Abstract
Objectives
Tannins with complex structures are important plant resources, which are abundant in the genus Terminalia. Various Terminalia species have been playing an important role in traditional medicine system. A systematic scoping review of Terminalia Linn. research literature for tannins was conducted to summarize the structures of tannins and analysis fragmentation pathway characteristics, which could provide references for the structural analysis of tannins from Terminalia Linn.
Methods
After an update of the literature search up to September 2018, the terms of Terminalia in all publications were analyzed. Electronic searches were conducted in scifinder and PubMed, and the information from 197 articles in all with regard to the tannin structure study was extracted.
Results
The compounds of 82 tannins from the genus Terminalia were reviewed. According to the structural differences, they can be divided into three categories, hydrolysable tannins, condensed tannins, and complex tannins, respectively. The fragmentation pathways of 46 identified tannins were analyzed, and the fragmentation rules of tannins were speculated according to different types.
Conclusion
This review has attracted attention to the active substances in this species such as the tannins summarized in further study. How to improve the extraction and purification technology of tannins from genus Terminalia is an urgent problem to be solved.
1. Introduction
Plants of the genus Terminalia (family Combretaceae) are widely used in traditional medicine all over the world [1]. There are about 250 Terminalia species, of which at least 50 are used as food [2]. Many species have biological activities including antitumor, anti-inflammatory, wound healing, antifungal, antibacterial, and antiviral activities [3–7]. In particular, Terminalia chebula, an Indian species, is well noted as the king of plants in Ayurveda for its extensive medicinal use [8]. The plants mainly include tannins, polyphenols, triterpenoids, flavonoids, aliphatic compounds, and other active ingredients, among which tannins and polyphenols are the main constituents [9].
Tannins are a kind of polyphenolic compounds with complex structures in plants. They are classified into three groups on the basis of their structures: hydrolysable tannins, condensed tannins, and complex tannins. Usually, their molecular weights are greater than 500 Da. Tannins are widely distributed in various plants, and they are considered defensive molecules to protect plant tissues from herbivorous attacks because of their astringent taste [10]. It has been reported that several natural tannins and related compounds have various biological activities, including antioxidant, antitumor, hypolipidemic, hypoglycemic, and antibacterial activities [11–14]. Takashi Tananka isolated terflavin A and B, tercatain, and tergallagin from the leaves of Terminalia catappa Linn. in 1986 [15]. Since then, more than 82 tannins have been isolated from the fruits, barks, leaves, and galls in the plants of the genus Terminalia. The mass spectrometric data of these tannins and the structure analysis of the compounds are discussed. This review aims to provide references for the structure identification of tannin constituents in the plants of Terminalia Linn. In the further study of phytochemistry, the research field of medicinal activity of this important genus should be highlighted and guided.
2. Methods
2.1. Data Sources and Searches
Electronic searches were conducted in scifinder and PubMed for articles up to September 2018, using terms related to tannins, Terminalia, and MS. Searches were conducted with no date or language restrictions.
2.2. Eligibility and Selection
The titles and abstracts of 197 articles were screened, respectively, and the full text of the article was reviewed to obtain sufficient information. Any disagreements regarding the inclusion of articles were resolved through discussion and consensus.
2.3. Data Extraction
The final data extraction included the following five categories: (1) general characteristics (compound name, source, structure, and journal name); (2) MS data (compound name, ion Source, ion mode, fragments, and journal name); and (3) MS fragmentation pattern (fragmentation rules and journal name).
3. Results and Discussions
3.1. Tannins
Tannins are widely distributed in plants. They can be classified into three types according to their structural differences. Hydrolysable tannins are a group of compounds formed by phenolic acids and their derivatives through glycoside bonds or ester bonds with glucose or polyols. They are further divided into gallotannins containing only galloyl groups, ellagitannins containing hexahydroxydiphenoyl group(s), and hydrolysable tannin oligomers divided into dimers, trimers, and tetramers according to the number of glucose nuclei [16]. Condensed tannins are a class of compounds formed by the carbon-carbon bond polymerization of flavane-3-ol such as catechins or their derivative gallocatechin. Complex tannins are a class of compounds composed of flavane-3-ol, the unit of condensed tannins, and hydrolyzed tannins, which are partially linked by carbon-carbon bonds.
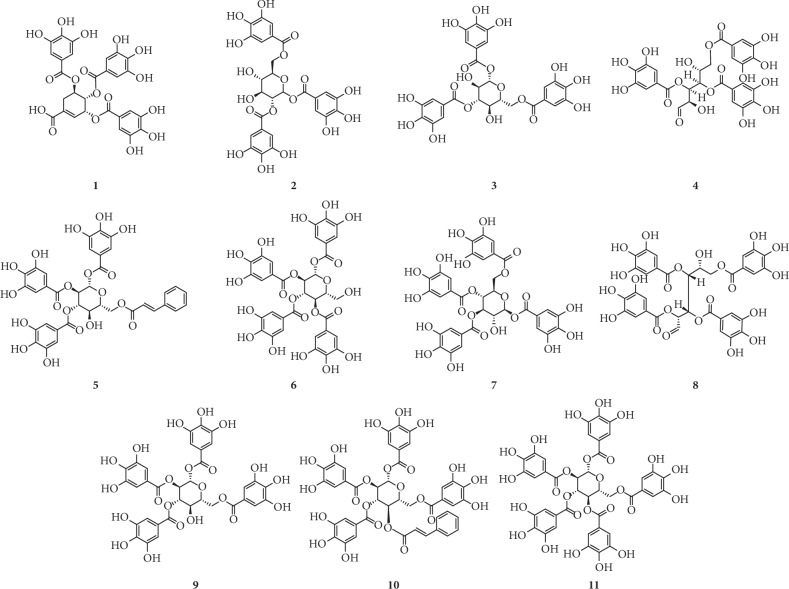
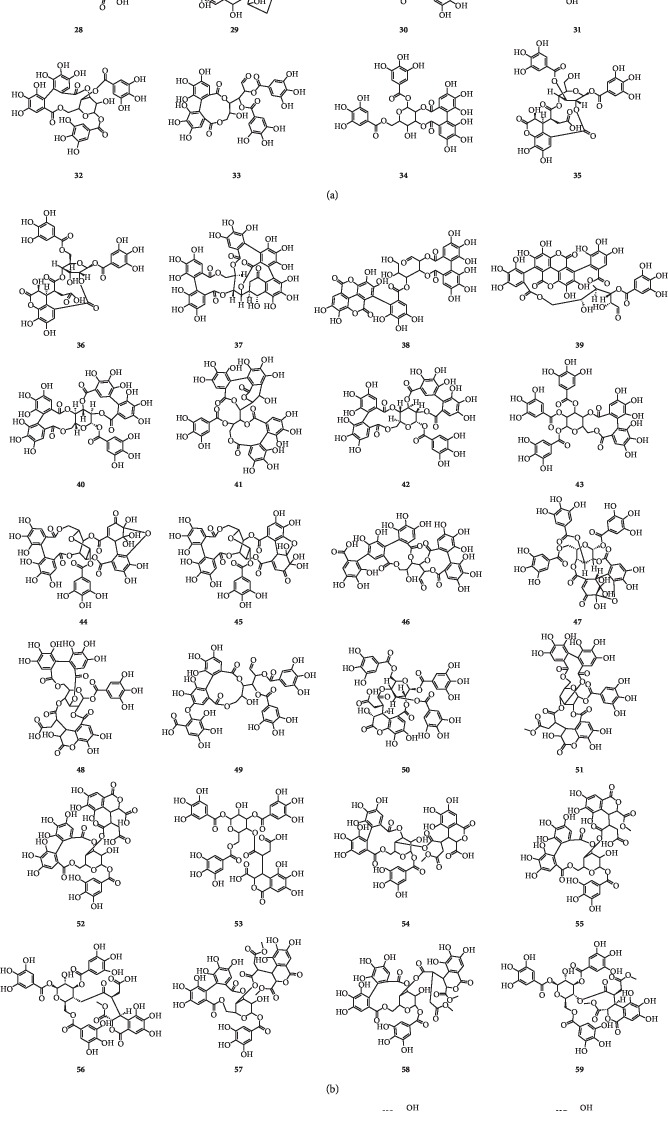
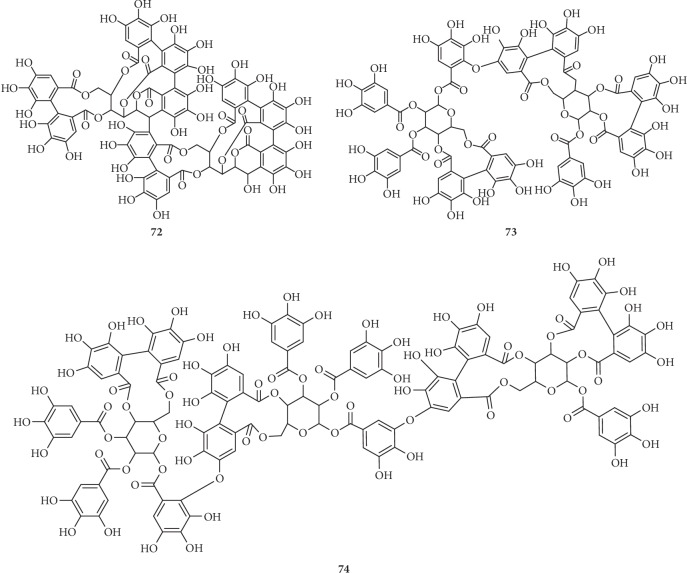
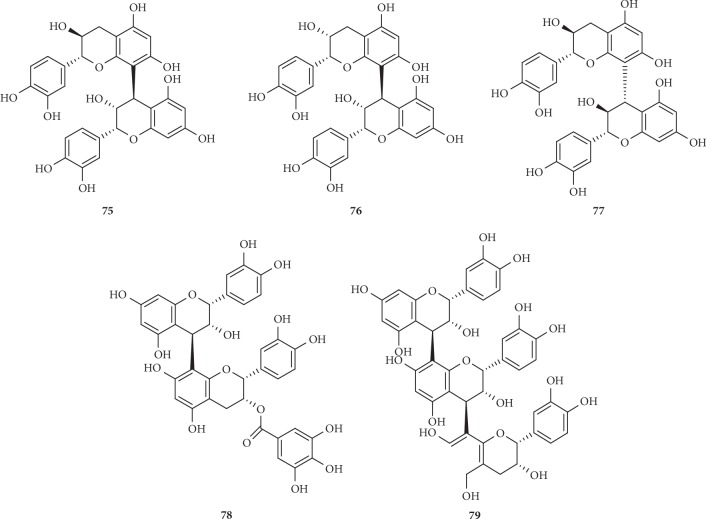
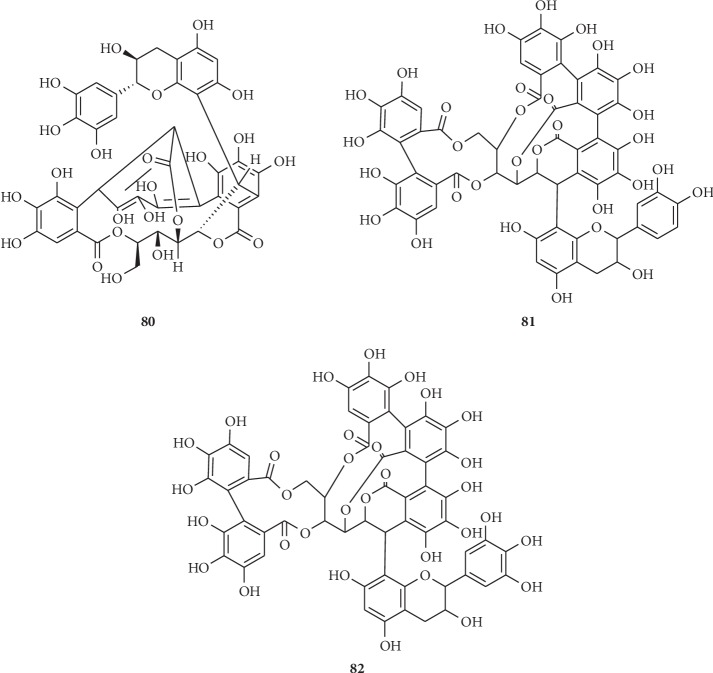
On the basis of the structural differences, they are divided into different types. Compounds 1–74 are hydrolysable tannins. Among them, compounds 1–11 (Figure 1) having only galloyl groups are gallotannins and compounds 12–71 (Figure 2) having hexahydroxydiphenoyl group(s) are ellagitannins. In addition, compounds 72 and 73 (Figure 3) possess two glucose nuclei, and compound 74 (Figure 3) possesses three glucose nuclei. Therefore, they are thought to be hydrolysable tannin dimers and hydrolysable tannin trimers, respectively. Meanwhile, compounds 75–79 (Figure 4) are condensed tannins. Compound 79 is further classified into condensed tannin trimers, and the others are condensed tannin dimers. Compounds 80–82 (Figure 5) possess the unit of condensed tannins and the unit of hydrolyzed tannins which are thought to be complex tannins. The names, corresponding plant resources, and related references of the compounds have been listed in Tables 1–5.
Figure 1.
Structures of compounds 1–11.
Figure 2.
Structures of compounds 12–71.
Figure 3.
Structures of compounds 72–74.
Figure 4.
Structures of compounds 75–79.
Figure 5.
Structures of compounds 80–82.
Table 1.
Gallotannins 1–11 in Figure 1.
| No. | Compound name | Source | Reference |
|---|---|---|---|
| 1 | Tri-O-galloylshikimic acid |
T. chebula Retz. (fruits) T. bellerica (fruits) |
[16] |
| 2 | 1,2,6-Tri-O-galloyl-β-D-glucopyranose | T. chebula Retz. (fruits) | [17] |
| 3 | 1,3,6-Tri-O-galloyl-β-D-glucose | T. citrina (fruits) | [18] |
| T. chebula Retz. (fruits) | [19–21] | ||
| T. catappa Linn. (the bark) | [22] | ||
| T. chebula Retz. (the gall) | [23, 24] | ||
| T. catappa Linn. (fruits) | [25] | ||
| T. chebula Retz. var. tomentella Kurt. (fruits) | [26] | ||
| 4 | 3,4,6-Tri-O-galloyl-D-glucose | T. chebula Retz. (fruits) | [19, 27–29] |
|
T. horrida (fruits) T. chebula Retz. (fruits) |
[16] | ||
| 5 | 1,2,3-Tri-O-galloyl-6-O-cinnamoyl-β-D-glucose | T. chebula Retz. (fruits) | [19] |
| 6 | 1,2,3,4-Tetra-O-galloyl-β-D-glucose | T. chebula Retz. (fruits) | [30] |
| 7 | 1,3,4,6-Tetra-O-galloyl-β-D-glucose | T. chebula Retz. (fruits) | [19, 28] |
|
T. bellerica (fruits) T. horrida (fruits) T. chebula Retz. (fruits) |
[16] | ||
| T. chebula Retz. var. tomentella Kurt. (fruits) | [26] | ||
| 8 | 2,3,4,6-Tetra-O-galloyl-D-glucose | T. arjuna (the bark) | [31] |
| 9 | 1,2,3,6-Tetra-O-galloyl-β-D-glucose |
T. chebula Retz. (fruits) T. bellirica (fruits) |
[32] |
| T. chebula Retz. (fruits) | [19] | ||
| T. bellirica (fruits) | [33] | ||
| 10 | 1,2,3,6-Tetra-O-galloyl-4-O-cinnamoyl-β-D-glucose | T. chebula Retz. (fruits) | [19] |
| 11 | 1,2,3,4,6-Penta-O-galloyl-β-D-glucose | T. chebula Retz. (fruits) | [19, 21, 27, 29, 34] |
|
T. chebula Retz. (fruits) T. bellirica (fruits) |
[32, 33] | ||
| T. arjuna (leaves) | [31] | ||
|
T. horrida (fruits) T. chebula Retz. (fruits) T. bellerica (fruits) |
[16] | ||
| T. chebula Retz. | [35] |
Table 2.
Ellagitannins 12–71 in Figure 2.
| No. | Compound name | Source | Reference |
|---|---|---|---|
| 12 | Galloyl-free chebulinic acid | T. chebula Retz. (fruits) | [36] |
| 13 | 4-O-(4″-O-Galloyl-α-L-rhamnopyranosy) ellagic acid | T. chebula Retz. (fruits) | [16] |
| 14 | 4′-O-Galloy-3,3′-di-O-methylellagic acid 4-O-β-D-xylopyranoside | T. superba (the bark) | [37] |
| 15 | Castalin |
T. catappa Linn. (the bark) T. parviflora (the bark) |
[22] |
| 16 | Terflavin D | T. chebula Retz. (fruits) | [38] |
| 17 | 2,3-(S)-HHDP-6-O-galloyl-D-glucose | T. parviflora (the bark) | [22] |
| 18 | Corilagin | T. chebula Retz. (fruits) | [19, 27, 29, 39–42] |
|
T. chebula Retz. (fruit rinds) T. bellirica (fruit rinds) |
[32] | ||
| T. citrina (fruits) | [18] | ||
| T. chebula Retz. (pericarps) | [18] | ||
|
T. chebula var. parviflora (fruits) T. chebula Retz. (fruits) |
[43] | ||
| T. chebula Retz. | [44] | ||
| T. catappa Linn. (leaves) | [45, 46] | ||
| T. catappa Linn. (bark) | [22] | ||
| T. catappa Linn. (fruits) | [25] | ||
| T. chebula Retz. (fruits) | [28, 47] | ||
|
T. bellerica (fruits) T. chebula Retz. (fruits) T. horrida (fruits) |
[16] | ||
| T. chebula Retz. (fruits and bark) | [48] | ||
| T. ferdinandiana (fruits) | [49] | ||
|
T. bellerica (fruits) T. chebula Retz. (fruits) |
[33] | ||
| 19 | Sanguiin H-4 | T. calamansanai (leaves) | [50, 51] |
| 20 | Gemin D | T. chebula Retz. (fruits) | [19] |
| 21 | Punicacortein A | T. catappa Linn. (fruit peels) | [52] |
| 22 | Chebulanin | T. chebula Retz. (fruits) | [19, 27, 29, 33, 34, 53, 54] |
|
T. chebula Retz. T. bellirica |
[55] | ||
| T. catappa Linn. (leaves) | [45] | ||
|
T. chebula Retz. var. parviflora (fruits) T. chebula Retz. (fruits) |
[43] | ||
|
T. brachystemma (leaves) T. mollis (leaves) |
[56] | ||
|
T. horrida (fruits) T. bellerica (fruits) T. chebula Retz. (fruits) |
[16] | ||
| 23 | Chebumeinin A | T. chebula Retz. (fruits) | [40] |
| 24 | Chebumeinin B | T. chebula Retz. (fruits) | [40, 41] |
| 25 | 4-O-(3″,4″-Di-O-galloyl-α-L-rhamnosyl) ellagic acid | T. catappa Linn. (leaves) | [45] |
| T. chebula Retz. (fruits) | [19] | ||
| T. brownii (the bark) | [57] | ||
|
T. horrida (fruits) T. chebula Retz. (fruits) |
[16] | ||
| 26 | 4-O-(2″,4″-Di-O-galloyl-α-L-rhamnosyl) ellagic acid | T. chebula Retz. (fruits) | [19] |
| 27 | 3′-O-Methyl-4-O-(3″,4″-di-O-galloyl-α-L-rhamnopyranosyl) ellagic acid | T. chebula Retz. (fruits) | [16] |
| 28 | Punicalin | T. catappa Linn. (leaves) | [45, 58] |
| T. arjuna (leaves) | [31] | ||
| T. chebula Retz. (fruits) | [38] | ||
| T. parviflora (the bark) | [22] | ||
| T. triflora (leaves) | [59] | ||
| T. horrida (fruits) | [16] | ||
| T. calamansanai (leaves) | [51] | ||
| 29 | 4,6-O-Isoterchebuloyl-D-glucose | T. macroptera (the bark) | [60] |
| 30 | Pedunculagin | T. chebula Retz. | [61] |
| 31 | Terflavin B | T. catappa Linn. (leaves) | [45, 58] |
| T. macroptera (the bark) | [60] | ||
| T. chebula Retz. (fruits) | [38] | ||
| T. horrida (fruits) | [16] | ||
| 32 | Tercatain | T. catappa Linn. (fruit peels) | [52] |
| T. chebula Retz. (fruits) | [19] | ||
| T. catappa Linn. (the bark) | [22] | ||
| T. catappa Linn. (leaves) | [45, 46] | ||
| 33 | Tellimagrandin I | T. catappa Linn. (bark) | [62] |
| T. muelleri (leaves) | [63] | ||
| T. chebula Retz. (fruits) | [19] | ||
| T. bellerica (fruits) | [16] | ||
| T. catappa Linn. (leaves) | [58] | ||
| T. calamansanai (leaves) | [51] | ||
| 34 | Sanguiin H-1 | T. calamansanai (leaves) | [51] |
| 35 | 1,3-Di-O-galloyl-2,4-chebuloyl-β-D-glucose |
T. horrida (fruits) T. chebula (fruits) |
[16] |
| 36 | 1,6-Di-O-galloyl-2,4-chebuloyl-β-D-glucose |
T. horrida (fruits) T. chebula Retz. (fruits) |
[16] |
| T. chebula Retz. | [64] | ||
| 37 | Castalagin | T. catappa Linn. (the bark) | [62] |
|
T. parviflora (the bark) T. catappa Linn. (the bark) |
[22] | ||
| 38 | Terflavin C | T. chebula Retz. (fruits) | [38] |
| 39 | 2-O-Galloylpunicalin | T. calamansanai (leaves) | [50] |
| T. arjuna (the bark) | [31] | ||
| T. triflora (leaves) | [59] | ||
| 40 | 2,3,4,6-bis-Hexahydroxydiphenyl-1-galloyl-β-glucose | T. arjuna (leaves) | [31] |
| 41 | Casuarinin | T. catappa Linn. (the bark) | [22, 51, 62] |
| T. chebula Retz. (fruits) | [27, 29, 40, 65] | ||
| T. arjuna Linn. (the bark) | [66, 67] | ||
| 42 | 1(α)-O-Galloylpedunculagin | T. calamansanai (leaves) | [51] |
| 43 | Tellimagrandin II | T. catappa Linn. (the bark) | [62] |
| T. catappa Linn. (leaves) | [45] | ||
| T. calamansanai (leaves) | [51] | ||
| 44 | Geraniin | T. chebula Retz. (fruits) | [68] |
| T. catappa Linn. (leaves) | [58] | ||
| 45 | Granatin B | T. catappa Linn. (leaves) | [58] |
| 46 | Praecoxin A | T. calamansanai (leaves) | [51] |
| 47 | Terchebin | T. chebula Retz var. tomentella Kurt. (fruits) | [26] |
| 48 | Chebulagic acid | T. chebula Retz. (fruits) | [19, 21, 27, 28, 39–41, 43, 53, 68–75] |
|
T. chebula Retz. (fruit rinds) T. bellirica (fruit rinds) |
[32] | ||
| T. citrina (fruits) | [18] | ||
| T. catappa Linn. (leaves) | [45, 46, 58] | ||
| T. chebula Retz. (pericarps) | [76] | ||
| T. muelleri (leaves) | [63] | ||
| T. chebula Retz. | [44, 76] | ||
| T. chebula Retz. (Galls) | [23, 24] | ||
|
T. bellerica (fruits) T. chebula Retz. (fruits) T. horrida (fruits) |
[16] | ||
| T. chebula Retz. (fruits and bark) | [48] | ||
|
T. arjuna (leaf, stem, root, bark, fruit) T. bellerica (leaf, stem, root, bark, fruit) T. chebula Retz. (leaf, stem, root, bark, fruit) |
[77] | ||
| T. bellerica (fruits) | [33] | ||
| 45 | Granatin B | T. catappa Linn. (leaves) | [58] |
| 46 | Praecoxin A | T. calamansanai (leaves) | [51] |
| 47 | Terchebin | T. chebula Retz var. tomentella Kurt. (fruits) | [26] |
| 48 | Chebulagic acid | T. chebula Retz. (fruits) | [19, 21, 27, 28, 39–41, 43, 53, 68–75] |
|
T. chebula Retz. (fruit rinds) T. bellirica (fruit rinds) |
[32] | ||
| T. citrina (fruits) | [18] | ||
| T. catappa Linn. (leaves) | [45, 46, 58] | ||
| T. chebula Retz. (pericarps) | [76] | ||
| T. muelleri (leaves) | [63] | ||
| T. chebula Retz. | [44, 77] | ||
| T. chebula Retz. (Galls) | [23, 24] | ||
|
T. bellerica (fruits) T. chebula Retz. (fruits) T. horrida (fruits) |
[16] | ||
| T. chebula Retz. (fruits and bark) | [48] | ||
|
T. arjuna (leaf, stem, root, bark, fruit) T. bellerica (leaf, stem, root, bark, fruit) T. chebula Retz. (leaf, stem, root, bark, fruit) |
[78] | ||
| T.bellerica (fruits) | [33] | ||
| T. chebula Retz var. tomentella Kurt. (fruits) | [26] | ||
| 49 | Rugosin B | T. calamansanai (leaves) | [51] |
| 50 | Chebulinic acid | T. chebula Retz. (fruits) | [19, 20, 27–30, 36, 39, 43, 53, 65, 68, 70, 73, 79–84] |
|
T. chebula Retz. (fruits) T. bellirica Roxb. (fruits) |
[32] | ||
| T. chebula Linn. (pericarps) | [85] | ||
| T. chebula Retz. (pericarps) | [76] | ||
| T. chebula Retz. | [44] | ||
| T. chebula Retz. (Galls) | [23, 24] | ||
|
T. bellerica (fruits) T. chebula Retz. (fruits) T. horrida (fruits) |
[16] | ||
| T. chebula Retz. (fruits and the bark) | [48] | ||
|
T. arjuna (leaf, stem, root, bark, fruit) T. bellerica (leaf, stem, root, bark, fruit) T. chebula Retz. (leaf, stem, root, bark, fruit) |
[78] | ||
|
T. bellereica (fruits) T. chebula Retz. (fruits) |
[33] | ||
| T. chebula Retz var. tomentella Kurt. (fruits) | [26] | ||
| 51 | Methyl chebulagate | T. chebula Retz. (fruits) | [19] |
| 52 | Neochebulagic acid | T. chebula Retz. (fruits) | [19] |
| 53 | Neochebulinic acid | T. chebula Retz. (fruits) | [27, 29, 43] |
| T. chebula Retz. var. tomentella Kurt. (fruits) | [26] | ||
| 54 | 6′-O-Methyl neochebulagate | T. chebula Retz. (fruits) | [19] |
| 55 | Methyl neochebulagate | T. chebula Retz. (the gall) | [24] |
|
T. horrida (fruits) T. chebula Retz. (fruits) |
[16] | ||
| 56 | Methyl neochebulinate | T. chebula Retz. (fruits) | [19] |
|
T. horrida (fruits) T. chebula Retz. (fruits) |
[16] | ||
| T. chebula Retz. var. tomentella Kurt. (fruits) | [26] | ||
| 57 | Dimethyl neochebulagate | T. chebula Retz. (fruits) | [19] |
| 58 | Dimethyl 4′-epineochebulagate | T. chebula Retz. (fruits) | [19] |
| 59 | Dimethyl neochebulinate | T. chebula Retz. (fruits) | [19] |
| 60 | Grandinin | T. catappa Linn. (the bark) | [22] |
| 61 | Calamanin A | T. calamansanai (leaves) | [51] |
| 62 | α-Punicalagin | T. oblongata (leaves) | [86] |
| T. myriocarpa Heurck (leaves) | [87] | ||
| T. chebula Retz. (fruits) | [28] | ||
| 63 | β-Punicalagin | T. oblongata (leaves) | [86] |
| T. myriocarpa Heurck (leaves) | [87] | ||
| 64 | Terchebulin | T. macroptera (roots) | [88] |
| T. chebula Retz. (fruits) | [27, 29, 38] | ||
| T. laxiflora (wood) | [89, 90] | ||
| 65 | Iso/terchebulin | T. catappa Linn. (the bark) | [62] |
| T. macroptera (the bark) | [60] | ||
| T. chebula Retz. (Galls) | [23, 24] | ||
| 66 | Terflavin A | T. catappa (the bark) | [62] |
| T. macroptera (the bark) | [60] | ||
| T. chebula Retz. (fruits) | [19, 38] | ||
| T. macroptera (roots) | [88] | ||
| T. catappa Linn. (leaves) | [58] | ||
| 67 | Eucalbanin A | T. muelleri (leaves) | [63] |
| 68 | Rugosin A | T. calamansanai (leaves) | [51] |
| 69 | tergallagin | T. catappa Linn. (leaves) | [58] |
| 70 | 1-α-O-Galloylpunicalagin | T. calamansanai (leaves) | [50, 51] |
| 71 | Calamansanin | T. calamansanai (leaves) | [51] |
Table 3.
Hydrolysable tannin polymers 72–74 in Figure 3.
Table 4.
Condensed tannins 75–79 in Figure 4.
| No. | Compound name | Source | Reference |
|---|---|---|---|
| 75 | Procyanidin B1 | T. tomentosa (the bark) | [91] |
| T. catappa Linn. (the bark) | [22] | ||
| 76 | Procyanidin B2 | T. tomentosa (the bark) | [91] |
| 77 | Procyanidin B3 | T. tomentosa (the bark) | [91] |
| 78 | 3′-O-Galloyl procyanidin B-2 | T. catappa Linn. (the bark) | [22] |
| 79 | Procyanidin C1 | T. tomentosa (bark) | [91] |
Table 5.
Complex tannins 80–82 in Figure 5.
3.2. MS Data of Tannins
The MS data of the tannins from the genus Terminalia (family Combretaceae) are shown in Table 6 as summarized. According to the compiled MS data, this review provides a useful and fast way for the identification of tannins.
Table 6.
The MS spectral data of compounds 1–82 except those which have no reported MS data.
| No. | Compound name | Molecular formula | Ion source | [M-H]– | Fragments | Reference |
|---|---|---|---|---|---|---|
| 1 | Tri-O-galloylshikimic acid | C28H22O17 | ESI | 628.9 | 477 (15), 325 (1), 169 (100) | [16] |
| 2 | 1,2,6-Tri-O-galloyl-β-D-glucose | C27H24O18 | 635 | 465 (100), 313 (20), 169 (10) | [92] | |
| 635.093 | 465.0479, 313.0427, 169.0061 | [93] | ||||
| 3 | 1,3,6-Tri-O-galloylglucose | C27H24O18 | 635.0895 | 465.06714 [C20H17O13]−, 211.02463 [C9H7O6]−, 169.01404 [C7H5O5]−, 125.02427 [C6H5O3]− | [94] | |
| 5 | 3,4,6-Tri-O-galloyl-D-glucose | C27H24O18 | ESI | 635.0882 | 169 (9), 235 (2), 271 (4), 295 (14), 313 (9), 405 (5), 423 (30), 465 (68), 483 (100), 617 (11) | [95] |
| 6 | 1,2,3,4-Tetra-O-galloyl-β-D-glucose | C34H28O22 | ESI | 787 | 617, 393, 169 | [32] |
| 7 | 1,3,4,6-Tetra-O-galloyl-β-D-glucose | C34H28O22 | 787 | 635, 617 | [96] | |
| 8 | 2,3,4,6-Tetra-O-galloyl-D-glucose | C34H28O22 | ESI | 787 | 617, 635 | [97] |
| ESI | 787.0914 | 635.0902, 617.0795,465.0709 | [98] | |||
| 787.0989 | 169.0158, 295.0297, 313.0570, 447.1352, 465.1383, 483.0638, 617.1949, 635.2112 | [99] | ||||
| 787.0996 | 617.0902 [M-H-GA]−, 447.0732 [M-H-2GA]−, 295.0418 [M-H-2GA-C7H4O4]−, 169.0140 [GA-H]− | [100] | ||||
| ESI | 787.1079 | 617.0834, 465.0731, 313.0606, 169.0177 | [101] | |||
| ESI | 787 | 635 [M-H-152]−, 617 [M-H-170]−, 483 [M-H-304]−, 465 [M-H-322]−, 447 [M-H-340]−, 169 [GA-H]− | [102] | |||
| 9 | 1,2,3,6-Tetra-O-galloyl-β-D-glucose | C34H28O22 | ESI | 787.0986 | 295 (1), 403 (2), 421 (0.4), 429 (1), 447 (2), 465 (3), 529 (0.2), 573 (4), 617 (100), 635 (31) | [95] |
| 11 | 1,2,3,4,6-Penta-O-galloyl-β-D-glucose | C41H32O26 | ESI | 939.1101 | 329 (0.4), 439 (0.4), 447 (0.2), 515 (0.2), 599 (1), 601 (0.2), 617 (3), 725 (1), 769 (100), 787 (8) | [95] |
| 939.111 | 787.1282 [M-H-C7H4O4]−, 769.1003 [M-H-GA]−, 617.0884 [M-H-GA-C7H4O4]−, 447.0593 [M-H-2GA-C7H4O4]−, 259.0248 [M-H-4GA]−, 169.0140 [GA-H]− | [103] | ||||
| ESI | 939 | 769[M-H-GA]–, 617[M-H + H2O-2GA]– | [104] | |||
| ESI | 939.11090 | 769.1, 617.1, 465.1, 447.1, 295.0, 169.0 | [105] | |||
| ESI | 939 | 769, 787, 617 | [97] | |||
| ESI | 939 | 939[M-H]−, 769[M-H-GA]−, 617[M-H + H2O-2GA]−, 447[M-H + H2O-3GA]−, 169[GA]−, 125[GA-CO2]− | [50] | |||
| ESI | 939 | 787, 769, 617, 599, 447 | [106] | |||
| 939.112 | 169, 617, 769 | [107] | ||||
| 939 | 469, 769, 629, 617, 465, 313, 169, 125 | [108] | ||||
| ESI | 939 | 787, 769, 635, 617 | [109] | |||
| ESI | 939.1104 | 787 [M-H-C7H4O4]−, 769 [M-H-C7H6O5]−, 635 [M-H-C14H8O8]−, 617 [M-H-C14H10O9]−, 465 [M-H-C21H14O13]−, 447 [M-H-C21H16O14]−, 313 [M-H-C28H18O17]−, 295 [M-H-C28H19O18]−, 169 [M-H-C34H26O21]−, 125 [C35H26O23]− | [110] | |||
| ESI | 939.3 | 169.0, 393.1, 769.2 | [111] | |||
| 13 | 4-O-(4″-O-Galloyl-α-L-rhamnopyranosyl) ellagic acid | C27H20O16 | ESI | 599 | 447 (23), 429 (2), 301 (100), 297 (6), 169 (3) | [16] |
| 15 | Castalin | C27H20O18 | 631 | 613 (100) | [112] | |
| ESI | 631.1 | 301 [EA-H]–, 331.0 [Galloylglu-H]−, 481.0 [HHDP-glu-H]− | [113] | |||
| 631 | 479, 317, 301 | [114] | ||||
| ESI | 631.0586 | 461.033 (71) [M-H–C7H4O4–H2O]−, 445.0461 (17) [M-H-C7H4O5-H2O]−, 300. 9986 (78) [ellagic acid]−, 273.0030, 245.0092 (44), 229.0142(45), 169.0143 (100) [GA]−, 125.0254 (30) | [115] | |||
| 18 | Corilagin | C27H22O18 | 633.0734 | 470.9841 | [116] | |
| 633.0762 | 463.0793, 300.9986, 169.0133 | [117] | ||||
| 633.0725 | 463 (7), 301 (100), 275 (30), 245 (5), 169 (7), 125 (4) | [118] | ||||
| ESI | 633 | 476, 454 | [32] | |||
| 19 | Sanguiin H-4 | C27H22O18 | ESI | 633.0719 | 327, 343, 177 | [119] |
| ESI | 633 | 481, 301, 275, 249, 635, 617, 465, 447, 353, 339, 321, 315, 303, 277, 257, 229, 211, 259, 231 | [120] | |||
| 22 | Chebulanin | C27H24O19 | 651 | 633, 481, 463, 291, 275 | [100] | |
| 651 | 481 [M-galloyl]−, 651 [M-H]−, 1303 [2M-H]− | [121] | ||||
| 651 | 633 [M-H-H2O]−, 405, 300, 275 | [122] | ||||
| 23 | Chebumeinin A | C29H30O18 | 669 | 366.9 | [123] | |
| 24 | Chebumeinin B | C28H28O19 | 669 | 366.8 | [123] | |
| 25 | 4-O-(3″,4″-Di-O-galloyl-α-L-rhamnopyranosyl) ellagic acid | C34H24O20 | ESI | 751.1 | 599 (22), 581 (6), 449 (30), 411 (4), 300 (100), 297 (8), 169 (6),151 (2) | [16] |
| 27 | 3′-O-Methyl-4-O-(3″,4″-di-O-galloyl-α-L-rhamnopyranosyl) ellagic acid | C35H26O20 | ESI | 765.2 | 613 (32), 595 (100), 461 (5), 449 (30), 443 (41), 425 (10), 315 (31), 169 (56) | [16] |
| 28 | Punicalin | C34H22O22 | 781 | 601, 301 | [124] | |
| ESI | 781.0531 | 721, 601, 271 | [125] | |||
| ESI | 781.5 | 299.4 | [126] | |||
| 781 | 721, 601, 557, 451, 299 | [100] | ||||
| 781 | 601, 299 | [127] | ||||
| 30 | Pedunculagin | C34H24O22 | 783.0673 | 300.9975 | [116] | |
| ESI | 783.07 | 1567.14 [2M-H]−, 391.03 [M-2H]2– | [128] | |||
| ESI | 783 | 481, 301, 257 | [129] | |||
| ESI | 783 | 391 [M-2H]2–, 783 [M-H]−, 1567 [2M-H]– | [121] | |||
| ESI | 783.2 | 783.2, 481.1, 301.0 | [130] | |||
| ESI | 783.0686 | 481.0516, 300, 9975 | [131] | |||
| 783 | 481, 301, 244 | [114] | ||||
| 783.068 | 481, 301, 275 | [125] | ||||
| ESI | 783.0692 | 935.0790, 613.0463, 300.9990 | [132] | |||
| ESI | 783.0679 | 481, 301 | [133] | |||
| 783.0699 | 481.0606, 391.0307,300.9999, 275.0191 | [134] | ||||
| 783 | 301, 481, 275 | [97] | ||||
| ESI | 783.06 | 481.06, 301.00, 275.02 | [135] | |||
| 31 | Terflavin B | C34H24O22 | ESI | 783 | 631 (11), 451 (100), 299 (1) | [16] |
| 33 | Tellimagrandin I | C34H26O22 | ESI | 785.08 | 301.00, 275.02, 169.01 | [135] |
| 784.6, 450.9, 402.6, 391.7, 214.7 | [136] | |||||
| ESI | 785 | 301, 483, 615 | [137] | |||
| ESI | 785 | 301, 483, 633, 615, 463, 419 | [97] | |||
| ESI | 785.0836 | 301, 483, 633 | [133] | |||
| ESI | 785.0866 | 633, 481,301, 275, 222 | [138] | |||
| ESI | 785 | 392 [M–2H]2–, 785 [M–H]−, 1571 [2M–H] − | [121] | |||
| ESI | 785.084 | 633.07, 615.06, 483.08, 300.99, 275.02 | [139] | |||
| ESI | 785 | 615,483,301 | [129] | |||
| 35 | 1,3-Di-O-galloyl-2,4-chebuloyl-β-D-glucose | C34H28O23 | ESI | 802.9 | 337 (100), 319 (47), 293 (41), 275 (61), 169 (8) | [16] |
| 37 | Castalagin | C41H26O26 | ESI | 933 | 915, 631, 451, 301 | [140] |
| ESI | 933.0644 | 915.0509, 631.0575, 479.0464, 461.0377, 300.9991 | [141] | |||
| ESI | 933 | 915, 631, 613, 569, 493, 301 | [142] | |||
| 915, 783, 631, 613, 569, 467, 493, 323, 301, 146 | ||||||
| ESI | 933 | 915 (95), 631 (100), 425 (20), 301 (5) | [112] | |||
| 933 | 181.1, 466.0 | [113] | ||||
| ESI | 933.0649 | 466.0299, 300.9968 | [134] | |||
| ESI | 933 | 915, 631, 613, 569 | [106] | |||
| 933 | 915, 871, 569, 301 | [114] | ||||
| ESI | 933.1 | 783.1, 631.1, 451.1, 301.0 | [130] | |||
| ESI | 466 [M-2H]2–, 933 [M-H]–, 933 [2M-2H]2–, 1867 [2M-H]– | [121] | ||||
| ESI | 935, 915, 613, 301 | [143] | ||||
| ESI | 933 | 631, 451, 301 | [144] | |||
| 39 | 2-O-Galloylpunicalin | C41H26O26 | 933 | 781, 721, 601 | [124] | |
| 41 | Casuarinin | C41H28O26 | ESI | 935.0796 | 785.1, 633.1, 483.1, 451.0, 425.0, 301.0, 275.0, 169.0 | [105] |
| ESI | 935 | 917, 633, 783, 301 | [137] | |||
| ESI | 935 | 467 [M-2H]2–, 935 [M-H]–, 1871 [2M-H]– | [121] | |||
| 935.076 | 633.075, 300.9999 | [145] | ||||
| 43 | Tellimagrandin II | C41H30O26 | ESI | 937.0953 | 301.0, 275.0, 249.0, 169.0 | [105] |
| ESI | 937 | 767, 741, 465, 301 | [97] | |||
| ESI | 937 | 785, 767, 635, 465, 301 | [106] | |||
| ESI | 937.0945 | 785, 633, 483, 301, 278, 237 | [138] | |||
| 44 | Geraniin | C41H28O27 | ESI | 951.0747 | 907.0849, 781.0537, 605.0788, 479, 425.0251, 298, 273.0042 | [141] |
| ESI | 951.0762 | 933.0717 (100) [M-H-H2O]–, 300.9991 (52), 169.0141 (2) | [115] | |||
| 951.6751 | 463.0505, 301.9987, 273.0040, 169.0132 | [146] | ||||
| ESI | 951 | 457 [M-2H2O-2H]2–, 466 [M-H2O-2H]2–, 951 [M-H]–, 1903 [2M-H]– | [121] | |||
| ESI | 951.0721 | 933.0770 [M-H-H2O]–, 300.9990, 169.0144 | [147] | |||
| ESI | 951.07 | 951.07 [M-H]–, 466.03 [M-2H]2–, 300.99 [EA-H]–, 633.07 [M-318-H]– | [128] | |||
| 45 | Granatin B | C41H27O27 | ESI | 951.0719 | 933 (7), 463 (20), 301 (100), 273 (32), 245 (17), 229 (3), 167 (3) | [118] |
| ESI | 951 | 933, 915, 301 | [148] | |||
| ESI | 951.0745 | 933.0604, 613.2044, 300.9980 | [131] | |||
| 46 | Praecoxin A | C41H28O27 | ESI | 951 | 783, 605, 889, 481, 301 | [149] |
| 48 | Chebulagic acid | C41H30O27 | ESI | 953 | 476, 169 | [32] |
| 953 | 935, 807, 633, 481, 463, 319, 301 | [100] | ||||
| ESI | 953 | 476 [M-2H]2–, 953 [M-H]– | [121] | |||
| 49 | Rugosin B | C41H30O27 | ESI | 953.0902 | 909.1, 785.1, 766.1, 597.0, 301.0, 275.0, 249.0, 169.0 | [105] |
| 953.2 | 909 (100), 883 (1), 785 (5) | [150] | ||||
| 50 | Chebulinic acid | C41H32O27 | 955 | 477 [M-2H]2–, 169 | [32] | |
| 955 | 937, 803, 785, 641, 607, 465, 337, 275, 131 | [100] | ||||
| 955 | 477 [M-2H]2–, 955 [M-H]– | [121] | ||||
| 955.1018 | 785, 169 | [151] | ||||
| 52 | Neochebulagic acid | C41H32O28 | 971 | 953 [M-H-H2O]–, 935 [M-H-H2O-H2O]–, 467 [M-2H-H2O-H2O]2–, 301 | [122] | |
| 56 | Methyl neochebulinate | C42H36O28 | ESI | 987.2 | 635 (100), 465 (1), 351 (3), 169 (1) | [16] |
| 60 | Grandinin | C46H34O30 | ESI | 1065 | 1047 (50), 1029 (50), 975 (100), | [112] |
| 62 | α-Punicalagin | C48H27O30 | ESI | 1083.056 | 781 (40), 601 (35), 575 (20), 301 (100), 275 (7), 249 (5) | [118] |
| 1083 | 781 (60), 601 (100), 575 (22) | [152] | ||||
| 1083.059 | 781.6071, 601.3680, 301.4796 | [131] | ||||
| ESI | 1083 | 781, 541, 301 | [153] | |||
| 63 | β-Punicalagin | C48H27O30 | ESI | 1083.054 | 1083 (43), 781 (55), 719 (29), 601 (86), 575 (29), 301 (100), 275 (43), 249 (15) | [118] |
| 1083 | 781 (35), 601 (100), 575 (15) | [152] | ||||
| 1083.059 | 781.6071, 601.3680, 301.4796 | [131] | ||||
| ESI | 1083 | 781, 541, 301 | [153] | |||
| 67 | Eucalbanin A | C48H30O30 | ESI | 1085 | 765, 633, 473 | [137] |
| ESI | 1085 | 933, 783, 765, 739, 633, 597, 469, 407 | [97] | |||
| ESI | 1085.074 | 783.07, 633.07, 450.99, 300.99 | [139] | |||
| 68 | Rugosin A | C48H34O31 | ESI | 1105.101 | 530.0, 891.1, 301.0, 169.0 | [105] |
| ESI | 1105.3 | 1061 (100), 937 (5), 935 (10), 917 (3) | [150] | |||
| 75 | Procyanidin B1 | C30H26O12 | 577.1344 | 577, 451, 425, 407, 289, 245, 161, 125 | [154] | |
| 577.16 | 287, 289, 425, 451 | [155] | ||||
| ESI | 577.1351 | 425.0875 (100), 451.1030 (90), 289.0713 (60), 407.0767 (60), 299.0556 (30), 287.0557 (10) | [156] | |||
| 76 | Procyanidin B2 | C30H26O12 | 577.152 | 287, 289, 425, 451 | [155] | |
| ESI | 577 | 451 (23.7), 425 (100), 407 (69.6), 289 (29.0), 408 (17.7), 407 (100), 289 (100), 281 (85.7), 256 | [157] | |||
| 77 | Procyanidin B3 | C30H26O12 | ESI | 577.1331 | 407 (75), 289 (81), 245 (67) | [158] |
| ESI | 577.1375 | 425, 407, 289, 287 | [159] | |||
| 78 | 3′-O-Galloyl procyanidin B2 | C37H30O16 | 729.1458 | 407.0766, 289.0716 | [160] | |
| 729.1471 | 303.05055, 364.58214, 441.08203 | [161] | ||||
| 79 | Procyanidin C1 | C45H38O18 | ESI | 865.1964 | 739.1640, 575.1171 | [162] |
| ESI | 865.195 | 865 (37), 695 (100), 577 (1), 407 (64), 289 (42) | [158] | |||
| MALDI | 865.191 | 287, 289, 575, 577, 713, 425, 739, 451, 413 | [155] | |||
| ESI | 865 | 675.3, 528.6 | [163] | |||
| ESI | 865.1984 | 739, 713, 577, 289 | [119] | |||
| ESI | 865.1985 | 739.1722, 577.1378, 451.1054, 407.0793, 287.0575, 245.0460 | [164] | |||
| 81 | Acutissimin A | C56H38O31 | ESI | 1205 | 1205, 915, 613, 602, 301 | [143] |
3.3. Fragmentation Pattern
3.3.1. Gallotannins
Most gallotannins produce major fragment ions [M-H-170]− and [M-H-152]−, which indicate the loss of gallic acid and galloyl residue. In addition, other fragment ions such as [M-H-170]−, [M-H-170-152]−, and [M-H-170-152-152]− are produced owing to the sequential losses of galloyl group and gallic acid.
Compound 7 (Figure 6) gave the [M-H]− ions at m/z 787 and displayed a fragmentation pattern similar to the successive neutral losses of gallic acids (170 Da) and galloyl radicals (152 Da). Due to the limited mass spectrometry information, it was difficult to distinguish the link position between galloyl groups and glucosyl unit [96].
Figure 6.
Fragmentation of compound 7.
Compound 8 (Figure 7) is characterized by fragment ions at m/z 635, corresponding to the loss of a galloyl residue ([M-H-152]−) and at m/z 617 owing to the loss of a gallic acid group ([M-H-170]−) [97].
Figure 7.
Fragmentation of compound 8.
Compound 11 (Figure 8) with the [M-H]− ion at m/z 939 and m/z 469 [M-2H]2−, showed typical fragments at m/z 769 [M-H-170]−, m/z 617 [M-H-170-152]−, m/z 465 [M-H-170-152-152]−, and m/z 313 [M-H-170-152-152-152]− which corresponded to the sequential losses of galloyl group and gallic acid [108].
Figure 8.
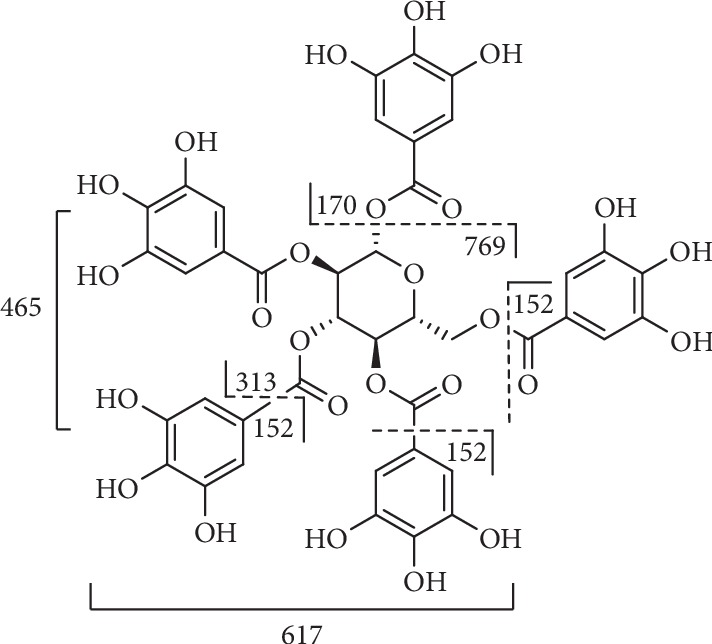
Fragmentation of compound 11.
3.3.2. Ellagitannins
Most ellagitannins produce major fragment ions [M-H-170]−, [M-H-170-162]−, and [M-H-302]−, which indicate the loss of gallic acid, galloylglucose group, and HHDP group. In addition, other fragment ions such as 151, 169, and 301 confirm the existence of galloyl group, gallic acid, and HHDP group, respectively.
Compound 18 (Figure 9) presented [M-H]− at m/z 633.0762 and MS2 fragments at m/z 463.0793 [M-H-152-H2O]−, which is consistent with sequential losses of galloyl and H2O and at m/z 300.9986 [M-H-152-180]− owing to the loss of a galloyl unit with a hexose [117].
Figure 9.
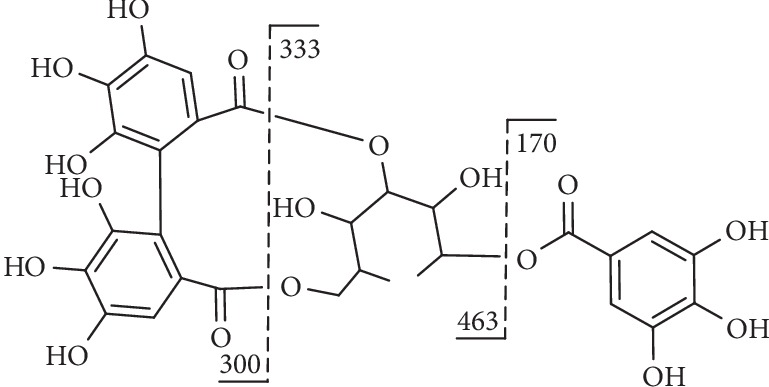
Fragmentation of compound 18.
Compound 37 (Figure 10) displayed [M-H]− at m/z 933 and MS2 ion at m/z 631 resulting from the loss of HHDP and presented MS3 ions at m/z 451 owing to the loss of glucosyl moiety and at m/z 301 which corresponded to the loss of galloyl-glucosyl moiety from the parent MS2 ion at m/z 631 [144].
Figure 10.
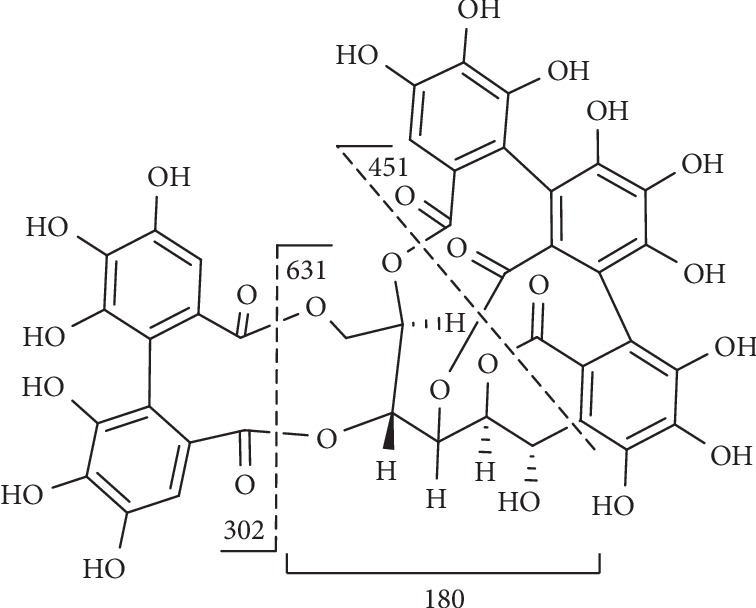
Fragmentation of compound 37.
Compound 39 (Figure 11) had an [M-H]− ion at m/z 933 and three mass fragments: one at 601 ([M-H-332]−) which corresponded to the loss of a galloylglucose unit and two others at m/z 781 ([M-H-152]−) which corresponded to the presence of a galloyl group and at m/z 721 after the cross-ring fragmentation of glucose ([M-H-152-60]−) [124].
Figure 11.
Fragmentation of compound 39.
Compound 42 (Figure 12) displayed molecular anions at m/z 935 and produced fragments at m/z 633 ([M-H-302]−), corresponding to the loss of an HHDP group and at m/z 301 ([M-H-634]−), indicating the presence of HHDP (302 Da), gallic acid (170 Da), and glucosyl (162 Da) groups [129].
Figure 12.
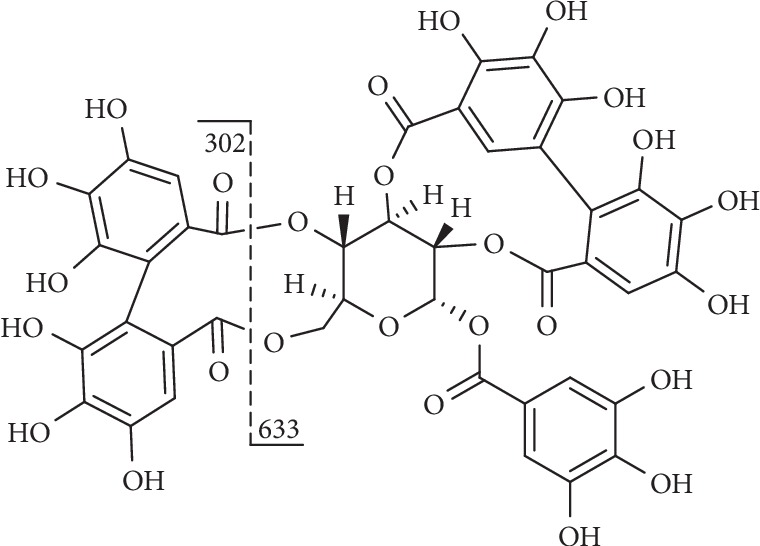
Fragmentation of compound 42.
Compounds 62 and 63 (Figure 13) are isomers, which had the same fragmentation behaviors, presented a same [M-H]− ion at m/z 1083, and further produced ions at m/z 781 ([M-H-302]−), m/z 601 ([M-H-302-180]−), and m/z 301, demonstrating the existence of HHDP and gallagic acid groups [165].
Figure 13.
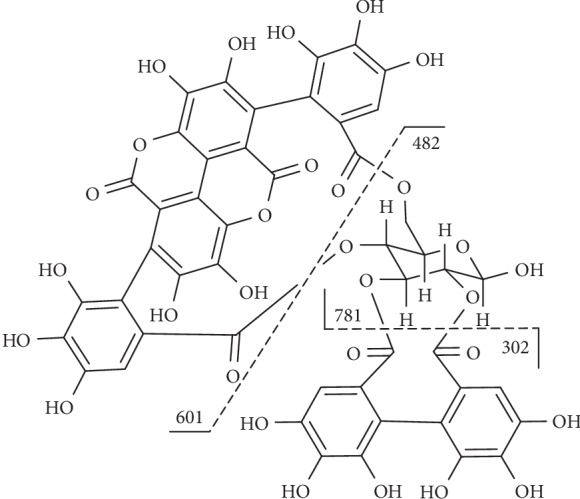
Fragmentation of compounds 62 and 63.
3.3.3. Condensed Tannins
Structurally significant product ions were produced by cleavages between monomeric subunits, which contain quinone methide (QM), heterocyclic ring fission (HRF), and retro-Diels–Alder (RDA) fragment ions.
QM fragmentation cleaves the single bond between subunits in B-type procyanidins to form a single quinone resulting in two possible product ions.
A second important structural fragmentation pathway for deprotonated procyanidins is heterocyclic ring fission (HRF), which results in the elimination of 1,3,5-trihydroxybenzene ([M-H-126]−).
Retro-Diels–Alder (RDA) fragmentation was distinguished by elimination of hydroxyvinyl benzenediol ([M-H-152]−), an extra water molecule ([M-H-152-18]−) simultaneously.
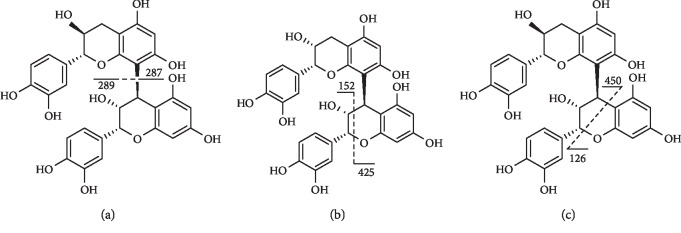
The dimeric procyanidins occur as the B-type procyanidins in nature, which contain four major isomers such as B1, B2, B3, and B4. We have sorted out compounds 75–77 which presented in Terminalia Linn. The three compounds presented the specific fragments of m/z 425 and 407, which corresponded to the characteristic fragmentations of procyanidin B-type dimmers [166].
Compound 76 (Figure 14) presented an [M-H]− ion at m/z 577, with fragment ions at 425 ([M-H-152]−), originated from Retro Diels–Alder (RDA) fragmentation of the heterocyclic ring. The fragment at m/z 407 ([M-H-170]−) resulted from both RDA rearrangement and loss of water molecule [155].
Figure 14.
Fragmentation of compound 76. (a) QM, (b) RDA, and (c) HRF.
Compound 77 had an [M-H]− ion at m/z 577 which presented a Retro-Diels–Alder (RDA) product with a neutral loss of 152 ([M-H-152]−) and subsequently loss of a water molecule [M-H-152-18]− [158].
Compound 79 (Figure 15) gave the [M-H]− ion at m/z 865 and showed fragment ions at m/z 287/577 and m/z 575/289 due to QM fragmentation. The fragment at m/z 713/695 corresponded to RDA fragmentation and at m/z 425/407 owing to RDA fragmentation of the QM product ion of m/z 577. It also formed ions of m/z 739, m/z 451, and m/z 413 through HRF fragmentation [155].
Figure 15.
Fragmentation of compound 79. (a) QM, (b) RDA, and (c) HRF.
3.3.4. Complex Tannins
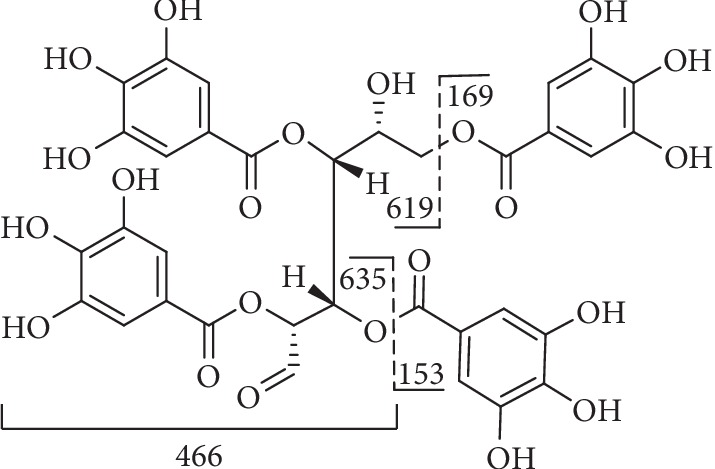
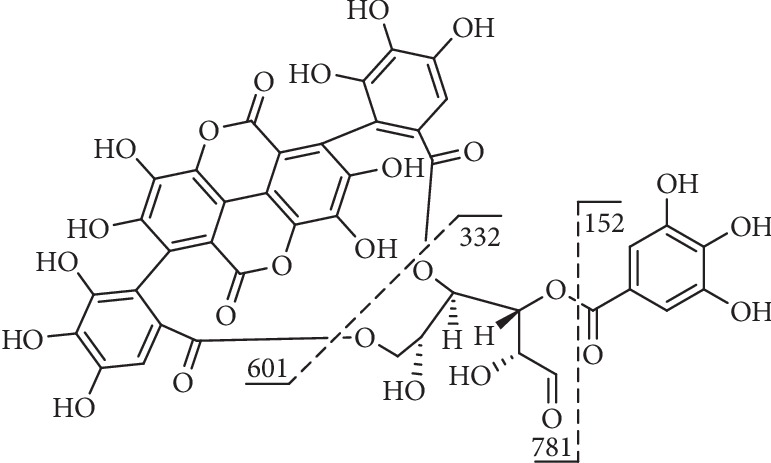
Compound 81 (Figure 16) had an [M-H]− ion at m/z 1205 and other fragments at m/z 915, due to the loss of the substituent at C-1 of the vescalagin-derived nuclei structure and at m/z 613 resulting from the loss of the 4,6-hexahydroxybiphenoyl unit from the latter fragment and at m/z 301 which corresponded to the existence of ellagic acid [143].
Figure 16.
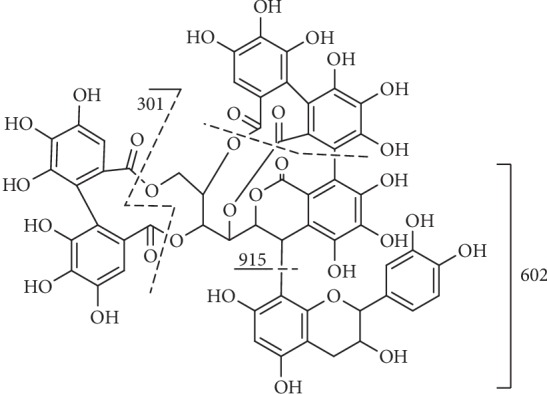
Fragmentation of compound 81.
4. Biological Activity
Natural compounds are important sources of drugs. More and more attention has been paid to the scientific investigation of natural bioactive compounds which may yield new compounds or leading compounds that can overcome the limitations of currently used drugs. At present, some achievements have been made in the study of tannins, but there are still some deficiencies. Tannins extracted from plants are often a collection of monomers of different kinds of tannins mentioned above. Their bioactivities are closely related to the action of these tannin monomers which need further studies. The reported biological activity of these tannins from the genus Terminalia (Family Combretaceae) was summarized briefly.
4.1. Antioxidant Activity
Ellagitannins such as compounds 18, 48, and 70 were found to be the major components in Terminalia bellirica, which exhibited the antioxidant and hepatoprotective activities [167]. Compounds 11, 20, 30, 33, and 43 exhibited great antioxidant activity in both chemical-based and cellular-based antioxidant assays, and compound 11 showed the highest cellular antioxidant activity [168]. Compound 11 has the highest potency for DPPH-, NO-, and ONOO-scavenging activity with IC50 ranging from 5 to 20 μM, 0.20, and 0.06 μM, respectively [169]. Compounds 33 and 43 showed the highest increase in GSH, and compound 30 produced the highest increase in SOD among four tannins [170]. Compounds 28 and 62 had in vitro antioxidant activity and in vivo antioxidative stress effects [171]. A lot of research showed that antioxidant compounds are related to a variety of oxidative stress-related diseases, such as cardiovascular diseases, neurodegenerative diseases, and cancer [172].
4.2. Anticancer Activity
It was confirmed that compound 18 could induce autophagy, apoptosis and ROS accumulation in gastric cancer cells in vitro [173]. IC50 values of HepG2, Molt-3, HL-60, NPC-BM1, HT 1080 and K562 were 1.42, 0.35, 0.12, 0.81, 1.02, 1.53 mg/mL in vivo, respectively [174]. A molecular mechanism study showed that the inhibition of the proliferation of ovarian cancer cells by compound 18 is mediated by blocking the TGF-beta/AKT/ERK/Smad signaling pathway [175]. Compound 11 could induce autophagy of HepG2, MCF-7, and A549 by activating MAPK 8/9/10 and JNK signaling pathways [176]. Compound 11 could also enhance GNMT promoter activity by downregulating MYC expression in hepatocellular carcinoma [177]. Compounds 70, 62, 63, 42, and 19 were isolated from Terminalia calamansanoi with the IC50 values of 65.2, 74.8, 42.2, 38.0, and >100 μM, respectively, for HL-60 cells [178]. It was confirmed that protective effects of compound 20 against DNA damage are induced by different mutagens [179]. The chemopreventive effect of compound 62/63 on H-ras-induced transformation may be due to inhibition of intracellular redox status and activation of JNK-1/p38 [180]. Compounds 30, 33, 49, and 68 could inhibit MCF-7/wt cell viability, and the inhibition ability is stronger with the number of functional units: hexahydroxydiphenoyl (HHDP) group [181]. Compound 50 was proven to have antiproliferative, proapoptotic, and antimigratory effects which are related to the PI3K/AKT and MAPK/ERK pathways [182].
4.3. Antimicrobial and Antivirus Activity
Compound 18 could inhibit biofilm formation, quorum sensing, and toxin secretion. This indicated that corilagin might be an effective antibacterial compound [183]. Compound 11 efficiently blocked entry of HCV of all major genotypes and also of the related flavivirus Zika virus [184]. Compound 11 could effectively inhibit the replication of RABV by the miR-455-5p/SOCS3/STAT3/IL-6-dependent pathway [185]. Compounds 28, 44, and 62 reduced the HCV replication [186] via a dual mechanism through preventing the formation of cccDNA and promoting cccDNA decay [187].
4.4. Antidiabetic Activity
It was confirmed that compound 18 can regulate diabetes, by exhibiting antidiabetic, antihyperlipidemic, and antioxidant properties in STZ-induced diabetic rats [188]. Compound 11 could maintain normal glycemia through the inhibitory action on alpha-amylases [189]. Compounds 22, 48, and 50 with the IC50 values of 690 μM, 97 M, and 361 μM could inhibit activity of mammalian intestinal maltase [53].
4.5. Other Therapeutic Activities
Compounds 20, 30, and 33 which have HHDP moiety decreased the ratio of MMPs/TIMPs to develop skin ageing [190]. Compound 48 was confirmed to inhibit TGF-beta 1-induced antifibrotic activity in choroid-retinal endothelial cells (RF/6A) [191] and inhibit TNF alpha induced proangiogenic and proinflammatory activities in retinal capillary endothelial cells [192].
The study of nanoparticles plays an important role in tannin activity and application. Bioavailability and bioactivity of a component are often altered once it is embedded into nanoparticles. Zheng Li fabricated the PPE with 16.6% (w/w) of punicalagin A, 32.5% (w/w) of punicalagin B, and a small amount of ellagic acid-hexoside and ellagic acid (1%, w/w). PPE-gelatin nanoparticle suspension had similar effects in inducing late stage of apoptosis and necrosis compared to PPE [193]. Guo-Bin Song fabricated a natural promising protein protective film through soluble dietary fiber (SDF)-tannin nanocluster self-assembly which characterized to possess a broad spectrum of antimicrobial properties and are beneficial to food preservation [194]. The field of nanoparticles plays an important role in the utilization of tannin activity with great development potential.
There is a lack of research on the interaction between proteins and tannins from Terminalia Linn., but the tannin extracted from persimmon fruits has been reported to have a high affinity to pancreatic lipase and possessed pancreatic lipase inhibition with IC50 of 0.44 mg/mL. Molecular docking showed that this interaction is mainly caused by the hydrogen bonding and π-π stacking [195]. It has been demonstrated that the very simple tannin methyl gallate was able to stack with itself or with caffeine [10]. The self-association of tannins should also take into account the interaction between tannins and proteins, as it governs their bioavailability. The interactions between tannin-tannin and tannin-protein are still unclear. Changes in protein bioactivity and structure induced by tannin binding need further studies.
Current limited metabolic studies showed that tannins are mainly metabolized as urolithins in the gut [196]. Urolithins are characterized to possess antitumor, antioxidative, and anti-inflammatory activities in vitro, which can be isolated and purified by high-speed counter-current chromatography. Urolithin A, a major punicalagin metabolite, could result in autophagy in SW620 colorectal cancer (CRC) cells at submicromolar concentrations [197]. It is very helpful for drug design to clarify the biotransformation of tannins in vivo.
Therefore, it is necessary to accelerate the development of the technical means for the analysis of bioactive compounds of natural medicines, so as to realize the large-scale development and utilization of tannin monomer compounds. The physiological activity of tannins has been fully confirmed, but the physiological mechanism of its various pharmacological effects is still not clear, limiting the development and utilization of tannins.
5. Conclusion
Terminalia species have been widely used in various traditional medical systems such as Siddha, Traditional Chinese Medicine (TCM), and Western, Southern, and Central African medicinal systems [8]. Apart from reports on the ethnopharmacological uses of many Terminalia species, few studies have carried out rigorous studies on the medical properties, mechanisms, and phytochemistry of this important genus. This may be due to the fact that tannins are the main active constituents in many Terminalia species. Tannins have strong polarity, high molecular weight, complex structure, active chemical properties, and are extremely difficult to crystallize which make them difficult to extract, separate, purify, and identify, and the quality standard is not easy to control. Therefore, they are so complex that they are not suitable for drug design and often overlooked as potential for drug discovery. Thus, how to improve the extraction and purification technology of tannins from genus Terminalia is an urgent problem to be solved. Researchers need to further determine the structure-activity relationship between tannins and their functions, clarify the mechanism of action, and carry out safety toxicological evaluation to ensure safety and stability, so as to make tannins hopeful to become new drugs on the market.
The structures of 82 tannins from the genus Terminalia were reviewed in this paper. The fragmentation pathways of identified tannins were analyzed, and the fragmentation rules of tannins were speculated, which could provide references for the structural analysis of natural medicines and their analogues. In further research, researchers may need to pay more attention to the species and the active substances such as the tannin summarized above.
Conflicts of Interest
No competing financial interest exists.
Authors' Contributions
Zihao Chang and Qiunan Zhang contributed equally to this work.
References
- 1.McGaw L. J., Rabe T., Sparg S. G., Jager A. K., Eloff J. N., van Staden J. An investigation on the biological activity of Combretum species. Journal of Ethnopharmacology. 2001;75(1):45–50. doi: 10.1016/s0378-8741(00)00405-0. [DOI] [PubMed] [Google Scholar]
- 2.Saleem A., Husheem M., Härkönen P., Pihlaja K. Inhibition of cancer cell growth by crude extract and the phenolics of Termnalia chebula Retz. fruit. Journal of Ethnopharmacology. 2002;81(3):327–336. doi: 10.1016/s0378-8741(02)00099-5. [DOI] [PubMed] [Google Scholar]
- 3.Konczak I., Zabaras D., Dunstan M., Aguas P. Antioxidant capacity and hydrophilic phytochemicals in commercially grown native Australian fruits. Food Chemistry. 2010;123(4):1048–1054. doi: 10.1016/j.foodchem.2010.05.060. [DOI] [Google Scholar]
- 4.Aneja K. R., Sharma C., Joshi R. Antimicrobial activity of Terminalia arjuna Wight & Arn: an ethnomedicinal plant against pathogens causing ear infection. Brazilian Journal of Otorhinolaryngology. 2012;78(1):68–74. doi: 10.1590/s1808-86942012000100011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hivrale M. G., Bandawane D. D., Mali A. A. Anti-inflammatory and analgesic activities of petroleum ether and ethyl acetate fractions of Tamarindus indica seeds. Oriental Pharmacy and Experimental Medicine. 2013;13(4):319–326. doi: 10.1007/s13596-013-0116-4. [DOI] [Google Scholar]
- 6.Kaur S., Michael H., Arora S., Harkonen P. L., Kumar S. The in vitro cytotoxic and apoptotic activity of Triphala-an Indian herbal drug. Journal of Ethnopharmacology. 2005;97(1):15–20. doi: 10.1016/j.jep.2004.09.050. [DOI] [PubMed] [Google Scholar]
- 7.Mohanty S., Cock I. E. The chemotherapeutic potential of Terminalia ferdinandiana: phytochemistry and bioactivity. Pharmacognosy Reviews. 2012;6(11):29–36. doi: 10.4103/0973-7847.95855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cock I. E. The medicinal properties and phytochemistry of plants of the genus Terminalia (Combretaceae) Inflammopharmacology. 2015;23(5):203–229. doi: 10.1007/s10787-015-0246-z. [DOI] [PubMed] [Google Scholar]
- 9.Gang D., Yanze L., Quanbin H. Advances in chemical constituents and biological activities of genus Terminalia. Foreign Pharmaceuticals. 1996;11(6):255–258. [Google Scholar]
- 10.Baxter N. J., Williamson M. P., Lilley T. H., Haslam E. Stacking interactions between caffeine and methyl gallate. Journal of the Chemical Society, Faraday Transactions. 1996;92(2):231–234. [Google Scholar]
- 11.Pajari A.-M., Päivärinta E., Paavolainen L., et al. Ellagitannin-rich cloudberry inhibits hepatocyte growth factorinduced cell migration and phosphatidylinositol 3-kinase/AKT activation in colon carcinoma cells and tumors in Min mice. Oncotarget. 2016;7(28):43907–43923. doi: 10.18632/oncotarget.9724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Benhong Z., Huiyuan W., Zhilei G., Qi F., Gang L. The scavenging effect of pomegranate pericarps extract tannins on active oxygen radicals. Chinese Journal of Hospital Pharmacy. 2008;28(17):1442–1445. [Google Scholar]
- 13.Zou B., Ge Z. Z., Zhang Y., Du J., Xu Z., Li C. M. Persimmon tannin accounts for hypolipidemic effects of persimmon through activating of AMPK and suppressing NF-kappaB activation and inflammatory responses in high-fat diet rats. Food & Function. 2014;5(7):1536–1546. doi: 10.1039/c3fo60635j. [DOI] [PubMed] [Google Scholar]
- 14.Singh B., Singh J. P., Kaur A., Singh N. Phenolic compounds as beneficial phytochemicals in pomegranate (Punica granatum L.) peel: a review. Food Chemistry. 2018;261:75–86. doi: 10.1016/j.foodchem.2018.04.039. [DOI] [PubMed] [Google Scholar]
- 15.Tanaka T., Nokaka G.-I., Nishioka I. Tannins and related compounds XL I. Isolation and characterization of novel ellagitannins, punicacorteins A, B, C and D, and puniglucon from the bark of Punica granatum L. Chemical & Pharmaceutical Bulletin. 1986;34(2):656–663. doi: 10.1248/cpb.34.656. [DOI] [Google Scholar]
- 16.Pfundstein B., El Desouky S. K., Hull W. E., Haubner R., Erben G., Owen R. W. Polyphenolic compounds in the fruits of Egyptian medicinal plants (Terminalia bellerica, Terminalia chebula and Terminalia horrida): characterization, quantitation and determination of antioxidant capacities. Phytochemistry. 2010;71(10):1132–1148. doi: 10.1016/j.phytochem.2010.03.018. [DOI] [PubMed] [Google Scholar]
- 17.Gang D., Yanze L., Maoping S., Dapeng Z., Longsheng S. Polyphenols in Terminalia chebula. Journal of China Pharmaceutical University. 2001;32(3):35–38. [Google Scholar]
- 18.Burapadaja S., Bunchoo A. Antimicrobial activity of tannins from Terminalia citrina. Planta Medica. 1995;61:365–366. doi: 10.1055/s-2006-958103. [DOI] [PubMed] [Google Scholar]
- 19.Lee D. Y., Kim H. W., Yang H., Sung S. H. Hydrolyzable tannins from the fruits of Terminalia chebula Retz and their alpha-glucosidase inhibitory activities. Phytochemistry. 2017;137:109–116. doi: 10.1016/j.phytochem.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 20.Yanze L., Gang D., Bingwu Y., Guihua N., Wenling W. Determination of three hydrolyzable tannins in Terminalia chebula by RP-HPLC. Journal of Pharmaceutical Analysis. 2000;20(3):189–191. [Google Scholar]
- 21.Ahn M.-J., Kim C. Y., Lee J. S., Kim T. G. Inhibition of HIV-1 integrase by galloyl glucoses from Terminalia chebula and flavonol glycoside gallates from Euphorbia pekinensis. Planta Medica. 2002;68(5):457–459. doi: 10.1055/s-2002-32070. [DOI] [PubMed] [Google Scholar]
- 22.Lin T.-C., Hsu F.-L. Tannin and related compounds from Terminalia catappa and Terminalia parviflora. Journal of the Chinese Chemical Society. 1999;46(4):613–618. doi: 10.1002/jccs.199900085. [DOI] [Google Scholar]
- 23.Manosroi A., Jantrawut P., Akazawa H., Akihisa T., Manosroi J. Biological activities of phenolic compounds isolated from galls of Terminalia chebula Retz. (Combretaceae) Natural Product Research. 2010;24(20):1915–1926. doi: 10.1080/14786419.2010.488631. [DOI] [PubMed] [Google Scholar]
- 24.Manosroi A., Jantrawut P., Ogihara E., et al. Biological activities of phenolic compounds and triterpenoids from the galls of Terminalia chebula. Chemistry & Biodiversity. 2013;10(8):1448–1463. doi: 10.1002/cbdv.201300149. [DOI] [PubMed] [Google Scholar]
- 25.Sari B. L., Mun’im A., Yanuar A., Riadhi R. Screening of α-glucosidase inhibitors from Terminalia catappa L. Fruits using molecular docking method and in vitro test. International Journal of Pharmacy and Pharmaceutical Sciences. 2016;8(12):p. 184. doi: 10.22159/ijpps.2016v8i12.14800. [DOI] [Google Scholar]
- 26.Jinghao Q., Xian W., Guilin C., Li B. Studies on polyphenolic components and antioxidant activities of Terminalia chebula Retz. Var. tomentella Kurt. Modern Chinese Medicine. 2013;15(12):1036–1041. [Google Scholar]
- 27.Juang L. J., Sheu S. J., Lin T. C. Determination of hydrolyzable tannins in the fruit of Terminalia chebula Retz. by high-performance liquid chromatography and capillary electrophoresis. Journal of Separation Science. 2004;27(9):718–724. doi: 10.1002/jssc.200401741. [DOI] [PubMed] [Google Scholar]
- 28.Pellati F., Bruni R., Righi D., et al. Metabolite profiling of polyphenols in a Terminalia chebula Retzius ayurvedic decoction and evaluation of its chemopreventive activity. Journal of Ethnopharmacology. 2013;147(2):277–285. doi: 10.1016/j.jep.2013.02.025. [DOI] [PubMed] [Google Scholar]
- 29.Singh G., Kumar P. Extraction, gas chromatography-mass spectrometry analysis and screening of fruits of Terminalia chebula Retz. for its antimicrobial potential. Pharmacognosy Research. 2013;5(3):162–168. doi: 10.4103/0974-8490.112421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mahajan A., Pai N. Simultaneous isolation and identification of phytoconstituents from Terminalia chebula by preparative chromatography. Journal of Chemical & Pharmaceutical Research. 2010;2(5):97–103. [Google Scholar]
- 31.Kandil F. E., Nassar M. I. A tannin anti-cancer promotor from Terminalia arjuna. Phytochemistry. 1998;47(8):1567–1568. doi: 10.1016/s0031-9422(97)01078-9. [DOI] [PubMed] [Google Scholar]
- 32.Avula B., Wang Y. H., Wang M., Shen Y. H., Khan I. A. Simultaneous determination and characterization of tannins and triterpene saponins from the fruits of various species of Terminalia and Phyllantus emblica using a UHPLC-UV-MS method: application to triphala. Planta Medica. 2012;79(2):181–188. doi: 10.1055/s-0032-1321082. [DOI] [PubMed] [Google Scholar]
- 33.Yang M. H., Vasquez Y., Ali Z., Khan I. A., Khan S. I. Constituents from Terminalia species increase PPARalpha and PPARgamma levels and stimulate glucose uptake without enhancing adipocyte differentiation. Journal of Ethnopharmacology. 2013;149(2):490–498. doi: 10.1016/j.jep.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 34.Kim M.-S., Lee D. Y., Sung S. H., Jeon W. K. Anti-cholinesterase Activities of Hydrolysable Tannins and Polyhydroxytriterpenoid Derivatives from Terminalia chebula Retz. Fruit. Records of Natural Products. 2018;12(3):284–289. doi: 10.25135/rnp.29.17.07.130. [DOI] [Google Scholar]
- 35.Sancheti S., Sancheti S., Um B. H., Seo S. Y. 1,2,3,4,6-penta-O-galloyl-β-d-glucose: a cholinesterase inhibitor from Terminalia chebula. South African Journal of Botany. 2010;76(2):285–288. doi: 10.1016/j.sajb.2009.11.006. [DOI] [Google Scholar]
- 36.Klika K. D., Saleem A., Sinkkonen J., et al. The structural and conformational analyses and antioxidant activities of chebulinic acid and its thrice-hydrolyzed derivative, 2,4-chebuloyl-β-D-glucopyranoside, isolated from the fruit of Terminalia chebula. Arkivoc. 2004;2004(7):83–105. doi: 10.3998/ark.5550190.0005.708. [DOI] [Google Scholar]
- 37.Kuete V., Tabopda T. K., Ngameni B., Nana F., Tshikalange T. E., Ngadjui B. T. Antimycobacterial, antibacterial and antifungal activities of Terminalia superba (Combretaceae) South African Journal of Botany. 2010;76(1):125–131. doi: 10.1016/j.sajb.2009.09.009. [DOI] [Google Scholar]
- 38.Lin T.-C., Nonaka G.-i., Nishioka I., Ho F.-c. Tannins and related compounds. C II. structures of Terchebulin, an ellagitannin having a novel tetraphenylcarboxylic acid (terchebulic acid) moiety, and biogenetically related Tannins from Terminalia chebula RETZ. Chemical and Pharmaceutical Bulletin. 1990;38(11):3004–3008. [Google Scholar]
- 39.Chandrasekhar Y., Phani Kumar G., Navya K., Ramya E. M., Anilakumar K. R. Tannins from Terminalia chebula fruits attenuates GABA antagonist-induced anxiety-like behaviour via modulation of neurotransmitters. Journal of Pharmacy and Pharmacology. 2018;70(12):1662–1674. doi: 10.1111/jphp.13007. [DOI] [PubMed] [Google Scholar]
- 40.Ajala O. S., Jukov A., Ma C. M. Hepatitis C virus inhibitory hydrolysable tannins from the fruits of Terminalia chebula. Fitoterapia. 2014;99:117–123. doi: 10.1016/j.fitote.2014.09.014. [DOI] [PubMed] [Google Scholar]
- 41.Gao J., Ajala O. S., Wang C. Y., et al. Comparison of pharmacokinetic profiles of Terminalia phenolics after intragastric administration of the aqueous extracts of the fruit of Terminalia chebula and a Mongolian compound medicine-Gurigumu-7. Journal of Ethnopharmacology. 2016;185:300–309. doi: 10.1016/j.jep.2016.03.016. [DOI] [PubMed] [Google Scholar]
- 42.Sheng Z., Yan X., Zhang R., et al. Assessment of the antidiarrhoeal properties of the aqueous extract and its soluble fractions of Chebulae Fructus (Terminalia chebula fruits) Pharmaceutical Biology. 2016;54(9):1847–1856. doi: 10.3109/13880209.2015.1131993. [DOI] [PubMed] [Google Scholar]
- 43.Juang L. J., Sheu S.-J. Chemical identification of the sources of commercial fructus chebulae. Phytochemical Analysis. 2005;16(4):246–251. doi: 10.1002/pca.823. [DOI] [PubMed] [Google Scholar]
- 44.Kalra P., Karwasra R., Gupta Y. K., Ray S. B., Singh S. Terminalia chebula supplementation attenuates cisplatin-induced nephrotoxicity in Wistar rats through modulation of apoptotic pathway. Natural Product Research. 2019;33(11):1641–1645. doi: 10.1080/14786419.2018.1425843. [DOI] [PubMed] [Google Scholar]
- 45.Chandrasekhar Y., Ramya E. M., Navya K., Phani Kumar G., Anilakumar K. R. Antidepressant like effects of hydrolysable tannins of Terminalia catappa leaf extract via modulation of hippocampal plasticity and regulation of monoamine neurotransmitters subjected to chronic mild stress (CMS) Biomedicine & Pharmacotherapy. 2017;86:414–425. doi: 10.1016/j.biopha.2016.12.031. [DOI] [PubMed] [Google Scholar]
- 46.Kinoshita S., Inoue Y., Nakama S., Ichiba T., Aniya Y. Antioxidant and hepatoprotective actions of medicinal herb, Terminalia catappa L. from Okinawa Island and its tannin corilagin. Phytomedicine. 2007;14(11):755–762. doi: 10.1016/j.phymed.2006.12.012. [DOI] [PubMed] [Google Scholar]
- 47.Rangsriwong P., Rangkadilok N., Satayavivad J., Goto M., Shotipruk A. Subcritical water extraction of polyphenolic compounds from Terminalia chebula Retz. fruits. Separation and Purification Technology. 2009;66(1):51–56. doi: 10.1016/j.seppur.2008.11.023. [DOI] [Google Scholar]
- 48.Pugazhendhi A., Beema Shafreen R., Pandima Devi K., Suganthy N. Assessment of antioxidant, anticholinesterase and antiamyloidogenic effect of Terminalia chebula , Terminalia arjuna and its bioactive constituent 7-methyl gallic acid–an in vitro and in silico studies. Journal of Molecular Liquids. 2018;257:69–81. doi: 10.1016/j.molliq.2018.02.081. [DOI] [Google Scholar]
- 49.Sirdaarta J., Matthews B., Cock I. E. Kakadu plum fruit extracts inhibit growth of the bacterial triggers of rheumatoid arthritis: identification of stilbene and tannin components. Journal of Functional Foods. 2015;17:610–620. doi: 10.1016/j.jff.2015.06.019. [DOI] [Google Scholar]
- 50.Chen L., Qi J., Chang Y. X., Zhu D., Yu B. Identification and determination of the major constituents in Traditional Chinese Medicinal formula Danggui-Shaoyao-San by HPLC-DAD-ESI-MS/MS. Journal of Pharmaceutical and Biomedical Analysis. 2009;50(2):127–137. doi: 10.1016/j.jpba.2009.03.039. [DOI] [PubMed] [Google Scholar]
- 51.Tanaka T., Morita A., Nonaka G.-i. Tannins and related compounds. CIII. isolation and characterization of new monomeric, dimeric and trimeric ellagitannins, calamansanin and calamanins A, B and C, from Terminalia calamansanai (BLANCO) ROLFE. Chemical and Pharmaceutical Bulletin. 1991;39(1):60–63. doi: 10.1248/cpb.39.60. [DOI] [Google Scholar]
- 52.Kaneria M. J., Rakholiya K. D., Marsonia L. R., Dave R. A., Golakiya B. A. Nontargeted metabolomics approach to determine metabolites profile and antioxidant study of tropical almond (Terminalia catappa L.) fruit peels using GC-QTOF-MS and LC-QTOF-MS. Journal of Pharmaceutical and Biomedical Analysis. 2018;160:415–427. doi: 10.1016/j.jpba.2018.08.026. [DOI] [PubMed] [Google Scholar]
- 53.Gao H., Huang Y., Xu P., Kawabata J. Inhibitory effect on α-glucosidase by the fruits of Terminalia chebula Retz. Food Chemistry. 2007;105(2):628–634. doi: 10.1016/j.foodchem.2007.04.023. [DOI] [Google Scholar]
- 54.Cheng H.-Y., Lin T.-C., Yu K.-H., Yang C.-M., Lin C.-C. Antioxidant and free radical scavenging activities of Terminalia chebula. Biological & Pharmaceutical Bulletin. 2003;26(9):1331–1335. doi: 10.1248/bpb.26.1331. [DOI] [PubMed] [Google Scholar]
- 55.Avula B., Wang Y. H., Isaac G., et al. Metabolic profiling of hoodia, chamomile, Terminalia species and evaluation of commercial preparations using ultrahigh-performance liquid chromatography quadrupole-time-of-flight mass spectrometry. Planta Medica. 2017;83(16):1297–1308. doi: 10.1055/s-0043-109239. [DOI] [PubMed] [Google Scholar]
- 56.Liu M., Katerere D. R., Gray A. I., Seidel V. Phytochemical and antifungal studies on Terminalia mollis and Terminalia brachystemma. Fitoterapia. 2009;80(6):369–373. doi: 10.1016/j.fitote.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 57.Machumi F., Midiwo J. O., Jacob M. R., et al. Phytochemical, antimicrobial and antiplasmodial investigations of Terminalia brownii. Natural Product Communications. 2013;8(6):761–764. doi: 10.1177/1934578x1300800619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tanaka T., Nonaka G.-I., Nishioka I. Tannins and related compounds. XLII. Isolation and characterization of four new hydrolyzable tannins, terflavins A and B, tergallagin and tercatain fron the leaves of Terminalia catappa L. Chemical and Pharmaceutical Bulletin. 1986;34(3):1039–1049. doi: 10.1248/cpb.34.1039. [DOI] [Google Scholar]
- 59.Martino V., Morales J., Martinez-Irujo J. J., Font M., Monge A., Coussio J. Two ellagitannins from the leaves of Terminalia triflora with inhibitory activity on HIV-1 reverse transcriptase. Phytotherapy Research. 2004;18(8):667–669. doi: 10.1002/ptr.1504. [DOI] [PubMed] [Google Scholar]
- 60.Conrad J., Vogler B., Reeb S., et al. Isoterchebulin and 4,6-O-isoterchebuloyl-D-glucose, novel hydrolyzable tannins from Terminalia macroptera. Journal of Natural Products. 2001;64(3):294–299. doi: 10.1021/np000506v. [DOI] [PubMed] [Google Scholar]
- 61.Grasel F. S., Ferrão M. F. Rapid discrimination of natural polyphenols (vegetable tannins) from different plants by molecular spectroscopy and PLS-DA. Analytical Methods. 2018;10(9):968–974. doi: 10.1039/c8ay00075a. [DOI] [Google Scholar]
- 62.Abiodun O. O., Rodriguez-Nogales A., Algieri F., et al. Antiinflammatory and immunomodulatory activity of an ethanolic extract from the stem bark of Terminalia catappa L. (Combretaceae): In vitro and in vivo evidences. Journal of Ethnopharmacology. 2016;192:309–319. doi: 10.1016/j.jep.2016.07.056. [DOI] [PubMed] [Google Scholar]
- 63.Fahmy N. M., Al-Sayed E., Abdel-Daim M. M., Karonen M., Singab A. N. Protective effect of Terminalia muelleri against carbon tetrachloride-induced hepato and nephro-toxicity in mice and characterization of its bioactive constituents. Pharmaceutical Biology. 2016;54(2):303–313. doi: 10.3109/13880209.2015.1035794. [DOI] [PubMed] [Google Scholar]
- 64.Pellikaan W. F., Stringano E., Leenaars J., et al. Evaluating effects of tannins on extent and rate of in vitro gas and CH4 production using an automated pressure evaluation system (APES) Animal Feed Science and Technology. 2011;166-167:377–390. doi: 10.1016/j.anifeedsci.2011.04.072. [DOI] [Google Scholar]
- 65.Cheng H. Y., Lin T. C., Yu K. H., Yang C. M., Lin C. C. Antioxidant and Free Radical Scavenging Activities of Terminalia chebula. Biological & Pharmaceutical Bulletin. 2003;26(9):1331–1335. doi: 10.1248/bpb.26.1331. [DOI] [PubMed] [Google Scholar]
- 66.Cheng H. Y., Lin C. C., Lin T. C. Antiherpes simplex virus type 2 activity of casuarinin from the bark of Terminalia arjuna Linn. Antiviral Research. 2002;55(3):447–455. doi: 10.1016/s0166-3542(02)00077-3. [DOI] [PubMed] [Google Scholar]
- 67.Kuo P. L., Hsu Y. L., Lin T. C., Chang J. K., Lin C. C. Induction of cell cycle arrest and apoptosis in human non-small cell lung cancer A549 cells by casuarinin from the bark of Terminalia arjuna Linn. Anti-Cancer Drugs. 2005;16(4):409–415. doi: 10.1097/00001813-200504000-00007. [DOI] [PubMed] [Google Scholar]
- 68.Lee Y., Byun H. S., Seok J. H., et al. Terminalia chebula provides protection against dual modes of necroptotic and apoptotic cell death upon death receptor ligation. Science Reports. 2016;6:p. 25094. doi: 10.1038/srep25094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hamada S.-i., Kataoka T., Woo J.-T., Yamada A. Immunosuppressive effects of gallic acid and chebulagic acid on CTL-mediated cytotoxicity. Biological & Pharmaceutical Bulletin. 1997;20(9):1017–1019. doi: 10.1248/bpb.20.1017. [DOI] [PubMed] [Google Scholar]
- 70.Han Q., Song J., Qiao C., Wong L., Xu H. Preparative isolation of hydrolysable tannins chebulagic acid and chebulinic acid from Terminalia chebula by high-speed counter-current chromatography. Journal of Separation Science. 2006;29(11):1653–1657. doi: 10.1002/jssc.200600089. [DOI] [PubMed] [Google Scholar]
- 71.Huang Y. N., Zhao D. D., Gao B., et al. Anti-hyperglycemic effect of chebulagic acid from the fruits of Terminalia chebula Retz. International Journal of Molecular Sciences. 2012;13(5):6320–6333. doi: 10.3390/ijms13056320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kumar N., Gangappa D., Gupta G., Karnati R. Chebulagic acid from Terminalia chebula causes G1 arrest, inhibits NFκB and induces apoptosis in retinoblastoma cells. BMC Complementary and Alternative Medicine. 2014;14(1) doi: 10.1186/1472-6882-14-319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lee S.-H., Ryu S. Y., Choi S. U., et al. Hydrolysable tannins and related compound having cytotoxic activity from the fruits of Terminalia chebula. Archives of Pharmacal Research. 1995;18(2):118–120. doi: 10.1007/bf02979144. [DOI] [Google Scholar]
- 74.Reddy D. B., Reddy T. C., Jyotsna G., et al. Chebulagic acid, a COX-LOX dual inhibitor isolated from the fruits of Terminalia chebula Retz., induces apoptosis in COLO-205 cell line. Journal of Ethnopharmacology. 2009;124(3):506–512. doi: 10.1016/j.jep.2009.05.022. [DOI] [PubMed] [Google Scholar]
- 75.Shyni G. L., Kavitha S., Indu S., et al. Chebulagic acid from Terminalia chebula enhances insulin mediated glucose uptake in 3T3-L1 adipocytes via PPARgamma signaling pathway. BioFactors. 2014;40(6):646–657. doi: 10.1002/biof.1193. [DOI] [PubMed] [Google Scholar]
- 76.Ekambaram S. P., Babu K. B., Perumal S. S., Rajendran D. Repeated oral dose toxicity study on hydrolysable tannin rich fraction isolated from fruit pericarps of Terminalia chebula Retz in Wistar albino rats. Regulatory Toxicology and Pharmacology. 2018;92:182–188. doi: 10.1016/j.yrtph.2017.12.001. [DOI] [PubMed] [Google Scholar]
- 77.Gao H., Huang Y.-N., Gao B., Kawabata J. Chebulagic acid is a potent α-glucosidase inhibitor. Bioscience, Biotechnology and Biochemistry. 2008;72(2):601–603. doi: 10.1271/bbb.70591. [DOI] [PubMed] [Google Scholar]
- 78.Singh A., Bajpai V., Kumar S., Kumar B., Srivastava M., Rameshkumar K. B. Comparative profiling of phenolic compounds from different plant parts of six Terminalia species by liquid chromatography–tandem mass spectrometry with chemometric analysis. Industrial Crops and Products. 2016;87:236–246. doi: 10.1016/j.indcrop.2016.04.048. [DOI] [Google Scholar]
- 79.Dv S. P., Nandam S. S., Vangalapati M. Optimization of physico-chemical parameters for the extraction of phenolic components from Terminalia chebula species. Research in Pharmacy. 2018;2(5):01–08. [Google Scholar]
- 80.Chhabra S., Mishra T., Kumar Y., et al. Chebulinic acid isolated from the fruits of Terminalia chebula specifically induces apoptosis in acute myeloid leukemia cells. Phytotherapy Research. 2017;31(12):1849–1857. doi: 10.1002/ptr.5927. [DOI] [PubMed] [Google Scholar]
- 81.Kumar K. J. Effect of geographical variation on contents of tannic acid, gallic acid, chebulinic acid and ethyl gallate in Terminalia chebula fruits. Natural Products: An Indian Journal. 2006;2(3-4):100–104. [Google Scholar]
- 82.Mishra V., Agrawal M., Onasanwo S. A., et al. Anti-secretory and cyto-protective effects of chebulinic acid isolated from the fruits of Terminalia chebula on gastric ulcers. Phytomedicine. 2013;20(6):506–511. doi: 10.1016/j.phymed.2013.01.002. [DOI] [PubMed] [Google Scholar]
- 83.Saleem A., H. M. Inhibition of cancer cell growth by crude extract and the phenolics of Termnalia chebula Retz. fruit. Journal of Ethnopharmacology. 2012;81(3):327–336. doi: 10.1016/S0378-8741(02)00099-5. [DOI] [PubMed] [Google Scholar]
- 84.Gangliu D., Yanze L., Li W., Chunru J., Longsheng S. Structural identification of hydrolyzable tannins in Terminalia chebula. Journal of China Pharmaceutical University. 2001;32(9):91–93. [Google Scholar]
- 85.Singh S., Lal U. R. Evaluation of in-vitro anti-inflammatory activity of chebulinic acid from Terminalia chebula Linn. against the denaturation of protein. Proceedings of The 18th International Electronic Conference on Synthetic Organic Chemistry; November 2014; Spain. [DOI] [Google Scholar]
- 86.Seto H. ChemInform abstract: isolation and structure elucidation of punicalagin, a toxic hydrolysable tannin, from Terminalia oblongata. ChemInform. 2011;21(46):2317–2321. doi: 10.1002/chin.201101187. [DOI] [Google Scholar]
- 87.Marzouk M. S. A., EI-Toumy S. A. A., Moharram F. A. Pharmacologically active ellagitannins from Terminalia myriocarpa. Planta Medica. 2002;68:523–527. doi: 10.1055/s-2002-32549. [DOI] [PubMed] [Google Scholar]
- 88.Silva O., Gomes E. T., Wolfender J.-L., Marston A., Hostettmann K. Application of high performance liquid chromatography coupled with ultraviolet spectroscopy and electrospray mass spectrometry to the characterisation of ellagitannins from Terminalia macroptera roots. Pharmaceutical Research. 2000;17(11):1396–1401. doi: 10.1023/A:1007598922712. [DOI] [PubMed] [Google Scholar]
- 89.Mohieldin E. A. M., Muddathir A. M., Yamauchi K., Mitsunaga T. Anti-caries activity of selected Sudanese medicinal plants with emphasis on Terminalia laxiflora. Revista Brasileira de Farmacognosia. 2017;27(5):611–618. doi: 10.1016/j.bjp.2017.04.002. [DOI] [Google Scholar]
- 90.Muddathir A. M., Yamauchi K., Mitsunaga T. Anti-acne activity of tannin-related compounds isolated from Terminalia laxiflora. Journal of Wood Science. 2013;59(5):426–431. doi: 10.1007/s10086-013-1344-4. [DOI] [Google Scholar]
- 91.Gahlaut A., Sharma A., Shirolkar A., Dabur R. Non-targeted identification of compounds from regenerated bark of Terminalia tomentosa by HPLC- (+) ESI-QTOFMS. Journal of Pharmacy Research. 2013;6(4):415–418. doi: 10.1016/j.jopr.2013.04.010. [DOI] [Google Scholar]
- 92.Jin W., Wang Y. F., Ge R. L., Shi H. M., Jia C. Q., Tu P. F. Simultaneous analysis of multiple bioactive constituents in Rheum tanguticum Maxim. ex Balf. by high-performance liquid chromatography coupled to tandem mass spectrometry. Rapid Communications in Mass Spectrometry. 2007;21(14):2351–2360. doi: 10.1002/rcm.3086. [DOI] [PubMed] [Google Scholar]
- 93.Zhu T., Liu X., Wang X., et al. Profiling and analysis of multiple compounds in rhubarb decoction after processing by wine steaming using UHPLC-Q-TOF-MS coupled with multiple statistical strategies. Journal of Separation Science. 2016;39(15):3081–3090. doi: 10.1002/jssc.201600256. [DOI] [PubMed] [Google Scholar]
- 94.Wang J., Jia Z., Zhang Z., et al. Analysis of Chemical Constituents of Melastoma dodecandrum Lour. by UPLC-ESI-Q-Exactive Focus-MS/MS. Molecules. 2017;22(3):p. 476. doi: 10.3390/molecules22030476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hooi Poay T., Sui Kiong L., Cheng Hock C. Characterisation of galloylated cyanogenic glucosides and hydrolysable tannins from leaves of Phyllagathis rotundifolia by LC-ESI-MS/MS. Phytochemical Analysis. 2011;22(6):516–525. doi: 10.1002/pca.1312. [DOI] [PubMed] [Google Scholar]
- 96.Wang S., Chen L., Leng J., Chen P., Fan X., Cheng Y. Fragment ion diagnostic strategies for the comprehensive identification of chemical profile of Gui-Zhi-Tang by integrating high-resolution MS, multiple-stage MS and UV information. Journal of Pharmaceutical and Biomedical Analysis. 2014;98:22–35. doi: 10.1016/j.jpba.2014.05.013. [DOI] [PubMed] [Google Scholar]
- 97.Boulekbache-Makhlouf L., Meudec E., Mazauric J. P., Madani K., Cheynier V. Qualitative and semi-quantitative analysis of phenolics in Eucalyptus globulus leaves by high-performance liquid chromatography coupled with diode array detection and electrospray ionisation mass spectrometry. Phytochemical Analysis. 2013;24(2):162–170. doi: 10.1002/pca.2396. [DOI] [PubMed] [Google Scholar]
- 98.Hu K., Dars A. G., Liu Q., Xie B., Sun Z. Phytochemical profiling of the ripening of Chinese mango (Mangifera indica L.) cultivars by real-time monitoring using UPLC-ESI-QTOF-MS and its potential benefits as prebiotic ingredients. Food Chemistry. 2018;256:171–180. doi: 10.1016/j.foodchem.2018.02.014. [DOI] [PubMed] [Google Scholar]
- 99.Li S. L., Song J. Z., Choi F. F., et al. Chemical profiling of Radix Paeoniae evaluated by ultra-performance liquid chromatography/photo-diode-array/quadrupole time-of-flight mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis. 2009;49(2):253–266. doi: 10.1016/j.jpba.2008.11.007. [DOI] [PubMed] [Google Scholar]
- 100.Li D. Q., Zhao J., Xie J., Li S. P. A novel sample preparation and on-line HPLC-DAD-MS/MS-BCD analysis for rapid screening and characterization of specific enzyme inhibitors in herbal extracts: case study of alpha-glucosidase. Journal of Pharmaceutical and Biomedical Analysis. 2014;88:130–135. doi: 10.1016/j.jpba.2013.08.029. [DOI] [PubMed] [Google Scholar]
- 101.Liu E. H., Qi L. W., Li B., et al. High-speed separation and characterization of major constituents in Radix Paeoniae Rubra by fast high-performance liquid chromatography coupled with diode-array detection and time-of-flight mass spectrometry. Rapid Communications in Mass Spectrometry. 2009;23(1):119–130. doi: 10.1002/rcm.3848. [DOI] [PubMed] [Google Scholar]
- 102.Xu S. J., Yang L., Zeng X., Zhang M., Wang Z. T. Characterization of compounds in the Chinese herbal drug Mu-Dan-Pi by liquid chromatography coupled to electrospray ionization mass spectrometry. Rapid Communications in Mass Spectrometry. 2006;20(22):3275–3288. doi: 10.1002/rcm.2717. [DOI] [PubMed] [Google Scholar]
- 103.Li P., Su W., Xie C., Zeng X., Peng W., Liu M. Rapid Identification and simultaneous quantification of multiple constituents in Nao-Shuan-Tong Capsule by ultra-fast liquid chromatography/diode-array detector/quadrupole time-of-flight tandem mass spectrometry. Journal of Chromatographic Science. 2015;53(6):886–897. doi: 10.1093/chromsci/bmu137. [DOI] [PubMed] [Google Scholar]
- 104.Chen X., Zhang L., Zhou S., Zhu Y., Liu C. Identification and characterization of constituents in Si-Wu-Tang by liquid chromatography connected with time of flight mass spectrometry and ion trap mass spectrometry. Asian Journal of Chemistry. 2013;25(11):6263–6266. doi: 10.14233/ajchem.2013.14346. [DOI] [Google Scholar]
- 105.Bijttebier S., Van der Auwera A., Voorspoels S., et al. A first step in the quest for the active constituents in filipendula ulmaria (meadowsweet): comprehensive phytochemical identification by liquid chromatography coupled to quadrupole-orbitrap mass spectrometry. Planta Medica. 2016;82(6):559–572. doi: 10.1055/s-0042-101943. [DOI] [PubMed] [Google Scholar]
- 106.Fernandes A., Sousa A., Mateus N., Cabral M., de Freitas V. Analysis of phenolic compounds in cork from Quercus suber L. by HPLC–DAD/ESI–MS. Food Chemistry. 2011;125(4):1398–1405. doi: 10.1016/j.foodchem.2010.10.016. [DOI] [Google Scholar]
- 107.Gomez-Caravaca A. M., Lopez-Cobo A., Verardo V., Segura-Carretero A., Fernandez-Gutierrez A. HPLC-DAD-q-TOF-MS as a powerful platform for the determination of phenolic and other polar compounds in the edible part of mango and its by-products (peel, seed, and seed husk) Electrophoresis. 2016;37(7-8):1072–1084. doi: 10.1002/elps.201500439. [DOI] [PubMed] [Google Scholar]
- 108.Hofmann T., Nebehaj E., Albert L. Antioxidant properties and detailed polyphenol profiling of European hornbeam (Carpinus betulus L.) leaves by multiple antioxidant capacity assays and high-performance liquid chromatography/multistage electrospray mass spectrometry. Industrial Crops and Products. 2016;87:340–349. doi: 10.1016/j.indcrop.2016.04.037. [DOI] [Google Scholar]
- 109.Iwanaga C. C., Ferreira L., Bernuci K. Z., et al. In vitro antioxidant potential and in vivo effects of Schinus terebinthifolia Raddi leaf extract in diabetic rats and determination of chemical composition by HPLC-ESI-MS/MS. Natural Product Research. 2019;33(11):1655–1658. doi: 10.1080/14786419.2018.1425848. [DOI] [PubMed] [Google Scholar]
- 110.Liu J.-h., Sun H., Zhang A.-h., et al. Serum pharmacochemistry combined with multiple data processing approach to screen the bioactive components and their metabolites in mutan cortex by ultra-performance liquid chromatography tandem mass spectrometry. Biomedical Chromatography. 2014;28(4):500–510. doi: 10.1002/bmc.3060. [DOI] [PubMed] [Google Scholar]
- 111.Salminen J.-P., Karonen M., Lempa K., et al. Characterisation of proanthocyanidin aglycones and glycosides from rose hips by high-performance liquid chromatography–mass spectrometry, and their rapid quantification together with vitamin C. Journal of Chromatography A. 2005;1077(2):170–180. doi: 10.1016/j.chroma.2005.04.073. [DOI] [PubMed] [Google Scholar]
- 112.Cardullo N., Muccilli V., Saletti R., Giovando S., Tringali C. A mass spectrometry and (1)H NMR study of hypoglycemic and antioxidant principles from a Castanea sativa tannin employed in oenology. Food Chemistry. 2018;268:585–593. doi: 10.1016/j.foodchem.2018.06.117. [DOI] [PubMed] [Google Scholar]
- 113.Comandini P., Lerma-Garcia M. J., Simo-Alfonso E. F., Toschi T. G. Tannin analysis of chestnut bark samples (Castanea sativa Mill.) by HPLC-DAD-MS. Food Chemistry. 2014;157:290–295. doi: 10.1016/j.foodchem.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 114.García-Villalba R., Espín J. C., Tomás-Barberán F. A., Rocha-Guzmán N. E. Comprehensive characterization by LC-DAD-MS/MS of the phenolic composition of seven Quercus leaf teas. Journal of Food Composition and Analysis. 2017;63:38–46. doi: 10.1016/j.jfca.2017.07.034. [DOI] [Google Scholar]
- 115.Kumar S., Chandra P., Bajpai V., et al. Rapid qualitative and quantitative analysis of bioactive compounds from Phyllanthus amarus using LC/MS/MS techniques. Industrial Crops and Products. 2015;69:143–152. doi: 10.1016/j.indcrop.2015.02.012. [DOI] [Google Scholar]
- 116.Di Stefano V., Pitonzo R., Novara M. E., et al. Antioxidant activity and phenolic composition in pomegranate (Punica granatum L.) genotypes from south Italy by UHPLC-Orbitrap-MS approach. Journal of the Science of Food and Agriculture. 2019;99(3):1038–1045. doi: 10.1002/jsfa.9270. [DOI] [PubMed] [Google Scholar]
- 117.Dos Reis Luz L., Porto D. D., Castro C. B., et al. Metabolomic profile of Schinopsis brasiliensis via UPLC-QTOF-MS for identification of biomarkers and evaluation of its cytotoxic potential. Journal of Chromatography B. 2018;1099:97–109. doi: 10.1016/j.jchromb.2018.09.019. [DOI] [PubMed] [Google Scholar]
- 118.Abdulla R., Mansur S., Lai H., et al. Qualitative analysis of polyphenols in macroporous resin pretreated pomegranate husk extract by HPLC-QTOF-MS. Phytochemical Analysis. 2017;28(5):465–473. doi: 10.1002/pca.2695. [DOI] [PubMed] [Google Scholar]
- 119.Dai W., Qi D., Yang T., et al. Nontargeted analysis using ultraperformance liquid chromatography-quadrupole time-of-flight mass spectrometry uncovers the effects of harvest season on the metabolites and taste quality of tea (Camellia sinensis L.) Journal of Agricultural and Food Chemistry. 2015;63(44):9869–9878. doi: 10.1021/acs.jafc.5b03967. [DOI] [PubMed] [Google Scholar]
- 120.Lee J.-H., Johnson J. V., Talcott S. T. Identification of ellagic acid conjugates and other polyphenolics in muscadine grapes by HPLC-ESI-MS. Journal of Agricultural and Food Chemistry. 2005;53(15):6003–6010. doi: 10.1021/jf050468r. [DOI] [PubMed] [Google Scholar]
- 121.Moilanen J., Sinkkonen J., Salminen J.-P. Characterization of bioactive plant ellagitannins by chromatographic, spectroscopic and mass spectrometric methods. Chemoecology. 2013;23(3):165–179. doi: 10.1007/s00049-013-0132-3. [DOI] [Google Scholar]
- 122.Yang B., Kortesniemi M., Liu P., Karonen M., Salminen J. P. Analysis of hydrolyzable tannins and other phenolic compounds in emblic leafflower (Phyllanthus emblica L.) fruits by high performance liquid chromatography-electrospray ionization mass spectrometry. Journal of Agricultural and Food Chemistry. 2012;60(35):8672–8683. doi: 10.1021/jf302925v. [DOI] [PubMed] [Google Scholar]
- 123.Changxi W., Lili Y., Haiyan X., et al. Simultaneous quantification of 7 components in different parts of Punica granatum fruits using ultra-high performance liquid chromatography-triple quadrupole mass spectrometry (UPLC-QQQMS) Food Science. 2016;37(04):139–143. [Google Scholar]
- 124.Borges G., Crozier A. HPLC-PDA-MS fingerprinting to assess the authenticity of pomegranate beverages. Food Chemistry. 2012;135(3):1863–1867. doi: 10.1016/j.foodchem.2012.05.108. [DOI] [PubMed] [Google Scholar]
- 125.Fernandez-Arroyo S., Barrajon-Catalan E., Micol V., Segura-Carretero A., Fernandez-Gutierrez A. High-performance liquid chromatography with diode array detection coupled to electrospray time-of-flight and ion-trap tandem mass spectrometry to identify phenolic compounds from a Cistus ladanifer aqueous extract. Phytochemical Analysis. 2009;21(4):307–313. doi: 10.1002/pca.1200. [DOI] [PubMed] [Google Scholar]
- 126.Benhong Z., Huilan Y., Xianxi G., Qi F., Gang L. Preliminary analysis of tannin-related constituents in pomegranate peel by HPLC-ESI-MS. China Pharmacist. 2015;18(2):201–204. [Google Scholar]
- 127.Maggi F., Lucarini D., Papa F., Peron G., Dall’Acqua S. Phytochemical analysis of the labdanum-poor Cistus creticus subsp. eriocephalus (Viv.) Greuter et Burdet growing in central Italy. Biochemical Systematics and Ecology. 2016;66:50–57. doi: 10.1016/j.bse.2016.02.030. [DOI] [Google Scholar]
- 128.Tuominen A., Sundman T. Stability and oxidation products of hydrolysable tannins in basic conditions detected by HPLC/DAD-ESI/QTOF/MS. Phytochemical Analysis. 2013;24(5):424–435. doi: 10.1002/pca.2456. [DOI] [PubMed] [Google Scholar]
- 129.Teixeira Lde L., Bertoldi F. C., Lajolo F. M., Hassimotto N. M. Identification of ellagitannins and flavonoids from Eugenia brasilienses Lam. (Grumixama) by HPLC-ESI-MS/MS. Journal of Agricultural and Food Chemistry. 2015;63(22):5417–5427. doi: 10.1021/acs.jafc.5b01195. [DOI] [PubMed] [Google Scholar]
- 130.Hager T. J., Howard L. R., Liyanage R., Lay J. O., Prior R. L. Ellagitannin composition of Blackberry as determined by HPLC-ESI-MS and MALDI-TOF-MS. Journal of Agricultural and Food Chemistry. 2008;56(3):661–669. doi: 10.1021/jf071990b. [DOI] [PubMed] [Google Scholar]
- 131.Gomez-Caravaca A. M., Verardo V., Toselli M., Segura-Carretero A., Fernandez-Gutierrez A., Caboni M. F. Determination of the major phenolic compounds in pomegranate juices by HPLC-DAD-ESI-MS. Journal of Agricultural and Food Chemistry. 2013;61(22):5328–5337. doi: 10.1021/jf400684n. [DOI] [PubMed] [Google Scholar]
- 132.D’Urso G., Pizza C., Piacente S., Montoro P. Combination of LC-MS based metabolomics and antioxidant activity for evaluation of bioactive compounds in Fragaria vesca leaves from Italy. Journal of Pharmaceutical and Biomedical Analysis. 2018;150:233–240. doi: 10.1016/j.jpba.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 133.D’Urso G., Sarais G., Lai C., Pizza C., Montoro P. LC-MS based metabolomics study of different parts of myrtle berry from Sardinia (Italy) Journal of Berry Research. 2017;7(3):217–229. doi: 10.3233/jbr-170158. [DOI] [Google Scholar]
- 134.Díaz-de-Cerio E., Gómez-Caravaca A. M., Verardo V., Fernández-Gutiérrez A., Segura-Carretero A. Determination of guava (Psidium guajava L.) leaf phenolic compounds using HPLC-DAD-QTOF-MS. Journal of Functional Foods. 2016;22:376–388. doi: 10.1016/j.jff.2016.01.040. [DOI] [Google Scholar]
- 135.Al-Sayed E., Singab A.-N., Ayoub N., Martiskainen O., Sinkkonen J., Pihlaja K. HPLC–PDA–ESI–MS/MS profiling and chemopreventive potential of Eucalyptus gomphocephala DC. Food Chemistry. 2012;133(3):1017–1024. doi: 10.1016/j.foodchem.2011.09.036. [DOI] [Google Scholar]
- 136.Anjum S., Gani A., Ahmad M., et al. Antioxidant and antiproliferative activity of walnut extract (Juglans regia L.) processed by different methods and identification of compounds using GC/MS and LC/MS technique. Journal of Food Processing and Preservation. 2017;41(1) doi: 10.1111/jfpp.12756.e12756 [DOI] [Google Scholar]
- 137.Boulekbache-Makhlouf L., Meudec E., Chibane M., et al. Analysis by high-performance liquid chromatography diode array detection mass spectrometry of phenolic compounds in fruit of Eucalyptus globulus cultivated in Algeria. Journal of Agricultural and Food Chemistry. 2010;58(24):12615–12624. doi: 10.1021/jf1029509. [DOI] [PubMed] [Google Scholar]
- 138.Grace M. H., Warlick C. W., Neff S. A., Lila M. A. Efficient preparative isolation and identification of walnut bioactive components using high-speed counter-current chromatography and LC-ESI-IT-TOF-MS. Food Chemistry. 2014;158:229–238. doi: 10.1016/j.foodchem.2014.02.117. [DOI] [PubMed] [Google Scholar]
- 139.Regueiro J., Sanchez-Gonzalez C., Vallverdu-Queralt A., Simal-Gandara J., Lamuela-Raventos R., Izquierdo-Pulido M. Comprehensive identification of walnut polyphenols by liquid chromatography coupled to linear ion trap-orbitrap mass spectrometry. Food Chemistry. 2014;152:340–348. doi: 10.1016/j.foodchem.2013.11.158. [DOI] [PubMed] [Google Scholar]
- 140.Del Bubba M., Checchini L., Chiuminatto U., Doumett S., Fibbi D., Giordani E. Liquid chromatographic/electrospray ionization tandem mass spectrometric study of polyphenolic composition of four cultivars of Fragaria vesca L. berries and their comparative evaluation. Journal of Mass Spectrometry. 2012;47(9):1207–1220. doi: 10.1002/jms.3030. [DOI] [PubMed] [Google Scholar]
- 141.La Barbera G., Capriotti A. L., Cavaliere C., et al. Comprehensive polyphenol profiling of a strawberry extract (Fragaria × ananassa) by ultra-high-performance liquid chromatography coupled with high-resolution mass spectrometry. Analytical and Bioanalytical Chemistry. 2017;409(8):2127–2142. doi: 10.1007/s00216-016-0159-8. [DOI] [PubMed] [Google Scholar]
- 142.Abdel-Hameed E.-S., Bazaid S., Shohayeb M. RP-HPLC-UV-ESI-MS phytochemical analysis of fruits of Conocarpus erectus L. Chemical Papers. 2014;68(10) doi: 10.2478/s11696-014-0570-6. [DOI] [Google Scholar]
- 143.Saucier C., Jourdes M., Glories Y. Extraction, detection, and quantification of flavano-ellagitannins and ethylvescalagin in a bordeaux red wine aged in oak barrels. Journal of Agricultural and Food Chemistry. 2006;54(19):7349–7354. doi: 10.1021/jf061724i. [DOI] [PubMed] [Google Scholar]
- 144.Simirgiotis M. J., Schmeda-Hirschmann G. Determination of phenolic composition and antioxidant activity in fruits, rhizomes and leaves of the white strawberry (Fragaria chiloensis spp. chiloensis form chiloensis) using HPLC-DAD–ESI-MS and free radical quenching techniques. Journal of Food Composition and Analysis. 2010;23(6):545–553. doi: 10.1016/j.jfca.2009.08.020. [DOI] [Google Scholar]
- 145.Oszmianski J., Wojdylo A., Nowicka P., Teleszko M., Cebulak T., Wolanin M. Determination of phenolic compounds and antioxidant activity in leaves from wild Rubus L. species. Molecules. 2015;20(3):4951–4966. doi: 10.3390/molecules20034951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Mediani A., Abas F., Khatib A., et al. Phytochemical and biological features of Phyllanthus niruri and Phyllanthus urinaria harvested at different growth stages revealed by 1 H NMR-based metabolomics. Industrial Crops and Products. 2015;77:602–613. doi: 10.1016/j.indcrop.2015.09.036. [DOI] [Google Scholar]
- 147.Sousa A. D., Maia I. V., Ribeiro P. R. V., Canuto K. M., Zocolo G. J., Sousa de Brito E. UPLC-QTOF-MSE-based chemometric approach driving the choice of the best extraction process for Phyllanthus niruri. Separation Science and Technology. 2017;52(10):1696–1706. doi: 10.1080/01496395.2017.1298612. [DOI] [Google Scholar]
- 148.Abid M., Yaich H., Cheikhrouhou S., et al. Antioxidant properties and phenolic profile characterization by LC-MS/MS of selected Tunisian pomegranate peels. Journal of Food Science and Technology. 2017;54(9):2890–2901. doi: 10.1007/s13197-017-2727-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Kejian W., Ming D., Xiaosong H., Jianxun Q., Yanbin H. Analysis of polyphenols in walnut kernel by liquid chromatography/electrospray ionization mass spectrometry. Analytical Chemistry. 2009;37(6):867–872. [Google Scholar]
- 150.Katanic J., Matic S., Pferschy-Wenzig E. M., et al. Filipendula ulmaria extracts attenuate cisplatin-induced liver and kidney oxidative stress in rats: In vivo investigation and LC-MS analysis. Food and Chemical Toxicology. 2017;99:86–102. doi: 10.1016/j.fct.2016.11.018. [DOI] [PubMed] [Google Scholar]
- 151.Ying X., Liu M., Liang Q., et al. Identification and analysis of absorbed components and their metabolites in rat plasma and tissues after oral administration of ‘Ershiwuwei Shanhu’ pill extracts by UPLC-DAD/Q-TOF-MS. Journal of Ethnopharmacology. 2013;150(1):324–338. doi: 10.1016/j.jep.2013.08.046. [DOI] [PubMed] [Google Scholar]
- 152.Brighenti V., Groothuis S. F., Prencipe F. P., Amir R., Benvenuti S., Pellati F. Metabolite fingerprinting of Punica granatum L. (pomegranate) polyphenols by means of high-performance liquid chromatography with diode array and electrospray ionization-mass spectrometry detection. Journal of Chromatography A. 2017;1480:20–31. doi: 10.1016/j.chroma.2016.12.017. [DOI] [PubMed] [Google Scholar]
- 153.Romani A., Campo M., Pinelli P. HPLC/DAD/ESI-MS analyses and anti-radical activity of hydrolyzable tannins from different vegetal species. Food Chemistry. 2012;130(1):214–221. doi: 10.1016/j.foodchem.2011.07.009. [DOI] [Google Scholar]
- 154.Ismail B. B., Pu Y., Guo M., Ma X., Liu D. LC-MS/QTOF identification of phytochemicals and the effects of solvents on phenolic constituents and antioxidant activity of baobab (Adansonia digitata) fruit pulp. Food Chemistry. 2019;277:279–288. doi: 10.1016/j.foodchem.2018.10.056. [DOI] [PubMed] [Google Scholar]
- 155.Rush M. D., Rue E. A., Wong A., Kowalski P., Glinski J. A., van Breemen R. B. Rapid determination of procyanidins using MALDI-ToF/ToF mass spectrometry. Journal of Agricultural and Food Chemistry. 2018;66(43):11355–11361. doi: 10.1021/acs.jafc.8b04258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Heras R. M.-L., Quifer-Rada P., Andrés A., Lamuela-Raventós R. Polyphenolic profile of persimmon leaves by high resolution mass spectrometry (LC-ESI-LTQ-orbitrap-MS) Journal of Functional Foods. 2016;23:370–377. doi: 10.1016/j.jff.2016.02.048. [DOI] [Google Scholar]
- 157.Gouveia-Figueira S. C., Castilho P. C. Phenolic screening by HPLC–DAD–ESI/MSn and antioxidant capacity of leaves, flowers and berries of Rubus grandifolius Lowe. Industrial Crops and Products. 2015;73:28–40. doi: 10.1016/j.indcrop.2015.03.022. [DOI] [Google Scholar]
- 158.Di Lecce G., Arranz S., Jauregui O., Tresserra-Rimbau A., Quifer-Rada P., Lamuela-Raventos R. M. Phenolic profiling of the skin, pulp and seeds of Albarino grapes using hybrid quadrupole time-of-flight and triple-quadrupole mass spectrometry. Food Chemistry. 2014;145:874–882. doi: 10.1016/j.foodchem.2013.08.115. [DOI] [PubMed] [Google Scholar]
- 159.Fetni S., Bertella N., Ouahab A., Martinez Zapater J. M., De Pascual-Teresa Fernandez S. Composition and biological activity of the Algerian plant Rosa canina L. by HPLC-UV-MS. Arabian Journal of Chemistry. 2017 doi: 10.1016/j.arabjc.2017.09.013. [DOI] [Google Scholar]
- 160.Chen H., Li M., Zhang C., et al. Isolation and identification of the anti-oxidant constituents from Loropetalum chinense (R. Brown) oliv. based on UHPLC(-)Q-TOF-MS/MS. Molecules. 2018;23(7):p. 1720. doi: 10.3390/molecules23071720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Han F., Li Y., Mao X., Xu R., Yin R. Characterization of chemical constituents in Rhodiola Crenulate by high-performance liquid chromatography coupled with Fourier-transform ion cyclotron resonance mass spectrometer (HPLC-FT-ICR MS) Journal of Mass Spectrometry. 2016;51(5):363–368. doi: 10.1002/jms.3764. [DOI] [PubMed] [Google Scholar]
- 162.Delgado de la Torre M. P., Priego-Capote F., Luque de Castro M. D. Tentative identification of polar and mid-polar compounds in extracts from wine lees by liquid chromatography-tandem mass spectrometry in high-resolution mode. Journal of Mass Spectrometry. 2015;50(6):826–837. doi: 10.1002/jms.3592. [DOI] [PubMed] [Google Scholar]
- 163.Chen B., Long P., Sun Y., et al. The chemical profiling of loquat leaf extract by HPLC-DAD-ESI-MS and its effects on hyperlipidemia and hyperglycemia in rats induced by a high-fat and fructose diet. Food & Function. 2017;8(2):687–694. doi: 10.1039/c6fo01578f. [DOI] [PubMed] [Google Scholar]
- 164.Ashida H., Lv Q., Luo F., et al. Identification of proanthocyanidins from litchi (Litchi chinensis Sonn.) Pulp by LC-ESI-Q-TOF-MS and their antioxidant activity. PLoS One. 2015;10(3) doi: 10.1371/journal.pone.0120480.e0120480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Du L. J., Huang J. P., Wang B., et al. Carbon molecular sieve based micro-matrix-solid-phase dispersion for the extraction of polyphenols in pomegranate peel by UHPLC-Q-TOF/MS. Electrophoresis. 2018;39(17):2218–2227. doi: 10.1002/elps.201800045. [DOI] [PubMed] [Google Scholar]
- 166.Ren Q., Wu C., Ren Y., Zhang J. Characterization and identification of the chemical constituents from tartary buckwheat (Fagopyrum tataricum Gaertn) by high performance liquid chromatography/photodiode array detector/linear ion trap FTICR hybrid mass spectrometry. Food Chemistry. 2013;136(3-4):1377–1389. doi: 10.1016/j.foodchem.2012.09.052. [DOI] [PubMed] [Google Scholar]
- 167.Sobeh M., Mahmoud M. F., Hasan R. A., et al. Chemical composition, antioxidant and hepatoprotective activities of methanol extracts from leaves of Terminalia bellirica and Terminalia sericea (Combretaceae) PeerJ. 2019;7:p. e6322. doi: 10.7717/peerj.6322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Chen Y., Wang J., Ou Y., et al. Cellular antioxidant activities of polyphenols isolated from Eucalyptus leaves (Eucalyptus grandis × Eucalyptus urophylla GL9) Journal of Functional Foods. 2014;7:737–745. doi: 10.1016/j.jff.2013.12.003. [DOI] [Google Scholar]
- 169.Torres-León C., Ventura-Sobrevilla J., Serna-Cock L., Ascacio-Valdés J. A., Contreras-Esquivel J., Aguilar C. N. Pentagalloylglucose (PGG): a valuable phenolic compound with functional properties. Journal of Functional Foods. 2017;37:176–189. doi: 10.1016/j.jff.2017.07.045. [DOI] [Google Scholar]
- 170.Al-Sayed E., Esmat A. Hepatoprotective and antioxidant effect of ellagitannins and galloyl esters isolated from Melaleuca styphelioides on carbon tetrachloride-induced hepatotoxicity in HepG2 cells. Pharmaceutical Biology. 2016;54(9):1727–1735. doi: 10.3109/13880209.2015.1125933. [DOI] [PubMed] [Google Scholar]
- 171.Sun Y.-q., Tao X., Men X.-m., Xu Z.-w., Wang T. In vitro and in vivo antioxidant activities of three major polyphenolic compounds in pomegranate peel: ellagic acid, punicalin, and punicalagin. Journal of Integrative Agriculture. 2017;16(8):1808–1818. doi: 10.1016/s2095-3119(16)61560-5. [DOI] [Google Scholar]
- 172.Roleira F. M., Tavares-da-Silva E. J., Varela C. L., et al. Plant derived and dietary phenolic antioxidants: anticancer properties. Food Chemistry. 2015;183:235–258. doi: 10.1016/j.foodchem.2015.03.039. [DOI] [PubMed] [Google Scholar]
- 173.Xu J., Zhang G., Tong Y., Yuan J., Li Y., Song G. Corilagin induces apoptosis, autophagy and ROS generation in gastric cancer cells in vitro. International Journal of Molecular Medicine. 2018;43(2):967–979. doi: 10.3892/ijmm.2018.4031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Huang S.-T., Yang R.-C., Pang J.-H. S. Aqueous extract of Phyllanthus urinaria induces apoptosis in human cancer cells. The American Journal of Chinese Medicine. 2004;32(2):175–183. doi: 10.1142/s0192415x04001849. [DOI] [PubMed] [Google Scholar]
- 175.Jia L., Jin H., Zhou J., et al. A potential anti-tumor herbal medicine, Corilagin, inhibits ovarian cancer cell growth through blocking the TGF-β signaling pathways. BMC Complementary and Alternative Medicine. 2013;13(1) doi: 10.1186/1472-6882-13-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Javelaud D., Mauviel A. Crosstalk mechanisms between the mitogen-activated protein kinase pathways and Smad signaling downstream of TGF-beta: implications for carcinogenesis. Oncogene. 2005;24(37):5742–5750. doi: 10.1038/sj.onc.1208928. [DOI] [PubMed] [Google Scholar]
- 177.Kant R., Yen C. H., Hung J. H., et al. Induction of GNMT by 1,2,3,4,6-penta-O-galloyl-beta-D-glucopyranoside through proteasome-independent MYC downregulation in hepatocellular carcinoma. Science Reports. 2019;9(1) doi: 10.1038/s41598-018-37292-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Chen L. G., Huang W. T., Lee L. T., Wang C. C. Ellagitannins from Terminalia calamansanai induced apoptosis in HL-60 cells. Toxicology in Vitro. 2009;23(4):603–609. doi: 10.1016/j.tiv.2009.01.020. [DOI] [PubMed] [Google Scholar]
- 179.Carneiro C. C., de Moraes-Filho A. V., Fernandes A. S., da Costa Santos S., de Melo e Silva D., Chen L. C. Cytotoxic and chemopreventive effects of gemin D against different mutagens using in vitro and in vivo assays. Anti-Cancer Agents in Medicinal Chemistry. 2017;17(5):712–718. doi: 10.2174/1871520616666160906092502. [DOI] [PubMed] [Google Scholar]
- 180.Chen P. S., Li J. H. Chemopreventive effect of punicalagin, a novel tannin component isolated from Terminalia catappa, on H-ras-transformed NIH3T3 cells. Toxicology Letters. 2006;163(1):44–53. doi: 10.1016/j.toxlet.2005.09.026. [DOI] [PubMed] [Google Scholar]
- 181.Berdowska I., Zieliński B., Saczko J., Sopel M., Gamian A., Fecka I. Modulatory impact of selected ellagitannins on the viability of human breast cancer cells. Journal of Functional Foods. 2018;42:122–128. doi: 10.1016/j.jff.2017.12.053. [DOI] [Google Scholar]
- 182.Wang R., Ma M., Gong X., Fan X., Walsh P. J. Reductive cross-coupling of aldehydes and imines mediated by visible light photoredox catalysis. Organic Letters. 2019;21(1):27–31. doi: 10.1021/acs.orglett.8b03394. [DOI] [PubMed] [Google Scholar]
- 183.Li K., Han X., Li R., et al. Composition, antivirulence activity, and active property distribution of the fruit of Terminalia chebula Retz. Journal of Food Science. 2019;84(7):1721–1729. doi: 10.1111/1750-3841.14655. [DOI] [PubMed] [Google Scholar]
- 184.Behrendt P., Perin P., Menzel N., et al. Pentagalloylglucose, a highly bioavailable polyphenolic compound present in Cortex moutan, efficiently blocks hepatitis C virus entry. Antiviral Research. 2017;147:19–28. doi: 10.1016/j.antiviral.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 185.Tu Z., Xu M., Zhang J., et al. Pentagalloylglucose inhibits the replication of rabies virus via mediation of the miR-455/SOCS3/STAT3/IL-6 pathway. Journal of Virology. 2019;93(18) doi: 10.1128/jvi.00539-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Reddy B. U., Mullick R., Kumar A., Sudha G., Srinivasan N., Das S. Small molecule inhibitors of HCV replication from pomegranate. Science Reports. 2015;4(1):p. 5411. doi: 10.1038/srep05411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Liu C., Cai D., Zhang L., et al. Identification of hydrolyzable tannins (punicalagin, punicalin and geraniin) as novel inhibitors of hepatitis B virus covalently closed circular DNA. Antiviral Research. 2016;134:97–107. doi: 10.1016/j.antiviral.2016.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Nandini H. S., Naik P. R. Action of corilagin on hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced diabetic rats. Chem Chemico-Biological Interactions. 2019;299:186–193. doi: 10.1016/j.cbi.2018.12.012. [DOI] [PubMed] [Google Scholar]
- 189.Kato-Schwartz C. G., Bracht F., Gonçalves G. d. A., et al. Inhibition of α-amylases by pentagalloyl glucose: kinetics, molecular dynamics and consequences for starch absorption. Journal of Functional Foods. 2018;44:265–273. doi: 10.1016/j.jff.2018.03.025. [DOI] [Google Scholar]
- 190.Yin J., Ahn H. S., Ha S. Y., et al. Anti-skin ageing effects of phenolic compounds from Carpinus tschonoskii. Natural Product Research. 2019;33(22):3317–3320. doi: 10.1080/14786419.2018.1497026. [DOI] [PubMed] [Google Scholar]
- 191.Shanmuganathan S., Angayarkanni N. Chebulagic acid and chebulinic acid inhibit TGF-beta1 induced fibrotic changes in the chorio-retinal endothelial cells by inhibiting ERK phosphorylation. Microvascular Research. 2019;121:14–23. doi: 10.1016/j.mvr.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 192.Shanmuganathan S., Angayarkanni N. Chebulagic acid chebulinic acid and gallic acid, the active principles of triphala, inhibit TNFalpha induced pro-angiogenic and pro-inflammatory activities in retinal capillary endothelial cells by inhibiting p38, ERK and NFkB phosphorylation. Vascular Pharmacology. 2018;108:23–35. doi: 10.1016/j.vph.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 193.Li Z., Percival S. S., Bonard S., Gu L. Fabrication of nanoparticles using partially purified pomegranate ellagitannins and gelatin and their apoptotic effects. Molecular Nutrition & Food Research. 2011;55(7):1096–1103. doi: 10.1002/mnfr.201000528. [DOI] [PubMed] [Google Scholar]
- 194.Song G. B., Xu J., Zheng H., et al. novel soluble dietary fiber-tannin self-assembled film: a promising protein protective material. Journal of Agricultural and Food Chemistry. 2015;63(24):5813–5820. doi: 10.1021/acs.jafc.5b00192. [DOI] [PubMed] [Google Scholar]
- 195.Zhu W., Jia Y., Peng J., Li C. M. Inhibitory effect of persimmon tannin on pancreatic lipase and the underlying mechanism in vitro. Journal of Agricultural and Food Chemistry. 2018;66(24):6013–6021. doi: 10.1021/acs.jafc.8b00850. [DOI] [PubMed] [Google Scholar]
- 196.Landete J. M. Ellagitannins, ellagic acid and their derived metabolites: a review about source, metabolism, functions and health. Food Research International. 2011;44(5):1150–1160. doi: 10.1016/j.foodres.2011.04.027. [DOI] [Google Scholar]
- 197.Zhao W., Shi F., Guo Z., Zhao J., Song X., Yang H. Metabolite of ellagitannins, urolithin A induces autophagy and inhibits metastasis in human sw620 colorectal cancer cells. Molecular Carcinogenesis. 2018;57(2):193–200. doi: 10.1002/mc.22746. [DOI] [PMC free article] [PubMed] [Google Scholar]