Abstract
There is a substantial interest in the development of NK1 substance P antagonists as potential treatments for various neuropsychiatric and somatic disorders. The aim of this study was to determine whether [18F]SPA-RQ can be utilized as a tool for studying the whole body distribution and function of NK1 receptors in preclinical settings. The compound was injected into guinea pigs with or without premedication with a NK1 receptor antagonist (NK1A-2). For comparison, we included two rats in the study, as the affinity of antagonists for NK1 receptors is known to vary between species. The whole body biodistribution of the tracer was determined at several time points. The tracer showed specific binding in organs compatible with the known location of NK1-receptors. Premedication with a NK1 antagonist led to an inhibited uptake of [18F]SPA-RQ in several organs of guinea pigs, notably intestine, pancreas, urinary bladder, uterus, skin and lung. Specific binding was also seen in both cortex and striatum. In contrast, negligible specific binding was observed in the rat brain with [18F]SPA-RQ, whereas the tracer uptake in peripheral tissues was similar to that seen in guinea pigs. We conclude that [18F]SPA-RQ/PET is a useful tool to study the distribution and function of peripherally located NK1 receptors e.g. in different disease models.
Subject terms: Target validation, Experimental models of disease
Introduction
The mammalian tachykinins (TKs) are a family of closely related peptides traditionally classified as neurotransmitters that includes substance P (SP), neurokinin A (NKA) and neurokinin B (NKB). Tachykinins are distributed throughout the central and peripheral nervous system and many different physiological functions, such as e.g. inflammation and smooth muscle contraction, have been linked to them1. The TKs mediate their effect via specific G-protein coupled receptors using predominantly the phosphoinositol system as a second messenger pathway. Three types of tachykinin receptors, NK1, NK2 and NK3 are heterogeneously distributed within species. SP is the natural endogenous ligand of NK1 receptors while NKA and NKB are the preferential ligands of NK2 and NK3 receptors, respectively2.
For many years, the tachykinins were considered almost exclusively as peptides of neuronal origin. However, the concept has changed due to evidence showing that TKs can be produced also by non-neuronal cells such as immune and inflammatory cells3,4. Moreover, an advance in the field of tachykinins has been the discovery of three new mammalian TKs, hemokinin-1 (HK1) and its human orthologs, endokinin A (EKA) and endokinin B (EKB)5–7. A common feature for HK1, EKA and EKB is that they are all primarily expressed in non-neuronal cells and they have shown a high selectivity for the NK1 receptor8,9.
SP is by far the best characterized TK and on the basis of anatomical localization and functional studies there is considerable interest in the development of NK1 substance P antagonists (SPAs) for treatments of a variety of disorders. Such disease processes includes inflammation, pain, affective and addictive disorders, as well as functional disorders of the intestine and urine bladder1.
Clinical trials with a NK1 receptor antagonist, aprepitant (Emend®), has proven to be an effective antiemetic drug in humans and this drug is now available for the treatment of chemotherapy-induced nausea and vomiting10,11. Aprepitant was not effective in the treatment of depression but has shown antitumor activity and proven to be effective for the therapy of itch12–15. Indeed, overexpression of NK1 receptors has been implicated in cancer progression and poor overall prognosis16. Research for new targets continues including receptors in peripheral regions such as urinary and gastrointestinal tracts.
We have previously reported on the synthesis of fluorine-18 labelled SPA-RQ ([2-[18F]fluoromethoxy-5-(5-trifluoromethyl-tetrazol-1-yl)-benzyl]([2S,3S]2-phenyl-piperidin-3-yl)-amine, [18F]SPA-RQ), a selective NK1 receptor antagonist with a very high affinity for this receptor17. Preliminary studies showed that [18F]SPA-RQ selectively bound to NK1 receptors in guinea pig (GP) brain and later it has shown to be a useful tracer for in vivo imaging of human brain NK1 receptors with PET17–22. The radiation dosimetry of [18F]SPA-RQ in human has also been reported23.
We here report the specific binding of [18F]SPA-RQ in the whole body of GPs and rats as to provide a tool to study the function and expression of NK1 receptors outside the brain. The structure of TK receptors, as well as the affinity of non-peptide antagonists for NK1 receptors is known to vary between species24,25. The receptors in the human resembles those in GP and therefore GP is an ideal species to study the function of TKs. For comparison, we also include rats in this study.
An ex vivo experimental PET study approach enables to investigate NK1 receptors at higher spatial resolution and sensitivity than in an in vivo setting. Furthermore, we illustrate the power of the ex vivo autoradiography methodology for determining the intra-organ distribution of the tracer. To define the specific binding of [18F]SPA-RQ the biodistribution was also determined in GPs premedicated with a selective NK1 antagonist.
Results
Synthesis of [18F]SPA-RQ
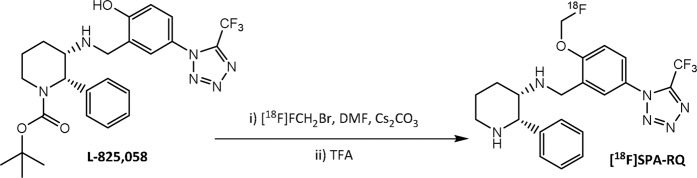
The radiotracer [18F]SPA-RQ was synthesized via O-fluoromethylation of the L-825,058 precursor in DMF (Fig. 1). The synthesis route yielded [18F]SPA-RQ at very high molar activity, ranging from 180 to 2480 GBq/µmol, with a mean value of 920 GBq/µmol at the end of synthesis. The radiochemical purity exceeded 98.5% in all cases.
Figure 1.
Radiochemical synthesis of [18F]SPA-RQ. [18F]FCH2Br = [18F]bromofluoromethane, Cs2CO3 = cesium carbonate, DMF = dimethylformamide, TFA = trifluoroacetic acid.
Peripheral biodistribution of [18F]SPA-RQ
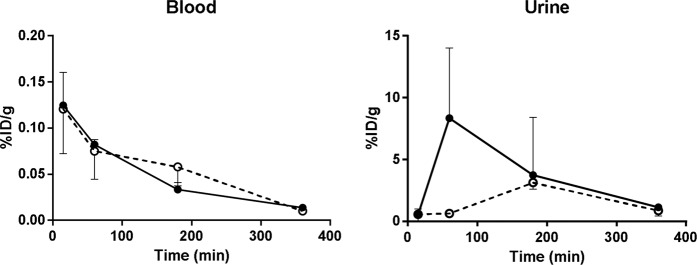
The peripheral biodistribution results of [18F]SPA-RQ from GPs that were either nonmedicated or premedicated with the NK1 selective antagonist are shown in Supplemental Table 1. The uptake of the tracer from blood into tissues was fast (Fig. 2). [18F]SPA-RQ and its radioactive metabolites were moderately bound to plasma proteins. Of the total 18F-radioactivity, free radioactivity in GP plasma was 35–41% at 15 min after the [18F]SPA-RQ injection. The fraction of free 18F-radioactivity increased slightly over time, being 46% and 51% at 60 and 180 min after the injection, respectively.
Figure 2.
18F-radioactivity uptake in blood and urine as a function of time. Data originating from nonmedicated GPs are shown in black symbols and premedicated in white symbols (dashed line). Data in all groups are for three animals per time point and treatment, if not otherwise stated in the Supplementary Table 1. Note that the y-scale (% ID/g) varies between the tissues.
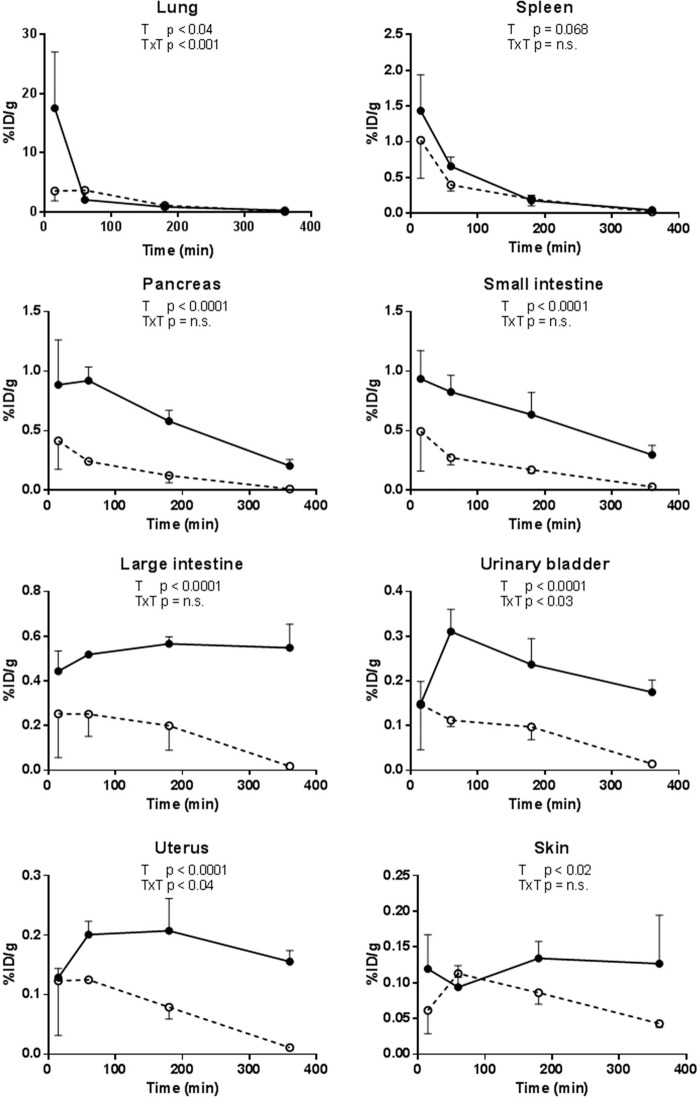
Up to 60 min post injection the highest 18F-radioactivity uptakes were seen in lungs, pancreas, small intestine, spleen, and kidneys (Figs. 3 and 4). At 360 min post injection high uptake of the tracer was still seen in lungs, pancreas and small intestine. Throughout the study high uptake of the tracer was also seen in the large intestine, urinary bladder, uterus, skin, urine and bone. The tracer was primarily excreted through the urinary pathway as a high amount of the tracer was measured from the urine (Fig. 2). Increasing amounts of 18F-radioactivity was measured in the bone (parietal bone in the skull) as a function of time, indicating that some defluorination of the tracer occurred, as earlier reported17.
Figure 3.
18F-radioactivity uptake in selected organs from nonmedicated (black symbols), and GPs premedicated (2 mg/kg) with a NK1 antagonist (white symbols, dashed line) as a function of time. Data in all groups are for three animals per time point and treatment. Note that the y-scale (% ID/g) varies between the different tissues. ANOVA: T, treatment effect; T x T, time x treatment interaction.
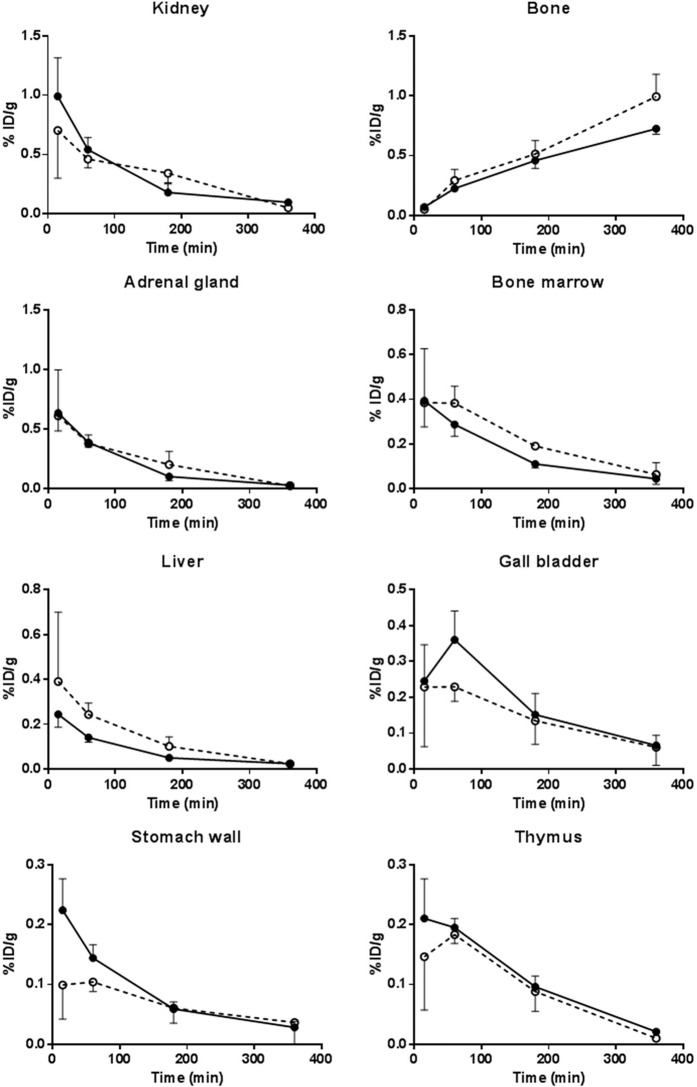
Figure 4.
18F-radioactivity uptake in selected organs unaffected by the NK1 antagonist pretreatment (2 mg/kg) as a function of time. Organs originating from nonmedicated GPs are shown in black symbols and premedicated in white symbols (dashed line). Data in all groups are for three animals per time point and treatment, if not otherwise stated in the Supplementary Table 1. Note that the y-scale (% ID/g) varies between the different tissues. ANOVA: T, treatment effect; T × T, time × treatment interaction.
Several tissues were clearly affected by the treatment (NK1A-2; 2 mg/kg) (Fig. 3). A clear effect on the 18F-radioactivity uptake was seen in small intestine, large intestine, pancreas, uterus, urinary bladder and skin. Furthermore, a moderate effect was seen in lung and a trend toward an effect was detected in the spleen.
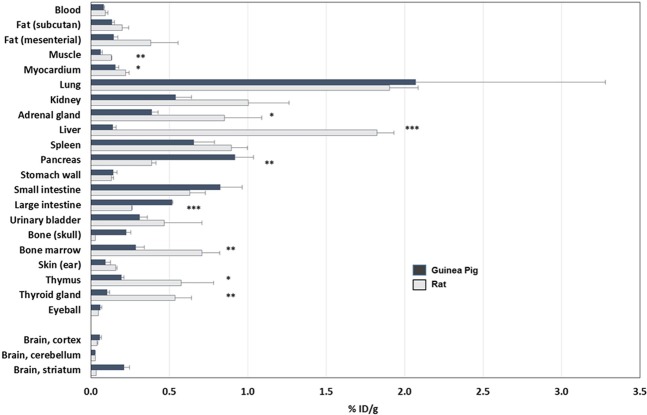
The biodistribution results of [18F]SPA-RQ from rats are shown in Supplemental Table 2. The uptake pattern of [18F]SPA-RQ in tissues of rats compared to GPs is shown in Fig. 5. A significantly higher uptake was seen in the rat heart (p < 0.05), liver (p < 0.001), bone marrow (p < 0.01), adrenal glands (p < 0.05), thymus (p < 0.05) and thyroid glands (p < 0.01), whereas a significantly lower uptake was detected in the pancreas (p < 0.01) and large intestine (p < 0.001). The rat kidney, spleen, fat and urinary bladder showed a trend toward a significantly higher uptake in the rats compared to GPs.
Figure 5.
18F-radioactivity uptake in GP and rat tissues at 60 min post injection of [18F]SPA-RQ. Values are from three GPs and two rats (rat striatum, muscle and eyeball; n = 1). Significances are marked as *p < 0.05, **p < 0.01 and ***p < 0.001.
Regional brain distribution
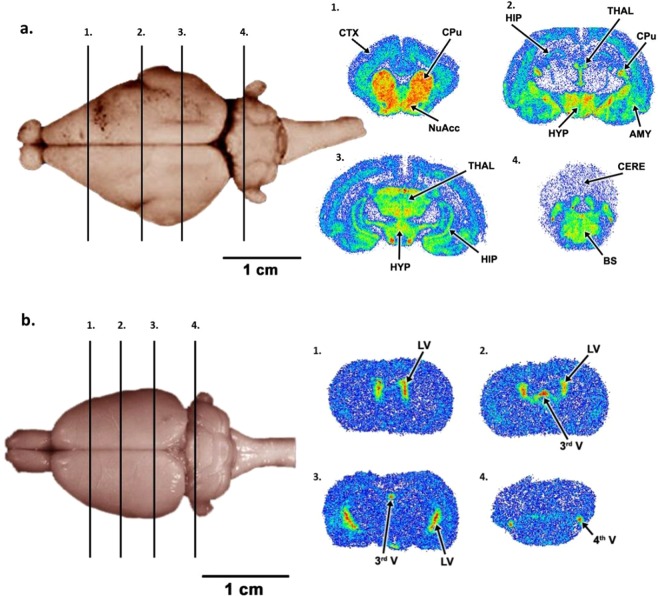
A typical example of the uptake of [18F]SPA-RQ into GP brain at 180 min post injection is shown in Fig. 6a. Nonmedicated animals showed fast and high uptake of the tracer in the caudate/putamen region as well as a very low uptake in the cerebellum. [18F]SPA-RQ also showed high uptakes in regions of thalamus, hypothalamus and midbrain regions. Uptake of 18F-radioactivity was also seen in the cortex (Fig. 6a). [18F]SPA-RQ uptakes in different brain regions from nonmedicated and premedicated GPs expressed as % ID/g tissue calculated from the biodistribution data are shown in Supplemental Table 1. The uptake of [18F]SPA-RQ into rat brain is shown in Fig. 6b. High 18F-radioactivity uptake was seen in lateral ventricle, 3rd ventricle and 4th ventricle and a clear lack of uptake was seen in the areas of caudate-putamen, cortex, thalamus and hypothalamus. Lack of specific binding in the caudate/putamen (striatum) region was also seen in the biodistribution data (Fig. 5).
Figure 6.
Uptake of [18F]SPA-RQ in (a) GP and b) rat brain. The numbered (1–4) bars (on the left) indicate the position from where the four brain slices shown at right were cut. The regional biodistribution of [18F]SPA-RQ in coronal slices of the GP brain at 180 min post injection is shown on the upper right (AMY = amygdala, BS = brainstem, CERE = cerebellum CPu = caudate-putamen, CTX = cortex, HIP = hippocampus, HYP = hypothalamus, NuAcc = nucleus accumbens and THAL = thalamus). The regional biodistribution of [18F]SPA-RQ in coronal slices of the rat brain at 60 min post injection is shown on the lower right (LV = lateral ventricle, 3rd V = 3rd ventricle, 4th V = 4th ventricle).
Discussion
TKs represent one of the largest known peptide families and can be found in different animal species from low invertebrates to mammals. The TKs and their receptors have been highly conserved throughout evolution, which argues for an important biological function that is probably still only partly understood2. They are widely distributed throughout the mammalian body, being primarily distributed in the central and peripheral nervous system but also in non-neuronal cells. In neuronal cells tachykinins acts as neurotransmitters or neuromodulators, whereas they function as autocrine, paracrine or endocrine regulators in non- neuronal cells.
As the NK1 receptor is a potential treatment target for a variety of disorders, we evaluated [18F]SPA-RQ as a tool to investigate the whole body expression and function of NK1 receptors. It has been reported that there are many-fold inter-species differences in the activity of non-peptide NK1 ligands26,27. In order to visualize the inter-species differences in the NK1 receptor distribution, two rats were also studied. Very weak [18F]SPA-RQ specific binding to NK1 receptors in the rat CNS was indeed detected.
Autoradiographic studies using [125I]SP as a NK1 probe have shown high binding in the amygdala, nucleus accumbens, septum, caudate, putamen, hippocampus, hypothalamus, locus coeruleus, substantia nigra, and the frontal, entorhinal, and olfactory cortices in monkey and human28. In line with these findings, we and others have previously reported on a high affinity and selective binding of [18F]SPA-RQ to NK1 receptors in GP and human brain17–22,28. However, in the current study we demonstrate the usage of [18F]SPA-RQ for imaging peripherally located NK1 receptors.
In peripheral neuronal cells, tachykinins can have sensory or motor functions. The source of tachykinins in the peripheral nervous system is a population of sensory nerves, which are activated by capsaicin29,30. When stimulated, a number of neuropeptide transmitters, including SP and NKA are released, which initiate functional responses.
Our results with GPs show that the peripheral uptake of [18F]SPA-RQ is in good agreement with the known biodistribution of SP and NK1 receptors. The highest 18F-radioactivity uptake in the periphery was seen in the lung. SP has been suggested to be a sensory neuropeptide in the lung, where it increases vascular permeability and produces smooth muscle constriction31. Recently, it has become clear that neuropeptides, including SP, are released not only from nerve endings but also from inflammatory immune cells, which affect nervous or immune cells in a paracrine or autocrine fashion32–34. It has also been reported that the immune and nervous systems together optimizes defence systems within the lung35. The uptake of [18F]SPA-RQ was especially high in the lungs at 15 min post injection and continuously high uptake was seen throughout the study, even though the uptake did decrease as a function of time. In premedicated GPs this uptake was significantly affected (p < 0.04) as shown in Fig. 3. This effect was probably mainly due to the apparently reduced 18F-radioactivity uptake at 15 min post injection in premedicated GPs, meanwhile it did slightly increase at 60 and 180 min after injection of [18F]SPA-RQ. This might be due to the complex role of SP, acting both as a neurotransmitter and an autocrine regulator in the lung.
18F-radioactivity uptake of [18F]SPA-RQ in organs of the immune system was measured in bone marrow, thymus and spleen. At early time points, a quite high uptake was seen in the spleen. This uptake decreased as a function of time and significantly reduced uptake (p < 0.001) in premedicated animals was only seen at 360 min post injection. The overall treatment effect did, however, show a trend toward significance (p = 0.068).
We also found, in agreement with the known function of SP, a high 18F-radioactivity uptake in both small- and large intestine. The uptake in the both intestines behaved quite similar and was significantly (p < 0.0001) decreased in premedicated GPs. In the intestine SP is believed to be released into the gut by neurons found in the sympathetic and parasympathetic autonomic nervous system as well as in the enteric nervous system. The released SP will activate cells which participate in the control of gastro-intestinal motility, secretion, circulation and tissue homoeostasis36. A high 18F-radioactivity uptake was also seen in the pancreas. This uptake was also significantly decreased (p < 0.0001) in premedicated GPs. SP released from the primary sensory fibers has been identified as one of the key neurotransmitters in pancreas. Receptors for SP have been detected on pancreatic acinar cells where they stimulate enzyme secretion37. SP did also promote proliferation of pancreatic ductal cells but not their differentiation into β-cells38, and preserved pancreatic β-cells in type 1 and type 2 diabetic mice39. Other gastro-intestinal tissues measured were the stomach wall, liver and the gall bladder. None of these tissues showed high accumulation of [18F]SPA-RQ, nor could the 18F-radioactivity uptake be blocked by the NK1 antagonist, although SP containing nerve fibers has been located in periportal and intralobular regions in the liver of different species (including GP) and in bile duct smooth muscle of GP40.
A moderate 18F-radioactivity uptake could also be detected in the urinary bladder wall, uterus and skin. Our results indicate a NK1 receptor specific uptake in these organs, since the uptake was significantly reduced by the NK1 antagonist. Sensory nerves in the guinea pig bladder wall are known to contain SP41. The precise function of SP as a neurotransmitter in the bladder wall and the uterus is unknown, but it has been suggested to contribute to physiological mechanisms such as the contraction of the bladder and uterus42,43. Sensory nerves in the skin control the vascular permeability and SP is also believed to be involved in neurogenic inflammations of skin44.
Even though a lack of uptake in the brain was detected in rats, the tracer showed quite similar peripheral tissue uptakes compared to GPs. The limited number of rats included in this study does, however, most probably affects our results to some degree. Generally, most studies have focused on developing new antagonist for NK1 receptors in the brain and hence the rat, as a NK1 receptor model, is less used. Our preliminary results does, however, indicate that [18F]SPA-RQ can be used for studying the function of peripheral NK1 receptors in the rat, as well.
In summary, PET imaging of NK1 receptors in peripheral organs will be useful in studies resolving the role of NK1 receptors in pathophysiological processes and diseases as well as in drug development. Based on our findings NK1 receptor imaging in peripheral tissues could be used e.g. for imaging of inflammatory processes33 in the gastrointestinal tract (IBD)36, respiratory system (asthma, chronic bronchitis)4 and pancreas (pancreatitis)45 providing possible targets for therapeutic interventions. Even though not evaluated in the current study, imaging of NK1 receptors in cancer is also an attractive field of application16,46.
Applying NK1 imaging to human peripheral tissues might need further characterization of tissue specific pharmacodynamic properties of the tracer, especially if aiming for full quantitation of receptor characteristics in proof-of concept type studies or NK1 receptor occupancy for dose-finding studies in drug development. However, for some applications semi-quantitative measures might be sufficient and easily translated to human applications.
Conclusions
Our results in GPs demonstrate that the uptake of [18F]SPA-RQ is consistent with earlier localization of NK1 receptors and indicate that [18F]SPA-RQ can be useful for studying the expression and function of peripherally located NK1 receptors e.g. in different disease models.
Materials and Methods
Synthesis of [18F]SPA-RQ
High molar activity [18F]bromofluoromethane was used as the labelling reagent. After the removal of DMF, hydrolysis was carried out in TFA and [18F]SPA-RQ was purified with semi-preparative HPLC. More detailed descriptions of the synthesis of [18F]bromofluoromethane and the synthesis of [18F]SPA-RQ can be found in literature17,47.
Animals
Female GP (Mol:Dunkin-Hartley, Möllegaard Breeding & Research Centre A/S, Skensved, Denmark) and male rats (Harlan Sprague-Dawley, Central Animal Laboratory, University of Turku, Turku, Finland) were used in this study. The animals were housed under standard conditions (temperature 21 °C; humidity 55 ± 5%; 12 h light/dark cycle) with free access to standard food and tap water. Animals were cared for in accordance with the Directives 2012/707/EU, 2014/11/EU and of the European Parliament and of the Council for the Care and Use of Laboratory Animals. Licenses were obtained from the Regional State Administrative Agencies in Finland.
Biodistribution study
Altogether twenty four GPs (530 ± 29 g) and two rats (295 ± 7 g) were used. GPs were anesthetized with i.m. injections of Ketamine/Xylazine (Ketamine 30 mg/kg, Xylazine 5 mg/kg) given prior to the administration of the tracer. In order to occupy the target receptors, twelve GPs were premedicated (2 mg/kg) with NK1A-2, a potent brain penetrant NK1 selective antagonist, in 10% ethanol/saline 5 min before the injection of [18F]SPA-RQ48. The tracer and the antagonist were injected through a cannula inserted in the jugularis. The injected doses varied depending on the distribution time and were 6.4 ± 2.6 MBq, 14 ± 5.1 MBq, 48 ± 26 MBq and 82 ± 19 MBq for GPs studied at 15, 60, 180 and 360 minutes, respectively. In rats, the tracer was administered (35 ± 25.2 MBq) via a tail vein and studied at 60 min post injection without pretreatment. The administrated amounts of non-radioactive SPA-RQ, calculated from the injected radioactivity and from the molar activity of [18F]SPA-RQ, varied from 20 to 200 ng. At each time point three nonmedicated and three premedicated GPs were studied. The animals were sacrificed and the organs of interest were immediately dissected and measured for 18F-radioactivity in a well counter (3 × 3 inches NaI (Tl) crystal, Bicron 3MW3/3 P, Bicron Inc., Newbury, Ohio, USA) and weighed. Brains were also rapidly removed, frozen in isopentane chilled with dry ice and prepared as described in the section on regional brain distribution. In addition, pieces of brain tissue from cortex, cerebellum and striatum from each animal were measured separately for absolute radioactivity. All data were corrected for background radioactivity and radioactivity decay. The uptake of 18F-radioactivity in tissues was expressed as percentage of injected dose per gram tissue weight (% ID/g).
Plasma protein binding
Cardiac blood samples were collected from GPs at 15, 60 and 180 min after [18F]SPA-RQ injection into gel-lithium heparin tubes (Terumo Europe, Leuven, Belgium), and plasma was separated by centrifugation (1 300 g, 10 min). The protein free fraction was separated from plasma by filtration with Filtron Microsep 30 K ultrafiltration tubes (3 030 g, 2 × 15 min; Filtron Technology Corporation, Northborough, MA) and the amount of radioactivity was measured before and after filtration to assess the free fraction of 18F-radioactivity in plasma.
Regional brain distribution
The regional brain distribution of [18F]SPA-RQ was studied using a digital autoradiography technique (Fuji BAS 5000, Fuji Photo Film Co. Ltd. Japan) in one nonmedicated GP and one rat. This technique has the advantages of being very sensitive for detecting the β+ particles emitted in the decay of 18F as well as having comparatively good spatial resolution (pixel size 25 × 25 µm). Serial coronal brain sections (20 μm) were obtained using a cryomicrotome. The brain sections were thaw-mounted onto microscope slides, air dried and apposed to an imaging plate (Fuji Imaging Plate BAS-TR2025, Fuji Photo Film Co., Ltd., Japan). The exposure time was 4 ± 0.5 h. The imaging plates were then scanned with the Fuji Analyzer BAS-5000. Regions were anatomically identified from the sections using a GP brain atlas49.
Statistics
Data are given as mean ± standard deviation (SD). Two-way analysis of variance (ANOVA) was used with the factors treatment (nonmedicated, premedicated) and time (15, 60, 180 and 360 min after injection of [18F]SPA-RQ) and their interaction. Square-root- and log-transformations were used to meet assumptions of ANOVA. If the interaction was not significant it was removed from analysis and only main effects were included in the final model. For simplicity, mean ± SD are used in figures. The analyses were performed with SAS System software, version 9.3 for Windows (SAS Institute, Cary, NC, USA). Differences in [18F]SPA-RQ uptake between GPs and rats were tested with an unpaired t-test and calculations carried out with use of GraphPad Prism software v. 6.07 (GraphPad Software, San Diego, CA, USA). Differences were considered significant if p < 0.05 (two-tailed).
Supplementary information
Acknowledgements
The authors want to gratefully acknowledge the contribution of Dr Donald Burns (Merck & Co, Inc., West Point, PA, USA) for the development of [18F]SPA-RQ. We thank Tarja Marttila, B.Sc., for excellent technical assistance and Heikki Hiekkanen, M.Sc., for consultation on the statistical analyses.
Author contributions
T.J.G., J.H., M.H.-S. and O.S. designed the experiments, participated in the interpretation of data and were responsible for writing the manuscript; S.F., O.E., J.B. and P.M. performed the experiments and participated in the interpretation of data; E.L. performed statistical analyses. All authors read and approved the final version of the manuscript.
Competing interests
This study was supported by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-019-56848-3.
References
- 1.Steinhoff MS, von Mentzer B, Geppetti P, Pothoulakis C, Bunnett NW. Tachykinins and their receptors: contributions to physiological control and the mechanisms of disease. Physiol. Rev. 2014;94:265–301. doi: 10.1152/physrev.00031.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pennefather JN, et al. Tachykinins and tachykinin receptors: a growing family. Life Sci. 2004;74:1445–1463. doi: 10.1016/j.lfs.2003.09.039. [DOI] [PubMed] [Google Scholar]
- 3.Weinstock JV, Blum A, Walder J, Walder R. Eosinophils from granulomas in murine schistosomiasis mansoni produce substance P. J. Immunol. 1988;141:961–966. [PubMed] [Google Scholar]
- 4.Joos GF, Pauwels RA. Pro-inflammatory effects of substance P: new perspectives for the treatment of airway diseases? Trends Pharmacol. Sci. 2000;21:131–133. doi: 10.1016/S0165-6147(00)01458-9. [DOI] [PubMed] [Google Scholar]
- 5.Zhang Y, Lu L, Furlonger C, Wu GE, Paige CJ. Hemokinin is a hematopoietic-specific tachykinin that regulates B lymphopoiesis. Nature Immunol. 2000;1:392–397. doi: 10.1038/80826. [DOI] [PubMed] [Google Scholar]
- 6.Kurtz MM, et al. Identification, localization and receptor characterization of novel mammalian substance P-like peptides. Gene. 2002;296:205–212. doi: 10.1016/S0378-1119(02)00861-2. [DOI] [PubMed] [Google Scholar]
- 7.Page NM, et al. Characterization of the endokinins: human tachykinins with cardiovascular activity. Proc Natl Acad Sci USA. 2003;100:6245–6250. doi: 10.1073/pnas.0931458100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Morteau O, Lu B, Gerard C, Gerard NP. Hemokinin 1 is a full agonist at the substance P receptor. Nat Immunol. 2001;2:1088. doi: 10.1038/ni1201-1088. [DOI] [PubMed] [Google Scholar]
- 9.Bellucci F, et al. Pharmacoligal profile of the novel mammalian tachykinin, hemokinin 1. Br J Pharmacol. 2002;135:266–274. doi: 10.1038/sj.bjp.0704443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Navari RM, et al. Reduction of cisplatin-induced emesis by a selective neurokinin-1-receptor antagonist. N Engl J Med. 1999;340:190–195. doi: 10.1056/NEJM199901213400304. [DOI] [PubMed] [Google Scholar]
- 11.Hesketh PJ, et al. The oral neurokinin-1 antagonist aprepitant for the prevention of chemotherapy-induced nausea and vomiting: A multinational, randomized, double-blind, placebo-controlled trial in patients receiving high-dose cisplatin - The Aprepitant Protocol 052 Study Group. J Clin Oncol. 2003;21:4112–4119. doi: 10.1200/JCO.2003.01.095. [DOI] [PubMed] [Google Scholar]
- 12.Keller M, et al. Lack of efficacy of the substance P (neurokinin 1 receptor) antagonist aprepitant in the treatment of major depressive disorder. Biol Psychiatry. 2006;59:216–223. doi: 10.1016/j.biopsych.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 13.Coveñas R, Muñoz M. Cancer progression and substance P. Histol Histopathol. 2014;29:881–90. doi: 10.14670/HH-29.881. [DOI] [PubMed] [Google Scholar]
- 14.Duval A, Dubertret L. Aprepitant as an antipruritic agent? N Engl J Med. 2009;361:1415–1416. doi: 10.1056/NEJMc0906670. [DOI] [PubMed] [Google Scholar]
- 15.Ständer S, Siepmann D, Herrgott I, Sunderkötter C, Luger TA. Targeting the neurokinin receptor 1 with aprepitant: a novel antipruritic strategy. PLoS One. 2010;5:e10968. doi: 10.1371/journal.pone.0010968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Majkowska-Pilip A, Halik PK, Gniazdowska E. The significance of NK1 receptor ligands and their application in targeted radionuclide tumour therapy. Pharmaceutics. 2019;11:E443. doi: 10.3390/pharmaceutics11090443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Solin O, et al. Synthesis and characterization of a potent, selective radiolabeled substance-P antagonist for NK1 receptor quantitation: ([18F]SPA-RQ) Mol Imaging Biol. 2004;6:373–384. doi: 10.1016/j.mibio.2004.08.001. [DOI] [PubMed] [Google Scholar]
- 18.Bergström M, et al. Human positron emission tomography studies of brain neurokinin 1 receptor occupancy by Aprepitant. Biol Psychiatry. 2004;55:1007–1012. doi: 10.1016/j.biopsych.2004.02.007. [DOI] [PubMed] [Google Scholar]
- 19.Hietala J, et al. Visualization and quantification of neurokinin-1 (NK1) receptors in the human brain. Mol Imaging Biol. 2005;7:262–272. doi: 10.1007/s11307-005-7001-6. [DOI] [PubMed] [Google Scholar]
- 20.Nyman MJ, et al. Gender and age affect NK1 receptors in the human brain - a positron emission tomography study with [18F]SPA-RQ. Int J Neuropsychopharmacol. 2007;10:219–229. doi: 10.1017/S1461145706006572. [DOI] [PubMed] [Google Scholar]
- 21.Fujimura Y, Yasuno F, Farris A. Decreased neurokinin-1 (substance P) receptor binding in patients with panic disorder: positron emission tomographic study with [18F]SPA-RQ. Biol Psychiatry. 2009;66:94–97. doi: 10.1016/j.biopsych.2008.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nyman M, et al. Brain neurokinin-1 receptor availability in never-medicated patients with major depression - A pilot study. J Affect Disord. 2019;242:188–194. doi: 10.1016/j.jad.2018.08.084. [DOI] [PubMed] [Google Scholar]
- 23.Sprague DR, et al. Human biodistribution and radiation dosimetry of the tachykinin NK1 antagonist radioligand [18F]SPA-RQ: comparison of thin-slice, bisected, and 2-dimensional planar image analysis. J Nucl Med. 2007;48:100–107. [PMC free article] [PubMed] [Google Scholar]
- 24.Petitet F, et al. NK-1 tachykinin receptor in rat and guinea pig brains: pharmacological and autoradiographical evidence for a species difference. Peptides. 1993;14:551–9. doi: 10.1016/0196-9781(93)90144-6. [DOI] [PubMed] [Google Scholar]
- 25.Rupniak NM, Kramer MS. Discovery of the antidepressant and anti-emetic efficacy of substance P receptor (NK1) antagonists. Trends Pharmacol Sci. 1999;20:485–90. doi: 10.1016/S0165-6147(99)01396-6. [DOI] [PubMed] [Google Scholar]
- 26.Otsuka M, Yoshioka K. Neurotransmitter functions of mammalian tachykinins. Physiol Rev. 1993;73:229–308. doi: 10.1152/physrev.1993.73.2.229. [DOI] [PubMed] [Google Scholar]
- 27.Ventura S, Lau WA, Buljubasich S, Pennefather JN. Species differences in the actions of sensory neuropeptides on contractility of the smooth muscle of the rat and guinea-pig prostate. Clin Exp Pharmacol Physiol. 2000;11:917–921. doi: 10.1046/j.1440-1681.2000.03361.x. [DOI] [PubMed] [Google Scholar]
- 28.Hargreaves R. Imaging substance P receptors (NK1) in the living human brain using positron emission tomography. J Clin Psychiatry. 2002;11:18–24. [PubMed] [Google Scholar]
- 29.Holzer P. Capsaicin: cellular targets, mechanism of action, and selectivity for thin sensory neurones. Pharmacol Rev. 1991;43:143–201. [PubMed] [Google Scholar]
- 30.Khawaja AM, Rogers DF. Tachykinins: receptor to effector. Int J Biochem Cell Biol. 1996;28:721–738. doi: 10.1016/1357-2725(96)00017-9. [DOI] [PubMed] [Google Scholar]
- 31.Lundberg JM, Saria A, Brodin E, Rosell S, Folkers K. A substance P antagonist inhibits vagally induced increase in vascular permeability and bronchial smooth muscle contraction in the guinea pig. Proc Natl Acad Sci USA. 1983;80:1120–1124. doi: 10.1073/pnas.80.4.1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lambrecht BN. Immunologists getting nervous: neuropeptides, dendritic cells and T cell activation. Respir Res. 2001;2:133–138. doi: 10.1186/rr49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.O’Connor TM, et al. The role of substance P in inflammatory disease. J Cell Physiol. 2004;201:167–180. doi: 10.1002/jcp.20061. [DOI] [PubMed] [Google Scholar]
- 34.Suvas S. Role of substance P neuropeptide in inflammation, wound healing, and tissue homeostasis. J Immunol. 2017;199:1543–1552. doi: 10.4049/jimmunol.1601751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Groneberg DA, Harrison S, Dinh QT, Geppetti P, Fischer A. Tachykinins in the respiratory tract. Curr Drug Targets. 2006;7:1005–10. doi: 10.2174/138945006778019318. [DOI] [PubMed] [Google Scholar]
- 36.Holzer P. Implications of tachykinins and calcitonin gene-related peptide in inflammatory bowel disease. Digestion. 1998;59:269–283. doi: 10.1159/000007504. [DOI] [PubMed] [Google Scholar]
- 37.Patto RJ, Vinayek R, Jensen RT, Gardner JD. Carbachol does not down-regulate substance P receptors in pancreatic acini. Pancreas. 1992;7:447–452. doi: 10.1097/00006676-199207000-00005. [DOI] [PubMed] [Google Scholar]
- 38.Zhang N, Gao D, Liu Y, Ji S, Sha L. Effects of Neuropeptide Substance P on Proliferation and β-Cell Differentiation of Adult Pancreatic Ductal Cells. Front Neurosci. 2018;12:806. doi: 10.3389/fnins.2018.00806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Um J, et al. Substance P preserves pancreatic β-cells in type 1 and type 2 diabetic mice. Biochem Biophys Res Commun. 2018;499:960–966. doi: 10.1016/j.bbrc.2018.04.028. [DOI] [PubMed] [Google Scholar]
- 40.Patacchini R, Bartho L, Maggim CA. Characterization of receptors mediating contraction induced by tachykinins in the guinea-pig isolated common bile duct. Br J Pharmacol. 1997;122:1633–1638. doi: 10.1038/sj.bjp.0701560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhou Y, Ling EA. Colocalization of nitric oxide synthase and some neurotransmitters in the intramural ganglia of the guinea pig urinary bladder. J Comp Neurol. 1998;394:496–505. doi: 10.1002/(SICI)1096-9861(19980518)394:4<496::AID-CNE7>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 42.Candenas L, et al. Tachykinins and tachykinin receptors: effects in the genitourinary tract. Life Sci. 2005;76:835–862. doi: 10.1016/j.lfs.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 43.Gillespie JI. Inhibitory actions of calcitonin gene-related peptide and capsaicin: evidence for local axonal reflexes in the bladder wall. BJU Int. 2005;95:149–156. doi: 10.1111/j.1464-410X.2005.05268.x. [DOI] [PubMed] [Google Scholar]
- 44.Maggi CA. The effects of tachykinins on inflammatory and immune cells. Regul Pept. 1997;70:75–90. doi: 10.1016/S0167-0115(97)00029-3. [DOI] [PubMed] [Google Scholar]
- 45.Liddle RA, Nathan JD. Neurogenic inflammation and pancreatitis. Pancreatology. 2004;4:551–559. doi: 10.1159/000082180. [DOI] [PubMed] [Google Scholar]
- 46.Munoz M, Covenas R, Esteban F, Redondo M. The substance P/NK-1 receptor system: NK-1 receptor antagonists as anti-cancer drugs. J Biosci. 2015;40:441–463. doi: 10.1007/s12038-015-9530-8. [DOI] [PubMed] [Google Scholar]
- 47.Bergman J, Eskola O, Lehikoinen P, Solin O. Automated synthesis and purification of [18F]bromofluoromethane at high specific radioactivity. Appl Radiat Isot. 2001;54:927–933. doi: 10.1016/S0969-8043(00)00358-4. [DOI] [PubMed] [Google Scholar]
- 48.Harrison T, et al. An orally active, water-soluble neurokinin-1 receptor antagonist suitable for both intravenous and oral clinical administration. J Med Chem. 2001;44:4296–4299. doi: 10.1021/jm0109558. [DOI] [PubMed] [Google Scholar]
- 49.Luparello, T. J. Stereotaxic atlas of the forebrain of the guinea pig. (New York, NY, 1967).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.