Abstract
Spontaneous resolution of non‐rate‐dependent left bundle branch block (LBBB) has been rarely reported. We present the case of a 74‐year‐old woman admitted with pulmonary edema, a newly diagnosed LBBB and severe left ventricular (LV) dysfunction. Five months later, the patient was asymptomatic, the ECG recording showed complete regression of the LBBB to narrow QRS and LV function completely recovered. However, at one‐year follow‐up LBBB reappeared together with mild LV dysfunction. Spontaneous resolution of LBBB may be responsible for LV electrical and mechanical reverse remodeling in dyssynchronopathies.
Keywords: dyssynchronopathy, ECG, heart failure, left bundle branch block
1. INTRODUCTION
Left bundle branch block (LBBB) is known to be a marker of the underlying structural heart disease, and it is considered a poor prognostic predictor in patients with heart failure (HF). In some cases, it is challenging to establish whether LBBB is the cause of left ventricular (LV) systolic impairment rather than the consequence of disease progression.
In some patients, the electrical and mechanical dyssynchrony induced by the presence of LBBB may be the causative mechanism of LV systolic dysfunction, therefore identifying a particular form of cardiomyopathy induced by LBBB, whose resolution (e.g., by cardiac resynchronization therapy) may induce a nearly complete regression of the condition. Spontaneous resolution of LBBB is a rare and poorly understood electrical phenomenon.
We present an unusual case of a woman with suspected LBBB‐induced cardiomyopathy and transient spontaneous resolution of LBBB together with improvement in LV systolic function and symptoms.
2. CASE REPORT
A 74‐year‐old woman was admitted to our hospital with acute pulmonary edema. Her past medical history was unremarkable. Blood pressure values were normal. The ECG showed sinus rhythm and a newly diagnosed LBBB (QRS duration 140 ms) (Figure 1). Transthoracic echocardiography (TTE) showed mild LV dilatation (end‐diastolic diameter 55 mm, end‐diastolic volume index 85 ml/m2) with normal absolute and relative wall thickness, significant intraventricular dyssynchrony and severely impaired systolic function (ejection fraction—EF calculated at 27%). Laboratory findings included a high‐sensitivity troponin value at 3.364 ng/ml (normal values < 0.056 ng/ml), while white blood cell count and C‐reactive protein values were within normal limits. Given the clinical presentation and the laboratory and ECG findings, the patient underwent urgent coronary angiography which demonstrated unobstructed coronary arteries. Guideline‐directed medical therapy (GDMT) for HF was started, and clinical stabilization was achieved. Her medications included an ACE‐inhibitor, a beta‐blocker (Carvedilol 6.25 mg bid), spironolactone, and loop diuretics. The patient was discharged after 6 days with the suspicion of a LBBB‐induced cardiomyopathy, and CRT implantation was scheduled after three months. At one‐month follow‐up, medical therapy was optimized and Sacubitril/Valsartan was prescribed, while beta‐blocker dosage remained unchanged due to the achieved target heart rate.
Figure 1.

ECG at first admission. Sinus rhythm and broad QRS with typical left bundle branch block morphology (QRS duration 140 ms, inferior axis). The heart rate was 66 bpm
At re‐admission for CRT implantation two months later, the patient was on NYHA II functional class. The ECG confirmed the presence of LBBB (QRS > 140 ms), and LV systolic function was still severely reduced. Unfortunately, the procedure could not successfully be carried out due to technical issues (no target vein other than the great cardiac vein), and the patient was discharged on medical therapy only and epicardial lead placement was considered.
During a scheduled follow‐up 5 months later, the ECG recording showed sinus rhythm at 79 bpm, complete regression of the LBBB to narrow QRS (QRS duration 90 ms) and otherwise normal findings (Figure 2). The patient functional capacity had subjectively improved, and no signs of congestion were detected. NT‐pro‐BNP levels were within normal limits. A repeated TTE demonstrated normal LV cavity dimensions and increased EF calculated at 55%. A subsequent cardiac magnetic resonance showed LV and right ventricular volumes within normal limits and calculated EF at 62%. Furthermore, it also revealed no presence at all of myocardial fibrosis or edema (Figure 3).
Figure 2.

ECG at 8 months. Complete resolution of the left bundle branch block (QRS 90 ms). The heart rate was 79 bpm
Figure 3.
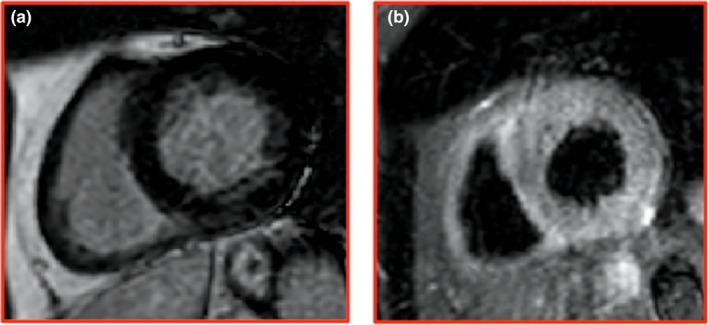
Cardiovascular magnetic resonance (CMR) at eight‐month follow‐up. Inversion recovery‐prepared gadolinium‐enhanced T1‐weighted short axis (a) views and T2‐weighted triple‐inversion recovery short axis view (b). The images demonstrate that there was neither late gadolinium enhancement nor myocardial edema on specific sequences
Surprisingly, a new ECG recorded at one‐year follow‐up showed sinus rhythm at 70 bpm and the recurrence of the same LBBB morphology (QRS duration 140 ms) (Figure 4), while TTE showed again mild LV dyssynchrony and subtle systolic impairment. Moreover, the conduction disturbance persisted irrespective of any heart rate increase or reduction, as seen on ECG exercise test.
Figure 4.

ECG at one year. Reoccurrence of left bundle branch block morphology. The heart rate was 70 bpm
3. DISCUSSION
We described the case of a patient suffering from de novo HF and LBBB‐induced LV dysfunction. Over follow‐up, we observed an on and off behavior of the LBBB accompanied by concordant changes in the patient's clinical status and LV function.
The presence of permanent LBBB is often considered a marker of the underlying structural cardiac abnormalities. However, there is plenty of literature describing intermittent and rate‐dependent intraventricular conduction disturbances whose appearance is related to changes in heart rate or in the cardiac cycle (Francia, Balla, Paneni, & Volpe, 2007). In our case though, no changes regarding heart rate or cardiac cycle length could be detected and there was not any beat‐to‐beat variation in the conduction through the left branch; moreover, LBBB morphology could be seen on multiple traces over a long period of time (3 months at least) and all these considerations led us to rule out the possibility that we might have been facing a properly defined intermittent LBBB.
Many acute conditions are traditionally related to the appearance of intraventricular conduction delay; indeed, LBBB is generally associated with myocardial injury, for example, due to ischemia or inflammation (Caforio et al., 2011). However, in our patient no certain cause for the appearance of LBBB could be identified.
Most importantly, our case demonstrates that the appearance of LBBB may prompt LV systolic function impairment whose main feature is dyssynchrony. Blanc, Fatemi, Bartault, Baraket, and Etienne (2005) already described this sort of cardiomyopathy when they found that LV pacing could potentially halt and reverse LV remodeling in patients with LBBB. The complete normalization of the LV systolic function and a significant improvement in our patient's clinical status when LBBB was not present strongly corroborates the hypothesis of a dyssynchrony‐induced form of cardiomyopathy.
Similarly to a few previous cases (Dizon, Horn, Neglia, Medina, & Garan, 2004; Kloosterman, Rienstra, van Gelder, & Maas, 2016), the mechanisms that may have counted for LBBB resolution and clinical improvement are uncertain. The patient was promptly started on HF‐GDMT and, when clinically indicated, Sacubitril/Valsartan was prescribed. Despite the lack of studies specifically investigating the role of GDMT for HF on conduction disturbances, QRS duration and LBBB‐induced cardiomyopathy, about 25% of patients with LBBB and HF with reduced ejection fraction may clinically respond to GDMT (Sze & Daubert, 2018). Although difficult to ascertain, this might have had a non‐negligible impact on electrical and mechanical LV remodeling. Finally, it is plausible that some idiopathic forms of LBBB, which are different from those induced by a defined pathological noxa (e.g., myocardial ischemia/inflammation), may occur with an on and off behavior resembling but different from classical intermittent forms (Bazoukis, Tsimos, & Korantzopoulos, 2016).
We discussed a rare case of electrical and mechanical LV reverse remodeling in a patient with suspected LBBB‐induced LV dysfunction with acute clinical presentation which apparently healed after spontaneous resolution of the LBBB. Despite being very uncommon, an apparently spontaneous regression of LBBB seems to be possible. Further studies are needed to investigate the complex relationship between LBBB and dyssynchrony‐induced cardiomyopathy.
Moccia E, Sanna GD, Parodi G. Transient left bundle branch block and intraventricular dyssynchrony as a cause of reversible left ventricular dysfunction: The “in vivo” documentation of spontaneous electrical remodeling. Ann Noninvasive Electrocardiol. 2019;24:e12667 10.1111/anec.12667
The authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
REFERENCES
- Bazoukis, G. , Tsimos, K. , & Korantzopoulos, P. (2016). Episodic left bundle branch block – a comprehensive review of the literature. Annals of Noninvasive Electrocardiology, 21(2), 117–125. 10.1111/anec.12361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanc, J. J. , Fatemi, M. , Bertault, V. , Baraket, F. , & Etienne, Y. (2005). Evaluation of left bundle branch block as a reversible cause of non‐ischaemic dilated cardiomyopathy with severe heart failure. A new concept of left ventricular dyssynchrony‐induced cardiomyopathy. Europace, 7(6), 604–610. 10.1016/j.eupc.2005.06005 [DOI] [PubMed] [Google Scholar]
- Caforio, A. L. , Pankuweit, S. , Arbustini, E. , Basso, C. , Gimeno‐Blanes, J. , Felix, S. B. , … Elliott, P. M. (2011). European Society of Cardiology working group on myocardial and pericardial diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. European Heart Journal, 34, 2636–2648. 10.1093/eurheartj/eht210 [DOI] [PubMed] [Google Scholar]
- Dizon, J. , Horn, E. , Neglia, J. , Medina, N. , & Garan, H. (2004). Loss of left bundle branch block follow biventricular pacing therapy for heart failure: Evidence for electrical remodelling? Journal of Interventional Cardiac Electrophysiology, 10, 47–50. 10.1023/B:JICE.0000011484.61659.b1 [DOI] [PubMed] [Google Scholar]
- Francia, P. , Balla, C. , Paneni, F. , & Volpe, M. (2007). Left bundle‐branch block‐pathophysiology, prognosis, and clinical management. Clinical Cardiology, 30, 110–115. 10.1002/clc.20034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kloosterman, M. , Rienstra, M. , Van Gelder, I. C. , & Maas, A. H. (2016). Spontaneous resolution of left bundle branch block and biventricular stimulation lead to reverse remodelling in dyssynchronopathy. Journal of Electrocardiology, 49, 696–698. 10.1016/j.jelectrocard.2016.08.007 [DOI] [PubMed] [Google Scholar]
- Sze, E. , & Daubert, J. P. (2018). Left bundle branch block‐induced left ventricular remodelling and its potential for reverse remodelling. Journal of Interventional Cardiac Electrophysiology, 52, 343–352. 10:1007/s10840-018-0407-2 [DOI] [PubMed] [Google Scholar]


