Abstract
Purpose
Nontraditional cardiovascular risk factors as apolipoprotein A (ApoA), apolipoprotein B (ApoB), and the proprotein convertase subtilisin/kexin type 9 (PCSK9) increase the prevalence of cardiovascular mortality in chronic kidney disease (CKD) or in end-stage renal disease (ESRD) through quantitative alterations. This review is aimed at establishing the biomarker (ApoA, ApoB, and PCSK9) level variations in uremic patients, to identify the studies showing the association between these biomarkers and the development of cardiovascular events and to depict the therapeutic options to reduce cardiovascular risk in CKD and ESRD patients.
Methods
We searched the electronic database of PubMed, Scopus, EBSCO, and Cochrane CENTRAL for studies evaluating apolipoproteins and PCSK9 in CKD and ESRD. Randomized controlled trials, observational studies (including case-control, prospective or retrospective cohort), and reviews/meta-analysis were included if reference was made to those keys and cardiovascular outcomes in CKD/ESRD.
Results
18 studies met inclusion criteria. Serum ApoA-I has been significantly associated with the development of new cardiovascular event and with cardiovascular mortality in ESRD patients. ApoA-IV level was independently associated with maximum carotid intima-media thickness (cIMT) and was a predictor for sudden cardiac death. The ApoB/ApoA-I ratio represents a strong predictor for coronary artery calcifications, cardiovascular mortality, and myocardial infarction in CKD/ESRD. Plasma levels of PCSK9 were not associated with cardiovascular events in CKD patients.
Conclusions
Although the “dyslipidemic status” in CKD/ESRD is not clearly depicted, due to different research findings, ApoA-I, ApoA-IV, and ApoB/ApoA-I ratio could be predictors of cardiovascular risk. Serum PCSK9 levels were not associated with the cardiovascular events in patients with CKD/ESRD. Probably in the future, the treatment of dyslipidemia in CKD/ESRD will be aimed at discovering new effective therapies on the action of these biomarkers.
1. Introduction
Worldwide, chronic kidney disease (CKD) represents a high public health priority [1]. Worldwide, over 2 million people require renal replacement therapy (hemodialysis (HD), peritoneal dialysis (PD), or kidney transplantation) to increase their survival rates [1, 2]. The prevalence of CKD has had an upward trend both in Europe and around the world, ESRD being merely the top of the iceberg [3]. CKD is an important cause of global mortality [1, 4]. The number of deaths caused by CKD has increased by 82.3% over the past two decades, being the third cause of the top 25 causes of deaths, after HIV/AIDS and diabetes [4].
Dyslipidemia in patients with impaired renal function is characterized by both qualitative changes in the cholesterol homeostasis and quantitative changes regarding the lipid parameters [5, 6]. Whereas in the general population dyslipidemia is described by the elevation of low-density lipoprotein cholesterol (LDL-C) [7], the progressive loss of renal function is associated with an increase of triglycerides, very low-density lipoprotein cholesterol (VLDL-C), and decreasing serum levels of the total cholesterol, HDL-C and LDL-C [5, 6].
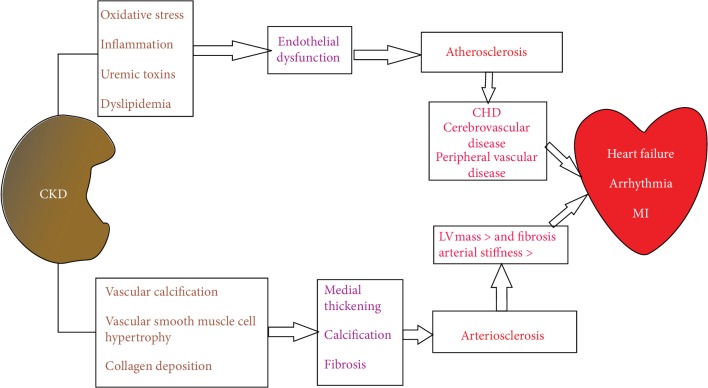
Cardiovascular mortality in dialysis patients is 10-20 times higher than that in the general population [1]. Cardiovascular death involves multiple pathogenic mechanisms: atherosclerosis, heart failure, and sudden death. Sudden death accounts for up to 25% of deaths from hemodialysis (HD) and occurs at the end of long-term HD and within the first 12 hours after HD [1]. Atherosclerosis and arteriosclerosis contribute to cardiovascular mortality in the general population [1, 8], while premature aging of the arteries, calcification, and arterial stiffness are characteristics of arteriosclerosis in chronic renal failure [1, 9]. Moreover, atherosclerosis affects arterial intima and is aggravated by CKD [1]. Several factors are involved in the pathogenesis of atherosclerosis and cardiovascular diseases: oxidative stress, inflammatory syndrome, malnutrition, arterial hypertension, endothelial dysfunction, vascular calcification, and dyslipidemia, both in the CKD and ESRD [7] (Figure 1).
Figure 1.
The pathophysiology of atherosclerosis and arteriosclerosis in patients with CKD. CHD: coronary heart disease; CKD: chronic kidney disease; LV: left ventricle; MI: myocardial infarction.
The common biomarkers involved in the evaluation of the “dyslipidemic status” in the general population and CKD/ESRD patients are total cholesterol, LDL-C, HDL-C, and triglycerides, for the assessment of CVD risk. In addition, other possible biomarkers are represented by apolipoproteins (ApoA, ApoB, and ApoB/ApoA-I ratio) or PCSK9.
2. Objectives
This review proposes (1) to identify the studies showing biomarker level modifications (serum PCSK9, apolipoprotein A, and apolipoprotein B) in “uremic milieu” and (2) to depict current evidence of the association between these biomarkers and the development of cardiovascular events (stroke, heart failure, coronary pathology, and cardiovascular mortality) and (3) proposes new therapeutic approaches to reduce cardiovascular risk in CKD or ESRD patients.
3. Method: Search Strategy
We searched the electronic database of PubMed, Scopus, EBSCO, and the Register of Controlled Trials (Cochrane CENTRAL) from 3 January 2018 to 30 December 2018 for studies that evaluated the apolipoprotein profile in patients with CKD and ESRD and its cardiovascular outcomes. The terms used for searching were “apolipoprotein A-I”, “apolipoprotein A-IV”, “apolipoprotein B”, “apolipoprotein B/apolipoprotein A-I ratio”, “PCSK9”, “end-stage renal disease”, “ESRD”, “chronic kidney failure”, “CKD”, “advanced CKD”, “hemodialysis”, and “peritoneal dialysis”. Relevant references in these articles were searched manually to identify possible additional studies [10]. Randomized controlled trials, observational studies, including case-control studies, prospective or retrospective cohort studies, reviews, and meta-analyses were included if reference was made to apolipoproteins and their cardiovascular outcomes in CKD/ESRD [10]. Case reports were excluded, and studies were selected by two independent reviewers by screening the title and abstract. In a second phase, the full articles which conformed to the selection criteria were obtained, the essential data was extracted independently, and the results were analysed [10]. Discrepancies were resolved by discussion and consensus, and duplicates were excluded both manually and through a reference manager software [10]. Of these, only 18 met the inclusion criteria (Table 1). For the selected studies, we reviewed the full-text article and additional relevant publications were added after screening the reference section.
Table 1.
Characteristics of the included studies for cardiovascular outcomes.
| Author | Study type | Apolipoprotein used | Outcomes | Population total | CKD patients | CKD stage | Dialysis type | Results |
|---|---|---|---|---|---|---|---|---|
| Kirmizis et al. [11] | Case-control study | ApoA-I ApoB/ApoA-I ratio |
Cardiovascular morbidity | 75 | 75 | G5D | HD | (i) In the ROC curve analysis, serum ApoA-I was shown to be inferior as a marker of cardiovascular morbidity, with a likelihood ratio of 2.8 (ii) On logistic regression analysis, the age- and sex-adjusted OR for the presence of CVD was 2.0 (95% CI: 1.6 to 2.4), when ApoB/ApoA-I ratio values above 1.13 were compared with values below this cut-off point (iii) For ApoB/ApoA-I ratio values above 1.13, the OR did not change essentially after controlling for various confounders: nonlipid risk factors (OR = 2; 95% CI: 1.7-2.3), Lp(a) (OR = 2; 95% CI: 1.7-2.2), or markers of inflammation (OR = 1.9; 95% CI: 1.5-2.3) |
|
| ||||||||
| Kim et al. [12] | Retrospective cross-sectional study | ApoB/ApoA-I ratio | Coronary artery calcification | 7780 | 7780 | G1-G3 | — | (i) In multivariate logistic regression analysis, the ApoB/ApoA-I ratio was significantly associated with an increased risk of coronary artery calcification in participants with normal kidney function (OR = 2.411, 95% CI: 1.224-4.748, p = 0.011), while in the participants with mild renal insufficiency, the ApoB/ApoA-I ratio was not associated with coronary artery calcification (OR = 1.074, 95% CI: 0.395-2.925, p = 0.888) |
|
| ||||||||
| Hung et al. [20] | Multicenter cross-sectional study | ApoA-I | Coronary heart disease | 995 | 995 | 5D | HD | (i) Univariate analysis revealed that ApoA-I was associated with CHD (ii) Multivariate logistic regression analysis showed that ApoA-I was associated with CHD (OR = 3.27, 95% CI: 1.96–5.43, p < 0.01) |
|
| ||||||||
| Cerezo et al. [21] | Prospective observational study | ApoA-I | New CV episodes | 331 | 331 | G3-G5 | Predialyzed | (i) In the ROC curve analyses, the ApoA-I concentrations were negatively associated with mortality, but with a lower level of significance (area below the curve = 0.372; p < 0.0001) (ii) The only parameter that was significantly associated with the development of new CV episodes was the concentration of ApoA-I (area below the curve = 0.410; p = 0.035) (iii) In a multivariate Cox model adjusted by confounders, the risk ratio (RR) for each 10 mg/dl of ApoA-I was 0.915, with 95% confidence intervals (CI) of 0.844 and 0.992 (p = 0.031) |
|
| ||||||||
| Honda et al. [22] | Prospective cohort study | ApoA-I | Composite cardiovascular events | 111 | 111 | G1-G5D | HD PD |
(i) ApoA-I was associated with composite CVD events (HR = 2.86, 95% CI: 1.75-4.5, p = 0.0002) (ii) ApoA-I did not predict CVD events |
|
| ||||||||
| Lamprea-Montealegre et al. [23] | Large multicenter cohort | ApoA-I ApoB/ApoA-I ratio |
Risk of coronary heart disease | 10137 | 1217 | G1-G4 | — | (i) CKD was associated with significantly higher concentrations of ApoB/ApoA-I ratios and significantly lower concentrations of ApoA-I (ii) ApoB/ApoA-I was associated with CHD risk (HR per one standard deviation = 1.22, 95% CI: 1.02-1.46) |
|
| ||||||||
| Cicero et al. [24] | Cohort study | ApoA-I | Arterial stiffness | 417 | 212 | G2-G3 | — | (i) In patients with CKD (G2-G3), the univariate analysis indicated that PWV was inversely related to ApoA-I (p < 0.05) (ii) In the stepwise multiple regression model that included all subjects (with normal function and CKD G2-G3), PWV was not associated with ApoA-I |
|
| ||||||||
| Zhan et al. [25] | Retrospective cohort | ApoA-I ApoB/ApoA-I ratio |
Cardiovascular events All-cause mortality |
860 | 860 | G5D | PD | (i) ApoA-I was correlated with all-cause mortality in model 2 (HR = 0.47, 95% CI: 0.25-0.89, p = 0.020) and model 3 (HR = 0.48, 95% CI: 0.24-0.94, p = 0.033) and with cardiovascular events in model 1 (HR = 0.47, 95% CI: 0.25-0.90, p = 0.022) and model 2 (HR = 0.39, 95% CI: 0.18-0.83, p = 0.015) (ii) In Cox regression analysis, after the adjustment in models, the ApoB/ApoA-I ratio was still associated with all-cause mortality (in model 3 HR = 1.60, 95% CI: 1.02-2.49, p = 0.040) and with cardiovascular events (in model 2: HR = 1.72, 95% CI: 1.05-2.81, p = 0.03 and in model 3: HR = 2.04, 95% CI: 1.21-3.44, p = 0.008) |
|
| ||||||||
| Sato et al. [26] | Prospective cohort | ApoA-I ApoB/ApoA-I ratio |
Cardiovascular disease- (CVD) related mortality | 1081 | 1081 | G5D | HD | (i) In the survival analyses, ApoA-I and the ApoB/ApoA-1 ratio were significantly related to all-cause and CVD-related mortality. Estimated survival curves by ApoA-I quartiles for all-cause and CVD-related mortality were significant (p = 0.001 and p = 0.001, respectively) (ii) In a multivariate Cox analysis, the ApoA-I (per 1-SD increase) was associated with all-cause mortality and CVD-related mortality (in model 2: HR = 0.75, 95% CI: 0.63–0.89, p = 0.001; HR = 0.77, 95% CI: 0.59–0.99, p = 0.04, respectively) (iii) In a multivariate Cox analysis, the ApoA-I (quartile IV versus quartile I) was associated with all-cause mortality and CVD-related mortality (in model 2: HR = 0.51, 95% CI: 0.32–0.81, p = 0.01; HR = 0.48, 95% CI: 0.24–0.98, p = 0.04, respectively) (iv) Survival curves by ApoB/ApoA-I ratio quartiles for all-cause and CVD-related mortality were significant (p = 0.001 and p = 0.02) (v) In a multivariate Cox analysis, the ApoB/ApoA-I ratio (per 1-SD increase) was associated with all-cause mortality and CVD-related mortality, even after adjustment in models (in model 3: HR = 1.16, 95% CI: 1.00–1.35, p = 0.046; HR = 1.38, 95% CI: 1.11–1.71, p = 0.004, respectively) (vi) In a multivariate Cox analysis, the ApoB/ApoA-I ratio (quartile IV versus quartile I) was associated with all-cause mortality and CVD-related mortality, even after adjustment in models (in model 3: HR = 1.65, 95% CI: 1.05–2.57, p = 0.03; HR = 2.56, 95% CI: 1.21–5.40, p = 0.01, respectively) |
|
| ||||||||
| Honda et al. [27] | Prospective cohort study | ApoA-I ApoB/ApoA-I ratio |
Death from all causes Composite CVD events |
412 | 412 | G5D | HD | (i) Quartiles of apolipoproteins were not associated with all-cause mortality (p > 0.05) (ii) Quartiles of ApoA-I were not associated with composite CVD events in models adjusted for age, sex, dialysis vintage, DM, history of CVD, and malnutrition (p > 0.05) (iii) ApoA-I was an independent risk factor in models adjusted for confounders including hs-CRP (HR = 0.62, 95% CI: 0.43-0.90, p < 0.05) (iv) Quartiles of ApoB/ApoA-I ratio was independently associated with CVD events in models adjusted with and without hs-CRP (HR = 2.21, 95% CI: 1.13-4.56, p < 0.05) and IL-6 (HR = 2.12, 95% CI: 1.09-4.33, p < 0.05) (v) Associations of apolipoproteins and ApoB/ApoA-I ratio with composite CVD events were also estimated in Cox hazards models of a 1-SD increase of variables (HR = 1.38, 95% CI: 1.04-1.85, p < 0.05) (vi) Each variable of ApoB/ApoA-I ratio was an independent biomarker of composite CVD events in this model adjusted for the time-varying covariates of HDL-C (HR = 5.80, 95% CI: 1.62-20.86, p < 0.05) and hs-CRP (HR = 5.52, 95% CI: 1.50-20.29, p < 0.05) (vii) The association of ApoB/ApoA-I ratio with composite CVD events disappeared when adjusted for IL-6 (p > 0.05) |
|
| ||||||||
| Bevc et al. [28] | Observational study | ApoA-I | Asymptomatic atherosclerosis (IMT, plaque occurrence, and number of plaques) | 91 | 91 | G5D | HD | (i) Multiple linear regression analysis of nontraditional risk factors showed no relationship between ApoA-I values and IMTc (p > 0.05), plaque occurrence (p > 0.05), and the number of plaques (p > 0.05) |
|
| ||||||||
| Kronenberg et al. [29] | Multicenter case-control study | ApoA-IV | Atherosclerotic complications | 454 | 227 | G1-G3 | — | (i) In the logistic regression analysis, ApoA-IV emerged as a significant and independent predictor for the presence of atherosclerotic events (OR = 0.92, 95% CI: 0.86–0.98, p = 0.011) |
|
| ||||||||
| Omori et al. [30] | Cross-sectional study | ApoA-IV | Cardiovascular disease Maximum cIMT |
116 | 116 | G5D | HD | (i) In a multivariable logistic regression analysis, after adjusting for confounders, high ApoA-IV concentration was associated with CVD and with maximum cIMT (OR = 0.24, 95% CI: 0.09–0.60, p < 0.005; OR = 0.33, 95% CI: 0.12–0.86, p < 0.05, respectively) (ii) In a stepwise multivariate regression analysis, A-IV concentrations were associated with maximum cIMT (p < 0.05) (iii) The serum ApoA-IV concentration was independently associated with maximum cIMT (adjusted r2 = 0.25) |
|
| ||||||||
| Kollerits et al. [31] | Post hoc analysis of prospective, randomized, controlled trial 4D | ApoA-IV | Death from all causes Death from cardiac causes Combined cardiac events Combined cerebrovascular events Combined cardiovascular events |
1224 | 1224 | G5D | HD | (i) At baseline, ApoA-IV was inversely associated with the prevalence of congestive heart failure (OR = 0.81 per 10 mg dl−1 increment in ApoA-IV, p < 0.001) (ii) At baseline, ApoA-IV was correlated with ECG abnormalities such as arrhythmia, atrial fibrillation/flutter, and right or left bundle branch block (iii) At baseline, associations between ApoA-IV and variables reflecting atherosclerotic disease were weaker than those for congestive heart failure (iv) Each 10 mg dl−1 increase in ApoA-IV concentration was associated with an 11% reduced risk of death during the observation period (p = 0.001) (v) A significant association between ApoA-IV and all-cause mortality was found in the nonwasting group (HR = 0.89, 95% CI: 0.84–0.96, p = 0.001) (vi) In patients with BMI > 23 kg m−2, there was a relationship between ApoA-IV concentrations and death from cardiac causes (HR = 0.88, 95% CI: 0.80–0.98, p = 0.02), sudden cardiac death (HR = 0.83, 95% CI: 0.72–0.95, p = 0.006), and combined cerebrovascular events (HR = 0.84, 95% CI: 0.73–0.96, p = 0.01) (vii) Atherogenic events (fatal and nonfatal myocardial infarction or cardiovascular interventions), which were included in the overall group with cardiac events, were not associated with ApoA-IV concentration (HR = 0.98, 95% CI: 0.92–1.05, p = 0.62) |
|
| ||||||||
| Luczak et al. [35] | Observational study | ApoA-IV | Formation of plaque | 125 | 74 | G1-G5 | — | (i) CKD and CVD groups revealed accumulation of two proteins: ApoA-IV and α-1-microglobulin (ii) The results showed that at least two processes differentially contribute to the plaque formation in CKD- and CVD-mediated atherosclerosis (iii) The downregulation and upregulation of ApoA-IV in CVD and CKD groups suggested that substantial differences exist in the efficacy of cholesterol transport in both groups of patients |
|
| ||||||||
| Holzmann et al. [43] | Large cohort | ApoB/ApoA-I ratio | Incidence of myocardial infarction | 142394 | 142394 | G1-G4 | — | (i) The ratio of ApoB/ApoA-I was a strong predictor of myocardial infarction, both among subjects with and without renal dysfunction (HR = 3.35, 95% CI: 2.25–4.91 and HR = 2.88, 95% CI: 2.54–3.26, p < 0.05, respectively) |
|
| ||||||||
| Rogacev et al. [6] | Cross-sectional observational CARE FOR HOMe Cross-sectional observational LURIC |
PCSK9 | (i) Acute myocardial infarction (ii) Surgical or interventional coronary/cerebrovascular/peripheral-arterial revascularization (iii) Stroke with symptoms > 24 hours (iv) Amputation above the ankle or death of any cause, cardiovascular death (v) Death immediately after intervention to treat CHD (vi) Fatal stroke (vii) Other causes of death due to CHD |
443 1450 |
443 1450 |
G1-G4 G1-G4 |
— | (i) Kaplan-Meier analysis demonstrated no significant association between tertiles of PCSK9 and CV outcomes (p = 0.62). Separate analyses stratified by statin intake did not yield different results (statin users: p = 0.367; statin nonusers: p = 0.834) (ii) In multivariate analyses, we adjusted for confounders; PCSK9 was not an independent predictor of CV events (p = 0.206) (iii) In Kaplan-Meier analysis, tertiles of PCSK9 were not associated with cardiovascular deaths (p = 0.729). Separate analyses stratified by statin intake did not yield different results (no statin: p = 0.772; statin: p = 0.611) |
|
| ||||||||
| Elewa et al. [61] | Cross-sectional observational study | PCSK9 | Cardiovascular risk | 134 | 134 | G1-G4 | — | (i) No relationship was observed between serum PCSK9 and cardiovascular risk |
4. Results and Discussion
Apolipoproteins A and B and the ApoB/ApoA-I ratio are predictors of cardiovascular outcomes and potential biomarkers for cardiovascular mortality in both the general population and CKD/ESRD patients [11, 12]. It is not currently clear whether these biomarkers represent cardiovascular risk factors or could help in CVD diagnosis and the setting of the therapeutic targets in CKD/ESRD patients. In a comprehensive review, Vlad et al. showed that lipoprotein(a) (Lp(a)), the genetic polymorphisms of apolipoprotein(a), apolipoprotein E (ApoE), and apolipoprotein B (ApoB) undergo modifications in uremic patients, being correlated with cardiovascular events [10]. Furthermore, it was pointed out that in ESRD patients, Lp(a) levels were independent risk factors for atherothrombosis and cardiovascular mortality, LMW apo(a) phenotype was the best predictor for coronary events, single nucleotide polymorphisms in ApoE gene increased the risk of cardiovascular events, and ApoB had a significant correlation with the value of carotid intima-media thickness and vascular stiffness [10].
Our search has led to several studies with different results and conclusions (Table 1), which has created confusion regarding the roles ApoA-I, ApoA-IV, ApoB/ApoA-I ratio, and PCSK9 within the CKD/ESRD framework. This lack of consistency could be caused by the methodology of different types of studies (cross-sectional/case-control studies), small numbers of patients in the study groups, different clinical and laboratory outcomes, lack of homogeneous criteria for inclusion/exclusion, different definitions of endpoints, various periods of follow-up, or different statistical approaches.
5. Apolipoprotein A-I
5.1. Background
Apolipoprotein A-I (ApoA-I) is secreted predominantly by the liver and intestine as lipid-free ApoA-I and constitutes approximately 70% of HDL protein, being required for the normal HDL biosynthesis [13]. ApoA-I levels are strongly associated with those of HDL-C [11]. ApoA-I binds to circulating phospholipids and forms pre-β HDL (lipid-poor nascent discoid HDL particles) [7]. ApoA-I is involved in the elimination of excess cholesterol in tissues, which it incorporates into HDL for direct, indirect, or reverse transport via LDL to the liver [11]. ApoA-I inhibits the expression of endothelial adhesion molecules such as intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1), while it prevents the production of monocyte chemoattractant protein-1 (MCP-1), which are critical steps for the production of reactive oxygen species (ROS) in the arterial wall [7]. Likewise, ApoA-I has anti-inflammatory properties, which may contribute to its cardioprotective role [14]. In addition, ApoA-I displays strong antioxidant properties [7] as well as numerous functions (Figure 2) [15–17].
Figure 2.
The biological functions of ApoA-I. In the liver, ApoA-I initiates the biogenesis of HDL and the lipid uptake and promotes cholesterol efflux. In the vascular endothelium, it maintains endothelial cell homeostasis. ApoE: apolipoprotein E; ABCA1: ATP-binding cassette transporters; LCAT: lecithin-cholesterol acyltransferase; SR-BI: scavenger receptor class B type I.
5.2. Apolipoprotein A-I in CKD and ESRD
ESRD is associated with a significant decrease in plasma ApoA-I and HDL-C [7, 18]. ApoA-I values in relation to ApoB values are used in estimating cardiovascular risk in patients with CKD/ESRD and CVD [19].
Thus, ESRD is associated with decreased levels of HDL-C and ApoA-I and may contribute to the atherogenic pathology [7]. In patients with CKD or ESRD, ApoA-I can efficiently evaluate the risk for cardiovascular disease [20], while its elevated level has been associated with a good survival rate [21].
5.3. Study Data
In a prospective cohort study conducted by Honda et al., the serum levels of ApoA-I were significantly decreased in patients with CKD 5D as compared to those with CKD stages 2-3 [22], in consent with the data reported by the ARIC study (Atherosclerosis Risk in Communities), in which CKD patients in stages 3-4 without coronary heart disease (CHD) had a low ApoA-I concentration [23].
5.3.1. Pro Studies
In a cross-sectional multicenter study enrolling 995 HD patients, Hung et al. found that ApoA-I was positively correlated with total cholesterol, HDL-C, blood urea (BUN), and serum albumin [20], but ApoA-I had a negative correlation with pulse wave velocity (PWV) [24].
ApoA-I was negatively associated with cardiovascular morbidity in both predialyzed CKD patients (area under the curve (AUC) = 0.372; p < 0.0001) [21] and HD patients [11]. Moreover, ApoA-I has been significantly associated with the development of a new cardiovascular event [21] and has had the strongest independent correlation for CHD, the cut-off value for ApoA-I being 216.2 mg/dl [20]. Therefore, Zhan et al. also identified an association between ApoA-I and cardiovascular events in PD patients [25]. In addition, the low level of ApoA-I and serum creatinine constituted significant predictors of coronary pathology [20].
ApoA-I was significantly associated with all-cause mortality and cardiovascular mortality in HD patients [26] and PD patients [25]. In CKD patients, Cerezo et al. revealed that high ApoA-I concentrations have been significantly associated with the development of new cardiovascular episodes and were negatively associated with mortality (but with a lower level of significance) [21].
5.3.2. Con Studies
Despite these findings, in an observational study that enrolled 412 HD patients, Honda et al. established that ApoA-I was not a risk factor for cardiovascular events [27]. Likewise, ApoA-I was correlated with cardiovascular events, but without any predictive strength in CKD patients (stages 2-5D) [22]. In an observational study with 91 HD patients, Bevc et al. revealed that ApoA-I did not correlate with carotid intima-media thickness (cIMT) [28]. Furthermore, in cohort CARE FOR HOMe (Cardiovascular and Renal Outcome in CKD 2–4 Patients—The Forth Homburg evaluation), which enrolled 443 patients, Rogacev et al. have revealed in a multivariate analysis after adjusting for confounders that ApoA-I levels were not associated with CV events (p = 0.483) [6].
6. Apolipoprotein A-IV
6.1. Background
Apolipoprotein A-IV (ApoA-IV) is a 46 kDa glycoprotein produced exclusively in the small intestine enterocytes during fat absorption and released into the lymph from the mesenteric duct, being incorporated into nascent chylomicrons [29–32]. In fasting plasma, ApoA-IV circulates as part of a lipid-poor, small HDL-like particle and ApoA-I free particles [29, 33]. ApoA-IV activates lecithin-cholesterol acyltransferase (LCAT) and modulates lipoprotein lipase (LPL) activation, favoring cholesteryl ester transfer from HDL to LDL, hence suggesting that ApoA-IV may behave like an antiatherogenic factor [29, 32]. ApoA-IV has antioxidant and antiatherogenic properties, and therefore, low levels of ApoA-IV increase the risk of CHD [31, 32] (Figure 3).
Figure 3.
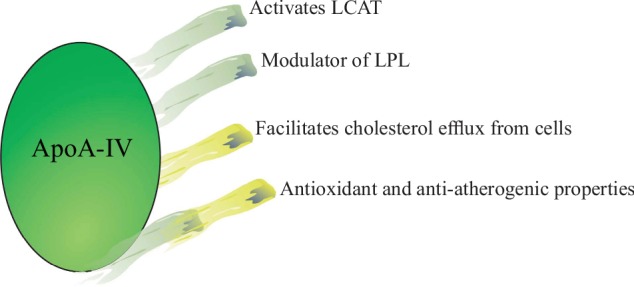
The roles of ApoA-IV. ApoA-IV has antioxidant and antiatherogenic functions. ApoA-IV activates LCAT and modulates LPL activation, favoring cholesteryl ester transfer from HDL to LDL. LCAT: lecithin-cholesterol acyltransferase; LPL: lipoprotein lipase.
6.2. Apolipoprotein A-IV in CKD and ESRD
ApoA-IV also plays a relevant role in the reverse cholesterol transport [30–34], which is affected in patients with CKD [34]. Few studies have investigated serum ApoA-IV in patients with CKD and have shown that the kidney plays a crucial part in its metabolism [30, 32]. Renal function parameters (GFR, creatinine, and BUN) were the most important determinants of serum ApoA-IV levels in patients with CKD [29]. Immunohistochemical studies have indicated that ApoA-IV is filtered in the glomerulus and is mostly reabsorbed by proximal tubular cells [33, 34]. ApoA-IV is significantly raised in HD and DP patients [29, 30, 32].
ApoA-IV begins to grow from the initial stages of CKD, becoming thus an early marker of renal failure [29]. Serum ApoA-IV is associated with the development of atherosclerotic lesions in HD patients and can be useful for estimating the cardiovascular risk [30]. Moreover, low levels of ApoA-IV have been validated as a risk predictor for all-cause mortality and sudden cardiac death, being adjusted by the nutritional status [31].
6.3. Study Data
ApoA-IV is a key link between the decreased GFR and the presence of cardiovascular events [29]. Kronenberg et al. revealed that ApoA-IV significantly increased with the decreasing glomerular filtration rate (GFR), especially in dialyzed patients [29]. Subjects with mild and moderate CKD [29] and ESRD (HD patients) [30], who developed atherosclerotic complications (carotid artery plaques and a low ankle-brachial index), had a decrease in ApoA-IV plasma concentrations [30] as compared to control participants (24.9 ± 8.7 versus 22.3 ± 7.7 mg/dl, p < 0.15 for mild-moderate CKD) [29]. Also, ApoA-IV had a 60% sensitivity, a 69% specificity, and a cut-off value of 321.92 μg/ml [30]. In addition, in a post hoc analysis performed in the Die Deutsche Diabetes Dialyse Studie (Study 4D), the scientists found the average ApoA-IV concentration of 49.8 ± 14.2 mg/dl about three times higher in HD patients than in the general population, which could not be influenced by statin administration [31].
6.3.1. Pro Studies
Patients with elevated ApoA-IV levels displayed a lower risk factor on coronary atherosclerosis and cardiovascular disease as compared to low ApoA-IV subjects [30]. In the post hoc 4D study, at baseline, ApoA-IV concentrations were closely associated with the congestive heart failure and with ECG changes (arrhythmia, atrial fibrillation, atrial flutter, left bundle branch block, or right bundle branch block) [31]. Kronenberg et al. identified that ApoA-IV was associated with atherosclerotic complications and each ApoA-IV increase of 1 mg/dl decreased odds ratio by 8% for these complications (p < 0.011) [29]. Likewise, Omori et al. indicated that the ApoA-IV level was independently associated with maximum carotid intima-media thickness (cIMT) and cardiovascular disease [30].
Furthermore, Luczak et al. have carried out a comparative proteomic analysis of plasma proteins isolated from 75 patients in different stages of renal disease, 25 patients with advanced cardiovascular disease, and 25 healthy volunteers [35]. A direct comparison between CKD and CVD groups revealed significant differences in the accumulation of 2 proteins: ApoA-IV and α-1-microglobulin [35]. These proteins individually contributed to the formation of atheroma plaque, yet the inflammatory process was more powerful in patients with CKD [35]. On the other hand, the stimulation or inhibition of ApoA-IV in CVD and CKD groups suggested differences in the cholesterol transport efficacy [35].
Following the body mass index (BMI) adjustment, in ESRD patients with BMI > 23 kg m2, ApoA-IV was associated with all-cause mortality, heart rate mortality, sudden cardiac death, cerebrovascular events, and cardiovascular disorders [31]. Also, Kollerits et al. showed that the increase of 10 mg/dl was associated with a reduced risk of death of 11% (p = 0.001) [31].
6.3.2. Con Studies
Contrary to these findings, in the same prospective randomized controlled trial, Kollerits et al. revealed that ApoA-IV did not impact upon the atherogenic risk: fatal myocardial infarction (p = 0.11) and nonfatal myocardial infarction (p = 0.14), fatal or nonfatal stroke (p = 0.18), or cardiovascular interventions (p = 0.62) [31]. Moreover, ApoA-IV did not associate with the cerebrovascular disease (p = 0.61) [31].
7. Apolipoprotein B and ApoB/ApoA-I Ratio
7.1. Background
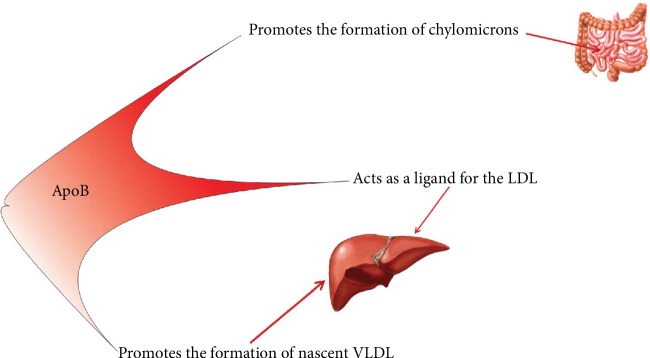
ApoB is a marker of dyslipidemia and is important in the binding of LDL particles to LDLR (receptor of LDL) for cellular absorption and degradation of LDL particles [11]. Apolipoprotein B (ApoB) is the primary protein component of very low-density lipoproteins (VLDL), intermediate-density lipoprotein (IDL), and LDL-C [11, 25, 26] and reflects the atherogenic particles from the body [11, 25, 26]. Other roles of ApoB are displayed in Figure 4 [36]. ApoB has two forms: ApoB-48 which is exclusively intestine secreted in chylomicrons and ApoB-100 which is only secreted by the liver in VLDL [37].
Figure 4.
The roles of ApoB. In the liver, ApoB promotes the formation of nascent VLDL and also is essential for the linking of LDL particles to LDLR for cellular absorption and degradation of LDL particles. In the intestine, ApoB stimulates the formation of chylomicrons. LDL: low-density lipoprotein; VLDL: very low-density lipoprotein.
In the general population, several clinical studies (e.g., AMORIS (Apolipoprotein-related MOrtality RISk) [38] or INTERHEART [39]) have shown that the ApoB/ApoA-I ratio is strongly correlated with cardiovascular events such as myocardial infarction and stroke [40].
7.2. Apolipoprotein B and ApoB/ApoA-I Ratio in CKD and ESRD
Patients with CKD (G1-G4) have elevated ApoB values [23, 24]. In HD patients, ApoB concentration is within the normal range, in contrast with PD patients, who have elevated ApoB levels (due to overproduction) [41]. HD patients display advanced atherosclerosis that is associated with nontraditional risk factors (ApoB) [11, 28]. The reduction of ApoB could be a critical risk marker for eccentric left ventricular remodeling and could be useful for cardiovascular risk stratification in PD patients [42].
In CKD patients, the ApoB/ApoA-I ratio reflects the cholesterol balance between atherogenic and antiatherogenic lipoprotein particles [12], and an increased ratio highlights the progression of atherosclerosis [25]. Moreover, the ApoB/ApoA-I ratio represents a strong predictor for coronary artery calcifications [12] and myocardial infarction in CKD or ESRD [43]. Also, ApoB/ApoA-I ratio measurement was significantly associated with all-cause mortality and cardiovascular mortality in HD and PD patients [25, 26].
7.3. Study Data
In the ARIC study (Atherosclerosis Risk in Communities), patients with CKD stages 3-4 recorded an important ApoB/ApoA-I ratio increment as compared to those without CKD [23].
7.3.1. Pro Studies
ApoB/ApoA-I ratio had a 100% sensitivity, a 77% specificity, and a cut-off value of 1.13 [11]. Besides, in the same case-control study conducted by Kirmizis et al., the ApoB/ApoA-I ratio was positively correlated with cardiovascular morbidity [11]. Moreover, in patients with stages 3-4 of CKD, the incidence of acute myocardial infarction was strongly associated with this ratio [43].
After adjustment for confounders, the ApoB/ApoA-I ratio was associated with cardiovascular events when serum levels exceeded the cut-off value, in patients with mild renal impairment [11], HD patients [27], and PD subjects [25]. Although the ApoB/ApoA-I ratio was associated with cardiovascular events, it displayed differences between the quartile models and the 1-SD increases, being influenced by interleukin-6 (IL-6) [27].
However, Sato et al. have found a substantial association of ApoB/ApoA-I ratio with all-cause and cardiovascular mortality in HD patients [26]. It appears that this ratio was significantly associated with all-cause mortality in PD patients [25].
7.3.2. Con Studies
In contrast, in an observational study, Kim et al. reported that ApoB/ApoA-I ratio was not associated with coronary artery calcification in patients with mild renal impairment [12]. Moreover, the ARIC study did not detect an association between the ApoB/ApoA-I ratio and the risk for coronary disease [23].
8. Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9)
8.1. Background
Proprotein convertase subtilisin/kexin type 9 (PCSK9) is a serine protease of the subtilase family [44, 45], secreted primarily by the liver [44–46] and containing 692 amino acids [46]. PCSK9 is an enzyme accepted as a new biomarker for the lipid metabolism, a novel therapeutic target for hypercholesterolemia, because the inhibition of PCSK9 may be one of the options for lowering cardiovascular risk [47–49].
It triggers the reduction of LDLR levels without affecting the LDLR mRNA (messenger ribonucleic acid). PCSK9 determines the degradation of LDLR [50] and inhibits receptor recycling in the hepatocyte membrane [6]. The human PCSK9 protein (hPCSK9) is synthesized mainly in the liver, the kidney, the small intestine [46], and the brain [44, 45] via sterol regulatory element-binding protein 2 (SREBP-2) regulation [46]. Sterol regulatory element-binding proteins (SREBPs) coordinate the synthesis and cellular uptake for cholesterol and fatty acids [51]. SREBP-2 is primarily responsible for the activation of genes involved in the cholesterol synthesis, as opposed to fatty acid synthesis [51]. Thus, by activating the SREBP-2 pathway, statins increase the level of PCSK9, limiting the efficiency of these drugs for lowering LDL cholesterol [46].
In the kidney, PCSK9 modulates the sodium absorption by degrading the epithelial sodium channel, and it also plays a part in the regulation of blood pressure [52]. PCSK9 has been identified in human pancreatic cells and does not modify endocrine pancreatic function [53]. Although adipocytes do not express PCSK9, they are rich in LDL and VLDL receptors, which play an important part in the hydrolysis of triglyceride-rich lipoproteins, and are helpful for fat storage in these cells [54]. In addition, PCSK9 is observed in carotid atherosclerotic lesions, especially in vascular smooth muscle cells [55]. Additional roles [56] are shown in Figure 5.
Figure 5.
The biological functions of PCSK9. LDLR: low-density lipoprotein receptor; VLDLRs: very low-density lipoprotein receptors.
The human PCSK9 gene is found on the human chromosome 1 and encodes the PCSK9 protein. The “gain-of-function” mutations of PCSK9 are associated with a rare form of autosomal dominant hypercholesterolemia (ADH), whereas “loss-of-function” mutations result in lowering cholesterol levels by reducing the CHD rate [46]. These genetic variants of PCSK9 affect both the plasma concentrations of PCSK9 and the serum level of LDL-C [49], thus becoming new targets for the treatment of hypercholesterolemia [57].
9. PCSK9 and CKD/ESRD
In patients with CKD, the available evidence for PCSK9 is insufficient, with very few observational studies and with a small number of patients. At the same time, there are no data on the use of PCSK9 inhibitors to them [57].
In HD patients, PCSK9 levels were close to control group levels [58], or a differentiation of serum PCSK9 values before and after HD was not identified [59]. In PD patients, PCSK9 levels were close to those measured in patients with nephrotic syndrome [60].
9.1. Study Data
9.1.1. Pro Studies
In a cross-sectional study comprising 134 diabetic patients with CKD, Elewa et al. identified that plasma PCSK9 was higher in patients with lipid lowering therapy, and the plasma PCSK9 values did not vary between patients with different eGFR or albuminuria categories [61]. During univariate analysis, Elewa et al. pointed out the significant positive correlation between the plasma PCSK9 level and the total iron binding capacity, vitamin E, renin, phosphaturia, and total serum cholesterol [61].
Also, Rogacev et al. have measured the serum level of PCSK9 in 2 independent cohorts: CARE FOR HOMe (Cardiovascular and Renal Outcome in CKD 2–4 Patients—The Forth Homburg evaluation), which included 443 patients, and the cohort LURIC (Ludwigshafen Risk and Cardiovascular Health Study) with 1450 patients [6], and they observed that plasma PCSK9 was poorly correlated with the total cholesterol, ApoB, and triglycerides [6].
9.1.2. Con Studies
Conflicting results were further reported by Rogacev et al. who found no significant correlation between PCSK9 and GRF in nonstatin users of the LURIC cohort (p = 0.733) [6]. In addition, in the same two independent cohorts, Rogacev et al. observed that plasma PCSK9 values were correlated neither with baseline GFR values nor with LDL-C [6]. Likewise, plasma levels of PCSK9 were not associated with cardiovascular events in patients with low renal function [6].
Although PCSK9 is a potential determinant of serum cholesterol, no relationship with early or current cardiovascular disease has been identified, and thus, it cannot be considered a cardiovascular risk factor in CKD or ESRD patients [61]. Furthermore, Kaplan-Meier analysis revealed that serum PCSK9 levels did not predict cardiovascular events in any cohort (CARE FOR HOMe p = 0.622; LURIC p = 0.729) [6].
10. Therapy Options in CKD/ESRD for Apolipoprotein and PCSK9
Treating dyslipidemia with statins and ezetimibe results in favorable effects for cardiovascular disease (CVD) prevention, in patients with moderate CKD [57]. This therapeutic strategy has not proven effective in HD patients as indicated in the cardiovascular outcomes of 4D, AURORA, and SHARP studies [7]. Furthermore, in CKD/ESRD patients treated with statin, PCSK9 concentration was higher compared to nonstatin subjects [6, 59, 61].
In patients with CKD/ESRD, the use of PPARα agonists is still controversial, and long-term safety and efficacy remain open to research [62]. By using niacin, besides having no additional benefit in patients with satisfactory control of LDL-C concentrations, tolerability was reduced [62]. ApoA-I mimetic is a new challenge for improving the lipid profile (in animal models), but clinical trials are still needed to confirm widespread use [62]. Although cholesteryl ester transfer protein (CETP) and cholesterol acyltransferase (ACAT) inhibitors have significantly improved HDL levels, results from major clinical trials have identified increased cardiovascular events [62].
Therefore, the need for an improvement of the lipid panel has emerged; new biomarkers (PCSK9) and new therapeutic strategies (monoclonal antibodies—evolocumab, alirocumab) are being further identified [57].
11. Key Points and Future Directions
The results of the studies focusing upon the association between apolipoproteins and CKD, after adjusting for cardiovascular events, suggest that supplementary mechanisms may be involved in addition to large vessel atherosclerosis. These include glomerulosclerosis, small vessel atherosclerosis, and direct toxic effects of apolipoproteins and lipids on the podocytes as well. The harmful effects are mediated by numerous mechanisms: expanded oxidative stress due to low levels of the apolipoprotein A1-enriched HDL fraction; the oxidized LDL-ApoB rich, local foam cell development; and the activation of inflammation [63].
The use of PCSK9 as a potential biomarker to identify patients with diabetic nephropathy who could benefit from anti-PCSK9 strategies and inhibition of PCSK9 could become an important treatment target in patients with CKD.
Patients with CKD should be enrolled in multicenter, randomized, double-blind trials (e.g., FOURIER, ODYSSEY) and closely monitored for the treatment efficacy against major cardiovascular events.
12. Conclusions
ESRD is associated with decreased levels of ApoA-I, with the increased levels ApoA-IV and ApoB, and with a high ApoB/ApoA-I ratio, but plasma PCSK9 levels are not associated with GFR decrease.
In patients with CKD/ESRD, even if there are the controversial results in the relationship between biomarkers and major cardiovascular events, ApoA-I, ApoA-IV, and ApoB/ApoA-I ratio are predictors of cardiovascular events. Nevertheless, these biomarkers could be useful for monitoring therapies with an impact upon cardiovascular morbimortality. Plasma PCSK9 levels are not associated with cardiovascular events in CKD or ESRD patients. However, circulating PSCK9 stands out as a promising biomarker for the diagnosis of dyslipidemia in patients with CVD and those affected by familial hypercholesterolemia.
In CKD patients, statins and ezetimibe can contribute to the prevention of CVD. The CETP, ACAT, and PCSK9 inhibitors have significantly improved HDL levels and reduced LDL-C. Also, ApoA-I mimetic is a new challenge for improving the lipid profile, but clinical trials are still needed to confirm widespread use.
Thus, in-depth studies are required on large cohorts of subjects along with the setting of clear targets of cardiovascular outcomes.
Abbreviations
- CKD:
Chronic kidney disease
- ESRD:
End-stage renal disease
- HD:
Hemodialysis
- PD:
Peritoneal dialysis
- ApoA:
Apolipoprotein A
- GFR:
Glomerular filtration rate
- PCSK9:
Proprotein convertase subtilisin/kexin type 9
- CVD:
Cardiovascular disease
- CHD:
Coronary heart disease
- cIMT:
Carotid intima-media thickness.
Contributor Information
Liliana Foia, Email: lilifoia@yahoo.co.uk.
Vasilica Toma, Email: vasilicatoma40@yahoo.com.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Authors' Contributions
Cristiana-Elena Vlad, Liliana Foia, and Laura Florea contributed equally to this work.
References
- 1.Ortiz A., Covic A., Fliser D., et al. Epidemiology, contributors to, and clinical trials of mortality risk in chronic kidney failure. The Lancet. 2014;383(9931):1831–1843. doi: 10.1016/S0140-6736(14)60384-6. [DOI] [PubMed] [Google Scholar]
- 2.Eggers P. W. Has the incidence of end-stage renal disease in the USA and other countries stabilized? Current Opinion in Nephrology and Hypertension. 2011;20(3):241–245. doi: 10.1097/MNH.0b013e3283454319. [DOI] [PubMed] [Google Scholar]
- 3.Couser W. G., Riella M. C. World Kidney Day 2011: protect your kidneys, save your heart. Nephron Clinical Practice. 2011;117(3):I–IV. doi: 10.1159/000323547. [DOI] [PubMed] [Google Scholar]
- 4.Lozano R., Naghavi M., Foreman K., et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. The Lancet. 2012;380(9859):2095–2128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vaziri N. D. Dyslipidemia of chronic renal failure: the nature, mechanisms, and potential consequences. American Journal of Physiology. Renal Physiology. 2006;290(2):F262–F272. doi: 10.1152/ajprenal.00099.2005. [DOI] [PubMed] [Google Scholar]
- 6.Rogacev K. S., Heine G. H., Silbernagel G., et al. PCSK9 plasma concentrations are independent of GFR and do not predict cardiovascular events in patients with decreased GFR. PLoS One. 2016;11(1, article e0146920) doi: 10.1371/journal.pone.0146920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moradi H., Vaziri N. D., Kashyap M. L., Said H. M., Kalantar-Zadeh K. Role of HDL dysfunction in end-stage renal disease: a double-edged sword. Journal of Renal Nutrition. 2013;23(3):203–206. doi: 10.1053/j.jrn.2013.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.London G. M., Drueke T. B. Atherosclerosis and arteriosclerosis in chronic renal failure. Kidney International. 1997;51(6):1678–1695. doi: 10.1038/ki.1997.233. [DOI] [PubMed] [Google Scholar]
- 9.Briet M., Boutouyrie P., Laurent S., London G. M. Arterial stiffness and pulse pressure in CKD and ESRD. Kidney International. 2012;82(4):388–400. doi: 10.1038/ki.2012.131. [DOI] [PubMed] [Google Scholar]
- 10.Vlad C., Burlacu A., Florea L., et al. A comprehensive review on apolipoproteins as nontraditional cardiovascular risk factors in end-stage renal disease: current evidence and perspectives. International Urology and Nephrology. 2019;51(7):1173–1189. doi: 10.1007/s11255-019-02170-w. [DOI] [PubMed] [Google Scholar]
- 11.Kirmizis D., Koutoupa E., Tsiandoulas A., et al. Serum lipid profile constituents as markers of cardiovascular morbidity in patients on chronic hemodialysis. Biomarker Insights. 2007;1:185–192. [PMC free article] [PubMed] [Google Scholar]
- 12.Kim S. H., Oh D., Jung K. S., et al. The association between the apolipoprotein B/A-I ratio and coronary calcification may differ depending on kidney function in a healthy population. PLoS One. 2017;12(9, article e0185522) doi: 10.1371/journal.pone.0185522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kosmas C. E., Silverio D., Sourlas A., Garcia F., Montan P. D., Guzman E. Primary genetic disorders affecting high density lipoprotein (HDL) Drugs in Context. 2018;7, article 212546:1–11. doi: 10.7573/dic.212546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Umemoto T., Han C. Y., Mitra P., et al. Apolipoprotein AI and high-density lipoprotein have anti-inflammatory effects on adipocytes via cholesterol transporters: ATP-binding cassette A-1, ATP-binding cassette G-1, and scavenger receptor B-1. Circulation Research. 2013;112(10):1345–1354. doi: 10.1161/CIRCRESAHA.111.300581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chroni A., Liu T., Gorshkova I., et al. The central helices of ApoA-I can promote ATP-binding cassette transporter A1 (ABCA1)-mediated lipid efflux. Amino acid residues 220-231 of the wild-type ApoA-I are required for lipid efflux in vitro and high density lipoprotein formation in vivo. The Journal of Biological Chemistry. 2003;278(9):6719–6730. doi: 10.1074/jbc.M205232200. [DOI] [PubMed] [Google Scholar]
- 16.Liadaki K. N., Liu T., Xu S., et al. Binding of high density lipoprotein (HDL) and discoidal reconstituted HDL to the HDL receptor scavenger receptor class B type I. Effect of lipid association and APOA-I mutations on receptor binding. The Journal of Biological Chemistry. 2000;275(28):21262–21271. doi: 10.1074/jbc.M002310200. [DOI] [PubMed] [Google Scholar]
- 17.Filou S., Lhomme M., Karavia E. A., et al. Distinct roles of apolipoproteins A1 and E in the modulation of high-density lipoprotein composition and function. Biochemistry. 2016;55(27):3752–3762. doi: 10.1021/acs.biochem.6b00389. [DOI] [PubMed] [Google Scholar]
- 18.Vaziri N. D. Lipotoxicity and impaired high density lipoprotein-mediated reverse cholesterol transport in chronic kidney disease. Journal of Renal Nutrition. 2010;20(5):S35–S43. doi: 10.1053/j.jrn.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 19.Mühlberger I., Mönks K., Fechete R., et al. Molecular pathways and crosstalk characterizing the cardiorenal syndrome. OMICS. 2012;16(3):105–112. doi: 10.1089/omi.2011.0121. [DOI] [PubMed] [Google Scholar]
- 20.Hung S. Y., Liou H. H., Ger L. P., et al. Clustering of unconventional cardiovascular risk factors among Taiwanese hemodialysis patients. American Journal of Nephrology. 2009;30(3):222–231. doi: 10.1159/000218105. [DOI] [PubMed] [Google Scholar]
- 21.Cerezo I., Fernández N., Romero B., Fernández-Carbonero E., Hernández-Gallego R., Caravaca F. Prognostic value of apolipoproteins A and B in the clinical course of patients with chronic kidney disease previous to dialysis. Nefrología. 2009;29(6):540–547. doi: 10.3265/Nefrologia.2009.29.6.5600.en.full. [DOI] [PubMed] [Google Scholar]
- 22.Honda H., Hirano T., Ueda M., et al. High-density lipoprotein subfractions and their oxidized subfraction particles in patients with chronic kidney disease. Journal of Atherosclerosis and Thrombosis. 2016;23(1):81–94. doi: 10.5551/jat.30015. [DOI] [PubMed] [Google Scholar]
- 23.Lamprea-Montealegre J. A., Sharrett A. R., Matsushita K., Selvin E., Szklo M., Astor B. C. Chronic kidney disease, lipids and apolipoproteins, and coronary heart disease: the ARIC study. Atherosclerosis. 2014;234(1):42–46. doi: 10.1016/j.atherosclerosis.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 24.Cicero A. F. G., Kuwabara M., Johnson R., et al. LDL-oxidation, serum uric acid, kidney function and pulse-wave velocity: data from the Brisighella Heart Study cohort. International Journal of Cardiology. 2018;261:204–208. doi: 10.1016/j.ijcard.2018.03.077. [DOI] [PubMed] [Google Scholar]
- 25.Zhan X., Chen Y., Yan C., et al. Apolipoprotein B/apolipoprotein A1 ratio and mortality among incident peritoneal dialysis patients. Lipids Health Dis. 2018;17(1):p. 117. doi: 10.1186/s12944-018-0771-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sato Y., Fujimoto S., Toida T., et al. Apoprotein B/apoprotein A-1 ratio and mortality among prevalent dialysis patients. Clinical Journal of the American Society of Nephrology. 2016;11(5):840–846. doi: 10.2215/CJN.09830915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Honda H., Hirano T., Ueda M., et al. Associations among apolipoproteins, oxidized high-density lipoprotein and cardiovascular events in patients on hemodialysis. PLoS One. 2017;12(5, article e0177980) doi: 10.1371/journal.pone.0177980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bevc S., Hojs R., Ekart R., Hojs-Fabjan T. Atherosclerosis in hemodialysis patients: traditional and nontraditional risk factors. Acta Dermatovenerologica Alpina Panonica et Adriatica. 2006;15(4):151–157. [PubMed] [Google Scholar]
- 29.Kronenberg F., Kuen E., Ritz E., et al. Apolipoprotein A-IV serum concentrations are elevated in patients with mild and moderate renal failure. Journal of the American Society of Nephrology. 2002;13(2):461–469. doi: 10.1681/ASN.V132461. [DOI] [PubMed] [Google Scholar]
- 30.Omori M., Watanabe M., Matsumoto K., Honda H., Hattori H., Akizawa T. Impact of serum apolipoprotein A-IV as a marker of cardiovascular disease in maintenance hemodialysis patients. Therapeutic Apheresis and Dialysis. 2010;14(3):341–348. doi: 10.1111/j.1744-9987.2010.00809.x. [DOI] [PubMed] [Google Scholar]
- 31.Kollerits B., Krane V., Drechsler C., et al. Apolipoprotein A-IV concentrations and clinical outcomes in haemodialysis patients with type 2 diabetes mellitus - a post hoc analysis of the 4D Study. Journal of Internal Medicine. 2012;272(6):592–600. doi: 10.1111/j.1365-2796.2012.02585.x. [DOI] [PubMed] [Google Scholar]
- 32.Stangl S., Kollerits B., Lamina C., et al. Association between apolipoprotein A-IV concentrations and chronic kidney disease in two large population-based cohorts: results from the KORA studies. Journal of Internal Medicine. 2015;278(4):410–423. doi: 10.1111/joim.12380. [DOI] [PubMed] [Google Scholar]
- 33.Lingenhel A., Lhotta K., Neyer U., et al. Role of the kidney in the metabolism of apolipoprotein A-IV: influence of the type of proteinuria. Journal of Lipid Research. 2006;47(9):2071–2079. doi: 10.1194/jlr.M600178-JLR200. [DOI] [PubMed] [Google Scholar]
- 34.Kronenberg F. Emerging risk factors and markers of chronic kidney disease progression. Nature Reviews Nephrology. 2009;5(12):677–689. doi: 10.1038/nrneph.2009.173. [DOI] [PubMed] [Google Scholar]
- 35.Luczak M., Formanowicz D., Pawliczak E., Wanic-Kossowska M., Wykretowicz A., Figlerowicz M. Chronic kidney disease-related atherosclerosis - proteomic studies of blood plasma. Proteome Science. 2011;9(1):p. 25. doi: 10.1186/1477-5956-9-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kypreos K. E., Bitzur R., Karavia E. A., Xepapadaki E., Panayiotakopoulos G., Constantinou C. Pharmacological management of dyslipidemia in atherosclerosis: limitations, challenges, and new therapeutic opportunities. Angiology. 2019;70(3):197–209. doi: 10.1177/0003319718779533. [DOI] [PubMed] [Google Scholar]
- 37.Ramasamy I. Update on the molecular biology of dyslipidemias. Clinica Chimica Acta. 2016;454:143–185. doi: 10.1016/j.cca.2015.10.033. [DOI] [PubMed] [Google Scholar]
- 38.Walldius G., Aastveit A. H., Jungner I. Stroke mortality and the apoB/apoA-I ratio: results of the AMORIS prospective study. Journal of Internal Medicine. 2006;259(3):259–266. doi: 10.1111/j.1365-2796.2005.01610.x. [DOI] [PubMed] [Google Scholar]
- 39.Yusuf S., Hawken S., Ounpuu S., et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. The Lancet. 2004;364(9438):937–952. doi: 10.1016/S0140-6736(04)17018-9. [DOI] [PubMed] [Google Scholar]
- 40.Tudor M. - N., Mitrea A., Georgiana Popa S., Zaharie S., Moţa M., Mola E. Apolipoproteins: good markers for cardiovacular risk in patients with chronic kidney disease and dyslipidemia. Romanian Journal of Diabetes Nutrition and Metabolic Diseases. 2014;21(3):185–191. doi: 10.2478/rjdnmd-2014-0023. [DOI] [Google Scholar]
- 41.Mikolasevic I., Žutelija M., Mavrinac V., Orlic L. Dyslipidemia in patients with chronic kidney disease: etiology and management. International Journal of Nephrology and Renovascular Disease. 2017;10:35–45. doi: 10.2147/IJNRD.S101808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ye M., Liu Y., Wang H., et al. Serum apolipoprotein B is inversely associated with eccentric left ventricular hypertrophy in peritoneal dialysis patients. International Urology and Nephrology. 2018;50(1):155–165. doi: 10.1007/s11255-017-1737-1. [DOI] [PubMed] [Google Scholar]
- 43.Holzmann M. J., Jungner I., Walldius G., et al. Dyslipidemia is a strong predictor of myocardial infarction in subjects with chronic kidney disease. Annals of Medicine. 2012;44(3):262–270. doi: 10.3109/07853890.2010.532153. [DOI] [PubMed] [Google Scholar]
- 44.Blanchard V., Khantalin I., Ramin-Mangata S., Chémello K., Nativel B., Lambert G. PCSK9: from biology to clinical applications. Pathology. 2019;51(2):177–183. doi: 10.1016/j.pathol.2018.10.012. [DOI] [PubMed] [Google Scholar]
- 45.Seidah N. G., Benjannet S., Wickham L., et al. The secretory proprotein convertase neural apoptosis-regulated convertase 1 (NARC-1): liver regeneration and neuronal differentiation. Proceedings of the National Academy of Sciences of the United States of America. 2003;100(3):928–933. doi: 10.1073/pnas.0335507100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tavori H., Fan D., Blakemore J. L., et al. Serum proprotein convertase subtilisin/kexin type 9 and cell surface low-density lipoprotein receptor: evidence for a reciprocal regulation. Circulation. 2013;127(24):2403–2413. doi: 10.1161/CIRCULATIONAHA.113.001592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bulbul M. C., Dagel T., Afsar B., et al. Disorders of lipid metabolism in chronic kidney disease. Blood Purification. 2018;46(2):144–152. doi: 10.1159/000488816. [DOI] [PubMed] [Google Scholar]
- 48.Bergeron N., Phan B. A., Ding Y., Fong A., Krauss R. M. Proprotein convertase subtilisin/kexin type 9 inhibition: a new therapeutic mechanism for reducing cardiovascular disease risk. Circulation. 2015;132(17):1648–1666. doi: 10.1161/CIRCULATIONAHA.115.016080. [DOI] [PubMed] [Google Scholar]
- 49.Nozue T. Lipid lowering therapy and circulating PCSK9 concentration. Journal of Atherosclerosis and Thrombosis. 2017;24(9):895–907. doi: 10.5551/jat.RV17012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Badescu C., Rezus E., Badescu L., Dima N., Rezus C. New drugs for lowering LDL-cholesterol. Medical Surgical Journal. 2016;120(3):485–490. [PubMed] [Google Scholar]
- 51.Madison B. B. Srebp2: a master regulator of sterol and fatty acid synthesis. Journal of Lipid Research. 2016;57(3):333–335. doi: 10.1194/jlr.C066712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sharotri V., Collier D. M., Olson D. R., Zhou R., Snyder P. M. Regulation of epithelial sodium channel trafficking by proprotein convertase subtilisin/kexin type 9 (PCSK9) The Journal of Biological Chemistry. 2012;287(23):19266–19274. doi: 10.1074/jbc.M112.363382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mbikay M., Sirois F., Mayne J., et al. PCSK9-deficient mice exhibit impaired glucose tolerance and pancreatic islet abnormalities. FEBS Letters. 2010;584(4):701–706. doi: 10.1016/j.febslet.2009.12.018. [DOI] [PubMed] [Google Scholar]
- 54.Roubtsova A., Munkonda M. N., Awan Z., et al. Circulating proprotein convertase subtilisin/kexin 9 (PCSK9) regulates VLDLR protein and triglyceride accumulation in visceral adipose tissue. Arteriosclerosis, Thrombosis, and Vascular Biology. 2011;31(4):785–791. doi: 10.1161/ATVBAHA.110.220988. [DOI] [PubMed] [Google Scholar]
- 55.Ferri N., Tibolla G., Pirillo A., et al. Proprotein convertase subtilisin kexin type 9 (PCSK9) secreted by cultured smooth muscle cells reduces macrophages LDLR levels. Atherosclerosis. 2012;220(2):381–386. doi: 10.1016/j.atherosclerosis.2011.11.026. [DOI] [PubMed] [Google Scholar]
- 56.Mombelli G., Castelnuovo S., Pavanello C. Potential of PCSK9 as a new target for the management of LDL cholesterol. Research Reports in Clinical Cardiology. 2015;6:73–86. doi: 10.2147/rrcc.s52961. [DOI] [Google Scholar]
- 57.Pavlakou P., Liberopoulos E., Dounousi E., Elisaf M. PCSK9 in chronic kidney disease. International Urology and Nephrology. 2017;49(6):1015–1024. doi: 10.1007/s11255-017-1505-2. [DOI] [PubMed] [Google Scholar]
- 58.Konarzewski M., Szolkiewicz M., Sucajtys-Szulc E., et al. Elevated circulating PCSK-9 concentration in renal failure patients is corrected by renal replacement therapy. American Journal of Nephrology. 2014;40(2):157–163. doi: 10.1159/000365935. [DOI] [PubMed] [Google Scholar]
- 59.Abujrad H., Mayne J., Ruzicka M., et al. Chronic kidney disease on hemodialysis is associated with decreased serum PCSK9 levels. Atherosclerosis. 2014;233(1):123–129. doi: 10.1016/j.atherosclerosis.2013.12.030. [DOI] [PubMed] [Google Scholar]
- 60.Jin K., Park B. S., Kim Y. W., Vaziri N. D. Plasma PCSK9 in nephrotic syndrome and in peritoneal dialysis: a cross- sectional study. American Journal of Kidney Diseases. 2014;63(4):584–589. doi: 10.1053/j.ajkd.2013.10.042. [DOI] [PubMed] [Google Scholar]
- 61.Elewa U., Fernández-Fernández B., Mahillo-Fernández I., et al. PCSK9 in diabetic kidney disease. European Journal of Clinical Investigation. 2016;46(9):779–786. doi: 10.1111/eci.12661. [DOI] [PubMed] [Google Scholar]
- 62.Khoueiry G., Abdallah M., Saiful F., et al. High-density lipoprotein in uremic patients: metabolism, impairment, and therapy. International Urology and Nephrology. 2014;46(1):27–39. doi: 10.1007/s11255-012-0366-y. [DOI] [PubMed] [Google Scholar]
- 63.Goek O. N., Köttgen A., Hoogeveen R. C., Ballantyne C. M., Coresh J., Astor B. C. Association of apolipoprotein A1 and B with kidney function and chronic kidney disease in two multiethnic population samples. Nephrology, Dialysis, Transplantation. 2012;27(7):2839–2847. doi: 10.1093/ndt/gfr795. [DOI] [PMC free article] [PubMed] [Google Scholar]