Abstract
Objectives
The HIV treatment cascade is a powerful framework for understanding progress from initial diagnosis to successful treatment. Data sources for cascades vary and often are based on clinical cohorts, population cohorts linked to clinics, or self-reported information. We use both biomarkers and self-reported data from a large population-based cohort of older South Africans to establish the first HIV cascade for this growing segment of the HIV-positive population and compare results using the different data sources.
Methods
Data came from the Health and Aging in Africa: A Longitudinal Study of an INDEPTH Community in South Africa (HAALSI) 2015 baseline survey of 5059 adults aged 40+ years. Dried blood spots (DBS) were screened for HIV, antiretroviral drugs and viral load. In-home surveys asked about HIV testing, diagnosis and antiretroviral therapy (ART) use. We calculated proportions and CIs for each stage of the cascade, conditional on attainment of the previous stage, using (1) biomarkers, (2) self-report and (3) both biomarkers and self-report, and compared with UNAIDS 90–90-90 targets.
Results
4560 participants had DBS results, among whom 1048 (23%) screened HIV-positive and comprised the denominator for each cascade. The biomarker cascade showed 63% (95% CI 60 to 66) on ART and 72% (95% CI 69 to 76) of those on ART with viral suppression. Self-reports underestimated testing, diagnosis and ART, with only 47% (95% CI 44 to 50) of HIV-positive individuals reporting ART use. The combined cascade indicated high HIV testing (89% (95% CI 87 to 91)), but lower knowledge of HIV-positive status (71% (95% CI 68 to 74)).
Conclusions
Older South Africans need repeated HIV testing and sustained ART to reach 90–90-90 targets. HIV cascades relying on self-reports are likely to underestimate true cascade attainment, and biomarkers provide substantial improvements to cascade estimates.
Introduction
The HIV treatment cascade, or care continuum, is a framework for understanding gaps in care and tracking the series of steps through which people living with HIV progress from initial diagnosis to achieving successful treatment. The treatment cascade is used worldwide for assessing progress in the scale-up of HIV testing and antiretroviral therapy (ART), and can be used to measure progress towards reaching the UNAIDS 90–90-90 targets for the year 2020: getting 90% of people with HIV to know their status, 90% of those diagnosed on sustained ART and 90% of those on ART to be virally suppressed.1 In South Africa, little is known about the coverage of HIV treatment among older HIV-positive adults. HIV testing and treatment initiatives have traditionally focused on adolescents and young adults, yet the older HIV-positive population is growing with the expansion of ART and interruption of HIV transmission in younger age groups.2–9 Older adults have been shown to be at risk of acquiring HIV and now present unique challenges to HIV care provision, such as rapid disease progression, slower ART response, complications from cognitive decline and a larger burden of comorbid chronic diseases.3,6,10–16 With a renewed focus needed on scaling up HIV programmes for older adults in South Africa, it is essential to understand gaps in care for this population.
Data sources for constructing cascades vary and often derive from clinical studies that are extrapolated to the larger population or include estimates that link different populations to define successive steps of the cascade.17,18 Outside of clinical data, population-based cascade analyses often rely on linkage to local clinic records to obtain estimates for testing and ART coverage.19,20 In South Africa, evidence from young adults shows that targets for HIV diagnoses and provision of ART are not being met. A population-based cascade estimate for adults ages 18–49 years in 2015 in Northwest Province showed that 58% of HIV-positive people were retained in care, and a 2017 study based on South Africa’s National Health Laboratory Service data across the country estimated that 58% of all HIV-positive people were linked to care and only 38% were on sustained ART.17–21 While viral suppression has been estimated to be low among the HIV-positive population overall in South Africa, some studies show evidence that progress towards the third 90 has been made, with high viral suppression found among those on ART. An estimate from 2012 in semi-urban South Africa showed that viral suppression was 27% among HIV-positive women and 18% among HIV-positive men age 18 and older, but found that 92% of those on ART had viral suppression (defined as <400 copies/mL).22 Similarly, a 2013 study in KwaZulu-Natal found 93% viral suppression among adults 15–59 years old on ART (defined as <1000 copies/mL); however, 2012–2014 data also from KwaZulu-Natal showed viral suppression (<400 copies/mL) was 71% among those on ART, or 34% of the HIV-positive population age 16 years and older.23,24
In this analysis, we constructed HIV care cascades from the first population-based cohort of older adults in southern Africa. We use self-reported survey data in conjunction with biomarker data for any step with a biomarker equivalent. Our primary aim was to develop a detailed characterisation of the treatment cascade for older adults, using both biomarkers and self-reported information about HIV testing and treatment, and to examine how the treatment cascade varies by demographics, mental health and social conditions. Our secondary aim was to compare the treatment cascade constructed from self-reported information and biomarkers, to demonstrate how cascades vary by source of information.
Methods
Data were from the Health and Aging in Africa: A Longitudinal Study of an INDEPTH Community in South Africa (HAALSI) 2015 baseline survey, which included a random sample of adults aged 40 years and older, and is nested within the Agincourt Health and Demographic Surveillance System in Mpumalanga Province, northeastern South Africa. There were 6281 men and women randomly selected for the study, and 5059 completed interviews for a response rate of 85.9%, while 391 (6%) were ineligible, 353 (6%) were not found, 48 (1%) were unable to participate and 430 (7%) refused. Those who refused were more likely to be younger, women and have higher education.25 The demographic profile of the study population is typical of rural South Africa, with low formal education and high unemployment, and some gaps in access to electricity and water. The study area covers 450 km2 and includes 31 villages and approximately 116 000 people. Six clinics, two health centres and three district hospitals make up the primary healthcare system.25 Face-to-face computer-assisted personal interviews (CAPIs) were conducted in the home with responses recorded on laptops. The survey asked questions about health and well-being, with measured performance tests of physical and cognitive function, and including a detailed module on HIV testing and treatment.25 Dried blood spots (DBS) were collected at the time of the interview and screened for HIV, ART and viral load. Ethical approval for HAALSI was obtained from the University of the Witwatersrand Human Research Ethics Committee (no. M141159), the Harvard T.H. Chan School of Public Health Office of Human Research Administration (no. 13–1608), and the Mpumalanga Provincial Research and Ethics Committee.
We calculated proportions and 95% CIs for each stage of the treatment cascade conditional on completion of the previous stage, starting from the HIV-positive population as defined by DBS screening. First, we examined the cascade using self-reported responses to these questions: “Have you ever been tested for HIV?”, “Do you know your HIV status?” and “Have you ever tested positive for HIV?”; and the following questions that were only asked of those who said they had tested positive for HIV: “Have you ever accessed an HIV treatment and care program?”, “Have you ever received ART?” and “Are you currently receiving ART?”. Second, we examined the cascade using DBS laboratory biomarker results for presence of ART and viral suppression. Third, we examined the combined cascade, where either self-report or biomarker evidence was positive for each stage. In the combined cascade, biomarker-measured positivity for ART was considered positive for all earlier cascade stages. DBS screening for ART was performed at the University of Cape Town to detect the presence of emtricitabine (FTC) or lamivudine (3TC), the second drugs of all standard three-drug regimens in first-line and second-line ART in South Africa. Presence of either drug at a concentration as low as 0.02 μg/mL was considered positive for ART. Viral suppression was defined as <100 copies/mL. In sensitivity analyses, we evaluated other thresholds for viral suppression at <1000 copies/mL and <400 copies/mL, and we evaluated the impact of considering everyone who was virally suppressed as positive for ART. Each cascade was compared with the UNAIDS 90–90-90 target.
We explored variations in the biomarker cascade across strata defined by key demographic and self-reported conditions obtained during the baseline interview, including age, sex, household wealth index, education level, marital status and depression score. We calculated adjusted predicted probabilities of taking ART among those who are HIV positive, and of being virally suppressed among those on ART, from logistic regression models and compared 95% CIs of estimates between groups. Household wealth index was calculated from household characteristics and ownership of household items, vehicles and livestock.26 Depression score was determined through an abbreviated eight-item Center for Epidemiologic Studies—Depression Scale questionnaire with dichotomous yes/no response options, where a score of 0–3 was considered low for depressive symptoms and a score of 4–8 was considered high for depressive symptoms.27
Results
The HAALSI baseline survey included 5059 participants, of whom 4707 (93%) consented to blood collection. There were 4582 individuals with blood test results (97% of those who consented), 22 of the HIV test results were indeterminate, and the remainder were missing due to insufficient sample or participants declining to give blood after consent. The proportion of individuals consenting to biomarker testing was nearly the same for women (93.4%) and men (92.4%), and was high for all age groups (91.1% for age 40–49, 92.2% for age 50–59, 94.7% for age 60–69, 95.0% for age 70–79, 91.4% for age 80+). The proportion consenting to blood collection was slightly higher for those who reported ever tested for HIV (94.2% vs 91.1%, χ2 p<0.001) and among those who reported being HIV positive (95.0% vs 92.8%, χ2 p=0.03).
We limited the analytical sample for this study to the 4560 with valid DBS HIV test results (90% of the baseline cohort) and defined the initial population for the cascades based on the 1048 (23.0%) with positive test results. A description of this population is shown in table 1. The HIV-positive population was similar to the HIV-negative population with respect to sex, but tended to be younger, more educated, less likely to be married and less wealthy (table 1).
Table 1.
Descriptive characteristics of the HAALSI baseline cohort members with valid HIV test results from dried blood spots
| HIV+ | HIV− | Total | ||
|---|---|---|---|---|
| N | 1048 | 3512 | 4560 | |
| Sex | Female | 565 (53.9%) | 1898 (54.0%) | 2463 (54.0%) |
| Age | 40–49 years | 310 (29.6%) | 504 (14.3%) | 814 (17.9%) |
| 50–59 years | 392 (37.4%) | 870 (24.8%) | 1262 (27.7%) | |
| 60–69 years | 244 (23.3%) | 959 (27.3%) | 1203 (26.4%) | |
| 70+ years | 102 (9.7%) | 1179 (33.6) | 1281 (28.1%) | |
| Education | None | 429 (40.9%) | 1660 (47.3) | 1089 (45.8%) |
| 1–7 years | 364 (34.7%) | 1225 (34.9%) | 1589 (34.9%) | |
| 8–11 years | 162 (15.5%) | 345 (9.8%) | 507 (11.1%) | |
| 12+ years | 89 (8.5%) | 272 (7.7%) | 361 (7.9%) | |
| Marriage | Currently married/cohabitating | 416 (39.7%) | 1936 (55.1%) | 2352 (51.6%) |
| Never married | 79 (7.5%) | 166 (4.7%) | 245 (5.4%) | |
| Separated/divorced | 212 (20.2%) | 373 (10.6%) | 585 (12.8%) | |
| Widowed | 340 (32.4%) | 1036 (29.5%) | 1376 (30.2%) | |
| Wealth index | Poorest fifth | 256 (24.4%) | 695 (19.8%) | 951 (20.9%) |
| Second fifth | 215 (20.5%) | 700 (19.9%) | 915 (20.1%) | |
| Third fifth | 215 (20.5%) | 679 (19.3%) | 894 (19.6%) | |
| Fourth fifth | 199 (19.0%) | 695 (19.8%) | 894 (19.6%) | |
| Wealthiest fifth | 163 (15.6%) | 743 (21.2%) | 906 (19.9%) | |
| Depression* | CES-D 8 score 0–3 | 953 (90.9%) | 3084 (87.8%) | 4037 (88.5%) |
| CES-D 8 score ≥4 | 84 (8.0%) | 351 (10.0%) | 435 (9.5%) | |
| Not assessed | 11 (1.0%) | 77 (2.2%) | 88 (1.9%) |
CES-D 8: Center for Epidemiologic Studies—Depression Scale with 8 items.27
Self-reported cascade
The cascade from the self-reported responses indicated that most of the HIV-positive population (81% (95% CI 78 to 83)) reported testing for HIV, but only around half of those who were DBS HIV-positive (51% (95% CI 48 to 54)) self-reported as being HIV positive. Among those who self-reported as HIV positive, nearly everyone reported being on treatment (95% (95% CI 93 to 97), leading to an estimate of 48% (95% CI 45 to 51) of all HIV-positive individuals currently on treatment in the self-report cascade (figure 1).
Figure 1.
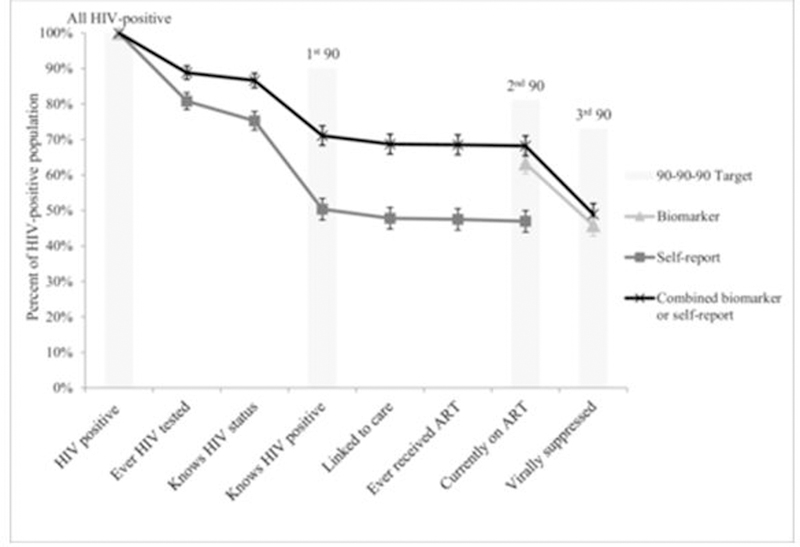
Stages of the HIV treatment cascade in older South African adults using (1) biomarkers, (2) self-report and (3) both biomarker and self-reported data, compared with the UNAIDS 90–90-90 targets. Error bars display 95% CIs for each proportion. ART, antiretroviral therapy.
Biomarker cascade
The cascade developed from the biomarker data showed a higher proportion on ART, with 63% of all persons measured as HIV positive (95% CI 60 to 66) screening positive for presence of ART in their blood and 73% (95% CI 69 to 76) of these, or 46% (95% CI: 43 to 49) overall, achieving viral suppression at <100 copies/mL, which falls substantially short of the UNAIDS target (figure 1). If we had considered viral suppression to indicate a positive biomarker for ART, the proportion on ART would increase substantially, from 63% to 71%, and the proportion suppressed among those on ART would increase slightly, from 73% to 75%.
Combined cascade
When combining self-report and biomarker data, we assumed that anyone with a positive biomarker for ART has tested positive for HIV in the past and is aware of their status. With this method, the cascade showed that the proportion who know they are HIV positive was 71% (95% CI 68 to 74), indicating that nearly three quarters of the HIV-positive population have either reported being HIV-positive or screened positive for being on ART. Similarly, using data from both sources showed that 68% (95% CI 65 to 71) of the HIV-positive population either reported being on ART or had screened positive for ART, compared with only 47% (95% CI 44 to 50) who self-reported current ART use. Viral suppression was lower than target levels and was detected for 72% (95% CI 68 to 75) of those who either reported current ART use or screened positive for ART. When changing the definition of viral suppression to <400 copies/mL or <1000 copies/mL, the proportion of those who reported or screened positive for ART who were virally suppressed increased to 81% and 88%, respectively, giving an overall estimate of 56% and 60% virally suppressed among the entire HIV-positive population for these more inclusive definitions.
Stratified cascades
In stratified analyses, we adjusted the biologic treatment cascades for age, but not for other demographic variables, which had little impact on our estimates. The cascade stratified by age group was unadjusted and this cascade showed slight variation by age group, wherein the youngest age group (age 40–49 years) had lower prevalence of ART (56% (95% CI 51 to 62) compared with 68% (95% CI 64 to 73) for age 50–59 years, 68% (95% CI 62 to 80) for age 60–69 years and 63% (95% CI 54 to 73) for age 70+ years) and slightly lower viral suppression (41% (95% CI 35 to 48) compared with 47% (95% CI 42 to 53) for age 50–59 years, 50% (95% CI 44 to 57) for age 60–69 years and 49% (95% CI 39 to 60) for age 70+ years; figure 2). We found cascades were similar for men and women and saw only minor differences in progression through other stratified biologic cascades (figure 2). The lowest wealth quintile had slightly lower attainment of steps in the cascade compared with the highest quintile (leading to 43% (95% CI 37 to 49) vs 49% (95% CI 41 to 57) of the HIV-positive population virally suppressed), and among those with high depression scores, 37% were virally suppressed (95% CI 24 to 53) compared with 47% (95% CI 41 to 54) among those with low depression score.
Figure 2.
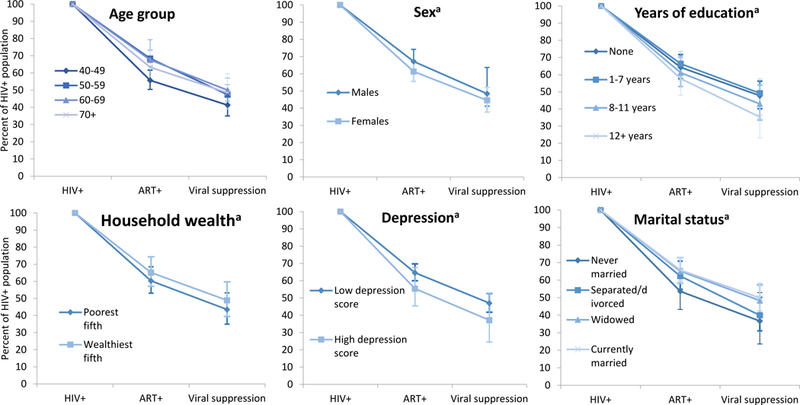
Biomarker cascades stratified by age group, sex, education level, household wealth quintile, depression score and marital status. ART+ is defined by the presence of 3TC or FTC in the dried blood spot. aAdjusted for age. ART, antiretroviral therapy.
Discussion
Globally, few countries have reached the UNAIDS 90–90-90 targets, and sustained ART remains a challenge.28,30 Our study underscores the need for improvements to increase diagnosis, provision of ART and viral suppression among the older HIV-positive population in South Africa. We found that overall viral suppression was low, at 49%, due to attrition that occurred throughout the cascade, which is consistent with previous studies of younger adults in low-income and middle-income countries.31
While we found evidence that having ever tested for HIV was common (nearly 90%), there is a critical decline from having an HIV test to awareness of HIV-positive status, which must be bridged in order to achieve the first 90 of the UNAIDS 90–90-90 target. Although the UNAIDS document uses ‘diagnosed’ and awareness of HIV-positive status interchangeably, we demonstrate that the question “Do you know your HIV status?”, when asked in an HIV-positive population, does not translate into acknowledgement of positive status or accessing care. There may be a number of reasons for the drop from “knows HIV status” to “knows HIV-positive”, such as (1) the participant knows they are HIV positive but does not report it due to stigma or shame given the interviews are face-to-face, (2) a participant may not have ever received a diagnosis but feels that “yes” is the correct or socially desirable response to knowing their status, (3) they may truly believe they “know” their status because of assumptions about exposure or lack or exposure, or (4) they have tested negative in the past and have recently acquired HIV. Repeat testing may improve knowledge of status, given the risk of acquisition of new HIV infections in this population.11 HIV-related stigma is an ongoing issue that is associated with ART non-initiation;32 thus, the 71% of the HIV-positive population that self-reported as HIV positive or had biomarker evidence of ART (presumed to signal awareness of status irrespective of self-reports) is likely a lower estimate for the total proportion of people who are actually aware of their positive status. Half of our study population self-reported their HIV-positive status, which is lower than some South African surveys of younger adults,23,24 and suggests that experienced or perceived HIV stigma persists for this population older adults.
A gap in care was also found at the viral suppression step for those currently on ART, and although the size of this gap varied greatly depending on the cut-off used for viral suppression, the 90–90-90 target was not met even with the least strict viral suppression definitions. When using comparable definitions, our estimates for viral suppression among those on ART were similar to or lower than estimates from studies of younger South African adults,22,24 which emphasises the need for treatment monitoring and improved ART adherence among the older adult population.
Estimates varied when using self-reported information only, which substantially underestimated the first two steps of the 90–90-90 target in comparison with the biomarker-based measures. The personal interaction during our CAPI may have enhanced social desirability biases. Additionally, ART self-report was only asked for those who reported testing positive for HIV, so self-report of ART could be higher if asked unconditionally. Combining self-report and biomarker information allowed us to gain a more nuanced understanding of the treatment cascade, relative to self-report or biomarkers alone. For reliable assessments of health system performance and monitoring of the 90–90-90 targets, self-report data are insufficient to be used alone without biomarker-based measurements, which are necessary for monitoring despite increased costs.
The stratified cascades showed only small differences and demonstrated that loss from care throughout the cascade is common for all segments of the older adult population. Our finding that the cascade was similar for men and women differs from studies of adults in younger age ranges in South Africa that demonstrate worse testing, linkage and mortality for men,19,22,33 and emphasises the need to target interventions towards both sexes. Both age and sex stratification of the cascade should be interpreted with caution, given the cross-sectional nature of this study, which ignores deaths that occur as people progress through the cascade.18,20 While our cross-sectional view shows that the viral suppression level is the same for men and women conditional on staying alive, if men have experienced more death throughout the cascade, we have an incomplete picture of their success in treatment. Similarly, given the more rapid disease progression and worse clinical outcomes that can occur in older HIV-positive individuals,12,14,15 our observation that the older age groups have higher viral suppression than those aged 40–49 years must also be interpreted as conditional on survival. Lastly, due to the geographically limited area covered by this population cohort, generalisability of the study findings should be considered restricted to high-HIV-prevalence rural South Africa populations.
Our population-based cohort with collection of survey and biomarker data allowed for an in-depth look at the treatment cascade for older South African adults, without relying on extrapolation from or linkage to clinical records, which can be incomplete. We demonstrate the advantage of using biomarkers in conjunction with self-reported data. We found that all demographic groups of this population require repeated HIV testing and improvements in linkage from testing to knowledge of HIV-positive status, and sustained ART to reach 90–90-90 targets. Interventions are needed to address these treatment needs of older South African adults. Future survey rounds are planned for the HAALSI study, and we plan to use this population-based longitudinal survey and biomarker data to examine the impact of death on the HIV treatment cascade as the cohort ages.
Key messages.
The HIV treatment cascade for older South Africans shows that UNAIDS 90–90-90 targets have not been met.
Report of HIV testing was high, but attrition throughout the cascade highlighted the need for repeat HIV testing and improved viral suppression on antiretroviral therapy.
Self-reported data drastically underestimated attainment at each step of the cascade compared with use of biomarker data.
Acknowledgments
We would like to thank the study team, participants and community of Bushbuckridge, South Africa, without whom this study would not have been possible. We would also like to thank the Division of Clinical Pharmacology Laboratory at the University of Cape Town for their assistance in performing tests of exposure to emtricitabine and lamivudine.
Funding The HAALSI study is funded by the National Institute on Aging (NIA) of the National Institutes of Health (NIH) (P01-AG041710), and is nested within the MRC/Wits Rural Public Health and Health Transitions Research Unit (Agincourt), funded by Wellcome Trust (058893/Z/99/A; 069683/Z/02/Z; 085477/Z/08/Z; 085477/B/08/Z) with important contributions from the University of the Witwatersrand, and the South African Medical Research Council. TB was supported by the Alexander von Humboldt Foundation through the Alexander von Humboldt Professor award, funded by the German Federal Ministry of Education and Research, as well as by NICHD of NIH (R01-HD084233), NIA of NIH (P01-AG041710), NIAID of NIH (R01-AI124389 and R01-AI112339) and FIC of NIH (D43-TW009775).
Footnotes
Handling editor Nicola Low
Competing interests None declared.
Patient consent for publication Not required.
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement Data are available in a public, open-access repository.
Request Permissions
If you wish to reuse any or all of this article please use the link below which will take you to the Copyright Clearance Center’s RightsLink service. You will be able to get a quick price and instant permission to reuse the content in many different ways.
References
- 1.Joint United National Programme on HIV/AIDS (UNAIDS). Ambitious treatment targets: writing the final chapter of the AIDS epidemic. Geneva, Switzerland, 2014. [Google Scholar]
- 2.Mahy M, Autenrieth CS, Stanecki K, et al. Increasing trends in HIV prevalence among people aged 50 years and older: evidence from estimates and survey data. AIDS 2014;28(Suppl 4):S453–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Negin J, Cumming RG. HIV infection in older adults in sub-Saharan Africa: extrapolating prevalence from existing data. Bull World Health Organ 2010;88:847–53. doi: 10.2471/BLT.10.076349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hontelez JAC, Lurie MN, Newell M-L, et al. Ageing with HIV in South Africa. AIDS 2011;25:1665–7. doi: 10.1097/QAD.0b013e32834982ea [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cornell M, Johnson LF, Schomaker M, et al. Age in antiretroviral therapy programmes in South Africa: a retrospective, multicentre, observational cohort study. The Lancet HIV 2015;2:e368–75. doi: 10.1016/S2352-3018(15)00113-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.UNAIDS. HIV and aging: a special supplement to the UNAIDS report on the global AIDS epidemic. Geneva: Joint UN Programme on HIV/AIDS, 2013. [Google Scholar]
- 7.Mutevedzi PC, Newell M-L. The changing face of the HIV epidemic in sub-Saharan Africa. Trop Med Int Health 2014;19:1015–28. doi: 10.1111/tmi.12344 [DOI] [PubMed] [Google Scholar]
- 8.Negin J, Bärnighausen T, Lundgren JD, et al. Aging with HIV in Africa: the challenges of living longer. AIDS 2012;26:S1–S5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gómez-Olivé FX, Angotti N, Houle B, et al. Prevalence of HIV among those 15 and older in rural South Africa. AIDS Care 2013;25:1122–8. doi: 10.1080/09540121.2012.750710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mills EJ, Rammohan A, Awofeso N. Ageing faster with AIDS in Africa. The Lancet 2011;377:1131–3. doi: 10.1016/S0140-6736(10)62180-0 [DOI] [PubMed] [Google Scholar]
- 11.Rosenberg M, Gomez-Olive FX, Rohr JK, et al. Sexual behaviors and HIV status: a population-based study among older adults in rural South Africa. J Acquir Immune Defic Syndr 2017;74:e9–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vinikoor MJ, Joseph J, Mwale J, et al. Age at antiretroviral therapy initiation predicts immune recovery, death, and loss to follow-up among HIV-infected adults in urban Zambia. AIDS Res Hum Retroviruses 2014;30:949–55. doi: 10.1089/aid.2014.0046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Greig J, Casas EC, O’Brien DP, et al. Association between older age and adverse outcomes on antiretroviral therapy: a cohort analysis of programme data from nine countries. AIDS 2012;26(Suppl1):S31–7. [DOI] [PubMed] [Google Scholar]
- 14.Mutevedzi PC, Lessells RJ, Rodger AJ, et al. Association of age with mortality and virological and immunological response to antiretroviral therapy in rural South African adults. PLoS ONE 2011;6:e21795. doi: 10.1371/journal.pone.0021795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maskew M, Brennan AT, MacPhail AP, et al. Poorer art outcomes with increasing age at a large public sector HIV clinic in Johannesburg, South Africa. J Int Assoc Physicians AIDS Care 2012;11:57–65.doi: 10.1177/1545109711421641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Negin J, Martiniuk A, Cumming RG, et al. Prevalence of HIV and chronic comorbidities among older adults. AIDS 2012;26:S55–S63. doi: 10.1097/QAD.0b013e3283558459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tavuka S, Brown AE, Pillay Y, et al. The continuum of HIV care in South Africa: implications for achieving the second and third UNAIDS 90–90-90 targets. AIDS 2017;31:545–52. [DOI] [PubMed] [Google Scholar]
- 18.Haber N, Pillay D, Porter K, et al. Constructing the cascade of HIV care: methods for measurement. Curr Opin HIV AIDS 2016;11:102–8. [DOI] [PubMed] [Google Scholar]
- 19.Bor J, Rosen S, Chimbindi N, et al. Mass HIV treatment and sex disparities in life expectancy: demographic surveillance in rural South Africa. PLoS Med 2015;12:e1001905. doi: 10.1371/journal.pmed.1001905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Haber N, Tanser F, Bor J, et al. From HIV infection to therapeutic response: a population-based longitudinal HIV cascade-of-care study in KwaZulu-Natal, South Africa. The Lancet HIV 2017;4:e223–30. doi: 10.1016/S2352-3018(16)30224-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lippman SA, Shade SB, El Ayadi AM, et al. Attrition and opportunities along the HIV care continuum: findings from a population-based sample, North West Province, South Africa. J Acquir Immune Defic Syndr 2016;73:91–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jean K, Puren A, Cutler E, et al. Level of viral suppression and the cascade of HIV care in a South African semi-urban setting in 2012. AIDS 2016;30:2107–16. doi: 10.1097/QAD.0000000000001155 [DOI] [PubMed] [Google Scholar]
- 23.Huerga H, Van Cutsem G, Ben Farhat J, et al. Progress towards the UNAIDS 90–90–90 goals by age and gender in a rural area of KwaZulu-Natal, South Africa: a household-based community cross-sectional survey. BMC Public Health 2018;18. doi: 10.1186/s12889-018-5208-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Iwuji CC, Orne-Gliemann J, Larmarange J, et al. Uptake of home-based HIV testing, linkage to care, and community attitudes about art in rural KwaZulu-Natal, South Africa: descriptive results from the first phase of the ANRS 12249 TASP cluster-randomised trial. PLoS Med 2016;13:e1002107. doi: 10.1371/journal.pmed.1002107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gómez-Olivé FX, Montana L, Wagner RG, et al. Cohort profile: health and ageing in Africa: a longitudinal study of an indepth community in South Africa (HAALSI). Int J Epidemiol 2018;47:689–90. doi: 10.1093/ije/dyx247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Filmer D, Pritchett LH. Estimating wealth effects without expenditure data—or tears: an application to educational enrollments in states of India. Demography 2001;38:115–32. [DOI] [PubMed] [Google Scholar]
- 27.Radloff LS. The CES-D scale: a self report depression scale for research in the general population. Appl Psychol Meas 1977;1:385–401. [Google Scholar]
- 28.Fox MP, Rosen S. Retention of adult patients on antiretroviral therapy in low-and middle-income countries: systematic review and meta-analysis 2008–2013. J Acquir Immune Defic Syndr 2015;69:98–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Granich R, Gupta S, Hall I, et al. Status and methodology of publicly available national HIV care continua and 90–90-90 targets: a systematic review. PLoS Med 2017;14:e1002253. doi: 10.1371/journal.pmed.1002253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Levi J, Raymond A, Pozniak A, et al. Can the UNAIDS 90–90-90 target be achieved? A systematic analysis of national HIV treatment cascades. BMJ Glob Health 2016;1:e000010. doi: 10.1136/bmjgh-2015-000010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kilmarx PH, Mutasa-Apollo T. Patching a leaky pipe: the cascade of HIV care. Curr Opin HIV AIDS 2013;8:59–64. [DOI] [PubMed] [Google Scholar]
- 32.Earnshaw VA, Bogart LM, Laurenceau J-P, et al. Internalized HIV stigma, art initiation and HIV-1 RNA suppression in South Africa: exploring avoidant coping as a longitudinal mediator. J Int AIDS Soc 2018;21:e25198. doi: 10.1002/jia2.25198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Camlin CS, Ssemmondo E, Chamie G, et al. Men “missing” from population-based HIV testing: insights from qualitative research. AIDS Care 2016;28:67–73. doi: 10.1080/09540121.2016.1164806 [DOI] [PMC free article] [PubMed] [Google Scholar]


