Abstract
Background
High-sensitivity troponin I (hs-Tnl) and B-type natriuretic peptide (BNP) are promising prognostic markers for coronary artery disease (CAD). This prospective cohort study investigated whether a combination of these cardiac biomarkers with conventional risk factors would add incremental value for the prediction of secondary major adverse cardiovascular events (MACEs) in patients with CAD, with and without type 2 diabetes mellitus (T2DM).
Methods
Baseline plasma level of hs-Tnl and BNP was measured in 2275 Chinese patients with stable CAD. Patients were monitored for new-onset of MACE over a median of 51 months. Cox proportional hazard model and area under the receiver operating characteristic curve (AUC) were used to assess the association of cardiac biomarkers with MACE and their predictive values in relationship with or without T2DM.
Results
During the follow up period 402 (18%) patients experienced a new-onset MACE with hs-Tnl and BNP level significantly higher than in those without MACE. In multivariable analyses, patients with elevated hs-Tnl (hazard ratio, 1.75 [95% CI 1.41–2.17]; P < 0.001) and BNP (hazard ratio, 1.42 [95% CI 1.15–1.75]; P = 0.001) were significantly associated with an increased risk of MACE after adjustment for variables of a risk factor model of age, sex, T2DM and hypertension. The risk factor model had an AUC of 0.64 for MACE prediction. The AUC significantly increased to 0.68 by the addition of hs-Tnl to the risk factor model. Subgroup analyses showed that hs-Tnl and BNP remained significant predictors of MACE in both patients with and without T2DM in multivariable models with higher risk of MACE evident in those without T2DM. Among patients without T2DM, addition of each biomarker yielded greater predictive accuracy than in T2DM patients, with AUC further increased to 0.75 when a combination of hs-Tnl and BNP was added to the risk factor model (age, sex and hypertension).
Conclusions
Elevated hs-Tnl and BNP level are independent predictors of new-onset MACE in CAD patients, irrespective of diabetes status. Among CAD patients without T2DM, a combination of cardiac biomarkers hs-Tnl and BNP yield the greatest predictive value beyond conventional risk factors.
Keywords: B-type natriuretic peptide, Cardiac troponin, Coronary artery disease, Risk prediction, Type 2 diabetes mellitus
Background
Conventional risk factors that include age, gender, smoking, glucose level, blood pressure and cholesterol level have historically been used to risk stratify subjects who are at risk of major adverse cardiovascular events (MACEs) [1, 2]. Nevertheless these clinical risk factors on their own have limited predictive value in patients with established coronary artery disease (CAD) and in whom more frequent surveillance and aggressive risk factor control is desired. Recently, cardiac biomarkers have been shown to be important in prediction of cardiovascular risk and superior to models based on only conventional risk factors [3, 4]. High-sensitivity troponin I (hs-Tnl), a well-known diagnostic marker of myocardial injury, has been shown to be associated with the burden of coronary atherosclerosis and impaired cardiac performance [5, 6]. Elevated hs-Tnl concentration has been demonstrated to be associated with cardiovascular comorbidities and a strong predictor of adverse cardiovascular outcomes, particularly in patients with stable CAD or type 2 diabetes mellitus (T2DM), and in the elderly population [7–11]. B‐type natriuretic peptide (BNP), a widely used marker for early diagnosis of acute heart failure and for risk stratification of patients with congestive heart failure [12, 13], has also been shown to be highly predictive of the occurrence of acute coronary syndromes (ACS) and mortality [14–17]. It is unknown whether a combination of hs-TnI and BNP can provide any incremental benefit for risk prediction in patients with established CAD. In addition, T2DM is a known predictor of elevated hs-Tnl and BNP [18, 19], and in patients with heart failure, the presence of diabetes is associated with a higher BNP level [20]. It remains unclear whether these cardiac biomarkers provide different predictive abilities for subsequent cardiovascular events in CAD patients with and without T2DM.
In this study, we sought to investigate the association of these cardiac biomarkers with long-term adverse cardiovascular outcomes and to determine whether adoption of a multiple cardiac biomarkers approach can provide incremental benefit beyond a conventional risk factor approach in patients with stable CAD, as well as their predictive value in subgroups of CAD patients with or without T2DM.
Methods
Study population
In this prospective cohort study, we recruited consecutive patients with stable CAD who attended follow-up at the Cardiac Clinic, Queen Mary Hospital, Hong Kong from December 2003 to December 2014. All study participants received evidence-based medical therapies including coronary revascularization and statins. Diagnosis of stable CAD was defined according to the guidelines of the American College of Cardiology [21]. The study was approved by the local Institutional Review Board and all patients provided written informed consent.
Baseline and laboratory measurements
Comprehensive data on demographics, medical history, medication use, smoking status and anthropometric parameters including body mass index and blood pressure were recorded at enrollment. Missing anthropometric measurements were imputed using multiple imputation in R package MICE [22]. Patients with any tobacco use in the last 30 days were considered a current smoker. T2DM was defined according to the World Health Organization 1998 diagnostic criteria [23], or regular prescription of anti-diabetic medication. Hypertension was considered present in patients with a history of hypertension or regularly prescribed anti-hypertensive medication.
Blood samples were drawn at recruitment from all patients following an overnight fast of 12 h to measure lipid profiles, glucose, hs-Tnl and BNP. Samples were stored at − 70 °C until analysis. Plasma hs-Tnl was measured by ARCHITECT STAT high-sensitive troponin-I assay (Abbott Laboratories, Abbott Park, IL, USA). The limit of detection was 1.9 pg/mL and the coefficient of variation at 99th percentile was 4%. Plasma BNP level was measured using ARCHITECT BNP assay (Abbott Laboratories, Abbott Park, IL, USA), measurement range 10–5000 pg/mL.
Outcomes
Follow-up for each patient began at enrollment and continued until diagnosis of a cardiovascular end-point, death, last visit, or end of study follow-up, whichever came first. Cardiovascular end-point was new-onset MACE during the follow-up period. Diagnosis of MACE was based on the International Classification of Disease Ninth Revision (ICD-9), and included acute myocardial infarction (ICD-9 410), ACS (ICD-9 411.1), stroke (ICD-9 430, 431, 433, 434, 436), peripheral vascular disease (ICD-9 443.9), and cardiovascular death (death certificate ICD-9 410-447). Follow-up information including dates of events and discharge diagnosis were verified from medical records of the Hong Kong Hospital Authority database. The main cause and date of death were obtained from the Hong Kong Death Registry for patients who died during the study period.
Statistical analysis
Data are presented as mean ± standard deviation or number and percentage, as appropriate. Kolmogorov–Smirnov test was used to determine the normality assumption for continuous variables. We transformed variables with a skewed distribution using natural logarithm transformation before analysis. Age was categorized as younger than 65 years or 65 years and over based on our recent study [4]. The optimal cutoff value for each biomarker was determined by Youden J index. Comparisons between groups were evaluated using Student’s t test for continuous variables and Chi squared test for categorical variables.
Survival curves were estimated using Kaplan–Meier method and the cumulative incidence of MACE for each biomarker was compared using log-rank test. Cox proportional hazard analyses were performed to examine the association between biomarker levels and MACE, adjusting for age, clinical risk factors including sex, current smoker, T2DM, hypertension and body mass index, as well as enrollment period, which was considered a confounder influencing prognosis. As opposed to baseline blood pressure and hemoglobin A1c measurements, history of T2DM and hypertension were used in the model selection to reduce variations caused by factors such as anti-hypertensive or anti-diabetic medication use. The proportional hazards assumption in each Cox regression model was tested using Schoenfeld residuals and no violation was observed. Tests for the interactions of age, sex and T2DM status with cardiac biomarkers were also performed. The independent association of biomarker level with MACE was evaluated in various models. Model 1: age ≥ 65 years. Model 2: age ≥ 65 years, clinical risk factors and enrollment period; variables were chosen if they were associated with MACE in the age-adjusted models with P < 0.10 to avoid over-adjustment. Model 3 to 5: additionally included cardiac biomarkers alone or in combination with the clinical risk factor model. Sensitivity analysis was conducted by using the competing risks regression model of Fine and Gray to estimate the sub-hazard ratios for MACE, with non-cardiovascular deaths treated as competing risk [24].
The area under the receiver operating characteristic curve (AUC), category-free net reclassification index and integrated discrimination improvement were used to assess the incremental predictive value of a model. The differences between AUCs were compared using DeLong’s test [25]. The performance of Cox regression models was evaluated by using the Akaike Information Criterion for model comparisons, where a lower Akaike Information Criterion value indicates a better fit. Based on the findings from a previous study [26], assuming the prediction model with cardiac biomarkers and clinical risk factor to detect a hazard ratio of 1.50 for cardiovascular outcomes, 198 events were required to provide an 80% power with a 2-sided significance level of 0.05. Effective sample size was achieved based on the rule of ten outcome events per predictor to develop the risk prediction model using Cox proportional hazards regression [27].
A two-sided P value less than 0.05 was considered statistically significant. All statistical tests were performed using STATA statistical software (version 14.0) and R-programming language (version 3.5.1).
Results
Baseline characteristics and biomarker levels
The demographic and biochemical characteristics with respect to incident MACE during follow-up are summarized in Table 1. Patients with missing clinical or biomarker data (n = 177) were excluded and 2275 patients (mean age: 67.7 ± 10.5 years) were included in the study. Among all patients with stable CAD, 402 experienced new-onset MACE after a median follow-up of 51 months (incidence rate, 4.2 per 100 patient-years). Based on the Youden J index, the optimal cutoff plasma level of hs-Tnl and BNP for prediction of MACE was 14.2 pg/mL and 56.7 pg/mL, respectively. Affected CAD patients were significantly older, and a higher proportion were female, and had T2DM or hypertension compared with those with no MACE during follow-up (all P < 0.01). Patients with CAD and incident MACE also had higher levels of plasma hs-Tnl and BNP, hemoglobin A1c and systolic blood pressure, but lower diastolic blood pressure at baseline (all P < 0.001). Nonetheless no differences in body mass index or cholesterol level were evident between CAD patients with or without incident MACE (P > 0.05).
Table 1.
Baseline characteristics of the study cohort
| Variable | All | Subjects with major adverse cardiovascular events | Subjects without major adverse cardiovascular events | P value |
|---|---|---|---|---|
| N | 2275 | 402 | 1873 | |
| Age ≥ 65 years | 1400 (62) | 313 (78) | 1087 (58) | < 0.001 |
| Male | 1664 (73) | 270 (67) | 1394 (74) | 0.003 |
| Current smoker | 289 (13) | 49 (12) | 240 (13) | 0.73 |
| T2DM | 1617 (71) | 328 (82) | 1289 (69) | < 0.001 |
| Hypertension | 1997 (88) | 380 (95) | 1617 (86) | < 0.001 |
| BMI, kg/m2 | 25.8 ± 3.6 | 25.4 ± 3.9 | 25.8 ± 3.6 | 0.054 |
| Systolic blood pressure, mmHg | 137 ± 20 | 140 ± 23 | 137 ± 20 | < 0.001 |
| Diastolic blood pressure, mmHg | 73 ± 11 | 70 ± 12 | 74 ± 11 | < 0.001 |
| Triglycerides, mmol/L | 1.49 ± 1.02 | 1.51 ± 1.11 | 1.49 ± 1.00 | 0.65 |
| Total cholesterol, mmol/L | 3.89 ± 0.89 | 3.94 ± 1.01 | 3.88 ± 0.87 | 0.17 |
| HDL-cholesterol, mmol/L | 1.18 ± 0.33 | 1.18 ± 0.33 | 1.18 ± 0.33 | 0.79 |
| LDL-cholesterol, mmol/L | 2.01 ± 0.73 | 2.06 ± 0.83 | 2.01 ± 0.71 | 0.22 |
| HbA1c, % | 7.07 ± 1.39 | 7.41 ± 1.51 | 7.00 ± 1.36 | < 0.001 |
| Ln-hs-Tnl, pg/mL | 2.1 ± 1.2 | 2.5 ± 1.4 | 2.1 ± 1.1 | < 0.001 |
| Above cutoff (> 14.2 pg/mL) | 511 (22) | 148 (37) | 363 (19) | < 0.001 |
| Ln-BNP, pg/mL | 3.8 ± 1.1 | 4.1 ± 1.3 | 3.7 ± 1.1 | < 0.001 |
| Above cutoff (> 56.7 pg/mL) | 877 (39) | 212 (53) | 665 (36) | < 0.001 |
| Ln-Enrollment period, month | 3.1 ± 1.0 | 3.5 ± 0.9 | 3.0 ± 1.0 | < 0.001 |
Values are mean ± SD or n (%)
BMI body mass index, BNP B-type natriuretic peptide, HbA1c hemoglobin A1c, HDL-C high density lipoprotein cholesterol, hs-Tnl high-sensitivity troponin I, Ln natural logarithm, LDL-C low density lipoprotein cholesterol, T2DM type 2 diabetes mellitus
Baseline characteristics of study participants according to T2DM status are presented in Table 2. There were 1617 (71%) CAD patients had T2DM. They were older and more often had a history of hypertension than those without T2DM. Patients with T2DM also had a higher BNP concentration and body mass index, lower total cholesterol and low-density lipoprotein and similar hs-Tnl concentration compared with patients without T2DM. The annual event rate of MACE in T2DM CAD patients (4.6 per 100 patient-years) was significantly higher than for those without T2DM (3.0 per 100 patient-years; P < 0.001).
Table 2.
Comparison of baseline characteristics between patients with and without T2DM
| Variables | T2DM | No T2DM | P value |
|---|---|---|---|
| n | 1617 | 658 | |
| Age ≥ 65 years | 1029 (64) | 371 (56) | 0.001 |
| Male | 1168 (72) | 496 (75) | 0.13 |
| Current smoker | 181 (11) | 108 (16) | 0.001 |
| Hypertension | 1557 (96) | 440 (67) | < 0.001 |
| BMI, kg/m2 | 26.0 ± 3.6 | 25.2 ± 3.6 | < 0.001 |
| Systolic blood pressure, mmHg | 140 ± 21 | 131 ± 18 | < 0.001 |
| Diastolic blood pressure, mmHg | 73 ± 11 | 74 ± 11 | 0.066 |
| Triglycerides, mmol/L | 1.50 ± 1.02 | 1.46 ± 1.02 | 0.37 |
| Total cholesterol, mmol/L | 3.80 ± 0.85 | 4.09 ± 0.97 | < 0.001 |
| HDL-cholesterol, mmol/L | 1.16 ± 0.33 | 1.23 ± 0.34 | < 0.001 |
| LDL-cholesterol, mmol/L | 1.95 ± 0.67 | 2.17 ± 0.84 | < 0.001 |
| HbA1c, % | 7.38 ± 1.36 | 6.34 ± 1.17 | < 0.001 |
| Ln-hs-Tnl, pg/mL | 2.2 ± 1.1 | 2.1 ± 1.4 | 0.44 |
| Above cut-off (> 14.2 pg/mL) | 376 (23) | 135 (21) | 0.16 |
| Ln-BNP, pg/mL | 3.8 ± 1.1 | 3.6 ± 1.2 | < 0.001 |
| Above cut-off (> 56.7 pg/mL) | 672 (42) | 205 (31) | <0.001 |
| Ln-enrollment period, month | 3.1 ± 0.8 | 2.9 ± 1.3 | <0.001 |
| Outcomes | |||
| Major adverse cardiovascular events | 328 (20) | 74 (11) | < 0.001 |
Values are mean ± SD or n (%)
BMI body mass index, BNP B-type natriuretic peptide, HbA1c hemoglobin A1c, HDL-C high density lipoprotein cholesterol, hs-Tnl high-sensitivity troponin I, Ln natural logarithm, LDL-C low density lipoprotein cholesterol, T2DM type 2 diabetes mellitus
Predictive values of cardiac biomarkers for MACE
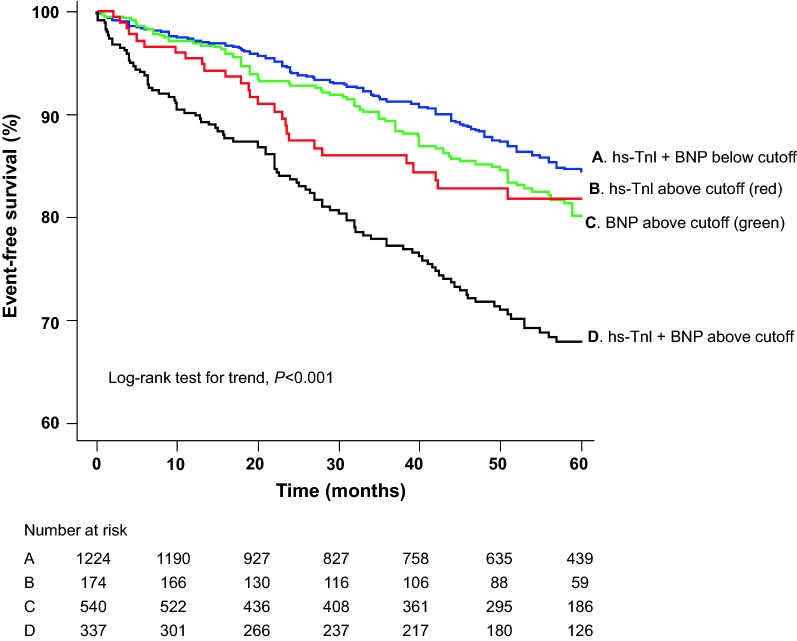
As shown in Fig. 1, Kaplan–Meier event-free curves for MACE show that patients with plasma hs-Tnl and BNP level above optimal cutoff value had significantly worse survival than those with levels below (log-rank test for trend P < 0.001). In the age-adjusted Cox regression analysis, sex, T2DM and hypertension were associated (P < 0.10) with an increased risk of MACE and were included in the final model (Additional file 1: Table S1). In the multivariable Cox proportional hazards model, higher level of individual biomarkers was strongly associated with an increased risk of subsequent MACE in all patients with stable CAD, after adjustment for advanced age, sex, T2DM and hypertension. The adjusted hazard ratio for patients with elevated level of hs-Tnl or BNP was 1.75 (95% CI 1.41–2.17; P < 0.001) and 1.42 (95% CI 1.15–1.75; P = 0.001), respectively (Table 3; Model 5).
Fig. 1.
Kaplan-Meier survival curves for MACEs in stable CAD patients. BNP B-type natriuretic peptide, hs-Tnl high-sensitivity troponin I
Table 3.
Multivariable Cox regression models predicting MACEs in stable CAD patients
| Variable | Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | HR (95% CI) | P value | HR (95% CI) | P value | HR (95% CI) | P value | |
| Age ≥ 65 years | 2.53 (2.00–3.21) | < 0.001 | 2.36 (1.85–2.99) | < 0.001 | 2.29 (1.80–2.91) | < 0.001 | 2.31 (1.82–2.93) | < 0.001 | 2.28 (1.79–2.90) | < 0.001 |
| Male | 0.83 (0.68–1.03) | 0.089 | 0.84 (0.68–1.04) | 0.098 | 0.85 (0.69–1.05) | 0.14 | 0.85 (0.69–1.06) | 0.15 | ||
| T2DM | – | – | 1.25 (0.95–1.65) | 0.11 | 1.24 (0.94–1.63) | 0.13 | 1.30 (0.99–1.71) | 0.062 | 1.28 (0.97–1.69) | 0.079 |
| Hypertension | – | – | 1.58 (1.00–2.50) | 0.051 | 1.54 (0.97–2.45) | 0.065 | 1.57 (0.99–2.49) | 0.055 | 1.55 (0.98–2.46) | 0.063 |
| BNP | – | – | – | – | 1.66 (1.36–2.02) | <0.001 | – | – | 1.42 (1.15–1.75) | 0.001 |
| hs-Tnl | – | – | – | – | – | – | 1.97 (1.60–2.41) | <0.001 | 1.75 (1.41–2.17) | < 0.001 |
BNP and hs-Tnl are at levels above optimal cutoffs
BNP B-type natriuretic peptide, CAD coronary artery disease, CI confidence interval, HR hazard ratio, hs-Tnl high-sensitivity troponin I, MACE major adverse cardiovascular event, T2DM type 2 diabetes mellitus
After accounting for the competing risk of non-cardiovascular death, the association of cardiac biomarkers with incident MACE remained significant and largely unchanged (Additional file 1: Table S2).
Incremental value of cardiac biomarkers over conventional risk factors for MACE
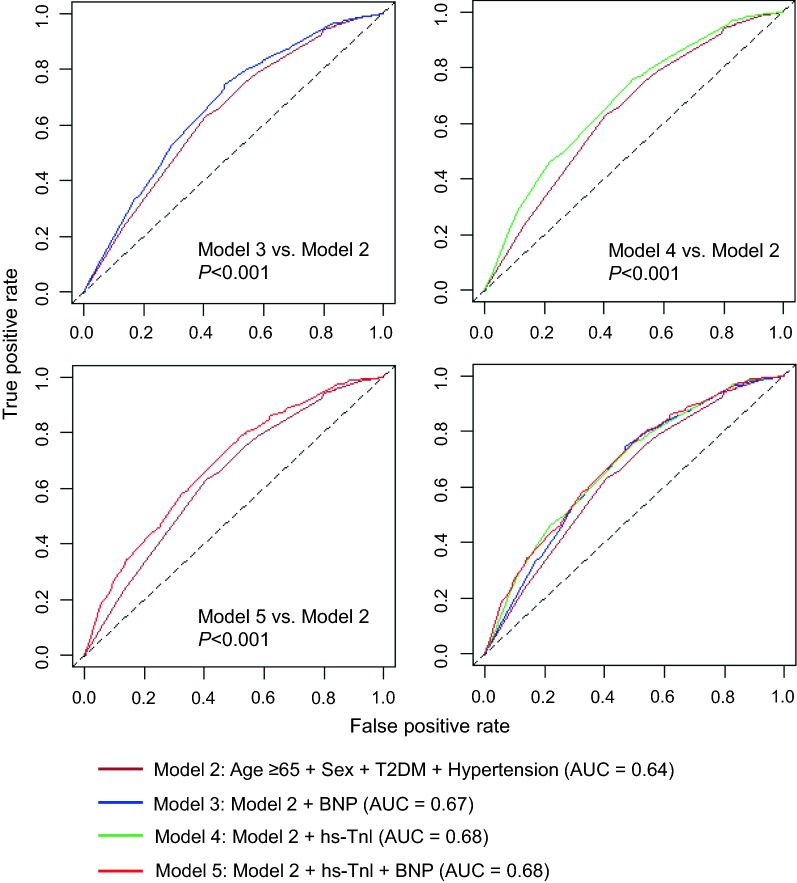
The receiver operating characteristic curve for different models are shown in Fig. 2. The AUC was 0.60 (95% CI 0.58–0.62) for model with age ≥ 65 years alone (Model 1). The addition of sex, T2DM and hypertension to the age model significantly increased the AUC to 0.64 (95% CI 0.61–0.67; Model 2). Moreover, significant increase in AUCs were observed when each biomarker was individually added to the risk factor model: hs-Tnl had the best single biomarker model with AUC further increased to 0.68 (95% CI 0.65–0.71; difference in AUCs, 0.04; P < 0.001; Model 4), whereas BNP yielded a slightly lower value of 0.67 (95% CI 0.64–0.69; difference in AUCs, 0.03; P = 0.001; Model 3) (Fig. 2). As shown in Table 4, both single biomarker models significantly improved risk reclassification and discrimination (hs-Tnl: net reclassification index [NRI], 34.9%; 95% CI 24.8–45.0; integrated discrimination improvement [IDI], 2.4%; 95% CI 1.6–3.2; BNP: NRI, 34.5%; 95% CI 23.8–45.1; IDI, 1.2%; 95% CI 0.7–1.7). According to the Akaike Information Criterion, the addition of a single or combination of biomarkers to the risk factor model improved model prediction for MACE (Table 4). In contrast, incorporating a combination of hs-Tnl and BNP into the age and clinical risk factor model (Model 5: AUC, 0.68; 95% CI 0.65–0.71) did not result in an increase in AUC when compared with the single biomarker model (Model 5 versus Model 4: DeLong’s test, P = 0.55).
Fig. 2.
Area under the receiver operating characteristic curves for predicting MACEs in stable CAD patients. BNP B-type natriuretic peptide, hs-Tnl high-sensitivity troponin I, T2DM type 2 diabetes mellitus
Table 4.
Performance of models predicting MACEs in stable CAD patients
| Model | Feature | AIC | NRI (95% CI) | P value | IDI (95% CI) | P value |
|---|---|---|---|---|---|---|
| Model 2 | Age ≥ 65 years + Male + T2DM + Hypertension | 5701.9 | – | – | – | – |
| Model 3 | Model 2 + BNP | 5678.3 | 34.5% (23.8–45.1%) | < 0.001 | 1.2% (0.7–1.7%) | < 0.001 |
| Model 4 | Model 2 + hs-Tnl | 5664.5 | 34.9% (24.8–45.0%) | < 0.001 | 2.4% (1.6–3.2%) | < 0.001 |
| Model 5 | Model 2 + hs-Tnl + BNP | 5655.5 | 29.7% (19.2–40.2%) | < 0.001 | 2.9% (2.1–3.8%) | < 0.001 |
Model 2: incorporated variables of age ≥ 65 years, male sex, presence of T2DM and hypertension; Model 3: variables in Model 2 and addition of BNP above optimal cutoff; Model 4: variables in Model 2 and addition of hs-Tnl above optimal cutoff; Model 5: variables in Model 2 and addition of hs-Tnl and BNP above cutoffs
AIC Akaike Information Criteria, BNP B-type natriuretic peptide, CAD coronary artery disease, CI confidence interval, hs-Tnl high-sensitivity troponin I, IDI integrated discrimination improvement, MACE major adverse cardiovascular event, NRI net reclassification index, T2DM type 2 diabetes mellitus
Interaction analyses showed significant interactions of T2DM status with hs-Tnl (Pinteraction = 0.046), and BNP (Pinteraction = 0.010), whereas no interactions of age and sex with biomarkers (all Pinteraction > 0.05) on MACE were found in the multivariable models.
Cardiac biomarkers for patients with or without T2DM
In multivariable Cox regression analyses, baseline hs-Tnl and BNP level remained significant predictors of MACE in both patients with and without T2DM after adjustment for age, sex and hypertension with higher risk of MACE evident in those without T2DM (Additional file 1: Table S3).
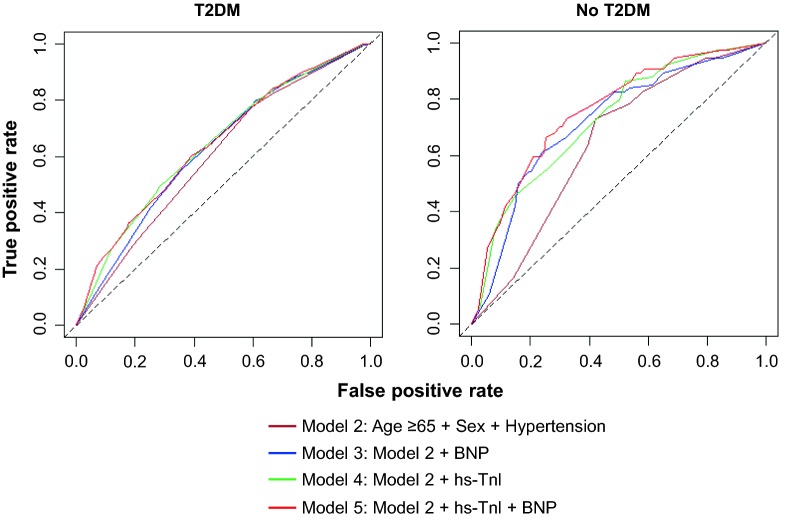
The predictive value of different models for MACE according to T2DM status are presented in Table 5 and Fig. 3. In patients with or without T2DM, the addition of any biomarker to the age and clinical risk factor model showed a significant increase in AUC for the prediction of MACE. Among patients with T2DM, the AUCs increased from 0.60 (95% CI 0.57–0.63) to 0.63 (95% CI 0.59–0.66) and 0.64 (95% CI 0.61–0.67) for BNP and hs-Tnl (DeLong’s test, both P < 0.001), respectively. Interestingly, the predictive performance of single biomarker models in patients without T2DM were significantly better than for those with T2DM (T2DM versus no T2DM, Model 3–5, all P < 0.05), with AUC increased from 0.64 (95% CI 0.59–0.70) to 0.72 (95% CI 0.66–0.78; DeLong’s test, both P < 0.01).
Table 5.
Subgroup analysis for performance of models predicting MACEs in patients with and without T2DM
| Model | Feature | T2DM, AUC (95% CI) | No T2DM, AUC (95% CI) | P value* |
|---|---|---|---|---|
| Model 2 | Age ≥ 65 years + Male + hypertension | 0.60 (0.57–0.63) | 0.64 (0.59–0.70) | 0.23 |
| Model 3 | Model 2 + BNP | 0.63 (0.59–0.66) | 0.72 (0.66–0.78) | 0.009 |
| Model 4 | Model 2 + hs-Tnl | 0.64 (0.61–0.67) | 0.72 (0.66–0.78) | 0.016 |
| Model 5 | Model 2 + hs-Tnl + BNP | 0.64 (0.61–0.68) | 0.75 (0.69–0.81) | <0.001 |
BNP and hs-Tnl are at levels above optimal cutoffs
AUC area under the curve, BNP B-type natriuretic peptide, CI confidence interval, hs-Tnl high-sensitivity troponin-I, T2DM type 2 diabetes mellitus
*P value from DeLong’s test for difference between AUCs of T2DM versus no T2DM
Fig. 3.
Area under the receiver operating characteristic curves for predicting MACEs according to T2DM status. BNP B-type natriuretic peptide, hs-Tnl high-sensitivity troponin I, T2DM type 2 diabetes mellitus
Adding a combination of hs-Tnl and BNP to the risk factor model further increased the AUC to 0.75 (95% CI 0.69–0.81), incremental benefit was observed in patients without T2DM when compared with the single biomarker model (Model 5 versus Model 4: DeLong’s test, P = 0.047). In contrast, among patients with T2DM, the combination of two cardiac biomarkers offered no significant incremental value over the single biomarker model in the risk prediction of MACE (Model 5 versus Model 4: DeLong’s test, P = 0.45).
Discussion
Main findings
In this cohort study of 2275 patients with stable CAD, higher baseline levels of hs-Tnl and BNP were strongly associated with subsequent risk of MACE. Baseline hs-Tnl and BNP level were independent predictors of incident MACE in both patients with and without T2DM, and remained significant after adjustment for age and clinical risk factors. The addition of each biomarker to the age and clinical risk factor model offered individual incremental benefit over conventional risk factors for predicting MACE. Among patients without T2DM, these two cardiac biomarkers, alone and in combination, provided greater improvements in risk prediction than in those with T2DM. It is of note that the incremental benefit of a combination of the two cardiac biomarkers over individual cardiac biomarker model of hs-Tnl was observed only in patients without T2DM. Our findings confirm the incremental prognostic value of hs-Tnl and BNP beyond conventional risk factors, and the superior predictive performance of the combined cardiac biomarkers models in patients with stable CAD without T2DM.
Predictive values of cardiac biomarkers for MACE
In this study, we examined the prognostic value of two cardiac biomarkers that reflect different pathophysiological mechanisms, including myocardial injury (hs-Tnl) and wall stress (BNP) for prediction of MACE. Prior studies have shown that hs-Tnl is a highly specific marker for cardiac damage and is associated with increased risk of cardiovascular events and mortality in the elderly and patients with chest pain [7, 9, 28, 29]. Our recent study also demonstrated that elevated hs-Tnl was associated with increased MACE in CAD patients with statin intolerance [30]. Moreover, BNP has been shown to be an important prognostic marker of left ventricular dysfunction as well as a predictor of cardiovascular morbidity and mortality in patients with stable vascular disease [31, 32]. Previous studies have demonstrated that plasma BNP level is increased in the presence of heart failure, myocardial infarction, left ventricular hypertrophy and diabetes [33–35]. In patients with prior ACS, recent studies [36] show that elevated levels of cardiac troponin and BNP are independently associated with higher risk of recurrent cardiovascular events. Similarly, these biomarkers have shown to be an independent prognostic marker for cardiovascular events and all-cause mortality in patients with stable CAD [37, 38].
In this study, we confirmed these findings and demonstrated that elevated hs-Tnl and BNP level was associated with increased risk of MACE in CAD patients with or without T2DM. The annual incidence rate of MACE was 8.0% among patients with elevated levels of hs-Tnl and BNP at baseline, compared with 3.0% among those without elevated cardiac biomarkers, supporting the utility of hs-Tnl and BNP as prognostic markers for future cardiovascular events in patients with stable CAD. Moreover, we performed a direct comparison of cardiac biomarkers for risk prediction of MACE in these patients. Among them, elevated hs-Tnl level provided a higher AUC and was associated with 1.9-fold increased risk of MACE, whereas a slightly lower AUC was observed for elevated BNP with an association of 1.6-fold increased risk.
Predictive values of cardiac biomarkers in CAD patient with or without T2DM
It is well known that T2DM is a predictor of elevated hs-Tnl [18, 19]. On the other hand, an elevated hs-TnI level was associated with the occurrence of MACE in patients with T2DM [30, 39]. Similarly, elevated BNP was associated with adverse cardiovascular outcomes in patients with T2DM [40]. Moreover, in T2DM patients with recent ACS, elevated BNP level was predictive of MACE [41]. Nevertheless, the predictive values of these biomarkers in patients with stable CAD and T2DM remains unclear.
To the best of our knowledge, this is the first study to evaluate whether the combined cardiac biomarkers approach incorporating hs-Tnl and BNP can provide incremental benefit in risk prediction of MACE in stable CAD patients with and without T2DM. In the present study, patients with T2DM were older and had a higher body mass index and prevalence of hypertension than those without T2DM. Furthermore, we observed a higher prevalence of elevated levels of hs-Tnl and BNP in patients with T2DM. Although the BNP concentration was significantly higher in T2DM patients, there was no significant difference in hs-Tnl level between patients with and without T2DM. Both elevated hs-Tnl and BNP were independently associated with the development of MACE among patients with and without T2DM. Interestingly, the predictive values of these cardiac biomarkers were stronger among CAD patients without T2DM. Our results revealed a more than twofold higher risk of MACE in patients without T2DM with elevated levels of hs-Tnl and BNP compared with patients with values below the cutoff level (Additional file 1: Table S3). Indeed, the prognostic value of these cardiac biomarkers was significantly higher in patients without T2DM than those with T2DM, after controlling for clinical risk factors.
Multi-biomarker approach in CAD with or without T2DM
The multi-biomarker approach could be useful for stratification of high risk population for intensity risk factors control such as lipid lowering therapies [3, 4]. Prior studies [26] revealed that the combination of troponin and BNP provide the best prediction for cardiovascular events or death in patients with T2DM. Nevertheless, the predictive performance of these multi-biomarkers was not found to be better than the BNP alone for MACE. Indeed, we demonstrated that a combination of hs-TnI and BNP offered greater incremental value in risk prediction compared with the single biomarker models only in patients without T2DM. Furthermore, this multiple cardiac biomarker approach in patients without T2DM also provided significantly superior predictive power than in patients with T2DM, both for a single and combination of biomarkers approach. The overall effect of the proposed approach in T2DM patients follows the same trend as in those without T2DM but with a smaller magnitude as in previous studies [26]. The reasons for the lack of incremental predictive values of combined cardiac biomarkers in CAD patients with T2DM is unknown.
First, it is possible that similar mechanisms, such as microvascular injuries contribute to elevated hs-TnI and BNP in CAD and T2DM, and thus limited the incremental predictive values in using both biomarkers. In animal model of myocardial injury induced by carbon monoxide, there was a positive correlation between the levels of troponin and BNP [42]. Moreover, the levels of hs-Tnl and BNP are closely correlated with each other after myocardial infarction, and high levels of troponin may reflect the severity of heart failure [43]. Second, this may be due to factors such as renal impairment and hyperglycemia that contribute to both elevated hs-Tnl and BNP in patients with T2DM [44, 45]. Nevertheless, recent studies demonstrated that perioperative glycemic control with exenatide infusion does not affect troponin and BNP release after cardiac surgery [46]. Third, the presence of T2DM might induce different protective mechanisms, such as a more favorable lipoprotein prolife associated with elevated BNP via adiponectin signaling as cardioprotective effects [47]; and modifying myocardial response to ischemia/reperfusion mediated by dynamin-related protein 1 mitochondrial fission [48].
Study limitations
This study has several limitations. First, there are no data that allow us to adjust for the severity of CAD, control of cardiovascular risk factors, such as blood pressure, smoking and lipid level, medications use and diabetes-related factors such as severity, glycemic controls and duration of diabetes that are known to influence the level of hs-Tnl and BNP [49, 50]. Second, we cannot conclude whether the biomarkers are causally related to the development of cardiovascular events. Third, in this study, other traditional risk factors, such as high-sensitive C-reactive protein and renal function were not included. Forth, it remains unclear whether repeated measurement of BNP and hs-TnI can provide further incremental value for prediction of MACE. Final, validation studies are needed to confirm our findings in CAD patients with and without T2DM.
Conclusions
Elevated hs-Tnl and BNP levels are independent predictors of new-onset MACE in patients with stable CAD, the prognostic values of which are greater in patients without T2DM. Among patients without T2DM, risk factor model incorporating clinical risk factor and combination of biomarkers yielded the greatest predictive value. These results support the rationale of utilizing the multiple cardiac biomarkers approach as a prognostic tool for risk stratification in high-risk patients, and may contribute to the optimization of patient treatment and outcomes.
Supplementary information
Additional file 1: Table S1. Unadjusted and age-adjusted Cox regression analysis predicting MACEs in stable CAD patients. Table S2. Competing risks regression models predicting MACEs in stable CAD patients. Table S3. Multivariable Cox regression models predicting MACEs in patients with and without T2DM.
Acknowledgements
Not applicable.
Abbreviations
- ACS
acute coronary syndrome
- AUC
area under the curve
- BNP
B-type natriuretic peptide
- CAD
coronary artery disease
- CI
confidence interval
- hs-Tnl
high-sensitivity troponin I
- HR
hazard ratio
- IDI
integrated discrimination improvement
- MACE
major adverse cardiovascular event
- NRI
net reclassification index
- T2DM
type 2 diabetes mellitus
Authors’ contributions
YKW contributed to the analysis and interpretation of data, and manuscript writing. CYYC, CST, JSHH, CHL, KKL, KWA, BMYC, PCS and AX contributed to the study concept and acquisition of data. KSLL and HFT contributed equally to the supervision of this work and are co-corresponding authors. All authors read and approved the finial manuscript.
Funding
This study was supported by the Hong Kong Research Grant Council: Theme Based Research Scheme (T12-705/11) and Collaborative Research Fund (HKU2/CRF/12R).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster. Written informed consent was obtained from all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Karen S. L. Lam and Hung-Fat Tse contributed equally to the supervision of this work and are co-corresponding authors
Contributor Information
Karen S. L. Lam, Email: ksllam@hku.hk
Hung-Fat Tse, Email: hftse@hkucc.hku.hk.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12933-019-0974-2.
References
- 1.Wilson PW, D’Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation. 1998;97(18):1837–1847. doi: 10.1161/01.CIR.97.18.1837. [DOI] [PubMed] [Google Scholar]
- 2.Goff DC, Jr, Lloyd-Jones DM, Bennett G, Coady S, D’Agostino RB, Gibbons R, Greenland P, Lackland DT, Levy D, O’Donnell CJ, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S49–S73. doi: 10.1161/01.cir.0000437741.48606.98. [DOI] [PubMed] [Google Scholar]
- 3.Prugger C, Luc G, Haas B, Morange PE, Ferrieres J, Amouyel P, Kee F, Ducimetiere P, Empana JP, Group PS Multiple biomarkers for the prediction of ischemic stroke: the PRIME study. Arterioscler Thromb Vasc Biol. 2013;33(3):659–666. doi: 10.1161/ATVBAHA.112.300109. [DOI] [PubMed] [Google Scholar]
- 4.Wong YK, Cheung CYY, Tang CS, Au KW, Hai JSH, Lee CH, Lau KK, Cheung BMY, Sham PC, Xu A, et al. Age-biomarkers-clinical risk factors for prediction of cardiovascular events in patients with coronary artery disease. Arterioscler Thromb Vasc Biol. 2018;38(10):2519–2527. doi: 10.1161/ATVBAHA.118.311726. [DOI] [PubMed] [Google Scholar]
- 5.Samman Tahhan A, Sandesara P, Hayek SS, Hammadah M, Alkhoder A, Kelli HM, Topel M, O’Neal WT, Ghasemzadeh N, Ko YA, et al. High-sensitivity troponin I levels and coronary artery disease severity, progression, and long-term outcomes. J Am Heart Assoc. 2018;7(5):e007914. doi: 10.1161/JAHA.117.007914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eggers KM, Lind L, Ahlstrom H, Bjerner T, Ebeling Barbier C, Larsson A, Venge P, Lindahl B. Prevalence and pathophysiological mechanisms of elevated cardiac troponin I levels in a population-based sample of elderly subjects. Eur Heart J. 2008;29(18):2252–2258. doi: 10.1093/eurheartj/ehn327. [DOI] [PubMed] [Google Scholar]
- 7.Eggers KM, Venge P, Lindahl B, Lind L. Cardiac troponin I levels measured with a high-sensitive assay increase over time and are strong predictors of mortality in an elderly population. J Am Coll Cardiol. 2013;61(18):1906–1913. doi: 10.1016/j.jacc.2012.12.048. [DOI] [PubMed] [Google Scholar]
- 8.Keller T, Zeller T, Peetz D, Tzikas S, Roth A, Czyz E, Bickel C, Baldus S, Warnholtz A, Frohlich M, et al. Sensitive troponin I assay in early diagnosis of acute myocardial infarction. N Engl J Med. 2009;361(9):868–877. doi: 10.1056/NEJMoa0903515. [DOI] [PubMed] [Google Scholar]
- 9.Januzzi JL, Jr, Suchindran S, Hoffmann U, Patel MR, Ferencik M, Coles A, Tardif JC, Ginsburg GS, Douglas PS, Investigators P. Single-Molecule hsTnI and short-term risk in stable patients with chest pain. J Am Coll Cardiol. 2019;73(3):251–260. doi: 10.1016/j.jacc.2018.10.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.White HD, Tonkin A, Simes J, Stewart R, Mann K, Thompson P, Colquhoun D, West M, Nestel P, Sullivan D, et al. Association of contemporary sensitive troponin I levels at baseline and change at 1 year with long-term coronary events following myocardial infarction or unstable angina: results from the LIPID Study (Long-Term Intervention With Pravastatin in Ischaemic Disease) J Am Coll Cardiol. 2014;63(4):345–354. doi: 10.1016/j.jacc.2013.08.1643. [DOI] [PubMed] [Google Scholar]
- 11.Cavender MA, White WB, Jarolim P, Bakris GL, Cushman WC, Kupfer S, Gao Q, Mehta CR, Zannad F, Cannon CP, et al. Serial Measurement of high-sensitivity troponin I and cardiovascular outcomes in patients with type 2 diabetes mellitus in the EXAMINE trial (Examination of Cardiovascular Outcomes With Alogliptin Versus Standard of Care) Circulation. 2017;135(20):1911–1921. doi: 10.1161/CIRCULATIONAHA.116.024632. [DOI] [PubMed] [Google Scholar]
- 12.Maisel AS, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, Omland T, Storrow AB, Abraham WT, Wu AH, et al. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med. 2002;347(3):161–167. doi: 10.1056/NEJMoa020233. [DOI] [PubMed] [Google Scholar]
- 13.Koglin J, Pehlivanli S, Schwaiblmair M, Vogeser M, Cremer P, vonScheidt W. Role of brain natriuretic peptide in risk stratification of patients with congestive heart failure. J Am Coll Cardiol. 2001;38(7):1934–1941. doi: 10.1016/S0735-1097(01)01672-2. [DOI] [PubMed] [Google Scholar]
- 14.Wang F, Wu Y, Tang L, Zhu W, Chen F, Xu T, Bo L, Li J, Deng X. Brain natriuretic peptide for prediction of mortality in patients with sepsis: a systematic review and meta-analysis. Crit Care. 2012;16(3):R74. doi: 10.1186/cc11331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Glaser R, Peacock WF, Wu AH, Muller R, Mockel M, Apple FS. Placental growth factor and B-type natriuretic peptide as independent predictors of risk from a multibiomarker panel in suspected acute coronary syndrome (Acute Risk and Related Outcomes Assessed With Cardiac Biomarkers [ARROW]) study. Am J Cardiol. 2011;107(6):821–826. doi: 10.1016/j.amjcard.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 16.Schnabel R, Lubos E, Rupprecht HJ, Espinola-Klein C, Bickel C, Lackner KJ, Cambien F, Tiret L, Munzel T, Blankenberg S. B-type natriuretic peptide and the risk of cardiovascular events and death in patients with stable angina: results from the AtheroGene study. J Am Coll Cardiol. 2006;47(3):552–558. doi: 10.1016/j.jacc.2005.09.039. [DOI] [PubMed] [Google Scholar]
- 17.Doust JA, Pietrzak E, Dobson A, Glasziou P. How well does B-type natriuretic peptide predict death and cardiac events in patients with heart failure: systematic review. BMJ. 2005;330(7492):625. doi: 10.1136/bmj.330.7492.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhong B, Wang Y, Zhang G. History of diabetes mellitus is associated with elevated cardiac troponin I levels in patients with chest pain but no coronary heart disease. Cardiol J. 2016;23(2):149–154. doi: 10.5603/CJ.a2015.0072. [DOI] [PubMed] [Google Scholar]
- 19.Verges B, Zeller M, Desgres J, Dentan G, Laurent Y, Janin-Manificat L, L’Huillier I, Rioufol G, Beer JC, Makki H, et al. High plasma N-terminal pro-brain natriuretic peptide level found in diabetic patients after myocardial infarction is associated with an increased risk of in-hospital mortality and cardiogenic shock. Eur Heart J. 2005;26(17):1734–1741. doi: 10.1093/eurheartj/ehi260. [DOI] [PubMed] [Google Scholar]
- 20.van der Horst IC, de Boer RA, Hillege HL, Boomsma F, Voors AA, van Veldhuisen DJ. Neurohormonal profile of patients with heart failure and diabetes. Neth Heart J. 2010;18(4):190–196. doi: 10.1007/BF03091760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Goff DC, Jr, Lloyd-Jones DM, Bennett G, Coady S, D’Agostino RB, Sr, Gibbons R, Greenland P, Lackland DT, Levy D, O’Donnell CJ, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63(25 Pt B):2935–2959. doi: 10.1016/j.jacc.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Buuren S, Groothuis-Oudshoorn K. MICE: Multivariate imputation by chained equations in R. J Stat Softw. 2011;45(3):1–68. doi: 10.18637/jss.v045.i03. [DOI] [Google Scholar]
- 23.Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15(7):539–553. doi: 10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 24.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. doi: 10.1080/01621459.1999.10474144. [DOI] [Google Scholar]
- 25.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–845. doi: 10.2307/2531595. [DOI] [PubMed] [Google Scholar]
- 26.Hillis GS, Welsh P, Chalmers J, Perkovic V, Chow CK, Li Q, Jun M, Neal B, Zoungas S, Poulter N, et al. The relative and combined ability of high-sensitivity cardiac troponin T and N-terminal pro-B-type natriuretic peptide to predict cardiovascular events and death in patients with type 2 diabetes. Diabetes Care. 2014;37(1):295–303. doi: 10.2337/dc13-1165. [DOI] [PubMed] [Google Scholar]
- 27.Vittinghoff E, McCulloch CE. Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol. 2007;165(6):710–718. doi: 10.1093/aje/kwk052. [DOI] [PubMed] [Google Scholar]
- 28.Zhu K, Knuiman M, Divitini M, Murray K, Lim EM, St John A, Walsh JP, Hung J. High-sensitivity cardiac troponin I and risk of cardiovascular disease in an Australian population-based cohort. Heart. 2018;104(11):895–903. doi: 10.1136/heartjnl-2017-312093. [DOI] [PubMed] [Google Scholar]
- 29.Omland T, Pfeffer MA, Solomon SD, de Lemos JA, Rosjo H, Saltyte Benth J, Maggioni A, Domanski MJ, Rouleau JL, Sabatine MS, et al. Prognostic value of cardiac troponin I measured with a highly sensitive assay in patients with stable coronary artery disease. J Am Coll Cardiol. 2013;61(12):1240–1249. doi: 10.1016/j.jacc.2012.12.026. [DOI] [PubMed] [Google Scholar]
- 30.Hai JJ, Wong YK, Wong CK, Un KC, Chan PH, Siu CW, Yiu KH, Lau CP, Tse HF. Prognostic implications of statin intolerance in stable coronary artery disease patients with different levels of high-sensitive troponin. BMC Cardiovasc Disord. 2019;19(1):168. doi: 10.1186/s12872-019-1152-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tsutamoto T, Wada A, Maeda K, Hisanaga T, Mabuchi N, Hayashi M, Ohnishi M, Sawaki M, Fujii M, Horie H, et al. Plasma brain natriuretic peptide level as a biochemical marker of morbidity and mortality in patients with asymptomatic or minimally symptomatic left ventricular dysfunction. Comparison with plasma angiotensin II and endothelin-1. Eur Heart J. 1999;20(24):1799–1807. doi: 10.1053/euhj.1999.1746. [DOI] [PubMed] [Google Scholar]
- 32.Wright GA, Struthers AD. Natriuretic peptides as a prognostic marker and therapeutic target in heart failure. Heart. 2006;92(2):149–151. doi: 10.1136/hrt.2003.018325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mukoyama M, Nakao K, Hosoda K, Suga S, Saito Y, Ogawa Y, Shirakami G, Jougasaki M, Obata K, Yasue H, et al. Brain natriuretic peptide as a novel cardiac hormone in humans. Evidence for an exquisite dual natriuretic peptide system, atrial natriuretic peptide and brain natriuretic peptide. J Clin Invest. 1991;87(4):1402–1412. doi: 10.1172/JCI115146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morita E, Yasue H, Yoshimura M, Ogawa H, Jougasaki M, Matsumura T, Mukoyama M, Nakao K. Increased plasma levels of brain natriuretic peptide in patients with acute myocardial infarction. Circulation. 1993;88(1):82–91. doi: 10.1161/01.CIR.88.1.82. [DOI] [PubMed] [Google Scholar]
- 35.Hasegawa K, Fujiwara H, Doyama K, Miyamae M, Fujiwara T, Suga S, Mukoyama M, Nakao K, Imura H, Sasayama S. Ventricular expression of brain natriuretic peptide in hypertrophic cardiomyopathy. Circulation. 1993;88(2):372–380. doi: 10.1161/01.CIR.88.2.372. [DOI] [PubMed] [Google Scholar]
- 36.Qamar A, Giugliano RP, Bohula EA, Park JG, Jarolim P, Murphy SA, Blazing MA, Califf RM, Cannon CP, Braunwald E, et al. Biomarkers and Clinical cardiovascular outcomes with Ezetimibe in the IMPROVE-IT Trial. J Am Coll Cardiol. 2019;74(8):1057–1068. doi: 10.1016/j.jacc.2019.06.038. [DOI] [PubMed] [Google Scholar]
- 37.Tonkin AM, Blankenberg S, Kirby A, Zeller T, Colquhoun DM, Funke-Kaiser A, Hague W, Hunt D, Keech AC, Nestel P, et al. Biomarkers in stable coronary heart disease, their modulation and cardiovascular risk: the LIPID biomarker study. Int J Cardiol. 2015;201:499–507. doi: 10.1016/j.ijcard.2015.07.080. [DOI] [PubMed] [Google Scholar]
- 38.Lyngbaek S, Winkel P, Gotze JP, Kastrup J, Gluud C, Kolmos HJ, Kjoller E, Jensen GB, Hansen JF, Hildebrandt P, et al. Risk stratification in stable coronary artery disease is possible at cardiac troponin levels below conventional detection and is improved by use of N-terminal pro-B-type natriuretic peptide. Eur J Prev Cardiol. 2014;21(10):1275–1284. doi: 10.1177/2047487313492099. [DOI] [PubMed] [Google Scholar]
- 39.Yiu K-H, Lau K-K, Zhao C-T, Chan Y-H, Chen Y, Zhen Z, Wong A, Lau C-P, Tse H-F. Predictive value of high-sensitivity troponin-I for future adverse cardiovascular outcome in stable patients with type 2 diabetes mellitus. Cardiovasc Diabetol. 2014;13(1):63. doi: 10.1186/1475-2840-13-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jarolim P, White WB, Cannon CP, Gao Q, Morrow DA. Serial measurement of natriuretic peptides and cardiovascular outcomes in patients with type 2 diabetes in the EXAMINE Trial. Diabetes Care. 2018;41(7):1510–1515. doi: 10.2337/dc18-0109. [DOI] [PubMed] [Google Scholar]
- 41.Wolsk E, Claggett B, Pfeffer MA, Diaz R, Dickstein K, Gerstein HC, Lawson FC, Lewis EF, Maggioni AP, McMurray JJV, et al. Role of B-type natriuretic peptide and N-terminal prohormone BNP as predictors of cardiovascular morbidity and mortality in patients with a recent coronary event and type 2 diabetes mellitus. J Am Heart Assoc. 2017;6(6):e004743. doi: 10.1161/JAHA.116.004743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yucel M, Avsarogullari L, Durukan P, Akdur O, Ozkan S, Sozuer E, Muhtaroglu S, Ikizceli I, Yurumez Y. BNP shows myocardial injury earlier than Troponin-I in experimental carbon monoxide poisoning. Eur Rev Med Pharmacol Sci. 2016;20(6):1149–1154. [PubMed] [Google Scholar]
- 43.Islam MN, Alam MF, Debnath RC, Aditya GP, Ali MH, Hossain MA, Siddique SR. Correlation between troponin-I and B-type natriuretic peptide level in acute myocardial infarction patients with heart failure. Mymensingh Med J. 2016;25(2):226–231. [PubMed] [Google Scholar]
- 44.Rubin J, Matsushita K, Ballantyne CM, Hoogeveen R, Coresh J, Selvin E. Chronic hyperglycemia and subclinical myocardial injury. J Am Coll Cardiol. 2012;59(5):484–489. doi: 10.1016/j.jacc.2011.10.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dal K, Ata N, Yavuz B, Sen O, Deveci OS, Aksoz Z, Yildirim AM, Uygungelen B, Akin KO, Beyan E, et al. The relationship between glycemic control and BNP levels in diabetic patients. Cardiol J. 2014;21(3):252–256. doi: 10.5603/CJ.a2013.0109. [DOI] [PubMed] [Google Scholar]
- 46.Besch G, Perrotti A, du Salomon Mont L, Puyraveau M, Ben-Said X, Baltres M, Barrucand B, Flicoteaux G, Vettoretti L, Samain E, et al. Impact of intravenous exenatide infusion for perioperative blood glucose control on myocardial ischemia-reperfusion injuries after coronary artery bypass graft surgery: sub study of the phase II/III ExSTRESS randomized trial. Cardiovasc Diabetol. 2018;17(1):140. doi: 10.1186/s12933-018-0784-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Masuch A, Pietzner M, Bahls M, Budde K, Kastenmuller G, Zylla S, Artati A, Adamski J, Volzke H, Dorr M, et al. Metabolomic profiling implicates adiponectin as mediator of a favorable lipoprotein profile associated with NT-proBNP. Cardiovasc Diabetol. 2018;17(1):120. doi: 10.1186/s12933-018-0765-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ding M, Dong Q, Liu Z, Liu Z, Qu Y, Li X, Huo C, Jia X, Fu F, Wang X. Inhibition of dynamin-related protein 1 protects against myocardial ischemia-reperfusion injury in diabetic mice. Cardiovasc Diabetol. 2017;16(1):19. doi: 10.1186/s12933-017-0501-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Brunstrom M, Carlberg B. Association of blood pressure lowering with mortality and cardiovascular disease across blood pressure levels: a systematic review and meta-analysis. JAMA Intern Med. 2018;178(1):28–36. doi: 10.1001/jamainternmed.2017.6015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rezende PC, Everett BM, Brooks MM, Vlachos H, Orchard TJ, Frye RL, Bhatt DL, Hlatky MA. Hypoglycemia and elevated troponin in patients with diabetes and coronary artery disease. J Am Coll Cardiol. 2018;72(15):1778–1786. doi: 10.1016/j.jacc.2018.07.067. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Unadjusted and age-adjusted Cox regression analysis predicting MACEs in stable CAD patients. Table S2. Competing risks regression models predicting MACEs in stable CAD patients. Table S3. Multivariable Cox regression models predicting MACEs in patients with and without T2DM.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.