Abstract
The dysfunction of placenta is common in somatic cell nuclear transfer (SCNT) cloned cattle and would cause aberrant fetal development and even abortion, which occurred with highest rate at the mid- to late gestation. However, the mechanism of abnormal placentas was unclear. To analyze the transcriptome-wide characteristics of abnormal placentas in SCNT cloned cattle, the mRNA, lncRNA and miRNA of placental cotyledon tissue at day 180 after gestation were sequenced. A total of 19,055 mRNAs, 30,141 lncRNAs and 684 miRNAs were identified. Compared with control group, 362 mRNAs, 1,272 lncRNAs and nine miRNAs (six known and three novel miRNAs) were differentially expressed (fold change ≥ 2 and P-value < 0.05). The differentially expressed genes were functionally enriched in urea and ions transmembrane transport, which indicated that the maternal-fetal interactions were disturbed in impaired placentas. Furthermore, the competing endogenous RNAs (ceRNAs) networks were identified to illustrate their roles in abnormal placental morphology. The present research would be helpful to discover the mechanism of late gestational abnormality of SCNT cattle by provides important genomic information and insights.
Subject terms: Developmental biology, RNA sequencing
Introduction
The establishment of somatic cell nuclear transfer (SCNT) technology supplies a powerful impetus for the development of animal cloning. The wide-spread application of SCNT made the propagation of genetics and production of gene modified livestock time-saving and efficient1. Although the improvements of this technology have been made over the past two decade, the clone efficiency of SCNT cattle remains relatively low. Series research showed that the birth rate of cloned embryos was 5 to 15%1–3. At the early stage of embryonic implantation (30 to 90 days after gestation), the pregnant losses of SCNT embryo were associated with various placental abnormalities, such as hypoplasia of trophoblastic epithelium, alteration of basement membrane, degeneration of allantoic vessel and developmental retardation of the villous3–6. During the mid- to late gestation period, large offspring syndrome (LOS) was caused by hydrallantois and placenta megaly, even the lethality of fetus and its recipient3,7–9.
The correlation between placental gene expression variation and aberrant pregnancy were followed with interest for the developmental abnormality partly induced by placental disorder in SCNT cattle10. Placental array analysis showed that placental failure of cloned bovine to be associated with abnormal embryo-maternal communication during the peri-implantation period11. Salilew-Wondim et al. found that compared with artificial inseminated (AI) pregnancy, the alterations of gene expression in SCNT bovine placenta were more significant than in vitro embryo production (IVP) pregnancy at day 50 of gestation. Further analysis showed that the dysregulation of 9% of these genes was caused by transcriptional reprogramming error12. RNA-seq of cloned bovine extraembryonic tissue showed that differentially expressed genes between SCNT and AI leaded to the inhibition of trophoblast and placental development at both preimplantation and postimplantation stages13. Besides, the placental tissue microRNAs (miRNAs) analysis of deceased cloned calves suggested that the abnormal miRNA expression play a role at aberrant epigenetic reprogramming and late-fetal and/or neonatal lethality14.
Our previous work found that aberrant large abdominal circumference of recipient cows carrying SCNT fetuses at 150 to 200 days of gestation had a tight connection with hydrops allantois, placental hypertrophy and fetal overgrowth. In this study, abnormal placentas at mid- to late gestation were sampled and the mRNAs, lncRNAs and miRNAs expression profiles were detected through RNA-seq. Subsequently, the function of key genes, lncRNAs, miRNAs and their potential interactions on placental abnormalities were analyzed using bioinformatics. This study will expand our knowledge on aberrant SCNT bovine pregnancy and would be helpful to discover the mechanism of late gestational abnormality of SCNT cattle.
Results
The development of SCNT bovine fetus was affected by placental abnormality
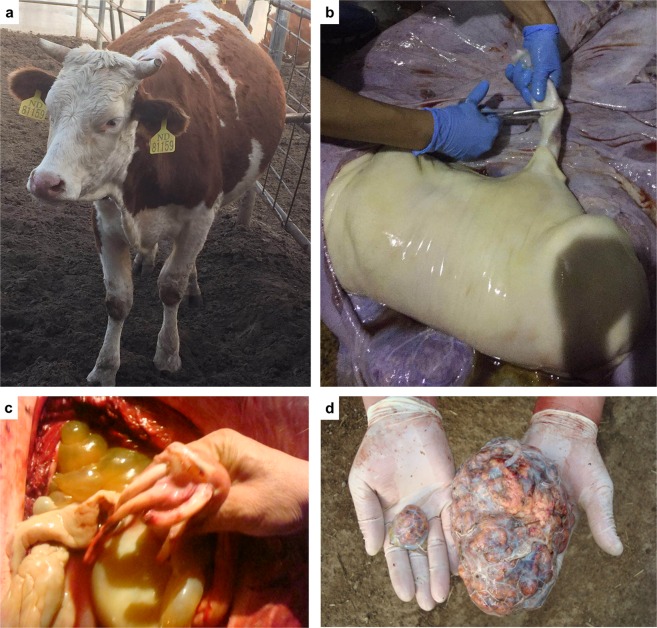
Our previous studies showed that the bovine embryos derived from artificial technology (especially asexual reproduction) with high abortion rate throughout the pregnancy process, particularly during the mid and late-gestation. According to our research data from 2008 to 2018, the rate of abortion was highest at 150 to 200 days of gestation. In one of experiments, the abortion rate reached to 24.00% in this period (unpublished data). The major symptom was that the abdominal circumferences of the recipient cows were aberrant large and aborted subsequently (Fig. 1a). Most of fetuses were oversized (Fig. 1b). A more extreme example was that the recipient cow delayed delivery for 30 days and signs of production appeared until 315 days after gestation. A huge calf was produced via caesarean section with the birth weight of 78 kg, which was 3 times to the normal calves (unpublished data). Unfortunately, this calf died less than a hundred days after birth.
Figure 1.
Clinical features of SCNT cattle during mid and late-pregnancy. (a) A case of SCNT fetus carried recipient cow with abnormally large abdominal circumference at 180 days of gestation. (b) A case of oversized SCNT bovine fetus. (c) The umbilical cord enlargement of the SCNT bovine fetus. (d) Placental hypertrophy and abnormal size of placental cotyledons.
The SCNT bovine abortions caused by LOS were also accompanied by placental abnormalities, which usually represented as enlargement of the umbilical cord, placental hypertrophy and abnormal size of placental cotyledons (Fig. 1c,d). In order to investigate the abnormal placentas at RNA level, we took out the fetuses and placentas after slaughter the recipients when the recipient cow was experientially observed to show abnormal abdominal circumference at day 180 ± 2 of gestation.
A total of 40 cows were used as recipients and 32 of which were estrous-synchronized. These recipients were transferred with SCNT (23 recipient cows) or in vivo produced embryos (9 recipient cows). At day 45 of gestation, the pregnancy rates of SCNT and in vivo produced embryos were 39.13% and 55.56%, respectively. At day 180 of gestation, the pregnancy rates of SCNT and in vivo produced fetuses were 13.04% and 55.56%, respectively (Table 1).
Table 1.
Statistics on pregnancy at gestation days after embryo transplant.
| Gestation day | SCNT cloned | In vivo derived | ||
|---|---|---|---|---|
| Pregnancy number | Pregnancy rate | Pregnancy number | Pregnancy rate | |
| Day 7 | 23 | — | 9 | — |
| Day 45 | 9 | 39.13% | 5 | 55.56% |
| Day 180 | 3 | 13.04% | 5 | 55.56% |
The days of gestation were counted starting at the day of the heat (Day 0). Embryos are transferred at Day 7. The line of Day 7 indicates recipient cows numbers used for embryo transplant.
Sequencing and mapping of mRNAs and lncRNAs
In order to develop a comprehensive catalogue of mRNAs and lncRNAs of bovine placentas, trascriptome data sets were generated by RNA-seq. Illumina sequencing of bovine placental cotyledons tissues yielded a total of 708,477,746 raw reads. The Phred Quality Score of the samples were more than 91% for Q30. Clean data included 133,443,802 reads at average and the minimum Q30 of clean data was 92.03%. Subsequently, the clean reads were mapped into the bovine reference genome using HiSAT215. The average mapping rate of the five samples was 94.11% and the unmapped rates were between 5.51% and 6.82%. Detail summary of the sequencing results were showed in Table S1, which could indicate the high quality of transcriptome sequencing data with suitable mapping. A total of 19,055 mRNAs were recognized from these data. Furthermore, after a series of basic and coding potential screening, 30,141 lncRNA loci were identified by overlapped the predicted results from CNCI, CPC, PFAM and CPAT.
Identification of miRNAs
To identify miRNAs of bovine normal and abnormal placentas, five small RNA libraries were constructed and sequenced independently. A total of 138,867,057 raw reads were generated. And 113,588,641 clean reads were obtained, which account for more than 77% of the raw data. Among five individual libraries, the minimum rates of removed reads were at a suitable level and the Q30 of raw or clean data were high enough to indicate the high-quality of small RNA sequencing (Table S2). The length of most sequences was distributed at the range of 21–23 nt and the highest percentage was 22nt, which is consistent with the common size of miRNAs. The total clean reads between 15–35 nt were aligned with the bovine genome using Bowtie software. The average perfect match rate was 72.70%. In total, 488 known and 196 novel miRNAs were identified using miRBase (Release 21) and miRDeep216.
Identification of differentially expressed mRNAs, lncRNAs and miRNAs between bovine normal and abnormal placentas
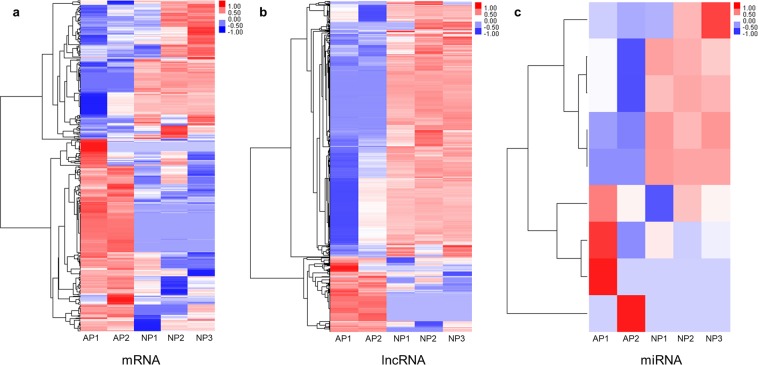
The identification of differentially expressed mRNAs, lncRNAs and miRNAs were calculated using edgeR with the threshold of fold change ≥2 and P-value < 0.05. 362 mRNAs, 1,272 lncRNAs and 9 miRNAs (6 known and 3 novel miRNAs) were found to be differentially expressed between abnormal and normal groups. Compared with normal group, 208 mRNAs, 283 lncRNAs and 4 miRNAs were up regulated, as well as 154 mRNAs, 989 lncRNAs and 5 miRNAs were down regulated in abnormal group. Expression patterns of mRNA, lncRNA and miRNA were shown by hierarchical clustering (Fig. 2). Then these differentially expressed non-coding RNAs (ncRNAs) were intersected with predicted target genes. As a result, 2,836 and 1,241 genes targeted with lncRNA and miRNA were screened out, respectively (Tables S3 and S4).
Figure 2.
Hierarchical clustering of mRNA, lncRNA and miRNA expression patterns compared between abnormal (AP) and normal placentas (NP). Expression values are represented in shades of red and blue, indicating expression above and below the mean expression value, respectively.
Functional enrichment analyses of differentially expressed ncRNAs target genes and differentially expressed mRNAs
To investigate the function of differentially expressed lncRNAs and miRNAs, the enrichment analyses of their target genes were performed. For target genes of differentially expressed lncRNAs, substance metabolism pathways were significantly enriched, including metabolic pathways (KO01100), carbon metabolism (KO01200) and glycerol phospholipid metabolism (KO00564). Besides, transcription associated functions were also high representation, such as Poly(A) RNA binding (GO: 0044822), catalytic step 2 spliceosome (GO: 0071013), mRNA splicing via spliceosome (GO: 0000398), RNA splicing (GO: 0008380), mRNA processing (GO: 0006397) and RNA binding (GO: 0003723). For target genes of differentially expressed miRNAs, the most significantly enriched pathway was ECM-receptor interaction (KO04512), which involved functions of tissue structural maintaining and organ morphogenesis. Moreover, hormone related pathways could also be found, such as ovarian steroidogenesis (KO04913), GnRH signaling pathway (KO04912), estrogen signaling pathway (KO04915) and aldosterone synthesis & secretion (KO04925). The results suggested that the abnormal ncRNAs expression may affect the material metabolism and hormone secretion of bovine placenta via regulate target genes.
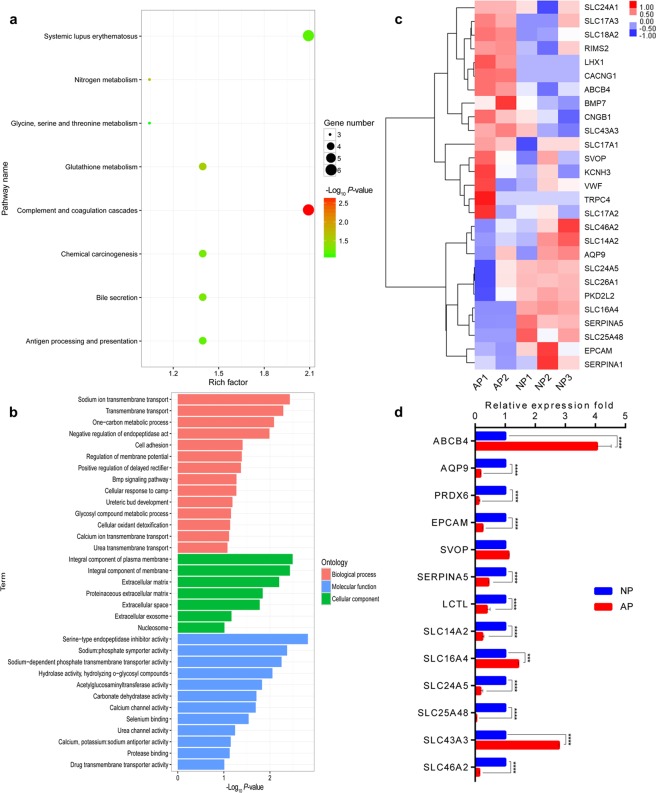
As the expression of mRNA is more directly related to the biological characters, the function of differentially expressed mRNAs was analyzed by KEGG and GO enrichment. A total of 8 KEGG pathways were enriched by the 362 differentially expressed genes (DEGs), of which the complement and coagulation cascades pathway was most significant (Fig. 3a). It was noteworthy that these DEGs were also functionally enriched in GO categories for the transmembrane transport of urea and ions (Fig. 3b). In the categories of ureteric bud development (GO: 0001657), urea transmembrane transport (GO: 0071918) and urea channel activity (GO: 0015265), the expressions of BMP7 and LHX1 were up-regulated in abnormal group, while SLC14A2, EPCAM and AQP9 were down-regulated (Fig. 3c). In the categories associated with ion transmembrane transport, 22 genes were involved. The expressions of 14 genes (SLC43A3, SVOP, VWF, RIMS2, ABCB4, SLC18A2, CNGB1, SLC24A1, KCNH3, SLC17A2, SLC17A1, SLC17A3, CACNG1, TRPC4) were up-regulated in abnormal group, and the other 8 genes (SLC16A4, PKD2L2, SLC25A48, SLC24A5,SLC26A1, SERPINA1, SLC46A2, SERPINA5) were down-regulated (Fig. 3c). To confirm the gene expression patterns, half of these genes were randomly selected to be validated by q-PCR. The results were in concordance with the RNA-seq data (Fig. 3d).
Figure 3.
The functional enrichment of the differentially expressed genes between between abnormal (AP) and normal placentas (NP). (a) The KEGG pathway enrichment of the DEGs. (b) The GO enrichment of the DEGs. (c) Expression patterns of selected genes associated with urea and ions transmembrane transport. (d) Candidate genes validated by q-PCR. ***P < 0.001, ****P < 0.0001.
The interaction network between ncRNAs and their target mRNAs
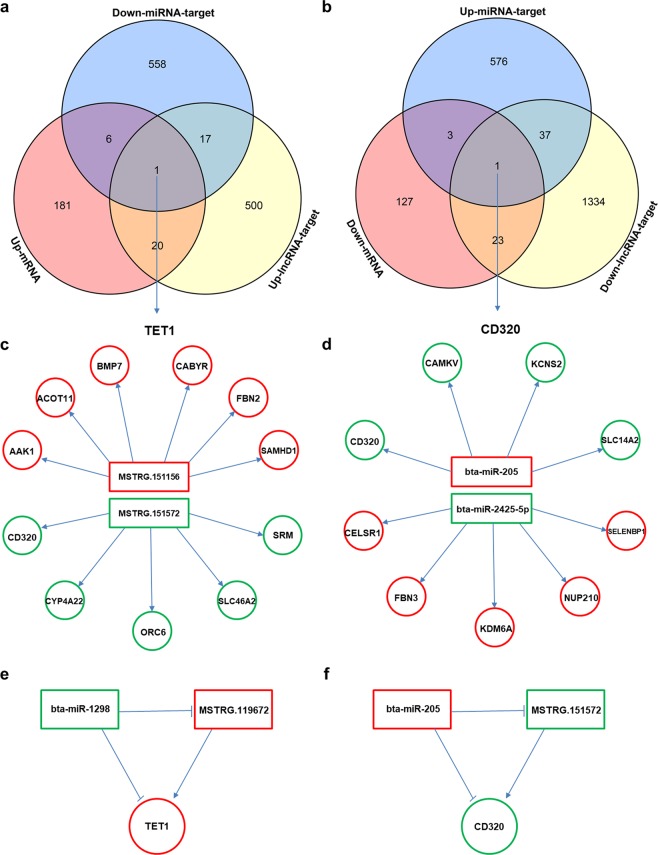
DEGs and their corresponding differentially expressed lncRNAs or miRNAs were selected according to the regulation relationship and expression trend between mRNAs and ncRNAs. In the abnormal group, target genes of up-regulated lncRNAs or down-regulated miRNAs were overlapped with up-regulated DEGs, as well as the target genes of down-regulated lncRNAs or up-regulated miRNAs were overlapped with down-regulated DEGs (Fig. 4a,b, Table 2). Based on the expression pattern of competing endogenous RNAs (ceRNAs), these overlapped DEGs were more likely to be regulated by lncRNAs or miRNAs17. Most of the up- and down-regulated DEGs were probably regulated by MSTRG.151156 and MSTRG.151572, respectively (Fig. 4c, Tables 2 and S5). Simultaneously, bta-miR-205 and bta-miR-2425-5p targeted more DEGs either (Fig. 4d, Table S5). It was worth noting that TET1 (up-regulated in abnormal group) and CD320 (down-regulated in abnormal placentas) were under the regulation of both lncRNA and miRNA (Fig. 4a,b), such as bta-miR-1298-MSTRG.119672-TET1 and bta-miR-205-MSTRG.151572-CD320 ceRNA networks (Fig. 4e,f). MSTRG.119672 serves as ceRNA to up regulate TET1. As a negative regulator, bta-miR-205 inhibits the expression of CD320.
Figure 4.
The interaction network between mRNAs and their target non-coding RNAs. (a) Overlap of up-regulated lncRNAs, up-regulated DEGs and down-regulated miRNAs in the abnormal placentas. (b) Overlap of down-regulated lncRNAs, down-regulated DEGsandup-regulated miRNAs in the abnormal placentas. (c) lncRNAs(in box) that potentially regulate most of the DEGs (in circle). Red indicates up- regulation, green indicates down- regulation. (d) miRNAs(in box) that potentially regulate most of the DEGs (in circle). Red indicates up- regulation, green indicates down- regulation. (e) The ceRNA interactions of bta-miR-1298-MSTRG.119672-TET1. (f) The ceRNA interactions of bta-miR-205-MSTRG.151572-CD320.
Table 2.
The differentially expressed candidate genes and their target non-coding RNAs in bovine placentas.
| Gene | target lncRNA | target miRNA | |||
|---|---|---|---|---|---|
| BMP7 | ↑ | MSTRG.119672 | ↑ | — | |
| MSTRG.161616 | ↓ | — | |||
| MSTRG.151156 | ↑ | — | |||
| MSTRG.236813 | ↑ | — | |||
| SLC14A2 | ↓ | — | bta-miR-205 | ↑ | |
| RIMS2 | ↑ | MSTRG.196921 | ↓ | ||
| ABCB4 | ↑ | MSTRG.161616 | ↓ | ||
| MSTRG.195717 | ↓ | ||||
| KCNH3 | ↑ | MSTRG.181130 | ↓ | ||
| TRPC4 | ↑ | — | Novel_97 | ↑ | |
| SLC16A4 | ↓ | — | bta-miR-1298 | ↓ | |
| SLC46A2 | ↓ | MSTRG.151572 | ↓ | — | |
| SERPINA5 | ↓ | MSTRG.181130 | ↓ | — | |
The arrows represent up or down expressions of RNAs in the abnormal placentas tissues.
Discussion
SCNT is one of the most useful embryo engineering techniques in basic research and livestock breeding. However, the bottleneck of low developmental efficiency and high incidence of abnormalities of SCNT offspring limits its development. Abortion of SCNT-derived fetuses usually caused by placental hypertrophy, allantoichydrops and placental edema at day 150–200 of pregnancy, and the placental hypertrophy is almost the universal complication of SCNT fetal death18. In this study, we focused on the SCNT bovine oversized placentas to understand the mechanism of abnormally SCNT bovine pregnant during mid to late gestation through the transcriptome analysis.
Clinical features of abnormal pregnant SCNT cattle during mid and late-gestation
During 2008 to 2018, a total of approximately 4,000 SCNT, in vitro fertilization (IVF) and in vivo produced bovine embryos were transferred by our team. We found that LOS and concomitant placental hypertrophy were occurred mainly in SCNT fetuses and some of IVF fetuses. About 1/4 recipient cows which carried SCNT fetuses exhibited symptom of larger abdominal circumference and eventually lead to the death of the recipients and the offspring. This problem has always been one of the great difficulties in animal husbandry19. Similar as Liu’s study, we found that this problem was donor cell-dependant9. In this study, several recipients exhibited the symptom with large abdominal circumference. And RNAs extracted from these abortive placentas failed to meet the criteria of transcriptome sequencing. Finally, we slaughtered the abnormal pregnant recipients to obtain hypertrophic placentas.
Functional affection of abnormal expressed genes of SCNT bovine placenta
Understanding of the transcriptome-wide landscape is conducive to discover the mechanism associated with placental morphological and functional changes. Similar with previous transcriptome or proteome studies, we also found that DEGs between abnormal and normal placentas were functionally enriched in complement and coagulation cascades pathway20,21. The result suggested that the regulatory changes of this pathway might influence the normal development of placentas. The functional GO terms of urea and ions transporting across membrane were associated with maternal-fetal interaction, in which a series of DEGs were involved.
In the categories of ureteric bud development and urea transport, BMP7 and LHX1 were up-regulated in abnormal placentas. BMP7 was shown to affect embryonic cloacal cavity separates, development of urethral system, as well as placental abnormalities22,23. Complete inactivation of BMP7 were found to restore ureteric bud outgrowth and branching24. Similarly, LHX1 was also considered to be associated with dysplasia of urogenital system development25. On the contrary, AQP9, EPCAM and SLC14A2 were identified to be expressed significantly lower in abnormal placentas by both RNA-seq and q-PCR. AQP9 encodes a water channel protein which participates in stimulating urea transport. Up-regulation of AQP9 were required when organismic repairation of pathological placental changes26. The abnormal amniotic fluid was considered to be related with the expression alterations of AQPs to in human, in which AQP9 played a role in the urea uptake and elimination across the placenta27. EPCAM were generally defined as a potential tumor marker and immunotherapy target, it was also suggested that inactivation of this gene could lead to prominent placental abnormalities. Placentas of EPCAM−/− mice exhibited thin labyrinthine layers and lacking of vascularity and induced early embryonic lethality28. However, EPCAM+/− mice were normal. SLC14A2 is one of the two major subgroups of the SLC14A urea transporter family, which is mainly expressed in kidney29. Down-regulation of SLC14A2 in abnormal placentas might cause difficulty of urea excreting. Therefore, abnormal expression of these genes in placentas might impact the transport of urea in SCNT bovine fetuses.
In the category of ions transmembrane transport, most of DEGs in this study were members of solute carrier family. Expression changes of these genes in abnormal placentas might be associated with maternal-fetal substances circulations for their crucial roles in transport of ions and drugs. The aberrant expression pattern of these genes not only causes the deliver obstruction of various substances including urate, but also may affect the placenta morphology and maternal-fetal interaction, which further aggravates placental dysfunction.
Effecting of ceRNA for SCNT bovine placental abnormity
Abnormal gene expression in SCNT bovine placenta could be attributable to the epigenome features of the somatic cell genome caused by the inappropriate reprogramming in SCNT embryos30,31. Besides, it is known that ncRNAs play diverse roles in regulating gene expression. In ceRNA network, miRNAs are usually considered to play negative regulations on mRNA, while lncRNAs can act as decoys of miRNAs to modulate gene expression32,33. Therefore, the expression of target lncRNAs is more similar with their corresponding genes.
In this study, some DEGs were likely to be regulated by lncRNAs or miRNAs, for the target genes of up-regulated lncRNAs or down-regulated miRNAs were overlapped with up-regulated DEGs, and the target genes of down-regulated lncRNAs or up-regulated miRNAs were overlapped with down-regulated DEGs. Two gene sets were obtained by above strategy, in which TET1 and CD320 were under the regulation by both lncRNA and miRNA. TET1 is a member of the TET family, the function contains the regulation of DNA methylation in mouse embryonic stem cells, placental trophoblasts and pathological placental tissue34–37. The expression of TET1 was under the negative regulation of miRNAs in cancer cells, such as miR-29, miR-26a, miR-767, miR-494 and miR-520b38. In current study, TET1 was found to be regulated by bta-miR-1298 and a lncRNA MSTRG.119672 in bovine placenta. In addition, the expression of CD320 was potentially co-regulated by bta-miR-205 and MSTRG.151572. CD320, also known as TCblR, is a transcobalam in receptor which is expressed in placenta with high quantities and mediates cobalamin (vitamin B12) maternal-fetal transport39. Impaired placental vascularization and endothelial dysfunction are considered to be associated with the concentration of placenta-related parameters in the circulation. Therefore soluble form of CD320 in serum was identified to be a potential biomarker for evaluating pregnancy risk40,41. We surmise that placental abnormity of SCNT cattle might be associated with the aberrant maternal-fetal transport of vitamin B12 by the altered expression of CD320 and/or its target ncRNAs.
Conclusions
In this study, we present the transcriptome-wide data of abnormal placental cotyledon tissues from SCNT cloned cattle at day 180 of gestation. The comparative analyses of mRNA, lncRNA, miRNA and ceRNA provide important genomic information and insights for further discovering the mechanism of abnormally SCNT bovine pregnant during late gestation.
Methods
Ethics approval and consent to participate
All experimental procedures and sample collections were conducted in accordance with the guidelines of the Inner Mongolia University Animal Care and Use Committee. The bovine ovaries used in this study were collected with permission of the Hohhot slaughterhouse. Experimental protocols were approved by the Institutional Animal Care and Use Committee at Inner Mongolia University.
Design of experiments
The donor cell line used in this study was designedly selected which could lead to higher rate of placental abnormalities according to our previous research. The tissues derived from in vivo produced embryo were set as control group. The abnormal and normal placentas were sampled at day 180 ± 2 of pregnancy. These samples were used for RNA-seq and quantitative real-time PCR (q-PCR) analyses.
Production of SCNT and in vivo embryos
In this study, fetal fibroblast of male Chinese Luxi cattle was used as donor for SCNT. The SCNT procedure was described as previously reported15. The protocols of nuclear transfer, fusion, activation, and embryo in vitro culture were followed as Wu et al.16. For in vivo embryo collection, donor cows (Chinese Simmental) were superovulated and inseminated with frozen/thawed semen from one Chinese Luxi bull with proven fertility. Morulae and blastocysts were collected by uterine flushing at day 7 or 8 after artificial insemination by routine nonsurgical procedure. Both SCNT and in vivo produced embryos were evaluated under a stereomicroscope and only morphologically intact embryos were selected for transfer.
Embryo transfer and pregnancy detection
The treatment of estrus synchronization was carried out according to previous17. Briefly, Simmental heifers of 18–20 month-old were chosen as recipients. At day 0 of estrus synchronization, EAZI BREED CIDR (Pfizer Pty, New Zealand) was used. At day 9, the recipients were injected with 0.5 mg cloprostenol (prostaglandin F2α) (Ningbo second hormone factory, China). At day 11, CIDR was removed. The recipients were observed estrus conditions at day 12–13. Each of estrous-synchronized recipient cow was transferred one or two blastocysts non-surgically at day 7 after estrus. Pregnancy was first detected by trans-rectal ultrasound at around 45 days after estrus, and placental samples were collected at 180 days after estrus.
Sample collection
At day 180 of gestation, three pregnant recipients which carried SCNT fetuses presented overgrowth of abdominal circumference compared to others. And the abnormal placental cotyledons were separated by caesarean section. In addition, three normal placental samples from in vivo produced embryo carrier recipients were collected as controls. These samples were snap-frozen in liquid nitrogen and stored for use towards the subsequent generation of RNA libraries.
RNA sequencing
Prior to the transcriptome sequencing, total RNA of the collected bovine placental tissues was extracted using Trizol reagent (Invitrogen, USA) and the quality was determined using agarose electrophoresis, Spectrophotometer and Agilent 2100 RNA Nano 6000 Assay Kit(Agilent Technologies, USA). The extracted RNAs were stored at −80 °C before the next use.
For mRNA and lncRNA sequencing, the RNA libraries of each sample were constructed separately. After removing ribosomal RNA by Ribo-ZeroTM Gold Kits (Epicentre, USA), libraries were prepared by select different index Tags according the manufacturer’s instructions of NEB Next Ultra Directional RNA Library Prep Kit for Illumina (NEB, USA).Then these libraries were sequenced on an Illumina Hiseq X ten system in PE150 mode.
For miRNA sequencing, small RNA fragments of 15–35 nt were isolated from total RNA, then ligated with adaptors and synthesized to cDNA for amplification. Then the prepared libraries were sequenced on an Illumina Hiseq. 2500 system in SE50 mode.
Quality control of sequencing data
The raw data were filtered under a series of steps as follow. For both RNA and miRNA data, low quality reads (>15% of bases whose Phred scores were ≤ 19) were removed. Then adapter-containing reads (the adapter sequence > 5 bp), unknown base calls (N) with the rate > 5% and rRNA matched reads were trimmed out from RNA raw data. For the raw data of miRNA, unknown base calls (N) with the rate > 10%, reads without 3’ adapters or insert fragments, reads containing poly-A/T or the length was not within the required range, were filtered to generate clean data. The Phred Quality Score (Q30) was calculated to assess the qualities of both raw and clean data. All subsequent analyses were based on the clean data.
Transcriptome assembly
The reference bovine genome and the annotation file were downloaded from ENSEMBL database (http://www.ensembl.org/index.html). And Clean Data were mapped to the reference genome using HISAT2 (v2.0.5) (http://ccb.jhu.edu/software/ hisat2/index.shtml). The mapped reads of each sample were assembled by using StringTie (v1.3.2d) with the parameter of -G ref.gtf -rf–l18,19.
Identification of lncRNA and miRNA
Bovine lncRNAs were identified from the assembled transcripts under the following conditions: (1) transcripts with length < 200 bp were removed; (2) transcripts with exon number < 2 were removed; (3) transcripts of all samples with reads coverage < 5 were removed; (4) compared with the annotation file of the species to screen the known mRNA and other non-coding RNA (rRNA, tRNA, snoRNA and snRNA) using the gffcompare software. (5) according to the information of class_code (“u”, “i”, “x”), the potential lincRNA, intronic lncRNAand anti-sense lncRNA were screened; (6) transcripts without coding potentials were removed byco-analysis of Coding-Non-Coding Index (CNCI), Coding Potential Calculator (CPC), PFAM database and Coding Potential Assessment Tool (CPAT)20.
The basic idea of known miRNA identification is to get overlap (100%) between genome location of mapping reads and the genome location of reference miRNA. If genome annotation data can be found in miRBase (Release 21), it just needs to get overlap by software Bed tools (v2.17.0)21. Otherwise, we will firstly map the reference miRNA to the reference genome to obtain location information, then get overlap. After excluded reads that mapped to known miRNA/ncRNA/repeat region/mRNA region, the remained reads were used to predict novel miRNA for animal by using software miRDeep222. The key of identification was the hairpin structure formation of reads stack and possibility and stability evaluation.
Quantification of RNAs expression level
For mRNAs and lncRNAs, read counts were counted by HTSeq23. FPKM (Fragments Per Kilobase Millon Mapped Reads) were calculated to represent the expression level in each sample24. For miRNAs, RPM (Reads Per Million total reads) values were considered as normalized count of sample, and can be directly used in inter-library comparison.
Target gene prediction of lncRNA and miRNA
For lncRNAs, mRNAs were selected with High Spearman correlation coefficient (P ≥ 0.9) as the trans-targets. And the mRNAs with distance less than 50 kb were selected as the cis-targets. For miRNAs, miRanda (3.3a) were used to predict targets of known or novel miRNA. The principle of miRanda prediction was on the basis of seed region sequence alignment. Results would be filtered by parameters -sc 160 -en −20.
Differential expressed RNAs and functional enrichment analyses
The differentially expressed RNAs (mRNAs, lncRNAs and miRNAs) between abnormal and control placental cotyledons were calculated by edgeR with P < 0.05 and |log2 ratio| ≥ 1. The enrichment analyses of KEGG (Kyoto Encyclopedia of Genes and Genomes) (https://www.kegg.jp/) and GO (Gene Ontology) (http://www.geneontology.org/) were performed by using DAVID (The Database for Annotation, Visualization and Integrated Discovery) (https://david.ncifcrf.gov/) under the background of Bostaurus species with the default parameters.
Supplementary information
Acknowledgements
We owe many thanks to Annoroad Gene Technology (Beijing) Co. Ltd. for the RNA sequencing and clean data processing. We are indebted to Gene Denovo (Guangzhou) Co. Ltd for their online Omic share tools which were used in this study. We are also grateful to Yunxi Wu for help in fieldworks of cattle farm. This research was partly funded by the National Natural Science Foundation of China (31460599) and the National Transgenic Animal Program (2016ZX08007002). Personally, as the writer of this article, I thank my four-year-old daughter, Shakira Gao, she is the source of my spiritual power.
Author contributions
L.Z., G.L. and G.G. conceived the study. S.W., X.S., C.B., Z.W. and Z.Z. prepared the samples. G.S. and L.Y. produced S.C.N.T. and in vivo derived embryos. L.Z., S.W. and J.Z. finished the embryo transfer. G.G. carried out the transcriptome analyses. X.L., Z.G. and X.S. carried out the q-PCR experiments. All authors contributed to data interpretation. G.G. and X.S. wrote the paper with significant contributions from G.L., L.Z. and X.W.
Data availability
All data generated or analyzed during this study are available from the corresponding author upon reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Guangqi Gao, Shenyuan Wang and Jiaqi Zhang.
Contributor Information
Guangpeng Li, Email: gpengli@imu.edu.cn.
Xiaohu Su, Email: 13947144670@139.com.
Li Zhang, Email: zhanglinmg@aliyun.com.
Supplementary information
is available for this paper at 10.1038/s41598-019-56566-w.
References
- 1.Keefer CL. Artificial cloning of domestic animals. Proc Natl Acad Sci USA. 2015;112:8874–8878. doi: 10.1073/pnas.1501718112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Long CR, Westhusin ME, Golding MC. Reshaping the transcriptional frontier: epigenetics and somatic cell nuclear transfer. Mol Reprod Dev. 2014;81:183–193. doi: 10.1002/mrd.22271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pozor MA, et al. Placental abnormalities in equine pregnancies generated by SCNT from one donor horse. Theriogenology. 2016;86:1573–1582. doi: 10.1016/j.theriogenology.2016.05.017. [DOI] [PubMed] [Google Scholar]
- 4.Palmieri C, Loi P, Ptak G, Della Salda L. Review paper: a review of the pathology of abnormal placentae of somatic cell nuclear transfer clone pregnancies in cattle, sheep, and mice. Vet Pathol. 2008;45:865–880. doi: 10.1354/vp.45-6-865. [DOI] [PubMed] [Google Scholar]
- 5.Wells DN. Animal cloning: problems and prospects. Rev Sci Tech. 2005;24:251–264. doi: 10.20506/rst.24.1.1566. [DOI] [PubMed] [Google Scholar]
- 6.Hill JR, et al. Evidence for placental abnormality as the major cause of mortality in first-trimester somatic cell cloned bovine fetuses. Biol Reprod. 2000;63:1787–1794. doi: 10.1095/biolreprod63.6.1787. [DOI] [PubMed] [Google Scholar]
- 7.Heyman Y, et al. Frequency and occurrence of late-gestation losses from cattle cloned embryos. Biol Reprod. 2002;66:6–13. doi: 10.1095/biolreprod66.1.6. [DOI] [PubMed] [Google Scholar]
- 8.Constant F, et al. Large offspring or large placenta syndrome? Morphometric analysis of late gestation bovine placentomes from somatic nuclear transfer pregnancies complicated by hydrallantois. Biol Reprod. 2006;75:122–130. doi: 10.1095/biolreprod.106.051581. [DOI] [PubMed] [Google Scholar]
- 9.Liu J, et al. Nuclear donor cell lines considerably influence cloning efficiency and the incidence of large offspring syndrome in bovine somatic cell nuclear transfer. Reprod Domest Anim. 2013;48:660–664. doi: 10.1111/rda.12140. [DOI] [PubMed] [Google Scholar]
- 10.Chavatte-Palmer P, et al. Review: Placental perturbations induce the developmental abnormalities often observed in bovine somatic cell nuclear transfer. Placenta. 2012;33(Suppl):S99–S104. doi: 10.1016/j.placenta.2011.09.012. [DOI] [PubMed] [Google Scholar]
- 11.Bauersachs S, et al. The endometrium responds differently to cloned versus fertilized embryos. Proc Natl Acad Sci USA. 2009;106:5681–5686. doi: 10.1073/pnas.0811841106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Salilew-Wondim D, et al. Aberrant placenta gene expression pattern in bovine pregnancies established after transfer of cloned or in vitro produced embryos. Physiol Genomics. 2013;45:28–46. doi: 10.1152/physiolgenomics.00076.2012. [DOI] [PubMed] [Google Scholar]
- 13.Biase FH, et al. Massive dysregulation of genes involved in cell signaling and placental development in cloned cattle conceptus and maternal endometrium. Proc Natl Acad Sci USA. 2016;113:14492–14501. doi: 10.1073/pnas.1520945114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Su J, et al. Identification of differentially expressed microRNAs in placentas of cloned and normally produced calves by Solexa sequencing. Anim Reprod Sci. 2015;155:64–74. doi: 10.1016/j.anireprosci.2015.02.001. [DOI] [PubMed] [Google Scholar]
- 15.Pertea M, Kim D, Pertea GM, Leek JT, Salzberg SL. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat Protoc. 2016;11:1650–1667. doi: 10.1038/nprot.2016.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Friedlander MR, Mackowiak SD, Li N, Chen W, Rajewsky N. miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucleic Acids Res. 2012;40:37–52. doi: 10.1093/nar/gkr688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Guil S, Esteller M. RNA-RNA interactions in gene regulation: the coding and noncoding players. Trends Biochem Sci. 2015;40:248–256. doi: 10.1016/j.tibs.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 18.Arnold DR, et al. Placental insufficiencies in cloned animals - a workshop report. Placenta. 2008;29(Suppl A):S108–110. doi: 10.1016/j.placenta.2007.11.010. [DOI] [PubMed] [Google Scholar]
- 19.Young LE, Sinclair KD, Wilmut I. Large offspring syndrome in cattle and sheep. Rev Reprod. 1998;3:155–163. doi: 10.1530/ror.0.0030155. [DOI] [PubMed] [Google Scholar]
- 20.Huang J, et al. A comparison of transcriptomic profiles in endometrium during window of implantation between women with unexplained recurrent implantation failure and recurrent miscarriage. Reproduction. 2017;153:749–758. doi: 10.1530/REP-16-0574. [DOI] [PubMed] [Google Scholar]
- 21.Jia R, et al. Comparative Proteomic Profile of the Human Umbilical Cord Blood Exosomes between Normal and Preeclampsia Pregnancies with High-Resolution Mass Spectrometry. Cell Physiol Biochem. 2015;36:2299–2306. doi: 10.1159/000430193. [DOI] [PubMed] [Google Scholar]
- 22.Xu K, et al. Bmp7 functions via a polarity mechanism to promote cloacal septation. PLoS One. 2012;7:e29372. doi: 10.1371/journal.pone.0029372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Monsivais D, et al. BMP7 Induces Uterine Receptivity and Blastocyst Attachment. Endocrinology. 2017;158:979–992. doi: 10.1210/en.2016-1629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goncalves A, Zeller R. Genetic analysis reveals an unexpected role of BMP7 in initiation of ureteric bud outgrowth in mouse embryos. PLoS One. 2011;6:e19370. doi: 10.1371/journal.pone.0019370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang W, et al. Identification and functional analysis of a novel LHX1 mutation associated with congenital absence of the uterus and vagina. Oncotarget. 2017;8:8785–8790. doi: 10.18632/oncotarget.14455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Castro-Parodi M, et al. Oxygen tension modulates AQP9 expression in human placenta. Placenta. 2013;34:690–698. doi: 10.1016/j.placenta.2013.04.017. [DOI] [PubMed] [Google Scholar]
- 27.Damiano AE. Review: Water channel proteins in the human placenta and fetal membranes. Placenta. 2011;32(Suppl 2):S207–211. doi: 10.1016/j.placenta.2010.12.012. [DOI] [PubMed] [Google Scholar]
- 28.Nagao K, et al. Abnormal placental development and early embryonic lethality in EpCAM-null mice. PLoS One. 2009;4:e8543. doi: 10.1371/journal.pone.0008543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klein JD, Blount MA, Sands JM. Molecular mechanisms of urea transport in health and disease. Pflugers Arch. 2012;464:561–572. doi: 10.1007/s00424-012-1157-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sim BW, Park CW, Kang MH, Min KS. Abnormal gene expression in regular and aggregated somatic cell nuclear transfer placentas. BMC Biotechnol. 2017;17:34. doi: 10.1186/s12896-017-0355-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Humpherys D, et al. Abnormal gene expression in cloned mice derived from embryonic stem cell and cumulus cell nuclei. Proc Natl Acad Sci USA. 2002;99:12889–12894. doi: 10.1073/pnas.192433399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ulitsky, I. Interactions between short and long noncoding RNAs. FEBS Lett, 10.1002/1873-3468.13085 (2018). [DOI] [PubMed]
- 33.Chan, J. J. & Tay, Y. Noncoding RNA:RNA Regulatory Networks in Cancer. Int J Mol Sci, 19, 10.3390/ijms19051310 (2018). [DOI] [PMC free article] [PubMed]
- 34.Koh KP, et al. Tet1 and Tet2 regulate 5-hydroxymethylcytosine production and cell lineage specification in mouse embryonic stem cells. Cell Stem Cell. 2011;8:200–213. doi: 10.1016/j.stem.2011.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yuen RK, Chen B, Blair JD, Robinson WP, Nelson DM. Hypoxia alters the epigenetic profile in cultured human placental trophoblasts. Epigenetics. 2013;8:192–202. doi: 10.4161/epi.23400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jin S, et al. Global DNA hypermethylation in down syndrome placenta. PLoS Genet. 2013;9:e1003515. doi: 10.1371/journal.pgen.1003515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ma M, Zhou QJ, Xiong Y, Li B, Li XT. Preeclampsia is associated with hypermethylation of IGF-1 promoter mediated by DNMT1. Am J Transl Res. 2018;10:16–39. [PMC free article] [PubMed] [Google Scholar]
- 38.Tian YP, Zhu YM, Sun XH, Lai MD. Multiple Functions of Ten-eleven Translocation 1 during Tumorigenesis. Chin Med J (Engl) 2016;129:1744–1751. doi: 10.4103/0366-6999.185873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arora K, Sequeira JM, Quadros EV. Maternofetal transport of vitamin B12: role of TCblR/CD320 and megalin. FASEB J. 2017;31:3098–3106. doi: 10.1096/fj.201700025R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Abuyaman O, Torring N, Obeid R, Nexo E. First trimester serum levels of the soluble transcobalamin receptor, holo-transcobalamin, and total transcobalamin in relation to preeclampsia risk. Scand J Clin Lab Invest. 2016;76:641–644. doi: 10.1080/00365513.2016.1230885. [DOI] [PubMed] [Google Scholar]
- 41.Abuyaman O, Andreasen BH, Kronborg C, Vittinghus E, Nexo E. The soluble receptor for vitamin B12 uptake (sCD320) increases during pregnancy and occurs in higher concentration in urine than in serum. PLoS One. 2013;8:e73110. doi: 10.1371/journal.pone.0073110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed during this study are available from the corresponding author upon reasonable request.