Abstract
Atherosclerosis is a chronic inflammatory disease of the vasculature characterised by the infiltration of activated neutrophils and macrophages at sites of damage within the vessel wall, which contributes to lesion formation and plaque progression. Selenomethionine (SeMet) is an organic form of selenium (Se), an essential trace element that functions in the regulation of the immune response by both bolstering the endogenous thioredoxin and glutathione antioxidant defence systems and by directly scavenging damaging oxidant species. This study evaluated the effect of dietary SeMet supplementation within a high fat diet fed apolipoprotein E deficient (ApoE−/-) mouse model of atherosclerosis. Dietary supplementation with SeMet (2 mg/kg) increased the tissue concentration of Se, and the expression and activity of glutathione peroxidase, compared to non-supplemented controls. Supplementation with SeMet significantly reduced atherosclerotic plaque formation in mouse aortae, resulted in a more stable lesion phenotype and improved vessel function. Concurrent with these results, SeMet supplementation decreased lesion accumulation of M1 inflammatory type macrophages, and decreased the extent of extracellular trap release from phorbol myristate acetate (PMA)-stimulated mouse bone marrow-derived cells. Importantly, these latter results were replicated within ex-vivo experiments on cultured neutrophils isolated from acute coronary syndrome patients, indicating the ability of SeMet to alter the acute inflammatory response within a clinically-relevant setting. Together, these data highlight the potential beneficial effect of SeMet supplementation as a therapeutic strategy for atherosclerosis.
Keywords: Atherosclerosis, Selenomethionine, Inflammation, Glutathione peroxidase
List of abbreviations
- ABAH
aminobenzoic acid hydrazide
- ACh
acetylcholine
- ACS
acute coronary syndrome
- αSMC
alpha smooth muscle cell
- ApoE−/
apolipoprotein E deficient
- ATP6
adenosine triphosphate synthase 6
- β-actin
beta actin
- CVD
cardiovascular disease
- CYTB
cytochrome B
- DAPI
4′,6-diamidino-2-phenylindole
- DTNB
5,5′-dithio-bis-[2-nitrobenzoic acid]
- Egr2
early growth response 2
- eNOS
endothelial nitric oxide synthase
- ET
extracellular trap
- GAPDH
glyceraldehyde 3-phosphate dehydrogenase
- GPX
glutathione peroxidase
- GPx1
glutathione peroxidase 1
- GSH
glutathione
- HFD
high-fat diet
- H2O2
hydrogen peroxide
- ICP-MS
inductively coupled mass spectrometry
- IL6
interleukin 6
- iNOS
inducible nitric oxide synthase
- MPO
myeloperoxidase
- NADPH
nicotinamide adenine dinucleotide phosphate
- NE
norepinephrine
- NETs
neutrophil extracellular traps
- NO
nitric oxide
- PMA
phorbol myristate acetate
- POSTN
periostin
- ROS
reactive oxygen species
- Se
selenium
- SeMet
selenomethionine
- SNP
sodium nitroprusside
- SOD2
superoxide dismutase 2
- TGFβ
transforming growth factor beta
- TMB
3,3′,5,5′-tetramethylbenzidine
- TNFα
tumour necrosis factor alpha
- TRX
thioredoxin
1. Introduction
Atherosclerosis is the major cause of cardiovascular disease and is characterised by the deposition of fatty material and cholesterol within the arterial wall [1,2]. Atherosclerosis is a chronic inflammatory disease that features the ongoing infiltration of a variety of immune cells, in particular neutrophils and macrophages, which play an essential role during lesion formation and plaque development [3,4], functioning to both eliminate cell debris and promote beneficial vessel structural remodelling [[5], [6], [7]]. As a part of their inflammatory repertoire, neutrophils release extracellular traps, aimed to confine infection, signal to and promote macrophage infiltration [3,8]. Activation of infiltrated neutrophils within vessel intima produces excess reactive oxidant species (ROS) and results in the release of the enzyme myeloperoxidase (MPO). This exacerbates oxidant generation and contributes to lesion development by accelerating the formation of lipid-filled foam cells, which increases overall oxidative stress [8,9]. Monocyte-derived macrophages, recruited to sites of damage within the vessel wall and atherosclerotic lesions, can further differentiate into either pro-inflammatory M1 macrophages or alternatively-activated M2 macrophages [3,[10], [11], [12]]. These two macrophage phenotypes contribute to atherosclerosis development in distinct ways. For example, inflammatory M1 macrophages increase oxidant and pro-inflammatory cytokine production, including tumour necrosis factor alpha (TNFα) [10,13] and are generally the dominant macrophage phenotype in unstable lesions [11,13]. In contrast, M2 macrophages inhibit inflammation within the atherosclerotic environment and promote plaque stabilisation by eliminating cell debris and promoting tissue fibrosis [14,15]. Unlike their inflammatory counterpart, M2 macrophages tend to predominate in small and stable plaque regions, suggesting an anti-atherogenic function for this alternatively-activated subset of macrophages [15]. How the balance between populations of M1 and M2 macrophages relates to atherosclerotic disease development is complex and remains unclear and complex.
Selenium (Se) is a trace element essential for human health, with evidence for an inverse correlation between Se status and CVD risk in a number of randomised trials, although there is some inconsistency in the data. Nonetheless, it is generally accepted that Se supplementation can be beneficial in situations where basal levels of Se are low (reviewed in Ref. [16]), with low levels of Se linked to the development of a variety of diseases [17]. This is due to the fact that many selenoproteins within the body are involved in endogenous antioxidant defence systems, including the glutathione peroxidase (GPX) and thioredoxin (TRX) families of enzymes [18,19], which detoxify oxidants particularly during prolonged conditions of oxidative stress. This is relevant to many disease settings [20], including chronic inflammatory diseases such as atherosclerosis [17]. Early in vivo studies utilising rabbit models demonstrated that Se supplementation could reduce lesion formation in atherosclerosis. This was attributed to the ability of Se to inhibit oxidative stress within the atherosclerotic plaque [21,22]. In addition, these studies also indicated that Se supplementation may inhibit plaque formation by modulating key events during atherosclerosis development, such as decreasing the underlying inflammatory response and endothelial dysfunction [23]. In support of these data, and highlighting a pivotal role of GPX within this setting, subsequent clinical studies have demonstrated that patients with low baseline GPX activity have a higher incidence of recurrent cardiovascular events, whilst pharmacological intervention to upregulate GPX activity can lower the risk of such events [24,25].
The metabolism of Se by the body is dependent on its particular chemical form, with inorganic and organic forms incorporating into proteins by different pathways and rates, which influences cellular function [26,27]. The majority of previous studies have relied on Se supplementation in the form of a diet containing inorganic Se, such as sodium selenite [21,22]. However, dietary supplementation with inorganic forms of Se can be problematic, because absorption is mainly through simple diffusion processes, which are influenced by the molarity within any given microenvironment. This results in a low transportation efficiency of these compounds, requiring high concentrations in order to achieve a beneficial outcome [27]. In contrast, organic forms of Se, such as selenomethionine (SeMet), are more readily taken up by cells, resulting in comparatively low dietary levels being required to bolster and enhance endogenous GPX activity [26,28].
In the present study, we investigate the efficacy of SeMet dietary supplementation against the development of atherosclerosis within an apolipoprotein E deficient (ApoE−/-) mouse model, examining the ability of this compound to enhance GPX expression and modulate the neutrophil and macrophage inflammatory responses.
2. Materials and methods
2.1. Animals
All studies were performed in compliance with protocols approved by the Sydney Local Health District Animal Welfare Committee (Protocol # 2016/035) on male ApoE−/- mice. Following weaning, mice were housed in groups of three to five and fed with normal rodent/chow diet (Specialty Feeds, WA, Australia) for 3 weeks. At the commencement of the long-term feeding study, mice were randomly separated into two cohorts for 12 weeks, a high-fat diet (HFD) (21% total fat, 0.15% cholesterol; Specialty Feeds, SF00-219) control group and a HFD group supplemented with SeMet (2 mg/kg, AK Scientific, M598). In a separate study, mice received a HFD for 6 weeks before being separated into either a HFD control group or a HFD group supplemented with SeMet (2 mg/kg) for 6 weeks. Given that mice are known to eat 4–5 g of food a day, the supplementation of 2 mg/kg (w/w) in feed used in this study equates to a dietary supplementation of SeMet of 8–10 μg/day. This is in agreeance with ranges of supplementation within previous animal studies using both selenium and SeMet [29], as well as within the current recommended dietary allowance of selenium for humans of 55 μg [30].
2.2. Sample collection
Mice were anesthetized by inhalation with 2% isoflurane in 1L/min of oxygen. Blood was drawn by direct puncture of the right ventricle, and was then placed into heparinised or EDTA coated tubes (BD Biosciences, North Ryde, NSW, Australia, 367839) as applicable. Plasma was separated from whole blood by centrifugation for 15 min at 2000g and immediately frozen at −80 °C for later analysis of cytokine and MPO levels. Liver, kidney and tissue from the aortic root were either fixed in 10% neutral buffered formalin (Sigma-Aldrich, St. Louis, Missouri, HT501128) then further processed for histological analysis or snap-frozen in liquid nitrogen and stored at −80 °C for subsequent SeMet analysis. Whole aorta sections were also snap-frozen in liquid nitrogen and stored at −80 °C for subsequent protein and gene expression analysis.
2.3. Quantification of SeMet by ICP-MS
Quantification of SeMet in plasma and tissue homogenates was performed after separation using a Dionex Ultimate 3000 UPLC system (Thermo Scientific) in combination with inductively coupled plasma mass spectrometry (ICP-MS). Plasma and tissue samples were transferred to Vivaspin® 500 μL centrifugal filter units (Sartorius AG, Göttingen, Germany, VS0191) with a semi-permeable membrane with molecular mass cut-off of 3 kDa and centrifuged at 14000g for 30 min. The filtrates were injected onto a C18 Gemini UPLC column (5 μm, 250 × 2 mm (Phenomenex, Værløse, Denmark), and separated with a mobile phase containing 200 mM ammonium acetate in 5% v/v methanol (pH 7), and flow rate of 200 μL min−1. The SeMet was quantified with an Agilent 8800 ICP-MS Triple Quad equipped with a 1.5 mm ID quartz injector torch and sampler and skimmer cones made of platinum (Agilent Technologies, Santa Clara, USA). Daily optimization was performed using a Se-standard solution (50 μg L−1 Se in mobile phase), and the analyses were carried out in MS/MS mode with oxygen as reaction gas (monitoring 77Se → 93SeO and 80Se → 96SeO).
2.4. Analysis of atherosclerosis lesions
Characterisation and extent of lesion formation was examined by two approaches. Firstly, whole cleaned and formalin fixed aortic sections were stained with Oil Red O solution (Sigma-Aldrich, O0625) using the enface technique described by Lin and colleagues [31]. Images of Oil red O stained aortic sections were captured with a digital camera attached to a Nikon SMZ800 Zooming Body Microscope and staining area measured using ImageJ software (National Institutes of Health, MD) [32]. Overall lesion burden was expressed as a percentage of red staining area relative to the entire surface area of the aortic section. Secondly, lesion size was assessed in aortic root cross-sections following H&E staining (Hematoxylin: Sigma-Aldrich, H3136; Eosin: Polysciences Inc, Warrington Township, PA, 17269) and presented as percentage of total vessel aortic root area. Lesion cross-sections from the aortic root were also assessed using immunohistochemical methods described by Menon & Fisher [33]. For identification of macrophages, sections were stained with the pan-macrophage marker F4/80 (Abcam, Cambridge, UK, ab6640), macrophage phenotype markers iNOS (Abcam, ab15323) for M1 and Egr2 (Abcam, ab43020) for M2. For identification of fibrosis sections were stained with antibodies targeting either α-smooth muscle actin (Abcam, ab5694) or periostin (Abcam, ab215199). Images were captured on an Axio Scan. Z1 slide scanner (Zeiss, Oberkochen, Germany) and quantified using either ImagePro (Media Cybernetics, Rockville, Maryland) or Zen (Zeiss) software.
2.5. Detection of peroxidase activity and antioxidant content
For total peroxidase activity in whole blood a bioluminescence assay was used as described by Gross et al. [34], with minor modifications. In brief, whole blood was freshly drawn into heparinised coated collection tubes and used within 3 h. 10 μL heparinised whole blood was diluted with 190 μL MEBSS buffer pH 7.4 (Suppl Table 1.) supplemented with 100 μM luminol (Sigma-Aldrich, A4685) in the presence or absence of 5 μM phorbol 12-myristate 13-acetate (PMA; Sigma-Aldrich, P8139) was combined in black-coated 96-well plates and the luminescence signal recorded at 1 min intervals over a 45 min period using a CLARIOstar Microplate Reader (BMG Labtech, Sydney, NSW, Australia). MPO protein levels in plasma were measured by an MPO ELISA kit following manufacturer's instructions (Abcam, ab155458). The effect of SeMet on MPO activity was assessed in vitro using 10 μL of purified MPO (200 ng/mL; Abcam, ab91116), 80 μL 0.75 mM H2O2 (EMD Millipore, Bayswater, VIC, Australia, 1.07298.0250) and 110 μL 3,3′,5,5′-Tetramethylbenzidine solution (2.9 mM TMB in 14.5% DMSO, Sigma-Aldrich, T2885) as described previously [35,36].
Plasma thiol levels were measured using 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB; Sigma-Aldrich, D8130) as described previously [37] with thiol concentration calculated by comparison to authentic GSH standards. Of the different isoforms of GPx, GPx1 is the dominant isotype, displaying positive effects in CVD through increased antioxidant activity under inflammatory conditions [38]. GPx1 expression was assessed within the kidney by immunohistochemistry staining as previously described using a fluorescently-labelled anti- GPx1 primary antibody (Abcam, ab22604) [33]. GPx1 protein expression within the aorta and kidney was further assessed with Western blotting and densitometry utilising ImageJ [32].
2.6. Stimulation of extracellular trap formation
Bone marrow cells were isolated from mice at time of sacrifice using methods previously described by Zhang and co-workers [39], with slight modifications. Briefly, mice femurs were flushed with 5 mL of PBS until the bone cavity appeared clear. Eluted cells were then centrifuged at 500 g at room temperature for 10 min, supernatant was discarded and the cell pellet resuspended in DMEM media (Sigma-Aldrich, D5030) containing 3% foetal bovine serum (FBS). Identification of bone marrow cell types was achieved through Cytospin staining using a commercial DiffQuik kit following manufacturer's instructions (Polysciences Inc, Warrington, PA, USA, 24606). Neutrophils from healthy controls or acute coronary syndrome (ACS) patients were isolated from freshly collected blood as described previously [40]. All studies were performed in compliance with protocols approved by the Sydney Local Health District Human Research Ethics Committee, (Protocol #X12-0241 & HREC/12/RPAH/377). All cells were seeded at density of 1 × 106 cells per mL in DMEM with or without 25 nM PMA for 3 h in order to stimulate extracellular trap (ET) formation. Visualisation and quantification of ET formation was undertaken following SYTOX Green (Life Technologies, Carlsbad, CA, USA, S7020) and qPCR as described previously [41,42], using primer sequences outlined in Suppl. Table 3.
2.7. Ex-vivo aortic vessel function analysis
Pressurised wire myography was used to evaluate the vascular reactivity on mouse aortic ring segments as described previously with minor modifications [43]. In brief, experiments were performed in Krebs’ buffer (Sigma-Aldrich, K3753) supplemented with 3.4 μM CaCl2 and diffused with 95% O2, 5% CO2 at 37 °C. After mounting and stabilisation, 2 mm vessel segments were constricted with 1 × 10−6 M norepinephrine (NE, Sigma-Aldrich, 74480) and then assessment of relaxation potential in response to increasing doses of acetylcholine (Ach, Sigma-Aldrich, A6625) or sodium nitroprusside (SNP, Sigma-Aldrich, PHR1423). Experiments were performed on n ≥ 4 aortas in duplicate, from each of the respective feeding cohorts.
2.8. Statistical analyses
All statistical analyses were carried out using GraphPad Prism 7, with significance determined by either one-way or two-way ANOVA and appropriate post-hoc tests as detailed in the figure legends.
3. Results
3.1. SeMet supplementation decreases atherosclerotic lesion burden accompanied by increased plaque stability and restoration of vessel function
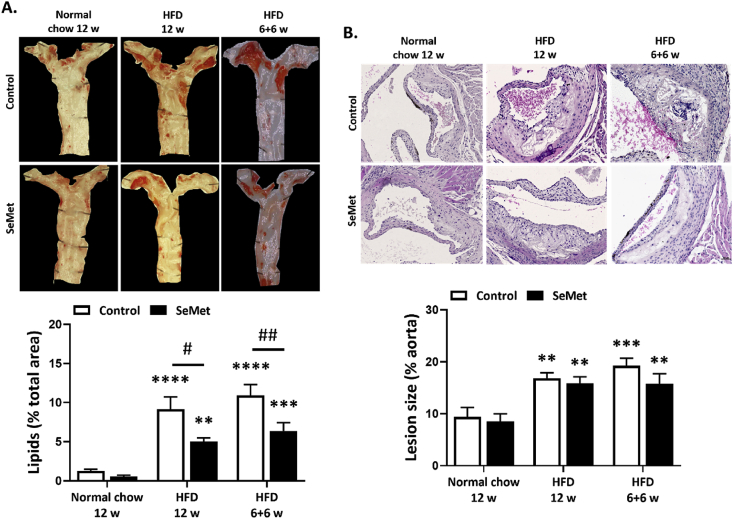
ApoE−/- mice are genetically susceptible to atherosclerotic lesion development and are a well characterised model for human atherosclerosis [44]. Total atherosclerotic lesion burden within mice aortae was examined following Oil red O staining for lipids (Fig. 1A). On a normal chow diet ApoE−/- mice develop plaques both within the aortic root and along the length of the aorta, which are reduced by ~50% in mice cohorts fed 2 mg/kg SeMet over a period of 12 weeks, although this did not reach statistical significance. In mice fed a HFD supplemented with SeMet for 12 weeks, along with the shorter duration of 6 weeks SeMet supplementation, following 6 weeks of HFD, there resulted a detectable level of SeMet within the mouse kidney of between 6 and 10-fold over controls, as assessed by ICP-MS (Suppl. Fig. 1). In the control cohort, a HFD caused a significant increase in lesion burden compared to a normal chow diet, equating to up to ~10% of the total aorta area when assessed at either 12 weeks or 6 weeks. Similar to the normal chow diet, supplementation of the HFD with 2 mg/kg SeMet resulted in a significant decrease in lesion burden, equating to a reduction of ~45% following 12 weeks on the HFD and ~42% in mice on supplementation with SeMet only in the last 6 weeks of the study (Fig. 1A). Measurement of lesion size within the aortic sinus of ApoE−/- mice demonstrated that with a 12 week normal chow diet the lesion size equated to ~10% of the vessel diameter, increasing to ~18% in the HFD cohorts, with slight, but not significant decreases evident within mice supplemented with SeMet (Fig. 1B).
Fig. 1.
SeMet supplementation reduces atherosclerotic lesion burden in mouse aortas. (A) Representative images of Oil Red O staining of lipids as a measure of lesion burden in aorta of mice receiving either normal chow or a high fat diet (HFD) without (open bars) or with (closed bars) 2 mg/kg SeMet (SeMet) supplementation for either 12 w or 6 w normal chow/HFD, followed by 6 w SeMet supplementation. (B) Representative images of H&E stained aortic root sections and quantification of lesion size from mice receiving either normal chow or a high fat diet (HFD) without (open bars) or with (closed bars) 2 mg/kg SeMet (SeMet) supplementation for either 12 w or 6 w normal chow/HFD, followed by 6 w SeMet supplementation. Data represent mean ± S.E.M from n ≥ 5 individual mice per group. **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001 indicates significant difference compared to respective normal chow cohorts, #p ≤ 0.05, ##p ≤ 0.01 indicates significant difference compared to respective control group by two-way ANOVA with Fisher's LSD test. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
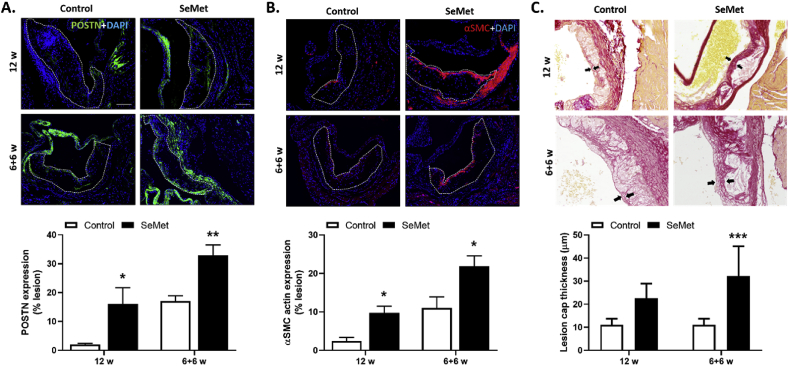
We next determined the vulnerability of lesions in our model, by assessing the extent of tissue fibrosis within the lesion, which is an important determinant of plaque stability [45]. Here we evaluated the extent of expression of two fibrotic markers, periostin (POSTN) and alpha smooth muscle cell (αSMC) actin, within aortic sinus sections by immunofluorescence staining. Mice fed a HFD supplemented with SeMet for 12 weeks demonstrated an approximate 8-fold increase in POSTN staining within the aortic sinus, equating to an increase from ~2% of the lesion area in the control cohort to ~16% of the lesion area in the SeMet fed cohort (Fig. 2A). Similarly, the extent of αSMC actin expression within lesions of the aortic sinus was increased approximately 4-fold in mice supplemented with SeMet for 12 weeks, equating to an increase from ~2% of the lesion area in the control group to ~9% of the lesion area within the SeMet fed group (Fig. 2B).
Fig. 2.
SeMet supplementation increases plaque stabilisation. Representative images of aortic root sections from mice receiving a HFD (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w normal chow, followed by 6 w SeMet supplementation stained for fibrosis markers (A) periostin (POSTN; green staining) or (B) alpha-smooth muscle cell actin (α-SMC; red staining) with DAPI (blue) counter-staining of nuclei. Dashed lines within the representative images indicate the plaque area within the aortic root. (C) Representative images of picrosirius red stained aortic root sections and quantification of lesion cap thickness from mice receiving a high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w normal chow, followed by 6 w SeMet supplementation, with arrows indicating the lesion cap of representative atherosclerotic plaques. Data represent mean ± S.E.M from n ≥ 5 individual mice per group. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001 indicates significant difference compared to respective control group by two-way ANOVA with Fisher's LSD test. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
Further analysis of the extent of fibrosis within aortic sinus tissue sections was undertaken using picrosirus red staining. Here, measurement of lesion cap thickness demonstrated that dietary supplementation with SeMet over the 12-week study period resulted in a doubling in lesion cap thickness from ~11 μm in the control cohort to ~22 μm in the SeMet mice, although this was not statistically significant (Fig. 2C). Interestingly, supplementation with SeMet for the final 6 weeks of the feeding regime, resulted in the lesions within the aortic sinus exhibiting a greater extent of fibrosis than their 12 week counterparts, with an approximate 50% increase in both POSTN (Fig. 2A) and αSMC actin (Fig. 2B) expression, up from ~16% to ~33% of the total lesion size in the former and from ~9% to ~22% in the latter SeMet supplemented cohorts. These fibrotic changes were accompanied by an approximate 3-fold statistically significant increase in lesion cap thickness, from ~11 μm in the control group to ~32 μm in the SeMet cohort (Fig. 2C).
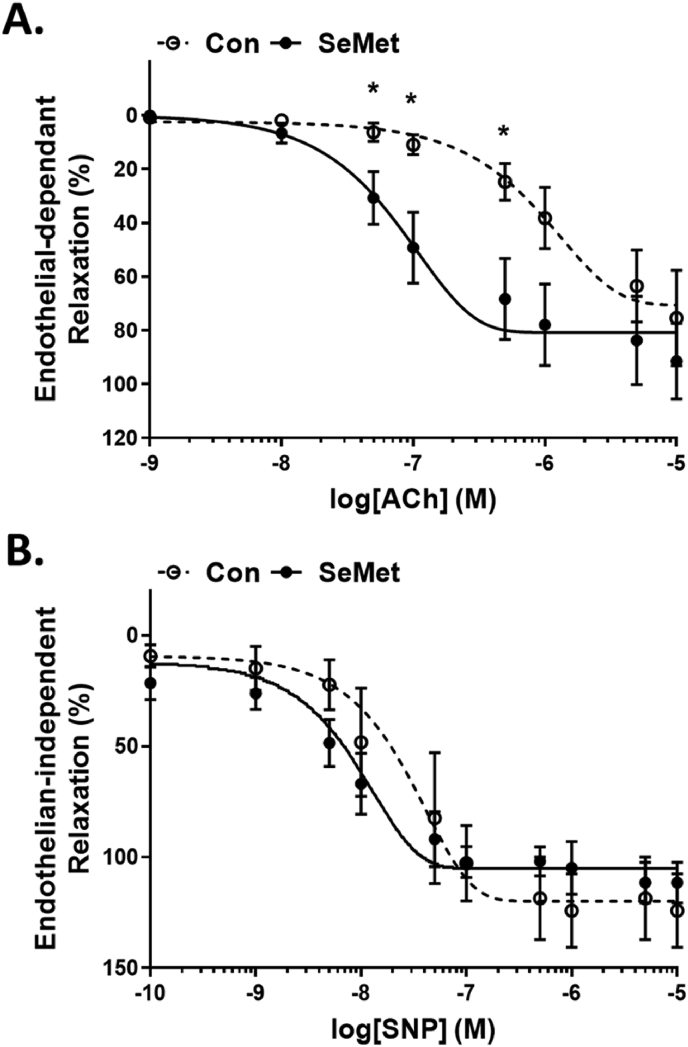
Detrimental vessel changes involving both endothelial dysfunction as well as fibrotic events that result in arterial wall thickening and reduced vessel lumen diameter, are a recognised hallmark of atherosclerosis [1,2]. Given that we had demonstrated that SeMet dietary supplementation resulted in an overall increase in the extent of fibrosis within atherosclerotic lesions, we next sought to establish the effect of SeMet on vessel function. Assessment of both endothelial-dependent (Fig. 3A) and -independent (Fig. 3B) relaxation of mouse aortic segments demonstrated a greater propensity for relaxation in vessels isolated from mice on a HFD supplemented with SeMet in the final 6 weeks of the study, compared to controls. Here, aortic segments from both cohorts responded identically to norepinephrine (NE)-induced constriction (data not shown), but vessels from the SeMet dietary supplemented cohort demonstrated significantly improved relaxation in response to acetylcholine (ACh) stimulation compared to controls, demonstrated by the left shift in the ACh dose-response curve (Fig. 3A). Conversely, despite a trend towards increased relaxation of vessels from the SeMet fed cohort in response to sodium nitroprusside (SNP) stimulation, these data did not reach statistical significance over the controls (Fig. 3B). Together, these data indicate an improvement in endothelial cell function upon SeMet supplementation.
Fig. 3.
SeMet supplementation improves aortic endothelial function through increased nitric oxide bioavailability. Examination of (A) endothelial-dependent relaxation to acetylcholine (Ach) and (B) endothelial-independent relaxation to sodium nitroprusside (SNP) in thoracic aorta segments from mice receiving a HFD for 12 w (Con; open circles) compared to mice receiving a HFD for 6 w, followed by 2 mg/kg SeMet (SeMet; closed circles) supplemented chow for 6 w. Data represent mean ± S.E.M from n ≥ 5 individual mice per group. *p ≤ 0.05 indicates significant difference compared to respective controls by unpaired Student's t-test.
3.2. SeMet supplementation favours macrophage polarisation towards the alternatively activated phenotype
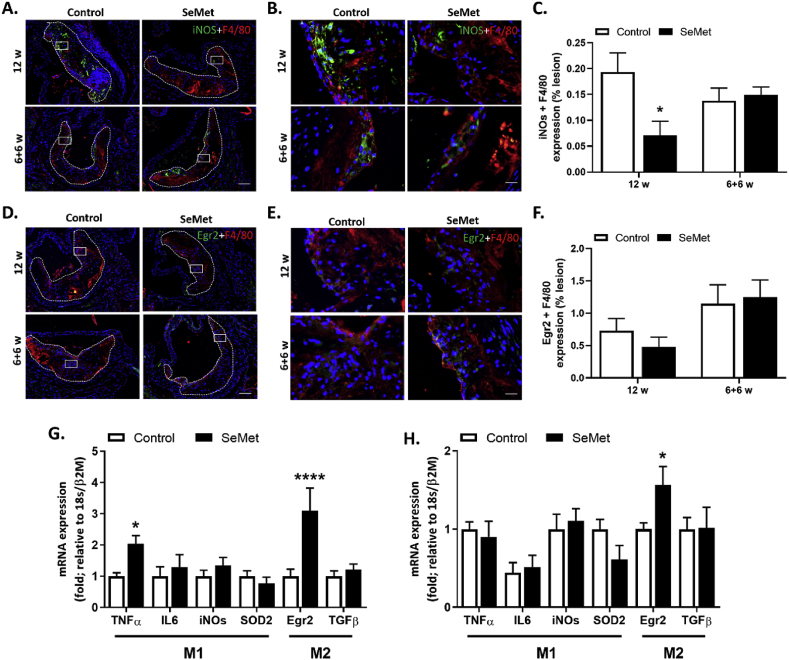
Macrophage infiltration, differentiation and activation all play contributing roles in atherosclerotic lesion development. As assessed by immunohistochemistry staining for macrophage-specific (F4/80) iNOS expression within mice atherosclerotic lesions, dietary SeMet supplementation over a 12 week period resulted in a significant decrease (65%) in pro-inflammatory M1 macrophages in lesions compared to controls, whilst lesion iNOS expression in mice supplemented with SeMet in the final 6 weeks of the study remained at the same level as controls (Fig. 4A–C). Conversely, macrophages within lesions from the SeMet cohort did not demonstrate any comparative difference in expression of the mouse-specific alternatively-activated M2 phenotype marker Egr2, regardless of feeding regime (Fig. 4D–F).
Fig. 4.
SeMet supplementation alters macrophage phenotype. (A) Representative aortic root images of immuno-histochemical staining for macrophages (red) with the M1 phenotype marker iNOS (green) with DAPI counter-staining of nuclei (blue) from mice on high fat diet (Control) compared to 2 mg/kg SeMet (SeMet) supplemented chow for either 12 w or 6 w chow, followed by 6 w SeMet supplementation. Dashed lines within the representative images indicate the plaque area within the aortic root with (B) high-power magnification image of white boxed area in (A) and (C) quantification of macrophage-specific iNOs staining from mice on high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w chow, followed by 6 w SeMet supplementation. (D) Representative aortic root images of immuno-histochemical staining for macrophages (red) with the M2 phenotype marker Egr2 (green) with DAPI counter-staining of nuclei (blue) from mice on high fat diet (Control) compared to 2 mg/kg SeMet (SeMet) supplemented chow for either 12 w or 6 w chow, followed by 6 w SeMet supplementation. Dashed lines within the representative images indicate the plaque area within the aortic root, with (E) high-power magnification image of white boxed area in (D) and (F) quantification of macrophage-specific Egr2 staining from mice on high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w chow, followed by 6 w SeMet supplementation. qPCR analysis of mRNA expression of M1 phenotype markers TNFα, IL-6, iNOs and SOD2, or M2 phenotype markers Egr2 and TGFβ within the descending aorta from mice on high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either (G) 12 w or (H) 6 w chow, followed by 6 w SeMet supplementation. Data expressed as mean ± S.E.M from n ≥ 5 individual mice per group. *p ≤ 0.05, ****p ≤ 0.001indicates significant difference compared to respective controls by two-way ANOVA with Fisher's LSD test. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
Further analysis of macrophage phenotype markers across the entirety of the aorta by qPCR analysis revealed both a significant 2-fold increase in M1 TNFα mRNA expression and a significant 3-fold increase in M2 Egr2 expression within aortae isolated from the 12 week SeMet fed cohort compared to controls (Fig. 4G). Erg2 mRNA was also significantly increased following SeMet supplementation only in the final 6 weeks of the study period. Conversely, there was no difference in aortic mRNA expression observed across all cohorts of the M1 markers IL6, iNOS and SOD2 or the M2 marker TGFβ (Fig. 4G/H).
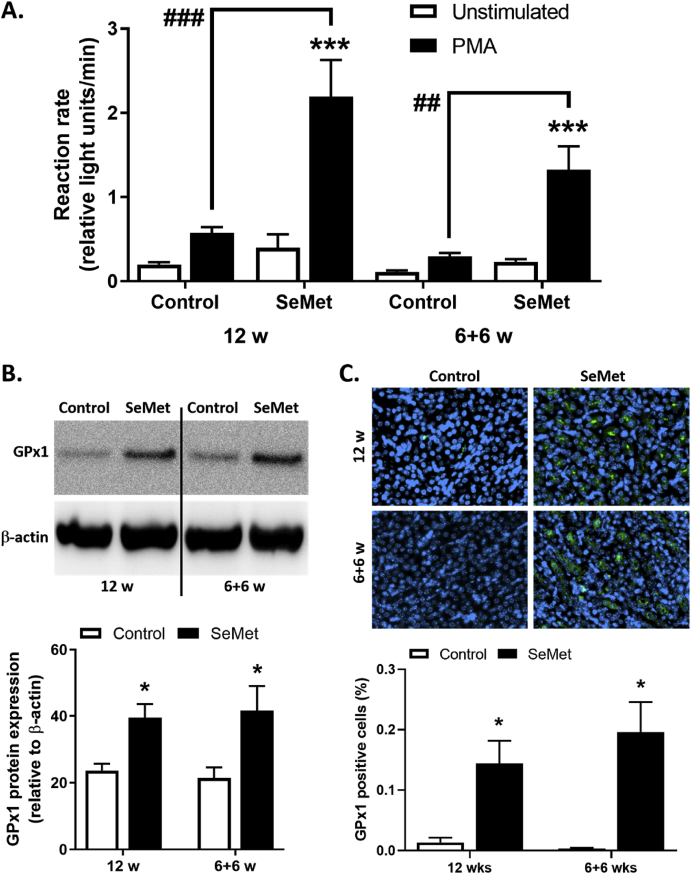
3.3. SeMet supplementation increases peroxidase expression and activity
SeMet supplementation is reported to increase the expression and activity selenium-dependent endogenous peroxidase systems [46]. In confirmation of this, we demonstrated that compared to controls, dietary supplementation with SeMet significantly increased the overall peroxidase activity within whole mouse blood, as determined by luminol luminescence following PMA stimulation. Here, in the presence of PMA, the luminol reaction rate was increased up to ~ 4-fold in the SeMet supplemented cohorts (Fig. 5A). Further investigation revealed that this increase in peroxidase activity within the blood was not due to an increase in circulating MPO release from activated inflammatory cells, with no difference in the mouse plasma MPO levels between cohorts (Suppl. Fig. 2A). In support of this, in vitro studies showed that SeMet supplementation of up to 25 μM had no effect on purified human MPO activity, assayed in the presence of H2O2 and TMB substrate (Suppl. Fig. 2B). In addition, no alternation of mouse plasma thiol levels was found in SeMet supplemented mice (Suppl. Fig. 2C). To identify a potential source of this increase relevant to our study, we next assessed the extent of GPx1 protein expression within the aorta of mice. Compared to other isoforms of GPx, GPx1 is the dominant isoform in mammalian tissues and its expression is highly sensitive to the availability of Se [38]. Here, using Western blotting to probe for GPx1 within homogenised aortic tissue, we demonstrate that cohorts of mice fed a diet supplemented with SeMet exhibited an approximate 2-fold increase in GPx1 expression, compared to the control-fed cohort (Fig. 5B). Similarly, evidence for whole-body increases in GPx1 was established by immunohistochemical analysis of kidney tissue. Compared to control mice, supplementation with SeMet resulted in a dramatic increase in the expression of GPx1 in the kidney, equating to ~10-fold increase in GPx1 expression, ranging from ~0.003 to 0.013% of overall kidney cells in control mice and increasing in range from 0.14 to 0.19% of the kidney cells in the SeMet supplemented cohorts (Fig. 5C).
Fig. 5.
SeMet supplementation increases GPx expression and activity. (A) Total peroxidase activity in mouse whole blood determined by the chemiluminescence reaction rate of luminol without (Unstimulated; open bars) or with 25 nM PMA (closed bars). (B) Representative Western blots and analysis of GPx-1 expression in aorta from mice on high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w normal chow, followed by 6 w SeMet supplementation. (C) Representative immuno-histochemical staining of kidney for GPx-1 (green) and DAPI staining of nuclei (blue) and quantification of GPx-1 positive cells from mice on high fat diet (Control; open bars) compared to 2 mg/kg SeMet (SeMet; closed bars) supplemented chow for either 12 w or 6 w normal chow, followed by 6 w SeMet supplementation. Data represent mean ± S.E.M from n ≥ 3 individual mice per group. *p ≤ 0.05, ***p ≤ 0.001 indicates significant difference compared to respective controls or ##p ≤ 0.01, ###p ≤ 0.001 between groups, by two-way ANOVA with Fisher's LSD Test. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
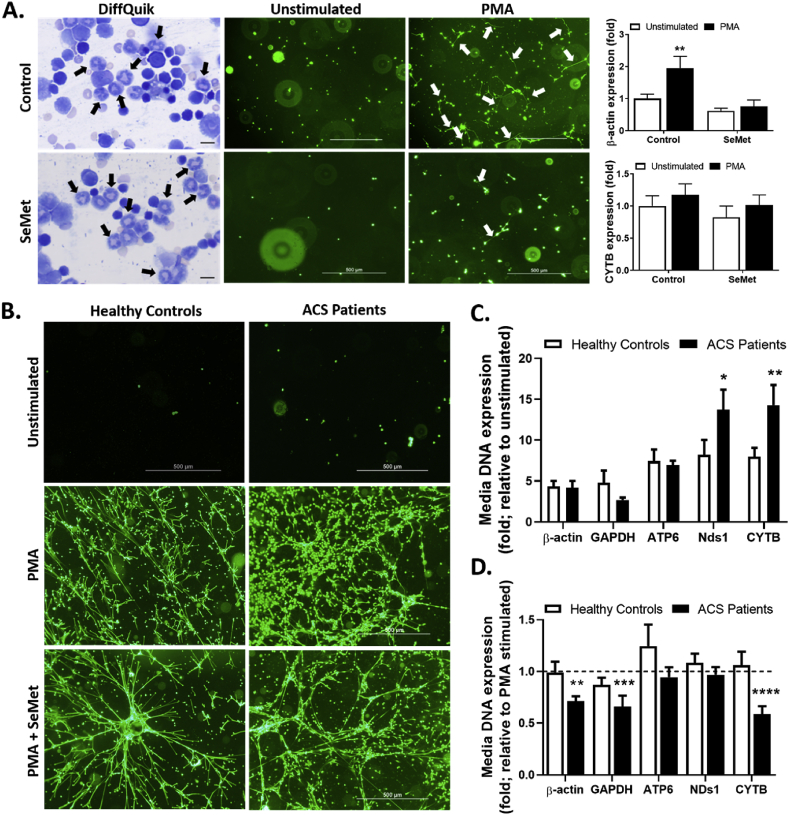
3.4. SeMet supplementation decreases extracellular trap formation
As a part of their inflammatory repertoire, neutrophils undergo a form of cell death known as NETosis, in which they produce an extracellular trap (NETs) consisting of extruded DNA and chromatin, decorated with enzymes, including neutrophil elastase and MPO [41,47]. Increasing evidence has implicated NETs in atherosclerotic lesion progression [48] and thrombosis [49]. Therefore, the extent of NET release was assessed in neutrophils isolated from bone marrow cells from mice fed a HFD with and without supplementation with SeMet for 12 weeks. In each case, the mouse bone marrow-derived cells contained a comparative abundance of neutrophils as assessed by DiffQuik staining (Fig. 6A). These cell cultures were then stimulated with 25 nM PMA for 3 h to invoke NET formation, as evidenced by increased SYTOX green staining of extracellular DNA (Fig. 6A). Cells isolated from mice receiving a diet supplemented with SeMet demonstrated a comparative decrease in the extent of NET formation, as indicated both from the live cell images (white arrows) and on quantification by qPCR analysis of genomic DNA present within the cell culture medium following stimulation. Here, the culture media of cells from control mice exhibited an approximately 2-fold increase in nuclear-derived β-actin DNA expression, whilst media from cells derived from the SeMet cohort remained at basal levels following PMA stimulation. This was distinct from qPCR analysis of the extent of mitochondria-derived CYTB genomic DNA within the culture media following PMA stimulation, which was comparative between both cohorts (Fig. 6A).
Fig. 6.
SeMet supplementation inhibits ET formation. (A) Bone marrow cells were isolated from mice on HFD (Control) or HFD supplemented with 2 mg/kg SeMet for 12 w and incubated without (Unstimulated; open bars) or with 25 nM PMA (closed bars) for 3 h to induce ET formation, with quantification of genomic DNA expression in the culture media of the nuclear encoded gene β-actin or the mitochondrial gene CYTB assessed by qPCR. Representative images of bone marrow cells stained with either DiffQuik (black arrows indicating neutrophils) or SYTOX green for extracellular DNA (white arrows). (B) Representative images of SYTOX green stained neutrophils isolated from either healthy donors (left panel) or acute coronary syndrome (ACS) patients (right panel) following stimulation with 25 nM PMA without or with 1 μM SeMet for 3 h. (C) Quantification of genomic DNA expression in the culture media following PMA stimulation of the nuclear encoded genes β-actin and GAPDH or the mitochondrial genes ATP6, MT- NDs1 and CYTB assessed by qPCR, from healthy donors (open bars) or ACS patients (closed bars). (D) same as for (C), except in the presence of 1 μM SeMet, with data represented here as a fold expression relative to healthy control or ACS patient neutrophils assayed in the absence of SeMet (dotted line). For the mice experiments, data represent mean ± S.E.M from n ≥ 4 individual mice per group assayed in triplicate. For human neutrophil studies, data represent mean ± S.E.M from n ≥ 5 individual donors per group assayed in triplicate. *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001 indicates significant difference compared to respective controls by two-way ANOVA with Fisher's LSD comparisons test. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
Translating the results from our mouse atherosclerosis study into a clinically-relevant scenario, we next isolated neutrophils from acute coronary syndrome (ACS) patients and determined the extent of NET formation from these cells and healthy donor neutrophils, in response to PMA stimulation both in the absence and presence of 1 μM SeMet added after neutrophil isolation. Both healthy donor and ACS patient neutrophils responded to PMA stimulation with NET production, as indicated by positive SYTOX green staining of extracellular DNA (Fig. 6B). Quantification of extracellular genomic DNA from healthy control stimulated neutrophils demonstrated an approximate 4-fold increase in media concentration of the nuclear genes β-actin and GAPDH, and an approximate 8-fold increase in media concentration of the mitochondrial genes ATP6, Nds1 and CYTB, compared to unstimulated controls (Fig. 6C). Similarly, PMA-stimulated neutrophils isolated form ACS patients exhibited a 4-fold increase in media expression of β-actin, a 3-fold increase in GAPDH, a 7-fold increase in ATP6, whilst extracellular expression of Nds1 and CYTB was significantly increased to ~14-fold over unstimulated controls (Fig. 6C). Interestingly, supplementation of the culture media with 1 μM SeMet had no effect on the extent of NET production from healthy donor neutrophils, as assessed through qPCR analysis of extracellular genomic DNA, but did reduce the expression of genomic DNA within the media from neutrophils isolated from ACS patients. Here, there was a significant decrease in the extracellular expression of nuclear genes β-actin and GAPDH as well as the mitochondria-derived gene CYTB from neutrophils assayed in the presence of SeMet, compared to control neutrophils stimulated without SeMet present (Fig. 6D).
4. Discussion
The aim of the present study was to investigate the effects of SeMet supplementation on the development of atherosclerosis in ApoE−/- mice. Mice were raised on a HFD supplemented with 2 mg/kg SeMet for either 12 weeks or a HFD for a period of 6 weeks, followed by a further 6 weeks with SeMet supplementation. These time points were chosen to reflect different stages of lesion development in ApoE−/- mice [44], with the latter cohort enabling the ability of SeMet to regress already established atherosclerotic plaques. We have demonstrated that SeMet supplementation reduces atherosclerotic lesion burden and plaque vulnerability, whilst improving vascular tone. Evidence was also obtained for an increase in the expression and activity of endogenous GPx antioxidant mechanisms. Furthermore, supplementation with SeMet decreased inflammatory M1 macrophages within the plaque and reduced the extent of NET release from bone marrow-derived cells isolated from mice following SeMet dietary intervention. Similarly, supplementation of neutrophils with SeMet ex vivo reduced NET release in cells isolated from ACS patients. Combined, these data indicate that SeMet supplementation can prevent atherosclerosis progression and potentially modulate the inflammatory response.
Previous studies have identified a crucial role for selenium in the regulation of endogenous antioxidant defence systems via alteration of GPx activity [20,50]. Dietary supplementation with selenium-enriched yeast sources or sodium selenite, an inorganic form of selenium, increases antioxidant activity, specifically GPx [51,52], with SeMet able to regulate the GPx pathway in tissue microenvironments in response to oxidative stress [27,53]. In the present study, we identified an increase in whole-blood peroxidase activity within SeMet supplemented mice. As with the earlier studies, our study specifies an increase in GPx1 expression, present in both the aorta and kidney of SeMet supplemented mice, indicating that this selenium-dependent antioxidant enzyme is the most likely source of the increased peroxidase activity evident and providing further evidence that selenium can increase antioxidant capacity via the GPx1 pathway [20,50]. To further support this, we also showed that SeMet supplementation had no effect on MPO expression or activity, which supports the conclusion that the increased peroxidase activity in whole-blood from SeMet supplemented mice is independent of MPO.
It has been previously shown that Se supplementation effectively reduced mortality in patients with severe systemic inflammatory response syndrome, suggesting a regulatory role for Se in inflammatory settings [54]. Despite these positive findings, the use of Se compounds as dietary supplements for the improvement of general health in humans remains controversial, as excess consumption may cause symptoms of toxicity, such as diarrhea and joint pain [55]. Similarly, in animal models an overdose of Se can exacerbate inflammatory responses, resulting in further damage to tissue [53]. However, compared to sodium selenite, SeMet supplementation at a comparable concentration to that used in the present study, did not enhance the extent of proinflammatory cytokine release from inflammatory cells [56].
Macrophages play an important role in chronic inflammatory diseases and its presence provides an indication of disease progression during atherogenesis [11,57,58]. In order to delineate the mechanisms attributable to the SeMet modulation of atherosclerosis we examined macrophage phenotypes within developed lesions. We showed reduced M1 proinflammatory macrophages within lesions that was accompanied by increased mRNA expression of the mouse-specific anti-inflammatory M2 marker Egr2 [59] throughout the aorta, indicating the modification of macrophage phenotypic differentiation as a potential mechanism by which SeMet supplementation can limit tissue damage and accompanying lesion development. This apparent accumulation of M2 polarised macrophages in the SeMet supplemented cohort may reflect the ROS-scavenging ability of the compound [60], favouring a less inflammatory environment. Additionally, the overall increase in M2 polarised macrophages in atherosclerosis function to remove tissue debris and are more resistant to foam cell formation, with their presence enhanced in the regression stage of the disease and particularly associated with stable plaques [14,61]. In support of this, our data further suggests that SeMet may promote tissue recovery by increasing atherosclerotic plaque stability, with notable increases in lesion cap thickness in mice supplemented with SeMet.
Alongside macrophages, neutrophils play a contributory role in atherosclerotic lesion development [3,8]. This is attributed in part to their ability to form NETs, which serve to exacerbate local inflammation, arterial intimal injury and thrombosis [49], with previous studies in mice demonstrating that inhibition of NET production results in a reduction in lesion burden [48]. We show that SeMet is able to modulate the neutrophil acute inflammatory response, with a decrease in NET production evident within mice fed a SeMet supplemented diet, as wells as in neutrophils isolated from ACS patients. NETosis occurs by two distinct forms: suicidal NETosis, as the name suggests, involves extrusion of nuclear components that is accompanied by cell death [62]. Vital NETosis, conversely involves the extracellular extrusion of DNA primarily from the mitochondria and is independent of cell death [3,63,64]. Our findings in mice suggest murine neutrophils undergo primarily suicidal NETosis during ET formation, whilst analysis of the genomic DNA extruded by human neutrophils provides evidence of both suicidal and vital NETosis occurring, with the latter form heightened in ACS patients compared to healthy controls. Regardless of species origin, we demonstrate that SeMet supplementation significantly ameliorates NET production, which may pertain to the compounds antioxidant and radical scavenging abilities [65] with NET generation largely dependent on ROS production, particularly from NADPH oxidase activity within the neutrophil [66].
The development of therapeutics promoting regression of atherosclerosis remain an important clinical objective particularly as humans typically do not present for treatment until well after the disease has substantially developed. In this study, cohorts of mice supplemented with SeMet only in the final 6 weeks of the HFD feeding regime not only demonstrated reduced lesion burden that was comparable to the 12 week SeMet study, but also had increased lesion expression of profibrotic markers and cap thickness. Furthermore, compared to controls, aorta from these mice displayed improved endothelial-dependent relaxation. In support of this, similar functional vessel studies of aortae isolated from rats fed a low Se diet demonstrated impaired relaxation that was mediated by reduced eNOS activity [67]. Similarly, within a rat model of homocysteine-induced endothelial dysfunction, Se supplementation increased NO within the plasma and improved endothelial-dependent vessel relaxation. Results that were confirmed within in vitro studies using HUVECs, which demonstrated increased eNOS expression and activity with accompanying NO production upon Se supplementation [68]. Conflicting data does exist, however, with studies utilising spontaneously hypertensive rats (SHR) demonstrating that SeMet supplementation decreased eNOS expression within the aortic wall [69]. A limitation of the current study is that eNOS activity and the levels of NO were not assessed, which may have particular relevance to the 12 w HFD cohort, given that SeMet supplementation decreased iNOs expression under these conditions.
Despite a recent study indicating an association between Se deficiency and metabolomic remodelling which influences longevity pathways in mice [30], low Se status is associated with CVD in humans [70] and evidence generated from a variety of animal studies consistently show that Se supplementation is able to prevent atherosclerotic lesion development and reduce the risk of CVD [71,72]. Much of these latter data, however, have been limited to the use of inorganic Se compounds, with underlying mechanisms of protection focused on the bolstering of endogenous antioxidant systems, while other important events associated with disease progression, such as the inflammatory response and resultant vascular cell dysfunction, remain uncharacterised. This study not only supports the inhibitory role of SeMet supplementation in atherosclerotic lesion progression and burden, but further highlights the anti-inflammatory aspects of the compound. Our data highlight the benefit of SeMet dietary supplementation in both preventing progression and promoting regression of atherosclerosis, together with improved vascular function, whilst providing new insights into the protective mechanisms of SeMet supplementation within the setting of inflammation.
Declaration of competing interestCOI
The authors of nothing to disclose.
Acknowledgements
This work was supported by a Perpetual IMPACT Grant (IPAP201601422). YZ acknowledges the receipt of an Australian Postgraduate Award from the University of Sydney. SP was supported by a Ramaciotti Health Investment Grant. BR was supported by a Commonwealth Scientific and Industrial Research Organisation (CSIRO) Science and Industry Endowment Fund (SIEF) STEM+ Business Fellowship. We would like to thank Mr Pat Pisansarakit and Ms Morgan Jones for assistance with the mouse bone marrow cell isolation and tissue culture.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.redox.2019.101409.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Cheung A.K., Sarnak M.J., Yan G., Dwyer J.T., Heyka R.J., Rocco M.V., Teehan B.P., Levey A.S. Atherosclerotic cardiovascular disease risks in chronic hemodialysis patients. Kidney Int. 2000;58(1):353–362. doi: 10.1046/j.1523-1755.2000.00173.x. [DOI] [PubMed] [Google Scholar]
- 2.Frostegard J. Immunity, atherosclerosis and cardiovascular disease. BMC Med. 2013;11:117. doi: 10.1186/1741-7015-11-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chavez-Sanchez L., Espinosa-Luna J.E., Chavez-Rueda K., Legorreta-Haquet M.V., Montoya-Diaz E., Blanco-Favela F. Innate immune system cells in atherosclerosis. Arch. Med. Res. 2014;45(1):1–14. doi: 10.1016/j.arcmed.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 4.Mallat Z., Tedgui A. Immunomodulation to combat atherosclerosis: the potential role of immune regulatory cells. Expert Opin. Biol. Ther. 2004;4(9):1387–1393. doi: 10.1517/14712598.4.9.1387. [DOI] [PubMed] [Google Scholar]
- 5.Hansson G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005;352(16):1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 6.Libby P. Inflammation in atherosclerosis. Nature. 2002;420(6917):868–874. doi: 10.1038/nature01323. [DOI] [PubMed] [Google Scholar]
- 7.Libby P., Ridker P.M., Maseri A. Inflammation and atherosclerosis. Circulation. 2002;105(9):1135–1143. doi: 10.1161/hc0902.104353. [DOI] [PubMed] [Google Scholar]
- 8.Soehnlein O. Multiple roles for neutrophils in atherosclerosis. Circ. Res. 2012;110(6):875–888. doi: 10.1161/CIRCRESAHA.111.257535. [DOI] [PubMed] [Google Scholar]
- 9.Hartwig H., Silvestre Roig C., Daemen M., Lutgens E., Soehnlein O. Neutrophils in atherosclerosis. A brief overview. Hämostaseologie. 2015;35(2):121–127. doi: 10.5482/HAMO-14-09-0040. [DOI] [PubMed] [Google Scholar]
- 10.De Paoli F., Staels B., Chinetti-Gbaguidi G. Macrophage phenotypes and their modulation in atherosclerosis. Circ. J. 2014;78(8):1775–1781. doi: 10.1253/circj.cj-14-0621. [DOI] [PubMed] [Google Scholar]
- 11.Leitinger N., Schulman I.G. Phenotypic polarization of macrophages in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2013;33(6):1120–1126. doi: 10.1161/ATVBAHA.112.300173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Decano J.L., Aikawa M. Dynamic macrophages: understanding mechanisms of activation as guide to therapy for atherosclerotic vascular disease. Front. Cardiovasc. Med. 2018;5:97. doi: 10.3389/fcvm.2018.00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bobryshev Y.V., Ivanova E.A., Chistiakov D.A., Nikiforov N.G., Orekhov A.N. Macrophages and their role in atherosclerosis: pathophysiology and transcriptome analysis. BioMed Res. Int. 2016;2016:1–13. doi: 10.1155/2016/9582430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tao B., Jin W., Xu J., Liang Z., Yao J., Zhang Y., Wang K., Cheng H., Zhang X., Ke Y. Myeloid-specific disruption of tyrosine phosphatase Shp2 promotes alternative activation of macrophages and predisposes mice to pulmonary fibrosis. J. Immunol. 2014;193(6):2801–2811. doi: 10.4049/jimmunol.1303463. [DOI] [PubMed] [Google Scholar]
- 15.Wilson H.M. Macrophages heterogeneity in atherosclerosis - implications for therapy. J. Cell Mol. Med. 2010;14(8):2055–2065. doi: 10.1111/j.1582-4934.2010.01121.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu H., Xu H., Huang K. Selenium in the prevention of atherosclerosis and its underlying mechanisms. Metallomics. 2017;9(1):21–37. doi: 10.1039/c6mt00195e. [DOI] [PubMed] [Google Scholar]
- 17.Fairweather-Tait S.J., Bao Y., Broadley M.R., Collings R., Ford D., Hesketh J.E., Hurst R. Selenium in human health and disease. Antioxidants Redox Signal. 2011;14(7):1337–1383. doi: 10.1089/ars.2010.3275. [DOI] [PubMed] [Google Scholar]
- 18.Labunskyy V.M., Hatfield D.L., Gladyshev V.N. Selenoproteins: molecular pathways and physiological roles. Physiol. Rev. 2014;94(3):739–777. doi: 10.1152/physrev.00039.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kryukov G.V., Castellano S., Novoselov S.V., Lobanov A.V., Zehtab O., Guigo R., Gladyshev V.N. Characterization of mammalian selenoproteomes. Science. 2003;300(5624):1439–1443. doi: 10.1126/science.1083516. [DOI] [PubMed] [Google Scholar]
- 20.Tinggi U. Selenium: its role as antioxidant in human health. Environ. Health Prev. Med. 2008;13(2):102–108. doi: 10.1007/s12199-007-0019-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wojcicki J., Rozewicka L., Barcew-Wiszniewska B., Samochowiec L., Juzwiak S., Kadlubowska D., Tustanowski S., Juzyszyn Z. Effect of selenium and vitamin E on the development of experimental atherosclerosis in rabbits. Atherosclerosis. 1991;87(1):9–16. doi: 10.1016/0021-9150(91)90227-t. [DOI] [PubMed] [Google Scholar]
- 22.Schwenke D.C., Behr S.R. Vitamin E combined with selenium inhibits atherosclerosis in hypercholesterolemic rabbits independently of effects on plasma cholesterol concentrations. Circ. Res. 1998;83(4):366–377. doi: 10.1161/01.res.83.4.366. [DOI] [PubMed] [Google Scholar]
- 23.Zhang F., Yu W., Hargrove J.L., Greenspan P., Dean R.G., Taylor E.W., Hartle D.K. Inhibition of TNF-alpha induced ICAM-1, VCAM-1 and E-selectin expression by selenium. Atherosclerosis. 2002;161(2):381–386. doi: 10.1016/s0021-9150(01)00672-4. [DOI] [PubMed] [Google Scholar]
- 24.Blankenberg S., Rupprecht H.J., Bickel C., Torzewski M., Hafner G., Tiret L., Smieja M., Cambien F., Meyer J., Lackner K.J., AtheroGene I. Glutathione peroxidase 1 activity and cardiovascular events in patients with coronary artery disease. N. Engl. J. Med. 2003;349(17):1605–1613. doi: 10.1056/NEJMoa030535. [DOI] [PubMed] [Google Scholar]
- 25.Schnabel R., Lackner K.J., Rupprecht H.J., Espinola-Klein C., Torzewski M., Lubos E., Bickel C., Cambien F., Tiret L., Munzel T., Blankenberg S. Glutathione peroxidase-1 and homocysteine for cardiovascular risk prediction: results from the AtheroGene study. J. Am. Coll. Cardiol. 2005;45(10):1631–1637. doi: 10.1016/j.jacc.2005.02.053. [DOI] [PubMed] [Google Scholar]
- 26.Wastney M.E., Combs G.F., Jr., Canfield W.K., Taylor P.R., Patterson K.Y., Hill A.D., Moler J.E., Patterson B.H. A human model of selenium that integrates metabolism from selenite and selenomethionine. J. Nutr. 2011;141(4):708–717. doi: 10.3945/jn.110.129049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mahima, Verma A.K., Kumar A., Rahal A., Kumar V., Roy D. Inorganic versus organic selenium supplementation: a review. Pak. J. Biol. Sci. 2012;15(9):418–425. doi: 10.3923/pjbs.2012.418.425. [DOI] [PubMed] [Google Scholar]
- 28.Thomson C.D., Robinson M.F., Butler J.A., Whanger P.D. Long-term supplementation with selenate and selenomethionine: selenium and glutathione peroxidase (EC 1.11.1.9) in blood components of New Zealand women. Br. J. Nutr. 1993;69(2):577–588. doi: 10.1079/bjn19930057. [DOI] [PubMed] [Google Scholar]
- 29.Gu Q.P., Xia Y.M., Ha P.C., Butler J.A., Whanger P.D. Distribution of selenium between plasma fractions in Guinea pigs and humans with various intakes of dietary selenium. J. Trace Elem. Med. Biol. 1998;12(1):8–15. doi: 10.1016/S0946-672X(98)80015-1. [DOI] [PubMed] [Google Scholar]
- 30.Yim S.H., Clish C.B., Gladyshev V.N. Selenium deficiency is associated with pro-longevity mechanisms. Cell Rep. 2019;27(9):2785–2797. doi: 10.1016/j.celrep.2019.05.001. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lin Y., Bai L., Chen Y., Zhu N., Bai Y., Li Q., Zhao S., Fan J., Liu E. Practical assessment of the quantification of atherosclerotic lesions in apoE(-)/(-) mice. Mol. Med. Rep. 2015;12(4):5298–5306. doi: 10.3892/mmr.2015.4084. [DOI] [PubMed] [Google Scholar]
- 32.Schneider C.A., Rasband W.S., Eliceiri K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods. 2012;9(7):671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Menon P., Fisher E.A. Immunostaining of macrophages, endothelial cells, and smooth muscle cells in the atherosclerotic mouse aorta. Methods Mol. Biol. 2015;1339:131–148. doi: 10.1007/978-1-4939-2929-0_9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gross S., Gammon S.T., Moss B.L., Rauch D., Harding J., Heinecke J.W., Ratner L., Piwnica-Worms D. Bioluminescence imaging of myeloperoxidase activity in vivo. Nat. Med. 2009;15(4):455–461. doi: 10.1038/nm.1886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pulli B., Ali M., Forghani R., Schob S., Hsieh K.L., Wojtkiewicz G., Linnoila J.J., Chen J.W. Measuring myeloperoxidase activity in biological samples. PLoS One. 2013;8(7) doi: 10.1371/journal.pone.0067976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Suzuki K., Ota H., Sasagawa S., Sakatani T., Fujikura T. Assay method for myeloperoxidase in human polymorphonuclear leukocytes. Anal. Biochem. 1983;132(2):345–352. doi: 10.1016/0003-2697(83)90019-2. [DOI] [PubMed] [Google Scholar]
- 37.Hawkins C.L., Morgan P.E., Davies M.J. Quantification of protein modification by oxidants. Free Radic. Biol. Med. 2009;46(8):965–988. doi: 10.1016/j.freeradbiomed.2009.01.007. [DOI] [PubMed] [Google Scholar]
- 38.Brigelius-Flohe R., Maiorino M. Glutathione peroxidases. Biochim. Biophys. Acta. 2013;1830(5):3289–3303. doi: 10.1016/j.bbagen.2012.11.020. [DOI] [PubMed] [Google Scholar]
- 39.Zhang X., Goncalves R., Mosser D.M. The isolation and characterization of murine macrophages. Curr. Protoc. Im. 2008 Nov doi: 10.1002/0471142735.im1401s83. (Chapter 14): p. Unit 14 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Oh H., Siano B., Diamond S. Neutrophil isolation protocol. J. Vis. Exp. 2008;(17) doi: 10.3791/745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Brinkmann V., Reichard U., Goosmann C., Fauler B., Uhlemann Y., Weiss D.S., Weinrauch Y., Zychlinsky A. Neutrophil extracellular traps kill bacteria. Science. 2004;303(5663):1532–1535. doi: 10.1126/science.1092385. [DOI] [PubMed] [Google Scholar]
- 42.Rayner B.S., Zhang Y., Brown B.E., Reyes L., Cogger V.C., Hawkins C.L. Role of hypochlorous acid (HOCl) and other inflammatory mediators in the induction of macrophage extracellular trap formation. Free Radic. Biol. Med. 2018;129:25–34. doi: 10.1016/j.freeradbiomed.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 43.Mian M.O., Idris-Khodja N., Li M.W., Leibowitz A., Paradis P., Rautureau Y., Schiffrin E.L. Preservation of endothelium-dependent relaxation in atherosclerotic mice with endothelium-restricted endothelin-1 overexpression. J. Pharmacol. Exp. Ther. 2013;347(1):30–37. doi: 10.1124/jpet.113.206532. [DOI] [PubMed] [Google Scholar]
- 44.Whitman S.C. A practical approach to using mice in atherosclerosis research. Clin. Biochem. Rev. 2004;25(1):81–93. [PMC free article] [PubMed] [Google Scholar]
- 45.Hansson G.K., Libby P., Tabas I. Inflammation and plaque vulnerability. J. Intern. Med. 2015;278(5):483–493. doi: 10.1111/joim.12406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Li K.X., Wang J.S., Yuan D., Zhao R.X., Wang Y.X., Zhan X.A. Effects of different selenium sources and levels on antioxidant status in broiler breeders. Asian-Australas. J. Anim. Sci. 2018;31(12):1939–1945. doi: 10.5713/ajas.18.0226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zawrotniak M., Rapala-Kozik M. Neutrophil extracellular traps (NETs) - formation and implications. Acta Biochim. Pol. 2013;60(3):277–284. [PubMed] [Google Scholar]
- 48.Knight J.S., Luo W., O'Dell A.A., Yalavarthi S., Zhao W., Subramanian V., Guo C., Grenn R.C., Thompson P.R., Eitzman D.T., Kaplan M.J. Peptidylarginine deiminase inhibition reduces vascular damage and modulates innate immune responses in murine models of atherosclerosis. Circ. Res. 2014;114(6):947–956. doi: 10.1161/CIRCRESAHA.114.303312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Martinod K., Wagner D.D. Thrombosis: tangled up in NETs. Blood. 2014;123(18):2768–2776. doi: 10.1182/blood-2013-10-463646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Brenneisen P., Steinbrenner H., Sies H. Selenium, oxidative stress, and health aspects. Mol. Asp. Med. 2005;26(4–5):256–267. doi: 10.1016/j.mam.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 51.Cheng W.H., Combs G.F., Jr., Lei X.G. Knockout of cellular glutathione peroxidase affects selenium-dependent parameters similarly in mice fed adequate and excessive dietary selenium. Biofactors. 1998;7(4):311–321. doi: 10.1002/biof.5520070403. [DOI] [PubMed] [Google Scholar]
- 52.Cheng W.H., Ho Y.S., Ross D.A., Valentine B.A., Combs G.F., Lei X.G. Cellular glutathione peroxidase knockout mice express normal levels of selenium-dependent plasma and phospholipid hydroperoxide glutathione peroxidases in various tissues. J. Nutr. 1997;127(8):1445–1450. doi: 10.1093/jn/127.8.1445. [DOI] [PubMed] [Google Scholar]
- 53.Tos-Luty S., Obuchowska-Przebirowska D., Latuszynska J., Musik I., Tokarska-Rodak M. Comparison of histological and ultrastructural changes in mice organs after supplementation with inorganic and organic selenium. Ann. Agric. Environ. Med. 2003;10(1):87–91. [PubMed] [Google Scholar]
- 54.Angstwurm M.W., Engelmann L., Zimmermann T., Lehmann C., Spes C.H., Abel P., Strauss R., Meier-Hellmann A., Insel R., Radke J., Schuttler J., Gartner R. Selenium in Intensive Care (SIC): results of a prospective randomized, placebo-controlled, multiple-center study in patients with severe systemic inflammatory response syndrome, sepsis, and septic shock. Crit. Care Med. 2007;35(1):118–126. doi: 10.1097/01.CCM.0000251124.83436.0E. [DOI] [PubMed] [Google Scholar]
- 55.MacFarquhar J.K., Broussard D.L., Melstrom P., Hutchinson R., Wolkin A., Martin C., Burk R.F., Dunn J.R., Green A.L., Hammond R., Schaffner W., Jones T.F. Acute selenium toxicity associated with a dietary supplement. Arch. Intern. Med. 2010;170(3):256–261. doi: 10.1001/archinternmed.2009.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Johnson V.J., Tsunoda M., Sharma R.P. Increased production of proinflammatory cytokines by murine macrophages following oral exposure to sodium selenite but not to seleno-L-methionine. Arch. Environ. Contam. Toxicol. 2000;39(2):243–250. doi: 10.1007/s002440010101. [DOI] [PubMed] [Google Scholar]
- 57.Colin S., Chinetti-Gbaguidi G., Staels B. Macrophage phenotypes in atherosclerosis. Immunol. Rev. 2014;262(1):153–166. doi: 10.1111/imr.12218. [DOI] [PubMed] [Google Scholar]
- 58.Chistiakov D.A., Bobryshev Y.V., Nikiforov N.G., Elizova N.V., Sobenin I.A., Orekhov A.N. Macrophage phenotypic plasticity in atherosclerosis: the associated features and the peculiarities of the expression of inflammatory genes. Int. J. Cardiol. 2015;184:436–445. doi: 10.1016/j.ijcard.2015.03.055. [DOI] [PubMed] [Google Scholar]
- 59.Jablonski K.A., Amici S.A., Webb L.M., Ruiz-Rosado Jde D., Popovich P.G., Partida-Sanchez S., Guerau-de-Arellano M. Novel markers to delineate murine M1 and M2 macrophages. PLoS One. 2015;10(12) doi: 10.1371/journal.pone.0145342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Reyes L., Hawkins C.L., Rayner B.S. Characterization of the cellular effects of myeloperoxidase-derived oxidants on H9c2 cardiac myoblasts. Arch. Biochem. Biophys. 2019;665:132–142. doi: 10.1016/j.abb.2019.03.004. [DOI] [PubMed] [Google Scholar]
- 61.Zhang X., Liu M.H., Qiao L., Zhang X.Y., Liu X.L., Dong M., Dai H.Y., Ni M., Luan X.R., Guan J., Lu H.X. Ginsenoside Rb1 enhances atherosclerotic plaque stability by skewing macrophages to the M2 phenotype. J. Cell Mol. Med. 2018;22(1):409–416. doi: 10.1111/jcmm.13329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jorch S.K., Kubes P. An emerging role for neutrophil extracellular traps in noninfectious disease. Nat. Med. 2017;23(3):279–287. doi: 10.1038/nm.4294. [DOI] [PubMed] [Google Scholar]
- 63.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018;18(2):134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 64.Qi H., Yang S., Zhang L. Neutrophil extracellular traps and endothelial dysfunction in atherosclerosis and thrombosis. Front. Immunol. 2017;8:928. doi: 10.3389/fimmu.2017.00928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Carroll L., Pattison D.I., Fu S., Schiesser C.H., Davies M.J., Hawkins C.L. Catalytic oxidant scavenging by selenium-containing compounds: reduction of selenoxides and N-chloramines by thiols and redox enzymes. Redox Biol. 2017;12:872–882. doi: 10.1016/j.redox.2017.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fuchs T.A., Abed U., Goosmann C., Hurwitz R., Schulze I., Wahn V., Weinrauch Y., Brinkmann V., Zychlinsky A. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 2007;176(2):231–241. doi: 10.1083/jcb.200606027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Stupin A., Cosic A., Novak S., Vesel M., Jukic I., Popovic B., Karalic K., Loncaric Z., Drenjancevic I. Reduced dietary selenium impairs vascular function by increasing oxidative stress in sprague-dawley rat aortas. Int. J. Environ. Res. Public Health. 2017;14(6) doi: 10.3390/ijerph14060591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ren H., Mu J., Ma J., Gong J., Li J., Wang J., Gao T., Zhu P., Zheng S., Xie J., Yuan B. Selenium inhibits homocysteine-induced endothelial dysfunction and apoptosis via activation of AKT. Cell. Physiol. Biochem. 2016;38(3):871–882. doi: 10.1159/000443041. [DOI] [PubMed] [Google Scholar]
- 69.Ruseva B., Atanasova M., Tsvetkova R., Betova T., Mollova M., Alexandrova M., Laleva P., Dimitrova A. Effect of selenium supplementation on redox status of the aortic wall in young spontaneously hypertensive rats. Oxid. Med. Cell Longev. 2015;2015:609053. doi: 10.1155/2015/609053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lymbury R., Tinggi U., Griffiths L., Rosenfeldt F., Perkins A.V. Selenium status of the Australian population: effect of age, gender and cardiovascular disease. Biol. Trace Elem. Res. 2008;126(Suppl 1):S1–S10. doi: 10.1007/s12011-008-8208-6. [DOI] [PubMed] [Google Scholar]
- 71.Dong H.L., Yuan N., Sun T., Dun A.S., Hou H.F. Effects of selenium supplement on atherogenesis of ApoE-knockout mice fed high fat diet. Zhonghua Xinxueguanbing Zazhi. 2016;44(3):244–249. doi: 10.3760/cma.j.issn.0253-3758.2016.03.011. [DOI] [PubMed] [Google Scholar]
- 72.Mehta U., Kang B.P., Kukreja R.S., Bansal M.P. Ultrastructural examination of rabbit aortic wall following high-fat diet feeding and selenium supplementation: a transmission electron microscopy study. J. Appl. Toxicol. 2002;22(6):405–413. doi: 10.1002/jat.877. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.