Abstract
Purpose
Long noncoding RNA 00703 (LINC00703) was found originating from a region downstream of Kruppel-like factor 6 (KLF6) gene, having 2 binding sites for miR-181a. Since KLF6 has been reported as a target of miR-181a in gastric cancer (GC), this study aims to investigate whether LINC00703 regulates the miR-181a/KLF6 axis and plays a functional role in GC pathogenesis.
Materials and Methods
GC tissues, cell lines, and nude mice were included in this study. RNA binding protein immunoprecipitation (RIP) and pull-down assays were used to evaluate interaction between LINC00703 and miR-181a. Quantitative real-time polymerase chain reaction and western blot were applied for analysis of gene expression at the transcriptional and protein levels. A nude xenograft mouse model was used to determine LINC00703 function in vivo.
Results
We revealed that LINC00703 competitively interacts with miR-181a to regulate KLF6. Overexpression of LINC00703 inhibited cell proliferation, migration/invasion, but promoted apoptosis in vitro, and arrested tumor growth in vivo. LINC00703 expression was found to be decreased in GC tissues, which was positively correlated with KLF6, but negatively with the miR-181a levels.
Conclusions
LINC00703 may have an anti-cancer function via modulation of the miR-181a/KLF6 axis. This study also provides a new potential diagnostic marker and therapeutic target for GC treatment.
Keywords: LincRNAs, KLF6, miRNA sponge, Gastric cancer
INTRODUCTION
Gastric cancer (GC), which is often diagnosed at an advanced stage accompanied by excessive proliferation and metastasis, ranks the fourth among the most commonly diagnosed cancers and is among the leading causes of cancer-related deaths worldwide [1]. Despite of great progress in chemotherapy, radiotherapy, and surgical techniques, the prognosis of GC patients remains poor [2,3]. A large number of studies have shown that various oncogenes or tumor suppressors play important roles in GC tumorigenesis. However, almost no well-accepted biomarkers have been established to facilitate the comprehensive management of GC patients so far [3]. Therefore, the discovery of novel molecular targets and understanding the underlying regulatory mechanisms in gastric carcinogenesis is of a great scientific and therapeutic interest.
Recently, long noncoding RNAs (LincRNAs) have emerged as crucial regulators in the pathology of various tumorigenesis [4,5]. Thus far, LincRNAs have been reported to modulate gene expression through gene imprinting, dosage compensation, and control of transcription or post-transcriptional processing [5,6,7]. Besides, a hypothesis of competing endogenous RNAs (ceRNAs) was proposed to describe a novel regulatory mechanism of LincRNAs [8,9], which assumes that LincRNAs and mRNAs can associate with each other by competing for common miRNA response elements [7,9]. For instance, Liu et al. [10] reported that LincRNA HOTAIR acts as a ceRNA by sponging miR-331-3p to regulate HER2 expression, thereby affecting proliferation, migration and invasion of gastric carcinoma cells. Further, LincRNA HOTAIR can also function as a sponge for miR-152 or miR-126 to modulate HLA-G or PI3K/AKT/MRP genes, respectively [11,12]. Similarly, it was found that LincRNA MEG3 can upregulate Bcl-2 via its ceRNA activity on miR-181a, and thus modulate cell proliferation, migration, invasion and apoptosis in GC [13].
The Kruppel-like factor (KLF) gene families have been shown to play essential roles in varieties of cellular processes and diseases [14]. One member of this family, KLF6, was reported to act as a tumor suppressor gene in the pathogenesis of GC [15,16]. Recent work has shown that KLF6 is a target gene of miR-181a in various diseases including GC [17,18,19,19]. By using bioinformatic analysis, we found a putative LincRNA LINC00703 [20], which is located approximately 600kb downstream of the KLF6 gene and has 2 binding sites for miR-181a. Thus, we hypothesized that LINC00703 might regulate the expression of KLF6 by competitively interacting with miR-181a. To verify this hypothesis, we utilized clinical GC tissues, cell lines, and nude mice in the present study. As a consequence, we demonstrate herein that LINC00703 acts as a ceRNA to regulate KLF6 expression by sponging miR-181a, thereby playing a functional role in the modulation of cell function and tumor growth both in vitro and in vivo. Our work suggested that the regulatory axis of LINC00703/miR-181a/KLF6 may provide novel diagnostic and therapeutic hints for effective therapy of GC.
MATERIALS AND METHODS
GC tissue preparation and cell culture
A total of 15 specimens of gastric adenocarcinoma and the adjacent noncancerous tissues (10 cm away from the tumor and harboring no cancer cells detected by histology) were collected by either surgical resection or endoscopy biopsy, at Tongji University Affiliated East Hospital. All the tissue samples were obtained with the informed consent of the patients, and the procedure was approved by the Medical Ethics Committee of Tongji University Affiliated East Hospital.
Human normal gastric epithelial cell line, GES-1, was purchased from Mingzhou Biotech (Ningbo, China). According to the provider's information, the GES-1 was established from normal gastric epithelial cells infected with the SV40 virus. This stable cell line was not cross contaminated, as characterized by polymorphic short tandem repeat (STR) authentication. The HEK 293T cells and human gastric adenocarcinoma cell lines, including SGC-7901, MGC-803, and BGC-823 were obtained from the Cell Bank of Chinese Academy of Sciences. All the cells were routinely cultured in RPMI1640 or DMEM medium supplemented with 10% fetal bovine serum (FBS) at 37°C in a humidified atmosphere with 5% CO2. The testing for mycoplasma was performed routinely, and no positive results were observed during the described studies.
Vectors construction and luciferase reporter activity assay
The full length of LINC00703 was amplified using cDNA as a template and then subcloned into the mammalian expression vector pcDNA3.1 (Invitrogen). The forward primers contained BamH1 restriction enzyme site: 5′-CGCGGATCCAATG TTTTTGCTCATTAATTC-3′; and reverse primers included XbaI restriction enzyme site: 5′-GCT CTAGACGACAAATTTTGTTGAATTTA-3′. To construct luciferase reporter vectors, the 3′-untranslated region (UTR) of KLF6 was amplified and cloned into psiCHECK2 luciferase vector (Ambion, Inc., Austin, TX, USA). For that, forward primers contained XhoI restriction enzyme site: 5′-CCGCTCGAGGGGAGCAGAGAGGTGGATCCT-3′ and reverse primers had NotI restriction enzyme site: 5′-TAGAGCGGCCGCTGGCAG TGATGTCATCTTTTATTT-3′. All the obtained constructs were verified by DNA sequencing. The luciferase activities were detected as described previously [21,22,23]. Chemically synthesized miR-181a mimic and the corresponding negative control (NC) were provided by Ruibobio (Guangzhou, China).
RNA binding protein immunoprecipitation (RIP) and pull-down assays
RIP assay was performed using the EZMagna RIP kit (Millipore, Billerica, MA, USA), following the manufacturer's protocol. The experimental procedure included cell lysis, magnetic beads preparation, immunoprecipitation, RNA purification and quantitative real-time polymerase chain reaction (qRT-PCR) analysis. Human anti-Ago2 antibody (Millipore) was used to enrich miR-181a and LINC00703, and normal mouse immunoglobulin G (IgG; Millipore) was used as a negative control.
For RNA pull-down assay, GC cells were transfected with biotinylated miR-181a (containing wild-type [WT] or mutated [Mut] sequences). After 48 hours, the cells were lysed in specific buffer (Ambion, Inc.) and then incubated with M-280 streptavidin beads (S3762, Sigma-Aldrich, St Louis, MO, USA), pre-coated with Rnase-free bovine serum albumin (BSA). After incubation for 3h at 4°C, the beads were rinsed 2 times with precooled lysis buffer, 3 times with low salt buffer and 1 time with high salt buffer. Combined RNA was purified for qRT-PCR analysis.
Cell proliferation and migration/invasion assays
After 24 hours of transfection, cells were trypsinized and seeded into 96-well culture plate at a density of 10,000 cells/well in growth medium supplemented with 10% serum. The cell proliferation was measured at different time points (24, 48, and 72 hours) using the 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) kit (Beyotime Biotechnology, Nanjing, China), following the manufacturer's protocol. Cell migration was determined using 8 μm pore size 96-well minimum inhibitory concentration (MIC) Transwell plates (Millipore), via measuring the number of migratory cells under a microscope in 5 fields (100×), as previously described [21]. For the invasion assay, the MIC plates were initially coated with matrigel (BD Biosciences, Bedford, MA, USA), diluted in serum free medium and followed the same procedures as migration assay.
Flow cytometry analysis of apoptosis
Twenty-four hours after transfection, the cells were collected and washed with PBS twice, and about 1-5×105 cells were resuspended in 400 μL Annexin V binding buffer (BD Biosciences, San Jose, CA, USA). The cells were stained with Annexin V-FITC (BD-Biosciences) for 15 minutes at 4°C in the dark. The reaction was stopped by the addition of propidium iodide (PI) and incubation for 5 minutes at 4°C in the dark. The samples were then subjected to flow cytometry analysis.
qRT-PCR
Total RNA from cell and tissue samples was extracted and quantified using the NanoDrop (Thermo Fisher Scientific, Rockford, IL, USA). For mRNA analysis, the first-strand cDNA was synthesized using reverse transcription kit according to the manufacturer's instruction. qRT-PCR analysis was performed on Step-One plus real-time PCR system (Applied Biosystems) using SYBR green-I Master PCR Mix with gene specific primers (Table 1). The expression level of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene was used as a reference. The PCR was performed at 95°C for 10 minutes, followed by 40 cycles of 95°C for 10 seconds and 60°C for 1 minutes. The miRNA expression was analyzed as described previously [21,22,23]. The expression level of small nucleolar RNA U44 was used for normalization. Finally, the relative expression was calculated using the 2−△△Ct method.
Table 1. Primer sequences used for qRT-PCR in this work.
| Gene name | Primer sequences (5′–3′) |
|---|---|
| KLF6 | Forward: AGTTAACCAGGCACTTCCG |
| Reverse: CTTTTAGCCTACAGGATCCACC | |
| LINC00703 | Forward: CTTAAAGGCCCCACGCTTCT |
| Reverse: GAGGTGGAGGTGTAGACTCG | |
| GAPDH | Forward: CATGAGAAGTATGACAACAGCCT |
| Reverse: AGTCCTTCCACGATACCAAAGT |
qRT-PCR = quantitative real-time polymerase chain reaction; KLF = Kruppel-like factor; LINC00703 = long noncoding RNA 00703; GAPDH = glyceraldehyde 3-phosphate dehydrogenase.
Western blot analysis
Total protein was extracted using radioimmunoprecipitation assay buffer and the protein concentration was measured by bicinchoninic acid method. Equal amounts of proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes (Millipore). The membranes were blocked with 5% non-fat milk powder at 20°C –25°C for 2 hours, and then incubated with a specific primary antibody at 4°C overnight. After the incubation with horseradish peroxidase conjugated secondary antibody for 1h at 20°C –25°C, the immune-blotting signals were visualized using an ECL kit (West-Pico, Super Signal; Pierce, Rockford, IL, USA).
Tumor formation assay in nude mice
Five-week-old female athymic BALB/c mice were purchased from the Beijing Vital River Laboratory (Beijing, China). All animal procedures were performed in accordance to the protocols approved by the Animal Care and Use Committees at the Shenzhen Institutes of Advanced Technology, Chinese Academy of Science. For xenograft models, 5×106 BGC-823 cells, transfected with pcDNA3.1 or LINC00703 overexpression vectors, were injected subcutaneously in the right flank of the mice (8 mice per group). Tumor volumes were examined every 3 days when the implantations were starting to grow bigger. Tumor volumes were calculated by using the equation V (mm3) =A×B2/2, where A is the largest diameter, and B is the perpendicular diameter. After 12 days, these mice were sacrificed to collect the tumor tissues for qRT-PCR analysis.
Statistical analysis
Data were expressed as the mean±standard deviation (SD) from at least three independent experiments, which included 3 replicate holes and 8 nude mice in each group of cell and animal experiments, respectively. Student's t-test (2-tailed), One-way analysis of variance and Mann-Whitney test were performed to analyze the in vitro and in vivo data. The expression difference between GC and matched normal tissues were analyzed by paired sample t-test. All the statistical analysis was performed using SPSS 15.0 software (SPSS Inc, Chicago, IL, USA). The P-values less than 0.05 were considered statistically significant.
RESULTS
LINC00703 interacts with miR-181a to regulate KLF6 expression
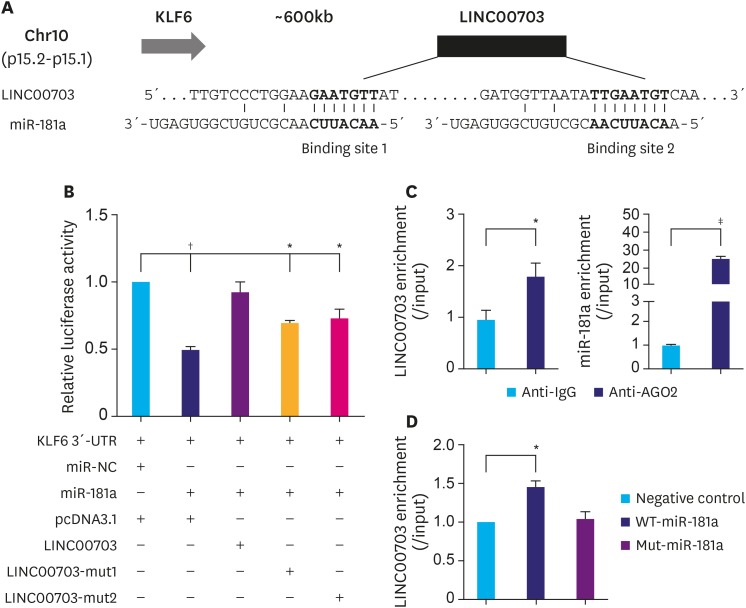
By searching in University of California Santa (UCSC) database (http://genome.ucsc.edu/), a putative LincRNA LINC00703 [20] was found located around 600 kb downstream of KLF6 gene at chromosome 10 (Fig. 1A). Further bioinformatics analysis (miRDB program and starBase v2.0) indicated that the transcript of LINC00703 has two binding sites of miR-181a (Fig. 1A). Since many studies report that KLF6 acts as tumor suppressor in GC and is a target of miR-181a [15,16,17,18,24], we thus hypothesized that LINC00703 may regulate the expression of KLF6 via binding to miR-181a. To validate our hypothesis, we first performed luciferase assays to evaluate whether LINC00703 can directly interact with miR-181a and affect the 3′-UTR reporter activity of KLF6. As shown in Fig. 1B, transfection of miR-181a significantly decreased the KLF6 luciferase activity, which, in turn, was recovered by the co-transfection of pcDNA3.1-based LINC00703 vector. Moreover, the luciferase activity was partially restored by the mutation of each binding site of miR-181a in LINC00703 (Fig. 1B). To further confirm the interaction between LINC00703 and miR-181a, RIP and RNA pulldown assays were performed. The results of RIP showed that both LINC00703 and miRNA-181a were enriched in Ago2-containing miRNAs, compared to control IgG immunoprecipitates (Fig. 1C). Moreover, RNA pulldown assay revealed that LINC00703 was co-precipitated with WT-miR-181a rather than Mut-miR-181a (Fig. 1D).
Fig. 1. LINC00703 interacts with miR-181a. (A) A schematic representation of LINC00703 location at chromosome 10 and 2 putative binding sites for miR-181a in LINC00703. (B) The luciferase reporter plasmid (KLF6 3′-UTR) was co-transfected with miR-181a and LINC00703-overexpressing plasmids into HEK-293T cells. The relative luciferase activity was measured 48h after transfection. (C) RIP with human monoclonal anti-Ago2, mouse preimmune IgG or 10% input from BGC-823 cell extracts. The qRT-PCR was performed to determine the RNA levels of miR-181a and LINC00703 in immunoprecipitates as compared to the input. (D) The expression of LINC00703 enriched by RNA pull-down assay. All data are presented as means±standard deviation (n=3, biological replicates).
LINC00703 = long noncoding RNA 00703; KLF = Kruppel-like factor; UTR = untranslated region; RIP = RNA binding protein immunoprecipitation; IgG = immunoglobulin G; qRT-PCR = quantitative real-time polymerase chain reaction.
*P<0.05, †P<0.01, and ‡P<0.001.
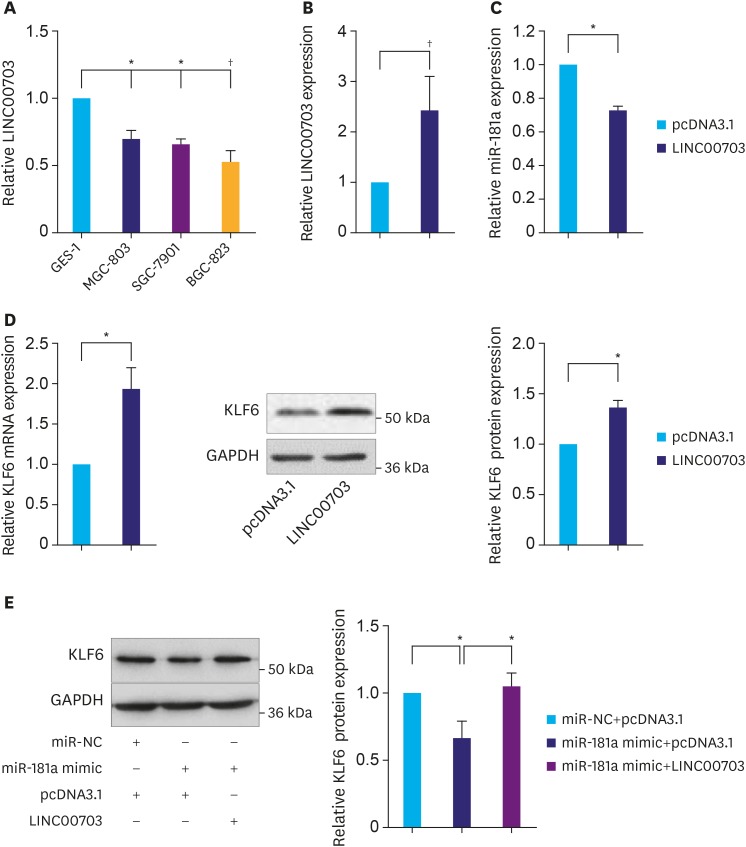
Next, we asked whether LINC00703 regulates KLF6 expression. Firstly, the expression of LINC00703 was analyzed in several common GC cell lines including MGC-803, SGC-7901 and BGC-823. As compared with GES-1 (a normal gastric mucosal epithelial cell line), the level of LINC00703 was significantly reduced in these cell lines, particularly in BGC-823 (Fig. 2A), which was used for the further investigations unless otherwise stated. Then, we transfected BGC-823 cells with pcDNA-LINC00703 vectors to achieve overexpression of LINC00703 (Fig. 2B). As a result, we observed a significant decrease of miR-181a level (Fig. 2C), but a significant increase of KLF6 expression at both mRNA and protein levels (Fig. 2D). Moreover, overexpression of LINC00703 partially rescued the effects of miR-181a mimic on KLF6 expression (Fig. 2E). Taken together, these data demonstrate that LINC00703 can directly bind to miR-181a to regulate the expression of KLF6 in GC cell.
Fig. 2. LINC00703 regulates KLF6 expression. (A) The qRT-PCR analysis, demonstrating LINC00703 expression in several gastric cancer cell lines (MGC-803, SGC-7901, and BGC-823), compared with normal gastric epithelium cell line (GES-1). (B) LINC00703 expression analysis in BGC-823 cells, transfected with pcDNA3.1 or LINC00703-overexpressing vector (LINC00703). (C and D) Expression levels of miR-181a and KLF6 after 48 hours, following the transfection of pcDNA3.1 or LINC00703 in BGC-823 cells. (E) The KLF6 protein level in BGC-823 cells, transfected with miR-181a mimic, LINC00703 or both miR-181a mimic and LINC00703. All data are presented as means±standard deviation (n=5, biological replicates).
LINC00703 = long noncoding RNA 00703; KLF = Kruppel-like factor; qRT-PCR = quantitative real-time polymerase chain reaction.
*P<0.05 and †P<0.01.
LINC00703 regulates proliferation, apoptosis and migration/invasion of GC cells
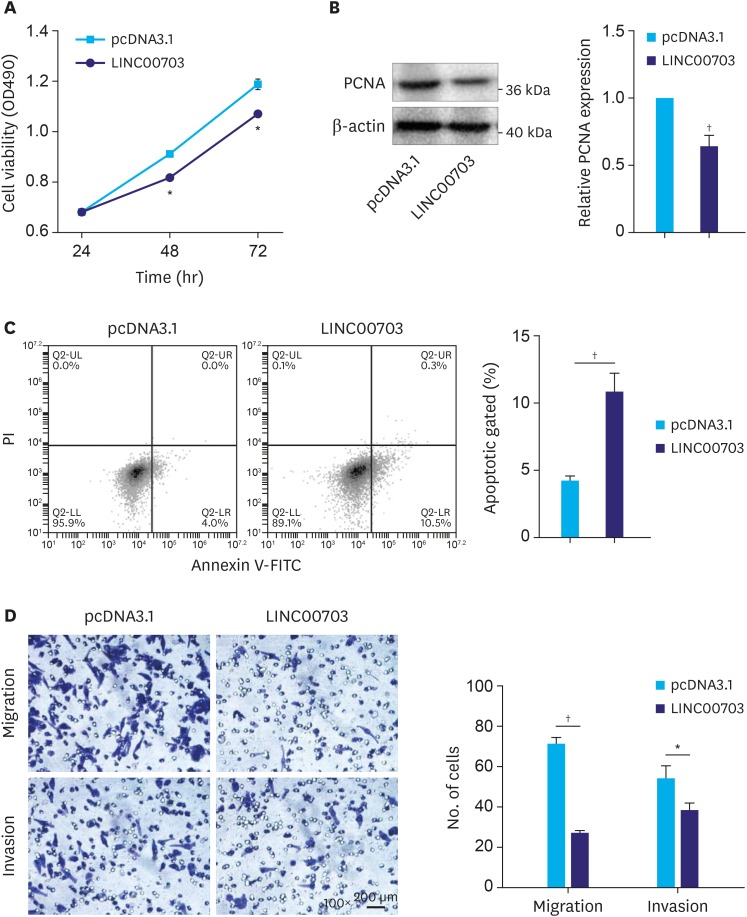
The above findings prompted us to explore the possible biological function of LINC00703. The results of MTT assay and proliferating cell nuclear antigen (PCNA) protein expression revealed that cell proliferation was significantly decreased in the cells, transfected with LINC00703 vector, as compared with controls (Fig. 3A and B). Notably, the flow-cytometric analysis showed that the fraction of early apoptotic cells was significantly increased in the cells, overexpressing LINC00703 (Fig. 3C). Moreover, the processes of cell migration and invasion were significantly reduced in BGC-823 cells, transfected with LINC00703 vector, as it was shown in transwell assays (Fig. 3D). Additionally, we found that transfection of miR-181a mimic can partially rescue the cellular function of LINC00703 (Supplementary Fig. 1). Thus, these results indicate that LINC00703 plays an inhibitory role in the regulation of cell proliferation, apoptosis, migration and invasion of GC cells.
Fig. 3. Functional analysis of LINC00703. BGC-823 cells were transfected with pcDNA3.1 or LINC00703 overexpressing vector (LINC00703). (A and B) Measurements of cell proliferation using MTT assay (A) and PCNA protein level (B). (C) Flow cytometry analysis of cell apoptotic rates. (D) Results of transwell assays reflecting the changes in cell migration and invasiveness (100×). All data are presented as means±standard deviation (n=3, biological replicates).
LINC00703 = long noncoding RNA 00703; MTT = 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; PCNA = proliferating cell nuclear antigen.
*P<0.05 and †P<0.01.
LINC00703 attenuates tumor growth in nude mice
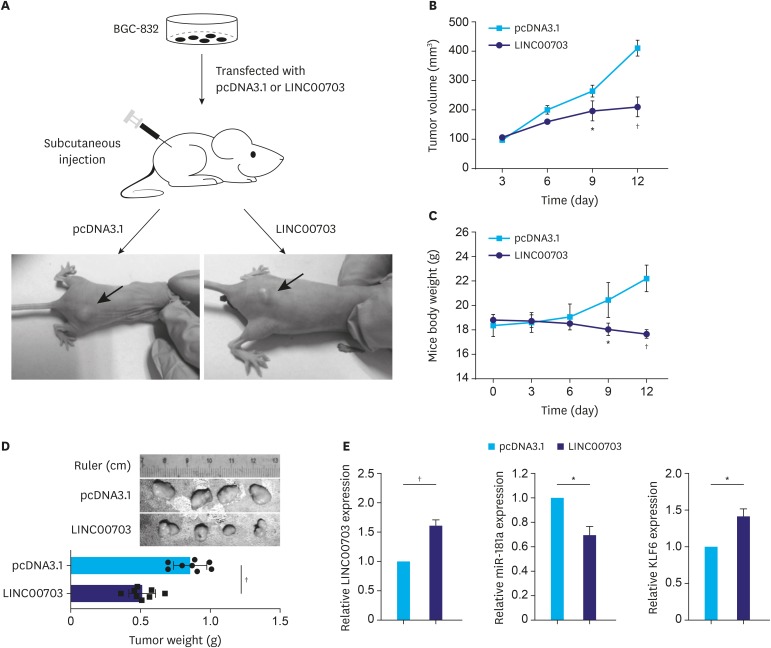
To explore whether LINC00703 influences GC tumorigenesis in vivo, BGC-823 cells transfected with pcDNA3.1 or LINC00703 vectors were injected in nude mice to induce subcutaneous tumors (Fig. 4A). Compared with the control mice, a significant decrease in tumor volume and mice weight were observed in mice overexpressing LINC00703 from day 9 (Fig. 4B and C). At the end of this experiment, the tumor tissues were harvested and weighed (Fig. 4D). Furthermore, the qRT-PCR analysis revealed increased expression of LINC00703 and KLF6, and decreased level of miR-181a in LINC00703 expressing tumor tissues (Fig. 4E).
Fig. 4. Effect of LINC00703 on tumor growth in vivo. (A) Tumors were subcutaneously induced in nude mice. BGC-832 cells that were stably transfected with pcDNA3.1 or LINC00703 overexpressing vectors (LINC00703) were injected into the flanks of nude mice for 12 days. (B and C) Dynamics of tumor volume (A) and mice body weight (B) according to the measurements every 3 days. (D) The tumors were dissected and weighed at the end of experiment. (E) qRT-PCR analysis of the expression of LINC00703, miR-181a and KLF6 in resected tumor tissues from the nude mice. All data are presented as means±standard deviation (n=8, for each group).
LINC00703 = long noncoding RNA 00703; qRT-PCR = quantitative real-time polymerase chain reaction; KLF = Kruppel-like factor.
*P<0.05 and †P<0.01.
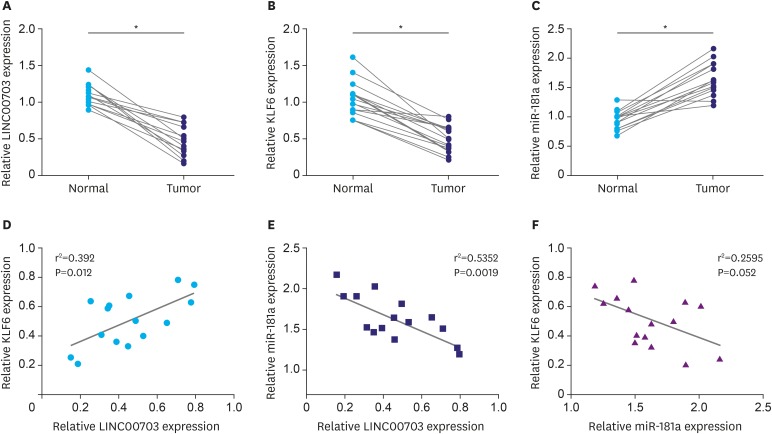
Correlation of LINC00703, KLF6 and miR-181a expression in GC tissues
To correlate LINC00703, KLF6 and miR-181a in clinical samples, we investigated their expression in 15 GC tissues and adjacent, histologically normal, tissues. It was found that LINC00703 and KLF6 levels were significantly decreased (Fig. 5A and B), while the miR-181a level was increased in cancerous tissues (Fig. 5C). Pearson correlation analysis showed that LINC00703 correlated positively with KLF6, but negatively with miR-181a at transcript level in GC tissues, compared with normal counterparts (Fig. 5D and E). Similar to previous results [19], miR-181a was negatively correlated with KLF6 (Fig. 5F).
Fig. 5. LINC00703 correlates positively with KLF6 but negatively with miR-181a. qRT-PCR analysis of LINC00703 (A), KLF6 (B), and miR-181a (C) in 15 GC tissues and their adjacent normal tissue. Pearson correlation analysis was performed between LINC00703 and KLF6 (D), LINC00703 and miR-181a (E), miR-181a and KLF6 (F). All data are presented as means±standard deviation.
LINC00703 = long noncoding RNA 00703; KLF = Kruppel-like factor; qRT-PCR = quantitative real-time polymerase chain reaction.
*P<0.01.
DISCUSSION
LincRNAs, with limited protein-coding capacity, are often expressed in a disease-, tissue-, or developmental stage-specific manner. Due to their vital functions in developmental processes and disease pathogenesis, LincRNAs have been regarded as attractive therapeutic targets [7]. Recently, an increasing number of studies have revealed that LincRNAs can compete with miRNAs, thereby affecting the expression of target genes [25,26,27,28]. However, the exact role of LincRNA and miRNA interactions in the GC tumorigenesis remains largely unknown. Therefore, understanding their interaction would facilitate the development of LincRNA/miRNA based diagnostics and therapeutics against cancers.
In this study, we found a novel LincRNA LINC00703 located in the downstream region of the KLF6 gene (Fig. 1A), suggesting a possible cis-regulatory mechanism between LINC00703 and KLF6 [29,30]. Further bioinformatics analysis showed that the LINC00703 transcript has four binding sites for miR-181a (Fig. 1A), which acts as a GC oncogene through targeting KLF6, as previously reported [19]. Inspired by the ‘competitive endogenous RNA regulatory network [7,9,31], we hypothesized that LINC00703 may serve as a ceRNA to regulate KLF6 through interaction with miR-181a. In support of this assumption, we found that LINC00703 indeed induces translational repression of a KLF6 3′-UTR luciferase reporter gene (Fig. 1B). In addition, RIP and RNA pulldown assays demonstrated a direct physical interaction between LINC00703 and miR-181a in GC cells (Fig. 1C and D), providing further evidence for LINC00703-mediated sequestering activity of miR-181a. Next, our results showed that the LINC00703 level was significantly reduced in the GC cell line, while overexpression of LINC00703 decreased miR-181a but increased KLF6 expression (Fig. 2). Moreover, LINC00703 could partially restore downregulation of the KLF6 expression level, caused by miR-181a (Fig. 2E). Based on these novel results of LINC00703 interaction with miR-181a, we propose that LINC00703 can regulate KLF6 by modulating miR-181a in GC cells.
Several studies show that LincRNA plays a crucial role in the regulation of cell function. For instance, LincRNA SNHG5 regulates GC cell proliferation and migration through targeting KLF4 [25]. By modulation of the miR-507-FOXM1 axis, LincRNA UCA1 is involved in cell proliferation, invasion, and G0/G1 cell cycle arrest in melanoma [32]. Similarly, LincRNA MEG3 upregulates Bcl-2 via its ceRNA activity on miR-181a, regulating cell proliferation, migration, invasion, and apoptosis in GC [13]. In this work, we demonstrated that overexpression of LINC00703 inhibited cell proliferation, migration, and invasion of GC cells (Fig. 3). Furthermore, the function of LINC00706 could be partially rescued by miR-181a (Supplementary Fig. 1). Importantly, LINC00703 also suppressed tumor growth in nude mice (Fig. 4). Together, these data suggest that LINC00703 may serve as a tumor suppressor in GC development.
To act as an endogenous ‘sponge’, the abundance of LINC00703 should be inversely related to miR-181a. We showed that LINC00703 expression was decreased, whereas the level of miR-181a was increased in GC tissue, in comparison with adjacent normal gastric tissues, showing a negative correlation (Fig. 5). These results are in accordance with the findings that overexpression of LINC00703 inhibited cell invasion and cell viability, and induced growth arrest both in vitro and in vivo (Figs. 3 and 4). Moreover, the expression level of LINC00703 positively correlates with KLF6 (Fig. 5), which is also in line with the results of the alteration of KLF6 expression by LINC00703 (Figs. 2 and 4E). Nevertheless, the tumorigenic properties and mechanistic heterogeneity of LINC00703 and the regulatory axis via LINC00703/miR-181a/KLF6 deserve to be elucidated more extensively in a larger number of clinical specimens.
In conclusion, our research demonstrated a novel regulatory axis that incorporates LINC00703 acting as a ceRNA in the regulation of KLF6 expression by sponging miR-181a, which indicates a key role of LINC00703 in human gastric carcinogenesis. Moreover, our results provide novel evidence that the downregulation of LINC00703 may serve as a prognostic or progression marker for GC.
ACKNOWLEDGEMENT
We are grateful for Tongji University Affiliated East Hospital to provide the GC tissue samples.
Footnotes
Funding: This study was financially supported by Guangdong Basic and Applied Basic Research Foundation (2019A1515012200), Natural Science Foundation of Guangdong Province (2018A030310456) and Shenzhen Municipal Basic Research Program (JCYJ20170818164405101).
- Conceptualization: Q.Z.
- Data curation: Y.H., L.X., Q.Z.
- Formal analysis: Y.H., L.X., Q.Z.
- Funding acquisition: Q.Z., L.Y., L.X.
- Investigation: Y.H., P.M.
- Software: P.M., Y.H.
- Supervision: Z.R., Q.Z.
- Writing - original draft: Y.H., L.Y., Q.Z.
- Writing - review & editing: Q.Z.
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
SUPPLEMENTARY MATERIAL
LINC00703 function is partially rescued by miR-181a mimic. BGC-823 cells were transfected with pcDNA3.1 or LINC007031 overexpressed vector (LINC00703) or LINC00703 + miR-181a mimic. (A and B) MTT assay and PCNA protein level were measured to determine cell proliferation, and cleaved caspase3 was used as an apoptotic marker. (C) Transwell assays were performed to determine cell migration and invasion (100×). All data are presented as means±standard deviation (n=3, biological replicates).
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12:354–362. doi: 10.3748/wjg.v12.i3.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hartgrink HH, Jansen EP, van Grieken NC, van de Velde CJ. Gastric cancer. Lancet. 2009;374:477–490. doi: 10.1016/S0140-6736(09)60617-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Matsunaga N, Wakasaki T, Yasumatsu R, Kotake Y. Long noncoding RNA, ANRIL, regulates the proliferation of head and neck squamous cell carcinoma. Anticancer Res. 2019;39:4073–4077. doi: 10.21873/anticanres.13564. [DOI] [PubMed] [Google Scholar]
- 5.Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136:629–641. doi: 10.1016/j.cell.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 6.Arita T, Ichikawa D, Konishi H, Komatsu S, Shiozaki A, Shoda K, et al. Circulating long non-coding RNAs in plasma of patients with gastric cancer. Anticancer Res. 2013;33:3185–3193. [PubMed] [Google Scholar]
- 7.Wilusz JE, Sunwoo H, Spector DL. Long noncoding RNAs: functional surprises from the RNA world. Genes Dev. 2009;23:1494–1504. doi: 10.1101/gad.1800909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ergun S, Oztuzcu S. Oncocers: ceRNA-mediated cross-talk by sponging miRNAs in oncogenic pathways. Tumour Biol. 2015;36:3129–3136. doi: 10.1007/s13277-015-3346-x. [DOI] [PubMed] [Google Scholar]
- 9.Cesana M, Cacchiarelli D, Legnini I, Santini T, Sthandier O, Chinappi M, et al. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell. 2011;147:358–369. doi: 10.1016/j.cell.2011.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liu XH, Sun M, Nie FQ, Ge YB, Zhang EB, Yin DD, et al. Lnc RNA HOTAIR functions as a competing endogenous RNA to regulate HER2 expression by sponging miR-331-3p in gastric cancer. Mol Cancer. 2014;13:92. doi: 10.1186/1476-4598-13-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yan J, Dang Y, Liu S, Zhang Y, Zhang G. LncRNA HOTAIR promotes cisplatin resistance in gastric cancer by targeting miR-126 to activate the PI3K/AKT/MRP1 genes. Tumour Biol. 2016;37:16345–16355. doi: 10.1007/s13277-016-5448-5. [DOI] [PubMed] [Google Scholar]
- 12.Song B, Guan Z, Liu F, Sun D, Wang K, Qu H. Long non-coding RNA HOTAIR promotes HLA-G expression via inhibiting miR-152 in gastric cancer cells. Biochem Biophys Res Commun. 2015;464:807–813. doi: 10.1016/j.bbrc.2015.07.040. [DOI] [PubMed] [Google Scholar]
- 13.Peng W, Si S, Zhang Q, Li C, Zhao F, Wang F, et al. Long non-coding RNA MEG3 functions as a competing endogenous RNA to regulate gastric cancer progression. J Exp Clin Cancer Res. 2015;34:79. doi: 10.1186/s13046-015-0197-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McConnell BB, Yang VW. Mammalian Krüppel-like factors in health and diseases. Physiol Rev. 2010;90:1337–1381. doi: 10.1152/physrev.00058.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sangodkar J, Shi J, DiFeo A, Schwartz R, Bromberg R, Choudhri A, et al. Functional role of the KLF6 tumour suppressor gene in gastric cancer. Eur J Cancer. 2009;45:666–676. doi: 10.1016/j.ejca.2008.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cho YG, Kim CJ, Park CH, Yang YM, Kim SY, Nam SW, et al. Genetic alterations of the KLF6 gene in gastric cancer. Oncogene. 2005;24:4588–4590. doi: 10.1038/sj.onc.1208670. [DOI] [PubMed] [Google Scholar]
- 17.Bi J, Zeng X, Zhao L, Wei Q, Yu L, Wang X, et al. miR-181a induces macrophage polarized to M2 phenotype and promotes M2 macrophage-mediated tumor cell metastasis by targeting KLF6 and C/EBPα. Mol Ther Nucleic Acids. 2016;5:e368. doi: 10.1038/mtna.2016.71. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 18.Lei Z, Ma X, Li H, Zhang Y, Gao Y, Fan Y, et al. Up-regulation of miR-181a in clear cell renal cell carcinoma is associated with lower KLF6 expression, enhanced cell proliferation, accelerated cell cycle transition, and diminished apoptosis. Urol Oncol. 2018;36:93.e23–93.e37. doi: 10.1016/j.urolonc.2017.09.019. [DOI] [PubMed] [Google Scholar]
- 19.Zhang X, Nie Y, Du Y, Cao J, Shen B, Li Y. MicroRNA-181a promotes gastric cancer by negatively regulating tumor suppressor KLF6. Tumour Biol. 2012;33:1589–1597. doi: 10.1007/s13277-012-0414-3. [DOI] [PubMed] [Google Scholar]
- 20.Liu H, Li J, Koirala P, Ding X, Chen B, Wang Y, et al. Long non-coding RNAs as prognostic markers in human breast cancer. Oncotarget. 2016;7:20584–20596. doi: 10.18632/oncotarget.7828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Qian Z, Li Y, Yang H, Chen J, Li X, Gou D. PDGFBB promotes proliferation and migration via regulating miR-1181/STAT3 axis in human pulmonary arterial smooth muscle cells. Am J Physiol Lung Cell Mol Physiol. 2018;315:L965–L976. doi: 10.1152/ajplung.00224.2018. [DOI] [PubMed] [Google Scholar]
- 22.Li Y, Li L, Qian Z, Lin B, Chen J, Luo Y, et al. Phosphatidylinositol 3-kinase-DNA methyltransferase 1-miR-1281-histone deacetylase 4 regulatory axis mediates platelet-derived growth factor-induced proliferation and migration of pulmonary artery smooth muscle cells. J Am Heart Assoc. 2018;7:e007572. doi: 10.1161/JAHA.117.007572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Qian Z, Li Y, Chen J, Li X, Gou D. miR-4632 mediates PDGF-BB-induced proliferation and antiapoptosis of human pulmonary artery smooth muscle cells via targeting cJUN. Am J Physiol Cell Physiol. 2017;313:C380–C391. doi: 10.1152/ajpcell.00061.2017. [DOI] [PubMed] [Google Scholar]
- 24.Ma J, Yao Y, Wang P, Liu Y, Zhao L, Li Z, et al. MiR-181a regulates blood-tumor barrier permeability by targeting Krüppel-like factor 6. J Cereb Blood Flow Metab. 2014;34:1826–1836. doi: 10.1038/jcbfm.2014.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhao L, Han T, Li Y, Sun J, Zhang S, Liu Y, et al. The lncRNA SNHG5/miR-32 axis regulates gastric cancer cell proliferation and migration by targeting KLF4. FASEB J. 2017;31:893–903. doi: 10.1096/fj.201600994R. [DOI] [PubMed] [Google Scholar]
- 26.Zhao H, Peng R, Liu Q, Liu D, Du P, Yuan J, et al. The lncRNA H19 interacts with miR-140 to modulate glioma growth by targeting iASPP. Arch Biochem Biophys. 2016;610:1–7. doi: 10.1016/j.abb.2016.09.014. [DOI] [PubMed] [Google Scholar]
- 27.Wang SH, Wu XC, Zhang MD, Weng MZ, Zhou D, Quan ZW. Long noncoding RNA H19 contributes to gallbladder cancer cell proliferation by modulated miR-194-5p targeting AKT2. Tumour Biol. 2016;37:9721–9730. doi: 10.1007/s13277-016-4852-1. [DOI] [PubMed] [Google Scholar]
- 28.Sun L, Sun P, Zhou QY, Gao X, Han Q. Long noncoding RNA MALAT1 promotes uveal melanoma cell growth and invasion by silencing of miR-140. Am J Transl Res. 2016;8:3939–3946. [PMC free article] [PubMed] [Google Scholar]
- 29.Wang KC, Yang YW, Liu B, Sanyal A, Corces-Zimmerman R, Chen Y, et al. A long noncoding RNA maintains active chromatin to coordinate homeotic gene expression. Nature. 2011;472:120–124. doi: 10.1038/nature09819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Guil S, Esteller M. Cis-acting noncoding RNAs: friends and foes. Nat Struct Mol Biol. 2012;19:1068–1075. doi: 10.1038/nsmb.2428. [DOI] [PubMed] [Google Scholar]
- 31.Wang KC, Chang HY. Molecular mechanisms of long noncoding RNAs. Mol Cell. 2011;43:904–914. doi: 10.1016/j.molcel.2011.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wei Y, Sun Q, Zhao L, Wu J, Chen X, Wang Y, et al. LncRNA UCA1-miR-507-FOXM1 axis is involved in cell proliferation, invasion and G0/G1 cell cycle arrest in melanoma. Med Oncol. 2016;33:88. doi: 10.1007/s12032-016-0804-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
LINC00703 function is partially rescued by miR-181a mimic. BGC-823 cells were transfected with pcDNA3.1 or LINC007031 overexpressed vector (LINC00703) or LINC00703 + miR-181a mimic. (A and B) MTT assay and PCNA protein level were measured to determine cell proliferation, and cleaved caspase3 was used as an apoptotic marker. (C) Transwell assays were performed to determine cell migration and invasion (100×). All data are presented as means±standard deviation (n=3, biological replicates).