Abstract
Background:
Patients with systemic lupus erythematosus (SLE) have a similar risk of myocardial infarction as those with diabetes mellitus (DM). Whether the risk of heart failure (HF) in SLE is similar to the elevated risk in DM is unknown. We sought to estimate the rates and risks for HF hospitalization among US Medicaid patients with SLE and to compare them to those for DM and the general Medicaid population.
Methods:
Using U.S. Medicaid data from 2007–2010, we identified patients with SLE or DM, and a matched cohort from the general Medicaid population and calculated incidence rates (IR), incidence rate ratios (IRR) and adjusted hazard ratios (HR) of a first HF hospitalization.
Results:
We identified 37,902 SLE (93% female, mean age 40.1±12.1), 76,657 DM (93% female, mean age 40.0±12.1), and 158,695 general Medicaid patients (93% female, mean age 40.2±12.1). The IR per 1000-person years was 6.9 (95% CI 6.3–7.5) for SLE, 6.6 (95% CI 6.2–7.0) for DM, and 1.6 (95% CI 1.5–1.8) for general Medicaid patients. The highest IRR compared to general Medicaid was seen among SLE patients in age group 18–39 (14.7, 95% CI 13.9–15.5). Multivariable-adjusted HRs for HF compared to general Medicaid population were similar for SLE (2.7, 95% CI 2.3–3.1) and DM (3.0, 95% CI 2.6–3.4).
Conclusion:
The incidence of HF among SLE patients was 2.7-fold higher than general Medicaid patients, and similar to DM. Further investigation into the biologic mechanism of HF among SLE compared to non-SLE and DM patients may shed light on the findings of this study.
Keywords: Heart Failure, Inflammation, SLE
Introduction
Atherosclerotic cardiovascular disease (ASCVD) risk and mortality are increased among patients with systemic lupus erythematosus (SLE), compared to patients without SLE1,2. Most studies demonstrating this increased risk have focused on myocardial infarction (MI) and stroke as the primary outcomes3. In contrast, few studies among SLE patients have examined the incidence and risk of heart failure (HF)4,5, which is associated with high morbidity and mortality and is often the end-stage of cardiac disease6.
Along with ischemic heart disease, hypertension, and smoking, type 2 diabetes mellitus (DM) is a strong risk factor for HF7. Patients with DM have been shown to have up to five-fold higher risk of HF compared to those without DM8. Recent studies have revealed that rates of ASCVD are similar among patients with SLE and age- and sex-matched DM patients, and both groups have rates that are higher than the general population9,10. How the risk of HF among SLE patients compares to that among patients with DM and to the general population is unknown.
The aim of this study was to investigate rates and risks of incident HF in patients with SLE compared to two groups: patients with DM and to individuals from the general Medicaid population. We hypothesized that the risk of HF in patients with SLE would be similar to that observed in patients with diabetes, and that both groups would have a substantially higher risk than that observed in the general Medicaid population.
Methods
Data source
We employed data from the Medicaid Analytic eXtract which includes billing claims, demographic information, and medication dispensing data for patients in Medicaid, the U.S. health insurance for low-income individuals. We identified adults of ages 18–65 years residing in the 29 most populated states in the U.S., who were enrolled in Medicaid between January 1, 2007 and December 31, 2010. We excluded patients >65 years old as > 90% are dually enrolled in Medicare and thus not all claims are captured in Medicaid.
Study design and cohort definition
Prevalent SLE was defined as having ≥ 3 International Classification of Diseases, Ninth Revision (ICD-9) codes specific for SLE (710.0) from hospital discharge diagnoses or physician visit claims, each ≥ 30 days apart, an approach used in prior studies11,12. We required a six-month period of continuous enrollment for collection of baseline covariable data prior to the index date, defined as the date on which SLE criteria was met. In the event that the date of the third ICD-9 code for SLE occurred prior to the required six-month continuous enrollment, the next SLE-related claim that would allow for a 6-month baseline period thereafter was selected as the index date.
We identified prevalent type 1 or type 2 DM patients as those having ≥ 3 ICD-9 codes for DM (249.XX, 250.XX, 357.2, 362.0×, 366.41) from hospital discharge diagnoses or physician visit claims each separated by ≥ 30 days, without any claims for SLE13,14. We required 6 months of continuous enrollment prior to the index date as the baseline period. The index date was the date of the third ICD-9 code, or in the event that the third ICD-9 code date occurred before the 6-month baseline period could be established, the next DM-related claim thereafter that would allow for a 6-month baseline period was selected as the index date. Patients who met criteria for both SLE and DM cohorts were not included in either cohorts.
We identified non-SLE and non-DM general Medicaid patients who had ICD-9 codes for any non-SLE, non-DM diagnoses from hospital discharge diagnoses or physician visit claims on the same index date as each SLE patient. We again required a 6-month baseline period of continuous enrollment prior to the index date, and patients with any SLE or DM ICD-9 codes during the baseline period were excluded from the general Medicaid cohort. We used a SAS greedy matching algorithm to identify the first closest match for each SLE patient by sex and age at index date (within 1 month) to two DM and four general Medicaid population patients15. For all three matched cohorts, we subsequently excluded patients with any diagnosis codes for HF (ICD-9 codes 402.01, 402.11, 402.91, 404.01, 404.11, 404.91, 404.03, 404.13, 404.93, and 428.xx, excluding 398.91) during the 6-month baseline period16.
Assessment of covariables
Patient characteristics for all cohorts were collected during the baseline period: age, sex, self-reported race/ethnicity, U.S. region of residence, and zip code-level median household income in quartiles from 2007–2010 U.S. Census data17. In the SLE cohort, lupus nephritis patients were defined as having ≥ 2 ICD-9 hospital discharge diagnoses or physician billing claims for nephritis, proteinuria, and/or renal failure, occurring ≥ 30 days apart, on or after the SLE criteria were met11,18. Among the DM patients, diabetic nephropathy patients were defined as having ≥ 2 ICD-9 hospital discharge or physician billing codes for nephritis, proteinuria, and/or renal failure ≥ 30 days apart on or after DM criteria was met19.
Using ICD-9, Diagnosis Related Group code and/or Current Procedural Terminology codes, we collected covariables in the baseline period, including the number of outpatient physician visits, smoking, obesity, hypertension, diabetes, hyperlipidemia, chronic kidney disease (CKD), thyroid disease (hypo- or hyperthyroidism), and valvular disease13,14,19–25. ASCVD at baseline was defined as the presence of any of the following diagnosis codes during the baseline period: acute MI, old MI, angina, percutaneous coronary intervention, coronary artery bypass graft, coronary atherosclerosis, cerebrovascular accident, peripheral vascular disease, and carotid artery stenosis24,26–30.
We calculated a Charlson comorbidity index for all patients31. We identified filled prescriptions using National Drug Codes and summed the number of unique medications filled per subject during the baseline period. We assessed baseline prescriptions filled for anticoagulants, beta-blockers, statins, and angiotensin converting enzyme inhibitor/angiotensin receptor blockers. We assessed glucocorticoid use of ≥ 10 mg/day of prednisone ever during the baseline period (prednisone, methylprednisolone, dexamethasone, hydrocortisone, prednisolone, and cortisone, defined as prednisone equivalents) during the baseline period.
Outcomes
The primary outcome was first HF hospitalization defined by ICD-9 codes (402.01, 402.11, 402.91, 404.01, 404.11, 404.91, 404.03, 404.13, 404.93, and 428.xx, excluding 398.91) for HF submitted as either primary or secondary hospital discharge diagnosis codes during the observation period16. Patients were followed from index date until first HF hospitalization, death, disenrollment or end of follow-up.
Statistical analysis
We calculated unadjusted first HF incidence rates (IRs) per 1,000 person-years with 95% confidence intervals (CI) using Poisson regression for all cohorts overall and stratified by covariates. We also calculated first HF incidence rate ratios (IRRs) with 95% CI for SLE and DM with general Medicaid as the reference, overall and stratified by covariates. For SLE, we calculated the HF IR after stratifying patients by hydroxychloroquine use versus non-use. We fit multivariable Cox sub-distribution proportional hazards models, calculating cause-specific risk while accounting for the competing risk of death, for incident HF hospitalization among SLE, DM and the general Medicaid cohort using the Fine and Gray model32. As our cohorts were no longer age- and sex-matched after excluding patients with baseline HF, our models were adjusted for age and sex. In our first model, we adjusted for age, sex, and race/ethnicity. We then examined the addition of other covariable in separate models, including region of residence, zip code level median household income, hypertension, obesity, hyperlipidemia, thyroid disease, valvular disease, peri/myocarditis, renal disease (lupus nephritis/diabetic nephropathy/CKD), Charlson comorbidity index, and ASCVD. We included the covariates that were found to be confounders, with significant change of greater than 10% in the effect estimates in the partial models. Our final Cox regression model included age, sex, race/ethnicity, hypertension, lupus nephritis/diabetic nephropathy/CKD, Charlson comorbidity index, and ASCVD.
Data were obtained from Centers for Medicare and Medicaid Services through approved Data Use Agreements and presented in accordance with CMS policies (cell sizes <11 were suppressed). We performed all analyses using SAS version 9.4. The Partners’ Institutional Review Board approved this study.
Results
We identified 40,212 SLE patients, who were matched by age at index date and sex to 80,424 DM patients, and 160,848 general Medicaid patients (Table 1). After excluding patients with baseline history of hospitalization for HF (5.7% for SLE, 4.6% for DM, and 1.3% for general Medicaid), our cohorts consisted of 37,902 SLE patients (93% female, mean age 40.1±12.1), 76,657 DM patients (93% female, mean age 40.0±12.1), and 158,695 general Medicaid patients (93% female, mean age 40.2±12.1). There was a higher proportion of Black patients within the SLE cohort compared to the DM and general Medicaid cohorts, while the general Medicaid cohort had the highest proportion of Hispanic patients. Renal disease due to lupus nephritis was present in 16% of SLE patients and 4% of the DM cohort had diabetic nephropathy. Hypertension, hyperlipidemia, and obesity were more common among patients with DM, while the presence of baseline ASCVD was higher among SLE patients compared to DM (10% vs 8%).
Table 1.
Baseline Characteristics of SLE, DM and General Medicaid cohorts, 2007–2010
| SLE | DM | General Medicaid | |
|---|---|---|---|
| Matched cohort size, after exclusion of baseline heart failure (N) | 37,902 | 76,657 | 158,695 |
| Female (%) | 93 | 93 | 93 |
| Mean age in years (age, SD) | 40.1 (12.1) | 40.0 (12.1) | 40.2 (12.1) |
| Race/Ethnicity | |||
| White (%) | 35 | 46 | 46 |
| Black (%) | 41 | 30 | 21 |
| Hispanic (%) | 16 | 18 | 26 |
| Asian (%) | 3 | 3 | 3 |
| American Indian/Alaskan Native (%) | 1 | 1 | 1 |
| U.S. Region of Residence | |||
| West (%) | 20 | 19 | 29 |
| Northeast (%) | 22 | 20 | 19% |
| South (%) | 38 | 39 | 33% |
| Midwest (%) | 20 | 22 | 19% |
| Lupus nephritis/Diabetic nephropathy/CKD (%) | 16 | 4 | 1% |
| Hypertension (%) | 31 | 37 | 11 |
| Hyperlipidemia (%) | 9 | 22 | 6 |
| Obesity (%) | 4 | 11 | 2 |
| Smoking (%) | 6 | 5 | 4 |
| Valvular disease (%) | 5 | 2 | 1 |
| Presence of ASCVD* (%) | 10 | 8 | 3 |
| Thyroid disease (%) | 7 | 7 | 3 |
| Pericarditis/Myocarditis (%) | 1 | <1 | <1 |
| Mean total number of medications (#, SD) | 9.7 (9.1) | 10.0 (9.1) | 3.4 (5.3) |
| Beta-blocker use (%) | 10 | 10 | 4 |
| ACE inhibitor/ARB use (%) | 18 | 31 | 5 |
| Glucocorticoid use (%) | 41 | 9 | 4 |
| Hydroxychloroquine use (%) | 37 | 0.3 | 0.2 |
| Charlson Comorbidity Index (mean, SD) | 1.7 (1.2) | 1.6 (1.2) | 0.4 (1.2) |
ASCVD: Baseline presence of any atherosclerotic cardiovascular disease by ICD-9 codes for angina, myocardial infarction, old myocardial infraction, percutaneous coronary intervention, coronary artery bypass graft, atherosclerosis, cerebrovascular accident, peripheral vascular disease, carotid stenosis
Abbreviations: ACE inhibitor/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; ASCVD, atherosclerotic cardiovascular disease; CKD, chronic kidney disease; DM, diabetes mellitus; SLE, systemic lupus erythematosus
During a mean follow-up time of 1.9±1.1 years for SLE, 1.8±1.1 years for DM, and 1.6±1.2 for general Medicaid cohort, there were 479 HF hospitalization events for SLE, 921 for DM and 397 for general Medicaid. There were 1,393 (3.7%) deaths for SLE, 1,949 (2.5%) deaths for DM, and 2,947 (1.9%) deaths for the general Medicaid cohort. The IR of HF per 1,000-person years was 6.9 (95% CI 6.3–7.5) among SLE, 6.6 (95% CI 6.2–7.0) among DM and 1.6 (95% CI 1.5–1.8) within general Medicaid (Table 2). The IR among SLE patients in the 18–39 age group was elevated at 6.1 (95% CI 5.3–7.0), compared to 6.2 (95% CI 5.2–7.4) in the 40–49 age group, and 8.7 (95% CI 7.4–10.2) in the 50–65 age group. Among DM patients, the IR increased more dramatically with age compared to the SLE patients, with IR of 2.8 (95% CI 2.4–3.2) in the 18–39 age group, 7.3 (95% CI 6.5–8.2) in the 40–49 age group, and 13.1 (95% CI 12.0–14.4) in the 50–65 age group. The IR of HF among the general Medicaid group remained lower than the IRs of the SLE and DM cohorts in all age groups.
Table 2.
Incidence Rates of First Hospitalizations for Heart Failure among SLE, DM and General Population Patients in Medicaid, stratified by baseline covariables
| Events | Number of Patients | Person-Years | Incidence Rate (95% CI) | |
|---|---|---|---|---|
| All patients | ||||
| SLE | 479 | 37,902 | 70,979 | 6.8 (6.2–7.4) |
| DM | 921 | 76,657 | 142,993 | 6.4 (6.0–6.9) |
| General Medicaid | 397 | 158,695 | 275,498 | 1.4 (1.3–1.6) |
| Age 18–39 | ||||
| SLE | 212 | 18,846 | 34,836 | 6.1 (5.3–7.0) |
| DM | 196 | 38,632 | 71,133 | 2.8 (2.4–3.2) |
| General Medicaid | 54 | 78,614 | 131,291 | 0.4 (0.3–0.5) |
| Age 40–49 | ||||
| SLE | 116 | 9,906 | 18,700 | 6.2 (5.2–7.4) |
| DM | 274 | 19,903 | 37,449 | 7.3 (6.5–8.2) |
| General Medicaid | 96 | 41,633 | 73,875 | 1.3 (1.1–1.6) |
| Age 50–65 | ||||
| SLE | 151 | 9,150 | 17,444 | 8.7 (7.4–10.2) |
| DM | 451 | 18,122 | 34,411 | 13.1 (12.0–14.4) |
| General Medicaid | 247 | 38,448 | 70,332 | 3.5 (3.1–4.0) |
| Sex Female | ||||
| SLE | 431 | 35,118 | 65,897 | 6.5 (6.0–7.2) |
| DM | 839 | 70,952 | 132,536 | 6.3 (5.9–6.8) |
| General Medicaid | 350 | 146,839 | 255,078 | 1.4 (1.2–1.5) |
| Sex Male | ||||
| SLE | 48 | 2,784 | 5,082 | 9.5 (7.1–12.5) |
| DM | 82 | 5,705 | 10,458 | 7.8 (6.3–9.7) |
| General Medicaid | 47 | 11,856 | 20,419 | 2.3 (1.7–3.1) |
| Race/Ethnicity White | ||||
| SLE | 94 | 13,422 | 24,393 | 3.9 (3.2–4.7) |
| DM | 363 | 35,099 | 66,209 | 5.5 (4.9–6.1) |
| General Medicaid | 176 | 73,389 | 130,436 | 1.4 (1.2–1.6) |
| Race/Ethnicity Black | ||||
| SLE | 295 | 15,608 | 29,955 | 10.0 (8.8–11.0) |
| DM | 381 | 22,630 | 42,916 | 8.9 (8.0–9.8) |
| General Medicaid | 169 | 33,160 | 61,763 | 2.7 (2.4–3.2) |
| Race/Ethnicity Hispanic | ||||
| SLE | 59 | 6,245 | 11,561 | 5.1 (4.0–6.6) |
| DM | 126 | 13,439 | 23,812 | 5.3 (4.4–6.3) |
| General Medicaid | 30 | 40,516 | 64,985 | 0.5 (0.3–0.7) |
| Race Ethnicity Asian | ||||
| SLE | - | - | - | 4.4 (2.4–8.1) |
| DM | 20 | 2,201 | 4,049 | 4.9 (3.2–7.7) |
| General Medicaid | - | - | - | 0.5 (0.2–1.3) |
| Race/Ethnicity American Indian/Alaskan Native | ||||
| SLE | - | - | - | 7.0 (2.9–16.8) |
| DM | 13 | 1,096 | 2,023 | 6.4 (3.7–11.1) |
| General Medicaid | - | - | - | 1.4 (0.5–3.7) |
| Without renal disease | ||||
| SLE | 240 | 31,992 | 57,854 | 4.2 (3.7–4.7) |
| DM | 611 | 73,217 | 135,012 | 4.5 (4.2–4.9) |
| General Medicaid | 366 | 157,367 | 273,455 | 1.3 (1.2–1.5) |
| With renal disease | ||||
| SLE | 239 | 5,910 | 13,124 | 18.2 (16.1–20.7) |
| DM | 310 | 3,440 | 7,981 | 38.8 (34.8–43.4) |
| General Medicaid | 31 | 1,328 | 2,043 | 15.2 (10.7–21.6) |
| Without valvular disease | ||||
| SLE | 414 | 35,937 | 67,378 | 6.1 (5.6–6.8) |
| DM | 862 | 75,148 | 140,242 | 6.2 (5.8–6.6) |
| General Medicaid | 371 | 157,046 | 272,760 | 1.4 (1.2–1.5) |
| With valvular disease | ||||
| SLE | 65 | 1,965 | 3,601 | 18.1 (14.2–23.0) |
| DM | 59 | 1,509 | 2,751 | 21.5 (16.6–27.7) |
| General Medicaid | 26 | 1,649 | 2,738 | 9.5 (6.5–14.0) |
| Without ASCVD* | ||||
| SLE | 387 | 34,296 | 64,364 | 6.0 (5.4–6.6) |
| DM | 663 | 70,552 | 131,638 | 5.0 (4.7–5.4) |
| General Medicaid | 327 | 153,620 | 266,926 | 1.2 (1.1–1.4) |
| With ASCVD* | ||||
| SLE | 92 | 3,606 | 6,614 | 13.9 (11.3–17.1) |
| DM | 258 | 6,105 | 11,355 | 22.7 (20.1–25.7) |
| General Medicaid | 70 | 5,075 | 8,572 | 8.2 (6.5–10.3) |
ASCVD: Baseline presence of any atherosclerotic cardiovascular disease by ICD-9 codes for angina, myocardial infarction, old myocardial infraction, percutaneous coronary intervention, coronary artery bypass graft, atherosclerosis, cerebrovascular accident, peripheral vascular disease, carotid stenosis
Abbreviations: ASCVD, atherosclerotic cardiovascular disease; DM, diabetes mellitus; SLE, systemic lupus erythematosus
Cell sizes under 11 are suppressed according to Federal Reporting Requirements.
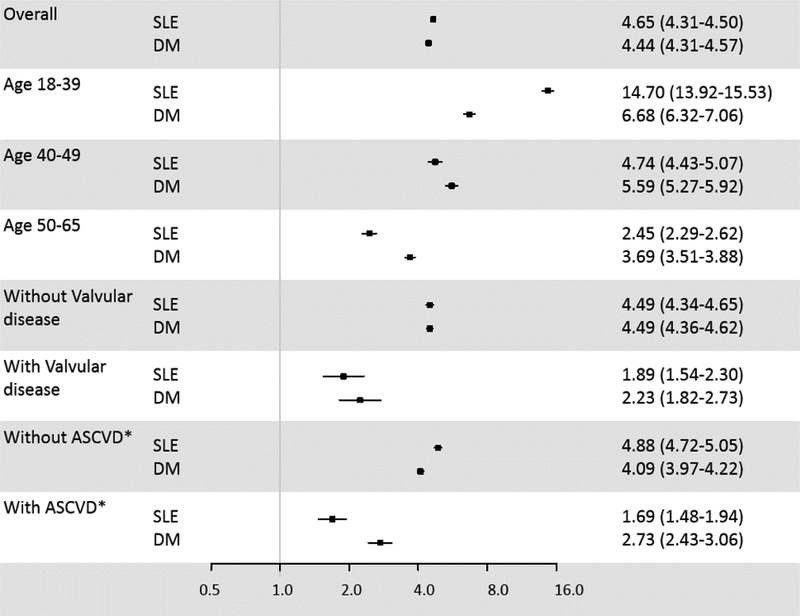
Unadjusted IRR of HF for SLE and DM compared to general Medicaid were similar at 4.65 (95% CI 4.31–4.50) and 4.44 (95% CI 4.31–4.57), respectively (Figure 1). However, in the 18–39 age group, the IRR of HF was 14.70 (95% CI 13.92–15.53) for SLE compared to 6.68 (95% CI 6.32–7.06) for DM, while in the higher age groups, the IRR were slightly higher for DM. The IR of HF were higher in patients with valvular disease for all cohorts (Table 2), but the IRR of SLE and DM compared to general Medicaid were higher in patients without valvular disease compared to those with valvular disease (Figure 1). A similar trend was noted in stratified analysis of patients with and without ASCVD. Among SLE patients, we stratified patients by baseline hydroxychloroquine use and found IR per 1,000 person-years of 6.88 (95% CI 6.14–7.71) for the 23,718 patients without baseline hydroxychloroquine use and 6.54 (95% CI 5.65–7.57) for the 14,184 patients with baseline use of hydroxychloroquine.
Figure 1. Incidence rate ratios (95% CI) of first hospitalization for heart failure for SLE and DM cohorts with general Medicaid as reference, overall and stratified by covariates.
*ASCVD: Baseline presence of any atherosclerotic cardiovascular disease by ICD-9 codes for angina, myocardial infarction, old myocardial infraction, percutaneous coronary intervention, coronary artery bypass graft, atherosclerosis, cerebrovascular accident, peripheral vascular disease, carotid stenosis
Abbreviation: ASCVD, atherosclerotic cardiovascular disease; DM, diabetes mellitus; SLE, systemic lupus erythematosus
In multivariable Cox regression models adjusted for age, sex, and race/ethnicity, the risk of incident HF was greater than four-fold in the SLE [subdistribution hazard ratio (HRSD) 4.2, 95% CI 3.6–4.7] and DM (HRSD 4.4, 95% CI 3.9–4.9) cohorts, with overlapping 95% CIs, compared to the age- and sex-matched general Medicaid patients as reference group (Table 3). When assessed individually in these models, baseline hypertension, renal disease (lupus nephritis/diabetic nephropathy/CKD), Charlson comorbidity index, and ASCVD variables were important confounders or mediators of the relationships between SLE and DM and risk of HF, significantly influencing comparative HRs for incident HF. In our final model, which additionally adjusted for baseline hypertension, renal disease, Charlson comorbidity index and ASCVD, the risk of HF hospitalization remained elevated in both disease groups: HRSD 2.7 (95% CI 2.3–3.1) for SLE and 3.0 (95% CI 2.6–3.4) for DM compared to general Medicaid patients.
Table 3.
Multivariable Hazard Ratios for Hospitalizations for Heart Failure among SLE, DM and General Medicaid Population, 2007–2010
| Cohort | HRSD (95% CI) Model 1 | HRSD (95% CI) Model 2 |
|---|---|---|
| General Medicaid | 1.0 (ref) | 1.0 (ref) |
| SLE | 4.2 (3.6–4.7) | 2.7 (2.3–3.1) |
| Diabetes Mellitus | 4.4 (3.9–4.9) | 3.0 (2.6–3.4) |
HRSD: Hazard ratio for first HF hospitalization event, competing risk analysis taking risk of death into account
Model 1: Adjusted for age, sex, and race/ethnicity
Model 2: Model 1 and additionally adjusted for hypertension, lupus nephritis/diabetic nephropathy/chronic kidney disease, Charlson comorbidity index and atherosclerotic cardiovascular disease
Abbreviations: DM, diabetes mellitus; SLE, systemic lupus erythematosus
Discussion
In this prospective cohort study of nearly 38,000 U.S. Medicaid patients with SLE, we found that the incidence rate of HF hospitalization was more than 4 times higher than among age- and sex-matched general Medicaid patients. Furthermore, SLE patients had a similar risk of HF hospitalization as patients with DM, a disease population with recognized elevated HF risk8. While the overall rate of HF was similar in patients with SLE and DM, the relative increase in HF risk for patients with SLE appeared to be concentrated among patients aged 18–39. In contrast, the relative increase in HF incidence among those with diabetes was evident first for patients in their 40s, and then continued among those aged 50–65. Finally, the increased risk of HF among patients with SLE persisted after adjustment for a number of important potential confounders, such as the Charlson comorbidity index and ASCVD.
Unlike in DM, a disease condition generally well recognized as associated with elevated risk of HF8,33,34, the increased risk of incident HF among SLE patients has not been well studied or recognized. HF has been reported previously to have increased prevalence among SLE patients with ASCVD events2,35–37. Female SLE patients had a 3.8-fold increased likelihood of being hospitalized for HF in a study of California hospital discharges from 1991–19944. A more recent analysis by Kim et al. of SLE patients and controls from 26 commercial U.S. healthcare systems from 1999 to 2016 also revealed that SLE increased the odds of HF by 3.2 fold compared to non-SLE controls5.
The higher relative IR of HF among young SLE patients compared to age-and sex-matched DM and non-SLE patients is consistent with previous findings. In the California study, SLE patients in the 18–44 year age group were 3.8-fold more likely than those without SLE to be hospitalized for HF, compared to 1.39-fold higher for those in the age group 45–64 and 1.25-fold higher for those aged ≥65 years old4. Similarly, in the study by Kim et al., the relative risk of HF was extremely high [49.5 (95% CI 37.3–65.5)] in the youngest age group of 20–24 and the relative gap decreased with increasing age5. These findings are consistent with the current study, in which young SLE patients in the 18–39 age group had over twice the IR of HF than did DM patients of the same age, demonstrating the excess risk among young patients with SLE. These data suggest that patients with SLE are at risk for a condition associated with significant morbidity and mortality at a much younger age than patients from the general population or with DM. By contrast, compared to general Medicaid patients, the IRR of HF for those with DM in the age group 50–65 was higher (HR 3.69, 95% CI 3.51–3.88) than those with SLE in that same age group (HR 2.45, 95% CI 2.29–2.62). The lower risk of HF among older SLE patients may be due to the fact that patients with SLE tend to develop HF at a younger age, and are then excluded from our analysis of older age groups because they have already developed prevalent HF.
In previous studies, IRs of HF in the general population have been demonstrated to be highest among Black patients compared to those of other races38,39. In the current study, the IR for incident HF was indeed highest among Black patients in all three cohorts, including the SLE cohort. As there was substantial variation in the racial make-up of our three cohorts, we adjusted our initial multivariable model for race/ethnicity among other demographic factors and SLE patients were still found to have a 4.2-fold increased risk of incident HF hospitalization compared to the general Medicaid patients, and a similar risk compared to the DM patients.
Known risk factors for HF include hypertension, ischemic heart disease, valvular heart disease, smoking, and obesity, and less common risk factors include myocarditis, which is rare, but is more prevalent among patients with SLE7,33,40–42. However, even after adjusting for covariates that were found to significantly lower the effect estimates for the risk of HF, the HR for incident HF for SLE remained elevated at 2.7, suggesting that these risk factors do not fully explain the increased HF risk in SLE. Renal impairment is also associated with risk of HF43, and was included as a confounder in our final model, which continued to reveal elevated HRs of HF for both SLE and DM.
In the general population, markers of inflammation including tumor necrosis factor-alpha, interleukin-6, and C-reactive protein, are associated with HF, suggesting inflammation may itself contribute to HF pathogenesis44,45. In DM, poor glycemic control has been associated with microvascular disease and increased HF risk46. Cardiac microvascular dysfunction likely plays a role in HF development, and in a prospective population-based study of patients who have had retinal photographs, retinopathy, revealing microvascular disease, has been a predictor of HF47. The etiology of HF in SLE is likely to include myocardial microvascular dysfunction resulting from chronic inflammation, as well as a combination of premature epicardial coronary disease and valvular heart disease48. Furthermore, while hydroxychloroquine use can lead to cardiomyopathy, we did not see a difference in IR of HF in SLE patients stratified by baseline hydroxychloroquine use49. Further research to clarify the pathobiological processes driving HF risk in SLE, and to identify strategies for HF prevention in this vulnerable population, are needed.
The strengths of our study include the large nationwide population of patients studied with SLE with age- and sex-matched comparison groups of DM and the general Medicaid populations. Our analysis included important covariables including race/ethnicity, previously associated with HF. However, although we included many baseline characteristics that are important factors of HF risk, use of administrative claims data may fail to adequately capture several important covariables such as obesity, smoking, and hyperlipidemia. We were also unable to include the presence of antiphospholipid syndrome, which may be particularly important among patients with SLE, although not so among diabetes or in the general population41.
We excluded patients who met criteria for both SLE and DM cohorts. However, our cohort inclusion criteria required ≥3 ICD-9 codes for DM at least 30 days apart, and a small proportion (0.4%) of our SLE patients had used insulin during the baseline period and may have had DM. Other limitations of our study include lack of information regarding disease duration for SLE and DM cohorts and disease activity among SLE patients, as well as inability to accurately assess severity, type (preserved or reduced ejection fraction), and stage of HF diagnosis in claims databases. Additionally, the findings of our study in the Medicaid database may not be generalizable to patients with commercial insurance with variations in access to care.
Conclusions
In this large U.S. Medicaid cohort study, we found that the adjusted risk for incident HF hospitalization among patients with SLE was almost 3-fold higher than general Medicaid patients, and was very similar to that seen among patients with DM. In line with previous reports of elevated rates of MI and stroke in SLE patients, this study demonstrates that patients with SLE are at particularly high risk for HF, similar to patients with DM, and that this risk is concentrated among young patients. Further studies to investigate the underlying mechanisms of HF in SLE, and strategies to target HF prevention in SLE patients are needed.
Acknowledgements
We would like to thank Chang Lu for programming review, and Cameron Speyer for technical review of this article.
Sources of Funding
This work was funded by NIH R01 AR057327 and K24 AR066109 (Dr. K Costenbader).
Disclosures
Dr. SK Chen received research support from T32 AR 7530-33. Dr. Barbhaiya is supported by the Rheumatology Rseearch Foundation Investigator Award. Dr. CH Feldman is supported by NIH K23 AR071500. She also receives research support from BMS and Pfizer for unrelated studies. Dr. K Costenbader has received research support for this study from the NIH R01 AR057327 and K24 AR066109, and research support from Rheumatology Research Foundation, Lupus Foundation of America, Pfizer, Biogen-Idec, Merck, Glaxo Smith Kline. Dr. B Everett’s work on this manuscript was supported by NIH R56 HL134810. He also reports grants and personal fees from Novartis Pharmaceuticals, and personal fees from Amgen, NIDDK, Roche Diagnostics, U.S. FDA, and UpToDate.
REFERENCES
- 1.Hak EA, Karlson EW, Feskanich D, Stampfer MJ, Costenbader KH. Systemic lupus erythematosus and the risk of cardiovascular disease: results from the nurses’ health study. Arthritis Rheum. 2009;61(10):1396–1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartels CM, Buhr KA, Goldberg JW, Bell CL, Visekruna M, Nekkanti S, Greenlee RT. Mortality and cardiovascular burden of systemic lupus erythematosus in a US population-based cohort. J Rheumatol. 2014;41(4):680–687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schoenfeld SR, Kasturi S, Costenbader KH. The epidemiology of atherosclerotic cardiovascular disease among patients with SLE: a systematic review. Semin Arthritis Rheum. 2013;43(1):77–95. [DOI] [PubMed] [Google Scholar]
- 4.Ward MM. Premature morbidity from cardiovascular and cerebrovascular diseases in women with systemic lupus erythematosus. Arthritis Rheum. 1999;42(2):338–346. [DOI] [PubMed] [Google Scholar]
- 5.Kim CH, Al-Kindi SG, Jandali B, Askari AD, Zacharias M, Oliveira GH. Incidence and risk of heart failure in systemic lupus erythematosus. Heart. 2017;103(3):227–233. [DOI] [PubMed] [Google Scholar]
- 6.Benjamin EJ, Virani SS, Callaway CW, Chamberlain AM, Chang AR, Cheng S, Chiuve SE, Cushman M, Delling FN, Deo R, de Ferranti SD, Ferguson JF, Fornage M, Gillespie C, Isasi CR, Jiménez MC, Jordan LC, Judd SE, Lackland D, et al. Heart Disease and Stroke Statistics—2018 Update: A Report From the American Heart Association. Circulation. 2018;137(12):e67–e492. doi: 10.1161/CIR.0000000000000558 [DOI] [PubMed] [Google Scholar]
- 7.He J, Ogden LG, Bazzano LA, Vupputuri S, Loria C, Whelton PK. Risk Factors for Congestive Heart Failure in US Men and Women. Arch Intern Med. 2001;161(7):996. [DOI] [PubMed] [Google Scholar]
- 8.Kannel WB, Hjortland M, Castelli WP. Role of diabetes in congestive heart failure: the Framingham study. Am J Cardiol. 1974;34(1):29–34. [DOI] [PubMed] [Google Scholar]
- 9.Koenig KF, Ribi C, Radosavac M, Zulewski H, Trendelenburg M. Prevalence of vascular disease in systemic lupus erythematosus compared with type-1 diabetes mellitus: a cross-sectional study of two cohorts. Lupus. 2015;24(1):58–65. [DOI] [PubMed] [Google Scholar]
- 10.Barbhaiya M, Feldman CH, Chen SK, Guan H, Lin TC, Fischer MA, Solomon DH, Everett BM CK. Risk of Cardiovascular Disease Events Among Patients with Systemic Lupus Erythematosus Compared to Those with Diabetes Mellitus in a Nationwide Medicaid Cohort [abstract]. Arthritis Rheumatol. 2016;68(Suppl 10). [Google Scholar]
- 11.Feldman CH, Hiraki LT, Liu J, Fischer MA, Solomon DH, Alarcón GS, Winkelmayer WC, Costenbader KH. Epidemiology and sociodemographics of systemic lupus erythematosus and lupus nephritis among US adults with Medicaid coverage, 2000–2004. Arthritis Rheum. 2013;65(3):753–763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bernatsky S, Linehan T, Hanly JG. The accuracy of administrative data diagnoses of systemic autoimmune rheumatic diseases. J Rheumatol. 2011;38(8):1612–1616. [DOI] [PubMed] [Google Scholar]
- 13.Condition Categories - Chronic Conditions Data Warehouse. https://www.ccwdata.org/web/guest/condition-categories. Accessed February 3, 2016.
- 14.Rector TS, Wickstrom SL, Shah M, Thomas Greeenlee N, Rheault P, Rogowski J, Freedman V, Adams J, Escarce JJ. Specificity and sensitivity of claims-based algorithms for identifying members of Medicare+Choice health plans that have chronic medical conditions. Health Serv Res. 2004;39(6 Pt 1):1839–1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Locally Written SAS Macros - Division of Biomedical Statistics and Informatics - Mayo Clinic Research. http://www.mayo.edu/research/departments-divisions/department-health-sciences-research/division-biomedical-statistics-informatics/software/locally-written-sas-macros?
- 16.Chen J, Hsieh AF-C, Dharmarajan K, Masoudi FA, Krumholz HM. National trends in heart failure hospitalization after acute myocardial infarction for Medicare beneficiaries: 1998–2010. Circulation. 2013;128(24):2577–2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Manson S, Schroeder J, Van Riper DRS. IPUMS National Historical Geographic Information System, Version 12.0 [Database]. 2017. [Google Scholar]
- 18.Chibnik L, Massarotti E, Costenbader K. Identification and validation of lupus nephritis cases using administrative data. Lupus. 2010;19(6):741–743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Winkelmayer WC, Schneeweiss S, Mogun H, Patrick AR, Avorn J, Solomon DH. Identification of Individuals With CKD From Medicare Claims Data: A Validation Study. Am J Kidney Dis. 2005;46(2):225–232. [DOI] [PubMed] [Google Scholar]
- 20.Wiley LK, Shah A, Xu H, Bush WS. ICD-9 tobacco use codes are effective identifiers of smoking status. J Am Med Inform Assoc. 2013;20(4):652–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Quan H, Li B, Saunders LD, Parsons GA, Nilsson CI, Alibhai A, Ghali WA, IMECCHI Investigators. Assessing validity of ICD-9-CM and ICD-10 administrative data in recording clinical conditions in a unique dually coded database. Health Serv Res. 2008;43(4):1424–1441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi J-C, Saunders LD, Beck CA, Feasby TE, Ghali WA. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–1139. [DOI] [PubMed] [Google Scholar]
- 23.Tamariz L, Palacio A, Denizard J, Schulman Y, Contreras G. The use of claims data algorithms to recruit eligible participants into clinical trials. Am J Manag Care. 2015;21(2):e114–8. [PubMed] [Google Scholar]
- 24.Birman-Deych E, Waterman AD, Yan Y, Nilasena DS, Radford MJ, Gage BF. Accuracy of ICD-9-CM codes for identifying cardiovascular and stroke risk factors. Med Care. 2005;43(5):480–485. [DOI] [PubMed] [Google Scholar]
- 25.Go AS, Hylek EM, Phillips KA, Chang Y, Henault LE, Selby JV, Singer DE. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA. 2001;285(18):2370–2375. [DOI] [PubMed] [Google Scholar]
- 26.Solomon DH, Schneeweiss S, Glynn RJ, Kiyota Y, Levin R, Mogun H, Avorn J. Relationship Between Selective Cyclooxygenase-2 Inhibitors and Acute Myocardial Infarction in Older Adults. Circulation. 2004;109(17):2068–2073. [DOI] [PubMed] [Google Scholar]
- 27.Varas-Lorenzo C, Castellsague J, Stang MR, Tomas L, Aguado J, Perez-Gutthann S. Positive predictive value of ICD-9 codes 410 and 411 in the identification of cases of acute coronary syndromes in the Saskatchewan Hospital automated database. Pharmacoepidemiol Drug Saf. 2008;17(8):842–852. [DOI] [PubMed] [Google Scholar]
- 28.Liao KP, Liu J, Lu B, Solomon DH, Kim SC. Association between lipid levels and major adverse cardiovascular events in rheumatoid arthritis compared to non-rheumatoid arthritis patients. Arthritis Rheumatol (Hoboken, NJ). 2015;67(8):2004–2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kumamaru H, Judd SE, Curtis JR, Ramachandran R, Hardy NC, Rhodes JD, Safford MM, Kissela BM, Howard G, Jalbert JJ, Brott TG, Setoguchi S. Validity of claims-based stroke algorithms in contemporary Medicare data: reasons for geographic and racial differences in stroke (REGARDS) study linked with medicare claims. Circ Cardiovasc Qual Outcomes. 2014;7(4):611–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fan J, Arruda-Olson AM, Leibson CL, Smith C, Liu G, Bailey KR, Kullo IJ. Billing code algorithms to identify cases of peripheral artery disease from administrative data. J Am Med Inform Assoc. 2013;20(e2):e349–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. [DOI] [PubMed] [Google Scholar]
- 32.Fine JP, Gray RJ. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J Am Stat Assoc. 1999;94(446):496–509. [Google Scholar]
- 33.Ho KK, Pinsky JL, Kannel WB, Levy D. The epidemiology of heart failure: the Framingham Study. J Am Coll Cardiol. 1993;22(4 Suppl A):6A–13A. [DOI] [PubMed] [Google Scholar]
- 34.Nichols GA, Hillier TA, Erbey JR, Brown JB. Congestive heart failure in type 2 diabetes: prevalence, incidence, and risk factors. Diabetes Care. 2001;24(9):1614–1619. [DOI] [PubMed] [Google Scholar]
- 35.Fernández-Nebro A, Rúa-Figueroa Í, López-Longo FJ, Galindo-Izquierdo M, Calvo-Alén J, Olivé-Marqués A, Ordóñez-Cañizares C, Martín-Martínez MA, Blanco R, Melero-González R, Ibáñez-Rúan J, Bernal-Vidal JA, Tomero-Muriel E, Uriarte-Isacelaya E, Horcada-Rubio L, Freire-González M, Narváez J, Boteanu AL, Santos-Soler G, et al. Cardiovascular Events in Systemic Lupus Erythematosus. Medicine (Baltimore). 2015;94(29):e1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pons-Estel GJ, Gonzalez LA, Zhang J, Burgos PI, Reveille JD, Vila LM, Alarcon GS. Predictors of cardiovascular damage in patients with systemic lupus erythematosus: data from LUMINA (LXVIII), a multiethnic US cohort. Rheumatology. 2009;48(7):817–822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Badui E, Garcia-Rubi D, Robles E, Jimenez J, Juan L, Deleze M, Diaz A, Mintz G. Cardiovascular Manifestations in Systemic Lupus Erythematosus. Prospective Study of 100 Patients. Angiology. 1985;36(7):431–441. [DOI] [PubMed] [Google Scholar]
- 38.Bahrami H, Kronmal R, Bluemke DA, Olson J, Shea S, Liu K, Burke GL, Lima JAC. Differences in the incidence of congestive heart failure by ethnicity: the multi-ethnic study of atherosclerosis. Arch Intern Med. 2008;168(19):2138–2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Loehr LR, Rosamond WD, Chang PP, Folsom AR, Chambless LE. Heart Failure Incidence and Survival (from the Atherosclerosis Risk in Communities Study). Am J Cardiol. 2008;101(7):1016–1022. [DOI] [PubMed] [Google Scholar]
- 40.Roldan CA, Shively BK, Crawford MH. An Echocardiographic Study of Valvular Heart Disease Associated with Systemic Lupus Erythematosus. N Engl J Med. 1996;335(19):1424–1430. [DOI] [PubMed] [Google Scholar]
- 41.Khamashta MA, Cervera R, Asherson RA, Hughes GRV, Coltart DJ, Font J, Ingelmo M, Paré C, Gil A, Vázquez JJ, Oliver J. Association of antibodies against phospholipids with heart valve disease in systemic lupus erythematosus. Lancet. 1990;335(8705):1541–1544. [DOI] [PubMed] [Google Scholar]
- 42.Levy D, Larson MG, Vasan RS, Kannel WB, Ho KKL. The Progression From Hypertension to Congestive Heart Failure. JAMA J Am Med Assoc. 1996;275(20):1557. [PubMed] [Google Scholar]
- 43.Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, McCullough PA, Kasiske BL, Kelepouris E, Klag MJ, Parfrey P, Pfeffer M, Raij L, Spinosa DJ, Wilson PW, American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology and E and P. Kidney Disease as a Risk Factor for Development of Cardiovascular Disease: A Statement From the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Circulation. 2003;108(17):2154–2169. [DOI] [PubMed] [Google Scholar]
- 44.Levine B, Kalman J, Mayer L, Fillit HM, Packer M. Elevated Circulating Levels of Tumor Necrosis Factor in Severe Chronic Heart Failure. N Engl J Med. 1990;323(4):236–241. [DOI] [PubMed] [Google Scholar]
- 45.Cesari M, Penninx BWJH, Newman, Kritchevsky, Nicklas, Sutton-Tyrrell, Rubin, Ding, Simonsick, Harris, Pahor. Inflammatory markers and onset of cardiovascular events: results from the Health ABC study. Circulation. 2003;108(19):2317–2322. [DOI] [PubMed] [Google Scholar]
- 46.Iribarren C, Karter AJ, Go AS, Ferrara A, Liu JY, Sidney S, Selby JV. Glycemic control and heart failure among adult patients with diabetes. Circulation. 2001;103(22):2668–2673. [DOI] [PubMed] [Google Scholar]
- 47.Wong TY, Rosamond W, Chang PP, Couper DJ, Sharrett AR, Hubbard LD, Folsom AR, Klein R. Retinopathy and Risk of Congestive Heart Failure. JAMA. 2005;293(1):63. [DOI] [PubMed] [Google Scholar]
- 48.Recio-Mayoral A, Mason JC, Kaski JC, Rubens MB, Harari OA, Camici PG. Chronic inflammation and coronary microvascular dysfunction in patients without risk factors for coronary artery disease. Eur Heart J. 2009;30(15):1837–1843. [DOI] [PubMed] [Google Scholar]
- 49.Tselios K, Deeb M, Gladman DD, Harvey P, Akhtari S, Mak S, Butany J, Urowitz MB. Antimalarial-induced Cardiomyopathy in Systemic Lupus Erythematosus: As Rare as Considered? J Rheumatol. 2019;46(4):391–396. [DOI] [PubMed] [Google Scholar]