Abstract
Atherosclerosis is characterized by a persistent, low-grade inflammation of the arterial wall. Monocytes and monocyte-derived macrophages play a pivotal role in the various stages of atherosclerosis. In the past few years, metabolic reprogramming has been identified as an important controller of myeloid cell activation status. In addition, metabolic and epigenetic reprogramming are key regulatory mechanisms of trained immunity, which denotes the non-specific innate immune memory that can develop after brief stimulation of monocytes with microbial or non-microbial stimuli. In this review, we build the case that metabolic reprogramming of monocytes and macrophages, and trained immunity in particular, contribute to the pathophysiology of atherosclerosis. We discuss the specific metabolic adaptations, including changes in glycolysis, oxidative phosphorylation, and cholesterol metabolism, that have been reported in atherogenic milieus in vitro and in vivo. In addition, we will focus on the role of these metabolic pathways in the development of trained immunity.
Keywords: Immunometabolism, Monocyte, Macrophage, Atherosclerosis, Trained immunity
Graphical Abstract
Graphical Abstract.
1. Background
Due to the growing and ageing world population, chronic diseases such as cardiovascular diseases (CVD) have become the most prevalent burden of disease. CVD, including myocardial infarction and stroke, are the leading cause of death worldwide.1 The vast majority of cardiovascular events is caused by rupture of atherosclerotic plaques in the arterial vessel wall and the subsequent formation of an occluding thrombus.
Monocytes and monocyte-derived macrophages play a pivotal role in the various stages of atherosclerosis.2,3 Atherosclerotic plaque formation is initiated by the recruitment of circulating monocytes, which are derived from myeloid progenitors in the bone marrow and a reservoir-population in the spleen, into the intimal layer of the vessel wall. Here, monocytes differentiate into macrophages that can develop into lipid-laden foam cells, which eventually contribute to the growth of the necrotic core. In addition, damage-associated molecular patterns can further activate plaque macrophages by binding to membrane-bound pattern recognition receptors (PRR), such as toll-like receptors (TLR) and scavenger receptors.2 Therefore, the process of atherosclerosis can be augmented in situations of monocytosis or in the presence of circulating monocytes with a specific pro-inflammatory phenotype.4,5
Recently, two novel mechanisms have been revealed that control the inflammatory and atherogenic function of myeloid cells. First, various intracellular metabolic pathways of monocytes and macrophages impact on the functional state of these cells.6 In addition, we and others reported that brief stimulation of myeloid cells can result in a long-term pro-inflammatory and pro-atherogenic phenotype, which is denoted trained immunity. Metabolic and epigenetic rewiring have been identified as important mechanisms driving trained immunity.7 In this review, we will focus on how changes in the metabolism of myeloid cells can influence the pathophysiology of atherosclerosis and how metabolic reprogramming drives trained immunity.
2. A short introduction to innate immune memory in atherosclerosis
In contrast to the traditional immunological paradigm, cells of the innate immune system such as monocytes and NK cells can build an immunological memory after previous encounters with micro-organisms.8–10 This is reflected by a persistent suppression (called immunotolerance) or a long-term augmentation of immune effector mechanisms, a phenomenon called ‘trained immunity’. Brief stimulation of monocytes with microbial products, such as the cell wall component of Candida albicans, β-glucan, or Bacille Calmette Guérin (BCG), induces epigenetic reprogramming at the level of chromatin architecture, which is associated with an increased production of cytokines and chemokines in response to a (similar or unrelated) secondary stimulus. It is important to realize that trained immunity is not only triggered by infectious stimuli such as β-glucan and BCG, but also by endogenous non-microbial atherogenic stimuli, such as oxidized LDL (oxLDL) and lipoprotein(a) [Lp(a)].11,12 A complete overview of all triggers of trained immunity has recently been provided by van der Heijden et al.13 Brief stimulation of isolated human monocytes with oxLDL and Lp(a) induces a macrophage phenotype that, upon restimulation with TLR2 and TLR4 ligands, responds with an augmented production of pro-atherogenic factors, such as the cytokines tumour necrosis factor-α (TNF-α) and interleukin (IL)-6.11,12 Moreover, oxLDL trained cells show increased foam cell formation capacity by up-regulating scavenger receptors CD36 and SR-A, as well as reducing the expression of cholesterol efflux transporters adenosine triphosphate (ATP)-binding cassette transporter-A1 and G1 (ABCA1 and ABCG1), processes that have previously been shown to accelerate atherosclerosis.14–16 Finally, trained macrophages produce more collagenases, such as matrix metalloproteinases 2 and 9 (MMP-2 and MMP-9), important in plaque destabilization.11 It is tempting to speculate that trained immunity could be a general underlying mechanism for other pro-atherogenic compounds, such as triglyceride-rich lipoproteins (TRLs), free fatty acids, uric acid, and advanced glycosylation end-products, that all elicit an inflammatory response in monocytes via PRR stimulation.17–20
Although trained immunity appears to have a protective effect against re-infections,21,22 it could play a detrimental role in situations in which myeloid cells themselves contribute to disease progression, e.g. in atherosclerosis. In that regard, trained immunity is likely to be an important mechanism linking infections and atherosclerosis.23
The relevance of trained immunity in the development of atherosclerosis and other inflammatory vascular wall disorders in humans is illustrated in several recent observational studies. In patients with established coronary artery disease, circulating monocytes show an enhanced cytokine production capacity, which was associated with changes in their epigenetic and metabolic signatures.24 Additionally, trained immunity characteristics are associated with the progression of cerebral small vessel disease, yet another prevalent vascular disorder that is associated with low-grade inflammation and shares risk factors with atherosclerosis.25
3. The mechanistic training tools in trained immunity
In order to promote trained immunity to boost resistance to infections in vulnerable patients or to dampen trained immunity in an attempt to prevent or reduce atherosclerosis, it is of crucial importance to thoroughly understand its underlying mechanism. In the past few years, we and others have tried to elucidate the intracellular mechanism that drives the memory function of trained immunity in mature circulating monocytes. To date, two key mechanisms have been identified: epigenetic and metabolic reprogramming. In this section, we will further delineate the involvement and interdependence of these mechanisms in trained immunity.
3.1 Epigenetic reprogramming
Epigenetic reprogramming at the level of the chromatin architecture, mainly occurring via histone modifications, is a central mechanism that underpins the enhanced functional state of trained innate immune cells (an in-depth review on epigenetics and trained immunity can be found elsewhere26). Epigenetic regulation is defined by the regulation of gene expression without an alteration in the DNA sequence itself. This can be due to DNA methylation, histone modifications, or post-translational modulation by non-coding RNAs. These methylation and acetylation marks can regulate the accessibility of the DNA for the transcriptional machinery. DNA hypermethylation is typically associated with gene silencing.27 Modifications of histones, however, can either lead to activation or repression of gene transcription. Histone acetylation neutralizes the positive charge of the lysine residues, which will stimulate the binding of transcription factors and, as such, stimulates activation of gene transcription.28 The effect of histone methylation on gene transcription is dependent on the specific lysine residue that is involved and the amount of methyl groups added. For example, trimethylation of lysine 4 at histone 3 (H3K4me3) is associated with an open chromatin that allows gene transcription, whereas H3K27me3 compact the chromatin, leading to gene silencing.29 Interestingly, individual histone modifications can influence each other and can also interact with DNA methylation marks, through modulation of the activity of protein complexes that bind these histone/DNA modifications.30
Analyses of several activating histone methylation and acetylation marks in the in vitro model for trained immunity showed distinct epigenetic characteristics for either naïve, tolerant, or β-glucan-trained macrophages.26 Notably, inhibition of epigenetic reprogramming with the non-specific histone methyltransferase inhibitor 5′-Methylthioadenosine completely prevented training.31 Regarding the role of DNA methylation in trained immunity, Novakovic et al.32 recently reported that DNA methylation was not significantly changed in β-glucan-trained macrophages.
In the context of atherosclerosis, in vitro training with oxLDL corresponds with enrichment of H3K4me3 at the promoters of at least TNFα, IL-6, MCP-1, IL-8, CD36, SR-A, MMP-2, and MMP-9.11 Moreover, in patients with established atherosclerosis, circulating monocytes demonstrate increased cytokine production capacity, which was associated with a lower presence of repressive histone modifications on the cytokine promoters.24 The differences in the specific histone modifications might be caused by the training stimulus, which is oxLDL in the in vitro experiments, but can also be yet another atherogenic stimulus in these patients in vivo. Additional studies using an unbiased whole genome approach to determine epigenetic reprogramming are clearly warranted in this context.
The importance of epigenetic regulation in atherosclerosis was also highlighted in a recent study by Fuster et al.,33 which investigated the role of clonal haematopoiesis induced by tet methylcytosine dioxygenase 2 (TET2) deficiency, a multifaceted transcriptional regulator, in Ldlr−/− mice. TET2 deficiency in macrophages promoted inflammation and aggravated atherosclerosis, which was mediated by increased production of IL-1β. Of note, Chromatin Immunoprecipitation (ChIP)-qPCR analysis revealed increased histone H3 acetylation at the Il1b gene promoter in TET2-deficient macrophages.33 With epigenetic reprogramming at the centre of trained immunity, it would be of great interest to explore whether clonal haematopoiesis could influence the induction of trained immunity.
3.2 Metabolic reprogramming
3.2.1 Differential routing of metabolic responses
Metabolic pathways play a major role in immune cell function. The glycolytic pathway, TCA cycle, pentose phosphate pathway (PPP), fatty acid oxidation (FAO), fatty acid synthesis (FAS) and amino acid metabolism each have a unique purpose in the cell. However, at the same time, they are closely intertwined, as a consequence of shared fuel inputs and the reliance on the products of other pathways to serve as precursors. For a detailed description of the function of the various metabolic pathways in immune cells, we refer to some excellent recent reviews.6,34 Immune cells distinctly use these metabolic pathways to fine tune their function and to produce energy and building blocks for cell maintenance and proliferation, and modulation of cellular signalling.6 As such, innate immune cells can mount distinct metabolic responses to different inflammatory stimuli. The classical dichotomy for macrophage activation is an illustrative example of this phenomenon. Stimulation with lipopolysaccharide (LPS) and interferon-γ (IFNγ) induces a profound metabolic rewiring of macrophages which is characterized by increased aerobic glycolysis and impaired OXPHOS via the TCA cycle, leading to an accumulation of intermediates such as succinate.35 This switch from OXPHOS to glycolysis closely resembles the Warburg effect in tumour cells, in which pyruvate produced by the glycolytic pathway is metabolized to lactate, while TCA cycle intermediates accumulate, resulting in a decreased oxidative phosphorylation.36 Other up-regulated metabolic pathways in LPS/IFNγ macrophages are the PPP and FAS. Activation via these metabolic routes prepares the macrophage for host defence against microbial pathogens. On the other hand, when macrophages are activated via IL-4, they exhibit increased FAO and OXPHOS and become instrumental in immune-regulation and wound healing.
Over the past few years, the classical dichotomy between M1 and M2 macrophages has been challenged and more metabolic blueprints for macrophage activation have been unravelled. Lachmandas et al. have recently studied the metabolic responses of human CD14+ monocytes to stimulation with several different ligands, including TLR ligands such as Pam3cys (TRL2), poly(I:C) (TLR3) and with whole pathogen lysates from Escherichia coli, Staphylococcus aureus, or Mycobacterium tuberculosis. Upon these stimulations, monocytes exhibited both increased glycolysis and oxidative phosphorylation.37 Also, IL-1β production of these cells in response to both LPS and Pam3cys (P3C) was restricted when glycolysis was blocked with 2-deoxyglucose (2-DG). Blocking the OXPHOS with rotenone, however, restricted IL-1β production only in P3C-stimulated cells. This and other studies35,38 have highlighted the complexity of metabolic rewiring in human innate immune cells and demonstrate that there is no universal response to pathogenic stimuli, but that different stimuli can induce divergent macrophage responses through the differential activation of metabolic routes.
3.2.2 Metabolic reprogramming in trained immunity
Two observations point towards an important role for the intracellular metabolism in the induction of trained immunity. First, pathway analysis of the top 500 up-regulated genes in β-glucan-trained monocytes displayed a marked activation of central metabolic pathways.26 Cheng et al.7 showed that trained immunity is mediated by increased aerobic glycolysis and lactate production via the activation of mammalian target of rapamycin (mTOR) and hypoxia-inducible factor-1α (HIF-1α). A close interaction between metabolism and epigenetics was subsequently illustrated by the observation that there was an enrichment of the activating histone modifications H3K4me3 and H3K27ac on the promoters of several key glycolytic enzymes in β-glucan-trained monocytes, 1 week after stimulation.7 Furthermore, it is increasingly appreciated that various intermediate metabolites serve as co-factors or substrates for epigenetic enzymes, which is reviewed in more detail by Keating and El-Osta.39 Genetic evidence for the impact of cellular metabolism on epigenetic remodelling in the myeloid lineage has previously been described.40,41 For instance, mutation of isocitrate dehydrogenase 1/2 (IDH1/2) enables this enzyme to convert α-ketoglutarate to 2-hydroxyglutarate in the citric acid cycle, which competitively acts on epigenetic modulators that normally depend on α-ketoglutarate as a substrate.40
In the context of trained immunity, the glycolytic switch itself was also shown to impact on the epigenetic remodelling through NAD+/NADH dependent effects on the sirtuin-1 class of histone deacetylase enzymes in β-glucan-trained cells.7 Moreover, an additional series of experiments illustrated that monocytes trained with β-glucan exhibit enhanced glutaminolysis, with a marked accumulation of fumarate and succinate, which was crucial for the induction of a trained macrophage phenotype.42 Notably, stimulation of monocytes with an excess of fumarate resulted in enhanced TNFα and IL-6 production 6 days later, which correlated with increased H3K4me3 at the promoters of their genes. It was demonstrated that fumarate directly inhibits histone demethylase KDM5, which correlated with increased training. Providing the macrophages with α-ketoglutarate, a co-factor for KDM5 activity, increased KDM5 biological activity and suppressed fumarate-induced training. Collectively, these experiments illustrate that various intermediate metabolites can function as substrates or co-factors for epigenetic enzymes.
In the following sections, we will describe the various metabolic pathways that can drive myeloid cell activation in the context of atherosclerosis. In particular, we will highlight the role of these pathways and the interaction with epigenetic reprogramming in trained immunity.
4. Immunometabolism in atherosclerosis and trained immunity
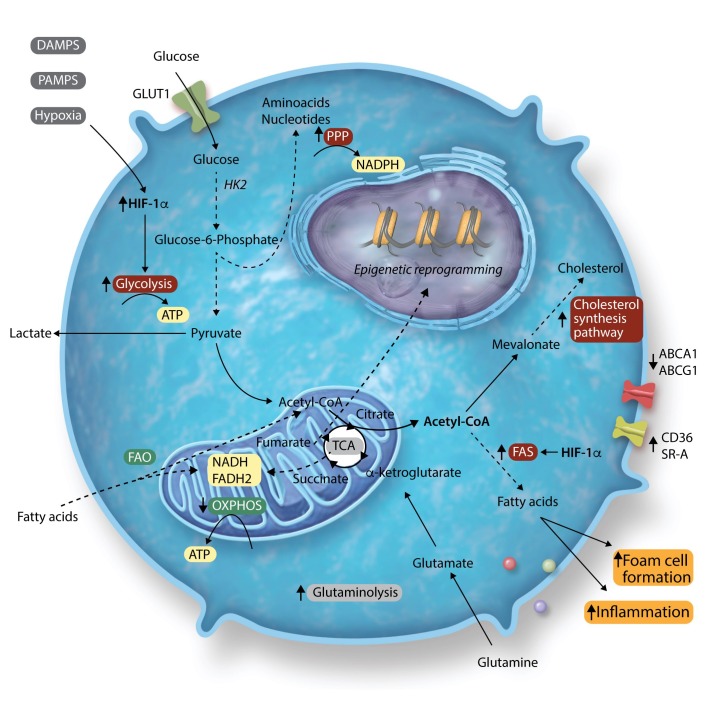
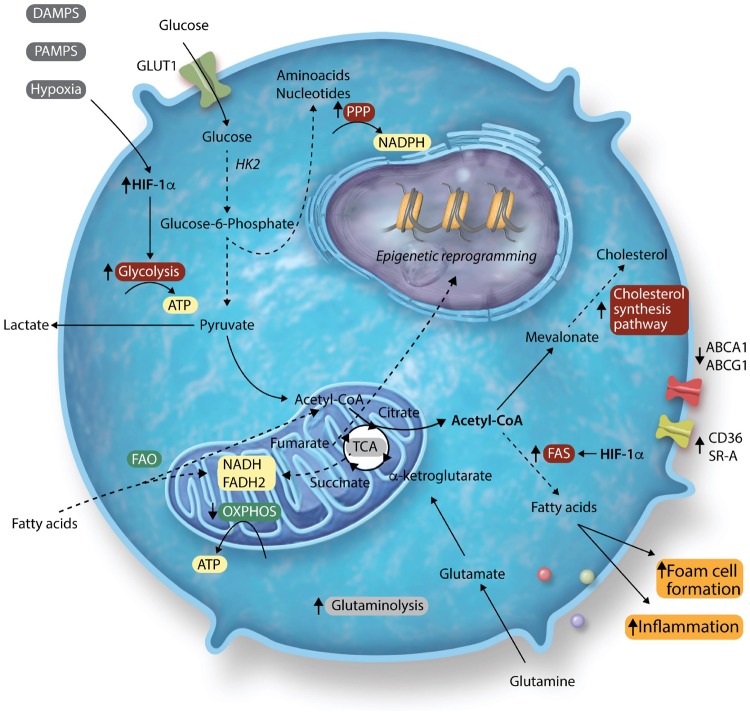
In the context of atherosclerosis, monocytes are exposed to a great variety of signals. Not only within the microenvironment of the atherosclerotic plaque itself, but also in the bone marrow niche and in the circulation. Over the past few years, many studies have sought to identify the distinct metabolic pathways induced by atherogenic stimuli, such as lipoproteins, glucose, and hypoxia. Here, we will discuss the preclinical and clinical data on the key metabolic pathways in myeloid cells that are modulated in the context of atherosclerosis and are important for trained immunity, including glycolysis, the PPP, and cholesterol metabolism (Figure 1).
Figure 1.
Overview of metabolic pathways in atherosclerosis and trained immunity. The arrows next to the pathway names indicate whether the pathways are up- or down-regulated in trained immunity. The colours indicate whether or not they affect atherosclerosis development: red indicates pro-atherogenic and green indicates anti-atherogenic. Grey indicates no clear impact on atherosclerosis or unknown impact.
4.1 Glycolysis is up-regulated in atherosclerosis
In monocytes and macrophages, glucose transporter 1 (GLUT-1) on the cell’s outer cell membrane initiates the uptake of glucose. Within the cell, glucose is phosphorylated by hexokinase to glucose-6-phosphate which is subsequently used in either the glycolysis, the PPP, or during FAS. During glycolysis, processing of glucose in the cytosol yields two ATPs as well as pyruvate. The pyruvate generated during glycolysis is either converted to lactate by lactate dehydrogenase or enters the TCA cycle in the mitochondria. Below, we will discuss the involvement of glycolysis in the various stages of atherosclerotic disease.
Atherosclerotic plaques are characterized by local hypoxic regions, where oxygen availability is limited. Under hypoxic conditions, HIF-1 is stabilized and activates the glycolytic pathway, thereby reducing the cell’s dependency on OXPHOS. The increased glycolytic flux occurs through increased expression of key glycolytic proteins GLUT1, hexokinase II (HK2), and 6-phosphofructo-2kinase/fructose-2, 6-biphosphatase 3 (PFKFB3).43 Genetic deletion of HIF-1α in myeloid cells from Ldlr−/− mice reduced atherosclerotic plaque formation, which was likely mediated by a reduction in glycolytic rate.44 By using 18F radiolabelled glucose analogue fluordeoxyglucose positron emission tomography CT (18F-FDG-PET/CT), several studies have assessed the involvement of glycolysis within human atherosclerotic plaques. Increased glucose uptake was found to co-localize with HIF-1α expression.45 Moreover, increased FDG uptake was associated with macrophage containing- and lipid-rich necrotic plaque areas.46,47 Tomas et al.48 performed metabolomics on human carotid atherosclerotic plaques, and revealed two distinct metabolic profiles associated with plaque vulnerability and inflammation. Vulnerable plaques were characterized by increased glycolysis, defective FAO, and increased anaplerosis of the TCA cycle. These plaques also had increased mRNA expression of glycolytic and PPP enzymes, next to increased protein levels of IL-1β, IL-6, and IL-18, but not TNFα, which resembles the cytokine production profile in macrophages of patients with atherosclerosis.49
Directing our scope from the plaque to the bone marrow, support for a role of myeloid cell glycolysis in myelopoiesis and atherogenesis is found in the observation that in hypercholesterolaemic apoE−/− mice, leucocytes and HSPCs exhibit an increased GLUT1-dependent glucose uptake, which is associated with increased mitochondrial potential. This implies that the influx of glycolytic metabolites in these cells fuels the mitochondria for oxidative phosphorylation and ATP generation.50 Transplantation of bone marrow derived from GLUT1−/+ mice into apoE−/− mice reversed HSPC proliferation and expansion, prevented myelopoiesis, and reduced atherosclerosis.50
In contrast to the pro-atherogenic role of GLUT1 in HSPCs, GLUT1 can also facilitate anti-atherosclerotic actions in plaque macrophages. GLUT1 (also known as Scl2a1) expression is increased during the process of efferocytosis, which fuels increased glucose uptake and a shift from oxidative phosphorylation towards increased aerobic glycolysis, which is needed for effective clearance of apoptotic cells.51 Bone marrow transplantation from myeloid-targeted LysM-Cre Slc2a1fl/fl mice into Ldlr−/− mice on a Western type diet (WTD) increased the necrotic core in the aorta.51
From a clinical perspective, circulating monocytes isolated from patients with atherosclerotic cardiovascular disease, showed an increased cytokine production capacity, which was associated with an up-regulation of glycolytic enzymes.24,49 This phenotype also persisted after in vitro differentiation into macrophages, showing a higher glycolytic flux accompanied by an increased oxygen consumption rate.49
Thus, there is ample evidence that atherosclerosis is associated with an increased glycolysis in myeloid cells. A series of recent studies reported that trained immunity is also critically dependent on activation of glycolysis.7,52 Of note, Arts et al.52 demonstrated that genetic variation in glycolytic genes affects the susceptibility of the innate immune system for BCG-induced training. The causal role for glycolysis in β-glucan-induced trained immunity has been validated by the observation that in vitro trained immunity is completely abrogated by the pharmacological inhibition of glycolysis with 2-deoxy-glucose.7 Moreover, administration of metformin, which also interferes with glycolysis, to mice during and after primary infection with C. albicans completely inhibited the protective effects leading to increased survival during disseminated candidiasis induced by a primary C. albicans injection.7 A possible role for the glycolysis-mediated induction of trained immunity in atherosclerosis specifically is evidenced by the observation that primary human monocytes trained with the common pro-atherogenic stimulus oxLDL display increased lactate production, suggesting increased glycolysis and subsequent pyruvate fermentation in vitro.11
The collection of evidence discussed above mounts the hypothesis that the up-regulated glycolysis observed in atherosclerosis, is intricately linked to trained immunity as an underlying mechanism. Whether this impacts on the progression of atherosclerosis in humans remains to be elucidated.
4.2 Cholesterol metabolism as central player linking lipid metabolism and inflammation
The synthesis of cholesterol is not only important in maintaining the structural integrity of cell membranes, but the isoprenoid intermediates derived from the cholesterol synthesis pathway are also needed for the prenylation of signalling and effector molecules involved in inflammatory responses. Therefore, the cholesterol homeostasis of a cell is tightly regulated by several transcription factors, including the Liver X receptor (LXR) and sterol regulatory element-binding protein (SREBP) family, which are both important in regulating inflammatory activity.
The importance of cholesterol homeostasis for immune function in the context of atherosclerosis is illustrated by various observations in murine models of atherosclerosis. Mice deficient in the cholesterol efflux transporters, ABCA1 and ABCG1, show accumulation of cholesterol in peritoneal macrophages and increased inflammatory responses to TLR ligands.15 In addition, Feig et al.53 showed that lipid-laden foam cells, residing in the atherosclerotic plaque environment, exhibit an increased inflammatory gene expression. Furthermore, Parathath et al.54 demonstrated that murine macrophages cultured under hypoxic conditions increase cellular sterol and triglycerides. This correlated with augmented activity and mRNA expression of 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase, thereby increasing the production of cholesterol. Moreover, hypoxia abrogated the ABCA1-mediated cholesterol efflux and these effects were reversed by reducing the expression of HIF-1α.54 Interestingly, Westerterp et al.14 demonstrated that Ldlr−/− mice transplanted with specific ABCA1 and ABCG1-deficient macrophages showed increased atherosclerotic lesions and a concomitant augmented expression of inflammatory chemokines within macrophage-containing areas.
Whilst these studies imply that cholesterol accumulation mounts pro-inflammatory responses, Spann et al.55 on the contrary reported that foam cells derived from murine peritoneal macrophages have an anti-inflammatory phenotype due to the accumulation of desmosterol. This finding was recently confirmed in lipid-laden peritoneal macrophages from Ldlr−/− mice on a high-fat diet.56 In addition to LXR-activation by desmosterol, suppression of the PPP attenuated the inflammatory responses. This suggests that foam cell formation per se results in a predominantly anti-inflammatory macrophage phenotype, but that it requires extrinsic pro-inflammatory stimuli from the atherosclerotic milieu to adopt a pro-inflammatory phenotype.
Cholesterol metabolism not only impacts on disease progression in the atherosclerotic plaque itself, but could also play a role in earlier disease stages, i.e. at the level of the bone marrow. Transplantation of Abca−/− and Abcg−/− bone marrow cells into Ldlr−/− mice resulted in increased haematopoietic stem and progenitor cells (HSPC) expansion and concurrent monocytosis and accelerated atherosclerosis.57 The same effects occurred in apoE−/− mice, in which HSPCs also demonstrated a reduced ABCA1- and ABCG1-mediated cholesterol efflux.58
Analysis of transcriptome data of β-glucan-trained macrophages revealed that the cholesterol synthesis pathway was also highly induced in trained cells. Bekkering et al. have further unravelled the role of this pathway in trained immunity and revealed that not the isoprenylation pathway, nor the synthesis of cholesterol itself, were responsible for the trained phenotype, but that the accumulation of mevalonate induced epigenetic reprogramming and subsequent trained immunity. This occurred via the activation of the insulin-like-growth factor-1 receptor pathway and subsequent induction of mTOR signalling and glycolysis.59 Monocytes from patients with mevalonate accumulation in the context of the Hyper-IgD syndrome showed a trained immunity phenotype, which was characterized by an increased expression of cytokines and genes involved in the glycolytic pathway. Inhibition of the cholesterol synthesis pathway by statins inhibited the trained phenotype. Whether intracellular cholesterol metabolism also induces trained immunity in patients with cardiovascular disease remains a subject of further investigation.
5. Getting to the core of the training course: trained immunity in the bone marrow
Atherosclerotic lesions grow through a combination of continuous recruitment of circulating monocytes60,61 as well as local macrophage proliferation.62 We previously hypothesized that trained circulating monocytes could therefore aggravate atherosclerotic lesion development.11 Interestingly, in humans in vivo, a trained immune phenotype of circulating monocytes remains apparent even 3 months after vaccination with BCG.8 Given the short circulating half-life of monocytes of only 1–4 days, it is likely that training already occurs at the level of myeloid progenitor cells in the bone marrow. Indeed, a recent series of papers unequivocally demonstrate that training occurs in myeloid progenitor cells in the bone marrow, at least in mouse models.63–65
Mitroulis et al. reported that in mice, administration of β-glucan induced a sustained increased proliferation and myeloid skewing of haematopoietic stem cells (HSCs) which was associated with mitigated LPS-induced DNA damage upon a subsequent LPS challenge. Moreover, the β-glucan-trained haematopoietic precursors were more efficiently protected against DNA damage and cell death induced by myeloablative chemotherapy. Interestingly, RNAseq analysis of long-term HSCs revealed profound metabolic reprogramming, especially involving genes in glycolysis, cholesterol biosynthesis, and the mevalonate pathway.64 Pharmacological inhibition with the IL-1 receptor antagonist Anakinra revealed that the up-regulation of glycolytic enzymes, via IL-1β signaling, was essential for HSC proliferation following β-glucan administration.64
Similar to β-glucan-induced reprogramming of HSCs, BCG vaccination in mice led to sustained enhanced myelopoiesis in HSCs and multipotent progenitor cells.65 Macrophages derived from BCG-educated HSCs showed profound reprogramming of activating histone modifications, showing that these cells are epigenetically primed to initiate a more protective response against a subsequent infection with M. tuberculosis.
That trained immunity at the bone marrow level also plays a role in the context of atherosclerosis, was recently shown by Christ et al.63 In an apoE−/− mouse model of atherosclerosis, a 4-week WTD induced a persistent pro-inflammatory reprogramming of innate immune progenitor cells in the bone marrow. This pro-atherogenic and pro-inflammatory phenotype persisted even 4 weeks after reversing to a normal chow diet. Gene Ontology term enrichment network analysis of differentially expressed genes in granulocyte-monocyte progenitor (GMP) cells from these WTD-fed mice revealed up-regulation of various metabolic processes, including the cholesterol biosynthesis pathway. Moreover, they investigated the epigenetic profiling of GMPs from mice fed a WTD compared with chow diet by assay for transposase-accessible chromatin sequencing (ATAC-seq) and revealed that the WTD induced changes in the chromatin landscape. Also in this model, activation of the NLRP3 inflammasome and IL-1β signalling was fundamental for persistent GMP reprogramming.63 Pietras et al.66 showed that IL-1 exposure can drive myeloid differentiation in HSCs. Whereas IL-1 administration always enhances myeloid cell production, it can severely compromise HSC function and blood regeneration in conditions of chronic exposure. However, when mice were injected with IL-1β for 20 days and allowed to rest without further treatment for 8 weeks, most of the bone marrow changes had reverted to untreated levels, with normal numbers of HSCs and multipotent progenitors. The only persisting differences were increased GMP and decreased MMP4 numbers.66 As the effects on the HSCs were largely resolved after IL-1 withdrawal, it remains elusive whether the epigenetic modifications seen in GMPs also occur in the HSCs of WTD-fed mice. If these are restricted to GMPs, the longevity of the epigenetic changes and increased inflammatory response in myeloid cells might be determined by the turnover of these progenitors or the stimulus.
Since increased levels of IL-1β and other pro-inflammatory cytokines are a hallmark of various chronic inflammatory conditions, including rheumatoid arthritis, inflammatory bowel disease, psoriasis, obesity and metabolic syndrome, trained immunity at the level of bone marrow myeloid progenitors might provide a general mechanism driving the progression of atherosclerosis.67–69 It was recently reported that in rheumatoid arthritis, atherosclerosis is accelerated due to increased proliferation and myeloid skewing of HSCs in the bone marrow.70 Notably, these HSCs were characterized by an increased cholesterol content, due to defects in the cholesterol efflux pathways. Even after mobilization into the circulation, the mature myeloid cells retained these defects in their cholesterol metabolism. Moreover, the HSCs showed increased cell surface levels of the common β-subunit of the IL-3/granulocyte-macrophage-colony stimulating factor (GM-CSF) receptor (IL-3Rβ),57,70 which phenocopies the HSC phenotype after β-glucan training.64
6. Clinical relevance and future directions
Evidence that central metabolic pathways, including glycolysis and cholesterol metabolism, are profoundly reprogrammed in myeloid cells in the different stages of atherosclerosis is rapidly accumulating. These processes also play major roles in the induction of trained immunity, with an intimate connection to epigenetic reprogramming.
To determine the point at which the metabolic adaptations of monocytes and macrophages become disease promoting, it is required to study the metabolic changes in the multiple phases of the development of atherosclerosis. These stages range from adaptation of myeloid precursors in the bone marrow to foam cell formation in atherosclerotic plaques, and most importantly rupture-prone plaques.
A potential non-invasive tool to visualize metabolic changes in atherosclerosis in humans is in vivo visualization of macrophage glycolysis with either FDG-PET/CT or hyperpolarized MRI.71–73 Lewis et al.71 used hyperpolarized [1-13C]pyruvate MR spectroscopy as a novel method to detect glycolytic metabolism in a macrophage like-cell suspension. Upon macrophage activation with LPS, the hyperpolarized lactate label flux rates almost doubled. Inhibition of glycolysis in these activated cells by 2-deoxyglucose normalized the rates and concurrently inhibited the production of key pro-inflammatory cytokines.
Targeting the disease-promoting metabolic changes in myeloid cells during atherogenesis could be an exciting novel therapeutic strategy. Notably, metformin and statins, drugs that have already been used in clinical practice for years, interfere with key metabolic pathways that also drive innate immune activation in atherosclerosis.7,52,59 In a study by Duivenvoorden et al.74 an injectable reconstituted high-density lipoprotein (rHDL) nanoparticle carrier vehicle was developed that is able to directly deliver statins to myeloid cells within atherosclerotic plaques. In vitro statin-rHDL showed an anti-inflammatory effect, which was mediated through the inhibition of the mevalonate pathway. When the statin-rHDL nanoparticles were subsequently applied in vivo in an apoE−/− mouse model, they accumulated in the atherosclerotic plaques, where they directly affected the plaque residing macrophages.74 A low-dose statin-rHDL treatment regimen during 3 months inhibited plaque progression, while in more advanced atherosclerotic plaques a 1-week high-dose regimen was able to markedly decrease inflammation.
In addition to drugs targeting intracellular metabolic pathways, epigenetic reprogramming is also amenable to pharmacological modulation and could serve as a potential tool to manipulate macrophage inflammatory responses in atherosclerosis (an extensive recent review can be found elsewhere75). Several drugs that inhibit epigenetic enzymes, such as histone deacetylase inhibitors, are already approved for clinical use in the fields of oncology and haematology and have been shown to also reduce inflammation in inflammatory diseases.76,77
Finally, since inflammatory cytokines have been identified as potent inducers of trained immunity, inhibition of these cytokines might also serve as a potential therapeutic target. For instance, the studies by Mitroulis et al.64 and Christ et al.63 have implied that inflammasome-mediated products such as IL-1β could be the central endogenous mediator in the induction of trained immunity. It is tempting to speculate that these mechanisms are part of the clinical benefit observed in the recent CANTOS trial, which studied IL-1β blockade in humans at cardiovascular risk.78 The CIRT study on the other hand, which was running parallel to the CANTOS trial, investigated whether inhibition of the CRP/IL-6/IL-1-axis with low-dose methotrexate (MTX) could reduce cardiovascular events. Among patients with stable atherosclerosis, low-dose MTX did not reduce levels of IL-1β, IL-6, or C-reactive protein and did not result in reduced cardiovascular events compared with placebo.79 Potentially, this could be explained by the fact that MTX affects S-adenosylmethionine levels, which is the main methyldonor for DNA and histone methylation, and as such may interfere with epigenetic remodelling and subsequent inflammatory responses.
In conclusion, further elucidation of the metabolic and epigenetic adaptations that control the development of atherosclerotic plaques will allow the future development of new classes of drugs to prevent or treat atherosclerosis.
Conflict of interest: none declared.
Funding
This work was supported by the Netherlands Organisation for Scientific Research (NWO) [Rubicon grant 452173113 to S.B.]; the Competitiveness Operational Programme grant of the Romanian Ministry of European Funds [HINT, P_37_762 to L.A.B.J.]; the Horizon 2020 REPROGRAM [667837 to N.P.R., L.A.B.J., and M.G.N.]; M.G.N. was supported by a Spinoza grant of the Netherlands Organization for Scientific Research.
This article is part of the Spotlight Issue on Immunometabolism.
References
- 1. GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017;390:1151–1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Moore KJ, Sheedy FJ, Fisher EA.. Macrophages in atherosclerosis: a dynamic balance. Nat Rev Immunol 2013;13:709–721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Boring L, Gosling J, Cleary M, Charo IF.. Decreased lesion formation in CCR2-/- mice reveals a role for chemokines in the initiation of atherosclerosis. Nature 1998;394:894–897. [DOI] [PubMed] [Google Scholar]
- 4. Sabatine MS, Morrow DA, Cannon CP, Murphy SA, Demopoulos LA, DiBattiste PM, McCabe CH, Braunwald E, Gibson CM.. Relationship between baseline white blood cell count and degree of coronary artery disease and mortality in patients with acute coronary syndromes: a TACTICS-TIMI 18 (Treat Angina with Aggrastat and determine Cost of Therapy with an Invasive or Conservative Strategy- Thrombolysis in Myocardial Infarction 18 trial) substudy. J Am Coll Cardiol 2002;40:1761–1768. [DOI] [PubMed] [Google Scholar]
- 5. Rogacev KS, Cremers B, Zawada AM, Seiler S, Binder N, Ege P, Große-Dunker G, Heisel I, Hornof F, Jeken J, Rebling NM, Ulrich C, Scheller B, Böhm M, Fliser D, Heine GH.. CD14++CD16+ monocytes independently predict cardiovascular events: a cohort study of 951 patients referred for elective coronary angiography. J Am Coll Cardiol 2012;60:1512–1520. [DOI] [PubMed] [Google Scholar]
- 6. O'Neill LA, Kishton RJ, Rathmell J.. A guide to immunometabolism for immunologists. Nat Rev Immunol 2016;16:553–565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cheng SC, Quintin J, Cramer RA, Shepardson KM, Saeed S, Kumar V, Giamarellos-Bourboulis EJ, Martens JH, Rao NA, Aghajanirefah A, Manjeri GR, Li Y, Ifrim DC, Arts RJ, van der Veer BM, Deen PM, Logie C, O'Neill LA, Willems P, van de Veerdonk FL, van der Meer JW, Ng A, Joosten LA, Wijmenga C, Stunnenberg HG, Xavier RJ, Netea MG.. mTOR- and HIF-1alpha-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 2014;345:1250684.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kleinnijenhuis J, Quintin J, Preijers F, Joosten LA, Ifrim DC, Saeed S, Jacobs C, van Loenhout J, de Jong D, Stunnenberg HG, Xavier RJ, van der Meer JW, van Crevel R, Netea MG.. Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci U S A 2012;109:17537–17542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Quintin J, Saeed S, Martens JHA, Giamarellos-Bourboulis EJ, Ifrim DC, Logie C, Jacobs L, Jansen T, Kullberg BJ, Wijmenga C, Joosten LAB, Xavier RJ, van der Meer JWM, Stunnenberg HG, Netea MG.. Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe 2012;12:223–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Kleinnijenhuis J, Quintin J, Preijers F, Joosten LA, Jacobs C, Xavier RJ, van der Meer JW, van Crevel R, Netea MG.. BCG-induced trained immunity in NK cells: role for non-specific protection to infection. Clin Immunol 2014;155:213–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bekkering S, Quintin J, Joosten LA, van der Meer JW, Netea MG, Riksen NP.. Oxidized low-density lipoprotein induces long-term proinflammatory cytokine production and foam cell formation via epigenetic reprogramming of monocytes. Arterioscler Thromb Vasc Biol 2014;34:1731–1738. [DOI] [PubMed] [Google Scholar]
- 12. van der Valk FM, Bekkering S, Kroon J, Yeang C, Van den Bossche J, van Buul JD, Ravandi A, Nederveen AJ, Verberne HJ, Scipione C, Nieuwdorp M, Joosten LA, Netea MG, Koschinsky ML, Witztum JL, Tsimikas S, Riksen NP, Stroes ES.. Oxidized phospholipids on lipoprotein(a) elicit arterial wall inflammation and an inflammatory monocyte response in humans. Circulation 2016;134:611–624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. van der Heijden C, Noz MP, Joosten LAB, Netea MG, Riksen NP, Keating ST.. Epigenetics and trained immunity. Antioxid Redox Signal 2018;29:1023–1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Westerterp M, Murphy AJ, Wang M, Pagler TA, Vengrenyuk Y, Kappus MS, Gorman DJ, Nagareddy PR, Zhu X, Abramowicz S, Parks JS, Welch C, Fisher EA, Wang N, Yvan-Charvet L, Tall AR.. Deficiency of ATP-binding cassette transporters A1 and G1 in macrophages increases inflammation and accelerates atherosclerosis in mice. Circ Res 2013;112:1456–1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Yvan-Charvet L, Ranalletta M, Wang N, Han S, Terasaka N, Li R, Welch C, Tall AR.. Combined deficiency of ABCA1 and ABCG1 promotes foam cell accumulation and accelerates atherosclerosis in mice. J Clin Invest 2007;117:3900–3908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Manning-Tobin JJ, Moore KJ, Seimon TA, Bell SA, Sharuk M, Alvarez-Leite JI, de Winther MP, Tabas I, Freeman MW.. Loss of SR-A and CD36 activity reduces atherosclerotic lesion complexity without abrogating foam cell formation in hyperlipidemic mice. Arterioscler Thromb Vasc Biol 2009;29:19–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. El-Osta A, Brasacchio D, Yao D, Pocai A, Jones PL, Roeder RG, Cooper ME, Brownlee M.. Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J Exp Med 2008;205:2409–2417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Crişan TO, Cleophas MCP, Novakovic B, Erler K, van de Veerdonk FL, Stunnenberg HG, Netea MG, Dinarello CA, Joosten LAB.. Uric acid priming in human monocytes is driven by the AKT-PRAS40 autophagy pathway. Proc Natl Acad Sci USA 2017;114:5485–5490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. van Diepen JA, Thiem K, Stienstra R, Riksen NP, Tack CJ, Netea MG.. Diabetes propels the risk for cardiovascular disease: sweet monocytes becoming aggressive? Cell Mol Life Sci 2016;73:4675–4684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Botham KM, Wheeler-Jones CP.. Postprandial lipoproteins and the molecular regulation of vascular homeostasis. Prog Lipid Res 2013;52:446–464. [DOI] [PubMed] [Google Scholar]
- 21. Kleinnijenhuis J, Quintin J, Preijers F, Benn CS, Joosten LA, Jacobs C, van Loenhout J, Xavier RJ, Aaby P, van der Meer JW, van Crevel R, Netea MG.. Long-lasting effects of BCG vaccination on both heterologous Th1/Th17 responses and innate trained immunity. J Innate Immun 2014;6:152–158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Arts RJW, Moorlag S, Novakovic B, Li Y, Wang SY, Oosting M, Kumar V, Xavier RJ, Wijmenga C, Joosten LAB, Reusken C, Benn CS, Aaby P, Koopmans MP, Stunnenberg HG, van Crevel R, Netea MG.. BCG vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe 2018;23:89–100.e5. [DOI] [PubMed] [Google Scholar]
- 23. Leentjens J, Bekkering S, Joosten LAB, Netea MG, Burgner DP, Riksen NP.. Trained innate immunity as a novel mechanism linking infection and the development of atherosclerosis. Circ Res 2018;122:664–669. [DOI] [PubMed] [Google Scholar]
- 24. Bekkering S, van den Munckhof I, Nielen T, Lamfers E, Dinarello C, Rutten J, de Graaf J, Joosten LA, Netea MG, Gomes ME, Riksen NP.. Innate immune cell activation and epigenetic remodeling in symptomatic and asymptomatic atherosclerosis in humans in vivo. Atherosclerosis 2016;254:228–236. [DOI] [PubMed] [Google Scholar]
- 25. Noz M, ter Telgte A, Wiegertjes K, Joosten LA, Netea MG, de Leeuw F, Riksen NP.. Trained immunity characteristics are associated with progressive cerebral small vessel disease. Stroke 2018;49:2910–2917. [DOI] [PubMed] [Google Scholar]
- 26. Saeed S, Quintin J, Kerstens HH, Rao NA, Aghajanirefah A, Matarese F, Cheng SC, Ratter J, Berentsen K, van der Ent MA, Sharifi N, Janssen-Megens EM, Ter Huurne M, Mandoli A, van Schaik T, Ng A, Burden F, Downes K, Frontini M, Kumar V, Giamarellos-Bourboulis EJ, Ouwehand WH, van der Meer JW, Joosten LA, Wijmenga C, Martens JH, Xavier RJ, Logie C, Netea MG, Stunnenberg HG.. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science 2014;345:1251086.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kouzarides T. Chromatin modifications and their function. Cell 2007;128:693–705. [DOI] [PubMed] [Google Scholar]
- 28. Bannister AJ, Kouzarides T.. Regulation of chromatin by histone modifications. Cell Res 2011;21:381–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Greer EL, Shi Y.. Histone methylation: a dynamic mark in health, disease and inheritance. Nat Rev Genet 2012;13:343–357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bernstein BE, Meissner A, Lander ES.. The mammalian epigenome. Cell 2007;128:669–681. [DOI] [PubMed] [Google Scholar]
- 31. Ifrim DC, Quintin J, Joosten LA, Jacobs C, Jansen T, Jacobs L, Gow NA, Williams DL, van der Meer JW, Netea MG.. Trained immunity or tolerance: opposing functional programs induced in human monocytes after engagement of various pattern recognition receptors. Clin Vaccine Immunol 2014;21:534–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Novakovic B, Habibi E, Wang SY, Arts RJW, Davar R, Megchelenbrink W, Kim B, Kuznetsova T, Kox M, Zwaag J, Matarese F, van Heeringen SJ, Janssen-Megens EM, Sharifi N, Wang C, Keramati F, Schoonenberg V, Flicek P, Clarke L, Pickkers P, Heath S, Gut I, Netea MG, Martens JHA, Logie C, Stunnenberg HG. β -glucan reverses the epigenetic state of LPS-induced immunological tolerance. Cell 2016;167:1354–1368.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Fuster JJ, MacLauchlan S, Zuriaga MA, Polackal MN, Ostriker AC, Chakraborty R, Wu CL, Sano S, Muralidharan S, Rius C, Vuong J, Jacob S, Muralidhar V, Robertson AA, Cooper MA, Andres V, Hirschi KK, Martin KA, Walsh K.. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 2017;355:842–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Tall AR, Yvan-Charvet L.. Cholesterol, inflammation and innate immunity. Nat Rev Immunol 2015;15:104–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Mills EL, Kelly B, Logan A, Costa ASH, Varma M, Bryant CE, Tourlomousis P, Däbritz JHM, Gottlieb E, Latorre I, Corr SC, McManus G, Ryan D, Jacobs HT, Szibor M, Xavier RJ, Braun T, Frezza C, Murphy MP, O’Neill LA.. Succinate dehydrogenase supports metabolic repurposing of mitochondria to drive inflammatory macrophages. Cell 2016;167:457–470.e13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Jha AK, Huang SC, Sergushichev A, Lampropoulou V, Ivanova Y, Loginicheva E, Chmielewski K, Stewart KM, Ashall J, Everts B, Pearce EJ, Driggers EM, Artyomov MN.. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 2015;42:419–430. [DOI] [PubMed] [Google Scholar]
- 37. Lachmandas E, Boutens L, Ratter JM, Hijmans A, Hooiveld GJ, Joosten LA, Rodenburg RJ, Fransen JA, Houtkooper RH, van Crevel R, Netea MG, Stienstra R.. Microbial stimulation of different Toll-like receptor signalling pathways induces diverse metabolic programmes in human monocytes. Nat Microbiol 2016;2:16246.. [DOI] [PubMed] [Google Scholar]
- 38. Zhou R, Yazdi AS, Menu P, Tschopp J.. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011;469:221–225. [DOI] [PubMed] [Google Scholar]
- 39. Keating ST, El-Osta A.. Epigenetics and metabolism. Circ Res 2015;116:715–736. [DOI] [PubMed] [Google Scholar]
- 40. Figueroa ME, Abdel-Wahab O, Lu C, Ward PS, Patel J, Shih A, Li Y, Bhagwat N, Vasanthakumar A, Fernandez HF, Tallman MS, Sun Z, Wolniak K, Peeters JK, Liu W, Choe SE, Fantin VR, Paietta E, Lowenberg B, Licht JD, Godley LA, Delwel R, Valk PJ, Thompson CB, Levine RL, Melnick A.. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell 2010;18:553–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Sasaki M, Knobbe CB, Munger JC, Lind EF, Brenner D, Brüstle A, Harris IS, Holmes R, Wakeham A, Haight J, You-Ten A, Li WY, Schalm S, Su SM, Virtanen C, Reifenberger G, Ohashi PS, Barber DL, Figueroa ME, Melnick A, Zúñiga-Pflücker J-C, Mak TW.. IDH1(R132H) mutation increases murine haematopoietic progenitors and alters epigenetics. Nature 2012;488:656–659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Arts RJ, Novakovic B, Ter Horst R, Carvalho A, Bekkering S, Lachmandas E, Rodrigues F, Silvestre R, Cheng SC, Wang SY, Habibi E, Goncalves LG, Mesquita I, Cunha C, van Laarhoven A, van de Veerdonk FL, Williams DL, van der Meer JW, Logie C, O'Neill LA, Dinarello CA, Riksen NP, van Crevel R, Clish C, Notebaart RA, Joosten LA, Stunnenberg HG, Xavier RJ, Netea MG.. Glutaminolysis and fumarate accumulation integrate immunometabolic and epigenetic programs in trained immunity. Cell Metab 2016;24:807–819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Tawakol A, Singh P, Mojena M, Pimentel-Santillana M, Emami H, MacNabb M, Rudd JHF, Narula J, Enriquez JA, Través PG, Fernández-Velasco M, Bartrons R, Martín-Sanz P, Fayad ZA, Tejedor A, Boscá L.. HIF-1alpha and PFKFB3 mediate a tight relationship between proinflammatory activation and anerobic metabolism in atherosclerotic macrophages. Arterioscler Thromb Vasc Biol 2015;35:1463–1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Aarup A, Pedersen TX, Junker N, Christoffersen C, Bartels ED, Madsen M, Nielsen CH, Nielsen LB.. Hypoxia-inducible factor-1alpha expression in macrophages promotes development of atherosclerosis. Arterioscler Thromb Vasc Biol 2016;36:1782–1790. [DOI] [PubMed] [Google Scholar]
- 45. Folco EJ, Sheikine Y, Rocha VZ, Christen T, Shvartz E, Sukhova GK, Di Carli MF, Libby P.. Hypoxia but not inflammation augments glucose uptake in human macrophages: implications for imaging atherosclerosis with 18fluorine-labeled 2-deoxy-D-glucose positron emission tomography. J Am Coll Cardiol 2011;58:603–614. [DOI] [PubMed] [Google Scholar]
- 46. Liu J, Kerwin WS, Caldwell JH, Ferguson MS, Hippe DS, Alessio AM, Martinez-Malo V, Pimentel K, Miyaoka RS, Kohler TR, Hatsukami TS, Yuan C.. High resolution FDG-microPET of carotid atherosclerosis: plaque components underlying enhanced FDG uptake. Int J Cardiovasc Imaging 2016;32:145–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Silvera SS, Aidi HE, Rudd JH, Mani V, Yang L, Farkouh M, Fuster V, Fayad ZA.. Multimodality imaging of atherosclerotic plaque activity and composition using FDG-PET/CT and MRI in carotid and femoral arteries. Atherosclerosis 2009;207:139–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Tomas L, Edsfeldt A, Mollet IG, Perisic Matic L, Prehn C, Adamski J, Paulsson-Berne G, Hedin U, Nilsson J, Bengtsson E, Gonçalves I, Björkbacka H.. Altered metabolism distinguishes high-risk from stable carotid atherosclerotic plaques. Eur Heart J 2018;39:2301–2310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Shirai T, Nazarewicz RR, Wallis BB, Yanes RE, Watanabe R, Hilhorst M, Tian L, Harrison DG, Giacomini JC, Assimes TL, Goronzy JJ, Weyand CM.. The glycolytic enzyme PKM2 bridges metabolic and inflammatory dysfunction in coronary artery disease. J Exp Med 2016;213:337–354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Sarrazy V, Viaud M, Westerterp M, Ivanov S, Giorgetti-Peraldi S, Guinamard R, Gautier EL, Thorp EB, De Vivo DC, Yvan-Charvet L.. Disruption of Glut1 in hematopoietic stem cells prevents myelopoiesis and enhanced glucose flux in atheromatous plaques of ApoE(-/-) mice. Circ Res 2016;118:1062–1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Morioka S, Perry JSA, Raymond MH, Medina CB, Zhu Y, Zhao L, Serbulea V, Onengut-Gumuscu S, Leitinger N, Kucenas S, Rathmell JC, Makowski L, Ravichandran KS.. Efferocytosis induces a novel SLC program to promote glucose uptake and lactate release. Nature 2018;563:714–718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Arts RJW, Carvalho A, La Rocca C, Palma C, Rodrigues F, Silvestre R, Kleinnijenhuis J, Lachmandas E, Goncalves LG, Belinha A, Cunha C, Oosting M, Joosten LAB, Matarese G, van Crevel R, Netea MG.. Immunometabolic pathways in BCG-induced trained immunity. Cell Rep 2016;17:2562–2571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Feig JE, Vengrenyuk Y, Reiser V, Wu C, Statnikov A, Aliferis CF, Garabedian MJ, Fisher EA, Puig O.. Regression of atherosclerosis is characterized by broad changes in the plaque macrophage transcriptome. PLoS One 2012;7:e39790.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Parathath S, Mick SL, Feig JE, Joaquin V, Grauer L, Habiel DM, Gassmann M, Gardner LB, Fisher EA.. Hypoxia is present in murine atherosclerotic plaques and has multiple adverse effects on macrophage lipid metabolism. Circ Res 2011;109:1141–1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Spann NJ, Garmire LX, McDonald JG, Myers DS, Milne SB, Shibata N, Reichart D, Fox JN, Shaked I, Heudobler D, Raetz CR, Wang EW, Kelly SL, Sullards MC, Murphy RC, Merrill AH Jr, Brown HA, Dennis EA, Li AC, Ley K, Tsimikas S, Fahy E, Subramaniam S, Quehenberger O, Russell DW, Glass CK.. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. Cell 2012;151:138–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Baardman J, Verberk SGS, Prange KHM, van Weeghel M, van der Velden S, Ryan DG, Wüst RCI, Neele AE, Speijer D, Denis SW, Witte ME, Houtkooper RH, O’neill LA, Knatko EV, Dinkova-Kostova AT, Lutgens E, de Winther MPJ, Van den Bossche J.. A defective pentose phosphate pathway reduces inflammatory macrophage responses during hypercholesterolemia. Cell Rep 2018;25:2044–2052.e5. [DOI] [PubMed] [Google Scholar]
- 57. Yvan-Charvet L, Pagler T, Gautier EL, Avagyan S, Siry RL, Han S, Welch CL, Wang N, Randolph GJ, Snoeck HW, Tall AR.. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 2010;328:1689–1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Murphy AJ, Akhtari M, Tolani S, Pagler T, Bijl N, Kuo CL, Wang M, Sanson M, Abramowicz S, Welch C, Bochem AE, Kuivenhoven JA, Yvan-Charvet L, Tall AR.. ApoE regulates hematopoietic stem cell proliferation, monocytosis, and monocyte accumulation in atherosclerotic lesions in mice. J Clin Invest 2011;121:4138–4149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Bekkering S, Arts RJW, Novakovic B, Kourtzelis I, van der Heijden C, Li Y, Popa CD, Ter Horst R, van Tuijl J, Netea-Maier RT, van de Veerdonk FL, Chavakis T, Joosten LAB, van der Meer JWM, Stunnenberg H, Riksen NP, Netea MG.. Metabolic induction of trained immunity through the mevalonate pathway. Cell 2018;172:135–146.e9. [DOI] [PubMed] [Google Scholar]
- 60. Swirski FK, Libby P, Aikawa E, Alcaide P, Luscinskas FW, Weissleder R, Pittet MJ.. Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J Clin Invest 2007;117:195–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Swirski FK, Pittet MJ, Kircher MF, Aikawa E, Jaffer FA, Libby P, Weissleder R.. Monocyte accumulation in mouse atherogenesis is progressive and proportional to extent of disease. Proc Natl Acad Sci USA 2006;103:10340–10345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Robbins CS, Hilgendorf I, Weber GF, Theurl I, Iwamoto Y, Figueiredo JL, Gorbatov R, Sukhova GK, Gerhardt LM, Smyth D, Zavitz CC, Shikatani EA, Parsons M, van Rooijen N, Lin HY, Husain M, Libby P, Nahrendorf M, Weissleder R, Swirski FK.. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nat Med 2013;19:1166–1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Christ A, Günther P, Lauterbach MAR, Duewell P, Biswas D, Pelka K, Scholz CJ, Oosting M, Haendler K, Baßler K, Klee K, Schulte-Schrepping J, Ulas T, Moorlag SJCFM, Kumar V, Park MH, Joosten LAB, Groh LA, Riksen NP, Espevik T, Schlitzer A, Li Y, Fitzgerald ML, Netea MG, Schultze JL, Latz E.. Western diet triggers NLRP3-dependent innate immune reprogramming. Cell 2018;172:162–175.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Mitroulis I, Ruppova K, Wang B, Chen LS, Grzybek M, Grinenko T, Eugster A, Troullinaki M, Palladini A, Kourtzelis I, Chatzigeorgiou A, Schlitzer A, Beyer M, Joosten LAB, Isermann B, Lesche M, Petzold A, Simons K, Henry I, Dahl A, Schultze JL, Wielockx B, Zamboni N, Mirtschink P, Coskun U, Hajishengallis G, Netea MG, Chavakis T.. Modulation of myelopoiesis progenitors is an integral component of trained immunity. Cell 2018;172:147–161.e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Kaufmann E, Sanz J, Dunn JL, Khan N, Mendonca LE, Pacis A, Tzelepis F, Pernet E, Dumaine A, Grenier JC, Mailhot-Leonard F, Ahmed E, Belle J, Besla R, Mazer B, King IL, Nijnik A, Robbins CS, Barreiro LB, Divangahi M.. BCG educates hematopoietic stem cells to generate protective innate immunity against tuberculosis. Cell 2018;172:176–190.e19. [DOI] [PubMed] [Google Scholar]
- 66. Pietras EM, Mirantes-Barbeito C, Fong S, Loeffler D, Kovtonyuk LV, Zhang SYi, Lakshminarasimhan R, Chin CP, Techner J-M, Will B, Nerlov C, Steidl U, Manz MG, Schroeder T, Passegué E.. Chronic interleukin-1 exposure drives haematopoietic stem cells towards precocious myeloid differentiation at the expense of self-renewal. Nat Cell Biol 2016;18:607–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Hoogeveen RM, Nahrendorf M, Riksen NP, Netea MG, de Winther MPJ, Lutgens E, Nordestgaard BG, Neidhart M, Stroes ESG, Catapano AL, Bekkering S.. Monocyte and haematopoietic progenitor reprogramming as common mechanism underlying chronic inflammatory and cardiovascular diseases. Eur Heart J 2018;39:3521–3527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Mason JC, Libby P.. Cardiovascular disease in patients with chronic inflammation: mechanisms underlying premature cardiovascular events in rheumatologic conditions. Eur Heart J 2015;36:482–489c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Libby P, Nahrendorf M, Swirski FK.. Leukocytes link local and systemic inflammation in ischemic cardiovascular disease: an expanded “cardiovascular continuum”. J Am Coll Cardiol 2016;67:1091–1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Dragoljevic D, Kraakman MJ, Nagareddy PR, Ngo D, Shihata W, Kammoun HL, Whillas A, Lee MKS, Al-Sharea A, Pernes G, Flynn MC, Lancaster GI, Febbraio MA, Chin-Dusting J, Hanaoka BY, Wicks IP, Murphy AJ.. Defective cholesterol metabolism in haematopoietic stem cells promotes monocyte-driven atherosclerosis in rheumatoid arthritis. Eur Heart J 2018;39:2158–2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Lewis AJM, Miller JJ, Lau AZ, Curtis MK, Rider OJ, Choudhury RP, Neubauer S, Cunningham CH, Carr CA, Tyler DJ.. Noninvasive immunometabolic cardiac inflammation imaging using hyperpolarized magnetic resonance. Circ Res 2018;122:1084–1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Tawakol A, Migrino RQ, Bashian GG, Bedri S, Vermylen D, Cury RC, Yates D, LaMuraglia GM, Furie K, Houser S, Gewirtz H, Muller JE, Brady TJ, Fischman AJ.. In vivo 18F-fluorodeoxyglucose positron emission tomography imaging provides a noninvasive measure of carotid plaque inflammation in patients. J Am Coll Cardiol 2006;48:1818–1824. [DOI] [PubMed] [Google Scholar]
- 73. Figueroa AL, Subramanian SS, Cury RC, Truong QA, Gardecki JA, Tearney GJ, Hoffmann U, Brady TJ, Tawakol A.. Distribution of inflammation within carotid atherosclerotic plaques with high-risk morphological features: a comparison between positron emission tomography activity, plaque morphology, and histopathology. Circ Cardiovasc Imaging 2012;5:69–77. [DOI] [PubMed] [Google Scholar]
- 74. Duivenvoorden R, Tang J, Cormode DP, Mieszawska AJ, Izquierdo-Garcia D, Ozcan C, Otten MJ, Zaidi N, Lobatto ME, van Rijs SM, Priem B, Kuan EL, Martel C, Hewing B, Sager H, Nahrendorf M, Randolph GJ, Stroes ES, Fuster V, Fisher EA, Fayad ZA, Mulder WJ.. A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation. Nat Commun 2014;5:3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Nicorescu I, Dallinga GM, Mpj D. W, Stroes ESG, Bahjat M.. Potential epigenetic therapeutics for atherosclerosis treatment. Atherosclerosis 2019;281:189–197. [DOI] [PubMed] [Google Scholar]
- 76. Mann BS, Johnson JR, He K, Sridhara R, Abraham S, Booth BP, Verbois L, Morse DE, Jee JM, Pope S, Harapanhalli RS, Dagher R, Farrell A, Justice R, Pazdur R.. Vorinostat for treatment of cutaneous manifestations of advanced primary cutaneous T-cell lymphoma. Clin Cancer Res 2007;13:2318–2322. [DOI] [PubMed] [Google Scholar]
- 77. Mehta S, Jeffrey KL.. Beyond receptors and signaling: epigenetic factors in the regulation of innate immunity. Immunol Cell Biol 2015;93:233–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ, Group CT.. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 2017;377:1119–1131. [DOI] [PubMed] [Google Scholar]
- 79. Ridker PM, Everett BM, Pradhan A, MacFadyen JG, Solomon DH, Zaharris E, Mam V, Hasan A, Rosenberg Y, Iturriaga E, Gupta M, Tsigoulis M, Verma S, Clearfield M, Libby P, Goldhaber SZ, Seagle R, Ofori C, Saklayen M, Butman S, Singh N, Le May M, Bertrand O, Johnston J, Paynter NP, Glynn RJ.. Investigators C. Low-Dose Methotrexate for the Prevention of Atherosclerotic Events. N Engl J Med 2019;380:752–762. [DOI] [PMC free article] [PubMed] [Google Scholar]