Abstract
Collective cell migration is required in a vast array of biological phenomena, including organogenesis and embryonic development. The mechanisms that underlie collective cell migration involve not only the morphogenetic changes associated with single cell migration, but also require the maintenance of cell-cell junctions during movement. Additionally, cell shape changes and polarity must be coordinated in a multicellular manner in order to preserve directional movement in the migrating cohort, and often relates to multiple functions of common signaling pathways. In this review, we summarize the current understanding of the mechanisms underlying higher order tissue organization during migration, with particular focus on the interplay between cell adhesion and signaling that we propose can be tuned to support different types of collective movements.
Keywords: collective cell migration, Drosophila, Xenopus, Ciona, adhesion, Integrin, Cadherin, Rap1, Myospheroid, signaling, FGF, mesoderm, epithelial sheet, border cells
Introduction
A fundamental goal of the field of developmental biology is building a more comprehensive understanding of the hierarchical relationship between single cells and groups of cells. Organismal development, which involves the interplay between gene regulation, patterning, and cell behavior, is a highly complex process that requires cells to move synchronously in highly ordered cohorts. The coordinated movement and maintenance of intercellular contact of two or more cells along a substrate is known as collective cell migration, and is required throughout development for the formation of complex biological structures, including tissues and organs. Collectively migrating cells typically maintain some form of intercellular adhesion in order to allow for movement either along an extracellular matrix (ECM) or in a three-dimensional space, such as between layers of tissue [1,2].
Migrating individual cells typically accomplish directional movement via the coordinated contraction of actomyosin networks in conjunction with polarized localization of actin nucleators and cell surface proteins. This remodeling of the cytoskeletal structure gives rise to different classes of protrusive behavior including lamellipodia, filopodia, lobopodia, invadopodia, and blebbing that are often found at the leading edge of a migrating cell [3,4]. These behaviors have been extensively studied in in vitro cell culture systems, and have provided a wealth of insight into the effectors that govern protrusive activity in migrating cells, including the activities of Rho GTPases and ligand/receptor pairs to allow for chemotaxis [5]. However, cell culture and ex vivo explant models provide limited insight into the dynamics of collective cell migration in vivo, as replication of the myriad extracellular factors and environment can prove incredibly difficult and may not offer an accurate simulacrum for the endogenous environment. Additionally, successful culture of cells and tissues of interest can prove to be a major challenge. This necessitates the use of model organisms, many of which have found extensive historical usage such that optimized techniques for testing observation can be employed relatively quickly and easily, and novel techniques intended to deepen the biological questions interrogated are continually being developed.
In general, migrating cell collectives utilize many of the same mechanisms for mediating directional movement as those of single cells, including dynamic remodeling of the cytoskeleton to form polarized protrusions, but must be able to coordinate these activities across a group of cells (Figure 1). Current research into collective cell migration emphasizes: 1) the necessity of maintaining cell adhesion and communication between cells that make up a cohort, which can either be transient or relatively stable [6–8], as well as 2) establishing spatial polarity within a cohort, which can often involve the assignment of so-called ‘leader’ and ‘follower’ cells [9,10]. In this minireview, we discuss insights obtained from in vitro, in vivo, and in silico models for studying collective cell migration, comparing and contrasting studies in different systems, with particular emphasis on findings from the past two years that have advanced understanding of spatial organization of tissues during collective cell migration.
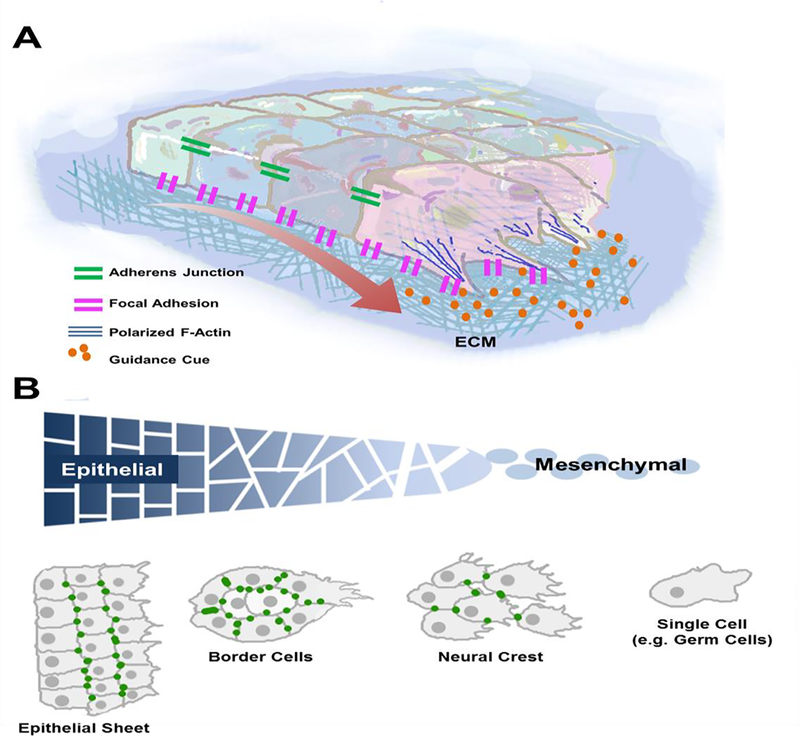
Figure 1. General model for collective cell migration and examples.
A) Directional movement of a cell collective requires both intercellular adhesions (via adherens junctions) and interactions with the substrate/environment (via focal adhesions). When exposed to a guidance cue, leader cells at the front typically lose epithelial character and become more protrusive, which is accompanied by a loss of intercellular adhesive contact and an increase in polarized F-actin to form protrusions. B) The epithelial to mesenchymal transition is marked by dynamic adjustments to adhesive states and changes to overall spatial arrangement. Shown are some examples of tissues that run the gamut of epithelial to mesenchymal characteristics, including epithelial sheets, Drosophila border cells, neural crest cells, and single cells (such as primordial germ cells).
Epithelial monolayer migration: an interplay of extracellular signaling and intracellular communication mediates differential cell behavior within a cohort
Migrating epithelial collectives require a highly coordinated remodeling of cell-cell adhesion, which is extensively described in multiple cell migration models. Two classes of adhesion molecules are typically associated with collective epithelial migration: Integrins and cadherins. Integrins and cadherins generally are not present in overlapping domains; integrins are found in focal adhesions (FAs) that can consist of a very diverse variety of integrin subunit combinations, while cadherins form homotypic interactions at adherens junctions (AJs). In addition to maintaining apicobasal polarity, the spatial distribution of intercellular adhesion across the epithelium, known as planar-polarized adhesion, can drive directed movement of groups of cells [11].
How multiple pathways influence collective motility was recently explored by Stedden et al., who used the Drosophila egg chamber to model the relationship between polarized signaling and collective migration of epithelial cells [12]. Each egg chamber consists of inner germ cells surrounded by a layer of follicle cells, which is in turn ensheathed by a basement membrane ECM. Each chamber forms one egg; in order to form the characteristic elongated shape of the egg, the follicle cells undergo collective unidirectional migration along the basement membrane [13,14]. This movement is mediated by cooperative signaling between basally-polarized receptor protein tyrosine phosphatase (RPTP) Lar at the leading edge of each follicle cell, and basally-polarized cadherin Fat2 at each cell’s trailing edge [15,16]. Stedden et al. demonstrate that in addition to the Fat2/Lar system, the semaphorin Sema-5C is autonomously required within the follicle cells and not the surrounding environment, and is basally enriched at the leading edge of each cell to suppress protrusion formation at the trailing edge of the cell ahead. This is accomplished via transduction by Plexin A as a likely receptor for the Sema-5C ligand. Additionally, there is apparent crosstalk between signaling via Sema-5C and Lar activity; removing one copy of Lar attenuates egg shape defects in Sema-5C mutants, demonstrating a genetic interaction between the two systems. This work demonstrates how multiple guidance pathways can coordinate collective epithelial motility within the same tissue type, of which there are few well-worked samples.
How to be a good follower: Adhesion dynamics regulate collective cell migration
To move efficiently as a group, migrating cells must balance cell-cell attachments to other cells in the migrating collective with the tendency to move in a directional manner, and in several recent studies cell signaling has been linked to modulation of adherens junction dynamics in the support of collective cell migration (Figure 2).
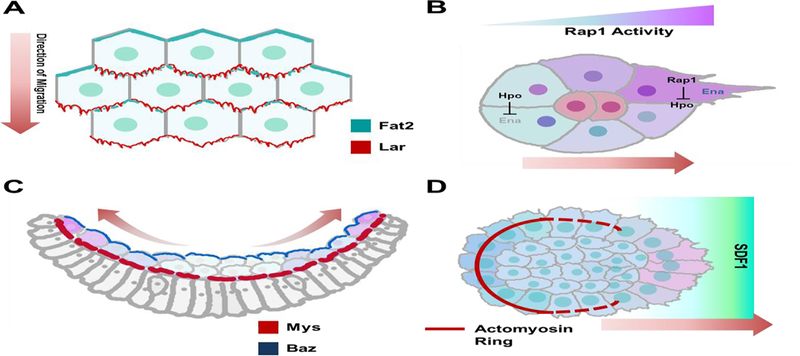
Figure 2. Different modes of collective migration mediated by different aspects of intracellular signaling.
A) Drosophila Follicle cell migration is mediated by planar polarized Fat2 and Lar. Fat2 expression in the trailing edge of cells ahead induce protrusive activity in the leading edge of cells behind, while Lar activity in the leading edge of the cells behind mediate trailing edge retraction in the preceding cell. B) High Rap1 activity suppresses Hpo activity in Drosophila border cells, which allows for Ena-mediated F-actin polymerization to form directional protrusions. In cells with low Rap1 activity, Hpo signaling suppresses Ena. C) Mesoderm spreading in gastrulating Drosophila embryos involves a MET that culminates in a single layer of cells. This process is supported by basal expression of the β-integrin subunit Mys. D) In neural crest cells, contraction of an actomyosin cable at the rear of the cell cohort drives intercalation, thus supporting directed movement. This contractility is inhibited in the front by exposure to SDF1 cue, ensuring that contractility is polarized to the rear of the group of cells.
The zebrafish posterior lateral line primordium is a group of cells that collectively migrate in embryos under the skin from near the ear to the tip of the tail, depositing neuromast cells periodically [1,17]. Wnt, Notch, FGF and chemokine signaling work together to support these cells’ fate, morphogenesis, and migration. Many prior studies have focused on how signals intersect to support directional movement and include self-generated chemokine gradients, that effectively steer migration of leading cells [18,19]. However, mechanical coupling of trailing cells to leading cells is also very important as it allows cells along the entire length of the primordium to move as a cohesive cohort [20,21].
Similarly, in the case of mesoderm migration at gastrulation in Drosophila, invaginated mesoderm cells move as a collective in a directed fashion, migrating directly upon the ectoderm that serves as substrate. This interaction initially occurs in the absence of a detectable ECM, which forms and becomes discernible only towards the end of the migration process [22]. Proper regulation of cell-cell adhesion in the mesoderm was determined to be crucial for supporting proper spreading of cells along this surface. Initially, it was proposed that the dorsally-directed movement exhibited by mesoderm cells as they migrate upon the ectoderm substratum is directed by FGF signaling functioning as a chemoattractive cue [23–25]. However, a recent study from our lab supports the view that a temporal decrease in adherens junctions may additionally (or alternatively) support this directional movement [26]. Essentially, mesoderm cells decrease cell-cell attachments to other mesoderm cells over time, and instead establish new connections to the ectodermal substratum, resulting ultimately in the formation of a monolayer of mesoderm cells important for proper specification of a number of distinct mesoderm cell types. Decreased FGF leads to a non-collective mesoderm migration phenotype, including decrease in proliferation; on the other other hand, increased FGF signaling leads to retention of high numbers of intercellular junctions and increased proliferation. It was proposed that FGF supports EMT both by decreasing junction numbers as well as increasing the tendency of cells to proliferate, and a loss of FGF signaling ablates the ability of cells to dynamically remodel adhesions to promote collective movement (Figure 2).
Parallels exist to a recent study by Kota et al., in which it was shown that the M-Ras GTPase and its effector Shoc-2, a scaffold protein, contribute to junction turnover by modulating the interaction of E-Cadherin with beta-catenin to affect intercellular adhesiveness in MCF10A cells, a non-transformed breast epithelial line [27]. It was suggested that Shoc-2 binds active M-Ras at cell-cell junctions to activate ERK, which acts on p120 catenin and/or other substrates, leading to dynamic turnover of cell-cell junctions.
It is possible that FGF signaling, and/or other receptor tyrosine kinase ERK-activating pathways, act directly on adherens junction components to regulate connections between cells in a migrating collective. Previous studies have alluded to such an idea [28]. Furthermore, collective cell migration by a dynamic exchange of homotypic cell-cell attachments for heterotypic ones may be a useful mechanism to support EMT when it is followed by a complementary mesenchymal to epithelial transition (MET), as relocating adherens junctions rather than clearing them completely may allow cells to reform attachments to reacquire epithelial characteristics more quickly.
Changing leadership positions: shifting paradigms in spatial organization of cell collectives
A prevailing view of collective cell migration involves a specialized group of cells (leader cells) primarily controlling directionality of the cohort, with follower cells subordinate to the actions of the leader cells. Epithelial monolayer migration presents several classic examples of this leader-follower cell model for directionality and arrangement. In recent years, the prevailing paradigm regarding leader-follower interactions has been expanded to account for a plethora of different migration interactions that do not necessarily fit this archetypal model for collective cell migration.
Migrating cells often encounter other cell types and navigate a variety of different topologies over the course of their movement. How cells modulate different responses when coming in contact with different cell types or tissues is a subject of extensive investigation, with multiple models postulating how a migrating collective of cells can coordinate and maintain cell-cell contacts while maintaining organization and directionality. The ability of migratory cells to form spatially organized patterns via heterotypic interactions with other cell types and by controlling intercellular adhesion is crucial to the formation of distinct organs. Multiple studies have demonstrated that substrate stiffness directly affects collective cell behavior -- stiffer, more rigid substrates generate a more effective scaffold for migratory behavior [29–31]. Neural crest (NC) cells, which are a population of precursor cells that give rise to a diverse number of tissues (including pigment cells, craniofacial skeletal and connective tissue, and various neuronal lineages) arise along the neural tube of the ectoderm and subsequently undergo an EMT to generate a population of cells that migrate at a distance as spatially distinct streams [32].
A number of different migratory cues are required for proper NC migration, including various combinations of ephrins as inhibitory signals [33]. As these ephrins are expressed in the NC and surrounding mesoderm in frogs, and in other vertebrates similar inhibitory cues are expressed in the placodal tissues surrounding the NC streams (e.g Semaphorin-3F in mice [34] and Robo2 in chick [35]), it was believed that a specific pattern of inhibitors in surrounding tissues shapes the migratory NC streams. However, as this inhibitor expression pattern arises at the onset of migration and is instead observed as a single, continuous band next to the NC prior to migration, it is unlikely that this streaming behavior arises from an inhibitory spatial pattern. Recently, the Mayor group utilized both in silico and in vivo methods to demonstrate that contrary to a patterned extracellular milieu directing formation of these NC cell streams, direct interactions with the placodes are sufficient to drive streaming behavior, which acts independently of any inhibitory pattern in surrounding cells [36]. Instead, a chase-and-run mechanism combining Sdf1-mediated chemotaxis and short-range repulsion in the placodes accounts for the formation of NC streams irrespective of proximity to nearby endoderm or mesoderm. Furthermore, the shapes of the cell streams are controlled by levels of intercellular adhesion: reducing the levels of adhesion by inhibiting N-cadherin expression within NCs resulted in thinner streams [36].
Most models of leader-follower interactions in migrating collectives describe interactions between the leader cells and follower cells as being mediated by an actomyosin network. In a recent manuscript authored by Shellard et al., researchers describe how the rear of a migrating cell cohort can drive collective chemotaxis in response to guidance cues [37]. By using ex vivo explants combined with in vivo live imaging of Xenopus and zebrafish cranial neural crest cells, Shellard et al. describe the existence of a multicellular actomyosin ring at the periphery of the migrating cell group (Figure 2). Using laser ablation techniques, Shellard et al. found that collective chemotaxis was impaired by disrupting the actomyosin ring at the rear of the cohort, while ablations in the front cells did not result in aberrant chemotaxis. Furthermore, by utilizing an optogenetic approach to increase or decrease contractility via timed exposure to blue light, they found that contractility at the rear of the neural crest cohort is sufficient to drive collective directional movement even in the absence of a chemotactic cue; this effect was not observed when contractility was induced in the front cells. These results demonstrate a shift in the current understanding of leader-follower interactions: cells in the front of a group of cells do not always drive directionality of the collective.
Drosophila border cells are another extensively-characterized system for investigating both autonomous and non-autonomous mechanisms of collective cell behavior that do not necessarily fit the traditional model of leader-follower interactions. Border cells are composed of about eight motile cells surrounding two immobile polar cells and migrate through a constrained space between much larger nurse cells to reach the developing oocyte [38]. Border cells delaminate from the follicular epithelium, maintaining apicobasal polarity while exhibiting canonical leader-follower protrusive dynamics, with a single front cell displaying high F-actin rich protrusive activity. [39,40]. During migration, an external oocyte-secreted gradient of platelet-derived/vascular endothelium-derived growth factor (PVF) and epidermal growth factor (EGF) signaling determines leader cell identity [41]; high levels of receptor tyrosine signaling within a cell directly relates to high levels of Rac1, which leads to increased protrusive activity [42,43]. Rac1 levels in the leader cell are maintained via the activity of the small GTPase Rab11, which functions in recycling endosomes to resolve spatial localization of Rac1 signaling, as well as by binding to the cytoskeleton regulator Moesin to facilitate cell-cell communication [44]. More recently, Chang et al. demonstrated that in addition to PVF/EGF-driven upregulation of Rac1, the formation of polarized protrusions in the leader cell is supported by negative regulation of the Hippo pathway by Rap1 [45]. The Hippo pathway was previously shown to support polarized actin assembly in border cells [46]; Chang et al. present evidence that Rap1 directly binds to Hippo to prevent its autophosphorylation, which interferes with its ability to polarize actin to support protrusive activity. As Hippo is a negative regulator of the actin elongation factor Enabled (Ena) [46,47], Rap1 relieves suppression of Ena to promote protrusive activity (Figure 2). These findings are consistent with another study by Sawant et al. demonstrating a requirement for Rap1 in ensuring that protrusions are limited to the front of the border cell cohort and promoting protrusion elongation [48]. Therefore, leader cell identity is not simply conferred solely by the upregulation of actin protrusions, but by a corresponding interplay with parallel signaling pathways that, in this case, suppress the activity of negative regulators of polarized actin assembly.
Multitasking skills required: New examples of a single pathway controlling multiple functions to drive spatial organization and adhesion remodeling
As we have described in the previous section, the spatial organization of a collective often requires input from multiple signaling pathways; these can work autonomously within the migrating collective, or via a non-autonomous mechanism via interaction with other tissues and the extracellular milieu. Recently, new examples of how a single signaling pathway can mediate multiple functions in order to direct a spatial logic within a migrating cohort of cells have been described. In this section, we describe two recent examples of one signaling pathway mediating multiple functions to drive collective migration: Discoidin domain receptor functions in Ciona cardiopharyngeal precursors, and FGF signaling in Drosophila longitudinal muscle precursors.
In the ascidian Ciona robusta, the cardiogenic progenitors also known as trunk ventral cells (TVCs) serve as a model for collective cell migration in the simplest case possible as two cells migrating in a concerted fashion [49]. Interactions with a number of tissues serve different roles in supporting migration of these cells: contact with the posterior mesenchyme provides signals that support specification of leader-trailer polarity, which is maintained by contact with the trunk endoderm, while adhesion to the epidermis provides a signal that supports their cohesion [49]. Recent studies have shed light on the mechanism, in that endodermal cells secrete a collagen, Col9-a1, that is deposited in the basal epidermal matrix and promotes collagen-binding Discoidin-domain receptor (Ddr) activation at the ventral membrane of migrating TVCs [50]. In addition, Ddr promotes leader-trailer polarized BMP-Smad signaling independent of its role in cell adhesion, demonstrating a novel example of how a single pathway can control multiple functions to promote polarized movement.
Parallels exist in the regulation of caudal visceral mesoderm (CVM) migration in Drosophila, as FGF signaling also plays a dual role in supporting cell adhesion as well as additional roles, possibly supporting cell polarity, within the migrating collective. Previously, a central role for FGF signaling was identified in ensuring that CVM cells move in a directional manner; these cells, which initially specify as a single cohort of cells before forming symmetrical streams that migrate over a span of six hours, migrate abnormally when FGF signaling components are ablated [51,52]. While restoring FGF signaling in the tissue serving as a substrate for the CVM (the trunk visceral mesoderm, or TVM) restores bilateral directional migration of the CVM, restoring FGF signaling in the CVM cells themselves similarly rescued migration defects. This suggests that the role FGF signaling plays in CVM migration is not simply a chemotactic one; it is likely that FGF plays additional functions in promoting forward movement. Most surprisingly, the relationship between the CVM and the TVM substrate is not necessarily one-sided -- while the TVM serves as a source of FGF ligand and as a substrate for migration, the CVM exerts significant morphogenetic influence on the TVM, manifesting as contralateral ‘bridges’ that correlate with abnormal CVM migration when FGF signaling is ablated [53]. Furthermore, these bridges do not manifest when the integrin βPS1 subunit (Mys) is expressed in the TVM in an FGF mutant background. As such, CVM migration represents an example of the interplay of multiple functions for a single pathway: FGF signaling plays a stabilizing role in controlling intercellular adhesion by controlling integrin levels, as well as ensuring proper pathfinding of the CVM cells.
In several cases, dual-function molecules that promote cell polarity (e.g. Ddr, FGF/Mys) have been implicated in supporting this process. It may be no accident that the same molecule is used to serve multiple roles in supporting collective cell migration: regulator of cell adhesion, both of migrating cell-cell and cell-substratum contacts; as well as regulator of spatial signaling pathway activation. Tight coordination of cell adhesion and signaling may be needed to control collective cell migration and using one molecule of dual-function may be pivotal to ensure both processes are in sync.
In this minireview, we have chosen to highlight recent studies that suggest the spatial regulation of signaling or cell adhesion is pivotal towards supporting collective cell movement. While it has long been demonstrated that migrating cells must remodel adhesion domains in order to facilitate directional movement, technical advancements in generating in vitro, in vitro, and in silico data that offer a more nuanced glimpse into the molecular mechanisms that couple signaling and adhesion within recent years have broadened our understanding of how complex cell behaviors are accomplished. In short, the field has improved its current understanding of how localized changes in adhesion and tension drive global migratory behaviors, as well as the intracellular dynamics that drive these changes via signal transduction. We also highlighted how multimodal functions of a common signaling pathway work together to control collective migration, facilitating intercellular adhesion and interaction with the extracellular substrate. While recent studies have demonstrated an essential role for the environment in controlling collective behavior, we have now seen that other behaviors (such as NC streaming) are regulated by other mechanisms that do not necessarily fit a traditional substrate-driven model. These advancements in our understanding demonstrate fertile ground for continued insights into collective cell behavior dynamics.
Acknowledgements
FM was funded by NIH grant R35GM118146 to AS. We are grateful to J. Sun and Y.Y. Bernadskaya for comments on the manuscript, and apologize to all colleagues whose work we could not cite due to space constraints.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of Interest: The authors declare no conflict of interest.
References and Recommended Reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
- 1.Friedl P, Gilmour D: Collective cell migration in morphogenesis, regeneration and cancer. Nature Reviews Molecular Cell Biology 2009, 10:445–457. [DOI] [PubMed] [Google Scholar]
- 2.Rørth P: Collective guidance of collective cell migration. Trends in Cell Biology 2007, 17:575–579. [DOI] [PubMed] [Google Scholar]
- 3.Ridley AJ: Life at the Leading Edge. Cell 2011, 145:1012–1022. [DOI] [PubMed] [Google Scholar]
- 4.Sixt M: Cell migration: fibroblasts find a new way to get ahead. J Cell Biol 2012, 197:347–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Y, Senoo H, Sesaki H, Iijima M: Rho GTPases orient directional sensing in chemotaxis. Proc Natl Acad Sci U S A 2013, 110:E4723–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Campbell K, Casanova J: A role for E-cadherin in ensuring cohesive migration of a heterogeneous population of non-epithelial cells. Nat Commun 2015, 6:7998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barriga EH, Mayor R: Embryonic Cell–Cell Adhesion. Current Topics in Developmental Biology 2015, doi: 10.1016/bs.ctdb.2014.11.023. [DOI] [PubMed] [Google Scholar]
- 8.Venhuizen J-H, Zegers MM: Making Heads or Tails of It: Cell–Cell Adhesion in Cellular and Supracellular Polarity in Collective Migration. Cold Spring Harbor Perspectives in Biology 2017, 9:a027854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Theveneau E, Linker C: Leaders in collective migration: are front cells really endowed with a particular set of skills? F1000Res 2017, 6:1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Etienne-Manneville S: Neighborly relations during collective migration. Curr Opin Cell Biol 2014, 30:51–59. [DOI] [PubMed] [Google Scholar]
- 11.Weber GF, Bjerke MA, DeSimone DW: Integrins and cadherins join forces to form adhesive networks. J Cell Sci 2011, 124:1183–1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stedden CG, Menegas W, Zajac AL, Williams AM, Cheng S, Özkan E, Horne-Badovinac S: Planar-Polarized Semaphorin-5c and Plexin A Promote the Collective Migration of Epithelial Cells in Drosophila. Curr Biol 2019, 29:908–920.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]; • This manuscript describes a role for Sema5C in supporting planar-polarized Fat2 and Lar-mediated protrusive activity in the leading edge of migrating Drosophila follicle cells by inhibiting protrusion formation in the trailing edge of cells ahead, likely via a Plexin A-dependent mechanism. This combination of polarized activating and inhibiting signals coordinates unidirectional movement of the follicle cell sheet.
- 13.Cetera M, Ramirez-San Juan GR, Oakes PW, Lewellyn L, Fairchild MJ, Tanentzapf G, Gardel ML, Horne-Badovinac S: Epithelial rotation promotes the global alignment of contractile actin bundles during Drosophila egg chamber elongation. Nat Commun 2014, 5:5511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Horne-Badovinac S, Bilder D: Mass transit: epithelial morphogenesis in the Drosophila egg chamber. Dev Dyn 2005, 232:559–574. [DOI] [PubMed] [Google Scholar]
- 15.Barlan K, Cetera M, Horne-Badovinac S: Fat2 and Lar Define a Basally Localized Planar Signaling System Controlling Collective Cell Migration. Dev Cell 2017, 40:467–477.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Viktorinová I, König T, Schlichting K, Dahmann C: The cadherin Fat2 is required for planar cell polarity in the Drosophila ovary. Development 2009, 136:4123–4132. [DOI] [PubMed] [Google Scholar]
- 17.Aman A, Piotrowski T: Cell migration during morphogenesis. Developmental Biology 2010, 341:20–33. [DOI] [PubMed] [Google Scholar]
- 18.Valentin G, Haas P, Gilmour D: The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr Biol 2007, 17:1026–1031. [DOI] [PubMed] [Google Scholar]
- 19.Dambly-Chaudière C, Cubedo N, Ghysen A: Control of cell migration in the development of the posterior lateral line: antagonistic interactions between the chemokine receptors CXCR4 and CXCR7/RDC1. BMC Dev Biol 2007, 7:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dalle Nogare D, Chitnis AB: A framework for understanding morphogenesis and migration of the zebrafish posterior Lateral Line primordium. Mech Dev 2017, 148:69–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dalle Nogare D, Somers K, Rao S, Matsuda M, Reichman-Fried M, Raz E, Chitnis AB: Leading and trailing cells cooperate in collective migration of the zebrafish posterior lateral line primordium. Development 2014, 141:3188–3196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Trisnadi N, Stathopoulos A: Ectopic expression screen identifies genes affecting Drosophila mesoderm development including the HSPG Trol. G3 2014, 5:301–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murray MJ, Saint R: Photoactivatable GFP resolves Drosophila mesoderm migration behaviour. Development 2007, 134:3975–3983. [DOI] [PubMed] [Google Scholar]
- 24.Stathopoulos A, Levine M: Whole-genome analysis of Drosophila gastrulation. Curr Opin Genet Dev 2004, 14:477–484. [DOI] [PubMed] [Google Scholar]
- 25.Clark IBN, Muha V, Klingseisen A, Leptin M, Müller H-AJ: Fibroblast growth factor signalling controls successive cell behaviours during mesoderm layer formation in Drosophila. Development 2011, 138:2705–2715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sun J, Stathopoulos A: FGF controls epithelial-mesenchymal transitions during gastrulation by regulating cell division and apicobasal polarity. Development 2018, 145. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• This manuscript describes a central role for FGF signaling in supporting a more prolonged EMT of migrating mesodermal cells during Drosophila gastrulation. Subsequent to invagination and collapse of the mesodermal tube, mesoderm cells slowly lose intercellular adhesion in order to support collective movement while maintaining cohesiveness. The mesoderm then undergoes a newly characterized MET to form a monolayer, whereupon integrin contacts with the substratum are established basally and the polarity protein Bazooka is localized apically.
- 27.Kota P, Terrell EM, Ritt DA, Insinna C, Westlake CJ, Morrison DK: M-Ras/Shoc2 signaling modulates E-cadherin turnover and cell-cell adhesion during collective cell migration. Proc Natl Acad Sci U S A 2019, 116:3536–3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Birchmeier W, Weidner KM, Behrens J: Molecular mechanisms leading to loss of differentiation and gain of invasiveness in epithelial cells. J Cell Sci Suppl 1993, 17:159–164. [DOI] [PubMed] [Google Scholar]
- 29.Barriga EH, Franze K, Charras G, Mayor R: Tissue stiffening coordinates morphogenesis by triggering collective cell migration in vivo. Nature 2018, 554:523–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Saxena N, Mogha P, Dash S, Majumder A, Jadhav S, Sen S: Matrix elasticity regulates mesenchymal stem cell chemotaxis. J Cell Sci 2018, 131. [DOI] [PubMed] [Google Scholar]
- 31.Alon R, Shulman Z: Chemokine triggered integrin activation and actin remodeling events guiding lymphocyte migration across vascular barriers. Exp Cell Res 2011, 317:632–641. [DOI] [PubMed] [Google Scholar]
- 32.Szabó A, Mayor R: Mechanisms of Neural Crest Migration. Annu Rev Genet 2018, 52:43–63. [DOI] [PubMed] [Google Scholar]
- 33.Smith A, Robinson V, Patel K, Wilkinson DG: The EphA4 and EphB1 receptor tyrosine kinases and ephrin-B2 ligand regulate targeted migration of branchial neural crest cells. Current Biology 1997, 7:561–570. [DOI] [PubMed] [Google Scholar]
- 34.Gammill LS, Gonzalez C, Bronner-Fraser M: Neuropilin 2/semaphorin 3F signaling guides peripheral nervous system segmentation. Developmental Biology 2006, 295:366. [Google Scholar]
- 35.Shiau CE, Lwigale PY, Das RM, Wilson SA, Bronner-Fraser M: Robo2-Slit1 dependent cell-cell interactions mediate assembly of the trigeminal ganglion. Nat Neurosci 2008, 11:269–276. [DOI] [PubMed] [Google Scholar]
- 36.Szabó A, Theveneau E, Turan M, Mayor R: Neural crest streaming as an emergent property of tissue interactions during morphogenesis. PLoS Comput Biol 2019, 15:e1007002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shellard A, Szabó A, Trepat X, Mayor R: Supracellular contraction at the rear of neural crest cell groups drives collective chemotaxis. Science 2018, 362:339–343. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Using a combination of in vivo live imaging, laser ablation techniques, and in silico modeling, the researchers demonstrate that in collective migration in Xenopus and zebrafish cranial neural crest cells is driven by a contractile actomyosin ring at the rear of the migrating cohort, which is excluded from the front cells in response to signaling by a gradient of SDF1. Local contraction of the ring at the rear of the collective drives cell intercalation, which propagates a wave of cell movement from the rear to the front.
- 38.Montell DJ, Yoon WH, Starz-Gaiano M: Group choreography: mechanisms orchestrating the collective movement of border cells. Nat Rev Mol Cell Biol 2012, 13:631–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bianco A, Poukkula M, Cliffe A, Mathieu J, Luque CM, Fulga TA, Rørth P: Two distinct modes of guidance signalling during collective migration of border cells. Nature 2007, 448:362–365. [DOI] [PubMed] [Google Scholar]
- 40.Veeman MT, McDonald JA: Dynamics of cell polarity in tissue morphogenesis: a comparative view from Drosophila and Ciona. F1000Research 2016, 5:1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Inaki M, Vishnu S, Cliffe A, Rørth P: Effective guidance of collective migration based on differences in cell states. Proc Natl Acad Sci U S A 2012, 109:2027–2032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fernández-Espartero CH, Ramel D, Farago M, Malartre M, Luque CM, Limanovich S, Katzav S, Emery G, Martín-Bermudo MD: GTP exchange factor Vav regulates guided cell migration by coupling guidance receptor signalling to local Rac activation. J Cell Sci 2013, 126:2285–2293. [DOI] [PubMed] [Google Scholar]
- 43.Wang X, He L, Wu YI, Hahn KM, Montell DJ: Light-mediated activation reveals a key role for Rac in collective guidance of cell movement in vivo. Nat Cell Biol 2010, 12:591–597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ramel D, Wang X, Laflamme C, Montell DJ, Emery G: Rab11 regulates cell-cell communication during collective cell movements. Nat Cell Biol 2013, 15:317–324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chang Y-C, Wu J-W, Hsieh Y-C, Huang T-H, Liao Z-M, Huang Y-S, Mondo JA, Montell D, Jang AC-C: Rap1 Negatively Regulates the Hippo Pathway to Polarize Directional Protrusions in Collective Cell Migration. Cell Rep 2018, 22:2160–2175. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• This manuscript describes an important role for Rap1 in supporting leader cell choice in Drosophila border cells. High Rap1 activity negatively regulates the Hippo pathway, which is responsible for suppressing the activity of the actin elongation factor Enabled. Leader cells have the highest Rap1 activity, allowing Ena to support protrusion formation in concert with Rac1 primarily in the front.
- 46.Lucas EP, Khanal I, Gaspar P, Fletcher GC, Polesello C, Tapon N, Thompson BJ: The Hippo pathway polarizes the actin cytoskeleton during collective migration of Drosophila border cells. J Cell Biol 2013, 201:875–885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lafuente EM, Puijenbroek AAFL, Krause M, Carman CV, Freeman GJ, Berezovskaya A, Constantine E, Springer TA, Gertler FB, Boussiotis VA: RIAM, an Ena/VASP and Profilin ligand, interacts with Rap1-GTP and mediates Rap1-induced adhesion. Dev Cell 2004, 7:585–595. [DOI] [PubMed] [Google Scholar]
- 48.Sawant K, Chen Y, Kotian N, Preuss KM, McDonald JA: Rap1 GTPase promotes coordinated collective cell migration in vivo. Mol Biol Cell 2018, 29:2656–2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gline S, Kaplan N, Bernadskaya Y, Abdu Y, Christiaen L: Surrounding tissues canalize motile cardiopharyngeal progenitors towards collective polarity and directed migration. Development 2015, 142:544–554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bernadskaya YY, Brahmbhatt S, Gline SE, Wang W, Christiaen L: Discoidin-domain receptor coordinates cell-matrix adhesion and collective polarity in migratory cardiopharyngeal progenitors. Nat Commun 2019, 10:57. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• In this manuscript, the authors use a combination of transgenesis, immunohistochemistry, live imaging, and cell tracking of Ciona trunk ventral cells (TVCs) to demonstrate how Discoidin-domain receptor (Ddr) has two different functions to promote collective directional movement: one function involves mediating integrin-based migration along the underlying ECM in response to activation by endoderm-derived type IX collagen Col9-a1, and the other is promoting polarized BMP-Smad signaling to establish leader and follower cell states via a mechanism independent of the integrin-ECM function.
- 51.Kadam S, Ghosh S, Stathopoulos A: Synchronous and symmetric migration of Drosophila caudal visceral mesoderm cells requires dual input by two FGF ligands. Development 2012, 139:699–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Reim I, Hollfelder D, Ismat A, Frasch M: The FGF8-related signals Pyramus and Thisbe promote pathfinding, substrate adhesion, and survival of migrating longitudinal gut muscle founder cells. Dev Biol 2012, 368:28–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Macabenta F, Stathopoulos A: Migrating cells control morphogenesis of substratum serving as track to promote directional movement of the collective. Development 2019, doi: 10.1242/dev.177295. [DOI] [PMC free article] [PubMed] [Google Scholar]; • This study describes how intersecting functions of FGF signaling in Drosophila trunk visceral mesoderm (TVM) and caudal visceral mesoderm (CVM) support gut muscle morphogenesis beyond a chemoattractive model. While FGF both supports integrin expression and confers directionality in migration, the earlier role supporting integrin is critical to ensuring stability of the TVM substratum, while the role in supporting directional migration is necessary for symmetric movement of the CVM. Ablating both functions uncovers a previously uncharacterized ability of the CVM to influence the morphogenesis of the TVM substrate.