Abstract
When breeding females compete for limited resources, the intensity of this reproductive conflict can determine whether the fitness benefits of current reproductive effort exceed the potential costs to survival and future fertility. In group-living species, reproductive competition can occur through post-natal competition among the offspring of co-breeding females. Spontaneous abortion could be a response to such competition, allowing females to curtail reproductive expenditure on offspring that are unlikely to survive and to conserve resources for future breeding opportunities. We tested this hypothesis using long-term data on banded mongooses, Mungos mungo, in which multiple females within a group give birth synchronously to a communal litter that is cared for by other group members. As predicted, abortions were more likely during dry periods when food is scarce, and in breeding attempts with more intense reproductive competition. Within breeding events, younger, lighter females carrying smaller fetuses were more likely to abort, particularly those that were also of lower rank. Our results suggest that abortion may be a means by which disadvantaged females conserve resources for future breeding attempts in more benign conditions, and highlight that female reproductive competition may be resolved long before the production of offspring.
Keywords: banded mongoose, abortion, female reproductive competition, cooperative species, reproductive suppression
1. Introduction
Female reproductive conflict, that is, competition over the distribution of reproduction among females in a social unit, is typically more subtle than the male–male competition and hence easily overlooked [1]. Yet, females use a variety of strategies to compete over access to and control of resources essential to the survival and reproductive success of their offspring. In reindeer (Rangifer tarandus), for example, dominant females monopolize access to superior foraging patches, gain weight during the winter, and consequently breed earlier and more often, producing calves that grow faster than those of subordinate females [2]. Cooperatively breeding animals exhibit more overt forms of competition, whereby dominant females and other non-breeding subordinate females aggressively suppress subordinate female reproduction through direct aggression and infanticide [3].
Spontaneous abortion in response to social cues can be a strategy for a female to save time and resources, in order to raise future offspring with better survival probabilities [4]. For example, in rodents, females commonly abort or reabsorb fetuses in the presence of a new unknown male (the ‘Bruce effect’ [5,6]). Spontaneous abortions have also been found to increase in wild female geladas (Theropithecus gelada) if the dominant male in the group is replaced [7]. We investigated whether patterns of abortion reflect the intensity of reproductive conflict in wild banded mongooses (Mungos mungo) in which offspring of several females compete for food and access to adult helpers or ‘escorts’ in communal litters [8]. We made two predictions. First, we predicted that abortion should be more frequent in breeding attempts with more intense reproductive competition, as measured by female group size and resource abundance. Second, within breeding attempts, we predicted that lower-rank females, females in poorer condition and those with smaller fetuses should be more likely to spontaneously abort, particularly when competition is high [8,9].
2. Material and methods
(a). Study population
We collected data from a wild population of banded mongooses living on and around the Mweya Peninsula, Queen Elizabeth National Park, Uganda (0°12′ S, 29°54′ E) between September 1999 and February 2015. For a detailed description of the climate, habitat and the population, see [10]. All individuals in the population are individually identified, and groups were visited daily to determine pregnancy, parturition and abortion dates. Pregnancy was confirmed by ultrasound scans and palpation of the abdomen during routine capture, and parturition and abortion detected from subsequent rapid changes in body shape and weight (for details of capture and anaesthesia, see [11]). Abortions were defined as confirmed pregnancies that lasted less than 60 days (mean gestation period [12]) and produced no viable pups. There were also nine direct observations of spontaneous abortion of unviable neonates. In the banded mongoose, groups of individuals are periodically evicted from their natal group, and the violent eviction events are known to increase the risk of abortion [13]. Therefore, to focus on spontaneous pregnancy loss, abortions from breeding attempts where an eviction event occurred were excluded from these analyses.
(b). Predictors of abortion
Rainfall in the study site correlates with invertebrate abundance (e.g. [14]), so cumulative rainfall for 60 days before the birth of the communal litter was used as a proxy for resource abundance during gestation. In the banded mongoose, pups born in communal litters compete for helpers postnatally, and the breeding success of dominant females declines with increasing female group size [15]. Probability of violent evictions where older females expel younger females also increases with group size [15,16], and the per capita reproductive success of the remaining females increases after eviction events [17], so the number of adult (over 1 year of age) females was used as a proxy of the intensity of reproductive competition. Ranked age (range 1–11, 1 = oldest female in the group) which describes vulnerability to eviction [9] was used as proxy of dominance status, by dividing it by the number of adult females in the group to get a relative rank score (range 0.09–1) that is comparable across different group sizes. Adult weight varies with rainfall and availability of invertebrate prey (e.g. [18]) and it predicts survival [18] as well as competitive ability [19]. Therefore, female weight on the closest weighing event prior to the estimated conception date was used as a proxy for female condition. Fetus size was measured from ultrasound scans (see [19] and electronic supplementary material) for the subset of data where this was available.
(c). Statistical analyses
Available data varied across females and breeding attempts, so in order to maximize sample size the analysis was conducted in three stages (see electronic supplementary material for full details, and [20] for the full datasets used in the analyses). First, we looked at whether any abortions occurred in a breeding event (Y/N, N = 461 communal litters from 18 groups; breeding events where an eviction occurred were left out), with rainfall during gestation (mm) and female group size as predictors; female group size was also fitted as a quadratic term, to allow for possible negative effects of both small and large group size on probability of abortion. Second, we looked at individual-level predictors of a particular female aborting (Y/N) in 207 confirmed pregnancies from 93 females in eight social groups, within the 57 litters in which abortions occurred and for which we had complete data for the predictor variables: female weight at conception (g), relative age rank, primiparity (Y/N), number of females and rainfall during gestation (mm). We included two-way interactions between weight and rainfall, female group size and rank, to test for condition-specific effects of resource levels and competitive environment. Third, we ran a separate model of whether a particular female aborted (Y/N), with average fetus size (mm2), relative age rank, number of females and two-way interactions between weight and rank, weight and female group size, and weight and fetus size as predictors, in the subset of data (76 pregnancies from 17 females, eight litters, six groups) where fetus size measurements were available; fetus gestational age was included as a covariate to control for capture at different times during pregnancy. Generalized linear mixed models (GLMMs) with a binomial error structure and logit link function were fitted using lme4 package [21] in R version 3.1.0 [22]. Group was included as a random factor in all analyses, and female and litter identity in the individual-level analyses (see electronic supplementary material for details). The significance of each fixed effect was assessed by comparing the likelihood ratio of the maximal model to that of the model without the fixed effect [21]. Non-significant interactions were removed to allow the significance of the main effects to be tested [23], but models were not simplified further, to avoid problems associated with stepwise model reduction (e.g. [23,24]).
3. Results
Abortions occurred in 133 (29%) out of 461 group breeding attempts, with 2.06 ± 1.26 (mean ± s.d.) females aborting in each. Out of 830 pregnancies that were confirmed by palpation and ultrasound, 361 (43%) were not carried to term. This is a conservative estimate of the overall abortion rate: most abortions occurred in the second trimester (average 43 days) and those occurring before pregnancy could be confirmed (less than 30 days) would go undetected.
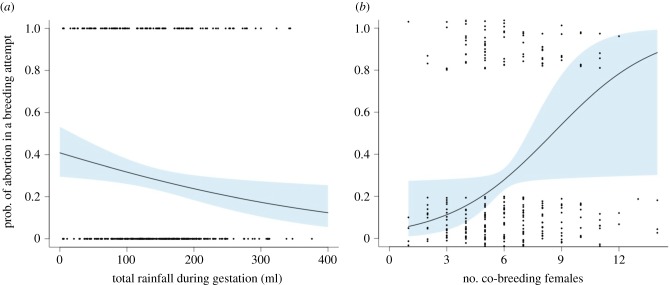
As predicted, among litters, abortion was more likely when rainfall was low (β ± s.e. = −0.272 ± 0.110, , p = 0.012; figure 1a) and when more females were co-breeding (β ± s.e. = 0.372 ± 0.180, , p = 0.033; figure 1b); the quadratic effect of female group size was not significant (β ± s.e. = −0.023 ± 0.013, , p = 0.065). Within litters, abortion probability was higher for females of lower rank, particularly in females that also had lower weight at conception (interaction weight × rank: β ± s.e. = −1.097 ± 0.552, , p = 0.042; figure 2a; all other terms p > 0.19, see electronic supplementary material, table S2). Females with larger fetuses were less likely to abort, and abortion probability declined with increasing fetus size more steeply in lighter than in heavier females (β ± s.e. = 0.025 ± 0.011, , N = 76, p = 0.002; figure 2b). No other terms predicted abortion probability at the individual level (electronic supplementary material, table S3).
Figure 1.
Litter-level predictors of abortion in wild banded mongooses. Abortions were more likely to occur in a given breeding attempt (a) when rainfall was low, and (b) in breeding attempts under more intense reproductive competition (when more females were co-breeding). The line and shaded area represent model predictions ±s.e. from a binomial GLMM, accounting for the random effect of group identity, and the points are raw data (jittered for clarity in b). (Online version in colour.)
Figure 2.
Individual-level predictors of abortion probability in wild banded mongooses. For a given female, in breeding attempts where abortions occurred, individual probability of abortion (a) increased with decreasing relative rank (1 = lowest rank), particularly in light females (blue) as compared to heavier females (red), and (b) decreased more steeply with increasing fetus size in light females (blue) than in heavy females (red). The lines and shaded areas represent model predictions ±s.e. from binomial GLMMs, plotted for light (25% quartile = 1200 g: dotted line, blue area) and heavy females (75% quartile = 1550 g: solid line, red area) after accounting for the random effects of female, litter and group identity. Dots are raw datapoints, jittered for clarity. Note that in the analyses, female weight was used as a continuous predictor, and weight categories are drawn for illustration only.
4. Discussion
Spontaneous abortion was common in banded mongooses, with 43% of detected pregnancies not carried to term. Equally high reproductive failure rates have been found in other mammals (e.g. [25]). Across litters, abortions were more common during dry periods, when invertebrate prey is scarce, suggesting that resource limitation is an important determinant of pregnancy outcome. However, controlling for the effect of rainfall, the probability of abortion also increased with increasing female group size, suggesting that reproductive competition is also an important determinant of abortion risk. Reproductive competition is intense among female banded mongooses and is manifested in conspicuous and violent behaviour such as infanticide and eviction of reproductive rivals. Our results show that conflict over reproduction may be resolved in subtle ways that are more difficult to detect, through termination of pregnancy before offspring are produced.
On an individual level, similar to red squirrels (Sciurus vulgaris [26]) and caribou (Rangifer tarandus [27]), female condition (measured as weight at conception) in conjunction with the females' social rank predicted abortion probability. As predicted, relatively young females were more likely to abort, and this effect of age rank on abortion probability was amplified for females that were in poor condition. We know from previous work that younger and lighter females are particularly responsive to reproductive competition, producing larger fetuses when female group size is large [19]. In this study, we have shown that larger fetuses are less likely to be aborted, and this relationship is particularly steep for lighter females. Taken together, these findings support the idea that those females that cannot (or do not) respond to reproductive competition by increasing their fetus size instead abort their litter, conserving resources for future reproductive events that are more likely to succeed. It is also the case that lighter and younger females are more likely to forego reproduction entirely, particularly when resources are scarce [28]. Our results highlight that reproductive conflict among females may often be resolved in subtle and complex ways, long before offspring are produced and without any associated aggression or agonistic behaviour.
At the study site, rainfall correlates with invertebrate prey abundance, and dry periods are linked with increased post-natal competition for food and access to helpers [8,14,18]. In such circumstances, low ranking and light females may gain from aborting their pregnancy to reallocate resources to a future breeding attempt in more benign and less competitive conditions [25], especially as their offspring tend to be smaller at birth and are particularly susceptible to infanticide by older, socially dominant females [12,15]. Breeding may also entail higher costs to lighter or less experienced females, and disproportionately compromise their survival or future reproduction [29,30]. The long-term fitness consequences of abortion remain unknown at present, but overall our results suggest that spontaneous abortion patterns may evolve as a response to potentially costly female reproductive conflict.
Supplementary Material
Acknowledgements
We are grateful to N. Guma, E. Asalu, M. Driciru and the wardens and staff of Queen Elizabeth National Park for continuous logistical support. We thank Francis Mwanguhya, Solomon Kyabulima, Kenneth Mwesige, Robert Businge and Solomon Ahabyona for assistance in the field, and previous Banded Mongoose Research Project researchers, particularly Jason Gilchrist, Matt Bell and Sarah Hodge and Neil Jordan, for their contribution to the long-term data. We thank four anonymous reviewers whose detailed comments greatly improved this manuscript.
Ethics
A previous study found no adverse effects of trapping and anaesthesia on pregnant females [31], and all methods were carried out in accordance with the Association for the Study of Animal Behaviour (ASAB) guidelines. All fieldwork was conducted under permissions from Uganda Wildlife Authority (UWA) and Uganda National Council for Science and Technology (UNCST), and animal research methods approved by the ethical review panel of the University of Exeter.
Data accessibility
Data supporting this manuscript are available on Figshare, at https://doi.org/10.6084/m9.figshare.5572408.v1 [20].
Authors' contributions
M.A.C. and E.I. conceived the study and designed it with guidance from G.K.-Z.; E.I., E.I.K.V., H.H.M. and F.J.T. conducted and interpreted analyses. E.I., E.I.K.V. and M.A.C. prepared the manuscript and G.K.-Z., H.H.M. and F.J.T. contributed to critical revision; all authors approved the final version and agree to be held accountable for the content therein.
Competing interests
We declare we have no competing interests
Funding
The study was funded by NERC (grant nos. NE/E015441/1; NE/G019657/1; NE/J010278/1) and an ERC grant (grant no. 309249) to M.A.C.
References
- 1.Stockley P, Bro-Jørgensen J. 2011. Female competition and its evolutionary consequences in mammals. Biol. Rev. 86, 341–366. ( 10.1111/j.1469-185X.2010.00149.x) [DOI] [PubMed] [Google Scholar]
- 2.Holand Ø, Gjøstein H, Losvar A, Kumpula J, Smith ME, Røed KH, Nieminen M, Weladji RB. 2004. Social rank in female reindeer (Rangifer tarandus): effects of body mass, antler size and age. J. Zool. 263, 365–372. ( 10.1017/S0952836904005382) [DOI] [Google Scholar]
- 3.Creel SR, Creel NM. 1991. Energetics, reproductive suppression and obligate communal breeding in carnivores. Behav. Ecol. Sociobiol. 28, 263–270. [Google Scholar]
- 4.Stearns SC. 1992. The evolution of life histories. Oxford, UK: Oxford University Press. [Google Scholar]
- 5.Bruce HM. 1959. An exteroceptive block to pregnancy in the mouse. Nature 184, 105 ( 10.1038/184105a0) [DOI] [PubMed] [Google Scholar]
- 6.Mahady SJ, Wolff JO. 2002. A field test of the Bruce effect in the monogamous prairie vole (Microtus ochrogaster). Behav. Ecol. Sociobiol. 52, 31–37. ( 10.1007/s00265-002-0484-0) [DOI] [Google Scholar]
- 7.Roberts EK, Lu A, Bergman TJ, Beehner JC. 2012. A Bruce effect in wild geladas. Science 335, 1222–1225. ( 10.1126/science.1213600) [DOI] [PubMed] [Google Scholar]
- 8.Hodge SJ, Bell MBV, Mwanguhya F, Kyabulima S, Waldick RC, Russell AF. 2009. Maternal weight, offspring competitive ability, and the evolution of communal breeding. Behav. Ecol. 20, 729–735. ( 10.1093/beheco/arp053) [DOI] [Google Scholar]
- 9.Cant MA, Nichols HJ, Johnstone RA, Hodge SJ. 2014. Policing of reproduction by hidden threats in a cooperative mammal. Proc. Natl Acad. Sci. USA 111, 326–330. ( 10.1073/pnas.1312626111) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cant MA, Vitikainen E, Nichols HJ. 2013. Chapter Six—Demography and Social Evolution of Banded Mongooses. Adv. Study Behav. 45, 407–445. ( 10.1016/B978-0-12-407186-5.00006-9) [DOI] [Google Scholar]
- 11.Jordan NR, Mwanguhya F, Kyabulima S, Ruedi P, Cant MA. 2010. Scent marking within and between groups of wild banded mongooses. J. Zool. 280, 72–83. ( 10.1111/j.1469-7998.2009.00646.x) [DOI] [Google Scholar]
- 12.Cant MA. 2000. Social control of reproduction in banded mongooses. Anim. Behav. 59, 147–158. ( 10.1006/anbe.1999.1279) [DOI] [PubMed] [Google Scholar]
- 13.Gilchrist JS. 2006. Female eviction, abortion, and infanticide in banded mongooses (Mungos mungo): implications for social control of reproduction and synchronized parturition. Behav. Ecol. 17, 664–669. ( 10.1093/beheco/ark012) [DOI] [Google Scholar]
- 14.Marshall HH, et al. 2017. Lifetime fitness consequences of early-life ecological hardship in a wild mammal population. Ecol. Evol. 57, 1–13. ( 10.1002/ece3.2747) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cant MA, Hodge SJ, Bell MBV, Gilchrist JS, Nichols HJ. 2010. Reproductive control via eviction (but not the threat of eviction) in banded mongooses. Proc. R. Soc. B 277, 2219–2226. ( 10.1098/rspb.2009.2097) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thompson FJ, Marshall HH, Sanderson JL, Vitikainen EIK, Nichols HJ, Gilchrist JS, Young AJ, Hodge SJ, Cant MA. 2016. Reproductive competition triggers mass eviction in cooperative banded mongooses. Proc. R. Soc. B 283, 20152607 ( 10.1098/rspb.2015.2607) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thompson FJ, Marshall HH, Vitikainen EIK, Cant M. 2017. A causes and consequences of intergroup conflict in cooperative banded mongooses. Anim. Behav. 126, 31–40. ( 10.1016/j.anbehav.2017.01.017) [DOI] [Google Scholar]
- 18.Marshall HH, et al. 2016. Variable ecological conditions promote male helping by changing banded mongoose group composition. Behav. Ecol. 27, 978–987. ( 10.1093/beheco/arw006) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Inzani EL, Marshall HH, Sanderson JL, Nichols HJ, Thompson FJ, Kalema-Zikusoka G, Hodge SJ, Cant MA, Vitikainen EIK. 2016. Female reproductive competition explains variation in prenatal investment in wild banded mongooses. Sci. Rep. 6, 20 013–20 016. ( 10.1038/srep20013) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Inzani EL, Marshall HH, Thompson FJ, Kalema-Zikusoka G, Cant MA, Vitikainen EIK. 2019. Data from: Spontaneous abortion as a response to reproductive conflict in the banded mongoose FigShare Digital Depository. ( 10.6084/m9.figshare.5572408.v1) [DOI] [PMC free article] [PubMed]
- 21.Bates D, Mächler M, Bolker B, Walker S. 2015. Fitting linear mixed-effects models using lme4. J. Stat. Soft. 67, 1–48. ( 10.18637/jss.v067.i01) [DOI] [Google Scholar]
- 22.R Core Team. 2016. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. [Google Scholar]
- 23.Engqvist L. 2005. The mistreatment of covariate interaction terms in linear model analyses of behavioural and evolutionary ecology studies. Anim. Behav. 70, 967–971. ( 10.1016/j.anbehav.2005.01.016) [DOI] [Google Scholar]
- 24.Forstmeier W, Schielzeth H. 2011. Cryptic multiple hypotheses testing in linear models: overestimated effect sizes and the winner's curse. Behav. Ecol. Sociobiol. 65, 47–55. ( 10.1007/s00265-010-1038-5) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wasser SK, Barash DP. 1983. Reproductive suppression among female mammals: implications for biomedicine and sexual selection theory. Q. Rev. Biol. 58, 513–538. ( 10.1086/413545) [DOI] [PubMed] [Google Scholar]
- 26.Wauters L, Dhondt AA. 1989. Body weight, longevity and reproductive success in red squirrels (Sciurus vulgaris). J. Anim. Ecol. 58, 637 ( 10.2307/4853) [DOI] [Google Scholar]
- 27.Cameron RD, Smith WT, Fancy SG, Gerhart KL, White RG. 2011. Calving success of female caribou in relation to body weight. Can. Zool. 71, 480–486. ( 10.1139/z93-069) [DOI] [Google Scholar]
- 28.Nichols HJ, Bell MBV, Hodge SJ, Cant MA. 2012. Resource limitation moderates the adaptive suppression of subordinate breeding in a cooperatively breeding mongoose. Behav. Ecol. 23, 635–642. ( 10.1093/beheco/ars008) [DOI] [Google Scholar]
- 29.Hanssen SA, Hasselquist D, Folstad I, Erikstad KE. 2005. Cost of reproduction in a long-lived bird: incubation effort reduces immune function and future reproduction. Proc. R. Soc. B 272, 1039–1046. ( 10.1098/rspb.2005.3057) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Reid JM, Bignal EM, Bignal S, McCracken DI, Monaghan P. 2003. Age-specific reproductive performance in red-billed choughs Pyrrhocorax pyrrhocorax: patterns and processes in a natural population. J. Anim. Ecol. 72, 765–776. ( 10.1046/j.1365-2656.2003.00750.x) [DOI] [Google Scholar]
- 31.Gilchrist JS, Otali E, Mwanguhya F. 2004. Why breed communally? Factors affecting fecundity in a communal breeding mammal: the banded mongoose (Mungos mungo). Behav. Ecol. Sociobiol. 57, 119–131. ( 10.1007/s00265-004-0837-y) [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Inzani EL, Marshall HH, Thompson FJ, Kalema-Zikusoka G, Cant MA, Vitikainen EIK. 2019. Data from: Spontaneous abortion as a response to reproductive conflict in the banded mongoose FigShare Digital Depository. ( 10.6084/m9.figshare.5572408.v1) [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Data supporting this manuscript are available on Figshare, at https://doi.org/10.6084/m9.figshare.5572408.v1 [20].