Abstract
As a result of societal changes, fathers participate more actively in child care than they used to. In this article, we propose a context‐dependent biobehavioral model of emergent fatherhood in which sociocultural, behavioral, hormonal, and neural factors develop and interact during the first 1,000 days of fatherhood. Sociocultural factors, including different expectations of fathers and varying opportunities for paternal caregiving through paid paternal leave, influence paternal involvement. Levels of hormones (e.g., testosterone, vasopressin, oxytocin, cortisol) predict fathers’ parenting behaviors, and involvement in caregiving in turn affects their hormones and brain responses to infant stimuli. The birth of the first child marks the transition to fatherhood and may be a critical period in men’s lives, with a smoother transition to fatherhood predicting more optimal involvement by fathers in subsequent years. A focus on prenatal and early postnatal fathering may pave the way for developing interventions that effectively support fathering during pregnancy and in the first years of their children’s lives.
Keywords: fathers, parenting, hormones, imaging
Fathers matter. The publication of Michael Lamb’s book, The Role of the Father in Child Development, in 1976 marked growing awareness of fathers’ role in the development of their offspring. Since its publication, fathers’ involvement in childrearing has increased substantially, at least as documented in Western, industrialized countries. In 1970, fathers in four such countries (Belgium, France, the German Democratic Republic, and the United States) worked 50.5 hr per week on average (Roby, 1975). On workdays, they spent an average of 11.8 min on child care (e.g., reading to, playing with, educating, supervising, or traveling with a child), and this doubled to an average of 25.3 min per day on the weekend. As a result of societal changes in these countries, including the increased participation of women and mothers in the labor force, this situation began to change between 1970 and 1980 and men became more active participants in child care. One generation later, in 2010, fathers in five countries (Australia, Denmark, France, Italy, and the United States) spent 34.5 hr per week on average on paid work, and on both weekdays and weekend days, they spent an average of 1.2 hr a day on child care (Craig & Mullan, 2010), a three‐ to six‐fold increase over what their own fathers typically did. Unfortunately, we know less about such changes in non‐Western and less industrialized countries.
Accumulating knowledge points to fathers’ role in early child development and highlights neurobiological changes in the transition to parenthood. In this article, we review this literature from the perspective of a biobehavioral model of emergent fatherhood (see Figure 1), starting from pregnancy with the transition to parenthood that marks the birth of the father through the first few years after birth. We focus on this phase because researchers and policymakers alike consider the first 1,000 days after conception critical for the child’s ability to grow, learn, and thrive (e.g., Berg, 2016; https://thousanddays.org; https://publications.parliament.uk/pa/cm201719/cmselect/cmhealth/1496/1496.pdf). In a similar vein, we suggest that the transition to fatherhood is a critical period in men’s lives, with a smoother transition to fatherhood predicting more optimal involvement of fathers in subsequent years. Longitudinal descriptive and experimental studies are needed to test this hypothesis; here, we propose a model with components essential for such studies (see Figure 1).
Figure 1.
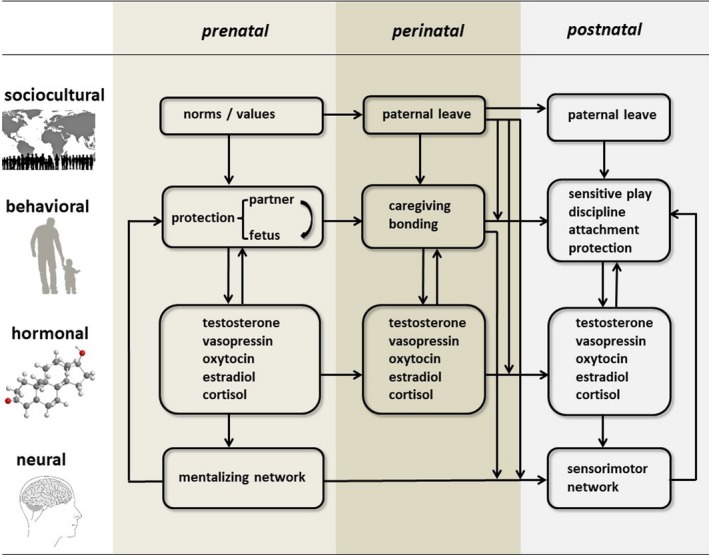
A biobehavioral model of emergent fatherhood.
Note. The transition to fatherhood varies at many levels: sociocultural, behavioral, hormonal, and neural. Bidirectional relations exist between fathering behaviors and hormonal and neural components in the prenatal, perinatal, and postnatal phases. For example, expectations of fathers on the sociocultural level may influence their bonding and involvement in caregiving behaviors, which may in turn influence but may also be influenced by hormonal and neural processes.
A Biobehavioral Model of Emergent Fatherhood
The transition to fatherhood is a major developmental milestone for men. Inspired by Bronfenbrenner’s bioecological perspective (Bronfenbrenner & Ceci, 1994), we propose a model of the transition to fatherhood that considers many levels, including the sociocultural level with respect to different expectations of fathers, childrearing attitudes, and involvement with infants. This level may influence the types of behavior fathers engage in with their infants (see Figure 1). As Figure 1 also shows, we propose bidirectional relations between fathering behaviors and hormonal and neural components in the prenatal, perinatal, and postnatal phases. We discuss each of these areas to show how they affect and are affected by the transition to parenthood. Research has described these transitions in mothers more frequently than in fathers, but the effects of fathering on child development, in and of itself and in interaction with maternal behavior, have also been shown (Dagan & Sagi‐Schwartz, 2018). Therefore, we need a complementary focus on fathers.
Sociocultural Factors
Sociocultural norms and values with regard to fathers’ involvement in child care have changed over time, affecting fathers’ personal norms and behaviors. Along with these changes, paid paternity leave has become more accessible, increasing opportunities for paternal caregiving that in turn may lead to hormonal and neural changes in fathers (see Figure 1). But significant differences in parental leave create disparities in opportunities for fathers’ involvement in infants’ care. Of the 186 countries examined in one study (Heymann & McNeill, 2013), 179 provided paid maternity leave, and 81 countries extended paid leave to new fathers through parental leave that could be taken by either parent, or through paternity leave specific to fathers. The United States is one of few industrialized countries without any statutory national paid family leave provisions for either parent. On the other end of the continuum, Sweden allows parents 540 days of paid parental leave per child, of which 90 days are nontransferable for each parent, with the right to return to employment. Not all fathers use the opportunities for paid leave, and in general, fathers take leave less frequently than mothers.
Why do couples use more maternal leave than paternal leave? First, breastfeeding favors the mother’s proximity to the infant, particularly during the first 6 months of the infant’s life. Second, because of income differences between men and women, it is often economically less feasible for fathers to reduce their working hours than it is for mothers. Third, work‐related and social expectations may push mothers into the role of primary caregivers and fathers into the role of secondary caregivers. Fourth, mothers may be (unconsciously) reluctant to delegate caregiving responsibilities to their partners (maternal gatekeeping; Gaunt, 2008). As a result, fathers spend less than half as much time in direct one‐on‐one interaction with their children as mothers, especially in early childhood (Wood & Repetti, 2004). Although quantity of time invested in parenting is considered less important than quality (Ainsworth, 1967), it takes time to get to know infants, become aware of their preferences, and read their (attachment) signals. Time spent directly responsible for infant care is related linearly to connectivity between parenting‐related brain areas in fathers (Abraham et al., 2014), showing that sociocultural norms that affect paternal involvement (indexing the behavioral level of our model) also affect the neural level indirectly.
Paternal Behavior
For at least two reasons, it is important to realize that fathering starts during pregnancy. First, the prenatal environment has far‐reaching consequences for child development (Glover, O'Donnell, O'Connor, & Fisher, 2018), and fathers can influence that environment positively (e.g., by quitting smoking) and negatively (e.g., by engaging in partner violence). Behaviors that protect the pregnant partner, such as ensuring that she gets sufficient rest and avoids pathogenic foods, protect the infant and benefit the baby’s development. Whether and how interindividual and intraindividual variance in such behaviors is related to hormonal and neural variation, and to variation in postnatal parenting behavior, remains to be determined.
Second, expectant fathers may experience somatic pregnancy symptoms, known as the couvade syndrome, including nausea, leg cramps, appetite and mood changes, and weight gain (Mason & Elwood, 1995). Estimates of incidence range from 11 to 79%, depending on what criteria studies use for inclusion. In nonindustrialized societies, the couvade syndrome may be a ritualization of the transition to fatherhood (e.g., in some cultures, the father remains in bed and is nurtured after the birth of the baby, while the mother resumes work), but it may also be related to typical physiological processes in fathers that eventually lead to parental responsiveness (Mason & Elwood, 1995). In one study, fathers with more couvade symptoms had a greater decrease in testosterone after exposure to infant cues (Storey, Walsh, Quinton, & Wynne‐Edwards, 2000). Thus, the symptoms may be observable phenomena resulting from underlying hormonal changes that also predict dimensions of caregiving. Researchers have not yet related the couvade syndrome to the quality of postnatal caregiving.
In the first year of an infant’s life, establishing an attachment relationship is an important developmental milestone. Although attachment theory has sometimes been criticized for emphasizing the traditional role of mothers as sole caregivers, both Bowlby (1969/1982) and Ainsworth (1967) made explicit that fathers were common and capable attachment figures. In fact, Bowlby argued, based on Harlow’s (1958) experiments with fur and wire rhesus monkey mothers, that (breast‐)feeding was not essential for the infant–parent relationship and that fathers could be capable caregivers of young infants. Indeed, the first study of the Strange Situation Procedure with fathers and mothers showed similar proportions of secure attachment with both parents (Main & Weston, 1981).
Parental responses to infants’ interactive behaviors are generally rated in terms of sensitivity or emotional support. Similar to the pattern of associations for mothers, higher levels of paternal sensitivity predict generally more favorable child outcomes. In correlational and experimental research, mothers’ sensitivity is associated modestly but robustly with secure infant–mother attachment (r = .24–.35; Verhage et al., 2016). For fathers, this meta‐analytic association is weaker (r = .12; Lucassen et al., 2011), with fathers’ observed sensitivity sometimes similar to but often lower than mothers’ sensitivity (e.g., Volling, McElwain, Notaro, & Herrera, 2002). Indeed, it is not uncommon for studies to report that fathers are substantially less sensitive and less involved than mothers, but that similar proportions of children are securely attached to these fathers and mothers (e.g., Lickenbrock & Braungart‐Rieker, 2015). This might suggest that the sensitivity or attachment measures used with mothers are less valid when used with fathers, or that different dimensions of parenting predict infant–father attachment (Grossmann et al., 2002). Given that the intergenerational transmission of attachment is similar in strength for fathers and mothers (Verhage et al., 2016), the search for paternal behavior underlying this transmission should get more attention. Stimulatory play and support of (cognitive) exploration may be paternal behaviors that promote secure infant–father attachment. Limit‐setting has also been suggested as a specific although not exclusive dimension of the father–child relationship (Grossmann et al., 2002).
Just like with mothers’ parenting, fathers’ parenting may be hampered by feelings of depression in the postnatal period. The prevalence of perinatal depression in fathers is 4–10% (Paulson, Bazemore, Goodman, & Leiferman, 2016), and fathers’ depression has been associated with problem behavior in children (Ramchandani, Stein, Evans, & O’Connor, 2005) and subsequent depression in the children themselves (Gutierrez‐Galve et al., 2018). Paternal perinatal depression influences the father–child relationship and is related to less optimal relationships between couples as well as to maternal depression (Paulson et al., 2016), doubling the risk for unfavorable child outcomes. As in mothers, in fathers, hormonal imbalances may be related to postpartum depression (Saxbe et al., 2018), but lack of sufficient sleep may also play a role: 35% of parents with children under age 2 report that they get only 5–6 hr of sleep per night (Krueger & Friedman, 2009). Identifying risk factors for paternal perinatal depression is an important step toward prevention, ideally before the baby is born.
Hormones
When women get pregnant, they experience hormonal changes. Oxytocin levels increase during pregnancy, as do levels of estradiol, testosterone, and cortisol (Edelstein et al., 2017). After a peak in oxytocin and cortisol around childbirth, levels decrease in the postpartum period. Do any hormonal changes prepare men for fatherhood?
Over the course of pregnancy, testosterone and estradiol decline in men, and in one study, men with greater declines were more involved in child care after birth (Edelstein et al., 2017). Indeed, testosterone is generally considered favorable to mating and unfavorable to parenting efforts, and in primary studies (e.g., Gettler, McDade, Feranil, & Kuzawa, 2011) and meta‐analyses, fathers tend to have lower levels of testosterone than nonfathers, but the effect size is modest (r = .11; Meijer, Van IJzendoorn, & Bakermans‐Kranenburg, 2019); this is probably because downregulation of testosterone levels depends on fathers’ actual involvement in child care and the presence or absence of other competitive demands. In one study, fathers’ lower basal testosterone in the immediate postnatal period predicted more involvement in child care 2–4 months later (Kuo et al., 2018). Fathers with lower basal testosterone levels tend to engage in higher‐quality parenting (meta‐analytic effect size r = .07; Meijer et al., 2019). However, testosterone may prepare fathers for caregiving; for example, exposure to cry stimuli increases fathers’ testosterone levels (Fleming, Corter, Stallings, & Steiner, 2002; Van Anders, Tolman, & Volling, 2012).
Monogamous male prairie voles have elevated levels of the hormone vasopressin after mating, leading to territoriality and partner protection (Winslow, Hastings, Carter, Harbaugh, & Insel, 1993). Similar preparatory mechanisms, including enhanced sensitivity to vasopressin, may be found in humans. Administering vasopressin to expectant fathers promoted attention to virtual baby‐related avatars (Cohen‐Bendahan, Beijers, Van Doornen, & de Weerth, 2015), and affected neural and behavioral responses to sounds of infants crying (Alyousefi‐Van Dijk et al., 2019; Thijssen et al., 2018), pointing to a role for vasopressin in responding to infant distress. Moreover, vasopressin levels may be related to fathers’ stimulatory interaction with their infants (Abraham & Feldman, 2018).
Levels of oxytocin, another hormone related to parenting (Feldman & Bakermans‐Kranenburg, 2017), increased over the first 6 months of fatherhood and after stimulatory play (Abraham & Feldman, 2018), while experimentally increased oxytocin levels led to more stimulatory play in fathers (Naber, Van IJzendoorn, Deschamps, Van Engeland, & Bakermans‐Kranenburg, 2010). This experimental study was also the first to show that administering oxytocin to fathers affected fathers’ behavior.
Cortisol may also play a role in fathers’ parenting. In mothers, higher levels of cortisol when babies are 2–24 months old are related to lower maternal sensitivity (Finegood, Blair, Granger, Hibel, & Mills‐Koonce, 2016), but directly after birth, high levels of cortisol are associated with more affectionate infant‐directed behavior (Fleming, Steiner, & Corter, 1997). In fathers, cortisol levels increase in response to infant crying (Fleming et al., 2002), and decrease when they hold their newborn (Kuo et al., 2018) or interact with their toddler (Storey, Noseworthy, Delahunty, Halfyard, & McKay, 2011). The distinction between basal cortisol levels and cortisol reactivity may be essential. Cortisol reactivity may be functional in responding to stressors such as the birth experience or infant distress, but (chronic) high cortisol levels may not be conducive to sensitive parenting. Indeed, in one study, fathers’ higher prenatal cortisol levels predicted lower quality of parenting 6 weeks postnatally (Bos et al., 2018). Moreover, cortisol may interact with testosterone in relation to parenting behavior. During prenatal care of a life‐like infant doll, cortisol was negatively associated with quality of caregiving in fathers with high testosterone levels (Bos et al., 2018).
In summary, hormonal changes in the transition to fatherhood seem related to parenting behavior bidirectionally (see Figure 1). Moreover, hormonal changes may induce or accompany changes in brain structure and functioning. Next, we turn to this issue.
Neural Networks
In mothers, reductions in grey matter volume in brain areas related to parenting have been observed from before to after pregnancy, while no such changes have been apparent in fathers (Hoekzema et al., 2016). However, changes in fathers’ grey matter volume in the postnatal period (between 2–4 weeks and 12–16 weeks after birth) have been seen (Kim et al., 2014). In animals, grey matter volume increased in brain regions involved in the detection of salient infant cues and regulating parenting behaviors, and that are especially sensitive to oxytocin and vasopressin through high densities of the pertinent receptors. Structural brain changes in new mothers can be induced by endocrine changes around pregnancy and childbirth, or by caregiving experiences after birth that may differ between mothers (who are often primary caregivers) and fathers (who are often secondary caregivers). To disentangle these two factors, and focusing on neural responses rather than morphology, one study compared primary caregiving mothers, secondary caregiving fathers, and primary caregiving (homosexual) fathers after the birth of their first child (Abraham et al., 2014). When watching themselves interact with their infant, primary caregiving fathers were similar to secondary caregiving fathers in the activation of their superior temporal sulcus (STS), the social understanding network, but similar to mothers in the activation of their amygdala, the emotional processing network. This points to the influence of caregiving experiences on brain functionality, which is corroborated by the finding that the connectivity between the STS and the amygdala increased linearly with time spent directly responsible for infant care.
In a meta‐analysis of brain responses of 350 people, 95 of whom were fathers, to sounds of infants’ cries, men showed more activity than women in the right inferior frontal gyrus (IFG), extending into the temporal pole and left angular gyrus (Witteman et al., 2019). The right IFG is involved in mentalizing, while the angular gyrus is involved in semantic processing. This suggests that men may preferentially activate a mentalizing network when processing infants’ cries. Women showed more activity in the insula (involved in emotional processing). The meta‐analysis also compared parents and (partnered) nonparents. Compared to adults without children, parents shifted toward more activity in a sensorimotor network including the insula, pre‐ and postcentral gyrus, and right putamen, enabling the integration of emotional information with somatosensory and motor information, and paving the way for behavioral responses (see Figure 1).
A study of processing threat to infants looked at the neural basis for protective parenting before and after the birth of fathers’ first child (Van 't Veer, Thijssen, Witteman, Van IJzendoorn, & Bakermans‐Kranenburg, 2019). In this imaging study, fathers viewed videos of an infant in danger and a matched control video without such danger, and were told to imagine that the infant was their own or someone else’s. Neural responses in bilateral motor areas, possibly indicating preparation for action, were stronger when fathers‐to‐be imagined that the threatened infant was their own rather than someone else’s, but after the birth of their baby, the distinction between responses to one’s own and someone else’s infant faded (Van 't Veer et al., 2019). This suggests that protective mechanisms present during pregnancy may broaden to include other babies after the experience of having an infant.
Directions for Research and Intervention: The Father‐to‐Be
After decades in which men and infants were often perceived as inhabiting different worlds, their worlds have met. The transition to fatherhood is a major life event that may predict parenting involvement and child development through toddlerhood and middle childhood into adolescence. In our review, we focused on fathering in the first 1,000 days, and father–infant interaction can be supported as early as pregnancy. In our lab, we tested a prenatal video‐feedback intervention program using ultrasounds between the 21st and 30th week of pregnancy. Each father is invited to interact with the fetus, verbally or by softly massaging the infant through the mother’s abdominal wall. The baby’s response is seen through ultrasound, a safe and noninvasive way to watch and wonder about the unborn child. The interaction is videotaped and reviewed with the father in three sessions, focusing on (a) the infant’s attachment versus exploration signals, (b) speaking for the baby (e.g., she seems really relaxed when you sing that song for her), and (c) sensitivity chains (movement of the fetus, parental response, potential response of the fetus). The parenting coach doing the intervention uses the video fragments to illustrate each theme. Seeing the infants respond to their fathers singing or reading to them is a moving experience, one that hopefully creates a head start for fathers.
Prenatal fathering experiences like this may support and extend the increased involvement of fathers in childrearing that we have seen in the past 50 years. Since involvement in caregiving affects fathers’ hormonal and neural functioning, such effects on fathers may be lasting, but this issue requires further research. The question of how changes in men’s behavior, and in their hormonal and neural functioning in the prenatal period are related to each other and to sociocultural factors warrants more attention. Sociocultural norms and expectations regarding fathers’ involvement during pregnancy may set the stage for fathers’ involvement after birth. Given the effects of fathering on child development, researchers and policymakers should give a more prominent place to fathers during pregnancy and the early postnatal period for the sake of the child and the family.
Marian J. Bakermans‐Kranenburg, Anna Lotz, Kim Alyousefi‐van Dijk, Vrije Universiteit Amsterdam; Marinus van IJzendoorn, Erasmus University.
Dr. Marian J. Bakermans‐Kranenburg’s work is supported by the European Research Council (ERC AdG 669249).
References
- Abraham, E. , & Feldman, R. (2018). The neurobiology of human allomaternal care: Implications for fathering, coparenting, and children’s social development. Physiology & Behavior, 193, 25–34. 10.1016/j.physbeh.2017.12.034 [DOI] [PubMed] [Google Scholar]
- Abraham, E. , Hendler, T. , Shapira‐Lichter, I. , Kanat‐Maymon, Y. , Zagoory‐Sharon, O. , & Feldman, R. (2014). Father’s brain is sensitive to childcare experiences. Proceedings of the National Academy of Sciences of the United States of America, 111, 9792–9797. 10.1073/pnas.1402569111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ainsworth, M. D. S. (1967). Infancy in Uganda: Infant care and the growth of love. Baltimore, MD: The Johns Hopkins Press. [Google Scholar]
- Alyousefi‐van Dijk, K. , Van 't Veer, A. E. , Meijer, W. M. , Lotz, A. M. , Rijlaarsdam, J. , Witteman, J. , & Bakermans‐Kranenburg, M. J. (2019). Vasopressin differentially affects handgrip force of expectant fathers in reaction to own and unknown infant faces. Frontiers in Behavioral Neuroscience, 13, 105 10.3389/fnbeh.2019.00105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg, A. (2016). The importance of the first 1,000 days of life. Journal of Child & Adolescent Mental Health, 28, iii–vi. 10.2989/17280583.2016.1223803 [DOI] [PubMed] [Google Scholar]
- Bos, P. A. , Hechler, C. , Beijers, R. , Shinohara, K. , Esposito, G. , & de Weerth, C. (2018). Prenatal and postnatal cortisol and testosterone are related to parental caregiving quality in fathers, but not in mothers. Psychoneuroendocrinology, 97, 94–103. 10.1016/j.psyneuen.2018.07.013 [DOI] [PubMed] [Google Scholar]
- Bowlby, J. (1982). Attachment, Attachment and loss (Vol. 1). Harmondsworth, UK: Penguin Books. (Original work published 1969) [Google Scholar]
- Bronfenbrenner, U. , & Ceci, S. J. (1994). Nature‐nurture reconceptualized in developmental perspective: A bioecological model. Psychological Review, 101, 568–586. 10.1037/0033-295X.101.4.568 [DOI] [PubMed] [Google Scholar]
- Cohen‐Bendahan, C. C. , Beijers, R. , Van Doornen, L. J. , & de Weerth, C. (2015). Explicit and implicit caregiving interests in expectant fathers: Do endogenous and exogenous oxytocin and vasopressin matter? Infant Behavior and Development, 41, 26–37. 10.1016/j.infbeh.2015.06.007 [DOI] [PubMed] [Google Scholar]
- Craig, L. , & Mullan, K. (2010). Parenthood, gender and work‐family time in the United States, Australia, Italy, France, and Denmark. Journal of Marriage and Family, 72, 1344–1361. 10.1111/j.1741-3737.2010.00769.x [DOI] [Google Scholar]
- Dagan, O. , & Sagi‐Schwartz, A. (2018). Early attachment network with mother and father: An unsettled issue. Child Development Perspectives, 12, 115–121. 10.1111/cdep.12272 [DOI] [Google Scholar]
- Edelstein, R. S. , Chopik, W. J. , Saxbe, D. E. , Wardecker, B. M. , Moors, A. C. , & LaBelle, O. P. (2017). Prospective and dyadic associations between expectant parents’ prenatal hormone changes and postpartum parenting outcomes. Developmental Psychobiology, 59, 77–90. 10.1002/dev.21469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman, R. , & Bakermans‐Kranenburg, M. J. (2017). Oxytocin: A parenting hormone. Current Opinion in Psychology, 15, 13–18. 10.1016/j.copsyc.2017.02.011 [DOI] [PubMed] [Google Scholar]
- Finegood, E. D. , Blair, C. , Granger, D. A. , Hibel, L. C. , & Mills‐Koonce, R. (2016). Psychobiological influences on maternal sensitivity in the context of adversity. Developmental Psychology, 52, 1073–1087. 10.1037/dev0000123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming, A. S. , Corter, C. , Stallings, J. , & Steiner, M. (2002). Testosterone and prolactin are associated with emotional responses to infant cries in new fathers. Hormones and Behavior, 42, 399–413. 10.1006/hbeh.2002.1840 [DOI] [PubMed] [Google Scholar]
- Fleming, A. S. , Steiner, M. , & Corter, C. (1997). Cortisol, hedonics, and maternal responsiveness in human mothers. Hormones and Behavior, 32, 85–98. 10.1006/hbeh.1997.1407 [DOI] [PubMed] [Google Scholar]
- Gaunt, R. (2008). Maternal gatekeeping: Antecedents and consequences. Journal of Family Issues, 29, 373–395. 10.1177/0192513X07307851 [DOI] [Google Scholar]
- Gettler, L. T. , McDade, T. W. , Feranil, A. B. , & Kuzawa, C. W. (2011). Longitudinal evidence that fatherhood decreases testosterone in human males. Proceedings of the National Academy of Sciences of the United States of America, 108, 16194–16199. 10.1006/hbeh.1997.1407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glover, V. , O'Donnell, K. J. , O'Connor, T. G. , & Fisher, J. (2018). Prenatal maternal stress, fetal programming, and mechanisms underlying later psychopathology—A global perspective. Development and Psychopathology, 30, 843–854. 10.1017/S095457941800038X [DOI] [PubMed] [Google Scholar]
- Grossmann, K. , Grossmann, K. E. , Fremmer‐Bombik, E. , Kindler, H. , Scheuerer‐Englisch, H. , & Zimmermann, A. P. (2002). The uniqueness of the child‐father attachment relationship: Fathers’ sensitive and challenging play as a pivotal variable in a 16‐year longitudinal study. Social Development, 11, 301–337. 10.1111/1467-9507.00202 [DOI] [Google Scholar]
- Gutierrez‐Galve, L. , Stein, A. , Hanington, L. , Heron, J. , Lewis, G. , O’Farrelly, C. , & Ramchandani, P. G. (2018). Association of maternal and paternal depression in the postnatal period with offspring depression at age 18 years. JAMA Psychiatry, 76, 290–296. 10.1001/jamapsychiatry.2018.3667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harlow, H. F. (1958). The nature of love. American Psychologist, 13, 673–685. 10.1037/h0047884 [DOI] [PubMed] [Google Scholar]
- Heymann, J. , & McNeill, K. (2013). Children’s chances: How countries can move from surviving to thriving. Cambridge, MA: Harvard University Press; 10.1111/gove.12168 [DOI] [Google Scholar]
- Hoekzema, E. , Barba‐Müller, E. , Pozzobon, C. , Picado, M. , Lucco, F. , García‐García, D. , … Vilarroya, O. (2016). Pregnancy leads to long‐lasting changes in human brain structure. Nature Neuroscience, 20, 287–296. 10.1038/nn.4458 [DOI] [PubMed] [Google Scholar]
- Kim, P. , Rigo, P. , Mayes, L. C. , Feldman, R. , Leckman, J. F. , & Swain, J. E. (2014). Neural plasticity in fathers of human infants. Social Neuroscience, 9, 522–535. 10.1080/17470919.2014.933713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krueger, P. M. , & Friedman, E. M. (2009). Sleep duration in the United States: A cross‐sectional population‐based study. American Journal of Epidemiology, 169, 1052–1063. 10.1093/aje/kwp023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo, P. X. , Braungart‐Rieker, J. M. , Burke Lefever, J. , Sarma, M. S. , O'Neill, M. , & Gettler, L. T. (2018). Fathers’ cortisol and testosterone in the days around infants’ births predict later paternal involvement. Hormones & Behavior, 106, 28–34. 10.1016/j.yhbeh.2018.08.011 [DOI] [PubMed] [Google Scholar]
- Lamb, M. E. (1976). The role of the father in child development. Ottawa, ON: Wiley. [Google Scholar]
- Lickenbrock, D. M. , & Braungart‐Rieker, J. M. (2015). Examining antecedents of infant attachment security with mothers and fathers: An ecological systems perspective. Infant Behavior and Development, 39, 173–187. 10.1016/j.infbeh.2015.03.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucassen, N. , Tharner, A. , Van IJzendoorn, M. H. , Bakermans‐Kranenburg, M. J. , Volling, B. L. , Verhulst, F. C. , … Tiemeier, H. (2011). The association between paternal sensitivity and infant‐father attachment security: A meta‐analysis of three decades of research. Journal of Family Psychology, 25, 986–992. 10.1037/a0025855 [DOI] [PubMed] [Google Scholar]
- Main, M. , & Weston, D. R. (1981). The quality of the toddler’s relationship to mother and to father: Related to conflict behavior and the readiness to establish new relationships. Child Development, 52, 932–940. 10.2307/1129097 [DOI] [Google Scholar]
- Mason, C. , & Elwood, R. (1995). Is there a physiological basis for the couvade and onset of paternal care? International Journal of Nursing Studies, 32, 137–148. 10.1016/0020-7489(94)00038-L [DOI] [PubMed] [Google Scholar]
- Meijer, W. M. , Van IJzendoorn, M. H. , & Bakermans‐Kranenburg, M. J. (2019). Challenging the challenge hypothesis on testosterone in fathers: A meta‐analysis. Psychoneuroendocrinology, 110, 104435 10.1016/j.psyneuen.2019.104435 [DOI] [PubMed] [Google Scholar]
- Naber, F. B. A. , Van IJzendoorn, M. H. , Deschamps, P. , Van Engeland, H. , & Bakermans‐Kranenburg, M. J. (2010). Intranasal oxytocin increases fathers’ observed responsiveness during play with their children: A double‐blind within‐subject experiment. Psychoneuroendocrinology, 35, 1583–1586. 10.1016/j.psyneuen.2010.04.007 [DOI] [PubMed] [Google Scholar]
- Paulson, J. F. , Bazemore, S. D. , Goodman, J. H. , & Leiferman, J. A. (2016). The course and interrelationship of maternal and paternal perinatal depression. Archives of Women's Mental Health, 19, 655–663. 10.1007/s00737-016-0598-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramchandani, P. , Stein, A. , Evans, J. , & O’Connor, T. G. (2005). Paternal depression in the postnatal period and child development: A prospective population study. The Lancet, 365, 2201–2205. 10.1016/s0140-6736(05)66778-5 [DOI] [PubMed] [Google Scholar]
- Roby, P. A. (1975). Shared parenting: Perspectives from other nations. School Review, 83, 415–431. 10.1086/443200 [DOI] [Google Scholar]
- Saxbe, D. , Corner, G. W. , Khaled, M. , Horton, K. , Wu, B. , & Khoddam, H. L. (2018). The weight of fatherhood: Identifying mechanisms to explain paternal perinatal weight gain. Health Psychology Review, 12, 1–18. 10.1080/17437199.2018.1463166 [DOI] [PubMed] [Google Scholar]
- Storey, A. E. , Noseworthy, D. E. , Delahunty, K. M. , Halfyard, S. J. , & McKay, D. W. (2011). The effects of social context on the hormonal and behavioral responsiveness of human fathers. Hormones and Behavior, 60, 353–361. 10.1016/j.yhbeh.2011.07.001 [DOI] [PubMed] [Google Scholar]
- Storey, A. E. , Walsh, C. J. , Quinton, R. L. , & Wynne‐Edwards, K. E. (2000). Hormonal correlates of paternal responsiveness in new and expectant fathers. Evolution and Human Behavior, 21, 79–95. 10.1016/S1090-5138(99)00042-2 [DOI] [PubMed] [Google Scholar]
- Thijssen, S. , Van 't Veer, A. E. , Witteman, J. , Meijer, W. M. , van IJzendoorn, M. H. , & Bakermans‐Kranenburg, M. J. (2018). Effects of vasopressin on neural processing of infant crying in expectant fathers. Hormones and Behavior, 103, 19–27. 10.1016/j.yhbeh.2018.05.014 [DOI] [PubMed] [Google Scholar]
- Van Anders, S. M. , Tolman, R. M. , & Volling, B. L. (2012). Baby cries and nurturance affect testosterone in men. Hormones and Behavior, 61, 31–36. 10.1016/j.yhbeh.2011.09.012 [DOI] [PubMed] [Google Scholar]
- Van 't Veer, A. E. , Thijssen, S. , Witteman, J. , Van Ijzendoorn, M. H. , & Bakermans‐Kranenburg, M. J. (2019). Exploring the neural basis for paternal protection: An investigation of the neural response to infants in danger. Social Cognitive and Affective Neuroscience, 14, 447–457. 10.1093/scan/nsz018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verhage, M. L. , Schuengel, C. , Madigan, S. , Fearon, R. M. P. , Oosterman, M. , Cassibba, R. , … Van IJzendoorn, M. H. (2016). Narrowing the transmission gap: A synthesis of three decades of research on intergenerational transmission of attachment. Psychological Bulletin, 142, 337–366. 10.1037/bul0000038 [DOI] [PubMed] [Google Scholar]
- Volling, B. L. , McElwain, N. L. , Notaro, P. C. , & Herrera, C. (2002). Parents' emotional availability and infant emotional competence: Predictors of parent‐infant attachment and emerging self‐regulation. Journal of Family Psychology, 16, 447–465. 10.1037//0893-3200.16.4.447 [DOI] [PubMed] [Google Scholar]
- Winslow, J. T. , Hastings, N. , Carter, C. S. , Harbaugh, C. R. , & Insel, T. R. (1993). Selective aggression and affiliation increase following mating in a monogamous mammal: A role for central vasopressin in pair bonding. Nature, 365, 545–548. [DOI] [PubMed] [Google Scholar]
- Witteman, J. , Van IJzendoorn, M. H. , Rilling, J. K. , Bos, P. A. , Schiller, N. O. , & Bakermans‐Kranenburg, M. J. (2019). Towards a neural model of infant cry perception. Neuroscience & Biobehavioral Reviews, 99, 23–32. 10.1016/j.neubiorev.2019.01.026 [DOI] [PubMed] [Google Scholar]
- Wood, J. J. , & Repetti, R. L. (2004). What gets dad involved? A longitudinal study of change in parental child caregiving involvement. Journal of Family Psychology, 18, 237–249. 10.1037/0893-3200.18.1.237 [DOI] [PubMed] [Google Scholar]


