Abstract
Background
Hepatic encephalopathy (HE) is a complication of transjugular intrahepatic portosystemic shunt (TIPS).
Aims
Extend the knowledge about the early detection of multiple brain metabolic abnormalities following TIPS; these abnormalities can be detected and managed prior to the clinical manifestation of HE with use of Multiparametric Magnetic Resonance with Spectroscopy.
Methods
12 cirrhotic Patients underwent TIPS; each Patient underwent a 3 T MRI evaluation before and after TIPS. The spectroscopic images were processed measuring the values of the metabolites N-acetylaspartate (NAA) - Glutamine / Glutamate (Glx) - Colina (Cho) - Myinositol (mI) at the level of the nuclei of the base.
Results
Spectroscopic examination performed before the TIPS procedure showed low values of Cho and Mi, instead following the procedure: an increase in the Glx value, a mean reduction in the values of Cho and mI, a statistically significant reduction in the Cho / Creatine ratio, in the mI / Creatine ratio and an increase of the Glx / Creatine ratio.
Conclusions
Our study demonstrated the efficacy of spectroscopy in Patient subjected to TIPS. MR 3 T with spectroscopy can become a valid tool for monitor the dynamics of changes in brain metabolism after TIPS and to provide an early diagnosis of HE allowing an early treatment.
Keywords: Hepatic encephalopathy (HE), Transjugular intrahepatic portosystemic shunt (TIPS), Multiparametric magnetic resonance, Spectroscopy
1. Introduction
Hepatic encephalopathy (HE) includes a broad spectrum of neuropsychiatric manifestations and is one of the most debilitating complications of chronic liver disease that worsens the quality of life and physical condition of the patient (Fig. 1). In spite of more than 100 years of research, the pathogenesis of HE is still not well understood. This reflects the limitation to study the brain of patients with HE in vivo. The most common suggestions include the role of neurotoxins, impaired neurotransmission due to metabolic changes in liver failure, changes in brain energy metabolism, systemic inflammatory response and alterations of the blood brain barrier [1].
Fig. 1.
Spectra obtained with Single-voxel Spin Echo (PRESS) in left globus pallidus in 68 years old woman. Can be osserved the 15 × 15 × 15 mm voxel in axial projection (a), sagittal (b) and coronal (c). Spectra obtained with tow different time of echo (TE), respectively 144 msec (d) e 35 msec (e).
It’s not excluded that many of the described abnormalities can be present at the same time and may ultimately be responsible for the development of HE.
However, is well recognized that ammonia is one of the major factor in its pathogenesis, inducing oxidative/nitrosative stress, mitochondrial abnormalities and astrocyte swelling.
Infact, the liver converts the ammonia to a non toxic compound, the urea. In case of reduced functionality of the liver and in porto-systemic shunt, this process is weak, developing hyperammonemia; this means that ammonia in excess can pass through blood brain barrier. In astrocytes, glutamine synthetase (GS) converts ammonia in excess and glutamate into glutamine.
Glutamine accumulation in astrocytes cause an higher intracellular osmolality, developing brain edema.
Cells exhibit short- and long-term adaptive mechanisms to adjust for changes in osmolarity.In the case of chronic adaptation, reduction of the levels of myoinositol, a key intracellular organic osmolyte, is accomplished slowly over several days. Such osmotic adaptation may be a factor that explains the lower frequency of brain edema in subacute hepatic failure.Consistent with these temporal changes in osmotic adaptation is the finding of an elevated glutamine and a low brain myoinositol in patients with cirrhosis, as seen with brain MRI spectroscopy [2].
Transjugular intrahepatic portosystemic shunt (TIPS) consists in the creation of percutaneous link between the hepatic vein and the intrahepatic segment of the portal vein. The aim of this procedure is to reducing portal venous pressure. To date, the procedure is considered an effective method for treating bleeding of esophageal varices and refractory ascites.
Hepatic encephalopathy is a very common complication in patients after TIPS procedure, 20–31% of the patients who undergo a TIPS procedure would demonstrate new or worsening HE after TIPS [3].
Radiology offers a variety of techniques for the non-invasive diagnosis of structural, functional and metabolic alterations involved in developing of hepatic encephalopathy in Patients after TIPS.
Actually there are few papers in the literature and there aren’t any standardized methods for an early recognition of alterations correlated with hepatic encephalopathy.
The aim of our study was to evaluate the effects of TIPS and the development of post-procedural HE by Multiparametric Magnetic Resonance and Spectroscopy (1H-MRS).
We have studyed the alteration of some brain metabolites pre and post TIPS and their correlation with clinical and morfological data.
2. Materials and methods
2.1. Patients and technique
Between January and October 2016, a total of 12 patients (10 men and 2 women) with a mean age of 592 years (range, 48–68 years), referred from the Diagnostic Department of the University Hospital of Tor Vergata, in collaboration with Hepatology Department, with a diagnosis of portal hypertension who have undergo to TIPS procedure, were enrolled in the study.
6 patients presented alcohol-related cirrhosis, 3 cryptogenic cirrhosis, 1 cirrhosis associated with congenital dilatation of the biliary tract and the remaining 2 autoimmune cirrhoses. The patients examined had a mean value of 7 at Child-Pugh Classification (range 6–8), a mean value of 15,5 at Model for End-Stage Liver Disease (MELD) (range 10–22) end 16,6 (range 9,9–22,4) at MELD-Na.
The demographic, clinical and biohumoral characteristics of the study population are shown in Table 1.
Table 1.
Population study: Demographic characteristics, clinical scores and biochemical values.
| Patients | |
|---|---|
| n | 12 |
| Sex (F/M) | 2/10 |
| Age | 59 ± 6.02 |
| Alcoholic Etiology (Yes/No) | 6/5 |
| MELD | 15.5 ± 3.91 |
| Child Pugh | 7 ± 0.57 |
| MELD-Na | 16.5 ± 4.44 |
| TIPS Indications: Hemorrhage/Ascites | 10/6 |
| Blood Creatinine level(mg/dl) | 2 ± 1.05 |
| Blood Sodium level | 137.8 ± 3.33 |
| Blood Albumin level | 3.43 ± 0.23 |
| Blood Urea level | 74.8 ± 28 |
| INR | 1.29 ± 0.04 |
| HE pre-TIPS | 4/8 |
| HE post-TIPS | 5/7 |
| Time from MRI 1- MRI 2 (gg) | 63 ± 25.5 |
Exclusion criteria:
-Age >75 years old;
-bilirubin levels > 5 mg/dl;
-Creatinina levels > 3 mg/dl;
- severe cardiac or pulmonary dysfunction;
-sepsi or spontaneous bacterial peritonitis.
The study was approved by the Ethics Committee of our institution, and informed consent was obtained from each participant before being introduced in the protocol.
All of the patient selected for the study have been subjected to a clinical assessment before TIPS.
Score for te Child-Pught Classification, Model for End-Stage Liver Disease (MELD) and MELD-Na, were calculated.
Indeed all of the patient made an MRI pre-TIPS.
All of the Tips procedure were performed by a single team of expert radiologist of the Policlinico Tor Vergata in a dedicated angiographic room.
2.2. MRI examination and analysis
The MR imaging studios were performed on 3.0 T magnet (Philips Achieva Intera 3 T; Philips Healthcare, Eindhoven, The Netherlands) equipped with a multichannel coil (8 channel).
Conventional MR imaging of the brain was performed initially, the protocol of the study is showed in Table 2.
Table 2.
MRI acquisition Protocol.
| Sequences | Planes | Parameters |
|---|---|---|
| T1-weighted Inversion Recovery (IR mode) | Axial, sagittal and coronal | repetition time msec/echo time msec/inversion time msec, 2000/10/800; field of view, 230 × 180 mm; matrix, 256 × 156; voxel size, 0.90 × 1.12 mm |
| T1-weighted Turbo Spin Echo | Axial | repetition time msec/echo time msec, 3000/80; field of view, 230 × 207 mm; matrix, 320 × 220; voxel size, 0.72 × 0.90 mm |
| T2 Fluid Attenuated Inversion Recovery (FLAIR) | Axial | repetition time msec/echo time msec/inversion time msec, 11 000/125/2800; field of view, 230 × 180 mm; matrix, 256 × 117; voxel size, 0.9 × 1.2 mm |
| DWI | Axial | Tr (ms) 2700; Te(ms)96; b-values 0, 500,1000; voxel size, 0.9 × 1.2 mm |
Proton MR spectroscopy (H-MRS) acquired with a Single Shot in Echo Planar Imaging sequence were performed (Table 3, Fig. 1)
Table 3.
Proton MR Spectroscopy protocol.
| Sequence | Region of Interest (ROI) | Parameters |
|---|---|---|
| Point-resolved spatially localized spectroscopy (PRESS) | Caudate nucleus | Short TE (35 sec); Long TR (144 sec); chemical shift with three saturation impulses for selective water signal |
The total time of the exame was performed in 45 min.
Then spectrum analysis was performed on a dedicate workstation with the Single Voxel technique (size 15 mm³).
Measurement were performed at the following resonaces: N-acetyl aspartate, glutamine/glutamate, choline and myo-inositol. Results are expressed as metabolic ratios with respect to the creatine + phosphocreatine resonance.
They have been positioned manually 7 ROIs on the FA (fractional Anisotropy) map at the level of: body callosum; right inner capsule; left internal capsule; caudate nucleus; putamen; white frontal substance; occipital white matter. The software then calculated for each ROI the relative FA values with the respective averages, standard deviations for each patient before and after TIPS.
A quantitative analysis was also performed by measuring the Pallidal Index (PI), which is calculated as the signal intensity ratio of GP relative to the frontal white matter (FWM) on T1-WI multiplied by 100.
Data processing was performed by two expert radiologists, independently and without knowing the pre- and post-procedural results. The obtained data have been saved and processed directly on a dedicated console with a specific program.
2.3. Statistical methods
Statistical analysis was conducted with the Graphpad Prism software, version 7.02 and Microsoft Excel.
The results were expressed as mean and deviation standard (SD) for continuous variables of values and percentages for variables categorical. The statistical comparison for the subgroups was made using the Student test (t-test).
Uni- and multivariate linear regression models were used to evaluate the relative prognostic significance of the various variables in predicting the possible onset of hepatic encephalopathy after TIPS.
In addition, contingency tables were created to correlate the MR and Spectroscopy findings, with the occurrence of HE in post-TIPS Patients.
When considered appropriated, the non-parametric sample test was used for independent sample of Mann-Whitney. ROC curves were developed to evaluate the sensitivity and specificity of neurometabolic changes identified with MRI pre and post-TIPS.
A p-value of less than 0.05 was considered significant in all statistical analyzes.
3. Results
Twelve patients were included in the study, 6 of them were affected by alcohol-related hepatic cirrhosis and remaining 6 pts were affected by cirrhosis not related to alcohol abuse (Non Alcohol-Related Hepatic Cirrhosis).
TIPS had been positioned successfully in all 12 patients. Consequently the portal-systemic gradient significantly decreased, with improvement of the underlying conditions (which were the reasons for planning intervention): isolated bleeding from esophageal varicous veins (n = 2 pts), congestive gastropathy (n = 4), refractory ascites (n = 4 pts) or isolated refractory ascites (n = 2 pts).
During follow up at least one episode of acute hepatic encephalopathy occurred in 5/12 patients. One out of these five patients had just one episode, whereas each of the remaining 4 patients experienced at least 5 episodes, these episodes required hospitalization for specific treatments. Two of 5 patients with multiple recurrent encephalopathy episodes were periodically using evacuative clysters in the attempt to reduce the frequency of recurrences.
Multivariate analysis considering age, episodes of acute hepatic encephalopathies prior to TIPS, and Child-Pugh score by using MELD a MELD-Na scales pre-TIPS, and new episodes of acute encephalopathy post-TIPS, showed statistically significant association (p < 0.05) between the latter and episodes acute encephalopathy prior to TIPS, as well as an association between post-TIPS episodes and Child-Pugh score pre-TIPS (Table 4).
Table 4.
Multivariate analysis showing how presence of HE before TIPS and an high Child Pugh score can be considered predictive factors associated with increased incidence of HE after TIP.
| Root-Mean-Square | F | p-Value | |
|---|---|---|---|
| Age | 1,867 | 0,040 | 0,845 |
| HE pre TIPS | 0,952 | 5,556 | 0,040 |
| Child Pugh Score | 1,371 | 5,217 | 0,045 |
| MELD | 53,571 | 4,139 | 0,069 |
| MELD-Na | 16,323 | 0,741 | 0,410 |
MRI was well tolerated by all patients and no procedural complication occurred
The MRI spectroscopy (IH-MRS) showed higher levels of glutamate + glutamine (Glx) in basal ganglia, in relationship to creatinine (Cr) values and lower levels of myoinositol (mI) and choline (Cho) (Fig. 2).
Fig. 2.
56 y.o. man before (A) and after (B) TIPS. Spectroscopy shows decreased level of myoinositol (mI) and choline(Cho) whereas there is an higher level of glutamate-glutamine (Glx).
In particular, the difference in the ratio Glx/Cr post-TIPS from pre-procedural ratio was statistically different, being the latter significantly higher (0.845 ± 0.04 vs 0.635 ± 0.04, p = 0.002) (Fig. 3).
Fig. 3.
Comparison between mean changing of GLX/Cr value pre and post TIPS, also considering the onset of HE. Patients in whom HE arose are the one with colored rectangle and circle.
Then, we evaluated the percentage differences between pre-TIPS levels and last post-TIPS levels, using ROC curves, of Glx/Cr ratio. A delta of at least 37.5% as increase in Glx/Cr ratio resulted to have sensitivity of 83.3% and specificity of 55.5% in predicting the onset of hepatic encephalopathy (Graph 1).
Graph 1.
Variations of Glx/Cr, of Cho/Cr and of mI/Cr value pre and post TIPS analysed with ROC curve, with related sensibility and specificity rate.
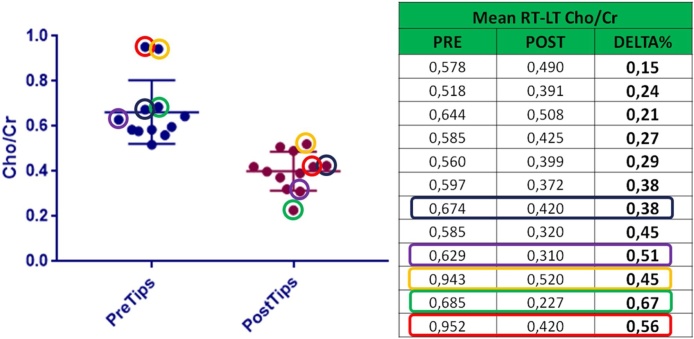
Choline levels decreased significantly from pre-TIPS levels of 0.662 ± 0.04 to 0.400 ± 0.02 post-procedure, as last observed values or include timing post-TIPS (p = 0.0001) (Fig. 4). Evaluating the percentage differences and using ROC curves, a delta of 41% as increase in Cho/Cr ratio between pre-TIPS levels and last post-TIPs values, resulted to have sensitivity of 83% and specificity of 77.7% in predicting the onset of hepatic encephalopathy (Graph 1).
Fig. 4.
Comparison between mean changing of Cho/Cr value pre and post TIPS, also considering the onset of HE. Patients in whom HE arose are the one with colored rectangle and circle.
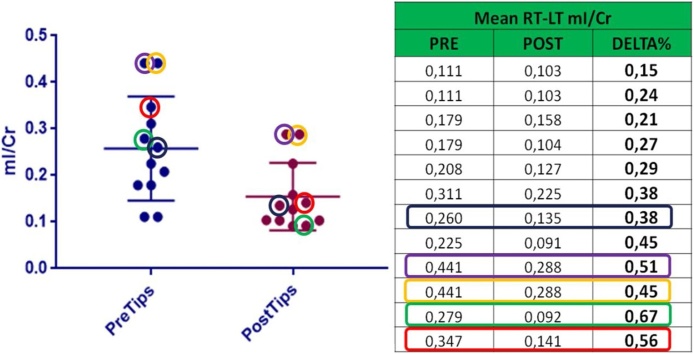
The myoinositol/Cr ratio decreased, due to a significant reduction of mI values from average pre-TIPSS levels of 0.257 ± 0.03 to 0.154 ± 0.02 post-TIPS (p = 0.013) (Fig. 5). The difference in pre-TIPS and final post-TIPS ratio myoinositol/Cr ratio with a delta ≥ 45%, the changes were able to predict the onset of hepatic encephalopathy (Graph 1) with a sensitivity of 66.6% and specificity of 77.7%.
Fig. 5.
Comparison between mean changing of mI/Cr value pre and post TIPS, also considering the onset of HE. Patients in whom HE arose are the one with colored rectangle and circle.
Statistical analysis of FA values did not show any significant difference in pre-TIPS vs post-TIPS values in encephalic regions, when patients with presence vs absence of hepatic encephalopathy were compared (Graph 2).
Graph 2.
Variations of FA value in different analyzed brain areas, before and after TIPS (P-value < 0.01).
Moreover, in patients who suffered from hepatic encephalopathy after TIPS implant procedure, a significantly greater bilateral decrease of myoinositol was observed when compared to patients without post-TIPS HE episodes (p < 0,01).
In T1- weighted images, bilateral hyperintense signals of globus pallidus were reported in 4/12 patients (Fig. 6) The Pallidal Index (PI) was 131.20 ± 12.24 (mean ± SD) at right side and 130.01 ± 10.68 (mean ± SD) at left side.
Fig. 6.
61 y.o. Man, bilateral hyperintensity can be seen in both globus pallidus, T-1 weighted -axial (A) and coronal (B).
After 63 ± ±25.5 days following the TIPS procedure, the mean PI value decreased to 129.50 ± 16.11 right-side and to 126.62 ± 19.25 left-side. The decrease did not reach statistical significance at Student t-test.
4. Discussion
Hepatic encephalopathy (HE) is a common complication of liver dysfunction, including acute liver failure and liver cirrhosis. HE presents as a spectrum of neuropsychiatric symptoms ranging from subtle fluctuating cognitive impairment to coma. It is a significant contributor of morbidity in Patients with liver disease. HE is observed in acute liver failure, liver bypass procedures, including trans-jugular intrahepatic portosystemic shunt, and cirrhosis.
The TIPS is usually efficacious in resolving bleeding from esophageal varices, when these are non-eligible for endoscopic treatment, refractory ascites. On the other hand, hepatic encephalopathy may occurred as post-procedural complications [4].
The possibility to predict onset of HE post-TIPS could be of great importance in order to start an early appropriate treatment or event preventive measures, if possible [5].
Pre-existing HE is considered a strong and independent predictive factor for post-TIPS HE [6]. Previous studies reported higher incidence in post-procedural HE in pts with pre-existing HE condition (about 25%) vs a frequency of less than 10% in patients free from HE prior to TIPS [7]. Our experience is in line with the literature data; we observed a correlation between CH-P score and post-TIPS HE incidence. We have not observed any relationship with other factors including MELD and MELD-Na scores, patients’ age and alcohol-related cirrhosis, though these factors were found to be associated with HE risk by other authors (Rigio et al, 2008, [8].
MRI T1- weighted images are routinely used in clinical practice for diagnosis of disorder in central nervous system [9]. Available literature describes hyperintense bilateral signal in basal nuclei, in particular in globus pallidus in 70–100% of patients with hepatic cirrhosis in T1- weighted images [10].
The intensity of the signal is progressively increasing with worsening of liver function and an increase was observed after TIPS placement. Nevertheless, it has been shown that the signal may decrease after improvement or normalization of liver function, e.g. after liver transplant or subsequently to portosystemic shunt occlusion. The increase in signal intensity following TIPS can be explained by manganese deposit, which is secondary to hepatocellular insufficiency and porto-systemic shunt [11,12].
In our study, the mean value of the pallidal index (PI) in our overall population of 12 pts resulted increased; a similar finding was reported by Krieger et al. [13]. However, in line with literature data, we have not found any correlation between the onset of HE and increase in intensity in globus pallidus.
MR spectroscopy is an useful non-invasive tool for assessment of brain metabolism. Abnormalities in spectroscopy were described in patients with liver cirrhosis regardless of presence of encephalopathy. Reduced ratio choline/creatinine (Cho/Cr) and mI/Cr have together with an increase in glutamate + glutamine/creatinine (Glx/Cr) ratio have been reported by using protonic spectroscopy (1H-MRS) in patients with cirrhosis [14]. Increase in Glx signal may be explained by increase in brain concentration due to ammoniac metabolism abnormalities, other neurotoxins and increase in oxidative stress in astrocytes.
Changes in choline and myoinositol levels could be part of a response to intracellular osmolality caused by glutamine accumulation in astrocytes [15]:
TIPS placement has been shown to worsen these changes which can be revealed at MRS [16]. Haussinger et al. [17] have described a case of a Patient with alcohol-induced liver cirrhosis, who had a normal Glx/mI ratio, and showed changes of that ratio, with an increased level of Glx and a lower level of mI, as early as one week after TIPS placement [17].
The authors have hypothesized that myoinositol release can be subsequent to cellular’s volume increase.The increase of Cellular volume can be related to an Intracellular accumulation of Glx, subsequent to an higher ammoniac circulating level. It has been hypothesized that increase in Glx signal may be compensatory to mI loss. [18]
The abnormalities revealed at spectroscopy seem to correlated with the severity of hepatic encephalopathy in cirrhotic patients undergoing TIPS. Interestingly, there is an evidence that TIPS occlusion has been followed by a rapid and complete resolution of clinical symptoms of encephalopathy and return/recovery of mI signal [14]. Tarasow et al have reported that metabolic abnormalities, which can be detected by spectroscopy, preceded clinical signs-symptoms of encephalopathy [19].
Our observations are in line with the literature data and Tarasow’s observation.
We have observed reduction of averaged Cho and mI levels, while Glx significantly increased after TIPSS. Patients with HE had also reduced Cho/Cr and mI/Cr ratios and increase in Glx levels, the latter being significantly associated with the onset of post-TIPS encephalopathy.
Kreis et al. performed H-1MRS of the brain in 11 patients with chronic hepatic encephalopathy (CHE) and the results were compared with those of patients with liver disease but without CHE; they found specific changes in the brain of CHE patients: elevation in cerebral glutamine levels (P less than or equal to .0001), a 23% reduction in choline metabolite levels (P less than or equal to .0001), and a more than 50% reduction in cerebral myo-inositol levels (P less than or equal to .0001). In four of the 15 patients with liver disease but without clinical CHE, they found a significant reduction in the myo-inositol level, and in two of these patients an elevation in the glutamine concentration was also observed [20].
These findings indicate a role for image-guided H-1 MR spectroscopy in the diagnosis and monitoring of both overt and preclinical-subclinical CHE.
5. Conclusions
Our study confirmed an important role of 1H-MRS in the assessment of hepatic encephalopathy in patients undergoing TIPS due to cirrhosis.
Data generated by our study extend the knowledge and the evidence of multiple brain metabolic abnormalities following TIPS; these abnormalities can be detected prior to the clinical manifestation of HE. However, our observations have to be confirmed by larger studies with longer follow up of patients post-TIPS. 1H-MRS can potentially become a valid tool for early detection of brain metabolic abnormalities and monitoring of their evolution. Further studies are needed d to understand if 1H-MSR can predict the risk of the onset of HE with a sufficient specificity and sensitivity.
Declaration of Competing Interest
We have no conflicts of interest to disclose.
References
- 1.Ferenci P. Hepatic encephalopathy. Gastroenterol. Rep. 2017;5(May 2):138–147. doi: 10.1093/gastro/gox013. Published: 18 April 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Javier Vaquero M.D., Chuhan Chung M.D., Michael E., Cahill B.A., Blei Andres T. Pathogenesis of hepatic encephalopathy in acute liver failure. MD Semin Liver Dis. 2003;23(3) doi: 10.1055/s-2003-42644. [DOI] [PubMed] [Google Scholar]
- 3.Bai M., Qi X., Yang Z., Yin Z., Nie Y., Yuan S., Wu K., Han G., Fan D. Predictors of hepatic encephalopathy after transjugular intrahepatic portosystemic shunt in cirrhotic patients: a systematic review. J. Gastroenterol. Hepatol. 2011;26(6):943–951. doi: 10.1111/j.1440-1746.2011.06663.x. [DOI] [PubMed] [Google Scholar]
- 4.Colombato L. The role of transjugular intrahepatic portosystemic shunt (TIPS) in the management of portal hypertension. J. Clin. Gastroenterol. 2007;41(3):S344–51. doi: 10.1097/MCG.0b013e318157e500. no. December. [DOI] [PubMed] [Google Scholar]
- 5.Hassoun Z., Deschênes M., Lafortune M., Dufresne M.P., Perreault P., Lepanto L., Gianfelice D., Bui B., Pomier-Layrargues G. Relationship between pre-TIPS liver perfusion by the portal vein and the incidence of post-TIPS chronic hepatic encephalopathy. Am. J. Gastroenterol. 2001;96(4):1205–1209. doi: 10.1111/j.1572-0241.2001.03704.x. [DOI] [PubMed] [Google Scholar]
- 6.Masson S., Mardini H.A., Rose J.D., Record C.O. Hepatic encephalopathy after transjugular intrahepatic portosystemic shunt insertion: a decade of experience. QJM. 2008;101(June 6):493–501. doi: 10.1093/qjmed/hcn037. [DOI] [PubMed] [Google Scholar]
- 7.Freedman A.M., Sanyal A.J., Tisnado J., Cole P.E., Shiffman M.L., Luketic V.A., Purdum P.P., Darcy M.D., Posner M.P. Complications of transjugular intrahepatic portosystemic shunt: a comprehensive review. Radiographics. 1993;13(November 6):1185–1210. doi: 10.1148/radiographics.13.6.8290720. [DOI] [PubMed] [Google Scholar]
- 8.Riggio O., Angeloni S., Salvatori F.M., De Santis A., Cerini F., Farcomeni A., Attili A.F., Merli M. Incidence, natural history, and risk factors of hepatic encephalopathyafter transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene- covered stent grafts. Am. J. Gastroenterol. 2008;103(11):2738–2746. doi: 10.1111/j.1572-0241.2008.02102.x. [DOI] [PubMed] [Google Scholar]
- 9.Fukuzawa T., Matsutani S., Maruyama H., Akiike T., Saisho H., Hattori T. Magnetic resonance images of the globus pallidus in patients with idiopathic portal hypertension: a quantitative analysis of the relationship between signal intensity and the grade of portosystemic shunt. J. Gastroenterol. Hepatol. 2006;(May) doi: 10.1111/j.1440-1746.2006.04226.x. [DOI] [PubMed] [Google Scholar]
- 10.Das K., Singh P., Chawla Y., Duseja A., Dhiman R.K., Suri S. Magnetic resonance imaging of brain in patients with cirrhotic and non-cirrhotic portal hypertension. Dig. Dis. Sci. 2008;53(10):2793–2798. doi: 10.1007/s10620-008-0383-y. [DOI] [PubMed] [Google Scholar]
- 11.Rovira A.1, Alonso J., Córdoba J. MR imaging findings in hepatic encephalopathy. AJNR Am. J. Neuroradiol. 2008;29(October 9):1612–1621. doi: 10.3174/ajnr.A1139. Epub 2008 Jun 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Inoue E., Hori S., Narumi Y., Fujita M., Kuriyama K., Kadota T., Kuroda C. Portal- systemic encephalopathy: presence of basal ganglia lesions with high signal intensity on MR images. Radiology. 1991;179(0033–8419):551–555. doi: 10.1148/radiology.179.2.2014310. SB–AIM SB–IM. [DOI] [PubMed] [Google Scholar]
- 13.Krieger S., Jauss M., Jansen O., Stiehl a, Sauer P., Geissler M., Theilmann L., Krieger D. MRI findings in chronic hepatic encephalopathy depend on portosystemic shunt: results of a controlled prospective clinical investigation. J. Hepatol. 1997;27:121–126. doi: 10.1016/s0168-8278(97)80290-5. [DOI] [PubMed] [Google Scholar]
- 14.Miese F., Kircheis G., Wittsack H.J., Wenserski F., Hemker J., Mödder U., Häussinger D., Cohnen M. 1H-MR spectroscopy, magnetization transfer, and diffusion-weighted imaging in alcoholic and nonalcoholic patients with cirrhosis with hepatic encephalopathy. Am. J. Neuroradiol. 2006;27(5):1019–1026. [PMC free article] [PubMed] [Google Scholar]
- 15.Butterworth R.F. Hepatic encephalopathy. Alcohol Res. Health. 2003;27(3):240–246. [PMC free article] [PubMed] [Google Scholar]
- 16.Häussinger D., Laubenberger J., Vom Dahl S., Ernst T., Bayer S., Langer M., Gerok W., Hennig J. Proton magnetic resonance spectroscopy studies on human brain Myo-inositol in hypo-osmolarity and hepatic encephalopathy. Gastroenterology. 1994;107(5):1475–1480. doi: 10.1016/0016-5085(94)90552-5. [DOI] [PubMed] [Google Scholar]
- 17.Häussinger D., Kircheis G., Fischer R., Schliess F., vom Dahl S. Hepatic encephalopathy in chronic liver disease: a clinical manifestation of astrocyte swelling 97 and low-grade cerebral edema? J. Hepatol. 2000;32(June 6):1035–1038. doi: 10.1016/s0168-8278(00)80110-5. [DOI] [PubMed] [Google Scholar]
- 18.Butterworth R.F. Pathogenesis of hepatic encephalopathy: new insights from neuroimaging and molecular studies. J. Hepatol. 2003;39(August 2):278–285. doi: 10.1016/s0168-8278(03)00267-8. [DOI] [PubMed] [Google Scholar]
- 19.Tarasow E., Panasiuk A., Siergiejczyk L., Orzechowska-Bobkiewicz A., Lewszuk A., Walecki J., Prokopowicz D. MR and 1H MR spectroscopy of the brain in patients with liver cirrhosis and early stages of hepatic encephalopathy. Hepatogastroenterology. 2003;50(54):2149–2153. [PubMed] [Google Scholar]
- 20.Kreis R., Ross B.D., Farrow N.A., Ackerman Z. Metabolic disorders of the brain in chronic hepatic encephalopathy detected with H-1 MR spectroscopy. Radiology. 1992 doi: 10.1148/radiology.182.1.1345760. Published Online: Jan 1. [DOI] [PubMed] [Google Scholar]