.
Selective Removal of Undifferentiated H1-iC9 Cells from the S7 Beta-like Cell Population
To determine whether contaminating undifferentiated stem cells can be removed from the S7 cell population, undifferentiated H1-iC9 cells were spiked into H1-iC9-derived S7 culture followed by AP1903 treatment. Undifferentiated H1-iC9 cells were identified by the expression of OCT4, a stem-cell-specific marker. As shown in Figure 6I, AP1903 treatment selectively eliminated undifferentiated H1-iC9 cells without affecting the viability of OCT4- S7 cells. These data also suggested that the S7 culture condition or the cell microenvironment was not inhibitory to AP1903-induced cell apoptosis. Based on these results, we concluded that the iC9 suicide system could be applied to selectively eliminate contaminating undifferentiated hPSCs without affecting differentiated beta-like cells.
Discussion
The genetic manipulation we carried out in the SOX2 locus did not affect the stemness of H1-iC9 cells, as they continued to express OCT4, SOX2, and NANOG at a similar level as the parental H1 cells (Figure 6F). The pluripotency of H1-iC9 cells was not affected neither as shown by their ability to form teratomas and to differentiate into hematopoietic cells, neurons, and pancreatic beta-like cells at similar efficiencies as H1 parental cells. Thus, ectopic expression of the iC9 gene product causes little adverse effect on the ability of self renewal and pluripotency of H1 cells. This approach therefore provides a platform to establish suicide-gene-containing hPSCs to facilitate their safe application in cell-based therapy.
In the current study, we chose the SOX2 locus for suicide gene insertion because of minimum potential off-target effect compared with the OCT4 and NANOG loci. By contrast, multiple genomic sites exhibit sequence homology with the on-target sites at the OCT4 and NANOG loci. It is therefore difficult to design sgRNAs that specifically cleave only the on-target sequence at these two loci. Zhu et al. previously reported the design of a sgRNA for in-frame insertion of fluorescence marker genes into the OCT4 locus in hESCs (Zhu et al., 2015). All potential OCT4 off-target sequences identified in our study except the one on chromosome 3 exhibit identical sequences to the OCT4 on-target sequence reported by Zhu et al. (Zhu et al., 2015). It is highly likely that the sgRNA they designed would generate double-strand breaks (DSBs) at these off-target sites, increasing the likelihood of indel formation or chromosome translocation. To reduce the risk of off-target cleavage, we used Cas9 nickase together with two sgRNAs targeting the site at the SOX2 locus for iC9 gene insertion. We sequenced the potential off-target site on chromosome 8 and the top five predicted off-target sites for each sgRNA in H1-iC9 cells and detected no sequence changes (Table S1), confirming the safety of using such a strategy to insert the suicide gene into a well-characterized stem cell locus.
Our result showed that although SOX2 expression was initially down-regulated during H1 differentiation into pancreatic beta-like cells, it was reactivated at stage 3 during posterior foregut formation, and SOX2+ cells persisted into S7 when beta-like cells emerged (Figure 6A). The emergence of these cells was not an artifact caused by gene editing, as they also existed in H1-derived S7 cells. In addition, our data showed that these SOX2+ cells were not undifferentiated stem cells or beta-like cells. SOX2 is strongly expressed in undifferentiated stem cells and becomes down-regulated during endoderm commitment. SOX2 expression reappears upon anterior-posterior patterning of the definitive endoderm to form foregut and persists in anterior foregut (Que et al., 2007). The co-expression of SOX2 and FOXA2 (Figure 6G) as well as the reactivation of HOXA1 and HOXA2 (Figure S11) suggested that SOX2+ cells generated during beta cell differentiation were anterior foregut endoderm cells. These results were consistent with the RNA-seq analysis for beta-cell differentiation published recently by Melton group (Veres et al., 2019). The production of these SOX2+ cells also reflects the fact that current differentiation protocols produced mixed cell population including small portion of non-endocrine cells (Veres et al., 2019). Although the differentiation protocol we used favors the formation of posterior foregut in S3 (Rezania et al., 2014), some anterior foregut cells could conceivably be generated by some small molecules. Our protocol includes bone morphogenetic protein (BMP) inhibitor LDN in S3 and S4 during differentiation. Inhibition of BMP signaling after specification of definitive endoderm from hPSCs has been shown to reactivate SOX2 expression in derived anterior foregut cells (Davenport et al., 2016, Green et al., 2011).
The finding that S7 cells expressed high levels of the anti-apoptotic BCL-2 protein could partially explain AP1903 resistance of SOX2+ cells. BCL-2 family proteins regulate the intrinsic apoptotic pathway by blocking the mitochondrial disruption that triggers the activation of Caspase 9 (Youle and Strasser, 2008). Although BCL-2 presumably acts upstream of Caspase, several studies showed that the Caspase 9 signaling cascade also induces mitochondrial disruption through the cleavage of BCL-2 (Chen et al., 2007, Cheng et al., 1997), thereby amplifying the intrinsic apoptotic pathway. High levels of BCL-2 protein may block this positive feedback loop and render the cells resistant to apoptosis. Down-regulation of BCL-2 or related anti-apoptotic proteins in the S7 culture with siRNA or shRNA may ultimately confirm the role of these proteins on the resistance to AP1903 treatment.
The use of various suicide gene systems to eliminate undifferentiated PSCs and prevent teratoma formation has been reported in several studies. For the iC9 suicide system, PSCs from different species transduced with lentivirus expressing iC9 could be effectively eradicated by AP 1903 (Wu et al., 2014, Yagyu et al., 2015). Another widely used suicide gene based on the HSV-TK/GCV induces cell killing by disruption of DNA synthesis and inhibition of cell proliferation (Cheng et al., 2012, Schuldiner et al., 2003). More recently, Qadir et al reported a new strategy by co-introduction of two suicide genes encoding HSV-TK and Escherichia coli nitroreductase (NTR) (Qadir et al., 2019). The strategy establishes a double-safe mechanism to ensure the eradication of any potential tumorigenic escapee while preserving differentiated cells through Cre recombination. An alternative approach using virus-like particles (VLPs) labeling with antibodies against a surface marker of hPSCs, SSEA-5, to deliver cytosine deaminase (CD) was designed to eliminate dividing cells with 5-fluorocytosine (5-FC) (Rampoldi et al., 2018). This strategy avoids the biosafety issue raised by transgenetic modification, but the efficiency of targeting undifferentiated cells remains to be tested. For example, because 3D culture systems are widely used in various cell differentiation protocols, whether VLPs can effectively introduce a toxic product into the core of cell aggregates from a 3D culture is unknown. To achieve reliable expression of suicide gene, instead of random transgene insertion mediated by a virus, site-specific insertion of a suicide gene by genome editing was applied. Ou et al. attempted to insert the HSV-TK gene into the OCT4 locus in hPSCs by zinc finger nucleases (ZFN) (Ou et al., 2013). However, the genetically modified hPSC clones were not sensitive to GCV and high rate of off-target insertion was also observed in their study. Our results that a selectable marker can interfere with the expression of the adjacent suicide gene and the detection of multiple genomic loci exhibiting similar or identical sequence as the OCT4 locus could provide possible explanations for Qu et al.’s observations. During the submission of the current work, Liang et al. reported a strategy of in-frame insertion of the HSV-TK gene into the cyclin-dependent kinase 1 (CDK1) locus in hESC lines (Liang et al., 2018). Because CDK1 is not expressed in non-dividing cells (Diril et al., 2012, Santamaria et al., 2007), their strategy ensures selective expression of the HSV-TK gene only in dividing cells, which can be eradicated by GCV. Although both the TK and our iC9 system are drug inducible, they use distinc mechanisms to induce cell death. The two systems may therefore complement each other to improve the safety of using stem cell-derived products in cell-based therapies.
In summary, our data demonstrate the feasibility of using an endogenous locus to modulate the expression of the iC9 gene for selective eradication of undifferentiated hPSCs. The three lineages we tested, including hematopoietic cells, neurons, and pancreatic beta-like cells, are potential candidates for clinical applications. AP1903 treatment of these cell lineages did not seem to affect cell viability or lineage commitment. Although residue SOX2+ cells remained in the derived S7 pancreatic beta-like cell population, they were not undifferentiated hPSCs and thus had no potential to form teratomas. This is consistent with the lack of teratoma formation in immunodeficient mice transplanted with the S7 cells from this differentiation protocol (Rezania et al., 2014). The strategy described here should therefore provide a layer of safety control to reduce the risk of teratoma from transplanted hPSC-derived cell products.
Limitations of the Study
In this study, SOX2 was used as the locus for insertion of a suicide gene due to the reduced off-target effect generated by CRISPR-Cas9 when compared with other two stem cell loci, OCT4 and NANOG. However, SOX2 is not only an important transcription regulator for stem cell pluripotency but is also involved in differentiation of neural progenitor cells and patterning of the foregut endoderm. Therefore, although we have demonstrated that this system can be applied in three clinically relevant hPSC-derived cell products, it is not applicable in cell types such as neuronal progenitor cells and anterior foregut cells that also express SOX2 at high levels. As shown in Figure 4E, neural progenitor cells rapidly became apoptotic upon AP1903 treatment. Finding a locus exclusively activated in hPSCs but not in these cells would circumvent this problem. In addition, our system is designed specifically to eradicate undifferentiated stem cells before transplantation to minimize the risk of teratoma formation. However, this strategy may not be sufficient to eliminate already formed tumors derived from the transplanted cell products due to the down-regulation of SOX2. The iC9 system and other TK gene-based strategies may therefore complement each other to improve the safety of stem-cell-derived products in transplantation therapy.
Methods
All methods can be found in the accompanying Transparent Methods supplemental file.
Acknowledgments
This work is supported by a grant from Wanek Innovation Program. Y.L. is support by the Project of International Cooperation and Exchanges of China, China [81520108002]. Research reported in this publication included work performed in the Animal Tumor Models Program, Pathology Solid Tumor Core, Integrative Genomics Core, and Analytical Cytometry Core supported by the National Cancer Institute of the National Institutes of Health, United States under award number P30CA033572. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Contributions
Y.W., F.K., and J.K.Y. conceived and designed experiments; Y.W., T.C., and Y.L. performed experiments; Y.W., F.K., H.H., and J.K.Y. provided data analysis and interpretation; Y.W. and J.K.Y. wrote the manuscript.
Declaration of Interests
The authors indicated no competing interests.
Published: December 20, 2019
Footnotes
Supplemental Information can be found online at https://doi.org/10.1016/j.isci.2019.11.038.
Supplemental Information
References
- Balboa D., Weltner J., Novik Y., Eurola S., Wartiovaara K., Otonkoski T. Generation of an OCT4 reporter human induced pluripotent stem cell line using CRISPR/SpCas9. Stem Cell Res. 2017;23:105–108. doi: 10.1016/j.scr.2017.07.006. [DOI] [PubMed] [Google Scholar]
- Boer B., Kopp J., Mallanna S., Desler M., Chakravarthy H., Wilder P.J., Bernadt C., Rizzino A. Elevating the levels of Sox2 in embryonal carcinoma cells and embryonic stem cells inhibits the expression of Sox2:Oct-3/4 target genes. Nucleic Acids Res. 2007;35:1773–1786. doi: 10.1093/nar/gkm059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bokhoven M., Stephen S.L., Knight S., Gevers E.F., Robinson I.C., Takeuchi Y., Collins M.K. Insertional gene activation by lentiviral and gammaretroviral vectors. J. Virol. 2009;83:283–294. doi: 10.1128/JVI.01865-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonini C., Ferrari G., Verzeletti S., Servida P., Zappone E., Ruggieri L., Ponzoni M., Rossini S., Mavilio F., Traversari C. HSV-TK gene transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science. 1997;276:1719–1724. doi: 10.1126/science.276.5319.1719. [DOI] [PubMed] [Google Scholar]
- Caserta T.M., Smith A.N., Gultice A.D., Reedy M.A., Brown T.L. Q-VD-OPh, a broad spectrum caspase inhibitor with potent antiapoptotic properties. Apoptosis. 2003;8:345–352. doi: 10.1023/a:1024116916932. [DOI] [PubMed] [Google Scholar]
- Chen M., Guerrero A.D., Huang L., Shabier Z., Pan M., Tan T.H., Wang J. Caspase-9-induced mitochondrial disruption through cleavage of anti-apoptotic BCL-2 family members. J. Biol. Chem. 2007;282:33888–33895. doi: 10.1074/jbc.M702969200. [DOI] [PubMed] [Google Scholar]
- Cheng E.H., Kirsch D.G., Clem R.J., Ravi R., Kastan M.B., Bedi A., Ueno K., Hardwick J.M. Conversion of Bcl-2 to a Bax-like death effector by caspases. Science. 1997;278:1966–1968. doi: 10.1126/science.278.5345.1966. [DOI] [PubMed] [Google Scholar]
- Cheng F., Ke Q., Chen F., Cai B., Gao Y., Ye C., Wang D., Zhang L., Lahn B.T., Li W. Protecting against wayward human induced pluripotent stem cells with a suicide gene. Biomaterials. 2012;33:3195–3204. doi: 10.1016/j.biomaterials.2012.01.023. [DOI] [PubMed] [Google Scholar]
- Clackson T., Yang W., Rozamus L.W., Hatada M., Amara J.F., Rollins C.T., Stevenson L.F., Magari S.R., Wood S.A., Courage N.L. Redesigning an FKBP-ligand interface to generate chemical dimerizers with novel specificity. Proc. Natl. Acad. Sci. U S A. 1998;95:10437–10442. doi: 10.1073/pnas.95.18.10437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davenport C., Diekmann U., Budde I., Detering N., Naujok O. Anterior-posterior patterning of definitive endoderm generated from human embryonic stem cells depends on the differential signaling of retinoic acid, Wnt-, and BMP-signaling. Stem Cells. 2016;34:2635–2647. doi: 10.1002/stem.2428. [DOI] [PubMed] [Google Scholar]
- Davis R.P., Costa M., Grandela C., Holland A.M., Hatzistavrou T., Micallef S.J., Li X., Goulburn A.L., Azzola L., Elefanty A.G. A protocol for removal of antibiotic resistance cassettes from human embryonic stem cells genetically modified by homologous recombination or transgenesis. Nat. Protoc. 2008;3:1550–1558. doi: 10.1038/nprot.2008.146. [DOI] [PubMed] [Google Scholar]
- Diril M.K., Ratnacaram C.K., Padmakumar V.C., Du T., Wasser M., Coppola V., Tessarollo L., Kaldis P. Cyclin-dependent kinase 1 (Cdk1) is essential for cell division and suppression of DNA re-replication but not for liver regeneration. Proc. Natl. Acad. Sci. U S A. 2012;109:3826–3831. doi: 10.1073/pnas.1115201109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham V., Khudyakov J., Ellis P., Pevny L. SOX2 functions to maintain neural progenitor identity. Neuron. 2003;39:749–765. doi: 10.1016/s0896-6273(03)00497-5. [DOI] [PubMed] [Google Scholar]
- Green M.D., Chen A., Nostro M.C., d'Souza S.L., Schaniel C., Lemischka I.R., Gouon-Evans V., Keller G., Snoeck H.W. Generation of anterior foregut endoderm from human embryonic and induced pluripotent stem cells. Nat. Biotechnol. 2011;29:267–272. doi: 10.1038/nbt.1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hentze H., Soong P.L., Wang S.T., Phillips B.W., Putti T.C., Dunn N.R. Teratoma formation by human embryonic stem cells: evaluation of essential parameters for future safety studies. Stem Cell Res. 2009;2:198–210. doi: 10.1016/j.scr.2009.02.002. [DOI] [PubMed] [Google Scholar]
- Hockemeyer D., Soldner F., Beard C., Gao Q., Mitalipova M., DeKelver R.C., Katibah G.E., Amora R., Boydston E.A., Zeitler B. Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nat. Biotechnol. 2009;27:851–857. doi: 10.1038/nbt.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klimanskaya I., Hipp J., Rezai K.A., West M., Atala A., Lanza R. Derivation and comparative assessment of retinal pigment epithelium from human embryonic stem cells using transcriptomics. Cloning Stem Cells. 2004;6:217–245. doi: 10.1089/clo.2004.6.217. [DOI] [PubMed] [Google Scholar]
- Kopp J.L., Ormsbee B.D., Desler M., Rizzino A. Small increases in the level of Sox2 trigger the differentiation of mouse embryonic stem cells. Stem Cells. 2008;26:903–911. doi: 10.1634/stemcells.2007-0951. [DOI] [PubMed] [Google Scholar]
- Krentz N.A., Nian C., Lynn F.C. TALEN/CRISPR-mediated eGFP knock-in add-on at the OCT4 locus does not impact differentiation of human embryonic stem cells towards endoderm. PLoS One. 2014;9:e114275. doi: 10.1371/journal.pone.0114275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBlanc L., Lee B.K., Yu A.C., Kim M., Kambhampati A.V., Dupont S.M., Seruggia D., Ryu B.U., Orkin S.H., Kim J. Yap1 safeguards mouse embryonic stem cells from excessive apoptosis during differentiation. Elife. 2018;7:e40167. doi: 10.7554/eLife.40167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee M.O., Moon S.H., Jeong H.C., Yi J.Y., Lee T.H., Shim S.H., Rhee Y.H., Lee S.H., Oh S.J., Lee M.Y. Inhibition of pluripotent stem cell-derived teratoma formation by small molecules. Proc. Natl. Acad. Sci. U S A. 2013;110:E3281–E3290. doi: 10.1073/pnas.1303669110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W., Xiang A.P. Safeguarding clinical translation of pluripotent stem cells with suicide genes. Organogenesis. 2013;9:34–39. doi: 10.4161/org.24317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang Q., Monetti C., Shutova M.V., Neely E.J., Hacibekiroglu S., Yang H., Kim C., Zhang P., Li C., Nagy K. Linking a cell-division gene and a suicide gene to define and improve cell therapy safety. Nature. 2018;563:701–704. doi: 10.1038/s41586-018-0733-7. [DOI] [PubMed] [Google Scholar]
- Menasche P., Vanneaux V., Hagege A., Bel A., Cholley B., Parouchev A., Cacciapuoti I., Al-Daccak R., Benhamouda N., Blons H. Transplantation of human embryonic stem cell-derived cardiovascular progenitors for severe ischemic Left ventricular dysfunction. J. Am. Coll. Cardiol. 2018;71:429–438. doi: 10.1016/j.jacc.2017.11.047. [DOI] [PubMed] [Google Scholar]
- Moolten F.L. Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: paradigm for a prospective cancer control strategy. Cancer Res. 1986;46:5276–5281. [PubMed] [Google Scholar]
- Ng E.S., Davis R.P., Azzola L., Stanley E.G., Elefanty A.G. Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood. 2005;106:1601–1603. doi: 10.1182/blood-2005-03-0987. [DOI] [PubMed] [Google Scholar]
- Ou W., Li P., Reiser J. Targeting of herpes simplex virus 1 thymidine kinase gene sequences into the OCT4 locus of human induced pluripotent stem cells. PLoS One. 2013;8:e81131. doi: 10.1371/journal.pone.0081131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagliuca F.W., Millman J.R., Gurtler M., Segel M., Van Dervort A., Ryu J.H., Peterson Q.P., Greiner D., Melton D.A. Generation of functional human pancreatic beta cells in vitro. Cell. 2014;159:428–439. doi: 10.1016/j.cell.2014.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qadir M.M.F., Alvarez-Cubela S., Belle K., Sapir T., Messaggio F., Johnson K.B., Umland O., Hardin D., Klein D., Perez-Alvarez I. A double fail-safe approach to prevent tumorigenesis and select pancreatic beta cells from human embryonic stem cells. Stem Cell Reports. 2019;12:611–623. doi: 10.1016/j.stemcr.2019.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Que J., Okubo T., Goldenring J.R., Nam K.T., Kurotani R., Morrisey E.E., Taranova O., Pevny L.H., Hogan B.L. Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm. Development. 2007;134:2521–2531. doi: 10.1242/dev.003855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rampoldi A., Crooke S.N., Preininger M.K., Jha R., Maxwell J., Ding L., Spearman P., Finn M.G., Xu C. Targeted elimination of tumorigenic human pluripotent stem cells using suicide-inducing virus-like particles. ACS Chem. Biol. 2018;13:2329–2338. doi: 10.1021/acschembio.8b00490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ran F.A., Hsu P.D., Lin C.Y., Gootenberg J.S., Konermann S., Trevino A.E., Scott D.A., Inoue A., Matoba S., Zhang Y. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell. 2013;154:1380–1389. doi: 10.1016/j.cell.2013.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reardon J.E. Herpes simplex virus type 1 and human DNA polymerase interactions with 2'-deoxyguanosine 5'-triphosphate analogues. Kinetics of incorporation into DNA and induction of inhibition. J. Biol. Chem. 1989;264:19039–19044. [PubMed] [Google Scholar]
- Rezania A., Bruin J.E., Arora P., Rubin A., Batushansky I., Asadi A., O'Dwyer S., Quiskamp N., Mojibian M., Albrecht T. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotechnol. 2014;32:1121–1133. doi: 10.1038/nbt.3033. [DOI] [PubMed] [Google Scholar]
- Santamaria D., Barriere C., Cerqueira A., Hunt S., Tardy C., Newton K., Caceres J.F., Dubus P., Malumbres M., Barbacid M. Cdk1 is sufficient to drive the mammalian cell cycle. Nature. 2007;448:811–815. doi: 10.1038/nature06046. [DOI] [PubMed] [Google Scholar]
- Schuldiner M., Itskovitz-Eldor J., Benvenisty N. Selective ablation of human embryonic stem cells expressing a “suicide” gene. Stem Cells. 2003;21:257–265. doi: 10.1634/stemcells.21-3-257. [DOI] [PubMed] [Google Scholar]
- Schwartz S.D., Regillo C.D., Lam B.L., Eliott D., Rosenfeld P.J., Gregori N.Z., Hubschman J.P., Davis J.L., Heilwell G., Spirn M. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. 2015;385:509–516. doi: 10.1016/S0140-6736(14)61376-3. [DOI] [PubMed] [Google Scholar]
- Song W.K., Park K.M., Kim H.J., Lee J.H., Choi J., Chong S.Y., Shim S.H., Del Priore L.V., Lanza R. Treatment of macular degeneration using embryonic stem cell-derived retinal pigment epithelium: preliminary results in Asian patients. Stem Cell Reports. 2015;4:860–872. doi: 10.1016/j.stemcr.2015.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straathof K.C., Pule M.A., Yotnda P., Dotti G., Vanin E.F., Brenner M.K., Heslop H.E., Spencer D.M., Rooney C.M. An inducible caspase 9 safety switch for T-cell therapy. Blood. 2005;105:4247–4254. doi: 10.1182/blood-2004-11-4564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talkhabi M., Aghdami N., Baharvand H. Human cardiomyocyte generation from pluripotent stem cells: a state-of-art. Life Sci. 2016;145:98–113. doi: 10.1016/j.lfs.2015.12.023. [DOI] [PubMed] [Google Scholar]
- Thomson J.A., Itskovitz-Eldor J., Shapiro S.S., Waknitz M.A., Swiergiel J.J., Marshall V.S., Jones J.M. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- Veres A., Faust A.L., Bushnell H.L., Engquist E.N., Kenty J.H., Harb G., Poh Y.C., Sintov E., Gurtler M., Pagliuca F.W. Charting cellular identity during human in vitro beta-cell differentiation. Nature. 2019;569:368–373. doi: 10.1038/s41586-019-1168-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wichterle H., Lieberam I., Porter J.A., Jessell T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell. 2002;110:385–397. doi: 10.1016/s0092-8674(02)00835-8. [DOI] [PubMed] [Google Scholar]
- Wu C., Hong S.G., Winkler T., Spencer D.M., Jares A., Ichwan B., Nicolae A., Guo V., Larochelle A., Dunbar C.E. Development of an inducible caspase-9 safety switch for pluripotent stem cell-based therapies. Mol. Ther. Methods Clin. Dev. 2014;1:14053. doi: 10.1038/mtm.2014.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagyu S., Hoyos V., Del Bufalo F., Brenner M.K. An inducible caspase-9 suicide gene to improve the safety of therapy using human induced pluripotent stem cells. Mol. Ther. 2015;23:1475–1485. doi: 10.1038/mt.2015.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J., Liu X., Bhalla K., Kim C.N., Ibrado A.M., Cai J., Peng T.I., Jones D.P., Wang X. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science. 1997;275:1129–1132. doi: 10.1126/science.275.5303.1129. [DOI] [PubMed] [Google Scholar]
- Youle R.J., Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 2008;9:47–59. doi: 10.1038/nrm2308. [DOI] [PubMed] [Google Scholar]
- Zarogoulidis P., Darwiche K., Sakkas A., Yarmus L., Huang H., Li Q., Freitag L., Zarogoulidis K., Malecki M. Suicide gene therapy for cancer - current strategies. J. Genet. Syndr. Gene Ther. 2013;4:16849. doi: 10.4172/2157-7412.1000139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Z., Verma N., Gonzalez F., Shi Z.D., Huangfu D. A CRISPR/Cas-Mediated selection-free knockin strategy in human embryonic stem cells. Stem Cell Reports. 2015;4:1103–1111. doi: 10.1016/j.stemcr.2015.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.