Abstract
A 53-year-old man presented with a 3-month history of progressive, non-productive cough followed by occasional swelling of the face and upper extremities. Physical examination on admission revealed prominent superficial vessels at the neck and upper extremity swelling. Bronchoscopy revealed the superior segment of the right lower lobe was narrow but without visible mass; cell block and biopsy done revealed signet-ring cell carcinoma with an immunohistochemistry pattern favouring the primary site of malignancy as either gastric or of the pancreaticobiliary tree. However, CT scan of the abdomen and pelvis with triple contrast revealed only slight gastric wall thickening; the pancreas was unremarkable. The patient underwent radiotherapy with noted improvement of symptoms. He was then discharged with plans for further work-up on an outpatient basis. This work highlights the importance of a comprehensive histopathological and immunohistochemical work-up which can help predict a site of origin to be able to provide the appropriate management.
Keywords: cancer intervention, lung cancer (oncology), radiotherapy, palliative procedures, pathology
Background
It is estimated that 15,000 people in the USA each year, with an incidence of 1 in 650 to 1 in 3100 patients, are diagnosed with superior vena cava (SVC) Syndrome.1 It is characterised by elevated venous pressure upstream brought about by the SVC obstacle, impaired venous return to the heart from the upper body (from the head to the upper extremities) and the formation of venous shunts and collateral vessels to bypass the obstacle.2 Patients present with face, neck and/or upper extremity swelling, engorged neck veins, cough, dyspnoea, orthopnoea, distended anterior chest vein collaterals and conjunctival suffusion. The diagnosis is usually made clinically along with imaging, such as a CT of the chest, that, along with the presence of collateral vessels, is sensitive and specific at 96% and 92%, respectively.3 Most of the SVC syndromes are caused by mediastinal malignancies, most of which are small cell bronchogenic carcinomas. The next most common cause is non-Hodgkin lymphoma followed by metastatic tumours. Furthermore, benign causes of SVC syndrome now consist of at least 40% of cases. The formation of a thrombus or stenosis from the placement of pacemaker wires and semipermanent intravascular catheters used for chemotherapy is increasing in incidence; these also cause the syndrome.4
The treatment options include percutaneous stent placement, corticosteroids, radiotherapy and chemotherapy. Thrombolytics and anticoagulation are indicated in cases when the obstacle is severe enough that it causes stasis and, eventually, formation of a thrombus. Treatment options for symptomatic relief are diuresis and elevating the head of the patient. The definitive treatment is based on histopathological diagnosis. However, randomised clinical trials on which to base ‘best therapy’ are still lacking.5–7
Despite the advancement of the technology to diagnose and work up malignancies, doctors still are unable to reveal a primary site in about 3%–5% of metastatic tumours.8 Signet-ring cell carcinoma (SRCC) is rare and poorly differentiated with a poor prognosis. Around 90% of these tumours arise from the stomach, breast or colon; however, almost every organ in the body could be the primary site. The characteristic histology of SRCCs is signet-ring cells in which the nucleus is pushed to the periphery by abundant intracytoplasmic mucin. SRCCs pose difficulties in the work-up and management since they are usually aggressive and present at an advanced stage.9 The majority of SRCCs originate from the gastrointestinal tract; thus, findings that point to a diagnosis of peritoneal carcinomatosis or omental ‘caking’ on CT imaging require immediate work-up that usually involves sending samples for cytological evaluation of peritoneal fluid, imaging, colonoscopy and upper endoscopy.10
There have been recent developments in terms of diagnostic tools, but in some patients with metastatic cancer, the site of the primary tumour still cannot be determined. These patients are defined as having cancer of unknown primary (CUP), and this is associated with a poor prognosis.11 CUP accounts for approximately 3%–5% of all cancer diagnoses. The most commonly reported subtype of CUP is adenocarcinoma.12 Although SRCC is a poorly differentiated aggressive subtype of adenocarcinoma, SRCC of unknown primary origin has rarely been reported.
Case presentation
A 53-year-old man, non-smoker, non-alcoholic beverage drinker, non-spicy food consumer, with no history of gastrointestinal diseases, presented to our hospital with a 3-month history of progressive, non-productive, on-and-off cough followed by upper back pain and occasional swelling of the face and upper extremities, with noted engorgement of neck veins. No anorexia, sudden weight loss, dyspnoea, nor fever was noted. He had consulted with a local doctor and was given levodropropizine and tramadol, which provided temporary relief. His symptoms worsened and were no longer relieved by the previously prescribed medications; thus, he sought consultation at the emergency department of a local hospital. A chest X-ray revealed a right paratracheal mass with right-sided pleural effusion. A diagnostic thoracentesis was done, which revealed findings consistent with metastatic adenocarcinoma, and thus, he was referred to our institution for further work-up and management. He had type 2 diabetes mellitus and stage 2 hypertension. He was a non-smoker, non-alcohol beverage drinker and non-illicit drug user, and occasionally drinks caffeinated and carbonated beverages. Family history was unremarkable for malignancy. He works as a farmer who occasionally used charcoal production through burning coconut husks as an additional means to make money.
Physical examination at the emergency department on admission revealed vital signs of 180/100 mm Hg, heart rate of 85 beats/min, respiratory rate of 19 breaths/min, and an oxygen saturation of 98%; distended neck veins, prominent superficial vessels at the neck, decreased breath sounds with decreased vocal fremiti at the base of the right lung and upper extremity swelling; the rest of physical examination was unremarkable.
The patient was managed as a case of SVC syndrome likely from the right paratracheal mass, which was probably a metastasis or a primary malignancy. Hydrocortisone 100 mg intravenous q8 was given, which afforded relief to the patient. He was also started on antihypertensives and antihyperglycaemics. Clinical chemistry tests and cultures were obtained. The patient underwent a CT scan of the abdomen with triple contrast to work-up for a possible primary or metastasis. He ultimately underwent a bronchoscopy to obtain samples for biopsy, then underwent three sessions of radiotherapy.
Investigations
His complete blood count, serum creatinine, lipid profile and coagulation tests were unremarkable. Urinalysis showed glucosuria, which was consistent with the patient’s condition of type 2 diabetes mellitus, along with his haemoglobin A1c of 9.4% and elevated random blood sugar. A CT of the chest with contrast done 1 month prior to admission revealed a 3.4×3.5×2.7 cm fairly defined, moderately enhancing mass lesion at the right lower lung, compressing the posterior aspect of the right lower bronchus, with associated atelectasis, fissural thickening and perilesional ground glass densities, prominent carinal lymph node with calcification and multiple subcentimetre right cervical and axillary lymph nodes were also noted, and a 3.7×4.3×4.5 cm heterogenous mildly enhancing mass in the right paratracheal region with associated mass effect. A repeat CT of the chest with contrast done 2 weeks later revealed no significant change in size of right paratracheal mass with compression of SVC; however, there was an increase in right pleural effusion with minimal left pleural effusion and an elevated right hemidiaphragm. A thoracentesis was done prior to admission, and pleural fluid studies revealed 8 mL yellow, cloudy, honey-coloured serous fluid. Pleural fluid cytology and cell block revealed rare atypical cells with specific gravity of 1.010; white blood cell count of 1500/µL; red blood cell count of 750/µL; polymorphonuclear cells, 5%; lymphocytes, 90%; monocytes, 5%; glucose, 12.69 mg/dL; total protein, 41.5 mg/dL; and lactate dehydrogenase, 140 U/L, consistent with an exudative picture.
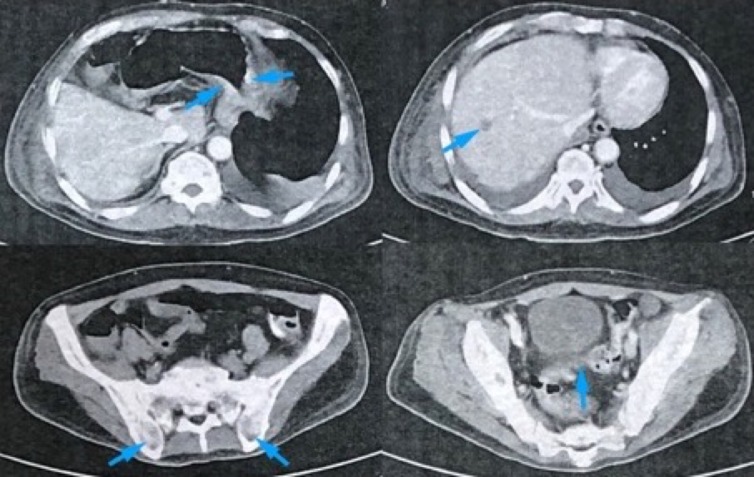
A CT of the abdomen and pelvis with triple (intravenous, oral and rectal) contrast was done, which revealed multiple fairly defined hypodense, peripherally enhancing nodular foci of different sizes in the bilateral psoas muscles, both gluteal muscle groups, paravertebral and bilateral proximal medial thigh muscles. The largest of these, measuring 3.3×1.6×1.3 cm with an approximate volume of 6.9 cc, was noted in the right psoas muscle. There was a slight diffuse thickening of the gastric fundus and sigmoid colonic walls (figure 1). There were noted hypodense nodules at the liver, right adrenal gland and spleen. Enlarged lymph nodes with necrotic centres were noted in the left iliac and superficial inguinal regions. A well-defined 0.7 cm partially exophytic, thinly septated hypodense cystic focus is seen in the cortex of the superior pole of the right kidney. Pericardial thickening, worrisome for metastasis, was appreciated. Subcutaneous fat stranding was noted in the anterolateral chest wall and in the right and left flank muscles also. Multiple fairly to well-defined areas of lysis were noted in the sacrum, bilateral iliac, right ischium, proximal femurs, and some of the posterior elements and bodies of the visualised vertebrae. An ill-defined enhancing isodense mass lesion measuring 3.0×3.2 cm is noted at the right parahilar region.
Figure 1.
An abdominal CT scan with triple contrast top to bottom (clockwise, blue arrows): slight gastric thickening at the fundus, hypodense nodule at the liver, well-defined areas of lysis at the iliac bones and thickening of the sigmoid colonic wall.
A bronchoscopy done revealed a pale and stunted trachea with multiple white nodules in the carina. The superior segment of the right lower lobe (B6) was narrow but without visible mass nor irregularities noted. Bronchial alveolar lavage was done, and samples were sent for further diagnostic studies.
We sent blood, sputum, and bronchial washing specimens for gram staining, cultures and acid-fast bacilli studies. These tests all turned out to be negative. The patient’s procalcitonin level was 0.05 ng/mL (reference value 0–0.25 ng/mL). Tumour marker carcinoembryonic antigen (CEA) was 28.69 ng/mL (reference value less than 5) ng/mL and CA 19–9 was 198.51 IU/mL (reference value less than 37 IU/mL).
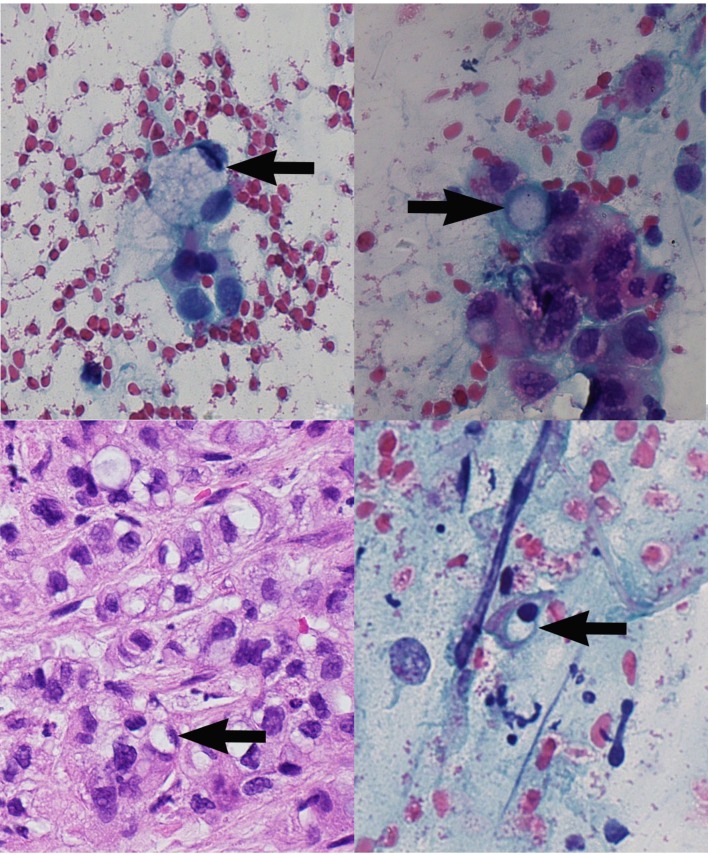
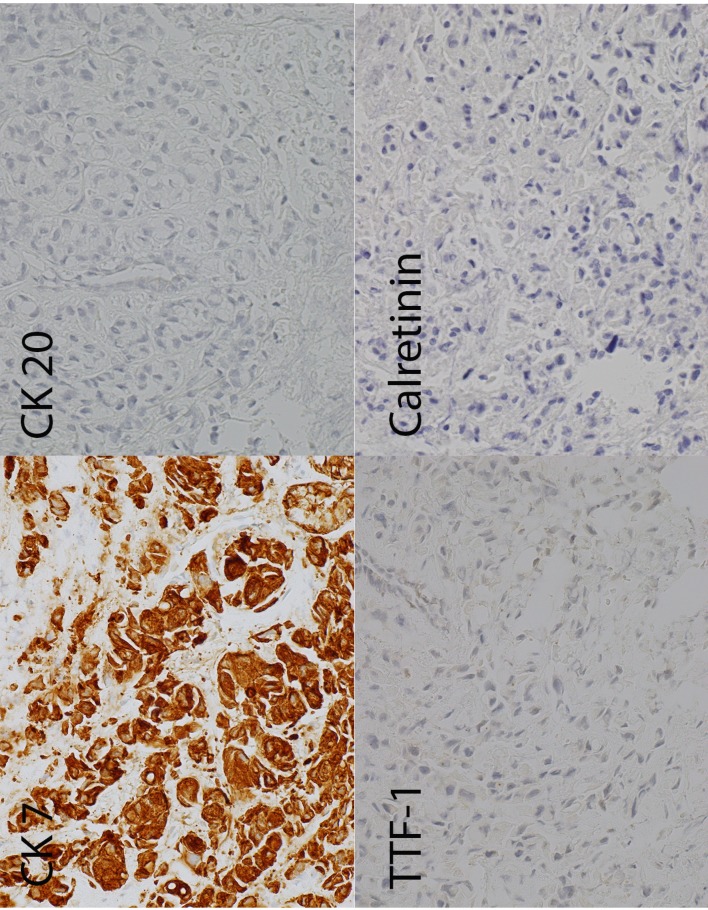
Bronchial alveolar lavage cytology stained with Papanicolaou smear and DiffQuick showed a fairly cellular smear. Cells were atypical, characterised by high nuclear to cytoplasmic ratio and pleomorphism. Some of the cells showed a signet-ring pattern, characterised by the presence of a mucin inclusion pushing the nucleus to the periphery (figure 2). The biopsy also showed similar findings. Given these features, the case was signed out as cell findings consistent with metastatic adenocarcinoma with signet-ring features. To ascertain the possible primary site of the tumour, a panel of immunohistochemistry studies was requested. The panel consisted of cytokeratin-7 (CK-7), cytokeratin-20 (CK-20), thyroid transcription factor-1 (TTF-1) and calretinin. Figure 3 shows the results of the immunostaining; this is consistent with a metastatic disease of gastric or pancreaticobiliary origin.
Figure 2.
Signet-ring cell carcinoma (×40). Top to bottom (clockwise, black arrows): (A) bronchial alveolar lavage specimen (H&E), immunohistochemistry; (B–D) bronchial alveolar lavage specimen (Papanicolau stain).
Figure 3.
Immunohistochemistry (clockwise top to bottom): the tumour cells are diffusely positive for CK-7, negative for CK-20, negative for TTF-1 and negative for calretinin. CK-7, cytokeratin-7; CK-20, cytokeratin-20; TTF-1, thyroid transcription factor-1.
Differential diagnosis
The patient came in with cough, engorged neck veins, swelling of the extremities with a chest X-ray showing a right paratracheal mass. A diagnostic thoracentesis was done in the previous hospital with findings consistent with metastatic adenocarcinoma. The initial consideration at the time was that the patient was in SVC syndrome; the challenge was determining if the mass was indeed metastatic or the primary. If ever it was metastatic, we had to determine the site of origin.
The patient underwent a CT of the chest with contrast, and with the pulmonary medicine specialist, we decided to do a fiberoptic bronchoscopy to visualise the pulmonary tree and also to obtain specimen for a biopsy. The biopsy was done, and the findings revealed metastatic signet-ring cell adenocarcinoma. We then decided to move forward with immunohistochemistry (IHC) staining to narrow down our differentials as to the site of origin, since the site of origin could be from the stomach, thyroid, a mesothelioma, pancreas or lung among others.
The IHC done was CK-7, CK-20, TTF-1 and calretinin. The sample only stained positive for CK-7. This led the us to believe that the site of origin was from the thyroid, a mesothelioma or gastric. Since the TTF-1 was negative, a thyroid primary was highly unlikely, and it stained negative for calretinin, which made mesothelioma also an unlikely site of origin. The CT of the abdomen and pelvis with contrast also only showed slight thickening of the gastric mucosa. This made a gastric primary also unlikely. After reviewing the criteria for diagnosing CUP, we decided to call the site of origin unknown in this case.
Treatment
The patient subsequently underwent radiation therapy daily for the next 3 days with 400 cGy each session with noted improvement of symptoms. The patient was also given enalapril and amlodipine for blood pressure control, insulin glargine for blood glucose control and dexamethasone, which alleviated the dyspnoea.
Outcome and follow-up
He was then discharged with plans for further work-up in locating the origin of the metastasis on an outpatient basis, and then for possible chemotherapy tailor-fit for him, but he was lost to follow-up.
Discussion
SVC syndrome is a medical emergency. The clinical presentation varies, which depends on the severity of the compression and the extent of collateral vessel formation; it is gradual, initially presenting with mild facial and upper extremity oedema to immediate and urgent with increased intracranial pressure, haemodynamic instability and upper airway obstruction.1 The subject of this case report presented with chronic intermittent, non-productive cough, facial plethora, upper extremity swelling and superficial blue vessels on the skin surface venous on the chest wall. A chest CT with intravenous contrast confirmed the diagnosis, which was due to external compression by a right paratracheal mass. A bronchoscopy was done, and bronchial alveolar lavage specimen were sent for biopsy and immunohistochemical staining. The biopsy was consistent with metastatic adenocarcinoma with signet-ring features. The immunohistochemistry done revealed the tumour cells to be diffusely positive for CK-7 and negative for CK-20, thyroid transcription factor - 1 and calretinin. Chemotherapy, radiation or intravascular stents are the mainstays of treatment.
A common problem in cases like this is being able to differentiate between primary pulmonary adenocarcinoma from adenocarcinoma metastatic to the lung. It is important to do so because the treatment and prognosis differ considerably. A useful method is by means of immunohistochemistry. Published studies have demonstrated that the combination of a positive CK-7, negative CK-20 immunophenotype, along with a positive TTF-1, was highly specific for primary pulmonary adenocarcinoma (specificity 100%).13 14 In a study done by Merchant et al, they investigated 17 cases of lung signet-ring cell adenocarcinoma. They showed that 82.4% (14 out of 17 cases) showed TTF-1 positivity, and a CK-7+/CK-20− pattern was seen in 94% (16/17).15 This was also the case in a study by Terada when a case of a primary SRCC of the lung was investigated by using immunohistochemical studies with a similar immunohistochemistry profile.16 A case of a primary pure signet-ring cell adenocarcinoma of the left main bronchus was reported by Terada et al. In this study, the immunohistochemical profile was positive for CK-7 and negative for CK-20, the TTF-1 was also negative. This meant that the site of origin is not of the lung. This is consistent with their bronchoscopy finding of a tumour in the extrapulmonary left main bronchus, which led them to conclude that that was the site of origin.17
A study by Moran showed that the primary lung tumours will react strongly with CK-7 and TTF-1, while those of gastrointestinal in origin will only be positive for CK-20 and CDX2.18 The findings in the studies mentioned are consistent with the statement by Ordóñez that TTF-1 is a specific transcription factor expressed in epithelial cells of the thyroid, lung and brain.19 A study done by Moldvay et al used TTF-1 on formalin-fixed, paraffin-embedded tissues of patients with primary bronchial adenocarcinomas; it stained positive for TTF-1 in 46/50 cases; among them, 30 cases revealed strong nuclear immunostaining. Furthermore, out of the metastatic adenocarcinomas, only two thyroid cancers were negative.20 This makes TTF-1 a useful immunohistochemical marker in the diagnosis of tumours originating from the thyroid or lung compared with those originating from other parts of the body.
In another study done by Castro et al, they investigated 15 cases of patients with primary signet-ring cell adenocarcinoma of the lung. The immunohistochemical staining done revealed that TTF-1 was positive in all cases, while three out of the six cases were only positive for CK-7 and all were negative for CK-20.21 This study shows us that while immunohistochemistry is a reliable tool in differentiating between primary and metastatic sources of signet-ring cell adenocarcinoma, it is not absolute, and that the use of immunohistochemistry must be coupled with other diagnostic tools to locate the site of origin.
Immunohistochemical studies done for this case are CK-7, CK-20, TTF-1 and calretinin. CK-20 is a low-molecular-weight cytokeratin that is normally expressed in the gastrointestinal epithelium, urothelium and Merkel cells. CK-7 is usually expressed by tumours of the lung, ovary, endometrium and breast, but not in tumours of the lower gastrointestinal tract. A combined CK-7+/CK-20− profile would suggest a primary site from the breast, thyroid, salivary glands, pancreatic, biliary tree and subset of gastric origin.22 Calretinin is a calcium-binding protein that is used to differentiate epithelioid pleural mesothelioma (+) and lung adenocarcinoma (−); it was ordered in this case to rule out a possible mesothelioma, which can have a CK-7+/CK-20− profile.23 TTF-1 is expressed in lung adenocarcinomas and thyroid carcinomas but not in adenocarcinomas arising from other sites. For patients with adenocarcinomas in the lung, TTF-1 staining is now routinely used to distinguish a primary lung cancer from a lung metastasis for which, in this case, was negative.24 This is summarised in table 1.
Table 1.
Immunohistochemical staining pattern for primary versus metastatic lung adenocarcinoma32
| Immunohistochemical stain | Primary lung adenocarcinoma | Metastatic adenocarcinoma |
| CK-7 | + | +/– |
| CK-20 | – | +/– |
| TTF-1 | + | +/– |
| Calretinin | – | – |
CK-7, cytokeratin-7; CK-20, cytokeratin-20; TTF-1, thyroid transcription factor-1.
A study by Tot evaluated 93 autopsy cases of patients with adenocarcinoma of a known primary site; 81% (17 out of 21) who had a colorectal primary revealed a CK-20-positive and CK-7-negative pattern and was later on proven to be accurately predicted. Furthermore, a pattern of CK-20-positive and CK-7-positive accurately predicted a cancer of pancreaticobiliary origin in 79% of the cases (11 out of 14).25 In this case, samples stained positive for CK-7 only (negative for CK-20, TTF-1 and calretinin), and along with the unremarkable findings of the pancreas and bile duct, an origin in the pancreaticobiliary tree is less likely. The samples did not stain positive for CK-20, which would make a colon primary less likely also, but this does not totally rule out a colorectal origin,26 especially since there was a finding of sigmoid wall thickening and an elevated CEA tumour marker. A negative TTF-1 means that the malignancy did not originate from the lung nor the thyroid gland and that it is a metastasis.
The sample had a CK-7+/CK-20− profile, with a signet ring feature, which suggests that it could be gastric in origin; with the CT of the abdomen and pelvis with contrast revealing slight gastric thickening at the fundus, this makes a gastric adenocarcinoma as the origin less likely since the most common CT finding of such a malignancy is ‘fungating’ followed by ‘diffuse infiltrative’.27 An endoscopy and colonoscopy were advised, but the patient did not consent. Despite the imaging and immunohistochemical work-up done, we were unable to locate the origin of the SRCC.
The European Society of Medical Oncology set diagnostic and staging guidelines for CUPs required in all patients, namely, a thorough medical history and physical examination, basic blood and biochemistry analyses, and CT scans with intravenous contrasts of the thorax, abdomen and pelvis. Thus, based on the clinical practice guidelines on CUPs site by the European Society of Medical Oncology, we have identified this as a CUP.28 Moreover, based on those guidelines, this case belongs to the non-specific subset of CUP, which generally has a poor prognosis with a median survival of around 4 months to 1 year.29 Modest survival prolongation and symptom palliation with preservation of quality of life are currently the only realistic aims of therapy.30 Low-toxicity palliative chemotherapy may have been an option in this case, comprising platinum salts, taxanes or new-generation cytotoxic compounds (gemcitabine, vinca alkaloids or irinotecan).31 A treatment protocol we could have given would be cisplatin 60–75 (day 1)+gemcitabine 1000 (days 1 and 8) every 3 weeks.
The researcher searched the PubMed database using the keywords ‘metastatic signet ring’, ‘unknown primary’ and ‘unknown origin’ using the All Fields in the advanced search option. The search returned eight case reports with a total of 10 patients that were published between 2002 and 2018, which are summarised in table 2.
Table 2.
List of case reports on patients with signet-ring cell carcinoma of unknown primary
| Author (year) | Age (years), gender | Chief complaints | Immunohistochemistry | Therapy | Survival |
| Gregoire et al (2014)8 | 66, female | Abdominal pain | Positive for ER and PR receptors | ECF, tamoxifen and letrozole | 24 months |
| 70, female | Dyspnoea | Positive for ER and PR receptors, CK-7 | Tamoxifen, letrozole and fulvestrant | 15 months | |
| Unknown, female | Abdominal pain | Positive for ER and PR receptors; CK-7 | Tamoxifen and letrozole | 37 months | |
| Al-Taee et al (2018)10 | 73, male | Nausea, vomiting and abdominal pain | Positive for CK-7, CK-20, and E-cadherin | None | Unknown |
| O’Kane et al (2018)9 | 70, male | Incidental finding on imaging after motor vehicle accident | Positive for CK-7 and CK-20 | None | 3 months |
| Kusakari et al (2007)33 | 46, male | Incidental mass on chest imaging | Positive for CK-7 | Cisplatin, irinotecan, and S-1 | 24 months |
| Heidemann et al (2002)34 | 71, female | Sanguineous discharge after defecation | Positive for pancytokeratin and EMA | 5-Fluorouracil and folinic acid | 36 months |
| Shin et al (2011)35 | 80, male | Back pain and jaundice, MAHA | Positive for CK-20 | None | 3 weeks |
| Lara et al (2016)36 | 34, female | Severe and progressive fatigue, MAHA | Positive for ER and PR receptors | None | Unknown |
| Handa et al (2018)37 | 63, female | Incidental finding after thyroidectomy, biopsy of regional lymph node showed SRCC | Positive for PAS, CK AE1/AE3 | Palliative radiotherapy, cisplatin and S-1 | 42 months |
| Danzinger et al
(2019)38 |
37, female | Dyspnoea, jaundice and epigastric pain; pregnant at 21 weeks and 5 days age of gestation | Positive for CK-7; negative for CK-20, CDX2, ER, PR, PAX8 and HER2 | Pembrolizumab | 14 days |
CDX2, caudal-type homeobox transcription factor 2; CK-7, cytokeratin-7; CK-20, cytokeratin-20; CK AE1/AE3, cytokeratin AE1/AE3; ECF, epirubicin, cisplatin, 5-fluorouracil; EMA, epithelial membrane antigen20–24; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; MAHA, microangiopathic haemolytic anaemia; PAS, periodic acid–Schiff; PAX8, paired-box gene 8; PR, progesterone receptor; SRCC, signet-ring cell carcinoma.
Out of the 11 reported patients with metastatic SRCC of unknown primary origin in table 2, seven patients received chemotherapeutic agents. Patients who received treatment had longer survival time. Not enough data are present for prognostication of patients with signet ring carcinoma of unknown primary. However, the prognosis of the SRCCs seems to be worse than tumours without an SRCC component for tumours with known primary origin.8
The patient in our study was given steroids and radiotherapy for the management of his symptoms. By the time the biopsy was done, and his symptoms (dyspnoea) lessened; the service (service consultants) deemed it better to have him discharged first and plan his chemotherapy on an outpatient basis since they will also have to prepare the funds necessary to procure the chemotherapeutic drug. It was also the patient's wishes that he sees his children first back in the province. Chemotherapy was not discarded; the patient was in distress and radiotherapy was the option deemed by the service to be the most important intervention at the time. The patient also had a poor performance status (Eastern Cooperative Oncology Group Performance Status 3), which did not make him a suitable candidate for chemotherapy. Also, the biopsy result had not come out then, so we could not give the appropriate chemotherapeutic regimen.
Patient’s perspective.
I didn’t know what was happening to my husband. His face suddenly grew and he had a hard time breathing. So we brought him to the best hospital we can find. It was explained to us that he had cancer that had spread to the area around his neck, which more or less caused the swelling. The doctors needed to get a biopsy first then went on to do radiotherapy to make my husband feel better. And he did. I asked where did the cancer come from? And the CT scan and other tests did not give us ananswer. So we were discharged and given instructions for follow-up so that we can continue to monitor and do more tests on my husband. They will also plan the chemotherapy as outpatient. They also explained to me that I should have my children screened when they grow of proper age because the cancer my husband had might be hereditary. I am thankful for my doctors who did everything for my husband.
Learning points.
Signet-ring cell carcinoma (SRCC) of unknown primary origin is a rare poorly differentiated aggressive subtype of adenocarcinoma that poses diagnostic and therapeutic difficulties as it tends to present at an advanced stage and is associated with a poor prognosis.
Patients who received therapy had better survival rates than patients who received conservative management based on the case reports gathered. Standards of care have yet to be established.
This work highlights the importance of a comprehensive histopathological and immunohistochemical work-up, which can help predict a site of origin to be able to provide appropriate management options to the patients.
Acknowledgments
We would like to extend gratitude to the department of laboratories, especially the surgical pathology department. We would also like to express our gratitude to our colleagues: Jose Antonio Ha-bana, Antonio Carlo De Guzman (radiation oncology) and Jesse Marie Denoga (family medicine).
Footnotes
Contributors: RBLJ took care of the patient and made the paper. PVCC was the senior author and also gave some inputs in editing the paper. AD-W was the consultant of the service and also provided valuable information regarding the case. DR was the pathology resident who aided in doing the biopsy and slide review of the patient in the case report.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Next of kin consent obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1. Wilson LD, Detterbeck FC, Yahalom J. Superior vena cava syndrome with malignant causes. N Engl J Med 2007;356:1862–9. 10.1056/NEJMcp067190 [DOI] [PubMed] [Google Scholar]
- 2. Rice TW, Rodriguez RM, Light RW. The superior vena cava syndrome: clinical characteristics and evolving etiology. Medicine 2006;85:37–42. 10.1097/01.md.0000198474.99876.f0 [DOI] [PubMed] [Google Scholar]
- 3. Watkinson AF, Yeow TN, Fraser C. Endovascular stenting to treat obstruction of the superior vena cava. BMJ 2008;336:1434–7. 10.1136/bmj.39562.512789.80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Cheng S. “Superior vena cava syndrome: a contemporary review of a historic disease”. Cardiology in Review 2009;1:16–23. [DOI] [PubMed] [Google Scholar]
- 5. Brzezniak C, Oronsky B, Carter CA, et al. Superior vena cava syndrome in a patient with small-cell lung cancer: a case report. Case Rep Oncol 2017;10:252–7. 10.1159/000464278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Cohen R, Mena D, Carbajal-Mendoza R, et al. Superior vena cava syndrome: a medical emergency? Int J Angiol 2008;17:43–6. 10.1055/s-0031-1278280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Charnsangavej C, Carrasco CH, Wallace S. Stenosis of the vena cava: preliminary assessment of treatment with expandable metallic stents. Radiology, U.S. National Library of Medicine, 1986. [DOI] [PubMed] [Google Scholar]
- 8. Gregoire C, Muller G, Machiels J-P, et al. Metastatic signet-ring cell carcinoma of unknown primary origin. Acta Clin Belg 2014;69:135–8. 10.1179/0001551213Z.0000000002 [DOI] [PubMed] [Google Scholar]
- 9. O’Kane D, Dean K, Nightingale R, et al. “Metastatic Signet Ring Cell Carcinoma of Unknown Primary Source. BMJ Case Reports 2018;9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Al-Taee A, Almukhtar R, Lai J, et al. “Metastatic Signet Ring Cell Carcinoma of Unknown Primary Origin: A Case Report and Review of the Literature”. Ann Transl Med 2018;15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Abbruzzese JL, Abbruzzese MC, Hess KR, et al. Unknown primary carcinoma: natural history and prognostic factors in 657 consecutive patients. JCO 1994;12:1272–80. 10.1200/JCO.1994.12.6.1272 [DOI] [PubMed] [Google Scholar]
- 12. Pavlidis N, Briasoulis E, Hainsworth J, et al. Diagnostic and therapeutic management of cancer of an unknown primary. Eur J Cancer 2003;39:1990–2005. 10.1016/S0959-8049(03)00547-1 [DOI] [PubMed] [Google Scholar]
- 13. Su Y-C, Hsu Y-C, Chai C-Y. Role of TTF-1, CK20, and CK7 immunohistochemistry for diagnosis of primary and secondary lung adenocarcinoma. Kaohsiung J Med Sci 2006;22:14–19. 10.1016/S1607-551X(09)70214-1 [DOI] [PubMed] [Google Scholar]
- 14. Jang KY, Kang MJ, Lee DG, et al. Utility of thyroid transcription factor-1 and cytokeratin 7 and 20 immunostaining in the identification of origin in malignant effusions. Anal Quant Cytol Histol 2001;23:400–4. [PubMed] [Google Scholar]
- 15. Merchant SH, Amin MB, Tamboli P, et al. Primary signet-ring cell carcinoma of lung: immunohistochemical study and comparison with non-pulmonary signet-ring cell carcinomas. Am J Surg Pathol 2001;25:1515–9. 10.1097/00000478-200112000-00007 [DOI] [PubMed] [Google Scholar]
- 16. Terada T. Primary signet-ring cell carcinoma of the lung: a case report with an immunohistochemical study. Int J Clin Exp Pathol 2012;5:171–4. [PMC free article] [PubMed] [Google Scholar]
- 17. Terada T. Primary pure signet-ring cell carcinoma of the extrapulmonary left main bronchus: a case report with an immunohistochemical study. Int J Clin Exp Pathol 2013;6:1138–41. [PMC free article] [PubMed] [Google Scholar]
- 18. Moran CA. Pulmonary adenocarcinoma: the expanding spectrum of histologic variants. Arch Pathol Lab Med 2006;130:958–62. 10.1043/1543-2165(2006)130[958:PATESO]2.0.CO;2 [DOI] [PubMed] [Google Scholar]
- 19. Ordóñez NG. Thyroid transcription factor-1 is a marker of lung and thyroid carcinomas. Adv Anat Pathol 2000;7:123–7. 10.1097/00125480-200007020-00007 [DOI] [PubMed] [Google Scholar]
- 20. Moldvay J, Jackel M, Bogos K, et al. P-641 the role of TTF-1 in differentiating primary and metastatic lung adenocarcinomas. Lung Cancer 2003;41 10.1016/S0169-5002(03)92608-0 [DOI] [PubMed] [Google Scholar]
- 21. Castro CY, Moran CA, Flieder DG, et al. Primary signet ring cell adenocarcinomas of the lung: a clinicopathological study of 15 cases. Histopathology 2001;39:397–401. 10.1046/j.1365-2559.2001.01224.x [DOI] [PubMed] [Google Scholar]
- 22. Cai Y-C, Banner B, Glickman J, et al. Cytokeratin 7 and 20 and thyroid transcription factor 1 can help distinguish pulmonary from gastrointestinal carcinoid and pancreatic endocrine tumors. Hum Pathol 2001;32:1087–93. 10.1053/hupa.2001.28245 [DOI] [PubMed] [Google Scholar]
- 23. Selves J, Long-Mira E, Mathieu M-C, et al. Immunohistochemistry for diagnosis of metastatic carcinomas of unknown primary site. Cancers 2018;10:108 10.3390/cancers10040108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Srodon M, Westra WH. Immunohistochemical staining for thyroid transcription factor-1: a helpful aid in discerning primary site of tumor origin in patients with brain metastases. Hum Pathol 2002;33:642–5. 10.1053/hupa.2002.124910 [DOI] [PubMed] [Google Scholar]
- 25. Tot T. Adenocarcinomas metastatic to the liver: the value of cytokeratins 20 and 7 in the search for unknown primary tumors. Cancer 1999;85:171–7. [DOI] [PubMed] [Google Scholar]
- 26. Bayrak R, Yenidünya S, Haltas H. Cytokeratin 7 and cytokeratin 20 expression in colorectal adenocarcinomas. Pathol Res Pract 2011;207:156–60. 10.1016/j.prp.2010.12.005 [DOI] [PubMed] [Google Scholar]
- 27. Lee JH, Park M-S, Kim KW, et al. Advanced gastric carcinoma with signet ring cell carcinoma versus non-signet ring cell carcinoma: differentiation with multidetector CT. J Comput Assist Tomogr 2006;30:880–4. 10.1097/01.rct.0000220800.76276.46 [DOI] [PubMed] [Google Scholar]
- 28. Fizazi K, Greco FA, Pavlidis N, et al. Cancers of unknown primary site: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2015;26:v133–8. 10.1093/annonc/mdv305 [DOI] [PubMed] [Google Scholar]
- 29. Bugat R, Bataillard A, Lesimple T, et al. Summary of the Standards, Options and recommendations for the management of patients with carcinoma of unknown primary site (2002). Br J Cancer 2003;89:S59–66. 10.1038/sj.bjc.6601085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Levy A, Massard C, Gross-Goupil M, et al. Carcinomas of an unknown primary site: a curable disease? Annals of Oncology 2008;19:1657–8. 10.1093/annonc/mdn430 [DOI] [PubMed] [Google Scholar]
- 31. Golfinopoulos V, Pentheroudakis G, Salanti G, et al. Comparative survival with diverse chemotherapy regimens for cancer of unknown primary site: Multiple-treatments meta-analysis. Cancer Treat Rev 2009;35:570–3. 10.1016/j.ctrv.2009.05.005 [DOI] [PubMed] [Google Scholar]
- 32. Jagirdar J. Application of immunohistochemistry to the diagnosis of primary and metastatic carcinoma to the lung. Arch Pathol Lab Med 2008;132:384–96. 10.1043/1543-2165(2008)132[384:AOITTD]2.0.CO;2 [DOI] [PubMed] [Google Scholar]
- 33. Kusakari C, Soda H, Nakamura Y, et al. Mediastinal signet-ring cell carcinoma of unknown primary: long-term survival by treatment with S-1, a novel derivative of 5-fluorouracil. Lung Cancer 2007;56:139–41. 10.1016/j.lungcan.2006.11.020 [DOI] [PubMed] [Google Scholar]
- 34. Heidemann J, Gockel HR, Winde G, et al. Signet-ring Cell Carcinoma of unknown primary Location. Metastatic to lower back Musculature - Remission following FU/FA Chemotherapy. Z Gastroenterol 2002;40:33–6. 10.1055/s-2001-19634 [DOI] [PubMed] [Google Scholar]
- 35. Shin S-Y, Park H, Chae SW, et al. Microangiopathic hemolytic anemia as the first manifestation of metastatic signet ring cell carcinoma of unknown origin: a case report and review of literature. Korean J Lab Med 2011;31:157–61. 10.3343/kjlm.2011.31.3.157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Lara K, Bae E, Park H, et al. “A rare case of concurrent signet-ring carcinoma of breast and microangiopathic hemolytic anemia”. J Surg Case Rep 2016;9:1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Handa H, Gomi D, Fukushima T, et al. Signet ring cell carcinoma of unknown primary origin detected incidentally by lymph node purification for thyroid carcinoma. Internal Medicine 2018;57:31–5. 10.2169/internalmedicine.9234-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Danzinger S, Köstler WJ, Funovics M, et al. Signet ring cell carcinoma of the lung: a diagnostic pitfall in pregnancy. Case Rep Obstet Gynecol 2019;2019:1–8. 10.1155/2019/9461579 [DOI] [PMC free article] [PubMed] [Google Scholar]