Abstract
Pollinators are crucial for ecosystem functionality; however, little is known about the plant species used by some of these, such as stingless bees. In this study, for the first time, pollen resources used by Melipona mimetica Cockerell (Hymenoptera: Apidae: Meliponini) and Scaptotrigona sp. Moure (Hymenoptera: Apidae: Meliponini) were identified through analysis of corbicular pollen found on worker bees in a dry forest in southern Ecuador. In total, 68 pollen types were identified belonging to 31 botanical families. The most represented plant families were Fabaceae (16%), Malvaceae (7%), and Boraginaceae (7%). Both stingless bee species exhibited a polylectic behavior, with an average of 16 pollen types collected by individual bees. Differences in abundances of pollen types collected by each species indicated distinct uses for these two bee species.
Keywords: stingless bee, corbicular pollen, Melipona, Scaptotrigona, dry forest
Pollinators play a critical functional role in most terrestrial ecosystems because plant persistence and regeneration depend on their activity (Klein et al. 2007, Absy et al. 2018). Additionally, they provide a key ecosystem service not only through the pollination of many crops but also through the direct resources they offer local human communities such as honey (Pasupuleti et al. 2017). Among pollinators, bees are particularly important in the reproduction of crops and most wild plants (Klein et al. 2007, Garibaldi et al. 2013). In parallel, plants are essential for bee growth and reproduction because, in all their life stages, bees are nutritionally dependent on floral resources, mainly nectar and pollen (Ogilvie and Forrest 2017, Absy et al. 2018). Unfortunately, pollinator decline is invoked as one of the most pervasive problems worldwide (Potts et al. 2010). Consequently, an understanding of bees’ performance and resource preferences in different ecosystems will permit the development of conservation actions to mend the decline in pollinators caused by human activities (Kleijn et al. 2008, Potts et al. 2010, Roubik and Patiño 2018).
Stingless bees comprise a diverse group of eusocial bees, especially diversified in the tropics (Slaa et al. 2006). They are considered one of the most important pollinators (Quezada-Euán 2018) and, also, the most relevant as ecosystem service providers in several tropical ecosystems. They are considered polylectic (i.e., generalists) because of their ability to collect pollen and nectar from an array of nonrelated plants (Eltz et al. 2001, Biesmeijer et al. 2005). Consequently, stingless bees constitute an important component in the complex pollinator networks of most tropical forest ecosystems (Schleuning et al. 2012). To know how these bees forage in tropical forests is a priority to guarantee their conservation and the services these insects provide (Murray et al. 2009).
Ecuador is considered one of the most diverse stingless bee hotspots with ca. 130 species representing 25% of the known species worldwide (Roubik 2018, Vit et al. 2018). An example of this extraordinary diversity was documented by Roubik (2018) in a 50 ha plot in Yasuní Biosphere Reserve, where 100 stingless bee species were identified. In addition, in the seasonal forests of southern Ecuador, 89 species have been recorded (Ramírez-Romero et al. 2013), some of which are traditionally managed in rural–urban areas and critical for the local economies (Martínez-Fortún et al. 2018). Surprisingly, information on the foraging behavior of stingless bees is almost entirely lacking for some Neotropical countries, including Ecuador.
Therefore, our research objective is to identify the pollen sources used by Scaptotrigona sp. Moure (Hymenoptera: Apidae: Meliponini) ‘catana’ and the Melipona mimetica Cockerell (Hymenoptera: Apidae: Meliponini) ‘bermejo’, which are two of the most important bees managed for honey production in seasonal dry forests in southern Ecuador (Vit et al. 2018). These forests are one of the most threatened ecosystem in Ecuador and are suffering a dramatic decline with unknown deforestation rates (Linares-Palomino et al. 2011, Tapia-Armijos et al. 2015).
Materials and Methods
The present study was conducted in the La manga-Garza Real area (85.1987 S; 72.712977 W) located in the municipality of Zapotillo, Loja Province, in the Southwestern of Ecuador. The regional climate is tropical semiarid, with an average annual temperature of 24°C and an average annual rainfall of 500 mm with a dry season between May and December and a rainy season between January and April (Maldonado 2002). This area is characterized by dry seasonal forests dominated by tree species such as Guayacán (Handroanthus chrysantha), Ceiba (Ceiba trichistandra), and Pretino (Cavanillesia platanifolia), which are interspersed with lowland scrub formations at elevations ranging between 525 and 547 m.a.s.l. (Cueva and Chalán 2010).
Sampling was carried out in the months of June, August, October, and November 2015 and May 2016, covering both dry and rainy seasons. We selected 3 meliponaries, spaces for the breeding of stingless bees for honey production, separated 500 m from each other, for evaluating the foraging behavior of both species. Each meliponary contained a colony of each stingless bee species. Pollen foragers returning to the hive were captured at the nest entrances using a hand-net between 8:30 and 14:00 h. From each colony, samples were collected from two pollen sacs of foragers bees, and each sample was individually placed in a 2-ml Eppendorf tube, duly labeled, and brought back to the laboratory. During each month, pollen was collected from 30 foragers for each colony and bee species.
Due to the lack of a pollen library for the plant species in our study region, pollen characterization of the vegetation was necessary. Floral resources identification in the surrounding habitat was done through a radial track of 2 km around the colonies; this distance is an approximate foraging range of some stingless bee species (Kuhn-Neto et al. 2009). For each plant species with floral resources, blossoms and flowers were taken for pollen characterization. We used these samples to create a pollen reference collection for our study.
In the laboratory, corbicular pollen samples from each individual were diluted in alcohol and mounted on three microscope slides. Once the alcohol evaporated, we set the pollen using 2 μl of glycerogelatin solution; the center of each slide was stained with fuchsin. The glycerogelatin solution was prepared according to Wodehouse (1935), diluting 7 g of gelatin, 50 ml of glycerin, and 1 g phenol in 42 ml of distilled water. The classification of pollen morphotypes was done using a Zeiss—Axiostar Plus optical microscope with a 40× objective lens. The number of grains of each morphotype was recorded for each slide, and relative abundance of each representative plant taxon found in the pollen sample was quantified for the three slides. The acetolysis method was not used; therefore, some morphological characteristics of the pollen were not described.
Taxonomic identification of pollen morphotypes found on stingless bees to the level of family, genus, or species was done by comparing them to the hand-collected pollen of plants in the area and, in some cases, with information about the characteristics of the family. Additionally, we obtained the mean and SD of pollen morphotypes visited by individual bees. We calculated the number of pollen types collected by an individual bee by summing the pollen types found on the three slides examined per bee.
Results
In total, 251 individuals of Melipona mimetica and 244 individuals of Scaptotrigona sp. that presented pollen loads were analyzed. In total, we identified 68 pollen morphotypes belonging to 31 families, 38 genera, 36 species, and 10 undetermined types (Table 1, Supp Plates 1–5 [online only]).
Table 1.
Frequency of pollen types present in samples of pollen loads of Melipona mimetica and Scaptotrigona sp.
| Family | Species/pollen type | Melipona mimetica | Scaptotrigona sp. | Plant type | Plate |
|---|---|---|---|---|---|
| Apocynaceae | |||||
| Nerium oleander | 0.34 | 0.69 | Tree | 1-p | |
| Arecaceae | |||||
| type 1 | 0.55 | 0.24 | 2-f | ||
| Asteraceae | |||||
| Parthenium hysterophorus | 3.32 | 2.57 | Herb | 1-r | |
| Jungia paniculata | 1.32 | 0.53 | Herb | 1-h | |
| Bidens pilosa | 0 | 0.001 | Herb | 5-i | |
| Bixaceae | |||||
| Cochlospermum vitifolium | 5.12 | 1.81 | Tree | 2-d | |
| Boraginaceae | |||||
| Heliotropium angiospermum | 0.68 | 0.54 | Herb | 1-g | |
| Cordia sp.1 | 0.44 | 0.57 | Shrub | 2-p | |
| Cordia lutea | 0.05 | 0.02 | Shrub | 2-g | |
| Heliotropium sp.1 | 3.68 | 2.71 | Herb | 2-a | |
| type 1 | 0.14 | 0.42 | 3-d | ||
| Cactaceae | |||||
| Armatocereus cartwrightianus | 0.66 | 0.41 | Succulent | 2-h | |
| Opuntia megasperma | 0.12 | 0.02 | Succulent | 5-e | |
| Convolvulaceae | |||||
| Ipomoea sp.1 | 0.19 | 1.52 | Shrub | 4-h | |
| Cunoniaceae | |||||
| type 1 | 12.58 | 0.77 | 5-b | ||
| Ericaceae | |||||
| type 1 | 1.39 | 0.78 | 2-o | ||
| Euphorbaceae | |||||
| Croton scouleri | 0.43 | 0.26 | Shrub | 3-f | |
| Fabaceae | |||||
| Caesalpinea glabrata | 4.13 | 1.2 | Tree | 5-c | |
| Pithecellobium sp.1 | 2.38 | 1.55 | Tree | 1-k | |
| Crotalaria sp.1 | 1.55 | 7.56 | Herb | 1-s | |
| Cajanus cajan | 0.99 | 0.54 | Shrub | 2-j | |
| Cercidium praecox | 0.95 | 0.97 | Tree | 3-e | |
| Senna incarnata | 0.57 | 0.81 | Shrub | 1-i | |
| Leucaena trichodes | 0.25 | 0.32 | Tree | 2-k | |
| Prosopis juliflora | 0.12 | 0.04 | Tree | 5-f | |
| Erythrina velutina | 0.05 | 0.16 | Tree | 1-m | |
| Acacia riparia | 0.007 | 0.01 | Tree/shrub | 3-a | |
| Bauhinia aculeata | 0.002 | 0 | Herb | 4-e | |
| Lamiaceae | |||||
| Clerodendrum molle | 4.6 | 4.44 | Shrub | 1-b | |
| type 1 | 0.65 | 0.29 | Shrub | 1-n | |
| type 2 | 0.03 | 0.02 | Shrub | 4-f | |
| Malvaceae | |||||
| Eriotheca ruizii | 0.43 | 0.63 | Tree | 1-c | |
| Ceiba insignis | 0.06 | 0.04 | Tree | 1-d | |
| Ceiba trichistandra | 0.06 | 0.03 | Tree | 2-q | |
| Ochroma pyramidale | 0.03 | 0.01 | Tree | 4-c | |
| Briquetia spicata | 0.02 | 0.01 | Herb | 4-b | |
| Melastomataceae | |||||
| Miconia gleasoniana | 0.7 | 1.64 | Shrub | 1-t | |
| Mimosaceae | |||||
| Inga coruscans | 1.16 | 0.4 | Tree | 1-l | |
| Inga sp. | 0.007 | 0.01 | Tree | 3-b | |
| Muntingiaceae | |||||
| type 1 | 3.42 | 10.24 | Shrub | 1-q | |
| Myrtaceae | |||||
| Psidium sp.1 | 0.08 | 0.03 | Tree | 4-g | |
| type 1 | 0 | 0.01 | 5-l | ||
| Nyctaginaceae | |||||
| Bougainvillea sp.1 | 0.22 | 0.13 | Shrub | 5-a | |
| Oleaceae | |||||
| type 1 | 0.17 | 0.12 | Shrub | 2-r | |
| Plumbaginaceae | |||||
| Plumbago scandens | 0.74 | 1.39 | Herb | 2-t | |
| Polygonaceae | |||||
| type 1 | 7.97 | 19.77 | 1-j | ||
| type 2 | 1.04 | 0.09 | 2-n | ||
| Proteaceae | |||||
| type 1 | 2.21 | 1.02 | Shrub | 2-b | |
| type 2 | 1.12 | 0.24 | Shrub | 2-c | |
| Pteridaceae | |||||
| Pteris altissima | 1.97 | 0.48 | Herb | 1-f | |
| Rhamnaceae | |||||
| Scutia spicata | 3.14 | 2.35 | Herb | 2-m | |
| Rosaceae | |||||
| Cydonia oblonga | 2.66 | 1.84 | tree | 1-a | |
| Rubiaceae | |||||
| type 1 | 0.15 | 0.14 | 1-e | ||
| Solanaceae | |||||
| Physalis pubescens | 0.96 | 0.2 | Herb | 5-h | |
| Sterculiaceae | |||||
| Byttneria flexuosa | 6.94 | 10.5 | Shrub | 2-e | |
| Tiliaceae | |||||
| Corchorus sp.1 | 13.99 | 13.25 | Herb | 5-j | |
| Heliocarpus sp.1 | 1.1 | 1.61 | Shrub | 2-l | |
| Undetermined | |||||
| type 1 | 1.29 | 0.57 | 1-o | ||
| type 2 | 1.16 | 0.13 | 2-i | ||
| type 3 | 0.19 | 0.37 | 2-s | ||
| type 4 | 0.13 | 0.35 | 3-c | ||
| type 5 | 0.13 | 0.02 | 3-g | ||
| type 6 | 0.11 | 0.28 | 4-a | ||
| type 7 | 0.04 | 0.03 | 4-d | ||
| type 8 | 0.03 | 0.02 | 5-d | ||
| type 9 | 0.009 | 0.006 | 5-g | ||
| type 10 | 0.009 | 0.005 | 5-k | ||
| Verbenaceae | |||||
| Verbena officinalis | 0.02 | 0.03 | Herb | 5-m |
Pollen type with frequency >5% for each bee species is in bold. Pollen structures ubication in Supp Mater. (online only) are included; the number represents the plate number and the letter the position.
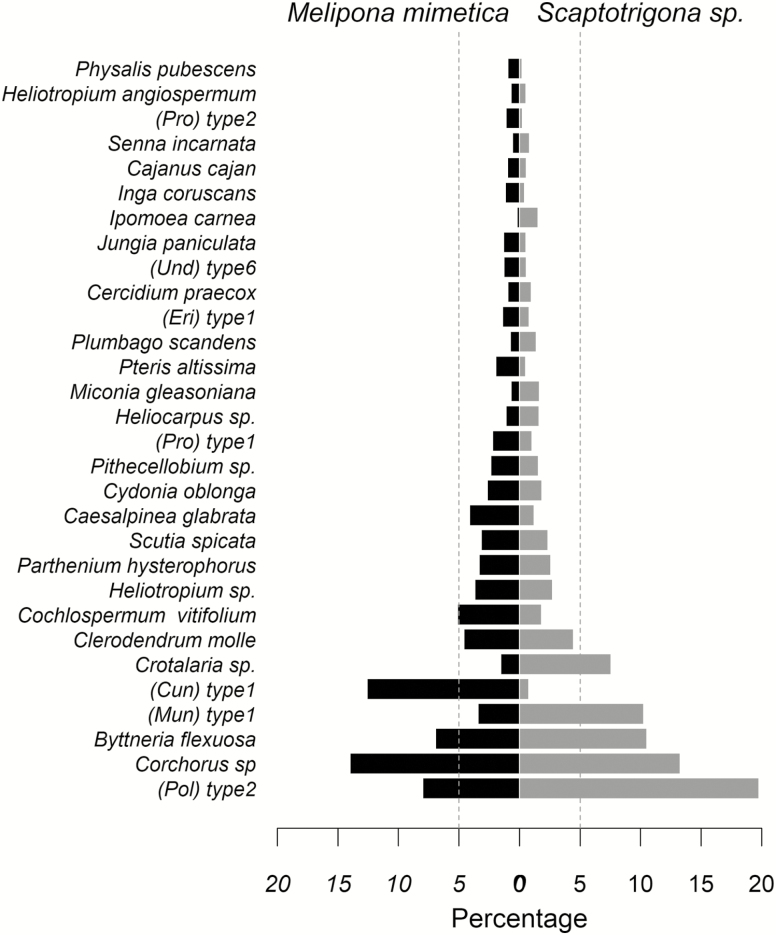
The occurrence of pollen morphotypes (for pooled samples) were almost identical for these two species: 66 for M. mimetica and 67 for Scaptotrigona sp. The botanical families that presented more pollen morphotypes were Fabaceae (11), Malvaceae and Boraginaceae (5), and Lamiaceae (3; Table 1). The two bee species collected similar pollen morphotypes although in different relative abundances (Fig. 1). For M. mimetica, the most abundant pollen morphotypes (>5%) were from Corchorus sp. (Tiliacea) with 14% of total corbicular pollen samples collected in the whole sampling period, 13% Cunoniaceae type 1, 8% Polygonaceae type 1, 7% Byttneria flexuosa (Sterculiaceae), and 5.2% Cochlospermum vitifolium (Bixaceae) (Table 1; Fig. 1). For Scaptotrigona sp., the most representative species were as follows: Polygonaceae type 1 with 20%, Corchorus sp. with 13%, Byttneria flexuosa with 10%, Muntingiaceae type 1 species with 10%, and Crotolaria sp. (Fabaceae) with 7% (Table 1; Fig. 1).
Fig. 1.
Percentage of pollen occurrence from most frequent plant species for Melipona mimetica and Scaptotrigona sp. in the Southern of Ecuador. Undetermined species are expressed as ‘type’ and their family is indicated in brackets. Pro = Proteaceae, Eri = Ericaceae, Cun = Cunoniaceae, Mun = Mundaceae, Pol = Polygonaceae, Und = Undetermined family.
Additionally, we found a polylectic behavior for both bee species, with a mean of 16.51 (±4.89 SD) pollen morphotypes collected by individuals of M. mimetica and 15.95 (±5.96 SD) for Scaptotrigona sp. Although both bee species collected the same number of pollen morphotypes, M. mimetica bees collected more than 20% of the pollen from a mean of 1.42 (±0.56 SD) morphotypes, in contrast to 1.28 (±0.62 SD) morphotypes for Scaptotrigona sp.
Discussion
In this study, the plant families Fabaceae, Malvaceae, and Boraginaceae represented the highest number of pollen morphotypes. These findings agree with other studies that have reported similar results for eusocial bees, such as those of the Meliponini tribe and Apis mellifera L. (Apini) both in genuine drylands and tropical regions (Dórea et al. 2010, Faria et al. 2012, de Novais et al. 2015). The Fabaceae family is identified in most previous studies as the most important pollen source both in terms of the number of pollen morphotypes and their relative abundances (e.g., Ramalho et al. 1990, Faria et al. 2012, de Novais et al. 2015). In the present study, these results are not unexpected as the three plant families are also the richest ones in the aboveground vegetation of our study area (Cueva-Ortíz et al. 2019). In contrast, pollen morphotypes of the other families, such as Tiliaceae, Cunoniaceae, Polygonaceae, Muntingiaceae, Sterculiaceae, which were identified as primary foraging resources for both M. mimetica and Scaptotrigona sp. in the present study, have not been previously reported for other stingless bee species. For example, Barros et al. (2013) and Ferreira and Absy (2017) in the Brazilian Amazon identified Melastomataceae and Solanaceae as the most representative resources for Melipona interrupta and Melipona fasciculata, respectively. Likewise, Rech and Absy (2011) determined that Fabaceae, Bignoniaceae, and Lamiaceae were important plant families for Scaptotrigona sp. However, most studies took place in wet regions and further research on stingless bees’ pollen resources in dry ecosystems is necessary to confirm the generality of the patterns reported in the present study.
Both stingless bees, M. mimetica and Scaptotrigona sp., exhibited a polylectic behavior with a total of ca. 65 pollen morphotypes collected for each bee species and with individual bees collecting on average 16 different pollen morphotypes. This polylectic behavior has been evidenced in other stingless bee species, with some species collecting 100 different pollen morphotypes (e.g., Heithaus 1979, Wilms et al. 1996). However, other bee species within the Melipona genus do not collect such a high number of pollen morphotypes. For example, Melipona subnitida collected only 14 different morphotypes in a dry forest in Brazil (Maia-Silva et al. 2018). In a similar fashion, Melipona interrupta (Ferreira and Absy 2017), Melipona fasciculata (Barros et al. 2013), and Melipona capixaba (Luz et al. 2011) from wetter tropical forests foraged on approximately 30 different pollen morphotypes, which is still far below what was observed in our species of Melipona. In Brazil, Scaptotrigona fulvicutis in a semideciduous forest (Marques-Souza et al. 2007) and Scaptotrigona aff. depilis in central Amazonian (Faria et al. 2012) also showed a very high number of resource plants, collecting 85 and 97 different pollen morphotypes, which is more pollen types than that reported for our Scaptotrigona species.
In conclusion, this study provides basic information on the plant resources visited by these two stingless bee species in southern Ecuador. Because of their endangered status and local economic importance, knowledge of the pollen resources used by these two bee species could help beekeepers better manage them and develop preservation programs, e.g., concentrating efforts to recover the vegetation in deforested areas using plant species commonly visited by these bees (Luz et al. 2011).
Supplementary Material
Acknowledgments
We thank Adrián Escudero for the friendly revision developed and May Platt for her help in the language revision. We also thank Dr. Johanne Brunet and two anonymous reviewers for their substantial improvement of the manuscript. This research was funded by Secretaria Nacional de Ciencia, Tecnología e Innovación (SENESCYT) through the Ensamble Scholarship, grant number PIC-17-BENS-003.
References Cited
- Absy M., Rech A. R., and Ferreira M. G.. . 2018. Pollen collected by stingless bees: a contribution to understanding Amazonian biodiversity, pp. 29–46. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Barros M. H. M. R., Luz C. F. P. D., and Albuquerque P. M. C. D.. . 2013. Pollen analysis of geopropolis of Melipona (Melikerria) fasciculata Smith, 1854 (Meliponini, Apidae, Hymenoptera) in areas of restinga, cerrado and flooded fields in the state of Maranhão, Brazil. Grana 52: 81–92. [Google Scholar]
- Biesmeijer J. C., Slaa E. J., Castro M. S., Viana B. F., Kleinert A. M. P., and Imperatriz-Fonseca V. L.. . 2005. Connectance of Brazilian social bee: food plant networks is influenced by habitat, but not by latitude, altitude or network size. Biota Neotrop. 5: 85–93. [Google Scholar]
- Cueva O. J., and Chalán L. A.. . 2010. Cobertura Vegetal y uso Actual del suelo de la Provincia de Loja. Informe Técnico, 1st ed Gráficas Amazonas, Loja, Ecuador. [Google Scholar]
- Cueva-Ortiz J., Espinosa C. I., Quiroz Dahik C., Aguirre Mendoza Z., Cueva Ortiz E., Gusmán E., Weber M., and Hildebrandt P.. . 2019. Influence of anthropogenic factors on the diversity and structure of a dry forest in the Central Part of the Tumbesian Region (Ecuador–Perú). Forests 10: 31. [Google Scholar]
- de Novais J. S., Garcêz A. C. A., Absy M. L., and Francisco de Assis R.. . 2015. Comparative pollen spectra of Tetragonisca angustula (Apidae, Meliponini) from the Lower Amazon (N Brazil) and caatinga (NE Brazil). Apidologie 46: 417–431. [Google Scholar]
- Dórea M. C., Aguiar C. M., Figueroa L. E., Lima L. C. L. E., and Francisco de Assis R.. . 2010. Pollen residues in nests of Centris tarsata Smith (Hymenoptera, Apidae, Centridini) in a tropical semiarid area in NE Brazil. Apidologie 41: 557–567. [Google Scholar]
- Eltz T., Brühl C. A., Van der Kaars S., Chey V. K., and Linsenmair K. E.. . 2001. Pollen foraging and resource partitioning of stingless bees in relation to flowering dynamics in a Southeast Asian tropical rainforest. Insect Soc. 48: 273–279. [Google Scholar]
- Faria L. B. D., Aleixo K. P., Garófalo C. A., Imperatriz-Fonseca V. L., and Silva C. I. D.. . 2012. Foraging of Scaptotrigona aff. depilis (Hymenoptera, Apidae) in an urbanized area: seasonality in resource availability and visited plants. Psyche 2012: 12: 1–12. [Google Scholar]
- Ferreira M. G., and Absy M. L.. . 2017. Pollen analysis of honeys of Melipona (Michmelia) seminigra merrillae and Melipona (Melikerria) interrupta (Hymenoptera: Apidae) bred in Central Amazon, Brazil. Grana 56: 436–449. [Google Scholar]
- Garibaldi L. A., Steffan-Dewenter I., Winfree R., Aizen M. A., Bommarco R., Cunningham S. A., Kremen C., Carvalheiro L. G., Harder L. D., Afik O., . et al. 2013. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339: 1608–1611. [DOI] [PubMed] [Google Scholar]
- Heithaus E. R. 1979. Flower visitation records and resource overlap of bees and wasps in northwest Costa Rica. Registros de visitas a flores y traslape de recursos de abejas y avispas en el noroeste de Costa Rica. Brenesia 16: 9–52. [Google Scholar]
- Kleijn D., and Raemakers I.. . 2008. A retrospective analysis of pollen host plant use by stable and declining bumble bee species. Ecology 89: 1811–1823. [DOI] [PubMed] [Google Scholar]
- Klein A. M., Vaissière B. E., Cane J. H., Steffan-Dewenter I., Cunningham S. A., Kremen C., and Tscharntke T.. . 2007. Importance of pollinators in changing landscapes for world crops. Proc. Biol. Sci. 274: 303–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhn-Neto B., Contrera F. A., Castro M. S., and Nieh J. C.. . 2009. Long distance foraging and recruitment by a stingless bee, Melipona mandacaia. Apidologie 40: 472–480. [Google Scholar]
- Linares-Palomino R., Oliveira-Filho A. T., and Pennington R. T.. . 2011. Neotropical seasonally dry forests: diversity, endemism, and biogeography of woody plants, pp. 3–21. InDirzo R., Young H. S., Mooney H. A., and Ceballos G. (eds.), Seasonally dry tropical forests: ecology and conservation. Island Press, Whasington, DC. [Google Scholar]
- Luz C. F. P. D., Fernandes-Salomao T. M., Lage L. G. A., Resende H. C., Tavares M. G., and Campos L. A. D. O.. . 2011. Pollen sources for Melipona capixaba Moure & Camargo: an endangered Brazilian stingless bee. Psyche 2011: 7. [Google Scholar]
- Maia-Silva C., Limão A. A. C., Hrncir M., da Silva Pereira J., and Imperatriz-Fonseca V. L.. . 2018. The contribution of palynological surveys to stingless bee conservation: a case study with Melipona subnitida, pp. 89–101. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Maldonado N. 2002. Clima y vegetación de la región sur del Ecuador. pp. 1–28. InAguirre Z., Madsen J., Cotton E., and Balslev H. (eds.), Botánica Austroecuatoriana. Ediciones Abya Ayala, Quito, Ecuador. [Google Scholar]
- Marques-Souza A. C., Absy M. L., and Kerr W. E.. . 2007. Pollen harvest features of the central Amazonian bee Scaptotrigona fulvicutis Moure 1964 (Apidae: Meliponinae), in Brazil. Acta Bot. Bras. 21: 11–20. [Google Scholar]
- Martínez-Fortún S., Ruiz C., Quijano N. A., and Vit P.. . 2018. Rural–urban meliponiculture and ecosystems in neotropical areas. Scaptotrigona, a resilient stingless bee?, pp. 421–434. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Murray T. E., Kuhlmann M., and Potts S. G.. . 2009. Conservation ecology of bees: populations, species and communities. Apidologie 40: 211–236. [Google Scholar]
- Ogilvie J. E., and Forrest J. R.. . 2017. Interactions between bee foraging and floral resource phenology shape bee populations and communities. Curr. Opin. Insect Sci. 21: 75–82. [DOI] [PubMed] [Google Scholar]
- Pasupuleti V. R., Sammugam L., Ramesh N., and Gan S. H.. . 2017. Honey, propolis, and royal jelly: a comprehensive review of their biological actions and health benefits. Oxid. Med. Cell. Longev. 2017: 1259510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potts S. G., Biesmeijer J. C., Kremen C., Neumann P., Schweiger O., and Kunin W. E.. . 2010. Global pollinator declines: trends, impacts and drivers. Trends Ecol. Evol. 25: 345–353. [DOI] [PubMed] [Google Scholar]
- Quezada-Euán J. J. G. 2018. Services provided by stingless bees, pp. 243–269. InQuezada-Euán J. J. G. (ed.), Stingless bees of Mexico. Springer, Cham, Switzerland. [Google Scholar]
- Ramalho M., Kleinert-Giovannini A., and Imperatriz-Fonseca V. I.. . 1990. Important bee plants for stingless bees (Melipona and Trigonini) and Africanized honeybees (Apis mellifera) in neotropical habitats: a review. Apidologie 21: 469–488. [Google Scholar]
- Ramírez-Romero J., Ureña-Alvarez J., and Camacho A.. . 2013. Las abejas sin aguijón (Apidae: Meliponini) de la región sur del Ecuador. Cedamaz 3: 81–92. [Google Scholar]
- Rech A. R., and Absy M.. . 2011. Pollen sources used by species of Meliponini (Hymenoptera: Apidae) along the Rio Negro channel in Amazonas, Brazil. Grana 50: 150–161. [Google Scholar]
- Roubik D. W. 2018. 100 species of meliponines (Apidae: Meliponini) in a parcel of western Amazonian forest at Yasuní Biosphere reserve, Ecuador, pp. 189–206. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Roubik D. W., and Patiño J. E. M.. . 2018. The stingless honey bees (Apidae, Apinae: Meliponini) in Panama and pollination ecology from pollen analysis, pp. 47–66. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Schleuning M., Fründ J., Klein A. M., Abrahamczyk S., Alarcón R., Albrecht M., Andersson G. K., Bazarian S., Böhning-Gaese K., Bommarco R., . et al. 2012. Specialization of mutualistic interaction networks decreases toward tropical latitudes. Curr. Biol. 22: 1925–1931. [DOI] [PubMed] [Google Scholar]
- Slaa E. J., Chaves L. A. S., Malagodi-Braga K. S., and Hofstede F. E.. . 2006. Stingless bees in applied pollination: practice and perspectives. Apidologie 37: 3–315. [Google Scholar]
- Tapia-Armijos M. F., Homeier J., Espinosa C. I., Leuschner C., and de la Cruz M.. . 2015. Deforestation and forest fragmentation in South Ecuador since the 1970s – losing a hotspot of biodiversity. PLoS One 10: e0133701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vit P., Pedro S. R., Maza F., Ramírez V. M., and Frisone V.. . 2018. Diversity of stingless bees in Ecuador, pot-pollen standards, and meliponiculture fostering a living museum Meliponini of the world, pp. 207–227. InVit P., Pedro S. R. M., and Roubik D. (eds.), Pot-pollen in stingless bee melittology. Springer, Cham, Switzerland. [Google Scholar]
- Wilms W., Imperatriz-Fonseca V. L., and Engels W.. . 1996. Resource partitioning between highly eusocial bees and possible impact of the introduced Africanized honey bee on native stingless bees in the Brazilian Atlantic rainforest. Stud. Neotrop. Fauna E 31: 137–151. [Google Scholar]
- Wodehouse R. P. 1935. Pollen grains, 1st ed.Mcgraw-Hill Book Company Inc., New York. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.